A Three-Domain Scoring System to Customize the Risk of Relapse of Differentiated Thyroid Carcinoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Definition of the Domains
2.3. Study Reference Standards
2.4. Statistical Considerations
3. Results
3.1. Characteristics of the Series
3.2. Training Set
3.3. Validation Set
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ATA | American Thyroid Association |
| DTC | Differentiated Thyroid Cancer |
| HR | Hazard ratio |
| NESD | No evidence of structural disease |
| PFS | Progression-free survival |
| PT | Post-therapy |
| RAI | Radioactive Iodine |
| REC | Structural recurrence |
| SPECT/CT | Single-photon emission computed tomography/X-ray computed tomography |
| Tg | Thyroglobulin |
| THW | Thyroid Hormone Withdrawal |
| TNM | Tumour-node-metastases |
| WBS | Whole-body scan |
References
- Seib, C.D.; Sosa, J.A. Evolving Understanding of the Epidemiology of Thyroid Cancer. Endocrinol. Metab. Clin. N. Am. 2019, 48, 23–35. [Google Scholar] [CrossRef]
- Vigneri, R.; Malandrino, P.; Vigneri, P. The changing epidemiology of thyroid cancer: Why is incidence increasing? Curr. Opin. Oncol. 2015, 27, 1–7. [Google Scholar] [CrossRef]
- Cabanillas, M.E.; McFadden, D.G.; Durante, C. Thyroid cancer. Lancet 2016, 388, 2783–2795. [Google Scholar] [CrossRef]
- Schmidbauer, B.; Menhart, K.; Hellwig, D.; Grosse, J. Differentiated Thyroid Cancer-Treatment: State of the Art. Int. J. Mol. Sci. 2017, 18, 1292. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef]
- Gallop, K.; Kerr, C.; Simmons, S.; McIver, B.; Cohen, E.E. A qualitative evaluation of the validity of published health utilities and generic health utility measures for capturing health-related quality of life (HRQL) impact of differentiated thyroid cancer (DTC) at different treatment phases. Qual. Life Res. 2015, 24, 325–338. [Google Scholar] [CrossRef]
- Trimboli, P.; Zilioli, V.; Imperiali, M.; Ceriani, L.; Giovanella, L. High-sensitive basal serum thyroglobulin 6-12 months after thyroid ablation is strongly associated with early response to therapy and event-free survival in patients with low-to-intermediate risk differentiated thyroid carcinomas. Eur. J. Endocrinol. 2017, 176, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Silaghi, C.A.; Lozovanu, V.; Silaghi, H.; Georgescu, R.D.; Pop, C.; Dobrean, A.; Georgescu, C.E. The Prognostic Value of MicroRNAs in Thyroid Cancers-A Systematic Review and Meta-Analysis. Cancers 2020, 12, 2608. [Google Scholar] [CrossRef] [PubMed]
- Nylen, C.; Mechera, R.; Marechal-Ross, I.; Tsang, V.; Chou, A.; Gill, A.J.; Clifton-Bligh, R.J.; Robinson, B.G.; Sywak, M.S.; Sidhu, S.B.; et al. Molecular Markers Guiding Thyroid Cancer Management. Cancers 2020, 12, 2164. [Google Scholar] [CrossRef] [PubMed]
- Cooper, D.S.; Doherty, G.; Haugen, B.R.; Kloos, R.T.; Lee, S.; Mandel, S.J.; Mazzaferri, E.L.; McIver, B.; Pacini, F.; Schlumberger, M.; et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 2009, 19, 1167–1214. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, S.; Agrawal, A.; Pantvaidya, G.; Shah, S.; Purandare, N.; Puranik, A.; Rangarajan, V. Assessment of the impact of 2015 American Thyroid Association guidelines in management of differentiated thyroid cancer patients. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Sawka, A.M.; Alexander, E.K.; Bianco, A.C.; Chou, R.; Haugen, B.R.; Kopp, P.A.; Pearce, E.N.; Ross, D.S.; Smallridge, R.C.; Jonklaas, J. Challenges in Developing Recommendations Based on Low-Quality Evidence in Thyroid Guidelines. Thyroid 2021, 31, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Bautista-Orduno, K.G.; Dorsey-Trevino, E.G.; Gonzalez-Gonzalez, J.G.; Castillo-Gonzalez, D.A.; Garcia-Leal, M.; Raygoza-Cortez, K.; Gionfriddo, M.R.; Rodriguez-Gutierrez, R. American thyroid association guidelines are inconsistent with Grading of Recommendations Assessment, Development, and Evaluations-A meta-epidemiologic study. J. Clin. Epidemiol. 2020, 123, 180–188.e182. [Google Scholar] [CrossRef] [PubMed]
- Lind, P.; Kohlfurst, S. Respective roles of thyroglobulin, radioiodine imaging, and positron emission tomography in the assessment of thyroid cancer. Semin. Nucl. Med. 2006, 36, 194–205. [Google Scholar] [CrossRef]
- Piccardo, A.; Siri, G.; Raffa, S.; Castellana, M.; Foppiani, L.; Bottoni, G.; Ugolini, M.; Cistaro, A.; Catrambone, U.; Altrinetti, V.; et al. How to better stratify the risk of differentiated thyroid carcinomas: The key role of radioactive iodine therapy, age, and gender. Eur. J. Nucl. Med. Mol. Imaging 2020, 48, 822–830. [Google Scholar] [CrossRef]
- Bychkov, A. AJCC/TNM Staging. Thyroid Gland, Miscellaneous. TNM Staging of Differentiated, Anaplastic and Medullary Thyroid Cancer, 8th ed.; AJCC: Chicago, IL, USA, 2017. [Google Scholar]
- Nixon, I.J.; Wang, L.Y.; Migliacci, J.C.; Eskander, A.; Campbell, M.J.; Aniss, A.; Morris, L.; Vaisman, F.; Corbo, R.; Momesso, D.; et al. An International Multi-Institutional Validation of Age 55 Years as a Cutoff for Risk Stratification in the AJCC/UICC Staging System for Well-Differentiated Thyroid Cancer. Thyroid 2016, 26, 373–380. [Google Scholar] [CrossRef]
- Trimboli, P.; Piccardo, A.; Signore, A.; Valabrega, S.; Barnabei, A.; Santolamazza, G.; Di Paolo, A.; Stati, V.; Chiefari, A.; Vottari, S.; et al. Patient Age Is an Independent Risk Factor of Relapse of Differentiated Thyroid Carcinoma and Improves the Performance of the American Thyroid Association Stratification System. Thyroid 2020, 30, 713–719. [Google Scholar] [CrossRef]
- Zahedi, A.; Bondaz, L.; Rajaraman, M.; Leslie, W.D.; Jefford, C.; Young, J.E.; Pathak, K.A.; Bureau, Y.; Rachinsky, I.; Badreddine, M.; et al. Risk for Thyroid Cancer Recurrence Is Higher in Men Than in Women Independent of Disease Stage at Presentation. Thyroid 2020, 30, 871–877. [Google Scholar] [CrossRef]
- Shi, R.L.; Qu, N.; Liao, T.; Wei, W.J.; Wang, Y.L.; Ji, Q.H. The Trend of Age-Group Effect on Prognosis in Differentiated Thyroid Cancer. Sci. Rep. 2016, 6, 27086. [Google Scholar] [CrossRef]
- Orosco, R.K.; Hussain, T.; Brumund, K.T.; Oh, D.K.; Chang, D.C.; Bouvet, M. Analysis of age and disease status as predictors of thyroid cancer-specific mortality using the Surveillance, Epidemiology, and End Results database. Thyroid 2015, 25, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Kuang, A.; Xie, J.; Ma, T. Possible explanations for patients with discordant findings of serum thyroglobulin and 131I whole-body scanning. J. Nucl. Med. 2005, 46, 1473–1480. [Google Scholar] [PubMed]
- Klain, M.; Pace, L.; Zampella, E.; Mannarino, T.; Limone, S.; Mazziotti, E.; De Simini, G.; Cuocolo, A. Outcome of Patients With Differentiated Thyroid Cancer Treated With 131-Iodine on the Basis of a Detectable Serum Thyroglobulin Level After Initial Treatment. Front. Endocrinol. 2019, 10, 146. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.Y.; Kim, W.B.; Kim, E.S.; Ryu, J.S.; Yeo, J.S.; Kim, S.C.; Hong, S.J.; Shong, Y.K. Serum thyroglobulin levels at the time of 131I remnant ablation just after thyroidectomy are useful for early prediction of clinical recurrence in low-risk patients with differentiated thyroid carcinoma. J. Clin. Endocrinol. Metab. 2005, 90, 1440–1445. [Google Scholar] [CrossRef]
- Piccardo, A.; Arecco, F.; Morbelli, S.; Bianchi, P.; Barbera, F.; Finessi, M.; Corvisieri, S.; Pestarino, E.; Foppiani, L.; Villavecchia, G.; et al. Low thyroglobulin concentrations after thyroidectomy increase the prognostic value of undetectable thyroglobulin levels on levo-thyroxine suppressive treatment in low-risk differentiated thyroid cancer. J. Endocrinol. Investig. 2010, 33, 83–87. [Google Scholar] [CrossRef]
- Piccardo, A.; Arecco, F.; Puntoni, M.; Foppiani, L.; Cabria, M.; Corvisieri, S.; Arlandini, A.; Altrinetti, V.; Bandelloni, R.; Orlandi, F. Focus on high-risk DTC patients: High postoperative serum thyroglobulin level is a strong predictor of disease persistence and is associated to progression-free survival and overall survival. Clin. Nucl. Med. 2013, 38, 18–24. [Google Scholar] [CrossRef]
- Kim, H.; Park, S.Y.; Choe, J.H.; Kim, J.S.; Hahn, S.Y.; Kim, S.W.; Chung, J.H.; Jung, J.; Kim, T.H. Preoperative Serum Thyroglobulin and Its Correlation with the Burden and Extent of Differentiated Thyroid Cancer. Cancers 2020, 12, 625. [Google Scholar] [CrossRef] [PubMed]
- Patell, R.; Mikhael, A.; Tabet, M.; Bena, J.; Berber, E.; Nasr, C. Assessing the utility of preoperative serum thyroglobulin in differentiated thyroid cancer: A retrospective cohort study. Endocrine 2018, 61, 506–510. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, D.; Szikszai, A.; Linke, R.; Bautz, W.; Kuwert, T. Impact of 131I SPECT/spiral CT on nodal staging of differentiated thyroid carcinoma at the first radioablation. J. Nucl. Med. 2009, 50, 18–23. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Avram, A.M. Radioiodine scintigraphy with SPECT/CT: An important diagnostic tool for thyroid cancer staging and risk stratification. J. Nucl. Med. Technol. 2014, 42, 170–180. [Google Scholar] [CrossRef]
- Tuttle, R.M.; Ahuja, S.; Avram, A.M.; Bernet, V.J.; Bourguet, P.; Daniels, G.H.; Dillehay, G.; Draganescu, C.; Flux, G.; Fuhrer, D.; et al. Controversies, Consensus, and Collaboration in the Use of (131)I Therapy in Differentiated Thyroid Cancer: A Joint Statement from the American Thyroid Association, the European Association of Nuclear Medicine, the Society of Nuclear Medicine and Molecular Imaging, and the European Thyroid Association. Thyroid 2019, 29, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Avram, A.M.; Fig, L.M.; Frey, K.A.; Gross, M.D.; Wong, K.K. Preablation 131-I scans with SPECT/CT in postoperative thyroid cancer patients: What is the impact on staging? J. Clin. Endocrinol. Metab. 2013, 98, 1163–1171. [Google Scholar] [CrossRef]
- Avram, A.M.; Esfandiari, N.H.; Wong, K.K. Preablation 131-I scans with SPECT/CT contribute to thyroid cancer risk stratification and 131-I therapy planning. J. Clin. Endocrinol. Metab. 2015, 100, 1895–1902. [Google Scholar] [CrossRef]
- Marotta, V.; Botti, G.; Ionna, F.; Pezzullo, L. TSH modulation in advanced differentiated thyroid cancer: A continuous walk on the tightrope between prognostic stratification, adverse events, and levothyroxine bioavailability. Minerva Endocrinol. 2020. [Google Scholar] [CrossRef]
- Marotta, V.; Sciammarella, C.; Chiofalo, M.G.; Gambardella, C.; Bellevicine, C.; Grasso, M.; Conzo, G.; Docimo, G.; Botti, G.; Losito, S.; et al. Hashimoto’s thyroiditis predicts outcome in intrathyroidal papillary thyroid cancer. Endocr. Relat. Cancer 2017, 24, 485–493. [Google Scholar] [CrossRef]
- Marotta, V.; Sciammarella, C.; Colao, A.; Faggiano, A. Application of molecular biology of differentiated thyroid cancer for clinical prognostication. Endocr. Relat. Cancer 2016, 23, R499–R515. [Google Scholar] [CrossRef]
| Variable | Subjects Included (n = 907) |
|---|---|
| Sex | |
| Female, no. (%) | 643 (70.9) |
| Male, no. (%) | 264 (29.1) |
| Age on diagnosis, median (IQR), years | 53 (41–63) |
| <55 years, no. (%) | 485 (53.5) |
| ≥55 years, no. (%) | 422 (46.5) |
| Pathology | |
| Papillary thyroid carcinoma, no. (%) | 791 (87.2) |
| Follicular thyroid carcinoma, no. (%) | 116 (12.8) |
| Clinico-pathological classification * | |
| T1, no. (%) | 458 (50.5) |
| T2, no. (%) | 137 (15.1) |
| T3, no. (%) | 247 (27.2) |
| T4, no. (%) | 65 (7.2) |
| N0, no. (%) | 669 (73.8) |
| N1, no. (%) | 238 (26.2) |
| M0, no. (%) | 887 (97.8) |
| M1, no. (%) | 20 (2.2) |
| TSH after THW, media (IQR), uIU/ml | 65.5 (49.2–89.7) |
| Radioactive iodine dose, median (IQR), GBq | 2.96 (2.96–3.7) |
| Outcome | |
| No evidence of structural disease, no. (%) | 674 (74.3) |
| Recurrence, no. (%) | 193 (21.3) |
| Death, no. (%) | 40 (4.4) |
| Descriptive Statistics (Absolute Number and Frequencies) According to the Onset of PFS during the Follow-up in the Training Set. | PFS | p-Value * | ||||
|---|---|---|---|---|---|---|
| Overall | No | Yes | ||||
| 635 (100%) | 495 (78.0) | 140 (22.1) | ||||
| Demographic domains | Age at diagnosis | <55 | 353 (55.6) | 283 (57.2) | 70 (50.0) | 0.149 |
| ≥55 | 212 (42.8) | 212 (42.8) | 70 (50.0) | |||
| Sex | F | 441 (69.5) | 362 (73.1) | 79 (56.4) | < 0.001 | |
| M | 194 (30.5) | 133 (26.9) | 61 (43.6) | |||
| Surgical domains | Histological type | Papillary CA | 549 (86.5) | 441 (89.1) | 108 (77.1) | 0.001 |
| Follicular CA | 86 (13.5) | 54 (10.9) | 32 (22.9) | |||
| ATA risk category | Low | 341 (53.7) | 321 (64.8) | 20 (14.3) | <0.001 | |
| Intermediate | 217 (34.2) | 148 (29.9) | 69 (49.3) | |||
| High | 77 (12.1) | 26 (5.2) | 51 (36.4) | |||
| RAI-related domains | Pre-RAI-Tg (ng/mL) | (0–5) | 392 (61.7) | 364 (73.5) | 28 (20.0) | <0.001 |
| (5–10) | 75 (11.8) | 63 (12.7) | 12 (8.6) | |||
| (10–50) | 90 (14.2) | 59 (11.9) | 31 (22.1) | |||
| >50 | 78 (12.3) | 9 (1.8) | 69 (49.3) | |||
| Post-therapeutic 131I WBS | R | 549 (86.5) | 473 (95.6) | 76 (54.3) | <0.001 | |
| Outside thyroid bed | 55 (8.7) | 21 (4.2) | 34 (24.3) | |||
| Distant metastases | 31 (4.9) | 1 (0.2) | 30 (21.4) | |||
| Prognostic Parameters | Model Estimates | |
|---|---|---|
| Hazard Ratio * (95%CI) | p-Value | |
| Progression-Free Survival | ||
| Histology | ||
| Papillary | 1 (reference) | |
| Follicular | 1.57 (1.0–2.57) | 0.047 |
| 2015 ATA risk category | ||
| Low-Risk | 1 (reference) | |
| Intermediate-Risk | 4.25 (2.52–7.14) | <0.001 |
| High-Risk | 4.52 (2.45–8.33) | <0.001 |
| Tg measured before RAI in THW (ng/mL) | ||
| (0–5) | 1 (reference) | |
| (5–10) | 1.47 (0.72–2.97) | 0.28 |
| (10–50) | 3.58 (2.11–6.05) | <0.001 |
| >50 | 7.15 (4.11–12.44) | <0.001 |
| Post-therapeutic WBS findings | ||
| Only remnant uptake | 1 (reference) | |
| Uptake outside thyroid bed | 2.49 (1.56–3.96) | <0.001 |
| Distant metastases | 3.23 (1.76–5.92) | <0.001 |
| Domains | Risk Factors | Levels | Score | |||
|---|---|---|---|---|---|---|
| Demographic | Age | <55 | ≥55 | |||
| 0 | 4 | |||||
| Sex | Female | Male | ||||
| 0 | 8 | |||||
| Surgical | Histology | Papillary Thyroid cancer | Follicular Thyroid cancer | |||
| 0 | 8 | |||||
| ATA 2015 | Low | Intermediate | High | |||
| 0 | 25 | 26 | ||||
| RAI related | Tg | (0–5) | (5–10) | (10–50) | ≥50 | |
| 0 | 7 | 22 | 34 | |||
| PT-WBS | Only remnant uptake | Uptake outside the thyroid bed | Distant metastases | |||
| 0 | 16 | 20 | ||||
| TOTAL | ||||||
| Risk categories | Mild risk | Moderate risk | Severe risk | |||
| 0–33 | 34–66 | 67–100 | ||||
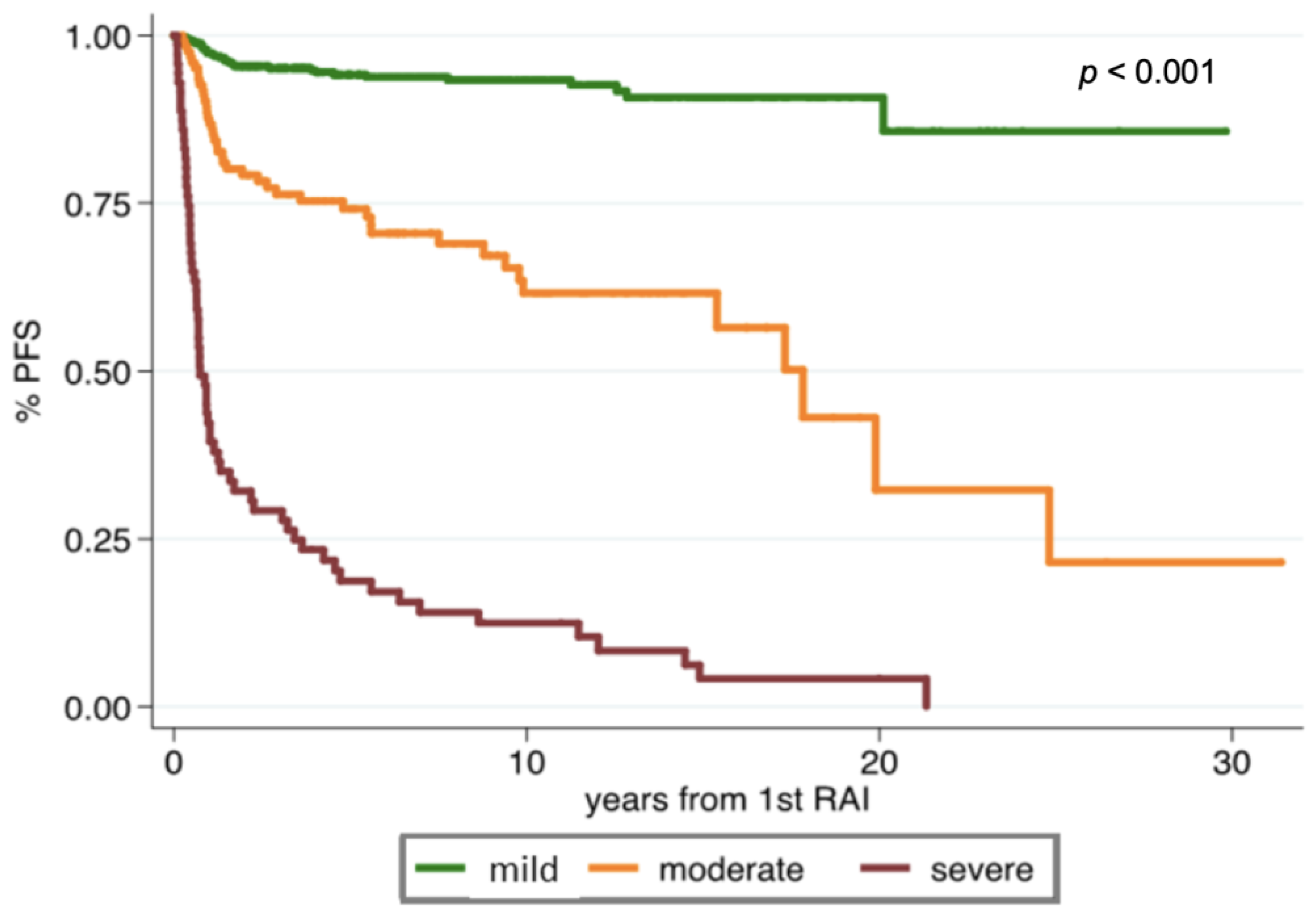
| Median PFS years (95%CI) | 25.7 (23.4–28.1) | 12.8 (8.5–17.1) | 0.7 (0.0–2.2) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piccardo, A.; Siri, G.; Ugolini, M.; Fiz, F.; Puntoni, M.; Bottoni, G.; Catrambone, U.; Pitoia, F.; Trimboli, P. A Three-Domain Scoring System to Customize the Risk of Relapse of Differentiated Thyroid Carcinoma. Cancers 2021, 13, 4335. https://doi.org/10.3390/cancers13174335
Piccardo A, Siri G, Ugolini M, Fiz F, Puntoni M, Bottoni G, Catrambone U, Pitoia F, Trimboli P. A Three-Domain Scoring System to Customize the Risk of Relapse of Differentiated Thyroid Carcinoma. Cancers. 2021; 13(17):4335. https://doi.org/10.3390/cancers13174335
Chicago/Turabian StylePiccardo, Arnoldo, Giacomo Siri, Martina Ugolini, Francesco Fiz, Matteo Puntoni, Gianluca Bottoni, Ugo Catrambone, Fabián Pitoia, and Pierpaolo Trimboli. 2021. "A Three-Domain Scoring System to Customize the Risk of Relapse of Differentiated Thyroid Carcinoma" Cancers 13, no. 17: 4335. https://doi.org/10.3390/cancers13174335
APA StylePiccardo, A., Siri, G., Ugolini, M., Fiz, F., Puntoni, M., Bottoni, G., Catrambone, U., Pitoia, F., & Trimboli, P. (2021). A Three-Domain Scoring System to Customize the Risk of Relapse of Differentiated Thyroid Carcinoma. Cancers, 13(17), 4335. https://doi.org/10.3390/cancers13174335







