Kidney Dysfunction Is Associated with Thrombosis and Disease Severity in Myeloproliferative Neoplasms: Implications from the German Study Group for MPN Bioregistry
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Study Population
2.2. Assessment of Kidney Function
2.3. Statistical Methods
3. Results
3.1. General Characteristics of Study Cohort
3.2. Differences among the eGFR Subgroups
3.2.1. Thrombosis and Bleeding
3.2.2. Comorbidities
3.2.3. Uric Acid and LDH Levels
3.2.4. Blood Counts and C-Reactive Protein
3.2.5. Driver Mutations
3.2.6. MPN Duration
3.3. Risk Factors for Decreased Kidney Function
3.4. Risk Factors for Developing Thrombosis and Bleeding in the Presence of Kidney Dysfunction
3.5. Treatment with Hydroxyurea, Ruxolitinib, and Anticoagulants Are Associated with Kidney Dysfunction
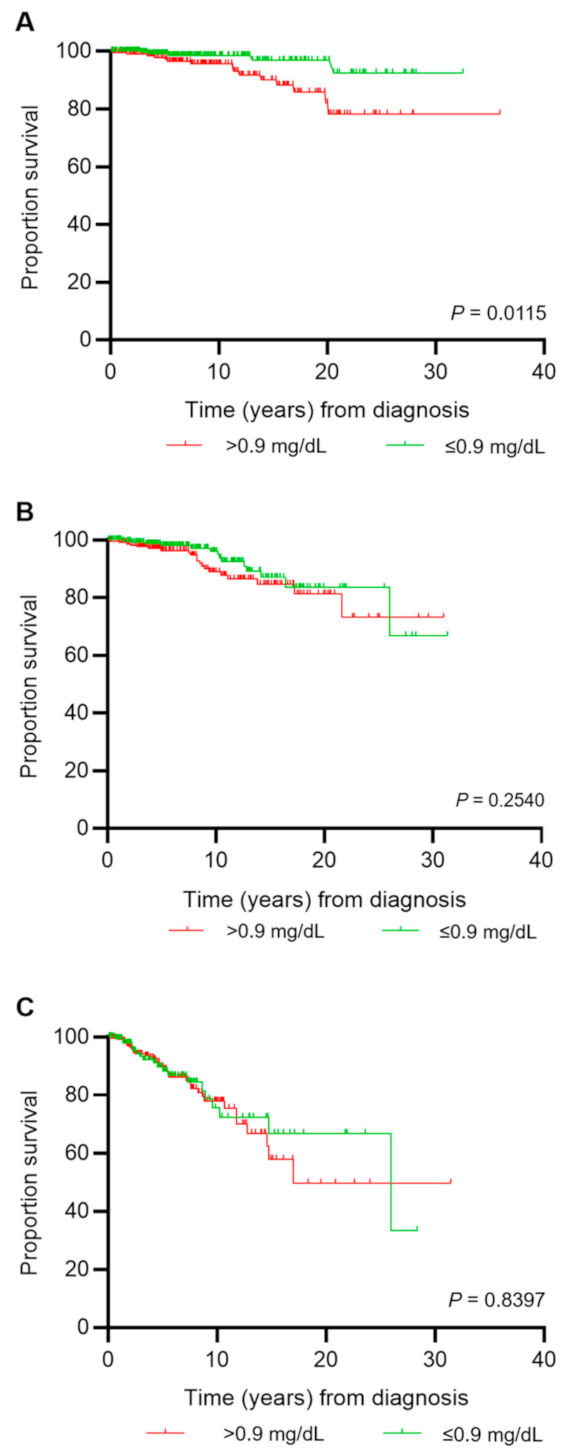
3.6. Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADL | Activities of daily living |
| ASA | Acetylsalicylic acid |
| CI | Confidence interval |
| CKD | Chronic kidney disease |
| CRP | C-reactive protein |
| ECOG PS | Eastern Cooperative Oncology Group Performance Status |
| eGFR | estimated glomerular filtration rate |
| ET | Essential thrombocythemia |
| FU | Follow-up |
| GSG-MPN bioregistry | Myeloproliferative neoplasms bioregistry study of the German Study Group for myeloproliferative neoplasms |
| HR | Hazard ratio |
| HU | Hydroxyurea |
| IMIDs | Immunomodulatory drugs |
| IWG-MRT | International Working Group-Myeloproliferative Neoplasms Research and Treatment |
| KDIGO | Kidney Disease Improving Global Outcomes |
| LDH | Lactate dehydrogenase |
| MDRD | Modification of Diet in Renal Disease |
| MF | Myelofibrosis |
| MPN | Myeloproliferative neoplasms |
| OR | Odds ratio |
| PMF | Primary myelofibrosis |
| PV | Polycythemia vera |
| Q1 | First quartile |
| Q3 | Third quartile |
| RUX | Ruxolitinib |
| Scr | Serum creatinine |
| WHO | World Health Organization |
| WW | Watch-and-wait |
References
- Barbui, T.; Finazzi, G.; Falanga, A. Myeloproliferative neoplasms and thrombosis. Blood 2013, 122, 2176–2184. [Google Scholar] [CrossRef] [PubMed]
- Hultcrantz, M.; Björkholm, M.; Dickman, P.W.; Landgren, O.; Derolf, Å.R.; Kristinsson, S.Y.; Andersson, T.M.L. Risk for Arterial and Venous Thrombosis in Patients With Myeloproliferative Neoplasms: A Population-Based Cohort Study. Ann. Intern. Med. 2018, 168, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Kaifie, A.; Kirschner, M.; Wolf, D.; Maintz, C.; Hänel, M.; Gattermann, N.; Gökkurt, E.; Platzbecker, U.; Hollburg, W.; Göthert, J.R.; et al. Bleeding, thrombosis, and anticoagulation in myeloproliferative neoplasms (MPN): Analysis from the German SAL-MPN-registry. J. Hematol. Oncol. 2016, 9, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McMahon, B.; Stein, B.L. Thrombotic and bleeding complications in classical myeloproliferative neoplasms. Semin. Thromb. Hemost. 2013, 39, 101–111. [Google Scholar] [PubMed]
- Reikvam, H.; Tiu, R.V. Venous thromboembolism in patients with essential thrombocythemia and polycythemia vera. Leukemia 2012, 26, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Price, G.L.; Davis, K.L.; Karve, S.; Pohl, G.; Walgren, R.A. Survival patterns in United States (US) medicare enrollees with non-CML myeloproliferative neoplasms (MPN). PLoS ONE 2014, 9, e90299. [Google Scholar] [CrossRef]
- Campbell, P.J.; MacLean, C.; Beer, P.A.; Buck, G.; Wheatley, K.; Kiladjian, J.-J.; Forsyth, C.; Harrison, C.N.; Green, A. Correlation of blood counts with vascular complications in essential thrombocythemia: Analysis of the prospective PT1 cohort. Blood 2012, 120, 1409–1411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carobbio, A.; Thiele, J.; Passamonti, F.; Rumi, E.; Ruggeri, M.; Rodeghiero, F.; Randi, M.L.; Bertozzi, I.; Vannucchi, A.M.; Antonioli, E.; et al. Risk factors for arterial and venous thrombosis in WHO-defined essential thrombocythemia: An international study of 891 patients. Blood 2011, 117, 5857–5859. [Google Scholar] [CrossRef]
- Griesshammer, M.; Kiladjian, J.-J.; Besses, C. Thromboembolic events in polycythemia vera. Ann. Hematol. 2019, 98, 1071–1082. [Google Scholar] [CrossRef] [Green Version]
- Landolfi, R.; Di Gennaro, L.; Barbui, T.; De Stefano, V.; Finazzi, G.; Marfisi, R.; Tognoni, G.; Marchioli, R.; European Collaboration on Low-Dose Aspirin in Polycythemia Vera (ECLAP). Leukocytosis as a major thrombotic risk factor in patients with polycythemia vera. Blood 2007, 109, 2446–2452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vannucchi, A.M.; Guglielmelli, P. JAK2 mutation-related disease and thrombosis. Semin. Thromb. Hemost. 2013, 39, 496–506. [Google Scholar] [PubMed]
- Zhang, Y.; Zhou, Y.; Wang, Y.; Teng, G.; Li, D.; Wang, Y.; Du, C.; Chen, Y.; Zhang, H.; Li, Y.; et al. Thrombosis among 1537 patients with JAK2(V617F) -mutated myeloproliferative neoplasms: Risk factors and development of a predictive model. Cancer Med. 2020, 9, 2096–2105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keith, D.S.; Nichols, G.A.; Gullion, C.M.; Brown, J.B.; Smith, D.H. Longitudinal follow-up and outcomes among a population with chronic kidney disease in a large managed care organization. Arch. Intern. Med. 2004, 164, 659–663. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Li, X.-C.; Lu, L.; Cao, Y.; Sun, R.-R.; Chen, S.; Zhang, P.-Y. Cardiovascular disease and its relationship with chronic kidney disease. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 2918–2926. [Google Scholar] [PubMed]
- Mathew, R.O.; Bangalore, S.; Lavelle, M.P.; Pellikka, P.A.; Sidhu, M.S.; Boden, W.E.; Asif, A. Diagnosis and management of atherosclerotic cardiovascular disease in chronic kidney disease: A review. Kidney Int. 2017, 91, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Provenzano, M.; Coppolino, G.; De Nicola, L.; Serra, R.; Garofalo, C.; Andreucci, M.; Bolignano, D. Unraveling Cardiovascular Risk in Renal Patients: A New Take on Old Tale. Front. Cell Dev. Biol. 2019, 7, 314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tonelli, M.; Wiebe, N.; Culleton, B.; House, A.; Rabbat, C.; Fok, M.; McAlister, F.; Garg, A.X. Chronic Kidney Disease and Mortality Risk: A Systematic Review. J. Am. Soc. Nephrol. 2006, 17, 2034–2047. [Google Scholar] [CrossRef] [Green Version]
- Hemmelgarn, B.R.; Manns, B.J.; Lloyd, A.; James, M.T.; Klarenbach, S.; Quinn, R.R.; Wiebe, N.; Tonelli, M.; Alberta Kidney Disease Network. Relation between kidney function, proteinuria, and adverse outcomes. JAMA 2010, 303, 423–429. [Google Scholar] [CrossRef] [Green Version]
- Ocak, G.; Lijfering, W.M.; Verduijn, M.; Dekker, F.; Rosendaal, F.R.; Cannegieter, S.C.; Vossen, C.Y. Risk of venous thrombosis in patients with chronic kidney disease: Identification of high-risk groups. J. Thromb. Haemost. 2013, 11, 627–633. [Google Scholar] [CrossRef]
- Menon, V.; Greene, T.; Wang, X.; Pereira, A.A.; Marcovina, S.M.; Beck, G.J.; Kusek, J.W.; Collins, A.J.; Levey, A.S.; Sarnak, M.J. C-reactive protein and albumin as predictors of all-cause and cardiovascular mortality in chronic kidney disease. Kidney Int. 2005, 68, 766–772. [Google Scholar] [CrossRef] [Green Version]
- Asaba, K.; Tojo, A.; Onozato, M.L.; Mimura, N.; Kido, M.; Goto, A.; Endo, H.; Fujita, T. Fibrillary glomerulonephritis associated with essential thrombocytosis. Clin. Exp. Nephrol. 2003, 7, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, S.; Hamai, K.; Fujishima, M.; Ohtani, H.; Komatsuda, A.; Sawada, K.; Wakui, H. Focal segmental glomerulosclerosis associated with polycythemia vera: Report of a case and review of the literature. Clin. Nephrol. 2007, 68, 412–415. [Google Scholar] [CrossRef]
- Rahimian, S.; Johnson, T.; Herb, R. A Case of Essential Thrombocythemia and IgA Nephropathy with Literature Review of the Concurrence. Case Rep. Oncol. Med. 2019, 2019, 5086963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christensen, A.S.; Møller, J.B.; Hasselbalch, H.C. Chronic kidney disease in patients with the Philadelphia-negative chronic myeloproliferative neoplasms. Leuk. Res. 2014, 38, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.-W.; Moon, J.Y.; Ryu, H.; Choi, Y.-S.; Song, I.-C.; Lee, H.-J.; Yun, H.J.; Kim, S.; Jo, D.-Y. Chronic kidney disease in the BCR-ABL1-negative myeloproliferative neoplasm: A single-center retrospective study. Korean J. Intern. Med. 2018, 33, 790–797. [Google Scholar] [CrossRef] [PubMed]
- Krečak, I.; Holik, H.; Martina, M.P.; Zekanović, I.; Coha, B.; Gverić-Krečak, V. Chronic kidney disease could be a risk factor for thrombosis in essential thrombocythemia and polycythemia vera. Int. J. Hematol. 2020, 112, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Lucijanic, M.; Galusic, D.; Krecak, I.; Sedinic, M.; Holik, H.; Perisa, V.; Peric, M.M.; Zekanovic, I.; Stoos-Veic, T.; Kusec, R. Reduced renal function strongly affects survival and thrombosis in patients with myelofibrosis. Ann. Hematol. 2020, 99, 2779–2785. [Google Scholar] [CrossRef]
- Strati, P.; Abdelrahim, M.; Selamet, U.; Page, V.D.; Pierce, S.A.; Verstovsek, S.; Abudayyeh, A. Ruxolitinib therapy is associated with improved renal function in patients with primary myelofibrosis. Ann. Hematol. 2019, 98, 1611–1616. [Google Scholar] [CrossRef]
- Fukuda, Y.; Araki, M.; Yamamoto, K.; Morishita, S.; Inano, T.; Misawa, K.; Ochiai, T.; Edahiro, Y.; Imai, M.; Yasuda, H.; et al. Evidence for prevention of renal dysfunction associated with primary myelofibrosis by cytoreductive therapy. Haematologica 2019, 104, e506–e509. [Google Scholar] [CrossRef] [PubMed]
- Tefferi, A.; Vardiman, J.W. Classification and diagnosis of myeloproliferative neoplasms: The 2008 World Health Organization criteria and point-of-care diagnostic algorithms. Leukemia 2008, 22, 14–22. [Google Scholar] [CrossRef] [Green Version]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., III; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Lente, F.V.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.; Stevens, P.E. Summary of KDIGO 2012 CKD Guideline: Behind the scenes, need for guidance, and a framework for moving forward. Kidney Int. 2014, 85, 49–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bar, C.; Huber, N.; Beier, F.; Blasco, M.A. Therapeutic effect of androgen therapy in a mouse model of aplastic anemia produced by short telomeres. Haematologica 2015, 100, 1267–1274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Passamonti, F.; Thiele, J.; Girodon, F.; Rumi, E.; Carobbio, A.; Gisslinger, H.; Kvasnicka, H.M.; Ruggeri, M.; Randi, M.L.; Gangat, N.; et al. A prognostic model to predict survival in 867 World Health Organization-defined essential thrombocythemia at diagnosis: A study by the International Working Group on Myelofibrosis Research and Treatment. Blood 2012, 120, 1197–1201. [Google Scholar] [CrossRef] [PubMed]
- Rumi, E.; Pietra, D.; Ferretti, V.; Klampfl, T.; Harutyunyan, A.S.; Milosevic, J.D.; Them, N.C.C.; Berg, T.; Elena, C.; Casetti, I.C.; et al. JAK2 or CALR mutation status defines subtypes of essential thrombocythemia with substantially different clinical course and outcomes. Blood 2014, 123, 1544–1551. [Google Scholar] [CrossRef] [PubMed]
- Arellano-Rodrigo, E.; Alvarez-Larran, A.; Reverter, J.C.; Colomer, D.; Villamor, N.; Bellosillo, B.; Cervantes, F. Platelet turnover, coagulation factors, and soluble markers of platelet and endothelial activation in essential thrombocythemia: Relationship with thrombosis occurrence and JAK2 V617F allele burden. Am. J. Hematol. 2009, 84, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Belotti, A.; Elli, E.; Speranza, T.; Lanzi, E.; Pioltelli, P.; Pogliani, E. Circulating endothelial cells and endothelial activation in essential thrombocythemia: Results from CD146+ immunomagnetic enrichment—Flow cytometry and soluble E-selectin detection. Am. J. Hematol. 2012, 87, 319–320. [Google Scholar] [CrossRef] [PubMed]
- Hauschner, H.; Horev, M.B.; Misgav, M.; Nagar, M.; Seligsohn, U.; Rosenberg, N.; Koren-Michowitz, M. Platelets from Calreticulin mutated essential thrombocythemia patients are less reactive than JAK2 V617F mutated platelets. Am. J. Hematol. 2019, 95, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Yamagata, K.; Ishida, K.; Sairenchi, T.; Takahashi, H.; Ohba, S.; Shiigai, T.; Narita, M.; Koyama, A. Risk factors for chronic kidney disease in a community-based population: A 10-year follow-up study. Kidney Int. 2007, 71, 159–166. [Google Scholar] [CrossRef] [Green Version]
- Coresh, J.; Astor, B.C.; Greene, T.; Eknoyan, G.; Levey, A.S. Prevalence of chronic kidney disease and decreased kidney function in the adult US population: Third National Health and Nutrition Examination Survey. Am. J. Kidney Dis. 2003, 41, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rifkin, D.E.; Shlipak, M.G.; Katz, R.; Fried, L.F.; Siscovick, D.; Chonchol, M.; Newman, A.B.; Sarnak, M.J. Rapid kidney function decline and mortality risk in older adults. Arch. Intern. Med. 2008, 168, 2212–2218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hapca, S.; Siddiqui, M.K.; Kwan, R.S.; Lim, M.; Matthew, S.; Doney, A.S.; Pearson, E.; Palmer, C.; Bell, S.; BEAt-DKD Consortium. The Relationship between AKI and CKD in Patients with Type 2 Diabetes: An Observational Cohort Study. J. Am. Soc. Nephrol. JASN 2020, 32, 138–150. [Google Scholar] [CrossRef]
- Patschan, D.; Muller, G.A. Acute Kidney Injury in Diabetes Mellitus. Int. J. Nephrol. 2016, 2016, 6232909. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skov, V.; Larsen, T.S.; Thomassen, M.; Riley, C.H.; Jensen, M.K.; Bjerrum, O.W.; Kruse, T.A.; Hasselbalch, H. Molecular profiling of peripheral blood cells from patients with polycythemia vera and related neoplasms: Identification of deregulated genes of significance for inflammation and immune surveillance. Leuk. Res. 2012, 36, 1387–1392. [Google Scholar] [CrossRef]
- Kato, S.; Chmielewski, M.; Honda, H.; Pecoits-Filho, R.; Matsuo, S.; Yuzawa, Y.; Tranaeus, A.; Stenvinkel, P.; Lindholm, B. Aspects of immune dysfunction in end-stage renal disease. Clin. J. Am. Soc. Nephrol. 2008, 3, 1526–1533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bazeley, J.; Bieber, B.; Li, Y.; Morgenstern, H.; De Sequera, P.; Combe, C.; Yamamoto, H.; Gallagher, M.; Port, F.K.; Robinson, B.M. C-reactive protein and prediction of 1-year mortality in prevalent hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2011, 6, 2452–2461. [Google Scholar] [CrossRef] [Green Version]
- Obermayr, R.P.; Temml, C.; Gutjahr, G.; Knechtelsdorfer, M.; Oberbauer, R.; Klauser-Braun, R. Elevated uric acid increases the risk for kidney disease. J. Am. Soc. Nephrol. 2008, 19, 2407–2413. [Google Scholar] [CrossRef] [Green Version]
- Brosius, F.C., 3rd; He, J.C. JAK inhibition and progressive kidney disease. Curr. Opin. Nephrol. Hypertens. 2015, 24, 88–95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brosius, F.C.; Tuttle, K.R.; Kretzler, M. JAK inhibition in the treatment of diabetic kidney disease. Diabetologia 2016, 59, 1624–1627. [Google Scholar] [CrossRef] [PubMed]
- Tabarroki, A.; Visconte, V.; Rogers, H.J.; Sekeres, M.A.; Samaras, C.; Lichtin, A.; Duong, H.K.; Englehaupt, R.; Cinalli, T.; Dodd, K.; et al. Clinical experiences with ruxolitinib in symptomatic patients with myeloproliferative neoplasm with chronic kidney disease. Leuk. Lymphoma. 2014, 55, 213–216. [Google Scholar] [CrossRef]
- Strati, P.; Glass, W.F.; Abdelrahim, M.; Selamet, U.; Tchakarov, A.; Workeneh, B.; Verstovsek, S.; Abudayyeh, A. Renal complications of primary myelofibrosis. Leuk. Lymphoma 2019, 60, 507–510. [Google Scholar] [CrossRef] [PubMed]
- Elliott, M.A.; Tefferi, A. Thrombosis and haemorrhage in polycythaemia vera and essential thrombocythaemia. Br. J. Haematol. 2005, 128, 275–290. [Google Scholar] [CrossRef] [PubMed]
- Kreher, S.; Ochsenreither, S.; Trappe, R.; Pabinger, I.; Bergmann, F.; Petrides, P.E.; Koschmieder, S.; Matzdorff, A.; Tiede, A.; Griesshammer, M.; et al. Prophylaxis and management of venous thromboembolism in patients with myeloproliferative neoplasms: Consensus statement of the Haemostasis Working Party of the German Society of Hematology and Oncology (DGHO), the Austrian Society of Hematology and Oncology (OGHO) and Society of Thrombosis and Haemostasis Research (GTH e.V.). Ann. Hematol. 2014, 93, 1953–1963. [Google Scholar] [PubMed]
- Carobbio, A.; Ferrari, A.; Masciulli, A.; Ghirardi, A.; Barosi, G.; Barbui, T. Leukocytosis and thrombosis in essential thrombocythemia and polycythemia vera: A systematic review and meta-analysis. Blood Adv. 2019, 3, 1729–1737. [Google Scholar] [CrossRef] [PubMed]
- Ronner, L.; Podoltsev, N.; Gotlib, J.; Heaney, M.L.; Kuykendall, A.T.; O’Connell, C.; Shammo, J.; Fleischman, A.G.; Scherber, R.M.; Mesa, R.; et al. Persistent leukocytosis in polycythemia vera is associated with disease evolution but not thrombosis. Blood 2020, 135, 1696–1703. [Google Scholar] [CrossRef]
- Madero, M.; Sarnak, M.J.; Stevens, L.A. Serum cystatin C as a marker of glomerular filtration rate. Curr. Opin. Nephrol. Hypertens. 2006, 15, 610–616. [Google Scholar] [CrossRef] [PubMed]
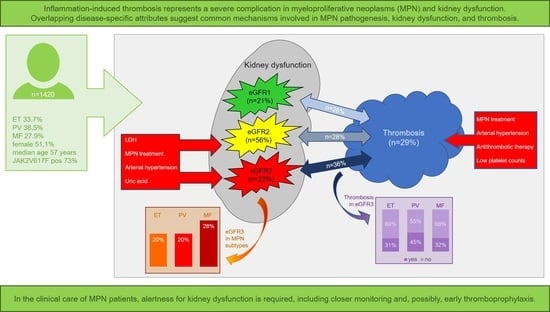
| Parameters (Available n) | All MPN | ET | PV | MF | P | |
|---|---|---|---|---|---|---|
| Overall | Between MPN Subtypes | |||||
| Diagnosis (n) | 1420 | 546 | 478 | 396 | - | - |
| Fractional Parameters; % | ||||||
| MPN subtype (n = 1420) | 100 | 38.5 | 33.7 | 27.9 | - | - |
| Male gender (n = 1420) | 48.9 | 41.6 | 49.8 | 57.8 | <0.0001 | EvP |
| JAK2V617F mutation pos (n = 1315) | 73.3 | 63.2 | 92.3 | 63.5 | <0.0001 | EvP/PvM |
| HU-treated (n = 1215) *,† | 39.6 | 35.9 | 53.4 | 28.9 | <0.0001 | EvP/PvM |
| RUX-treated (n = 1215) *,‡ | 18.8 | 4.7 | 16.7 | 40.1 | <0.0001 | EvP/EvM/PvM |
| Other MPN treatment (n = 1215) § | 21.3 | 30.2 | 12.4 | 19.5 | <0.0001 | EvP/EvM/PvM |
| ASA-treated (n = 1252) | 57.7 | 62.8 | 66.8 | 40.0 | <0.0001 | EvM/PvM |
| P2Y12-antagonist-treated (n = 1208) || | 5.3 | 8.1 | 4.2 | 2.7 | 0.0014 | EvM |
| Anticoagulant-treated (n = 1420) ¶ | 11.6 | 9.5 | 15.5 | 9.6 | 0.0061 | EvP/PvM |
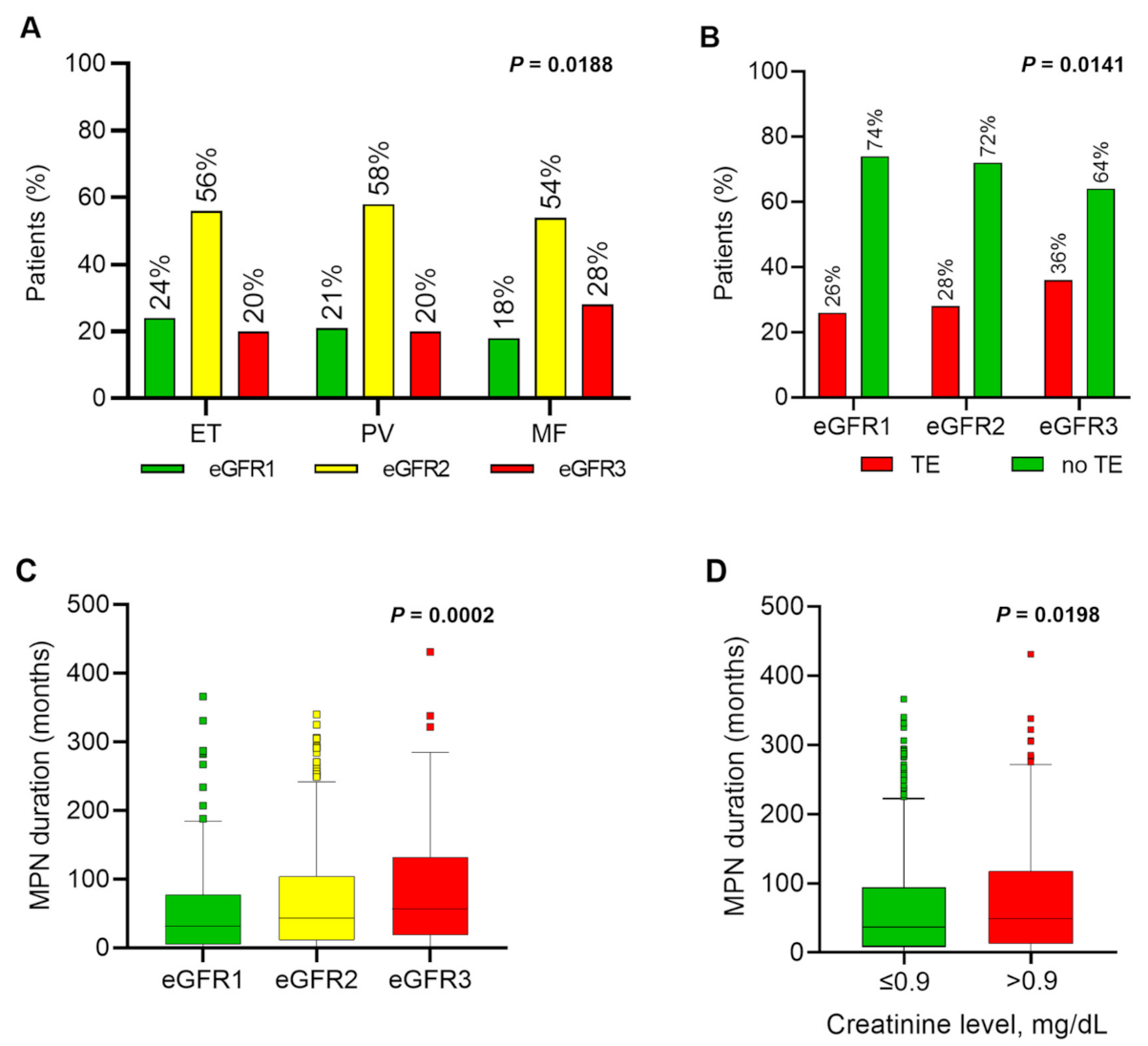
| Kidney function by eGFR subgroup (n = 1420) | - | - | - | - | 0.0188 | - |
| eGFR1 (n = 304) | 21.4 | 23.6 | 21.3 | 18.4 | 0.1589 | - |
| eGFR2 (n = 799) | 56.3 | 56.4 | 58.4 | 53.5 | 0.3565 | - |
| eGFR3 (n = 317) | 22.3 | 20.0 | 20.3 | 28.0 | 0.0057 | EvM/PvM |
| Thromboembolism, all (n = 1420) | 29.2 | 26.0 | 33.7 | 28.0 | 0.0223 | EvP |
| Arterial TE (n = 1409) | 18.5 | 17.6 | 22.2 | 15.4 | 0.0287 | PvM |
| Venous TE (n = 1409) | 12.3 | 9.7 | 14.2 | 13.6 | 0.0581 | - |
| Severe hemorrhage (n = 1420) | 3.9 | 3.1 | 4.2 | 4.6 | 0.4841 | - |
| Diabetes mellitus (n = 1251) | 7.8 | 6.4 | 9.5 | 7.5 | 0.2130 | - |
| Arterial hypertension | 49.0 | 45.6 | 62.8 | 38.6 | <0.0001 | EvP/PvM |
| Hyperlipidemia | 11.6 | 10.6 | 17.0 | 8.8 | 0.383 | - |
| ECOG-score (n = 985) #, ** | - | - | - | - | 0.0013 | EvM †† |
| ECOG 0 (n = 683) | 69.3 | 75.1 | 70.9 | 60.1 | - | - |
| ECOG 1 (n = 270) | 27.4 | 22.2 | 27.7 | 34.0 | - | - |
| ECOG 2 (n = 25) | 2.5 | 2.4 | 1.2 | 4.2 | - | - |
| ECOG 3 (n = 7) | 0.7 | 0.3 | 0.3 | 1.8 | - | - |
| Numeric Parameters; median Q1, Q3 | ||||||
| Age diagnosis, years (n = 1416) | 57 47, 68 | 54 43, 65 | 59 49, 70 | 60 50, 69 | <0.0001 | EvP/EvM |
| Age creatinine test, years (n = 1420) | 62 51, 72 | 59 48, 70 | 65 54, 74 | 63 54, 73 | <0.0001 | EvP/EvM |
| Age at first MPN treatment, years (n = 1215) | 60 49, 70 | 58 45, 67 | 63 52, 72 | 60 50, 71 | 0.0001 | EvP |
| MPN duration, months (n = 1183) | 42 11, 105 | 46 12, 125 | 57 14, 130 | 29 7, 68 | <0.0001 | EvM/PvM |
| Leukocytes, G/L (n = 1408) | 8.4 6.1, 11.7 | 8.2 6.2, 10.3 | 9.0 6.8, 12.8 | 7.9 5.4, 12.9 | <0.0001 | EvP/PvM |
| Platelets, G/L (n = 1395) | 492 290, 734 | 678 502, 924 | 419 278, 604 | 336 159, 547 | <0.0001 | EvP/EvM/PvM |
| Creatinine, mg/dL (n = 1420) | 0.9 0.8, 1.1 | 0.9 0.8, 1.0 | 0.90 0.8, 1.1 | 1.0 0.8, 1.2 | <0.0001 | EvM/PvM |
| eGFR, mL/min/1.73 m2 (n = 1420) | 74 62, 87 | 77 64, 89 | 74 63, 87 | 72 58, 86 | 0.0280 | EvM |
| Uric acid, mg/dL (n = 1012) | 5.7 4.5, 6.9 | 5.2 4.3, 6.3 | 5.9 4.7, 7.1 | 6.15 5.0, 7.3 | <0.0001 | EvP/EvM |
| LDH, U/L (n = 1356) | 268 15, 395 | 239 198, 293 | 258 213, 342 | 435 279, 698 | <0.0001 | EvP/EvM/PvM |
| CRP, mg/L (n = 356) | 1.4 0.6, 4.5 | 1.1 0.5, 3.8 | 1.3 0.6, 3.3 | 2.30 0.9, 5.7 | 0.0015 | EvM/PvM |
| Risk Factors for Decreased Kidney Function | Odds Ratio | 95% CI | P |
|---|---|---|---|
| Univariate regression (n = 1420) | |||
| Diagnosis (n = 1420) | - | - | 0.0065 |
| ET vs. MF (n = 942) | 0.674 | 0.525–0.867 | 0.0021 |
| ET vs. PV (n = 1024) | 0.926 | 0.730–1.174 | 0.5243 |
| MF vs. PV (n = 874) | 1.372 | 1.060–1.777 | 0.0160 |
| JAK2V617F mutation (yes vs. no; n = 1315) | 1.244 | 0.981–1.576 | 0.0711 |
| eGFR2 vs. eGFR1 (n = 1030) | 1.033 | 0.764–1.395 | 0.8347 |
| eGFR3 vs. eGFR1 (n = 576) | 1.435 | 0.982–2.097 | 0.0619 |
| eGFR3 vs. eGFR2 (n = 1024) | 1.390 | 1.005–1.922 | 0.0464 |
| Diabetes mellitus (yes vs. no; n = 1251) | 1.815 | 1.215–2.711 | 0.0036 |
| eGFR2 vs. eGFR1 (n = 976) | 0.663 | 0.382–1.153 | 0.1454 |
| eGFR3 vs. eGFR1 (n = 542) | 1.878 | 1.071–3.295 | 0.0279 |
| eGFR3 vs. eGFR2 (n = 984) | 2.830 | 1.763–4.543 | <0.0001 |
| Arterial hypertension (yes vs. no; n = 956) | 2.419 | 1.879–3.114 | <0.0001 |
| eGFR2 vs. eGFR1 (n = 739) | 1.405 | 1.016–1.943 | 0.0399 |
| eGFR3 vs. eGFR1 (n = 436) | 3.983 | 2.671–5.939 | <0.0001 |
| eGFR3 vs. eGFR2 (n = 737) | 2.836 | 2.023–3.976 | <0.0001 |
| Hyperlipidemia (yes vs. no; n = 181) | 3.570 | 1.445–8.819 | 0.0058 |
| eGFR2 vs. eGFR1 (n = 152) | 1.815 | 0.477–6.905 | 0.3820 |
| eGFR3 vs. eGFR1 (n = 81) | 6.220 | 1.501–25.779 | 0.0118 |
| eGFR3 vs. eGFR2 (n = 129) | 3.429 | 1.207–9.739 | 0.0207 |
| Leukocytes (>8.4 vs. ≤8.4 G/L; n = 1408) | 1.367 | 1.116–1.675 | 0.0025 |
| eGFR2 vs. eGFR1 (n = 1093) | 1.023 | 0.785–1.333 | 0.8670 |
| eGFR3 vs. eGFR1 (n = 619) | 1.624 | 1.181–2.231 | 0.0028 |
| eGFR3 vs. eGFR2 (n = 1104) | 1.587 | 1.218–2.067 | 0.0006 |
| Platelets (>492 vs. ≤492 G/L; n = 1395) | 0.832 | 0.679–1.019 | 0.0755 |
| eGFR2 vs. eGFR1 (n = 1080) | 1.078 | 0.827–1.406 | 0.5774 |
| eGFR3 vs. eGFR1 (n = 618) | 0.755 | 0.550–1.036 | 0.0814 |
| eGFR3 vs. eGFR2 (n = 1092) | 0.700 | 0.538–0.911 | 0.0079 |
| Uric acid (>5.7 vs. ≤5.7 mg/dL; n = 1012) | 2.548 | 1.986–3.268 | <0.0001 |
| eGFR2 vs. eGFR1 (n = 790) | 1.587 | 1.150–2.192 | 0.0050 |
| eGFR3 vs. eGFR1 (n = 444) | 4.351 | 2.923–6.476 | <0.0001 |
| eGFR3 vs. eGFR2 (n = 790) | 2.741 | 1.971–3.811 | <0.0001 |
| Absolute neutrophil count (>5.48 vs. ≤5.48 G/L; n = 1254) | 1.397 | 1.126–1.734 | 0.0024 |
| eGFR2 vs. eGFR1 (n = 981) | 1.077 | 0.814–1.426 | 0.6019 |
| eGFR3 vs. eGFR1 (n = 545) | 1.690 | 1.204–2.373 | 0.0024 |
| eGFR3 vs. eGFR2 (n = 982) | 1.569 | 1.183–2.081 | 0.0018 |
| Absolute monocyte count (>0.55 vs. ≤0.55 G/L; n = 1312) | 1.349 | 1.094–1.664 | 0.0052 |
| eGFR2 vs. eGFR1 (n = 1018) | 0.972 | 0.739–1.278 | 0.8382 |
| eGFR3 vs. eGFR1 (n = 579) | 1.589 | 1.144–2.207 | 0.0058 |
| eGFR3 vs. eGFR2 (n = 1027) | 1.635 | 1.243–2.150 | 0.0004 |
| LDH (>267.5 vs. ≤267.5 U/L; n = 1356) | 1.533 | 1.245–1.887 | <0.0001 |
| eGFR2 vs. eGFR1 (n = 1056) | 1.197 | 0.913–1.570 | 0.1940 |
| eGFR3 vs. eGFR1 (n = 592) | 1.949 | 1.405–2.703 | <0.0001 |
| eGFR3 vs. eGFR2 (n = 1064) | 1.628 | 1.241–2.136 | 0.0004 |
| CRP (>1.4 vs. ≤1.4 mg/L; n = 356) | 1.633 | 1.091–2.444 | 0.0172 |
| eGFR2 vs. eGFR1 (n = 273) | 0.980 | 0.579–1.660 | 0.9411 |
| eGFR3 vs. eGFR1 (n = 162) | 2.109 | 1.125–3.953 | 0.0200 |
| eGFR3 vs. eGFR2 (n = 277) | 2.151 | 1.269–3.645 | 0.0044 |
| MPN duration (>42 vs. ≤42 months; n = 1183) | 1.507 | 1.207–1.882 | 0.0003 |
| eGFR2 vs. eGFR1 (n = 904) | 1.482 | 1.101–1.996 | 0.0095 |
| eGFR3 vs. eGFR1 (n = 523) | 1.913 | 1.350–2.710 | 0.0003 |
| eGFR3 vs. eGFR2 (n = 939) | 1.290 | 0.973–1.710 | 0.0764 |
| MPN therapy (yes vs. WW; n = 1215) * | 1.784 | 1.428–2.230 | <0.0001 |
| eGFR2 vs. eGFR1 (n = 941) | 1.267 | 0.952–1.686 | 0.1049 |
| eGFR3 vs. eGFR1 (n = 535) | 2.514 | 1.762–3.587 | <0.0001 |
| eGFR3 vs. eGFR2 (n = 939) | 1.985 | 1.470–2.679 | <0.0001 |
| HU-treated (yes vs. WW; n = 780) | 1.734 | 1.286–2.338 | 0.0003 |
| eGFR2 vs. eGFR1 (n = 639) | 1.639 | 1.099–2.444 | 0.0153 |
| eGFR3 vs. eGFR1 (n = 316) | 2.421 | 1.493–3.926 | 0.0003 |
| eGFR3 vs. eGFR2 (n = 605) | 1.477 | 1.004–2.172 | 0.0475 |
| RUX-treated (yes vs. WW; n = 618) | 2.412 | 1.560–3.728 | <0.0001 |
| eGFR2 vs. eGFR1 (n = 506) | 1.517 | 0.810–2.842 | 0.1928 |
| eGFR3 vs. eGFR1 (n = 260) | 3.663 | 1.839–7.294 | 0.0002 |
| eGFR3 vs. eGFR2 (n = 470) | 2.413 | 1.446–4.027 | 0.0007 |
| Other MPN treatment (yes vs. WW; n = 865) | 1.737 | 1.331–2.267 | <0.0001 |
| eGFR2 vs. eGFR1 (n = 682) | 1.006 | 0.714–1.417 | 0.9734 |
| eGFR3 vs. eGFR1 (n = 389) | 2.344 | 1.557–3.527 | <0.0001 |
| eGFR3 vs. eGFR2 (n = 659) | 2.330 | 1.647–3.297 | <0.0001 |
| Antithrombotic therapy (yes vs. no; n = 1251) | 1.196 | 0.946–1.511 | 0.1348 |
| eGFR2 vs. eGFR1 (n = 967) | 1.296 | 0.958–1.752 | 0.0925 |
| eGFR3 vs. eGFR1 (n = 548) | 1.318 | 0.918–1.892 | 0.1314 |
| eGFR3 vs. eGFR2 (n = 939) | 1.017 | 0.750–1.380 | 0.9113 |
| Multiple Regression | |||
| eGFR2 vs. eGFR1 (n = 639) * | - | - | - |
| HU-treated (yes vs. WW) | 1.639 | 1.099–2.444 | 0.0153 |
| eGFR3 vs. eGFR1 (n = 278) †,‡ | - | - | - |
| Arterial hypertension (yes vs. no) | 3.073 | 1.746–5.405 | <0.0001 |
| Uric acid (>5.7 vs. ≤ 5.7 mg/dL) | 4.918 | 2.787–8.677 | <0.0001 |
| HU-treated (yes vs. WW) | 3.509 | 1.630–7.557 | 0.0013 |
| RUX-treated (yes vs. WW) | 5.416 | 1.927–15.223 | 0.0014 |
| Other MPN treatment (yes vs. WW) | 2.477 | 1.277–4.803 | 0.0073 |
| eGFR3 vs. eGFR2 (n = 589) § | - | - | - |
| Arterial hypertension (yes vs. no) | 2.004 | 1.440–2.789 | <0.0001 |
| Uric acid (>5.7 vs. ≤5.7 mg/dL) | 2.254 | 1.617–3.140 | <0.0001 |
| LDH (>267.5 vs. ≤267.5 U/L) | 1.448 | 1.036–2.023 | 0.0301 |
| HU-treated (yes vs. WW) | 1.797 | 1.162–2.780 | 0.0085 |
| Other MPN treatment (yes vs. WW) | 1.645 | 1.095–2.471 | 0.0165 |
| Risk Factors for Thromboembolic Event | Odds Ratio | 95% CI | P |
|---|---|---|---|
| Univariate regression (n = 1420) | |||
| eGFR group (n = 1420) | - | - | 0.0145 |
| eGFR3 vs. eGFR2 (n = 1116) | 1.449 | 1.098–1.912 | 0.0088 |
| eGFR3 vs. eGFR1 (n = 621) | 1.551 | 1.100–2.187 | 0.0123 |
| eGFR2 vs. eGFR1 (n = 1103) | 1.071 | 0.794–1.443 | 0.6544 |
| ET (n = 546) | - | - | 0.3744 |
| eGFR3 vs. eGFR2 (n = 417) | 1.408 | 0.870–2.280 | 0.1634 |
| eGFR3 vs. eGFR1 (n = 238) | 1.319 | 0.749–2.323 | 0.3381 |
| eGFR2 vs. eGFR1 (n = 437) | 0.936 | 0.583–1.503 | 0.7856 |
| PV (n = 478) | - | - | 0.0258 |
| eGFR3 vs. eGFR2 (n = 376) | 1.863 | 1.160–2.992 | 0.0100 |
| eGFR3 vs. eGFR1 (n = 199) | 1.901 | 1.063–3.400 | 0.0302 |
| eGFR2 vs. eGFR1 (n = 381) | 1.021 | 0.624–1.670 | 0.9355 |
| MF (n = 396) | - | - | 0.3649 |
| eGFR3 vs. eGFR2 (n = 323) | 1.167 | 0.708–1.923 | 0.5453 |
| eGFR3 vs. eGFR1 (n = 184) | 1.640 | 0.828–3.251 | 0.1561 |
| eGFR2 vs. eGFR1 (n = 285) | 1.406 | 0.749–2.640 | 0.2890 |
| Diagnosis (n = 1420) | - | - | 0.0227 |
| ET vs. PV (n = 1024) | 0.692 | 0.529–0.906 | 0.0074 |
| ET vs. MF (n = 942) | 0.902 | 0.675–1.207 | 0.4894 |
| MF vs. PV (n = 874) | 0.767 | 0.574–1.025 | 0.0727 |
| Sex (female vs. male; n = 1420) | 0.819 | 0.651–1.029 | 0.0869 |
| Age at diagnosis (>60 vs. ≤60 years; n = 1416) | 1.219 | 0.968–1.535 | 0.0924 |
| Age at creatinine test (>60 vs. ≤60 years; n = 1420) | 1.190 | 0.945–1.500 | 0.1397 |
| JAK2V617F mutation (yes vs. no; n = 1315) | 1.632 | 1.227–2.169 | 0.0007 |
| Diabetes mellitus (yes vs. no; n = 1251) | 1.245 | 0.803–1.930 | 0.3279 |
| Arterial hypertension (yes vs. no; n = 956) | 1.838 | 1.398–2.418 | <0.0001 |
| Hyperlipidemia (yes vs. no; n = 181) | 2.936 | 1.149–7.503 | 0.0245 |
| Leukocytes (>8.4 vs. ≤8.4 G/L; n = 1408) | 0.850 | 0.676–1.070 | 0.1668 |
| Platelets (>492 vs. ≤492 G/L; n = 1395) | 0.633 | 0.502–0.799 | 0.0001 |
| Uric acid (>5.7 vs. ≤5.7 mg/dL; n = 1012) | 1.166 | 0.888–1.531 | 0.2685 |
| Absolute neutrophil count (>5.48 vs. ≤5.48 G/L; n = 1254) | 0.807 | 0.632–1.030 | 0.0843 |
| Absolute monocyte count (>0.55 vs. ≤0.55 G/L; n = 1312) | 1.041 | 0.821–1.322 | 0.7385 |
| LDH (>267.5 vs. ≤267.5 U/L; n = 1356) | 1.246 | 0.986–1.574 | 0.0653 |
| CRP (>1.4 vs. ≤1.4 mg/L; n = 356) | 1.194 | 0.768–1.855 | 0.4308 |
| MPN therapy (yes vs. WW; n = 1215) | 1.516 | 1.183–1.944 | 0.0010 |
| HU-treated (yes vs. WW; n = 780) | 1.592 | 1.156–2.191 | 0.0043 |
| RUX-treated (yes vs. WW; n = 618) | 1.164 | 0.718–1.885 | 0.5382 |
| Other MPN treatment (yes vs. WW; n = 865) | 1.567 | 1.169–2.102 | 0.0027 |
| Antithrombotic therapy (yes vs. no; n = 1251) | 4.407 | 3.178–6.111 | <0.0001 |
| ASA (yes vs. no; n = 1029) | 2.622 | 1.862–3.694 | <0.0001 |
| P2Y12-antagonists (yes vs. no; n = 405) | 12.999 | 5.752–29.387 | <0.0001 |
| Anticoagulant-treated (yes vs. no; n = 497) | 22.867 | 13.583–38.498 | <0.0001 |
| Multiple regression (n = 911) *,† | |||
| Arterial hypertension (yes vs. no) | 1.800 | 1.349–2.401 | 0.0117 |
| Platelets (>492 vs. ≤492 G/L) | 0.699 | 0.524–0.931 | 0.0068 |
| ASA (yes vs. no) | 2.885 | 1.905–4.371 | <0.0001 |
| P2Y12-antagonists (yes vs. no) | 12.957 | 5.040–33.311 | <0.0001 |
| Anticoagulant-treated (yes vs. no) | 20.284 | 11.175–36.815 | <0.0001 |
| By Diagnosis | |||
| ET (n = 347) | - | - | - |
| Arterial hypertension (yes vs. no) | 1.913 | 1.099–3.330 | 0.0217 |
| RUX-treated (yes vs. WW) | 10.379 | 1.572–68.513 | 0.0151 |
| Other MPN treatment (yes vs. WW) | 2.013 | 1.108–3.656 | 0.0216 |
| ASA (yes vs. no) | 2.094 | 1.003 -4.374 | 0.0491 |
| P2Y12-antagonists (yes vs. no) | 12.235 | 3.362–44.524 | 0.0001 |
| Anticoagulant-treated (yes vs. no) | 29.068 | 9.167–92.171 | <0.0001 |
| PV (n = 377) | - | - | - |
| Other MPN treatment (yes vs. WW) | 2.270 | 1.250–4.120 | 0.0071 |
| Anticoagulant-treated (yes vs. no) | 12.732 | 5.271–30.756 | <0.0001 |
| MF (n = 267) | - | - | - |
| Arterial hypertension (yes vs. no) | 1.960 | 1.067–3.601 | 0.0301 |
| ASA (yes vs. no) | 5.546 | 2.837–10.843 | <0.0001 |
| Anticoagulant-treated (yes vs no) | 22.606 | 7.167–71.304 | <0.0001 |
| Risk Factors for Bleeding Event | ODDS RATIO | 95% CI | P |
|---|---|---|---|
| Univariate regression (n = 1420) | |||
| eGFR group (n = 1420) | - | - | 0.6469 |
| eGFR3 vs. eGFR2 (n = 1116) | 0.807 | 0.391–1.666 | 0.5624 |
| eGFR3 vs. eGFR1 (n = 621) | 0.675 | 0.295–1.543 | 0.3513 |
| eGFR2 vs. eGFR1 (n = 1103) | 0.836 | 0.438–1.594 | 0.5865 |
| ET (n = 546) | - | - | 0.2269 |
| eGFR3 vs. eGFR2 (n = 417) | 0.701 | 0.147–3.353 | 0.6563 |
| eGFR3 vs. eGFR1 (n = 238) | 0.326 | 0.066–1.602 | 0.1675 |
| eGFR2 vs. eGFR1 (n = 437) | 0.465 | 0.165–1.309 | 0.1471 |
| PV (n = 478) | - | - | 0.5219 |
| eGFR3 vs. eGFR2 (n = 376) | 0.431 | 0.095–1.944 | 0.2734 |
| eGFR3 vs. eGFR1 (n = 199) | 0.408 | 0.077–2.157 | 0.2916 |
| eGFR2 vs. eGFR1 (n = 381) | 0.948 | 0.329–2.729 | 0.9213 |
| MF (n = 396) | - | - | 0.6940 |
| eGFR3 vs. eGFR2 (n = 323) | 1.154 | 0.408–3.263 | 0.7867 |
| eGFR3 vs. eGFR1 (n = 184) | 2.029 | 0.398–10.337 | 0.3946 |
| eGFR2 vs. eGFR1 (n = 285) | 1.757 | 0.376–8.215 | 0.4736 |
| Diagnosis (n = 1420) | - | - | 0.4877 |
| ET vs. PV (n = 1024) | 0.736 | 0.381–1.422 | 0.3618 |
| ET vs. MF (n = 942) | 0.675 | 0.343–1.327 | 0.2543 |
| MF vs. PV (n = 874) | 1.091 | 0.569–2.091 | 0.7941 |
| Sex (female vs. male; n = 1420) | 0.533 | 0.305–0.933 | 0.0277 |
| Age at diagnosis (>60 vs. ≤60 years; n = 1416) | 0.756 | 0.432–1.323 | 0.0924 |
| Age at creatinine test (>60 vs. ≤60 years; n = 1420) | 0.784 | 0.457–1.344 | 0.3758 |
| JAK2V617F mutation (yes vs. no; n = 1315) | 1.759 | 0.813–3.809 | 0.1517 |
| Diabetes mellitus (yes vs. no; n = 1251) | 1.439 | 0.556–3.725 | 0.4534 |
| Arterial hypertension (yes vs. no; n = 956) | 1.302 | 0.678–2.499 | 0.4284 |
| Hyperlipoproteinemia (yes vs. no; n = 181) | 2.661 | 0.779–9.086 | 0.1182 |
| Leukocytes (>8.4 vs. ≤8.4 G/L; n = 1408) | 1.543 | 0.890–2.674 | 0.1223 |
| Platelets (>492 vs. ≤492 G/L; n = 1395) | 0.560 | 0.320–0.979 | 0.0421 |
| Uric acid (>5.7 vs. ≤5.7 mg/dL; n = 1012) | 1.685 | 0.869–3.269 | 0.1225 |
| Absolute neutrophil count (>5.48 vs. ≤5.48 G/L; n = 1254) | 1.697 | 0.963–2.992 | 0.0673 |
| Absolute monocyte count (>0.55 vs. ≤0.55 G/L; n = 1312) | 0.980 | 0.566–1.698 | 0.9424 |
| LDH (>267.5 vs. ≤267.5 U/L; n = 1356) | 3.292 | 1.748–6.201 | 0.0002 |
| CRP (>1.4 vs. ≤1.4 mg/L; n = 356) | 0.868 | 0.351–2.150 | 0.7603 |
| MPN therapy (yes vs. WW; n = 1215) | 1.694 | 0.927 -3.095 | 0.0865 |
| Antithrombotic therapy (yes vs. WW; n = 1251) | 0.903 | 0.500–1.628 | 0.7333 |
| Multiple regression (n = 1335) * | |||
| LDH (>267.5 vs. ≤267.5 U/L; n = 1356) | 3.292 | 1.748–6.201 | 0.0002 |
| Sample (n = 1215) | Therapy | n (%) | eGFR mL/min/1.73 m2 | P | |||
|---|---|---|---|---|---|---|---|
| Median | Q1 | Q3 | Overall | Pairwise * | |||
| All patients (n = 1215) | <0.0001 | ||||||
| HU only | 256 (21.07) | 71.90 | 60.58 | 84.39 | 0.0005 | ||
| RUX only | 94 (7.74) | 68.56 | 56.23 | 84.48 | 0.0002 | ||
| Other | 341 (28.07) | 72.77 | 57.40 | 85.95 | |||
| Multiple therapies | 250 (20.58) | 71.00 | 54.41 | 84.62 | 0.0021 | ||
| WW | 524 (43.13) | 77.81 | 66.48 | 90.23 | |||
| ET (n = 471) | 0.0013 | ||||||
| HU only | 88 (18.68) | 72.43 | 63.26 | 86.72 | 0.0168 | ||
| RUX only | 5 (1.06) | 60.79 | 58.00 | 61.52 | 0.0588 | ||
| Other | 148 (31.42) | 76.70 | 57.48 | 89.24 | |||
| Multiple therapies | 88 (18.68) | 74.37 | 55.71 | 89.24 | >0.9999 | ||
| WW | 230 (48.83) | 78.55 | 69.47 | 91.42 | |||
| PV (n = 395) | 0.6865 | ||||||
| HU only | 135 (34.18) | 72.88 | 60.43 | 84.79 | 0.3516 | ||
| RUX only | 11 (2.78) | 72.21 | 55.75 | 81.59 | 0.8951 | ||
| Other | 90 (22.78) | 72.63 | 59.32 | 89.69 | |||
| Multiple therapies | 77 (19.49) | 70.11 | 58.69 | 83.20 | 0.3453 | ||
| WW | 159 (40.25) | 74.36 | 63.05 | 87.20 | |||
| MF (n = 349) | 0.0073 | ||||||
| HU only | 33 (9.46) | 68.90 | 58.61 | 73.45 | 0.0488 | ||
| RUX only | 78 (22.35) | 68.85 | 56.23 | 84.69 | 0.0188 | ||
| Other | 103 (29.51) | 69.74 | 53.46 | 81.02 | |||
| Multiple therapies | 85 (24.36) | 69.74 | 52.67 | 80.35 | 0.2208 | ||
| WW | 135 (38.68) | 76.29 | 62.46 | 91.20 | |||
| Sample (n = 1251) | Therapy | n (%) | eGFR mL/min/1.73 m2 | P | |||
|---|---|---|---|---|---|---|---|
| Median | Q1 | Q3 | Overall | Pairwise * | |||
| All patients (n = 1251) | 0.1462 | ||||||
| ASA | 654 (52.28) | 75.00 | 63.18 | 86.25 | 0.9909 | ||
| P2Y12-antagonists | 30 (2.40) | 74.50 | 59.14 | 83.25 | 0.8892 | ||
| Anticoagulant-treated | 122 (9.75) | 70.65 | 55.36 | 86.30 | 0.1092 | ||
| No | 375 (29.98) | 75.23 | 62.37 | 89.19 | |||
| ET (n = 483) | 0.0234 | ||||||
| ASA | 272 (56.31) | 76.78 | 64.87 | 88.05 | 0.1026 | ||
| P2Y12-antagonists | 19 (3.93) | 74.47 | 52.69 | 85.74 | 0.1713 | ||
| Anticoagulant-treated | 34 (7.04) | 78.82 | 54.61 | 89.15 | 0.5919 | ||
| No | 124 (25.67) | 79.09 | 67.91 | 94.05 | |||
| PV (n = 415) | 0.3127 | ||||||
| ASA | 254 (61.20) | 74.97 | 63.44 | 86.25 | 0.7041 | ||
| P2Y12-antagonists | 7 (1.69) | 81.36 | 66.91 | 88.84 | 0.7107 | ||
| Anticoagulant-treated | 59 (14.22) | 70.81 | 57.15 | 89.84 | >0.9999 | ||
| No | 73 (17.59) | 70.50 | 59.60 | 83.97 | |||
| MF (n = 349) | 0.0849 | ||||||
| ASA | 128 (36.26) | 71.33 | 59.84 | 81.12 | 0.2907 | ||
| P2Y12-antagonists | 4 (1.13) | 65.46 | 49.50 | 76.41 | 0.8580 | ||
| Anticoagulant-treated | 29 (8.22) | 62.06 | 52.36 | 83.70 | 0.2262 | ||
| no | 178 (50.42) | 73.16 | 59.20 | 87.80 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gecht, J.; Tsoukakis, I.; Kricheldorf, K.; Stegelmann, F.; Klausmann, M.; Griesshammer, M.; Schulz, H.; Hollburg, W.; Göthert, J.R.; Sockel, K.; et al. Kidney Dysfunction Is Associated with Thrombosis and Disease Severity in Myeloproliferative Neoplasms: Implications from the German Study Group for MPN Bioregistry. Cancers 2021, 13, 4086. https://doi.org/10.3390/cancers13164086
Gecht J, Tsoukakis I, Kricheldorf K, Stegelmann F, Klausmann M, Griesshammer M, Schulz H, Hollburg W, Göthert JR, Sockel K, et al. Kidney Dysfunction Is Associated with Thrombosis and Disease Severity in Myeloproliferative Neoplasms: Implications from the German Study Group for MPN Bioregistry. Cancers. 2021; 13(16):4086. https://doi.org/10.3390/cancers13164086
Chicago/Turabian StyleGecht, Judith, Ioannis Tsoukakis, Kim Kricheldorf, Frank Stegelmann, Martine Klausmann, Martin Griesshammer, Holger Schulz, Wiebke Hollburg, Joachim R. Göthert, Katja Sockel, and et al. 2021. "Kidney Dysfunction Is Associated with Thrombosis and Disease Severity in Myeloproliferative Neoplasms: Implications from the German Study Group for MPN Bioregistry" Cancers 13, no. 16: 4086. https://doi.org/10.3390/cancers13164086
APA StyleGecht, J., Tsoukakis, I., Kricheldorf, K., Stegelmann, F., Klausmann, M., Griesshammer, M., Schulz, H., Hollburg, W., Göthert, J. R., Sockel, K., Heidel, F. H., Gattermann, N., Maintz, C., Al-Ali, H. K., Platzbecker, U., Hansen, R., Hänel, M., Parmentier, S., Bommer, M., ... Koschmieder, S. (2021). Kidney Dysfunction Is Associated with Thrombosis and Disease Severity in Myeloproliferative Neoplasms: Implications from the German Study Group for MPN Bioregistry. Cancers, 13(16), 4086. https://doi.org/10.3390/cancers13164086