Electrochemotherapy with Mitomycin C Potentiates Apoptosis Death by Inhibiting Autophagy in Squamous Carcinoma Cells
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Cultures
2.2. Flow Cytometry Analysis of P-gp Expression
2.3. In Vitro Electroporation
2.4. In Vitro Treatments
2.5. MTT Assay
2.6. Phase Contrast Microscopy
2.7. Scanning Electron Microscopy (SEM)
2.8. Determination of Apoptotic Cell Death by Annexin V-FITC/PI Staining by Flow Cytometry
2.9. Immunolabeling of Cytochrome C and Nuclear Staining
2.10. Western Blotting
2.11. Statistical Analysis
2.12. Clinical Application
2.13. Histological Analysis
3. Results
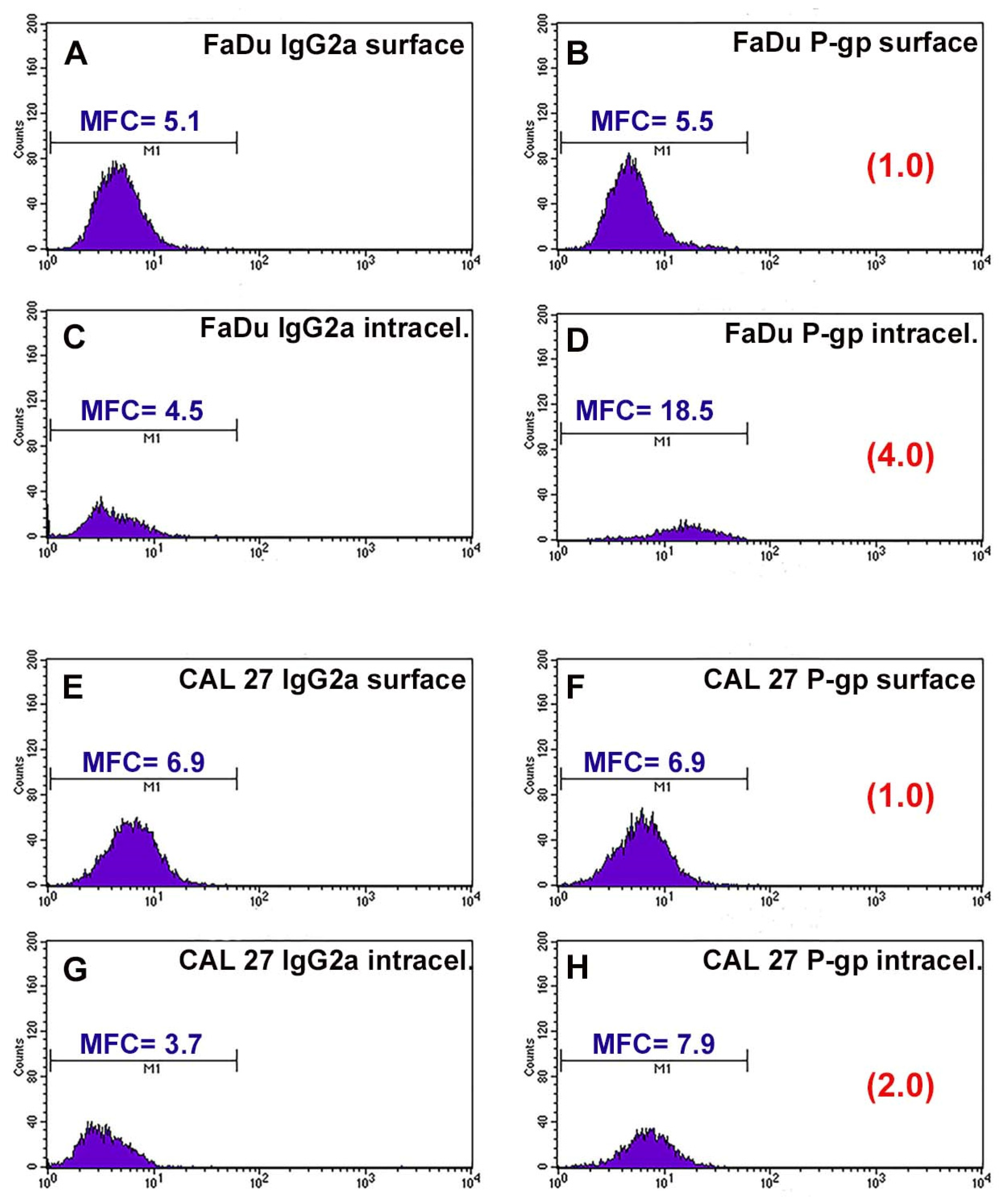
3.1. P-gp Expression Profiles of FaDu and CAL 27 Cells
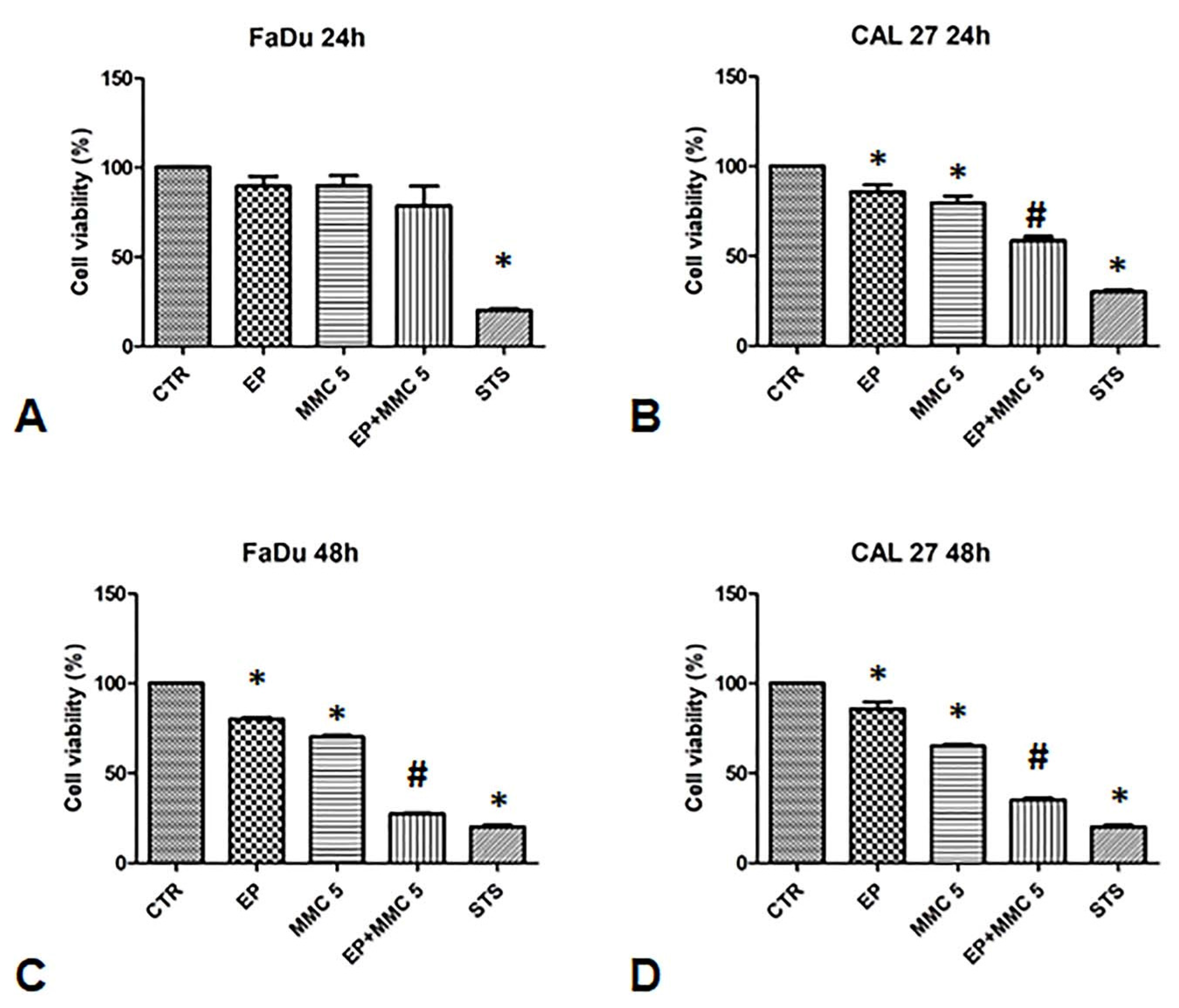
3.2. Cells Sensitivity after MMC Treatment Alone for Dose Selection
3.3. Observation of the Cytotoxic Effect of the EP+MMC Combination by Optical Microscopy
3.4. Morphological Changes Revealed by SEM Observations
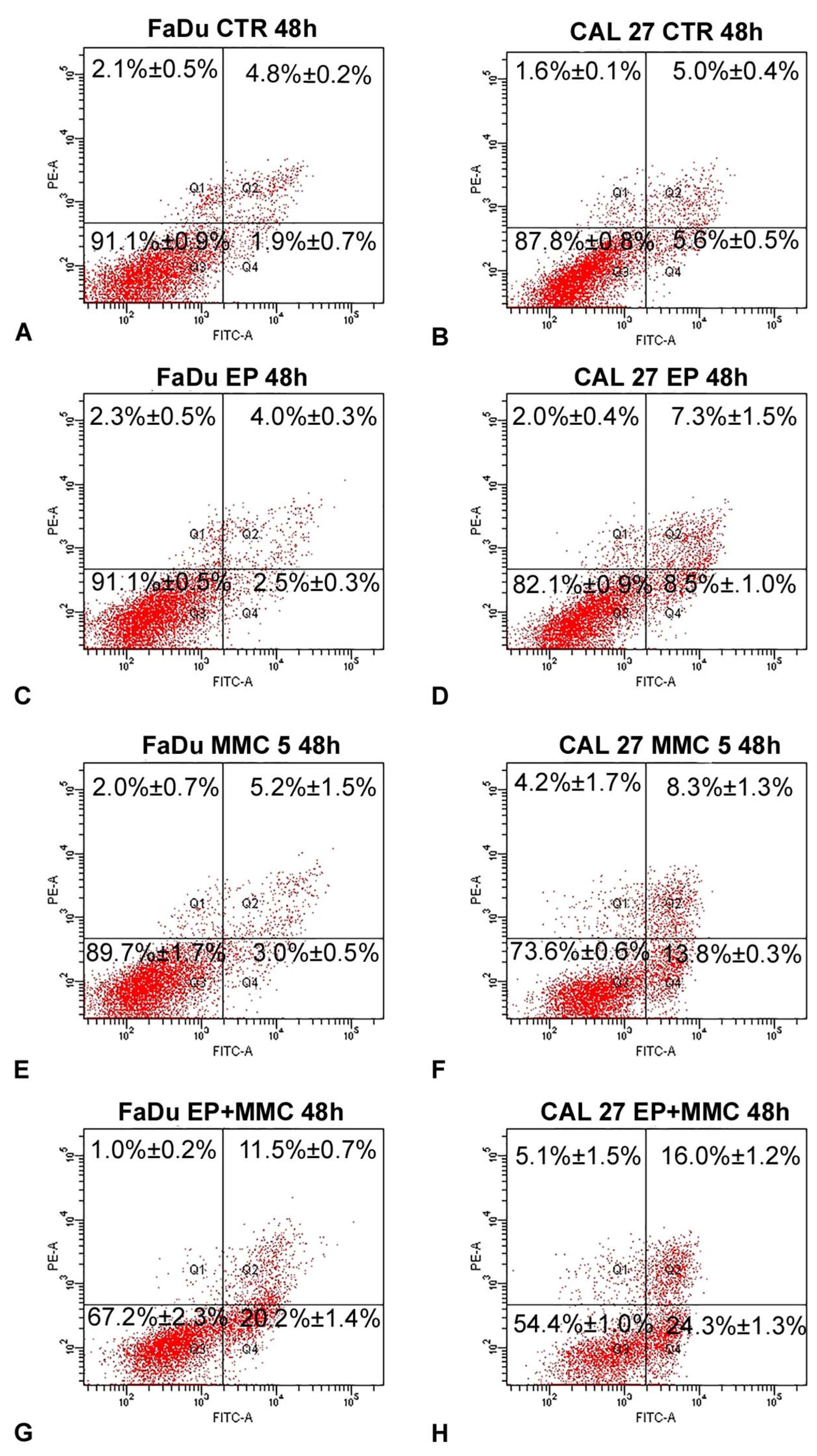
3.5. Apoptosis Detected by Flow Cytometry
3.6. Immunofluorescence Imaging of Cytochrome C Release for Apoptotic Studies
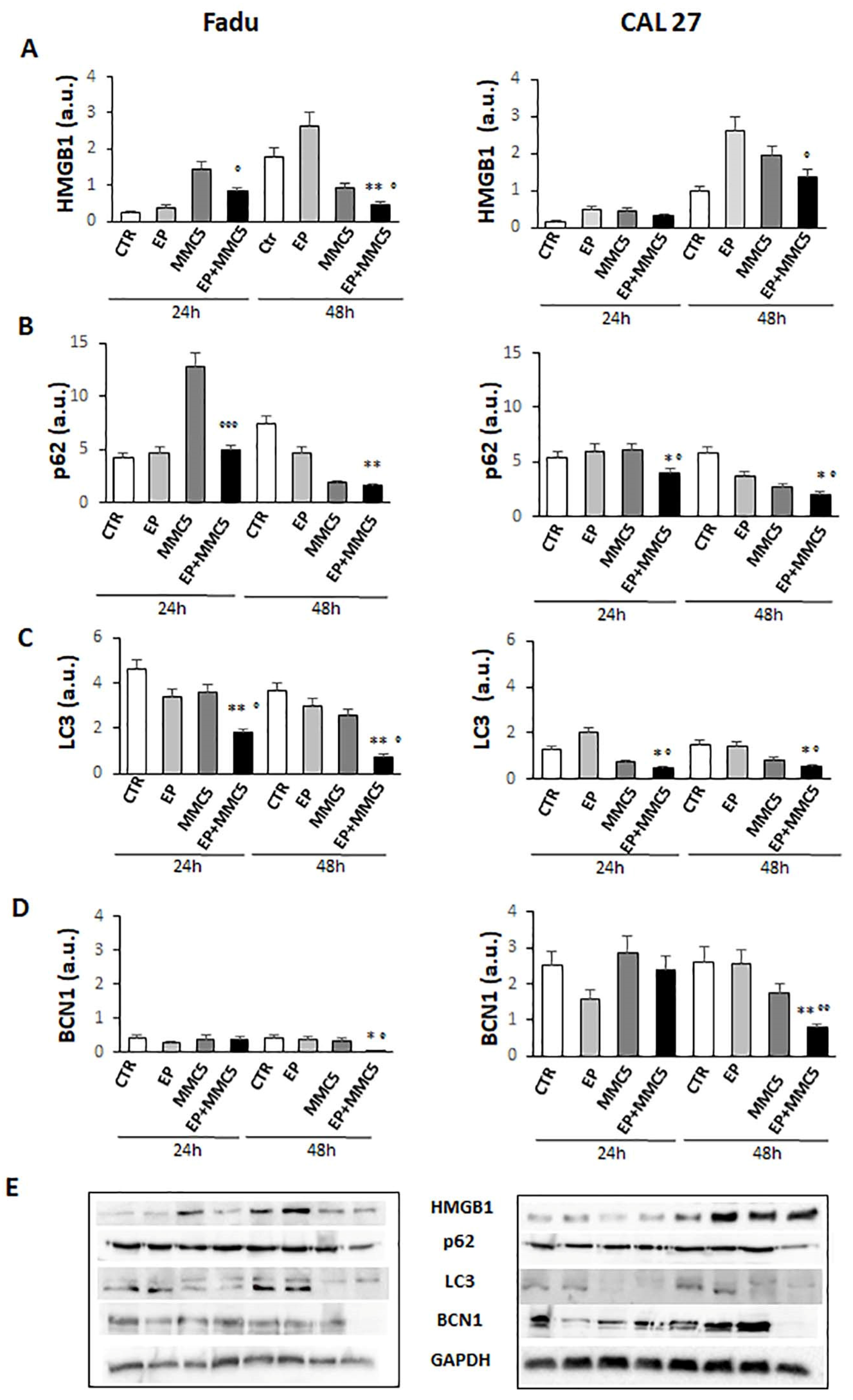
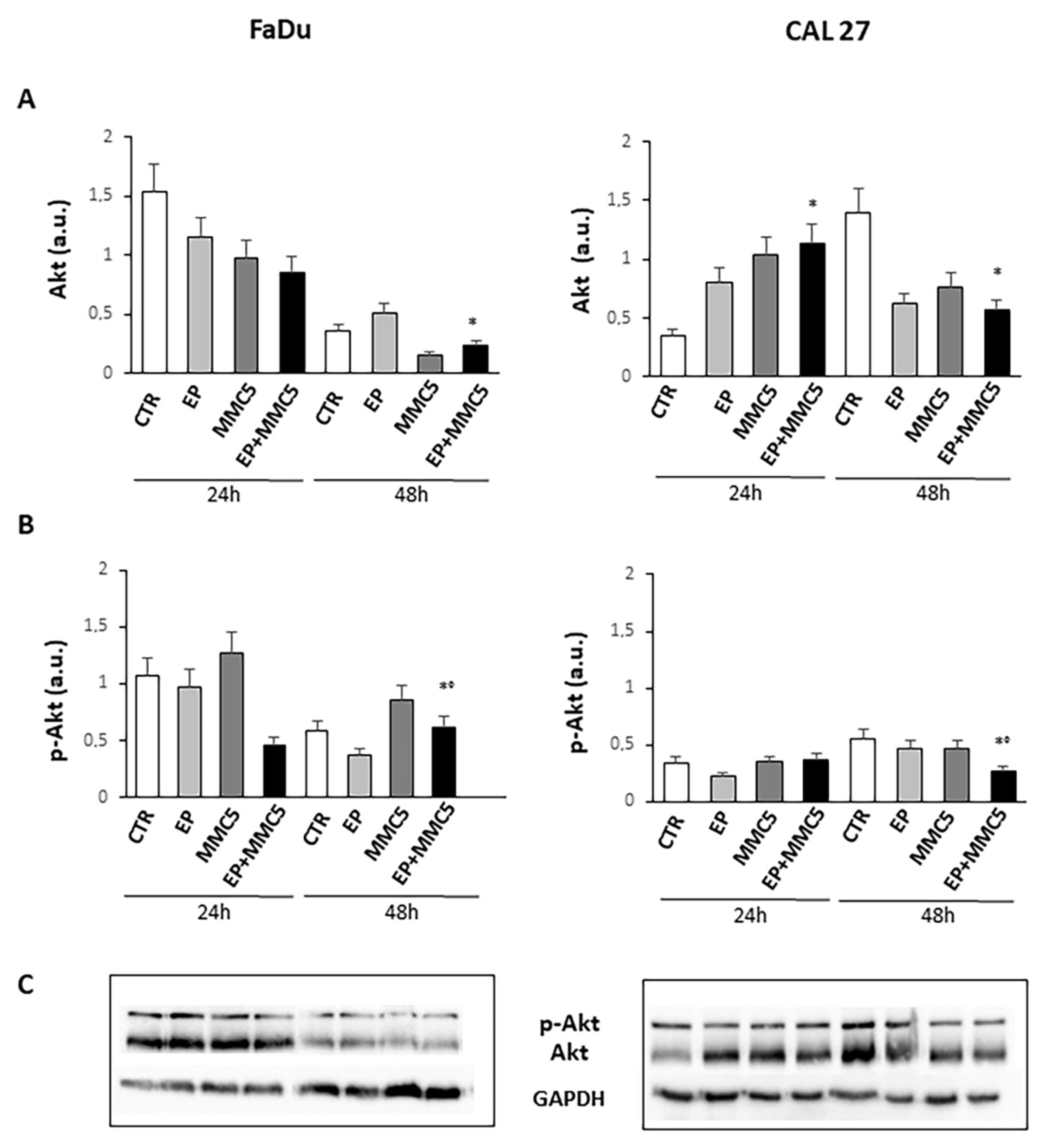
3.7. Analysis of Apoptotic and Autophagic-Associated Proteins Expression
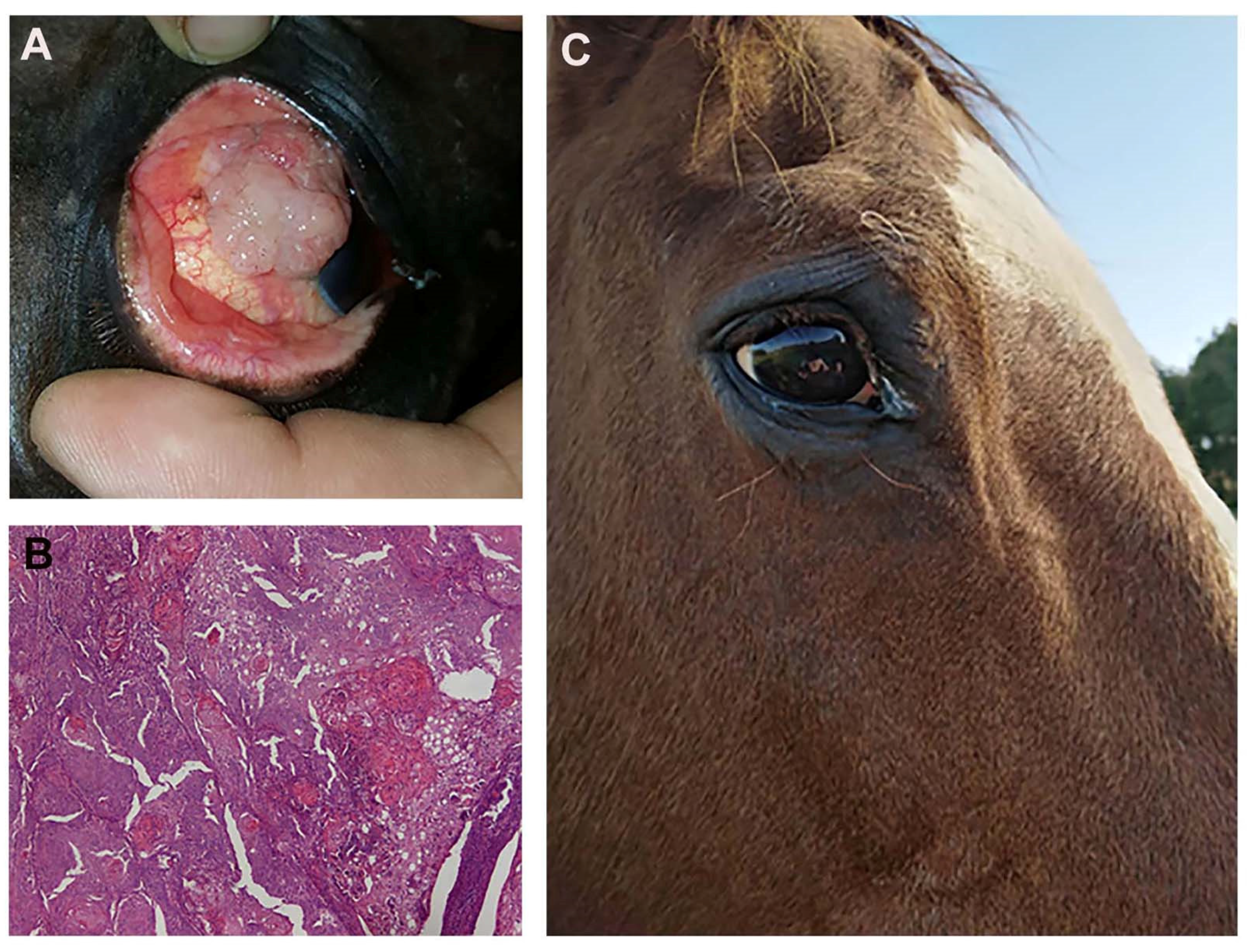
3.8. Clinical Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Campana, L.G.; Miklavčič, D.; Bertino, G.; Marconato, R.; Valpione, S.; Imarisio, I.; Dieci, M.V.; Granziera, E.; Cemazar, M.; Alaibac, M.; et al. Electrochemotherapy of superficial tumors—Current status: Basic Principles, operating procedures, shared indications, and emerging applications. Semin. Oncol. 2019, 46, 173–191. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Renaud, S.M.; Buglioni, S.; Carocci, F.; Dragonetti, E.; Murace, R.; Cardelli, P.; Vincenzi, B.; Baldi, A.; Citro, G. Electrochemotherapy with cisplatin enhances local control after surgical ablation of fibrosarcoma in cats: An approach to improve the therapeutic index of highly toxic chemotherapy drugs. J. Transl. Med. 2011, 9, 152. [Google Scholar] [CrossRef]
- Meschini, S.; Condello, M.; Lista, P.; Vincenzi, B.; Baldi, A.; Citro, G.; Arancia, G.; Spugnini, E.P. Electroporation adopting trains of biphasic pulses enhances in vitro and in vivo the cytotoxic effect of doxorubicin on multidrug resistant colon adenocarcinoma cells (LoVo). Eur. J. Cancer 2012, 48, 2236–2243. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Baldi, A. Electrochemotherapy in veterinary oncology: State-of-the-art and perspectives. Vet. Clin. N. Am. Small. Anim. Pract. 2019, 49, 967–979. [Google Scholar] [CrossRef] [PubMed]
- Probst, U.; Fuhrmann, I.; Beyer, L.; Wiggermann, P. Electrochemotherapy as a new modality in interventional oncology: A review. Technol. Cancer Res. Treat. 2018, 17. [Google Scholar] [CrossRef] [PubMed]
- Mittal, L.; Aryal, U.K.; Camarillo, I.G.; Ferreira, R.M.; Sundararajan, R. Quantitative proteomic analysis of enhanced cellular effects of electrochemotherapy with cisplatin in triple-negative breast cancer cells. Sci. Rep. 2019, 9, 13916. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, P.; O’Donovan, T.R.; McKenna, S.L.; Forde, P.F. Electrochemotherapy causes caspase-independent necrotic-like death in pancreatic cancer cells. Cancers 2019, 11, 1177. [Google Scholar] [CrossRef]
- Frandsen, S.K.; Gehl, J. A review on differences in effects on normal and malignant cells and tissues to electroporation-based therapies: A focus on calcium electroporation. Technol. Cancer Res. Treat. 2018, 17. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Baldi, F.; Mellone, P.; Feroce, F.; D’Avino, A.; Bonetto, F.; Vincenzi, B.; Citro, G.; Baldi, A. Patterns of tumor response in canine and feline cancer patients treated with electrochemotherapy: Preclinical data for the standardization of this treatment in pets and humans. J. Transl. Med. 2007, 5, 48. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Azzarito, T.; Fais, S.; Fanciulli, M.; Baldi, A. Electrochemotherapy as first line cancer treatment: Experiences from veterinary medicine in developing novel protocols. CCDT 2015, 16, 43–52. [Google Scholar] [CrossRef]
- Schüttrumpf, L.; Marschner, S.; Scheu, K.; Hess, J.; Rietzler, S.; Walch, A.; Baumeister, P.; Kirchner, T.; Ganswindt, U.; Zitzelsberger, H.; et al. Definitive chemoradiotherapy in patients with squamous cell cancers of the head and neck—Results from an unselected cohort of the clinical cooperation group “Personalized Radiotherapy in Head and Neck Cancer”. Radiat. Oncol. 2020, 15, 7. [Google Scholar] [CrossRef]
- Esmaeili, N.; Friebe, M. Electrochemotherapy: A review of current status, alternative IGP approaches, and future perspectives. J. Healthc. Eng. 2019, 2019, 2784516. [Google Scholar] [CrossRef]
- Ojha, R.; Jha, V.; Singh, S.K. Gemcitabine and mitomycin induced autophagy regulates cancer stem cell pool in urothelial carcinoma cells. Biochim. Biophys. Acta 2016, 1863, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Belmokhtar, C.A.; Hillion, J.; Ségal-Bendirdjian, E. Staurosporine induces apoptosis through both caspase-dependent and caspase-independent mechanisms. Oncogene 2001, 20, 3354–3362. [Google Scholar] [CrossRef] [PubMed]
- Meschini, S.; Pellegrini, E.; Condello, M.; Occhionero, G.; Delfine, S.; Condello, G.; Mastrodonato, F. Cytotoxic and Apoptotic Activities of Prunus Spinosa Trigno ecotype extract on human cancer cells. Molecules 2017, 22, 1578. [Google Scholar] [CrossRef] [PubMed]
- Condello, M.; Pellegrini, E.; Spugnini, E.P.; Baldi, A.; Amadio, B.; Vincenzi, B.; Occhionero, G.; Delfine, S.; Mastrodonato, F.; Meschini, S. Anticancer activity of “trigno m”, extract of Prunus Spinosa Drupes, against in vitro 3D and in vivo colon cancer models. Biomed. Pharmacother. 2019, 118, 109281. [Google Scholar] [CrossRef] [PubMed]
- Rayner, S.G.; Van Zyl, N. The use of mitomycin C as an adjunctive treatment for equine ocular squamous cell carcinoma. Aust Vet. J. 2006, 84, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Scacco, L.; Bolaffio, C.; Romano, A.; Fanciulli, M.; Baldi, A.; Spugnini, E.P. Adjuvant electrochemotherapy increases local control in a recurring equine anal melanoma. J. Equine Vet. Sci. 2013, 33, 637–639. [Google Scholar] [CrossRef]
- Spugnini, E.P.; D’Alterio, G.L.; Dotsinsky, I.; Mudrov, T.; Dragonetti, E.; Murace, R.; Citro, G.; Baldi, A. Electrochemotherapy for the treatment of multiple melanomas in a horse. J. Equine Vet. Sci. 2011, 31, 430–433. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Bolaffio, C.; Scacco, L.; Baldi, A. Electrochemotherapy Increases local control after incomplete excision of a recurring penile fibrosarcoma in a stallion. Open Vet. J. 2016, 6, 234–237. [Google Scholar] [CrossRef] [PubMed]
- Spugnini, E.P.; Bolaffio, C.; Scacco, L.; Baldi, A. Isolated limb perfusion electrochemotherapy for the treatment of an advanced squamous cell carcinoma of the hoof in a mare. Open Vet. J. 2017, 7, 192. [Google Scholar] [CrossRef]
- Xu, T.; Jiang, L.; Wang, Z. The progression of HMGB1-induced autophagy in cancer biology. OncoTargets Ther. 2019, 12, 365–377. [Google Scholar] [CrossRef] [PubMed]
- Tsong, T.Y. Electroporation of cell membranes. Biophys. J. 1991, 60, 297–306. [Google Scholar] [CrossRef]
- Batista Napotnik, T.; Miklavčič, D. In vitro electroporation detection methods—An overview. Bioelectrochemistry 2018, 120, 166–182. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Arancia, G.; Porrello, A.; Colone, M.; Formisano, G.; Stringaro, A.; Citro, G.; Molinari, A. Ultrastructural modifications of cell membranes induced by “electroporation” on melanoma xenografts. Microsc. Res. Tech. 2007, 70, 1041–1050. [Google Scholar] [CrossRef]
- Halaby, R. Influence of lysosomal sequestration on multidrug resistance in cancer cells. CDR 2019. [Google Scholar] [CrossRef]
- Molinari, A.; Calcabrini, A.; Meschini, S.; Stringaro, A.; Crateri, P.; Toccacieli, L.; Marra, M.; Colone, M.; Cianfriglia, M.; Arancia, G. Subcellular detection and localization of the drug transporter P-glycoprotein in cultured tumor cells. CPPS 2002, 3, 653–670. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, H.; Chen, X. Drug resistance and combating drug resistance in cancer. CDR 2019. [Google Scholar] [CrossRef] [PubMed]
- Stojanović, N.; Brozovic, A.; Majhen, D.; Bosnar, M.H.; Fritz, G.; Osmak, M.; Ambriović-Ristov, A. Integrin Avβ3 Expression in tongue squamous carcinoma cells Cal27 confers anticancer drug resistance through loss of PSrc(Y418). Biochim. Biophys. Acta Mol. Cell Res. 2016, 1863, 1969–1978. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Chen, Y.; Liu, G.; Li, C.; Song, Y.; Cao, Z.; Li, W.; Hu, J.; Lu, C.; Liu, Y. PI3K/AKT Pathway as a key link modulates the multidrug resistance of cancers. Cell Death Dis. 2020, 11, 797. [Google Scholar] [CrossRef]
- Sharabi, S.; Guez, D.; Daniels, D.; Cooper, I.; Atrakchi, D.; Liraz-Zaltsman, S.; Last, D.; Mardor, Y. The application of point source electroporation and chemotherapy for the treatment of glioma: A randomized controlled rat study. Sci. Rep. 2020, 10, 2178. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Wang, J.; Sun, S.; Zhang, Y.; Lin, X.; Lao, X.; Cui, B.; Li, S. Irreversible electroporation versus radiotherapy after induction chemotherapy on survival in patients with locally advanced pancreatic cancer: A propensity score analysis. BMC Cancer 2019, 19, 394. [Google Scholar] [CrossRef]
- Brentnall, M.; Rodriguez-Menocal, L.; De Guevara, R.L.; Cepero, E.; Boise, L.H. Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biol. 2013, 14, 32. [Google Scholar] [CrossRef] [PubMed]
- McComb, S.; Chan, P.K.; Guinot, A.; Hartmannsdottir, H.; Jenni, S.; Dobay, M.P.; Bourquin, J.-P.; Bornhauser, B.C. Efficient apoptosis requires feedback amplification of upstream apoptotic signals by effector caspase-3 or -7. Sci. Adv. 2019, 5, eaau9433. [Google Scholar] [CrossRef]
- Shen, C.-Y.; Chen, L.-H.; Lin, Y.-F.; Lai, L.-C.; Chuang, E.Y.; Tsai, M.-H. Mitomycin C treatment induces resistance and enhanced migration via phosphorylated akt in aggressive lung cancer cells. Oncotarget 2016, 7, 79995–80007. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.; Chang, Y.; Lu, W.; Sheng, X.; Wang, S.; Xu, H.; Ma, J. Regulation of autophagy by glycolysis in cancer. CMAR 2020, 12, 13259–13271. [Google Scholar] [CrossRef]
- Kang, R.; Livesey, K.M.; Zeh, H.J., III; Loze, M.T.; Tang, D. Metabolic regulation by HMGB1-mediated autophagy and mitophagy. Autophagy 2011, 7, 1256–1258. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Kang, R.; Livesey, K.M.; Cheh, C.-W.; Farkas, A.; Loughran, P.; Hoppe, G.; Bianchi, M.E.; Tracey, K.J.; Zeh, H.J.; et al. Endogenous HMGB1 regulates autophagy. J. Cell Biol. 2010, 190, 881–892. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Livesey, K.M.; Zeh, H.J.; Loze, M.T.; Tang, D. HMGB1: A novel beclin 1-binding protein active in autophagy. Autophagy 2010, 6, 1209–1211. [Google Scholar] [CrossRef]
- Hamurcu, Z.; Delibaşı, N.; Geçene, S.; Şener, E.F.; Dönmez-Altuntaş, H.; Özkul, Y.; Canatan, H.; Ozpolat, B. Targeting LC3 and beclin-1 autophagy genes suppresses proliferation, survival, migration and invasion by inhibition of cyclin-D1 and UPAR/integrin Β1/Src signaling in triple negative breast cancer cells. J. Cancer Res. Clin. Oncol. 2018, 144, 415–430. [Google Scholar] [CrossRef]
- Li, X.; He, S.; Ma, B. Autophagy and autophagy-related proteins in cancer. Mol. Cancer 2020, 19, 12. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.; Yu, H.; Gu, S.; Xu, Y.; Su, J.; Li, H.; Kang, J.; Cui, M. P62/SQSTM1 is involved in cisplatin resistance in human ovarian cancer cells via the Keap1-Nrf2-ARE system. Int. J. Oncol. 2014, 45, 2341–2348. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Pan, B.-Q.; Shi, F.; Xie, Z.-H.; Jiang, Y.-Y.; Shang, L.; Zhang, Y.; Xu, X.; Cai, Y.; Hao, J.-J.; et al. Sequestosome 1 protects esophageal squamous carcinoma cells from apoptosis via stabilizing SKP2 under serum starvation condition. Oncogene 2018, 37, 3260–3274. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.-Z.; Li, S.-S.; Zhou, W.; Kang, Z.-J.; Zhang, Q.-X.; Kamran, M.; Xu, J.; Liang, D.-P.; Wang, C.-L.; Hou, Z.-J.; et al. P62/SQSTM1 enhances breast cancer stem-like properties by stabilizing MYC MRNA. Oncogene 2017, 36, 304–317. [Google Scholar] [CrossRef] [PubMed]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Condello, M.; D’Avack, G.; Vona, R.; Spugnini, E.P.; Scacco, L.; Meschini, S. Electrochemotherapy with Mitomycin C Potentiates Apoptosis Death by Inhibiting Autophagy in Squamous Carcinoma Cells. Cancers 2021, 13, 3867. https://doi.org/10.3390/cancers13153867
Condello M, D’Avack G, Vona R, Spugnini EP, Scacco L, Meschini S. Electrochemotherapy with Mitomycin C Potentiates Apoptosis Death by Inhibiting Autophagy in Squamous Carcinoma Cells. Cancers. 2021; 13(15):3867. https://doi.org/10.3390/cancers13153867
Chicago/Turabian StyleCondello, Maria, Gloria D’Avack, Rosa Vona, Enrico Pierluigi Spugnini, Licia Scacco, and Stefania Meschini. 2021. "Electrochemotherapy with Mitomycin C Potentiates Apoptosis Death by Inhibiting Autophagy in Squamous Carcinoma Cells" Cancers 13, no. 15: 3867. https://doi.org/10.3390/cancers13153867
APA StyleCondello, M., D’Avack, G., Vona, R., Spugnini, E. P., Scacco, L., & Meschini, S. (2021). Electrochemotherapy with Mitomycin C Potentiates Apoptosis Death by Inhibiting Autophagy in Squamous Carcinoma Cells. Cancers, 13(15), 3867. https://doi.org/10.3390/cancers13153867








