Interrelations between Patients’ Clinicopathological Characteristics and Their Association with Response to Immunotherapy in a Real-World Cohort of NSCLC Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cohort Characteristics
2.2. Statistical Analysis
2.2.1. Cohort Pre-Processing
2.2.2. Age Stratification
2.2.3. PD-L1 Pre-Processing
2.2.4. PFS Computation
2.2.5. OS Computation
2.2.6. Survival Models
2.2.7. Interactions between Features
2.2.8. Feature Selection
3. Results
3.1. Description of the Cohort
3.2. Association of Clinical Features with Clinical Outcome
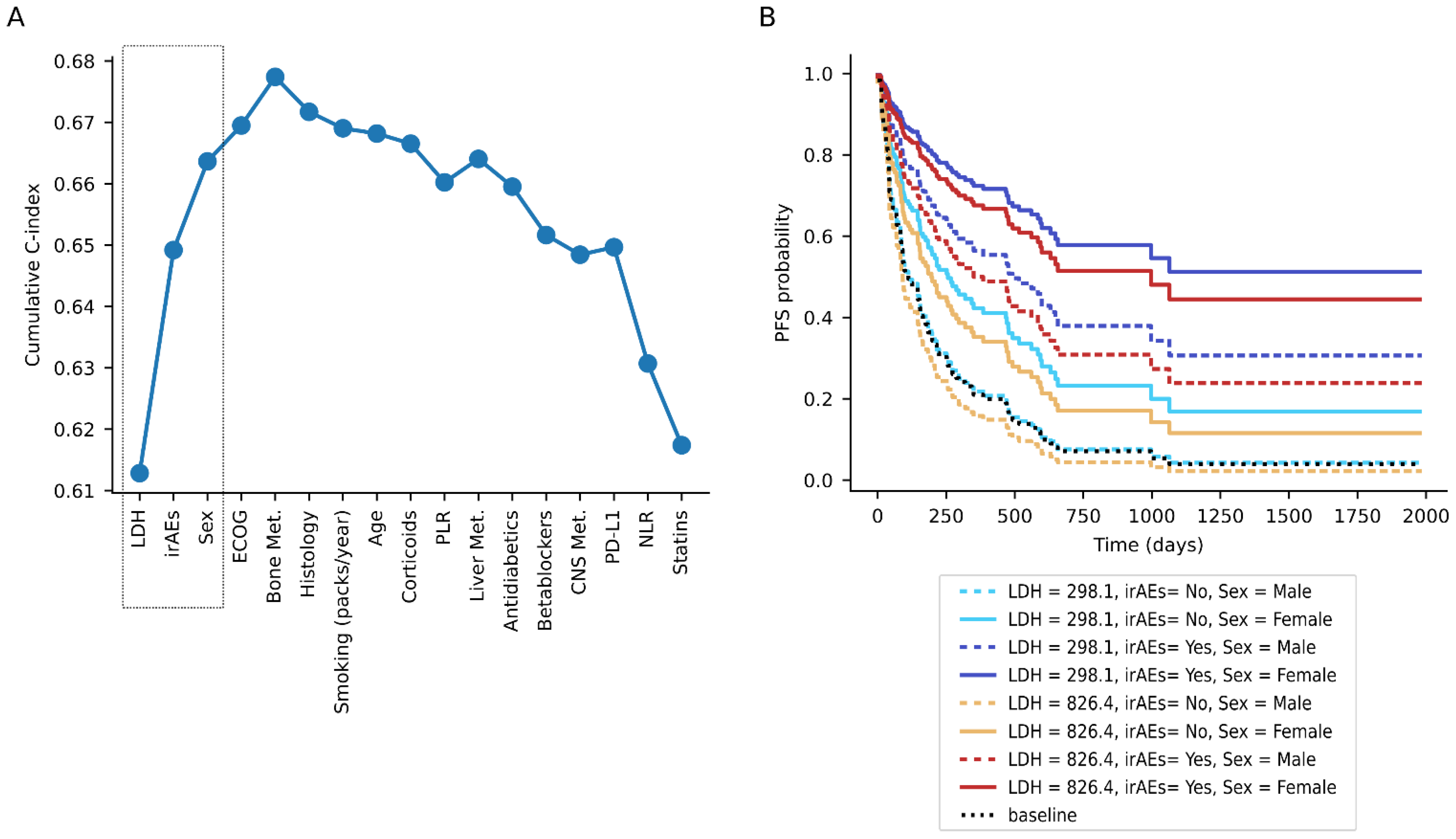
3.3. Interactions between Features and Proposed Compendium
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rizvi, H.; Sanchez-Vega, F.; La, K.; Chatila, W.; Jonsson, P.; Halpenny, D.; Plodkowski, A.; Long, N.; Sauter, J.L.; Rekhtman, N.; et al. Molecular determinants of response to anti-programmed cell death (PD)-1 and anti-programmed death-ligand 1 (PD-L1) blockade in patients with non-small-cell lung cancer profiled with targeted next-generation sequencing. J. Clin. Oncol. 2018, 36, 633–641. [Google Scholar] [CrossRef]
- Rizvi, N.A.; Hellmann, M.D.; Snyder, A.; Kvistborg, P.; Makarov, V.; Havel, J.J.; Lee, W.; Yuan, J.; Wong, P.; Ho, T.S.; et al. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 2015, 348, 124–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hellmann, M.D.; Nathanson, T.; Rizvi, H.; Creelan, B.C.; Sanchez-Vega, F.; Ahuja, A.; Ni, A.; Novik, J.B.; Mangarin, L.M.B.; Abu-Akeel, M.; et al. Genomic Features of Response to Combination Immunotherapy in Patients with Advanced Non-Small-Cell Lung Cancer. Cancer Cell 2018, 33, 843–852.e4. [Google Scholar] [CrossRef] [Green Version]
- Frigola, J.; Navarro, A.; Carbonell, C.; Callejo, A.; Iranzo, P.; Cedrés, S.; Martinez-Marti, A.; Pardo, N.; Saoudi-Gonzalez, N.; Martinez, D.; et al. Molecular profiling of long-term responders to immune checkpoint inhibitors in advanced non-small cell lung cancer. Mol. Oncol. 2020, 1–14. [Google Scholar] [CrossRef]
- Bagley, S.J.; Kothari, S.; Aggarwal, C.; Bauml, J.M.; Alley, E.W.; Evans, T.L.; Kosteva, J.A.; Ciunci, C.A.; Gabriel, P.E.; Thompson, J.C.; et al. Pretreatment neutrophil-to-lymphocyte ratio as a marker of outcomes in nivolumab-treated patients with advanced non-small-cell lung cancer. Lung Cancer 2017, 106, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mezquita, L.; Auclin, E.; Ferrara, R.; Charrier, M.; Remon, J.; Planchard, D.; Ponce, S.; Ares, L.P.; Leroy, L.; Audigier-Valette, C.; et al. Association of the lung immune prognostic index with immune checkpoint inhibitor outcomes in patients with advanced non-small cell lung cancer. JAMA Oncol. 2018, 4, 351–357. [Google Scholar] [CrossRef]
- Prelaj, A.; Ferrara, R.; Rebuzzi, S.E.; Proto, C.; Signorelli, D.; Galli, G.; De Toma, A.; Randon, G.; Pagani, F.; Viscardi, G.; et al. Epsilon: A prognostic score for immunotherapy in advanced non-small-cell lung cancer: A validation cohort. Cancers 2019, 11, 1954. [Google Scholar] [CrossRef] [Green Version]
- Sorich, M.J.; Rowland, A.; Karapetis, C.S.; Hopkins, A.M. Evaluation of the Lung Immune Prognostic Index for Prediction of Survival and Response in Patients Treated With Atezolizumab for NSCLC: Pooled Analysis of Clinical Trials. J. Thorac. Oncol. 2019, 14, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Kazandjian, D.; Gong, Y.; Keegan, P.; Pazdur, R.; Blumenthal, G.M. Prognostic Value of the Lung Immune Prognostic Index for Patients Treated for Metastatic Non-Small Cell Lung Cancer. JAMA Oncol. 2019, 5, 1481–1485. [Google Scholar] [CrossRef]
- Sato, K.; Akamatsu, H.; Murakami, E.; Sasaki, S.; Kanai, K.; Hayata, A.; Tokudome, N.; Akamatsu, K.; Koh, Y.; Ueda, H.; et al. Correlation between immune-related adverse events and efficacy in non-small cell lung cancer treated with nivolumab. Lung Cancer 2018, 115, 71–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grangeon, M.; Tomasini, P.; Chaleat, S.; Jeanson, A.; Souquet-Bressand, M.; Khobta, N.; Bermudez, J.; Trigui, Y.; Greillier, L.; Blanchon, M.; et al. Association Between Immune-related Adverse Events and Efficacy of Immune Checkpoint Inhibitors in Non–small-cell Lung Cancer. Clin. Lung Cancer 2019, 20, 201–207. [Google Scholar]
- Haratani, K.; Hayashi, H.; Chiba, Y.; Kudo, K.; Yonesaka, K.; Kato, R.; Kaneda, H.; Hasegawa, Y.; Tanaka, K.; Takeda, M.; et al. Association of immune-related adverse events with nivolumab efficacy in non-small cell lung cancer. JAMA Oncol. 2018, 4, 374–378. [Google Scholar] [CrossRef]
- Cortellini, A.; Chiari, R.; Ricciuti, B.; Metro, G.; Perrone, F.; Tiseo, M.; Bersanelli, M.; Bordi, P.; Santini, D.; Giusti, R.; et al. Correlations Between the Immune-related Adverse Events Spectrum and Efficacy of Anti-PD1 Immunotherapy in NSCLC Patients. Clin. Lung Cancer 2019, 20, 237–247.e1. [Google Scholar]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Li, Y.; Yan, X.; Song, Q.; Wang, G.; Hu, Y.; Jiao, S.; Wang, J. Pretreatment lactate dehydrogenase may predict outcome of advanced non small-cell lung cancer patients treated with immune checkpoint inhibitors: A meta-analysis. Cancer Med. 2019, 8, 1467–1473. [Google Scholar] [CrossRef] [PubMed]
- Diem, S.; Schmid, S.; Krapf, M.; Flatz, L.; Born, D.; Jochum, W.; Templeton, A.J.; Früh, M. Neutrophil-to-Lymphocyte ratio (NLR) and Platelet-to-Lymphocyte ratio (PLR) as prognostic markers in patients with non-small cell lung cancer (NSCLC) treated with nivolumab. Lung Cancer 2017, 111, 176–181. [Google Scholar] [CrossRef]
- Russo, A.; Russano, M.; Franchina, T.; Migliorino, M.R.; Aprile, G.; Mansueto, G.; Berruti, A.; Falcone, A.; Aieta, M.; Gelibter, A.; et al. Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Outcomes with Nivolumab in Pretreated Non-Small Cell Lung Cancer (NSCLC): A Large Retrospective Multicenter Study. Adv. Ther. 2020, 37, 1145–1155. [Google Scholar] [CrossRef]
- Omori, M.; Okuma, Y.; Hakozaki, T.; Hosomi, Y. Statins improve survival in patients previously treated with nivolumab for advanced non-small cell lung cancer: An observational study. Mol. Clin. Oncol. 2018, 10, 137–143. [Google Scholar] [CrossRef] [Green Version]
- Arbour, K.C.; Mezquita, L.; Long, N.; Rizvi, H.; Auclin, E.; Ni, A.; Martínez-Bernal, G.; Ferrara, R.; Victoria Lai, W.; Hendriks, L.E.L.; et al. Impact of baseline steroids on efficacy of programmed cell death-1 and programmed death-ligand 1 blockade in patients with non–small-cell lung cancer. J. Clin. Oncol. 2018, 36, 2872–2878. [Google Scholar] [CrossRef] [PubMed]
- Cortellini, A.; Tucci, M.; Adamo, V.; Stucci, L.S.; Russo, A.; Tanda, E.T.; Spagnolo, F.; Rastelli, F.; Bisonni, R.; Santini, D.; et al. Integrated analysis of concomitant medications and oncological outcomes from PD-1/PD-L1 checkpoint inhibitors in clinical practice. J. Immunother. Cancer 2020. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.C.; Pennell, N.A. Early Use of Systemic Corticosteroids in Patients with Advanced NSCLC Treated with Nivolumab. J. Thorac. Oncol. 2018, 13, 1771–1775. [Google Scholar] [CrossRef] [Green Version]
- Borghaei, H.; Gettinger, S.; Vokes, E.E.; Chow, L.Q.M.; Burgio, M.A.; de Castro Carpeno, J.; Pluzanski, A.; Arrieta, O.; Frontera, O.A.; Chiari, R.; et al. Five-Year Outcomes from the Randomized, Phase III Trials CheckMate 017 and 057: Nivolumab versus Docetaxel in Previously Treated Non-Small-Cell Lung Cancer. J. Clin. Oncol. 2021, 39, 723–733. [Google Scholar] [CrossRef]
- Gandhi, L.; Rodríguez-Abreu, D.; Gadgeel, S.; Esteban, E.; Felip, E.; De Angelis, F.; Domine, M.; Clingan, P.; Hochmair, M.J.; Powell, S.F.; et al. Pembrolizumab plus Chemotherapy in Metastatic Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 2078–2092. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, K.; Gill, R.R.; Widick, P.; Bindal, P.; McDonald, D.C.; Shea, M.; Rangachari, D.; Costa, D.B. Association of Performance Status with Survival in Patients with Advanced Non-Small Cell Lung Cancer Treated With Pembrolizumab Monotherapy. JAMA Netw. Open 2021, 4, e2037120. [Google Scholar] [CrossRef] [PubMed]
- Landi, L.; D’Incà, F.; Gelibter, A.; Chiari, R.; Grossi, F.; Delmonte, A.; Passaro, A.; Signorelli, D.; Gelsomino, F.; Galetta, D.; et al. Bone metastases and immunotherapy in patients with advanced non-small-cell lung cancer. J. Immunother. Cancer 2019, 7, 1–9. [Google Scholar] [CrossRef]
- Qiao, M.; Zhou, F.; Hou, L.; Li, X.; Zhao, C.; Jiang, T.; Gao, G.; Su, C.; Wu, C.; Ren, S.; et al. Efficacy of immune-checkpoint inhibitors in advanced non-small cell lung cancer patients with different metastases. Ann. Transl. Med. 2021, 9, 34. [Google Scholar] [CrossRef]
- Pérez-Díez, I.; Hidalgo, M.R.; Malmierca-Merlo, P.; Farràs, R.; de la Iglesia-Vayá, M.; Provencio, M.; Romero, A.; García-García, F.; Andreu, Z. Functional signatures in non-small-cell lung cancer: A systematic review and meta-analysis of sex-based differences in transcriptomic studies. medRxiv 2020. [Google Scholar] [CrossRef]
- Conforti, F.; Pala, L.; Bagnardi, V.; Viale, G.; De Pas, T.; Pagan, E.; Pennacchioli, E.; Cocorocchio, E.; Ferrucci, P.F.; De Marinis, F.; et al. Sex-Based Heterogeneity in Response to Lung Cancer Immunotherapy: A Systematic Review and Meta-Analysis. J. Natl. Cancer Inst. 2019, 111, 772–781. [Google Scholar] [CrossRef]
- Gainor, J.F.; Rizvi, H.; Jimenez Aguilar, E.; Skoulidis, F.; Yeap, B.Y.; Naidoo, J.; Khosrowjerdi, S.; Mooradian, M.; Lydon, C.; Illei, P.; et al. Clinical activity of programmed cell death 1 (PD-1) blockade in never, light, and heavy smokers with non-small-cell lung cancer and PD-L1 expression ≥50%. Ann. Oncol. 2020, 31, 404–411. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Ricciuti, B.; Nguyen, T.; Li, X.; Rabin, M.S.; Awad, M.M.; Lin, X.; Johnson, B.E.; Christiani, D.C. Association between smoking history and tumor mutation burden in advanced non–small cell lung cancer. Cancer Res. 2021, 81, 2566–2573. [Google Scholar] [CrossRef]
- Dudnik, E.; Kareff, S.; Moskovitz, M.; Kim, C.; Liu, S.V.; Lobachov, A.; Gottfried, T.; Urban, D.; Zer, A.; Rotem, O.; et al. Real-world survival outcomes with immune checkpoint inhibitors in large-cell neuroendocrine tumors of lung. J. Immunother Cancer 2021. [Google Scholar] [CrossRef]

| VARIBLE | Number of Pts |
| ECOG at ICIs start | |
| 1 | 190 |
| 0 | 53 |
| 2 | 18 |
| not available | 1 |
| Histology | |
| adk | 191 |
| scc | 58 |
| nos | 7 |
| lcnec | 6 |
| Sex | |
| male | 182 |
| female | 80 |
| Age category | |
| middle | 173 |
| young | 55 |
| elder | 34 |
| Smoking | |
| yes | 221 |
| no | 30 |
| not available | 11 |
| CNS Met. At ICIs start | |
| no | 203 |
| yes | 58 |
| not available | 1 |
| Liver Met. At ICIs start | |
| no | 198 |
| yes | 63 |
| not available | 1 |
| Bone Met. At ICIs start | |
| no | 167 |
| yes | 94 |
| not available | 1 |
| Corticoids pre-ICIs | |
| no | 211 |
| yes | 45 |
| not available | 6 |
| Statins | |
| no | 177 |
| yes | 85 |
| Antidiabetic drugs | |
| no | 222 |
| yes | 40 |
| Betablockers | |
| no | 227 |
| yes | 35 |
| ICIs line | |
| 2 | 101 |
| 1 | 90 |
| >2 | 53 |
| not available | 18 |
| Treatment type | |
| ICIs | 203 |
| CT+ICIs | 40 |
| TKI+ICIs | 19 |
| PD-L1 | |
| not available | 174 |
| positive | 68 |
| negative | 21 |
| Immune-related Adverse Events | |
| no | 195 |
| yes | 67 |
| VARIABLE | Value |
| Smoking (packs/year) | |
| Patients with data | 251 |
| mean | 40.33 |
| std | 29.69 |
| min | 0.00 |
| 25% | 20.00 |
| 50% | 35.00 |
| 75% | 54.00 |
| max | 200.00 |
| LDH before ICIs start | |
| Patients with data | 229 |
| mean | 589.02 |
| std | 692.50 |
| min | 211.00 |
| 25% | 354.00 |
| 50% | 441.00 |
| 75% | 591.00 |
| max | 7950.00 |
| Neutrophils to lymphocytes ratio | |
| Patients with data | 262 |
| mean | 5.71 |
| std | 6.85 |
| min | 0.10 |
| 25% | 2.41 |
| 50% | 3.69 |
| 75% | 6.49 |
| max | 72.00 |
| Platelets to lymphocytes ratio | |
| Patients with data | 262 |
| mean | 249.95 |
| std | 204.86 |
| min | 15.89 |
| 25% | 145.18 |
| 50% | 200.00 |
| 75% | 292.08 |
| max | 2160.00 |
| coef | exp(coef) | coef Lower 95% | coef Upper 95% | exp(coef) Lower 95% | exp(coef) Upper 95% | p | |
|---|---|---|---|---|---|---|---|
| irAEs (yes) | −1.058123091 | 0.347106687 | −1.510824435 | −0.605421746 | 0.220727927 | 0.54584417 | 4.62473 × 10−6 |
| Sex (female) | −0.65991903 | 0.516893185 | −1.099535233 | −0.220302828 | 0.333025827 | 0.80227581 | 0.003259403 |
| Age (elder) | −0.590496219 | 0.554052285 | −1.323552824 | 0.142560386 | 0.266187901 | 1.153222717 | 0.114381452 |
| PD-L1 (positive) | −0.33701656 | 0.713897018 | −0.79189525 | 0.11786213 | 0.452985461 | 1.125088984 | 0.146467095 |
| Platelets to lymphocytes ratio | −0.322180026 | 0.724567738 | −0.652891336 | 0.008531284 | 0.520538547 | 1.008567779 | 0.056210093 |
| Age (middle) | −0.276002656 | 0.758810916 | −0.722468713 | 0.170463402 | 0.485552087 | 1.185854251 | 0.225650961 |
| Smoking (packs/year) | −0.247906764 | 0.780432704 | −0.476616326 | −0.019197202 | 0.6208807 | 0.98098589 | 0.033630231 |
| First ICIs line (2) | −0.208104308 | 0.812122321 | −0.617793 | 0.201584384 | 0.539132992 | 1.223339463 | 0.319455347 |
| Histology (SCC) | −0.174563429 | 0.839823584 | −1.443825478 | 1.094698621 | 0.236023128 | 2.988281943 | 0.78750135 |
| Histology (ADK) | −0.11182882 | 0.894197313 | −1.305146045 | 1.081488405 | 0.271132935 | 2.949065689 | 0.854269791 |
| Corticoids pre-ICIs (yes) | −0.058363841 | 0.943306672 | −0.594921579 | 0.478193898 | 0.551605821 | 1.613158241 | 0.831175452 |
| Statins (yes) | −0.045064475 | 0.955935846 | −0.469399658 | 0.379270708 | 0.625377596 | 1.461218546 | 0.835112989 |
| Antidiabetic drugs (yes) | 0.124236215 | 1.132283302 | −0.417358939 | 0.665831369 | 0.658784414 | 1.946107783 | 0.653002448 |
| CNS Met. At ICIs start (yes) | 0.165526565 | 1.18001431 | −0.369831564 | 0.700884695 | 0.690850685 | 2.015535052 | 0.544515894 |
| Betablockers (yes) | 0.166015388 | 1.180591269 | −0.405754355 | 0.737785132 | 0.666473863 | 2.091298431 | 0.569300122 |
| Neutrophils to lymphocytes ratio | 0.206942426 | 1.229911758 | −0.157958907 | 0.571843759 | 0.85388487 | 1.771530317 | 0.266339197 |
| LDH before ICIs start | 0.212285577 | 1.236500951 | 0.033498548 | 0.391072605 | 1.034065943 | 1.478565861 | 0.019954947 |
| Liver Met. At ICIs start (yes) | 0.216398756 | 1.241597375 | −0.282048877 | 0.71484639 | 0.754236819 | 2.043872699 | 0.394819692 |
| First ICIs line (>2) | 0.226301968 | 1.253954263 | −0.292801411 | 0.745405348 | 0.746170307 | 2.107295451 | 0.392860157 |
| ECOG at ICIs start | 0.31212435 | 1.366324584 | 0.107503857 | 0.516744843 | 1.113495155 | 1.676561287 | 0.002792578 |
| Smoking (yes) | 0.323985338 | 1.382627035 | −0.36367997 | 1.011650646 | 0.695113616 | 2.750136773 | 0.355791634 |
| Bone Met. At ICIs start (yes) | 0.388614778 | 1.474936265 | 0.00245578 | 0.774773777 | 1.002458798 | 2.170101144 | 0.048560724 |
| PD-L1 (negative) | 0.651155188 | 1.917754917 | 0.028236571 | 1.274073805 | 1.028639002 | 3.575388368 | 0.040480882 |
| Histology (LCNEC) | 1.016874796 | 2.764541493 | −0.570709179 | 2.604458771 | 0.565124522 | 13.5239038 | 0.209337005 |
| coef | exp(coef) | coef Lower 95% | coef Upper 95% | exp(coef) Lower 95% | exp(coef) Upper 95% | p | |
|---|---|---|---|---|---|---|---|
| irAEs (yes) | −1.17629018 | 0.308420805 | −1.689034653 | −0.663545707 | 0.184697735 | 0.515021976 | 6.9125 × 10−6 |
| Sex (female) | −0.425381431 | 0.653520465 | −0.886418215 | 0.035655353 | 0.412129271 | 1.036298628 | 0.070546507 |
| Age (elder) | 0.020976826 | 1.021198387 | −0.748568714 | 0.790522367 | 0.473043129 | 2.204547708 | 0.957392402 |
| PD-L1 (positive) | −0.162555299 | 0.849969087 | −0.628550091 | 0.303439493 | 0.533364571 | 1.354509632 | 0.49416206 |
| Platelets to lymphocytes ratio | −0.301424347 | 0.73976379 | −0.635059432 | 0.032210738 | 0.529903994 | 1.032735119 | 0.076604011 |
| Age (middle) | −0.230925221 | 0.793798823 | −0.704497311 | 0.242646869 | 0.49435702 | 1.274618438 | 0.339210729 |
| Smoking (packs/year) | −0.083313356 | 0.920062795 | −0.334292255 | 0.167665543 | 0.715844543 | 1.182541035 | 0.515293654 |
| First ICIs line (2) | −0.190668303 | 0.826406659 | −0.631537143 | 0.250200537 | 0.53177376 | 1.284282938 | 0.396632245 |
| Histology (SCC) | −0.55449072 | 0.574364699 | −1.853459843 | 0.744478403 | 0.15669409 | 2.105343008 | 0.402789624 |
| Histology (ADK) | −0.378308731 | 0.685018982 | −1.565201451 | 0.80858399 | 0.209045896 | 2.244727179 | 0.53215713 |
| Corticoids pre-ICIs (yes) | 0.297373388 | 1.346317904 | −0.27292365 | 0.867670425 | 0.7611509 | 2.381356839 | 0.306782927 |
| Statins (yes) | −0.521778936 | 0.593463874 | −1.00774932 | −0.035808552 | 0.365039643 | 0.96482499 | 0.035345032 |
| Antidiabetic drugs (yes) | −0.245060142 | 0.782657466 | −0.900516883 | 0.410396599 | 0.406359565 | 1.507415506 | 0.4636894 |
| CNS Met. At ICIs start (yes) | −0.188443944 | 0.82824693 | −0.738623387 | 0.361735499 | 0.477771169 | 1.435819117 | 0.502020309 |
| Betablockers (yes) | −0.077242336 | 0.925665504 | −0.736352462 | 0.58186779 | 0.478857384 | 1.789377494 | 0.818331261 |
| Neutrophils to lymphocytes ratio | 0.379749171 | 1.461917852 | 0.020283753 | 0.739214589 | 1.020490866 | 2.094289989 | 0.038399938 |
| LDH before ICIs start | 0.203631208 | 1.225845988 | 0.013387751 | 0.393874665 | 1.013477768 | 1.482714701 | 0.03591487 |
| Liver Met. At ICIs start (yes) | 0.609518258 | 1.839545 | 0.11918694 | 1.099849576 | 1.126580502 | 3.00371416 | 0.014835056 |
| First ICIs line (>2) | −0.016989338 | 0.983154167 | −0.579587935 | 0.545609259 | 0.560129129 | 1.725659435 | 0.95280321 |
| ECOG at ICIs start | 0.332175729 | 1.393997792 | 0.109309707 | 0.55504175 | 1.115507778 | 1.742013713 | 0.003486043 |
| Smoking (yes) | 0.099953185 | 1.105119181 | −0.610449023 | 0.810355393 | 0.543106947 | 2.24870702 | 0.782727943 |
| Bone Met. At ICIs start (yes) | 0.662856619 | 1.940327201 | 0.25418509 | 1.071528149 | 1.289410439 | 2.919838039 | 0.001477737 |
| PD-L1 (negative) | 0.732340436 | 2.079942889 | −0.00562912 | 1.470309992 | 0.994386694 | 4.350583576 | 0.051773332 |
| Histology (LCNEC) | −0.311295214 | 0.732497601 | −2.056409813 | 1.433819386 | 0.127912376 | 4.194689775 | 0.72662371 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Callejo, A.; Frigola, J.; Iranzo, P.; Carbonell, C.; Diaz, N.; Marmolejo, D.; Assaf, J.D.; Cedrés, S.; Martinez-Marti, A.; Navarro, A.; et al. Interrelations between Patients’ Clinicopathological Characteristics and Their Association with Response to Immunotherapy in a Real-World Cohort of NSCLC Patients. Cancers 2021, 13, 3249. https://doi.org/10.3390/cancers13133249
Callejo A, Frigola J, Iranzo P, Carbonell C, Diaz N, Marmolejo D, Assaf JD, Cedrés S, Martinez-Marti A, Navarro A, et al. Interrelations between Patients’ Clinicopathological Characteristics and Their Association with Response to Immunotherapy in a Real-World Cohort of NSCLC Patients. Cancers. 2021; 13(13):3249. https://doi.org/10.3390/cancers13133249
Chicago/Turabian StyleCallejo, Ana, Joan Frigola, Patricia Iranzo, Caterina Carbonell, Nely Diaz, David Marmolejo, Juan David Assaf, Susana Cedrés, Alex Martinez-Marti, Alejandro Navarro, and et al. 2021. "Interrelations between Patients’ Clinicopathological Characteristics and Their Association with Response to Immunotherapy in a Real-World Cohort of NSCLC Patients" Cancers 13, no. 13: 3249. https://doi.org/10.3390/cancers13133249






