Familial Risk and Heritability of Hematologic Malignancies in the Nordic Twin Study of Cancer
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cumulative Incidence and Measures of Cross-Cancer Familial Dependence
2.2. Biometric Modelling
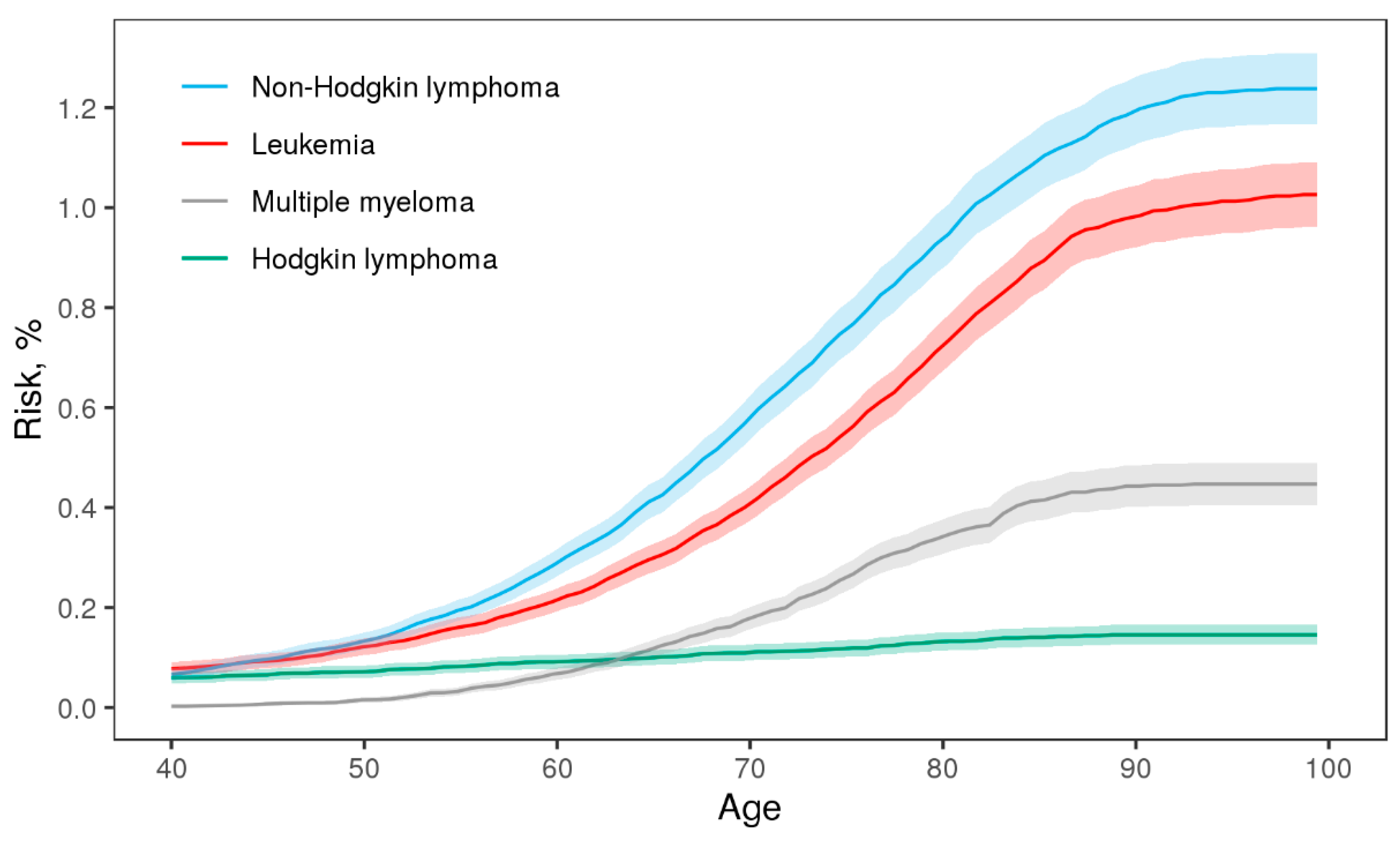
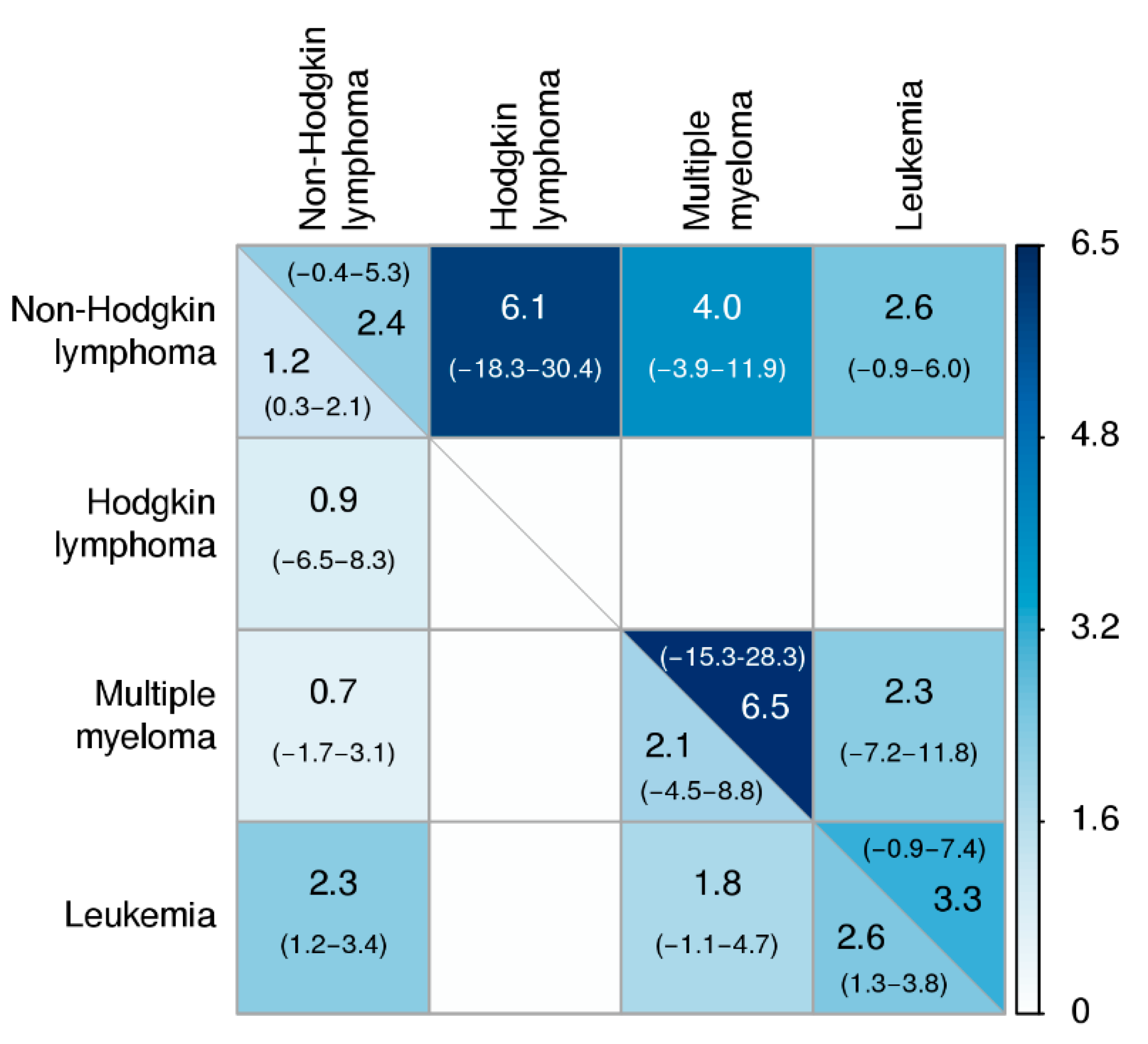
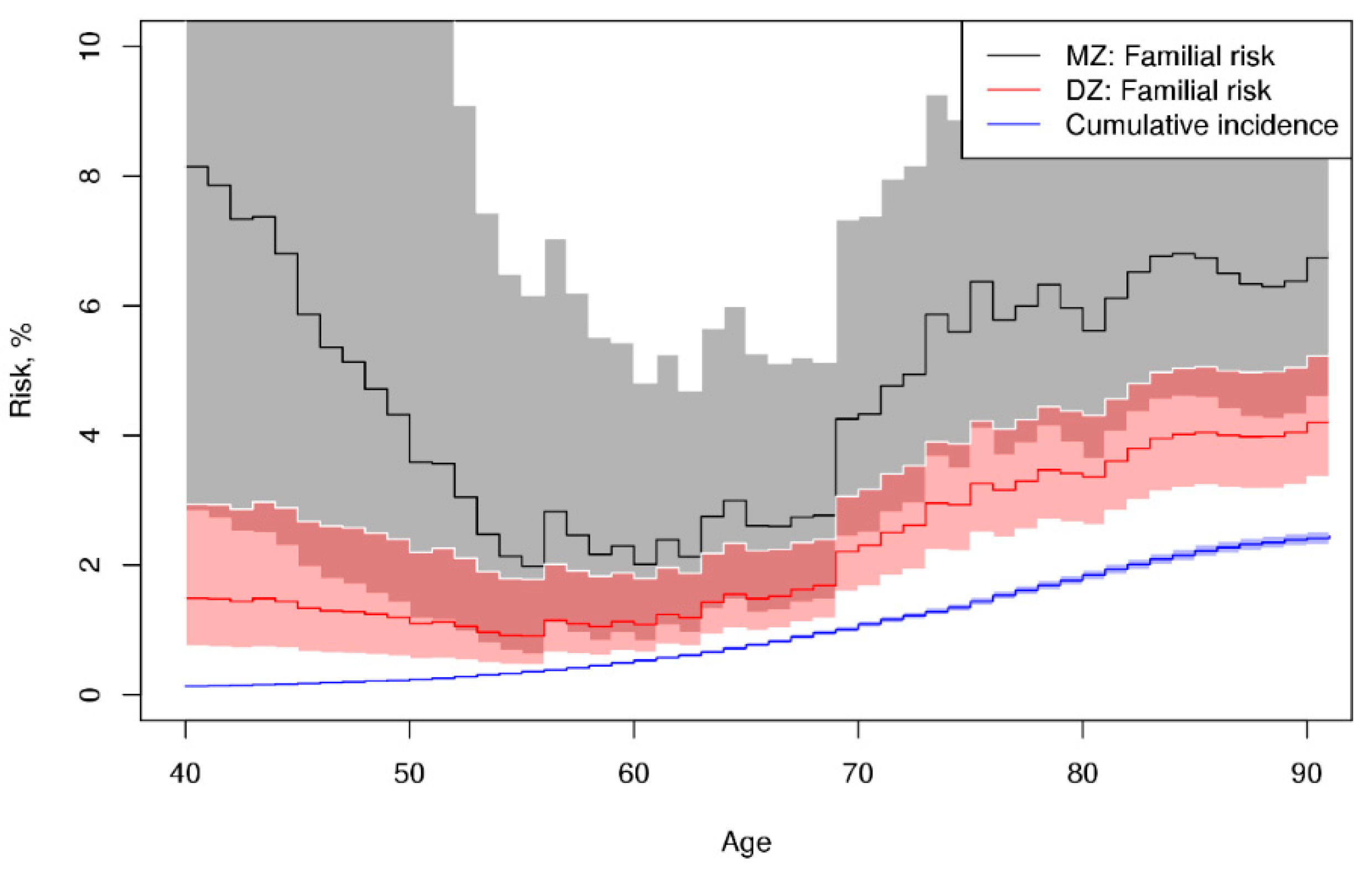
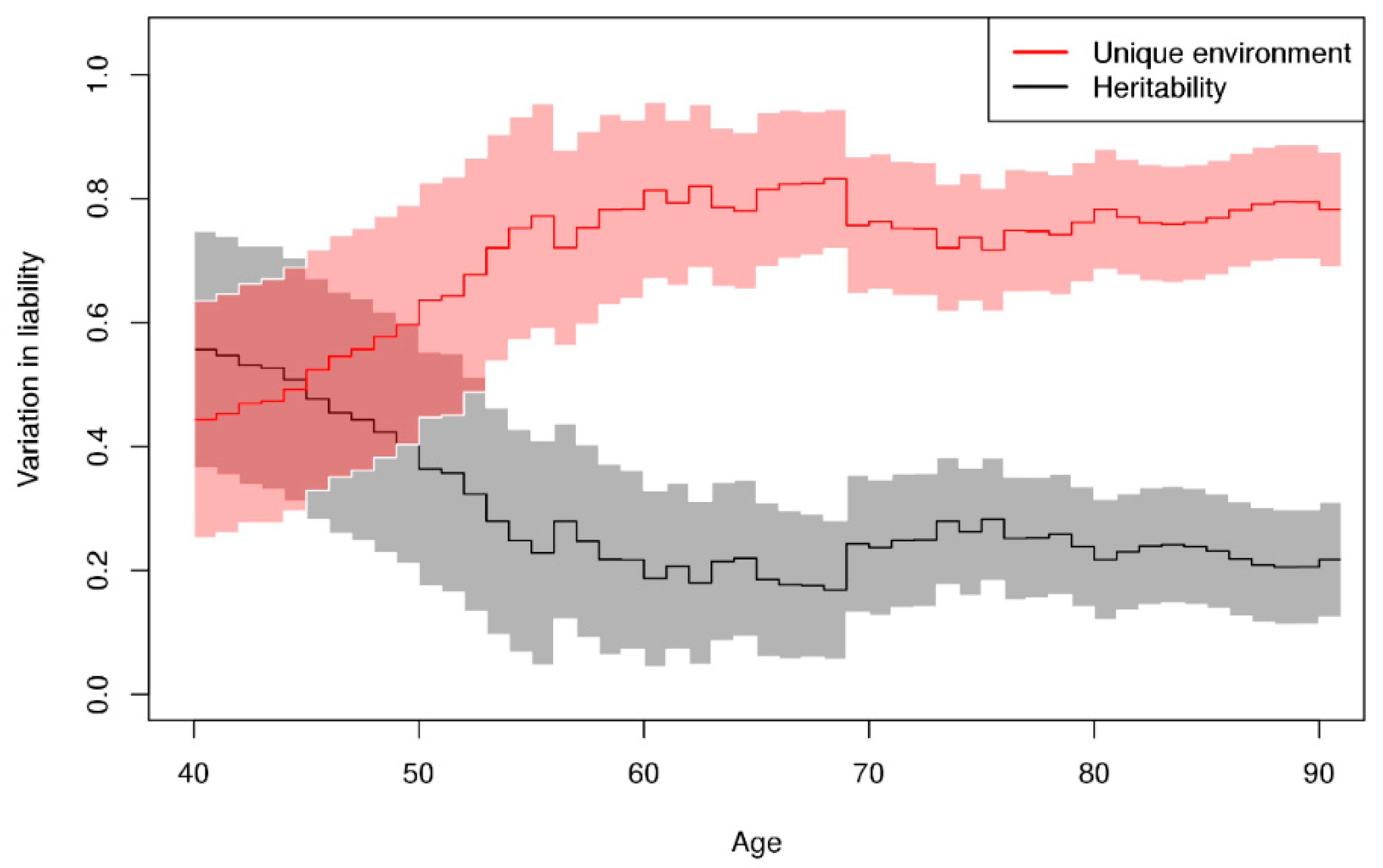
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Danckert, B.; Ferlay, J.; Engholm, G.; Hansen, H.L.; Johannesen, T.B.; Khan, S.; Køtlum, J.E.; Ólafsdóttir, E.; Schmidt, L.K.H.; Virtanen, A.; et al. NORDCAN: Cancer Incidence, Mortality, Prevalence and Survival in the Nordic Countries. Available online: https://www-dep.iarc.fr/nordcan/english/frame.asp (accessed on 3 May 2021).
- Global Burden of Disease Cancer Collaboration. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived with Disability, and Disability-Adjusted Life-Years for 29 Cancer Groups, 1990 to 2016: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol. 2018, 4, 1553–1568. [Google Scholar] [CrossRef]
- Hjalgrim, H. On the aetiology of Hodgkin lymphoma. Dan. Med. J. 2012, 59, 4485. [Google Scholar]
- Chiu, B.C.-H.; Weisenburger, D.D. An Update of the Epidemiology of Non-Hodgkin’s Lymphoma. Clin. Lymphoma 2003, 4, 161–168. [Google Scholar] [CrossRef]
- Juliusson, G.; Hough, R. Leukemia. Neuronal Act. Tumor Tissue 2016, 43, 87–100. [Google Scholar]
- Alexander, D.D.; Mink, P.J.; Adami, H.-O.; Cole, P.; Mandel, J.S.; Oken, M.M.; Trichopoulos, D. Multiple myeloma: A review of the epidemiologic literature. Int. J. Cancer 2007, 120 (Suppl. 12), 40–61. [Google Scholar] [CrossRef]
- Fallah, M.; Kharazmi, E.; Pukkala, E.; Tretli, S.; Olsen, J.H.; Tryggvadottir, L.; Sundquist, K.; Hemminki, K. Familial risk of non-Hodgkin lymphoma by sex, relationship, age at diagnosis and histology: A joint study from five Nordic countries. Leukemia 2015, 30, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.D.; Mink, P.J.; Adami, H.-O.; Chang, E.T.; Cole, P.; Mandel, J.S.; Trichopoulos, D. The non-Hodgkin lymphomas: A review of the epidemiologic literature. Int. J. Cancer 2007, 120, 1–39. [Google Scholar] [CrossRef]
- Kharazmi, E.; Fallah, M.; Pukkala, E.; Olsen, J.H.; Tryggvadottir, L.; Sundquist, K.; Tretli, S.; Hemminki, K. Risk of familial classical Hodgkin lymphoma by relationship, histology, age, and sex: A joint study from five Nordic countries. Blood 2015, 126, 1990–1995. [Google Scholar] [CrossRef] [PubMed]
- Mucci, L.A.; Hjelmborg, J.; Harris, J.R.; Czene, K.; Havelick, D.J.; Scheike, T.; Graff, R.E.; Holst, K.; Möller, S.; Unger, R.; et al. Familial Risk and Heritability of Cancer Among Twins in Nordic Countries. JAMA 2016, 315, 68–76. [Google Scholar] [CrossRef]
- Harris, J.R.; Hjelmborg, J.; Adami, H.-O.; Czene, K.; Mucci, L.; Kaprio, J. Nordic Twin Study of Cancer (NorTwinCan) Collaboration The Nordic Twin Study on Cancer—NorTwinCan. Twin Res. Hum. Genet. 2019, 22, 817–823. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, R.; Holford, T.R.; Leaderer, B.; Zahm, S.H.; Boyle, P.; Zhu, Y.; Qin, Q.; Zheng, T. Family history of hema-topoietic and non-hematopoietic malignancies and risk of non-Hodgkin lymphoma. Cancer Causes Control 2007, 18, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.T.; Smedby, K.E.; Hjalgrim, H.; Porwit-MacDonald, A.; Roos, G.; Glimelius, B.; Adami, H.-O. Family History of Hematopoietic Malignancy and Risk of Lymphoma. J. Natl. Cancer Inst. 2005, 97, 1466–1474. [Google Scholar] [CrossRef]
- Altieri, A.; Bermejo, J.L.; Hemminki, K. Familial risk for non-Hodgkin lymphoma and other lymphoproliferative malignancies by histopathologic subtype: The Swedish Family-Cancer Database. Blood 2005, 106, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Slager, S.L.; Brennan, P.; Holly, E.A.; De Sanjose, S.; Bernstein, L.; Boffetta, P.; Cerhan, J.R.; Maynadie, M.; Spinelli, J.J.; et al. Family history of hematopoietic malignancies and risk of non-Hodgkin lymphoma (NHL): A pooled analysis of 10 211 cases and 11 905 controls from the International Lymphoma Epidemiology Consortium (InterLymph). Blood 2006, 109, 3479–3488. [Google Scholar] [CrossRef]
- Schinasi, L.H.; Brown, E.E.; Camp, N.J.; Wang, S.S.; Hofmann, J.N.; Chiu, B.C.; Miligi, L.; Freeman, L.E.B.; De Sanjose, S.; Bernstein, L.; et al. Multiple myeloma and family history of lymphohaematopoietic cancers: Results from the International Multiple Myeloma Consortium. Br. J. Haematol. 2016, 175, 87–101. [Google Scholar] [CrossRef] [PubMed]
- VanValkenburg, M.E.; Pruitt, G.I.; Brill, I.K.; Costa, L.; Ehtsham, M.; Justement, I.T.; Innis-Shelton, R.D.; Salzman, D.; Reddy, E.S.P.; Godby, K.N.; et al. Family history of hematologic malignancies and risk of multiple myeloma: Differences by race and clinical features. Cancer Causes Control. 2015, 27, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Cerhan, J.R.; Slager, S.L. Familial predisposition and genetic risk factors for lymphoma. Blood 2015, 126, 2265–2273. [Google Scholar] [CrossRef] [PubMed]
- Sud, A.; Chattopadhyay, S.; Thomsen, H.; Sundquist, K.; Sundquist, J.; Houlston, R.S.; Hemminki, K. Analysis of 153115 patients with hematological malignancies refines the spectrum of familial risk. Blood 2019, 134. [Google Scholar] [CrossRef]
- Mensah, F.; Willett, E.; Ansell, P.; Adamson, P.; Roman, E. Non-Hodgkin’s Lymphoma and Family History of Hematologic Malignancy. Am. J. Epidemiol. 2006, 165, 126–133. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Goldgar, D.E.; Easton, D.F.; Cannon-Albright, L.A.; Skolnick, M.H. Systematic Population-Based Assessment of Cancer Risk in First-Degree Relatives of Cancer Probands. J. Natl. Cancer Inst. 1994, 86, 1600–1608. [Google Scholar] [CrossRef] [PubMed]
- Mack, T.M.; Cozen, W.; Shibata, D.K.; Weiss, L.M.; Nathwani, B.N.; Hernandez, A.M.; Taylor, C.R.; Hamilton, A.S.; Deapen, D.M.; Rappaport, E.B. Concordance for Hodgkin’s Disease in Identical Twins Suggesting Genetic Susceptibility to the Young-Adult Form of the Disease. N. Engl. J. Med. 1995, 332, 413–419. [Google Scholar] [CrossRef]
- Lichtenstein, P.; Holm, N.V.; Verkasalo, P.K.; Iliadou, A.; Kaprio, J.; Koskenvuo, M.; Pukkala, E.; Skytthe, A.; Hemminki, K. Environmental and Heritable Factors in the Causation of Cancer—Analyses of Cohorts of Twins from Sweden, Denmark, and Finland. N. Engl. J. Med. 2000, 343, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Skytthe, A.; Harris, J.R.; Czene, K.; Mucci, L.; Adami, H.-O.; Christensen, K.; Hjelmborg, J.; Holm, N.V.; Nilsen, T.S.; Kaprio, J.; et al. Cancer Incidence and Mortality in 260,000 Nordic Twins With 30,000 Prospective Cancers. Twin Res. Hum. Genet. 2019, 22, 99–107. [Google Scholar] [CrossRef]
- Engholm, G.; Ferlay, J.; Christensen, N.; Bray, F.; Gjerstorff, M.L.; Klint, Å.; Køtlum, J.E.; Ólafsdóttir, E.; Pukkala, E.; Storm, H. NORDCAN—A Nordic tool for cancer information, planning, quality control and research. Acta Oncol. 2010, 49, 725–736. [Google Scholar] [CrossRef]
- Scheike, T.H.; Holst, K.K.; Hjelmborg, J.B. Estimating heritability for cause specific mortality based on twin studies. Lifetime Data Anal. 2013, 20, 210–233. [Google Scholar] [CrossRef] [PubMed]
- Scheike, T.H.; Holst, K.K.; Hjelmborg, J. Estimating twin concordance for bivariate competing risks twin data. Stat. Med. 2013, 33, 1193–1204. [Google Scholar] [CrossRef] [PubMed]
- Holst, K.K.; Scheike, T.H.; Hjelmborg, J. The liability threshold model for censored twin data. Comput. Stat. Data Anal. 2016, 93, 324–335. [Google Scholar] [CrossRef]
- Sham, P. Statistics in Human Genetics; Wiley: London, UK, 1998. [Google Scholar]
- Peters, A.; Nawrot, T.S.; Baccarelli, A.A. Hallmarks of environmental insults. Cell 2021, 184, 1455–1468. [Google Scholar] [CrossRef] [PubMed]
- Hjelmborg, J.B.; Scheike, T.; Holst, K.; Skytthe, A.; Penney, K.L.; Graff, R.E.; Pukkala, E.; Christensen, K.; Adami, H.-O.; Holm, N.V.; et al. The Heritability of Prostate Cancer in the Nordic Twin Study of Cancer. Cancer Epidemiology Biomarkers Prev. 2014, 23, 2303–2310. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.R-project.org/ (accessed on 3 May 2021).
- Chang, E.T.; Smedby, K.E.; Hjalgrim, H.; Glimelius, B.; Adami, H.-O. Reliability of Self-Reported Family History of Cancer in a Large Case–Control Study of Lymphoma. J. Natl. Cancer Inst. 2006, 98, 61–68. [Google Scholar] [CrossRef]
- Godley, L.A.; Shimamura, A. Genetic predisposition to hematologic malignancies: Management and surveillance. Blood 2017, 130, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, E.; Alegre, V.; García-Melgares, M.; Laguna, C.; Martin, B.; Sánchez, J.; Oliver, V. Tattoo pigment in two lymph nodes in a patient with melanoma. J. Eur. Acad. Dermatol. Venereol. 2007, 22, 101–102. [Google Scholar] [CrossRef] [PubMed]
- Schreiver, I.; Hesse, B.; Seim, C.; Castillo-Michel, H.; Villanova, J.; Laux, P.; Dreiack, N.; Penning, R.; Tucoulou, R.; Cotte, M.; et al. Synchrotron-based ν-XRF mapping and µ-FTIR microscopy enable to look into the fate and effects of tattoo pigments in human skin. Sci. Rep. 2017, 7, 11395. [Google Scholar] [CrossRef]
- Singh, R.S.; Diwan, A.H.; Prieto, V.G. Potential diagnostic pitfalls in melanoma arising in a cutaneous tattoo. Histopathology 2007, 51, 283–285. [Google Scholar] [CrossRef]
- Sepehri, M.; Sejersen, T.; Qvortrup, K.; Lerche, C.M.; Serup, J. Tattoo Pigments Are Observed in the Kupffer Cells of the Liver Indicating Blood-Borne Distribution of Tattoo Ink. Dermatology 2017, 233, 86–93. [Google Scholar] [CrossRef] [PubMed]
| Country | Denmark | Finland | Norway | Sweden | Total |
|---|---|---|---|---|---|
| Birth Cohort | 1870–2004 | 1875–1957 | 1915–1991 | 1886–2008 | |
| n individual twins | 112,560 | 31,447 | 32,332 | 140,058 | 316,397 |
| n (%) MZ twins | 25,120 (22) | 8368 (27) | 13,100 (41) | 42,030 (30) | 88,618 (28) |
| n (%) female twins | 55,652 (49) | 15,717 (50) | 17,473 (54) | 73,640 (53) | 162,482 (51) |
| First date follow-up | January 1943 | February 1974 | April 1956 | April 1961 | |
| End of follow-up | December 2016 | December 2016 | December 2015 | December 2015 | |
| Median follow-up time (IQR), years | 46.6 (26.5–62.1) | 41.3 (31.7–41.3) | 52.9 (39.4–59.9) | 42.0 (22.2–58.2) | 41.8 (26.4–58.2) |
| Median entry age (IQR), years | 0 (0–0) | 31.8 (25.6–43.1) | 2.4 (0–16.3) | 1.7 (0–23.3) | 0 (0–24.6) |
| Number of incident cancers | |||||
| Any cancer site | 19,581 | 7044 | 5465 | 26,359 | 58,449 |
| Non-Hodgkin lymphoma | 424 | 261 | 184 | 645 | 1514 |
| Hodgkin lymphoma | 80 | 29 | 22 | 98 | 229 |
| Multiple myeloma | 144 | 72 | 53 | 253 | 522 |
| Leukemia | 406 | 141 | 102 | 545 | 1194 |
| Cancer Site | Lifetime Risk, % (95% CI) 1 | Number of Twin Pairs | Familial Risk, % (95% CI) 1 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MZ | DZ | ||||||||
| Conc. | Disc. | Conc. | Disc. | MZ | DZ | ||||
| Other 3 | None 4 | Other 3 | None 4 | ||||||
| Overall hematologic 2 | 2.5 (2.4–2.6) | 21 | 232 | 591 | 30 | 592 | 1653 | 7.6 (4.8–11.8) | 4.5 (3.1–6.5) |
| Non-Hodgkin lymphoma | 1.1 (1.0–1.2) | 4 | 115 | 246 | 8 | 279 | 732 | 5.2 (1.8–13.9) | 2.1 (1.0–4.3) |
| Hodgkin lymphoma | 0.1 (0.1–0.1) | 1–3 | 14 | 52 | 0 | 33 | 110 | 7.4 (1.8–25.6) | - |
| Multiple myeloma | 0.4 (0.3–0.4) | 1–3 | 49 | 94 | 1–3 | 92 | 244 | 2.6 (0.6–10.0) | 1.4 (0.2–9.2) |
| Leukemia | 0.9 (0.8–1.0) | 6 | 72 | 207 | 8 | 219 | 582 | 5.4 (2.1–13.3) | 3.6 (1.7–7.3) |
| Cancer Site | Tetrachoric Correlations, (95% CI) | Heritability, (95% CI) | Shared Environment, (95% CI) | Unique Environment, (95% CI) | |
|---|---|---|---|---|---|
| MZ | DZ | ||||
| Overall hematologic 1 | 0.24 (0.12–0.35) | 0.12 (0.03–0.20) | 0.24 (0.14–0.33) | 0.00 (NA–NA) | 0.76 (0.67–0.86) |
| Non-Hodgkin lymphoma | 0.28 (0.05–0.48) | 0.10 (−0.03–0.22) | 0.25 (0.08–0.42) | 0.00 (NA–NA) | 0.75 (0.58–0.92) |
| Hodgkin lymphoma | 0.57 (0.29–0.76) | - | 0.56 (0.32–0.80) | 0.00 (NA–NA) | 0.44 (0.20–0.68) |
| Multiple myeloma | 0.26 (0.02–0.48) | 0.17 (−0.12–0.43) | 0.19 (−0.54–0.93) | 0.07 (−0.54–0.69) | 0.74 (0.50–0.97) |
| Leukemia | 0.31 (0.10–0.49) | 0.23 (0.09–0.36) | 0.16 (−0.31–0.64) | 0.14 (−0.19–0.48) | 0.69 (0.50–0.89) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clemmensen, S.B.; Harris, J.R.; Mengel-From, J.; Bonat, W.H.; Frederiksen, H.; Kaprio, J.; Hjelmborg, J.v.B. Familial Risk and Heritability of Hematologic Malignancies in the Nordic Twin Study of Cancer. Cancers 2021, 13, 3023. https://doi.org/10.3390/cancers13123023
Clemmensen SB, Harris JR, Mengel-From J, Bonat WH, Frederiksen H, Kaprio J, Hjelmborg JvB. Familial Risk and Heritability of Hematologic Malignancies in the Nordic Twin Study of Cancer. Cancers. 2021; 13(12):3023. https://doi.org/10.3390/cancers13123023
Chicago/Turabian StyleClemmensen, Signe B., Jennifer R. Harris, Jonas Mengel-From, Wagner H. Bonat, Henrik Frederiksen, Jaakko Kaprio, and Jacob v. B. Hjelmborg. 2021. "Familial Risk and Heritability of Hematologic Malignancies in the Nordic Twin Study of Cancer" Cancers 13, no. 12: 3023. https://doi.org/10.3390/cancers13123023
APA StyleClemmensen, S. B., Harris, J. R., Mengel-From, J., Bonat, W. H., Frederiksen, H., Kaprio, J., & Hjelmborg, J. v. B. (2021). Familial Risk and Heritability of Hematologic Malignancies in the Nordic Twin Study of Cancer. Cancers, 13(12), 3023. https://doi.org/10.3390/cancers13123023






