Development of EndoScreen Chip, a Microfluidic Pre-Endoscopy Triage Test for Esophageal Adenocarcinoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
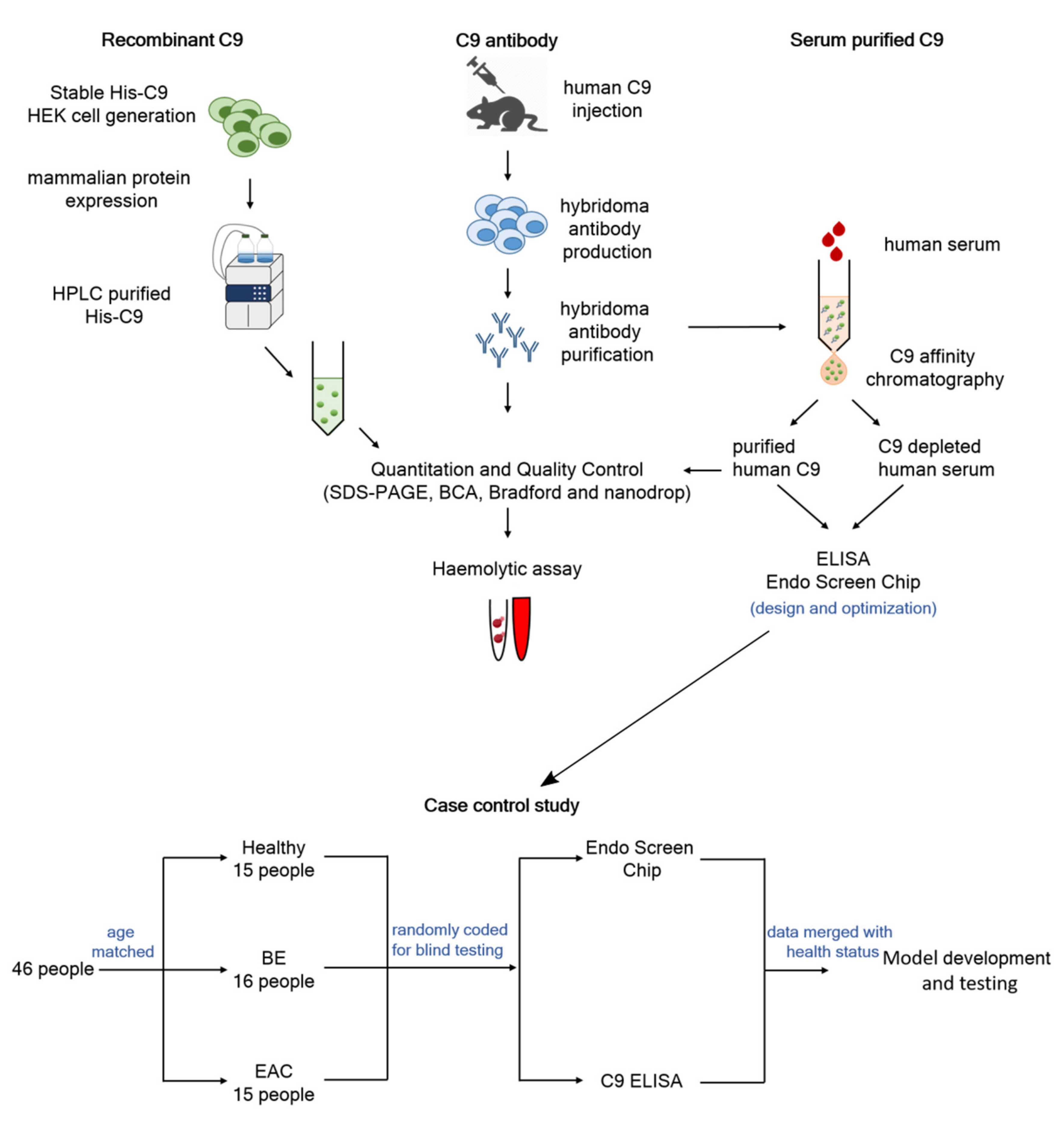
2.1. Study Cohort and Experimental Design
2.2. Recombinant Protein Expression and Purification
2.3. Antibody Production and Purification
2.4. Serum Purified C9 and Production of Depleted Serum
2.5. Characterization of mAb 26 by Western Blot
2.6. Hemolytic Assay
2.7. C9 Direct ELISA
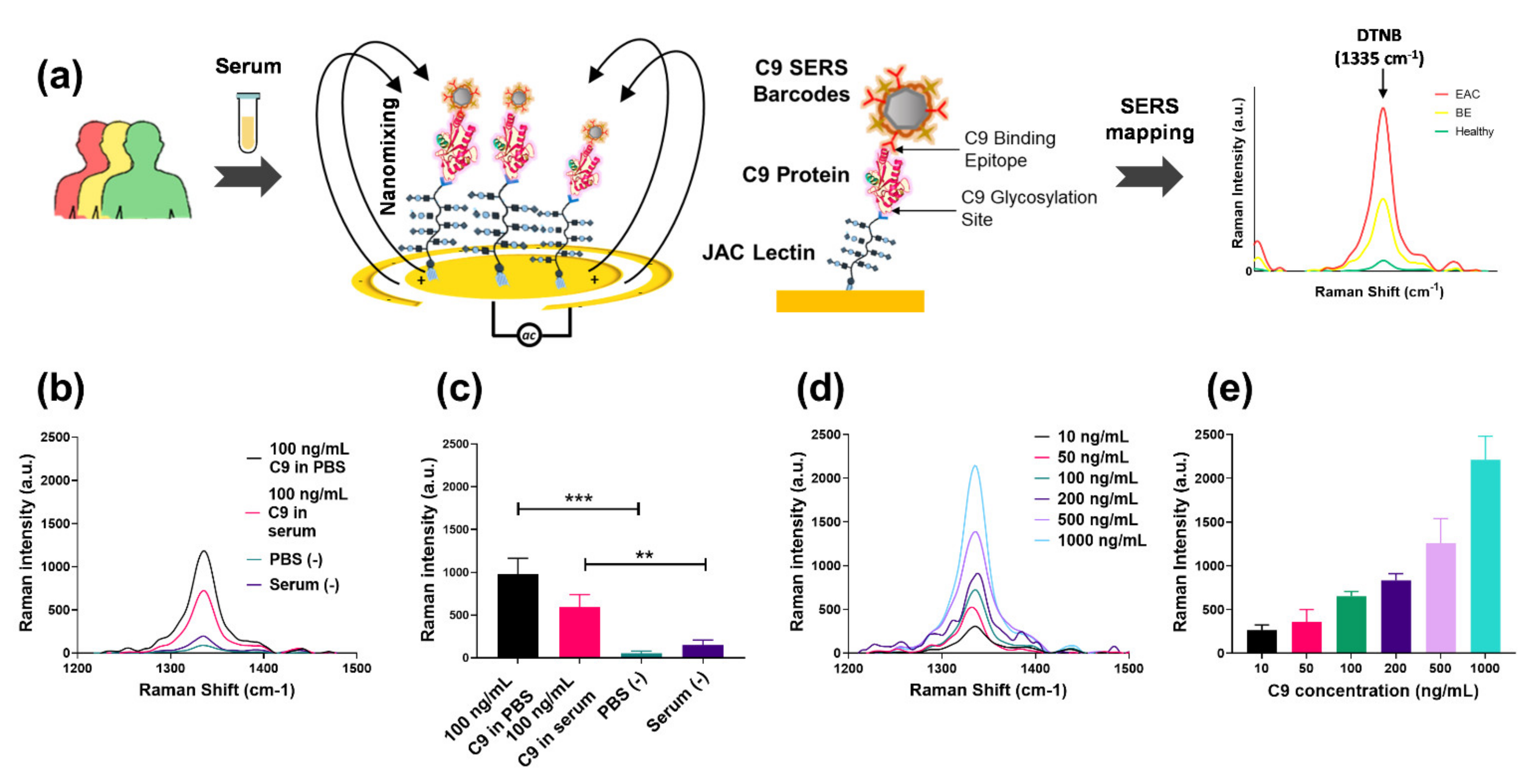
2.8. EndoScreen Chip Fabrication and Functionalization
2.9. SERS Nanotags Synthesis
2.10. C9 Sample Preparation for EndoScreen Chip Assay
2.11. EndoScreen Chip Assay
2.12. Statistical Analysis
3. Results
3.1. Validation of New C9 Monoclonal Antibody
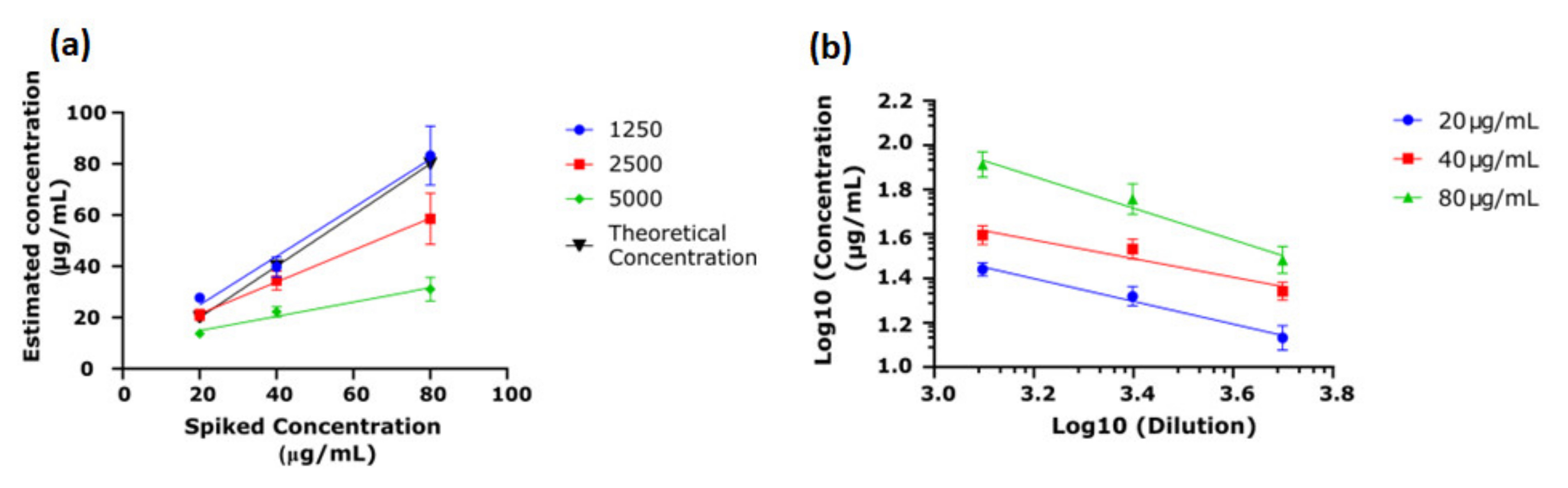
3.2. Establishing the Endo Screen Chip
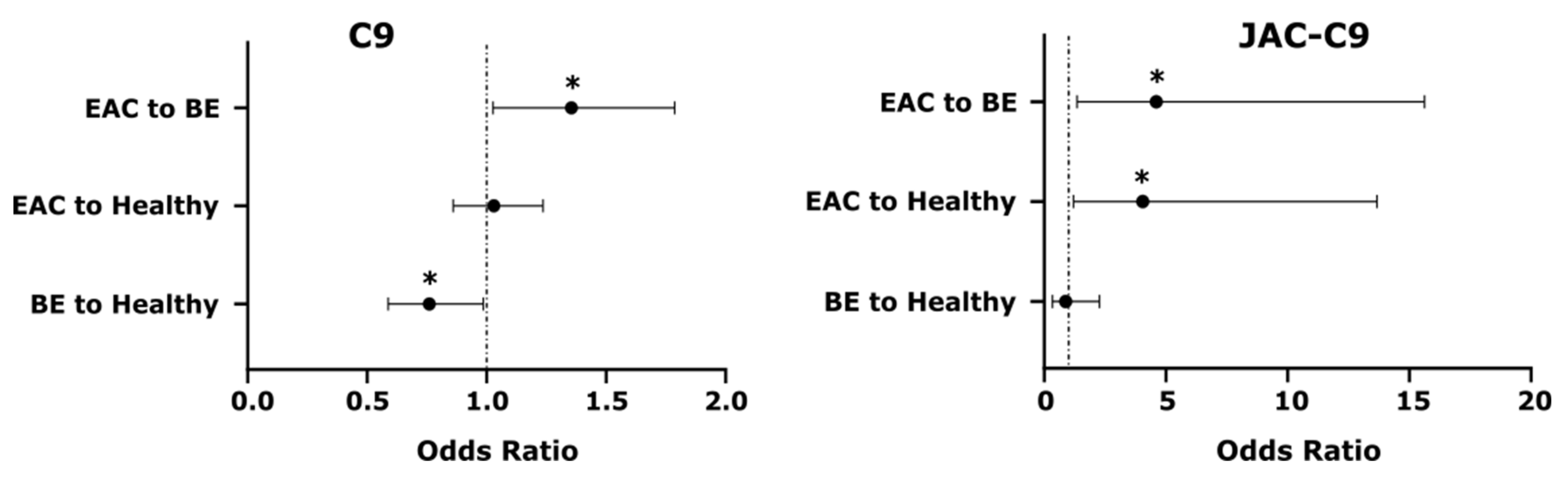
3.3. Total C9 and JAC-C9 Are Elevated in EAC Patient Serum
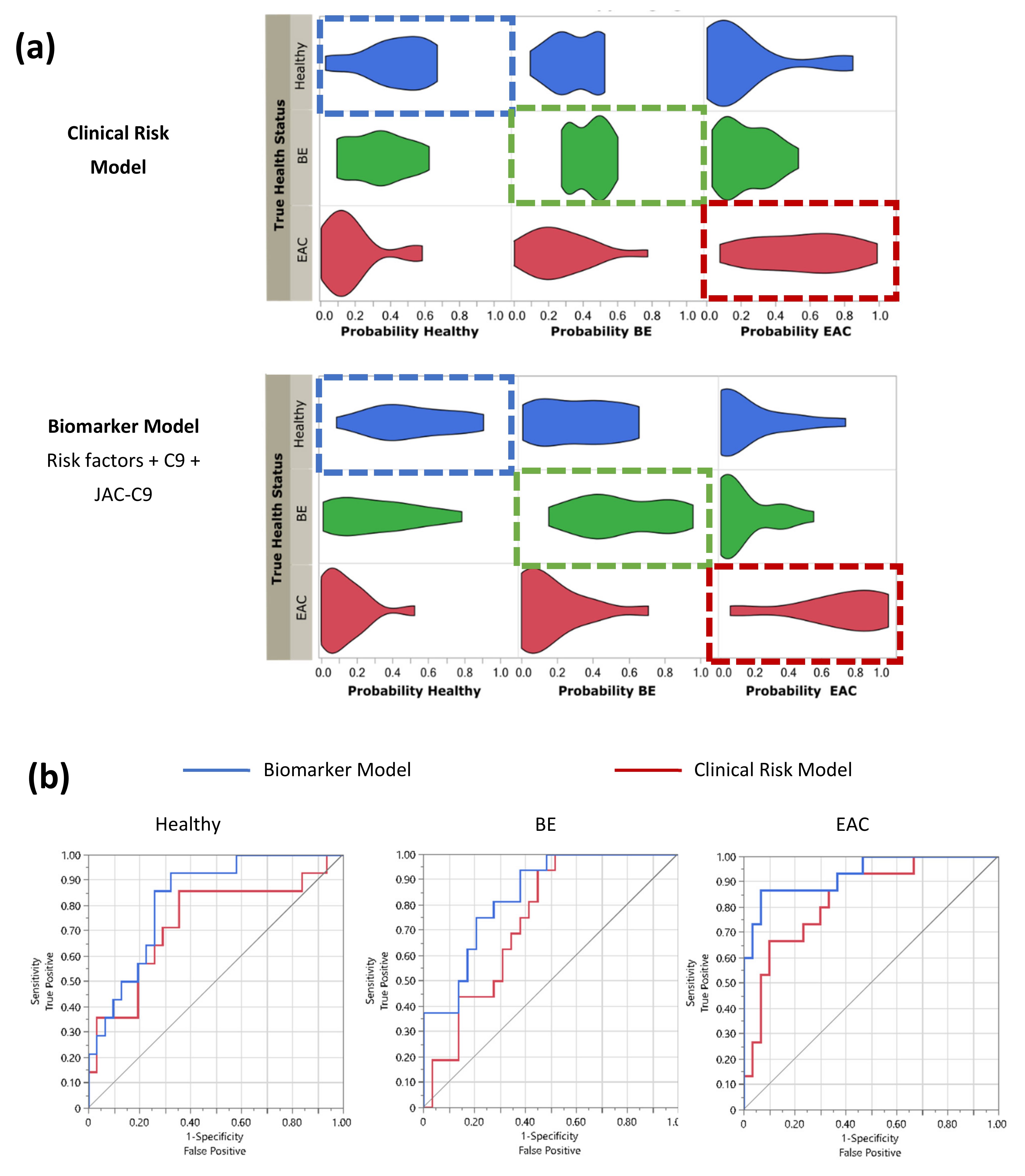
3.4. Serum C9 Biomarker Panel Improves the Detection of EAC over Clinical Risk Factors Alone
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Inadomi, J.M.; Saxena, N. Screening and Surveillance for Barrett’s Esophagus: Is It Cost-Effective? Dig. Dis. Sci. 2018, 63, 2094–2104. [Google Scholar] [CrossRef] [PubMed]
- Souza, R.F.; Spechler, S.J. Advances in Biomarkers for Risk Stratification in Barrett’s Esophagus. Gastrointest. Endosc. Clin. North Am. 2021, 31, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.K.; Sharma, A.N.; Chak, A.; Fitzgerald, R.C. Progress in Screening for Barrett’s Esophagus. Gastrointest. Endosc. Clin. North Am. 2021, 31, 43–58. [Google Scholar] [CrossRef] [PubMed]
- Thrift, A.P. Barrett’s Esophagus and Esophageal Adenocarcinoma: How Common Are They Really? Dig. Dis. Sci. 2018, 63, 1988–1996. [Google Scholar] [CrossRef]
- Ireland, C.J.; Thrift, A.P.; Esterman, A. Risk Prediction Models for Barrett’s Esophagus Discriminate Well and Are Generalizable in an External Validation Study. Dig. Dis. Sci. 2020, 65, 2992–2999. [Google Scholar] [CrossRef]
- Rubenstein, J.H.; Morgenstern, H.; Appelman, H.; Scheiman, J.; Schoenfeld, P.; McMahon, L.F.; Metko, V.; Near, E.; Kellenberg, J.; Kalish, T.; et al. Prediction of Barrett’s Esophagus Among Men. Am. J. Gastroenterol. 2013, 108, 353–362. [Google Scholar] [CrossRef]
- Wani, S.; Rubenstein, J.H.; Vieth, M.; Bergman, J. Diagnosis and Management of Low-Grade Dysplasia in Barrett’s Esophagus: Expert Review From the Clinical Practice Updates Committee of the American Gastroenterological Association. Gastroenterology 2016, 151, 822–835. [Google Scholar] [CrossRef]
- Rosenfeld, A.; Graham, D.G.; Jevons, S.; Ariza, J.; Hagan, D.; Wilson, A.; Lovat, S.J.; Sami, S.S.; Ahmad, O.F.; Novelli, M.; et al. Development and validation of a risk prediction model to diagnose Barrett’s oesophagus (MARK-BE): A case-control machine learning approach. Lancet Digit. Health 2020, 2, e37–e48. [Google Scholar] [CrossRef]
- Coleman, H.G.; Xie, S.-H.; Lagergren, J. The Epidemiology of Esophageal Adenocarcinoma. Gastroenterology 2018, 154, 390–405. [Google Scholar] [CrossRef]
- Cook, M.B.; Thrift, A.P. Epidemiology of Barrett’s Esophagus and Esophageal Adenocarcinoma. Gastrointest. Endosc. Clin. North Am. 2021, 31, 1–26. [Google Scholar] [CrossRef]
- Tan, M.C.; Mansour, N.; White, D.L.; Sisson, A.; El-Serag, H.B.; Thrift, A.P. Systematic review with meta-analysis: Prevalence of prior and concurrent Barrett’s oesophagus in oesophageal adenocarcinoma patients. Aliment. Pharmacol. Ther. 2020, 52, 20–36. [Google Scholar] [CrossRef]
- Grady, W.M.; Yu, M.; Markowitz, S.D.; Chak, A. Barrett’s Esophagus and Esophageal Adenocarcinoma Biomarkers. Cancer Epidemiol. Biomark. Prev. 2020, 29, 2486–2494. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Saunders, N.; Barbour, A.P.; Hill, M.M. Early Diagnostic Biomarkers for Esophageal Adenocarcinoma—The Current State of Play. Cancer Epidemiol. Biomark. Prev. 2013, 22, 1185–1209. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.; Loo, R.; Dennis, J.W.; O’Leary, C.A.; Hill, M.M. High-throughput lectin magnetic bead array-coupled tandem mass spectrometry for glycoprotein biomarker discovery. Electrophoresis 2011, 32, 3564–3575. [Google Scholar] [CrossRef]
- Shah, A.; Le Cao, K.-A.; Choi, E.; Chen, D.; Gautier, B.; Nancarrow, D.; Whiteman, D.C.; Saunders, N.; Barbour, A.P.; Joshi, V.; et al. Serum Glycoprotein Biomarker Discovery and Qualification Pipeline Reveals Novel Diagnostic Biomarker Candidates for Esophageal Adenocarcinoma. Mol. Cell. Proteom. 2015, 14, 3023–3039. [Google Scholar] [CrossRef]
- Shah, A.; Hartel, G.; Brown, I.; Winterford, C.; Na, R.; Cao, K.-A.L.; Spicer, B.A.; Dunstone, M.A.; Phillips, W.A.; Lord, R.V.; et al. Evaluation of Serum Glycoprotein Biomarker Candidates for Detection of Esophageal Adenocarcinoma and Surveillance of Barrett’s Esophagus. Mol. Cell. Proteom. 2018, 17, 2324–2334. [Google Scholar] [CrossRef]
- Li, J.; Wuethrich, A.; Dey, S.; Lane, R.E.; Sina, A.A.I.; Wang, J.; Wang, Y.; Puttick, S.; Koo, K.M.; Trau, M. The Growing Impact of Micro/Nanomaterial-Based Systems in Precision Oncology: Translating “Multiomics” Technologies. Adv. Funct. Mater. 2020, 30. [Google Scholar] [CrossRef]
- Zong, C.; Xu, M.; Xu, L.-J.; Wei, T.; Ma, X.; Zheng, X.-S.; Hu, R.; Ren, B. Surface-Enhanced Raman Spectroscopy for Bioanalysis: Reliability and Challenges. Chem. Rev. 2018, 118, 4946–4980. [Google Scholar] [CrossRef]
- Li, J.; Wuethrich, A.; Sina, A.A.I.; Cheng, H.-H.; Wang, Y.; Behren, A.; Mainwaring, P.N.; Trau, M. A digital single-molecule nanopillar SERS platform for predicting and monitoring immune toxicities in immunotherapy. Nat. Commun. 2021, 12, 1–12. [Google Scholar] [CrossRef]
- Khondakar, K.R.; Dey, S.; Wuethrich, A.; Ibn Sina, A.A.; Trau, M. Toward Personalized Cancer Treatment: From Diagnostics to Therapy Monitoring in Miniaturized Electrohydrodynamic Systems. Accounts Chem. Res. 2019, 52, 2113–2123. [Google Scholar] [CrossRef]
- Nancarrow, D.J.; Clouston, A.D.; Smithers, B.M.; Gotley, D.C.; Drew, P.A.; Watson, D.I.; Tyagi, S.; Hayward, N.; Whiteman, D.C.; For the Australian Cancer Study and the Study of Digestive Health. Whole Genome Expression Array Profiling Highlights Differences in Mucosal Defense Genes in Barrett’s Esophagus and Esophageal Adenocarcinoma. PLoS ONE 2011, 6, e22513. [Google Scholar] [CrossRef]
- Smith, K.J.; O’Brien, S.M.; Smithers, B.M.; Gotley, D.C.; Webb, P.M.; Green, A.C.; Whiteman, D.C. Interactions among smoking, obesity, and symptoms of acid reflux in Barrett’s esophagus. Cancer Epidemiol. Biomarkers Prev. 2005, 14, 2481–2486. [Google Scholar] [CrossRef]
- Xie, S.-H.; Lagergren, J. A global assessment of the male predominance in esophageal adenocarcinoma. Oncotarget 2016, 7, 38876–38883. [Google Scholar] [CrossRef]
- Dudkina, N.V.; Spicer, B.A.; Reboul, C.F.; Conroy, P.J.; Lukoyanova, N.; Elmlund, H.; Law, R.H.P.; Ekkel, S.M.; Kondos, S.C.; Goode, R.J.A.; et al. Structure of the poly-C9 component of the complement membrane attack complex. Nat. Commun. 2016, 7, 10588. [Google Scholar] [CrossRef] [PubMed]
- Köhler, G.; Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nat. Cell Biol. 1975, 256, 495–497. [Google Scholar] [CrossRef]
- Morgan, B.P. Immunoaffinity Methods for Purification of Complement Components and Regulators. In Complement Methods and Protocols; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2003; Volume 150, pp. 53–60. [Google Scholar]
- Zelek, W.M.; Harris, C.L.; Morgan, B.P. Extracting the barbs from complement assays: Identification and optimisation of a safe substitute for traditional buffers. Immunobiology 2018, 223, 744–749. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, J.; Shanmugasundaram, K.B.; Yeo, B.; Möller, A.; Wuethrich, A.; Lin, L.L.; Trau, M. Tracking Drug-Induced Epithelial–Mesenchymal Transition in Breast Cancer by a Microfluidic Surface-Enhanced Raman Spectroscopy Immunoassay. Small 2020, 16, 1905614. [Google Scholar] [CrossRef] [PubMed]
- Frens, G. Controlled Nucleation for the Regulation of the Particle Size in Monodisperse Gold Suspensions. Nat. Phys. Sci. 1973, 241, 20–22. [Google Scholar] [CrossRef]
- Tsao, S.C.-H.; Wang, J.; Wang, Y.; Behren, A.; Cebon, J.; Trau, M. Characterising the phenotypic evolution of circulating tumour cells during treatment. Nat. Commun. 2018, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Lui, H.; McLean, D.I.; Zeng, H. Automated Autofluorescence Background Subtraction Algorithm for Biomedical Raman Spectroscopy. Appl. Spectrosc. 2007, 61, 1225–1232. [Google Scholar] [CrossRef]
- Fenlon, C.; O’Grady, L.; Doherty, M.L.; Dunnion, J. A discussion of calibration techniques for evaluating binary and categorical predictive models. Prev. Veter-Med. 2018, 149, 107–114. [Google Scholar] [CrossRef]
- The United States Food and Drug Administration. Q2 (R1) Validation of Analytical Procedures: Text and Methodology. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/q2-r1-validation-analytical-procedures-text-and-methodology (accessed on 1 September 2020).
- Andreasson, U.; Perret-Liaudet, A.; Doorn, L.J.C.V.W.V.; Blennow, K.; Chiasserini, D.; Engelborghs, S.; Fladby, T.; Genc, S.; Kruse, N.; Kuiperij, B.; et al. A Practical Guide to Immunoassay Method Validation. Front. Neurol. 2015, 6, 179. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.; Creavin, A.; O’Connell, M.; O’Connor, B.; Clarke, P. Optimization of the enzyme-linked lectin assay for enhanced glycoprotein and glycoconjugate analysis. Anal. Biochem. 2011, 413, 114–122. [Google Scholar] [CrossRef]
- Ding, S.-Y.; Yi, J.; Li, J.-F.; Ren, B.; Wu, D.-Y.; Panneerselvam, R.; Tian, Z.-Q. Nanostructure-based plasmon-enhanced Raman spectroscopy for surface analysis of materials. Nat. Rev. Mater. 2016, 1, 16021. [Google Scholar] [CrossRef]
- Cotton, C.C.; Shaheen, N.J. Overutilization of Endoscopic Surveillance in Barrett’s Esophagus: The Perils of Too Much of a Good Thing. Am. J. Gastroenterol. 2020, 115, 1019–1021. [Google Scholar] [CrossRef] [PubMed]
- Australian Commission on Safety and Quality in Health Care. Third Australian Atlas of Healthcare Variation 2018; Australian Commission on Safety and Quality in Health Care: Sydney, Australia, 2018. [Google Scholar]
- Barbosa, A.I.; Gehlot, P.; Sidapra, K.; Edwards, A.D.; Reis, N.M. Portable smartphone quantitation of prostate specific antigen (PSA) in a fluoropolymer microfluidic device. Biosens. Bioelectron. 2015, 70, 5–14. [Google Scholar] [CrossRef]
- Hirst, N.G.; Gordon, L.; Whiteman, D.C.; Watson, D.I.; Barendregt, J.J. Is endoscopic surveillance for non-dysplastic Barrett’s esophagus cost-effective? Review of economic evaluations. J. Gastroenterol. Hepatol. 2011, 26, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Peters, Y.; Siersema, P.D. Public Preferences and Predicted Uptake for Esophageal Cancer Screening Strategies: A Labeled Discrete Choice Experiment. Clin. Transl. Gastroenterol. 2020, 11, e00260. [Google Scholar] [CrossRef]
- Lennon, A.M.; Buchanan, A.H.; Kinde, I.; Warren, A.; Honushefsky, A.; Cohain, A.T.; Ledbetter, D.H.; Sanfilippo, F.; Sheridan, K.; Rosica, D.; et al. Feasibility of blood testing combined with PET-CT to screen for cancer and guide intervention. Science 2020, 369, eabb9601. [Google Scholar] [CrossRef]
- Franc, V.; Yang, Y.; Heck, A.J.R. Proteoform Profile Mapping of the Human Serum Complement Component C9 Revealing Unexpected New Features of N-, O-, and C-Glycosylation. Anal. Chem. 2017, 89, 3483–3491. [Google Scholar] [CrossRef]
- Chong, P.-K.; Lee, H.; Loh, M.C.S.; Choong, L.-Y.; Lin, Q.; So, J.B.Y.; Lim, K.H.; Soo, R.A.; Yong, W.P.; Chan, S.P.; et al. Upregulation of plasma C9 protein in gastric cancer patients. Proteomics 2010, 10, 3210–3221. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, E.; Furuta, T.; Ohtsuki, S.; Tachikawa, M.; Uchida, Y.; Sabit, H.; Obuchi, W.; Baba, T.; Watanabe, M.; Terasaki, T.; et al. Identification of blood biomarkers in glioblastoma by SWATH mass spectrometry and quantitative targeted absolute proteomics. PLoS ONE 2018, 13, e0193799. [Google Scholar] [CrossRef] [PubMed]
- Murakoshi, Y.; Honda, K.; Sasazuki, S.; Ono, M.; Negishi, A.; Matsubara, J.; Sakuma, T.; Kuwabara, H.; Nakamori, S.; Sata, N.; et al. Plasma biomarker discovery and validation for colorectal cancer by quantitative shotgun mass spectrometry and protein microarray. Cancer Sci. 2010, 102, 630–638. [Google Scholar] [CrossRef]
- Narayanasamy, A.; Ahn, J.-M.; Sung, H.-J.; Kong, D.-H.; Ha, K.-S.; Lee, S.-Y.; Cho, J.-Y. Fucosylated glycoproteomic approach to identify a complement component 9 associated with squamous cell lung cancer (SQLC). J. Proteom. 2011, 74, 2948–2958. [Google Scholar] [CrossRef] [PubMed]
- Thurman, J.M.; Laskowski, J.; Nemenoff, R.A. Complement and Cancer—A Dysfunctional Relationship? Antibodies 2020, 9, 61. [Google Scholar] [CrossRef]
- Lindblad, M.; Bright, T.; Schloithe, A.; Mayne, G.C.; Chen, G.; Bull, J.; Bampton, P.A.; Fraser, R.J.L.; Gatenby, P.A.; Gordon, L.; et al. Toward More Efficient Surveillance of Barrett’s Esophagus: Identification and Exclusion of Patients at Low Risk of Cancer. World J. Surg. 2016, 41, 1023–1034. [Google Scholar] [CrossRef]
- Gordon, L.G.; Mayne, G.C.; Hirst, N.G.; Bright, T.; Whiteman, D.C.; Follow-Up, A.C.S.C.; Watson, D.I. Cost-effectiveness of endoscopic surveillance of non-dysplastic Barrett’s esophagus. Gastrointest. Endosc. 2014, 79, 242–256. [Google Scholar] [CrossRef]

| Parameter | Category | Healthy | BE | EAC | p Value (Healthy vs. BE vs. EAC) |
|---|---|---|---|---|---|
| Number | 15 | 16 | 15 | ||
| Age (years) | Median | 64.39 | 61.89 | 61.72 | 0.146 a |
| Range | 56–75 | 52–75 | 53–74 | ||
| BMI | Healthy wt (<25) | 10.9% (5) | 6.5% (3) | 2.2% (1) | 0.054 b |
| Overweight (<30) | 10.9% (5) | 23.9% (11) | 10.9% (5) | ||
| Obese I (<35) | 10.9% (5) | 4.3% (2) | 17.4% (8) | ||
| Obese II (<40) | 0.0% (0) | 0.0% (0) | 0.0% (0) | ||
| Obese III (≥40) | 0.0% (0) | 0.0% (0) | 2.2% (1) | ||
| Heartburn and Reflux History | Never | 11.1% (5) | 4.4% (2) | 2.2% (1) | 0.005 b |
| <Once/month | 6.7% (3) | 6.7% (3) | 2.2% (1) | ||
| Monthly (few times/month) | 8.9% (4) | 13.3% (6) | 2.2% (1) | ||
| Weekly (few times/wk) | 2.2% (1) | 0.0% (0) | 15.6% (7) | ||
| Daily | 2.2% (1) | 11.1% (5) | 11.1% (5) |
| Predictive model | Health Status: Healthy | ||||
|---|---|---|---|---|---|
| Predictor | AUC | StdError | Lower 95% | Upper 95% | Prob > ChiSq |
| Clinical Risk Model | 0.7350 | 0.0886 | 0.5322 | 0.8712 | |
| Biomarker Model | 0.8272 | 0.0621 | 0.6714 | 0.9181 | |
| Difference | −0.092 | 0.0635 | −0.217 | 0.0323 | 0.1465 |
| Health Status: BE | |||||
| Predictor | AUC | StdError | Lower 95% | Upper 95% | Prob > ChiSq |
| Clinical Risk Model | 0.7435 | 0.0726 | 0.5788 | 0.8595 | |
| Biomarker Model | 0.8405 | 0.0579 | 0.6934 | 0.9247 | |
| Difference | −0.097 | 0.0690 | −0.232 | 0.0382 | 0.1598 |
| Health Status: EAC | |||||
| Predictor | AUC | StdError | Lower 95% | Upper 95% | Prob > ChiSq |
| Clinical Risk Model | 0.8378 | 0.0624 | 0.6774 | 0.9270 | |
| Biomarker Model | 0.9311 | 0.0411 | 0.7936 | 0.9794 | |
| Difference | −0.093 | 0.0432 | −0.178 | −0.009 | 0.0309 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Webster, J.A.; Wuethrich, A.; Shanmugasundaram, K.B.; Richards, R.S.; Zelek, W.M.; Shah, A.K.; Gordon, L.G.; Kendall, B.J.; Hartel, G.; Morgan, B.P.; et al. Development of EndoScreen Chip, a Microfluidic Pre-Endoscopy Triage Test for Esophageal Adenocarcinoma. Cancers 2021, 13, 2865. https://doi.org/10.3390/cancers13122865
Webster JA, Wuethrich A, Shanmugasundaram KB, Richards RS, Zelek WM, Shah AK, Gordon LG, Kendall BJ, Hartel G, Morgan BP, et al. Development of EndoScreen Chip, a Microfluidic Pre-Endoscopy Triage Test for Esophageal Adenocarcinoma. Cancers. 2021; 13(12):2865. https://doi.org/10.3390/cancers13122865
Chicago/Turabian StyleWebster, Julie A., Alain Wuethrich, Karthik B. Shanmugasundaram, Renee S. Richards, Wioleta M. Zelek, Alok K. Shah, Louisa G. Gordon, Bradley J. Kendall, Gunter Hartel, B. Paul Morgan, and et al. 2021. "Development of EndoScreen Chip, a Microfluidic Pre-Endoscopy Triage Test for Esophageal Adenocarcinoma" Cancers 13, no. 12: 2865. https://doi.org/10.3390/cancers13122865
APA StyleWebster, J. A., Wuethrich, A., Shanmugasundaram, K. B., Richards, R. S., Zelek, W. M., Shah, A. K., Gordon, L. G., Kendall, B. J., Hartel, G., Morgan, B. P., Trau, M., & Hill, M. M. (2021). Development of EndoScreen Chip, a Microfluidic Pre-Endoscopy Triage Test for Esophageal Adenocarcinoma. Cancers, 13(12), 2865. https://doi.org/10.3390/cancers13122865










