Back to the Future: Rethinking the Great Potential of lncRNAS for Optimizing Chemotherapeutic Response in Ovarian Cancer
Abstract
1. Introduction
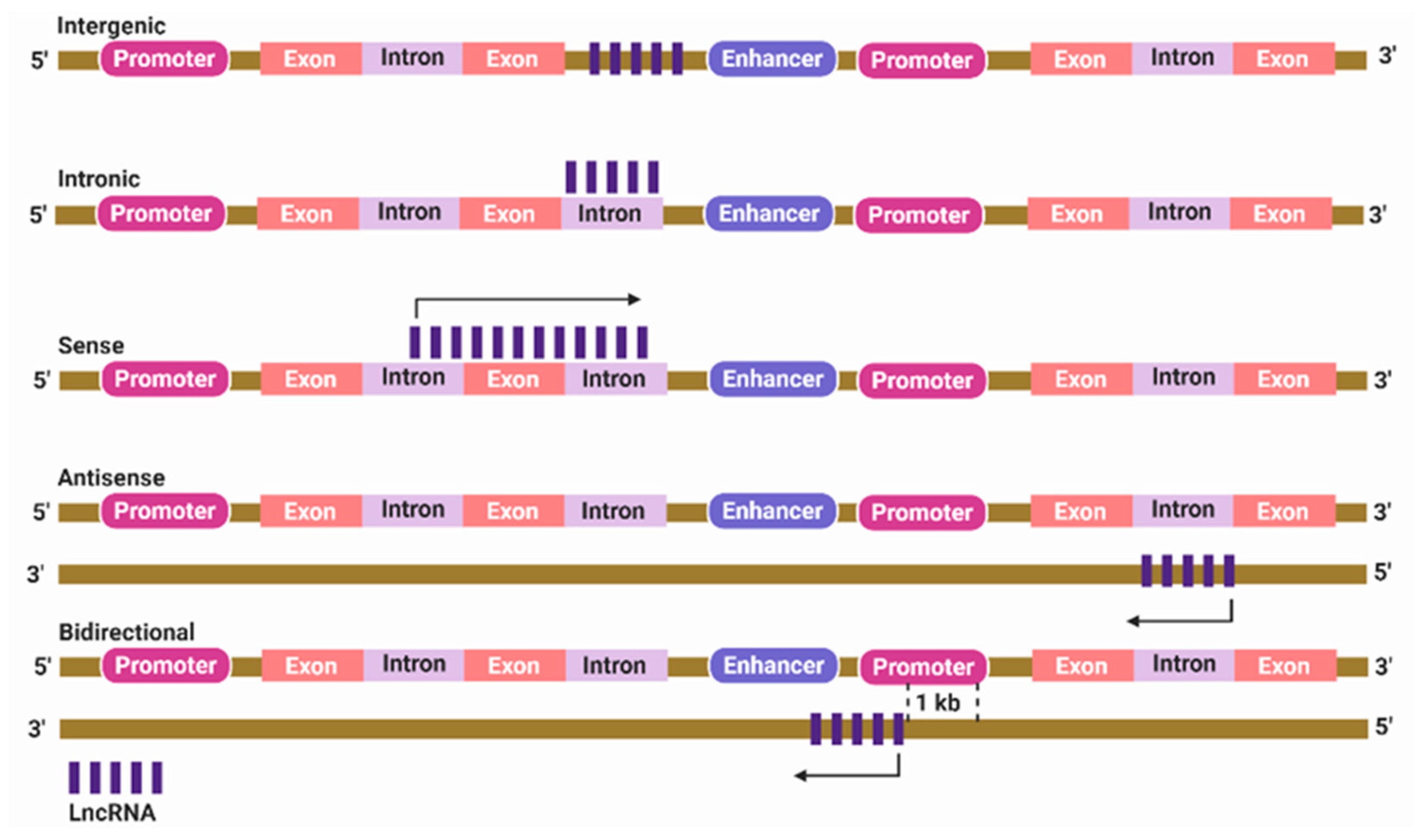
2. Classification of lncRNAs
3. Localization of lncRNAs
4. Major Biological Functions of lncRNAs
5. lncRNAs and Chemoresistance in Ovarian Cancer
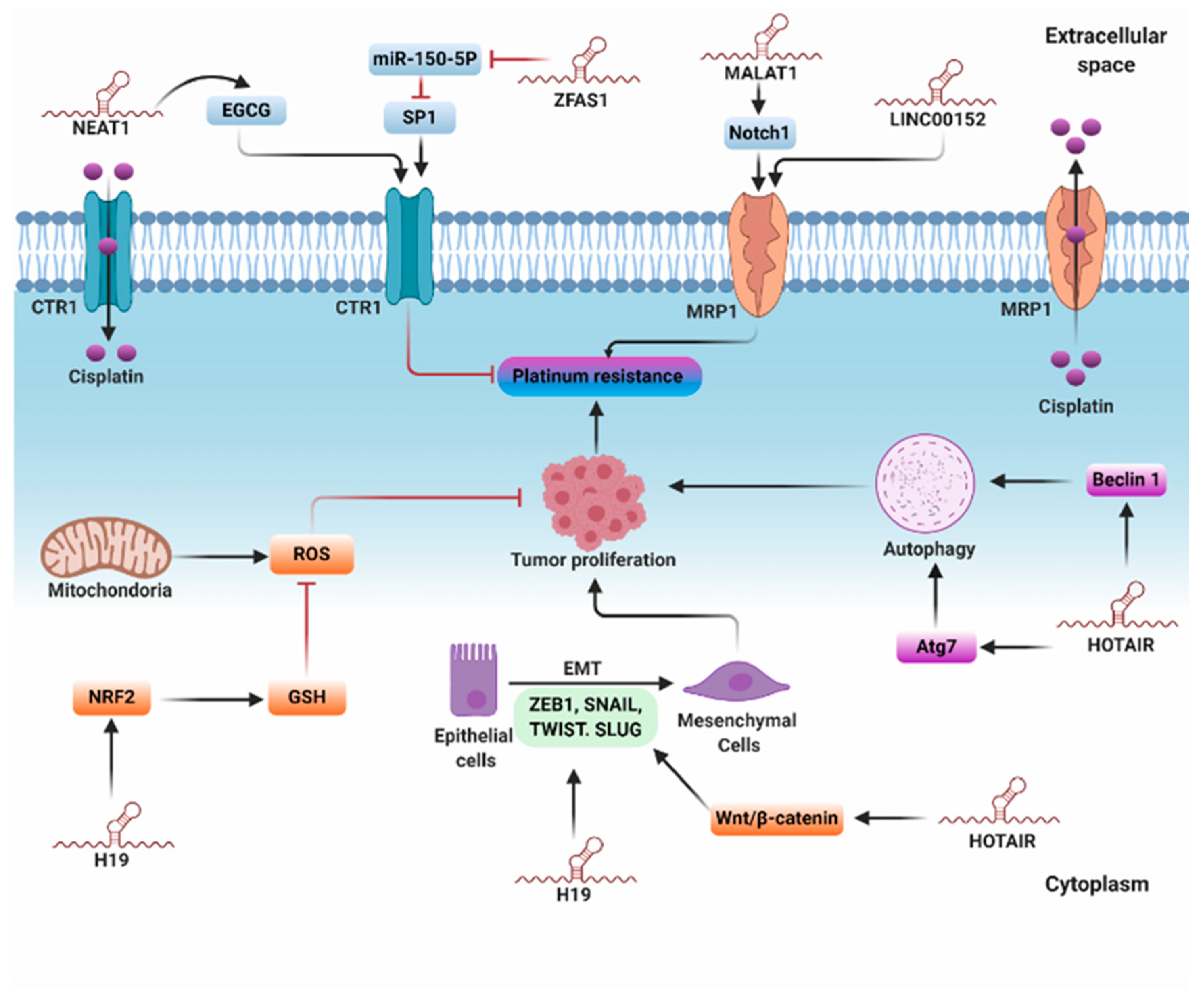
5.1. lncRNAs and Platinum Resistance
5.1.1. Reduced Intracellular Accumulation
5.1.2. Intracellular Detoxification
5.1.3. Regulation of Autophagy
5.1.4. Epithelial–Mesenchymal Transition
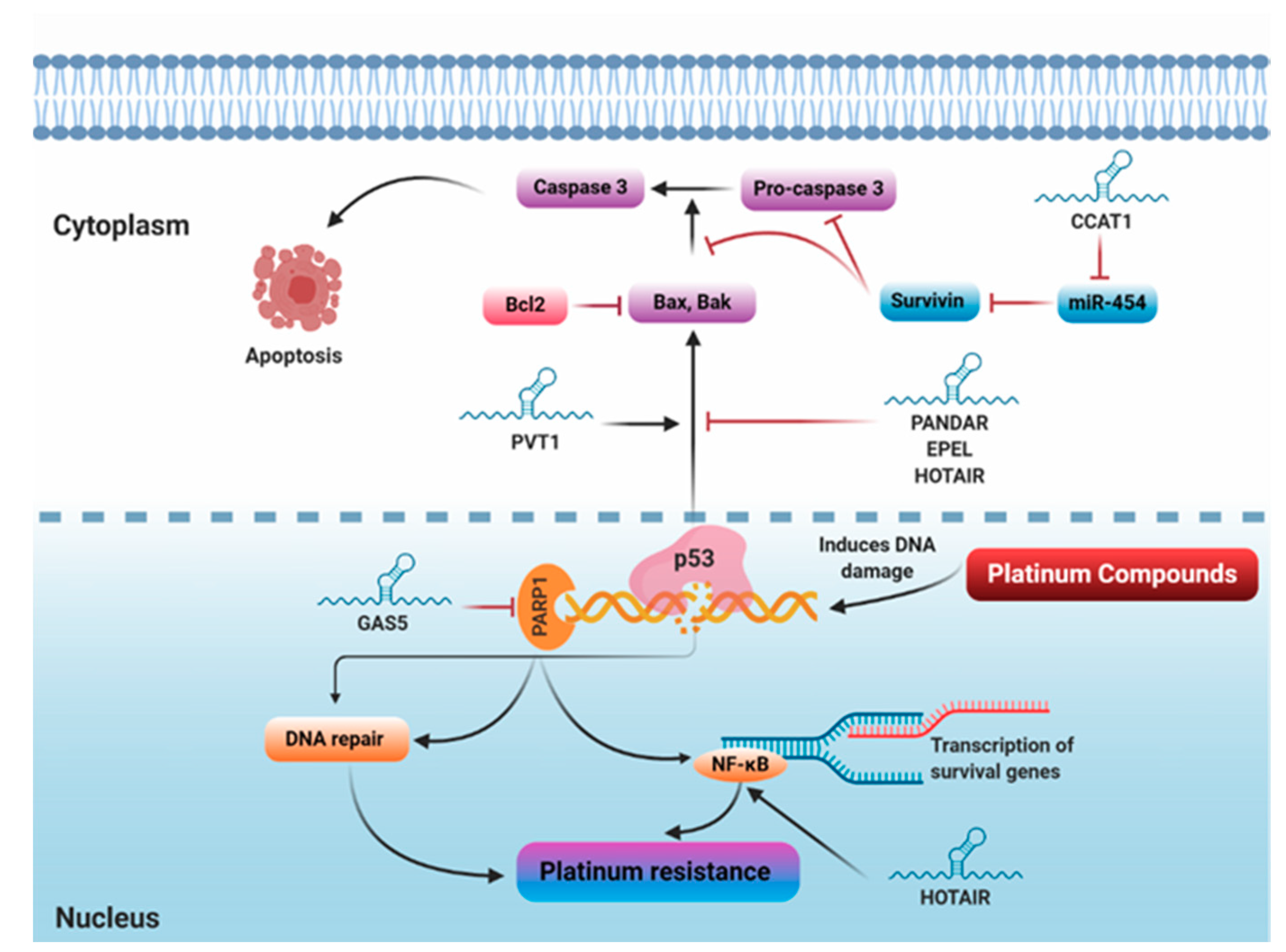
5.1.5. Repair of Damaged DNA
5.1.6. Regulation of Apoptosis
5.1.7. Sponging miRNAs
5.1.8. Other lncRNAs Implicated in Platinum Resistance
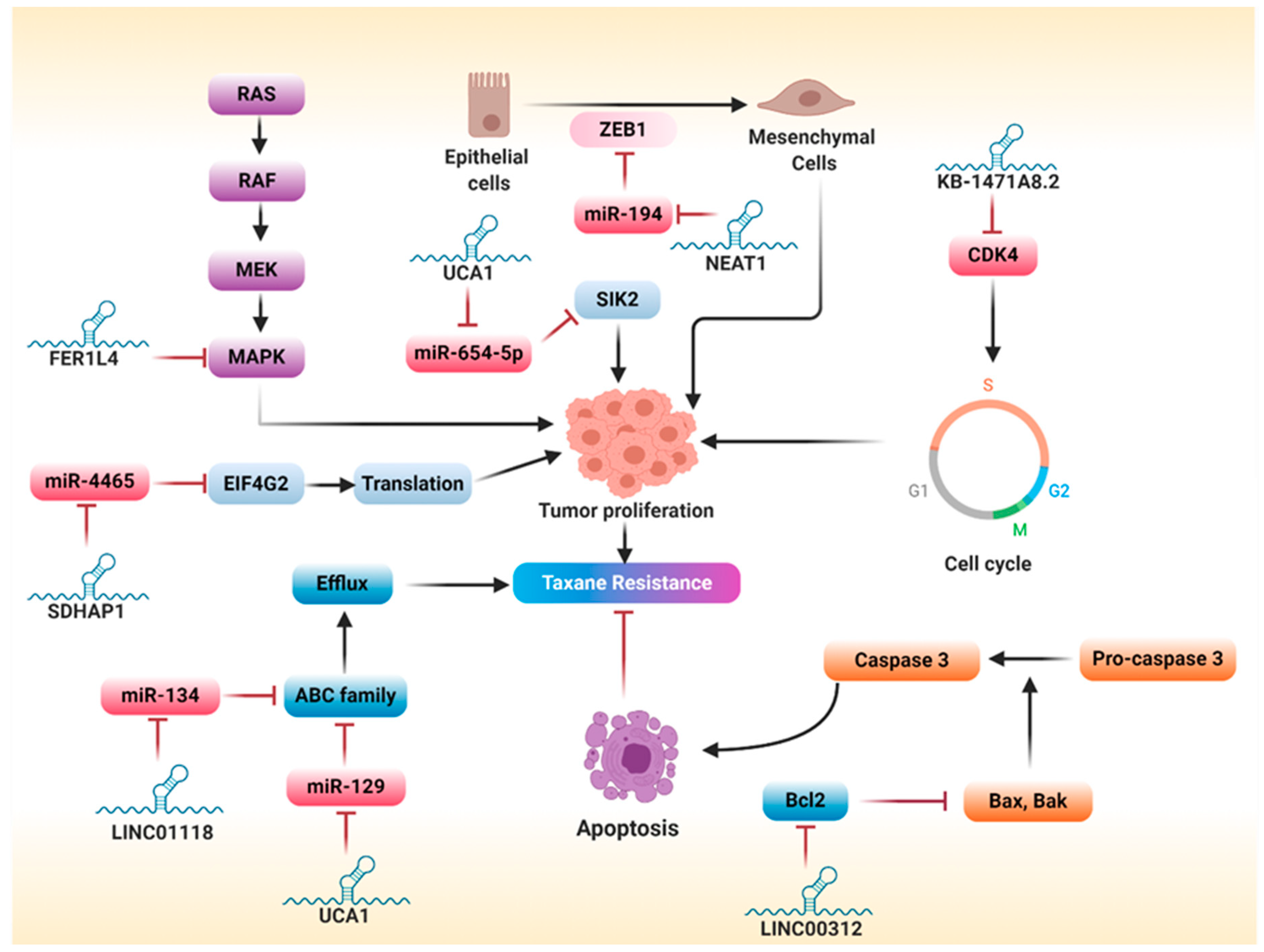
5.2. lncRNAs and Taxanes Resistance
5.2.1. Increased Drug Efflux
5.2.2. Epithelial–Mesenchymal Transition
5.2.3. Regulation of Survival Pathways
5.2.4. Regulation of Apoptosis
5.2.5. Regulation of Cell Cycle
5.2.6. Sponging miRNAs
5.2.7. Other lncRNAs Implicated in Paclitaxel Resistance
6. Therapeutic Approaches for Targeting lncRNAs
7. lncRNAs as Diagnostic and Prognostic Biomarkers
8. Challenges for lncRNAs Applications in Cancer
9. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABC | ATB binding cassette |
| AKT | Protein kinase B |
| Ago2 | Argonaute 2 |
| ASO | Antisense oligonucleotide |
| ATG7 | Autophagy related 7 homolog |
| BAD | Bcl2-associated death promoter |
| BAK | BCL-2 antagonist killer |
| BAX | BCL2-associated X protein |
| BCL-2 | B-cell lymphoma 2 |
| BCL-XL | B-cell lymphoma-extra-large protein |
| BIK | BCL-2 interacting killer |
| RAF | Rapidly accelerated fibrosarcoma |
| BRCA1/2 | Breast cancer susceptibility proteins 1 and 2 |
| CASC2 | Cancer susceptibility 2 |
| CDK4 | Cyclin dependent kinase 4 |
| CDKN1A (p21) | Cyclin dependent kinase inhibitor 1A |
| ceRNAs | Competitive endogenous RNAs |
| CHK1 | Checkpoint kinase 1 |
| CRISPR | Clustered regularly interspaced short palindromic repeats |
| CTNNB1 | Catenin beta 1 |
| CTR1 | Copper transporter 1 |
| DDR | DNA damage response |
| DOPC | 1,2-Dioleoyl-sn-Glycero-3-Phosphatidylcholine |
| DTA | Diphtheria toxin A |
| E2F4 | E2F transcription factor 4 |
| EC | Endometrial cancer |
| EGCG | Epigallocatechin-3-gallate |
| EMT | Epithelial mesenchymal transition |
| ERK | Extracellular signal-regulated kinase |
| eRNAs | Enhancer RNAs |
| EZH2 | Enhancer of the zeste 2 polycomb repressive complex 2 subunit |
| FISH | Fluorescent in situ hybridization |
| FOSL2 | FOS like 2 |
| FRA-2 | Fos-related antigen 2 |
| G6PD | Glucose-6-phosphate dehydrogenase |
| GCLC | Glutamate-cysteine ligase catalytic subunit |
| GCLM | Glutamate-cysteine ligase modifier subunit |
| GSH | Glutathione or reduced glutathione |
| GSR | Glutathione reductase |
| GSTP1 | Glutathione S-transferase Pi 1 |
| HIF1-α | Hypoxia-inducible factor 1 alpha |
| JNK | Jun N-terminal kinase |
| Kb | Kilobase |
| KPNA4 | Karyopherin α 4 |
| lncRNAs | Long non-coding RNAs |
| MAPK | Mitogen-activated protein kinase |
| MEF2D | Myocyte enhancer factor 2D |
| MEG3 | Maternally expressed 3 |
| miRNA | Micro RNA |
| MLH1 | MutL homolog 1 or DNA mismatch repair protein Mlh1 |
| MMR | Mismatch repair |
| MRPs | Multi-drug resistant proteins |
| MSH2 | MutS homolog 2 or DNA mismatch repair protein Msh2 |
| NF-κB | Nuclear factor kappa B |
| NQO1 | NADPH quinone oxidoreductase 1 |
| NRF2 | Nuclear factor erythroid 2 |
| OC | Ovarian cancer |
| OSCC | Ovarian squamous cell carcinoma |
| PARP | Poly (ADP-ribose) polymerase |
| PCAT1 | Prostate cancer associated transcript 1 |
| PEI | Polyethylenimine |
| PI3K | Phospho-inositide-3-kinase |
| PRC2 | Polycomb repressive complex 2 |
| PTEN | Phosphatase and tensin homolog |
| PTX | Paclitaxel |
| RAS | Rat sarcoma |
| RISC | RNA-induced silencing complex |
| RNAi | RNA interference |
| ROCK1 | Rho-associated coiled-coil-containing protein kinase 1 |
| ROS | Reactive oxygen species |
| SFRS2 | Arginine/serine-rich splicing factor 2 |
| shRNAs | Short hairpin RNA |
| SIK2 | Salt-inducible kinase 2 |
| SIRT1 | Sirtuin 1 |
| SP1 | The specific protein 1 |
| SRA1 | Steroid receptor RNA activator 1 |
| STAT3 | Signal transducer and activator of transcription 3 |
| TDRG1 | Testis development related 1 |
| TGF-β | Transforming growth factor beta 1 |
| TIMP1 | Tissue inhibitor of matrix metalloproteinase 1 |
| TP53 | Tumor protein 53 |
| WNT7A | Protein Wnt-7a |
| ZEB1 | Zinc finger E-box-binding homeobox 1 |
References
- Coleman, R.L.; Monk, B.J.; Sood, A.K.; Herzog, T.J. Latest research and treatment of advanced-stage epithelial ovarian cancer. Nat. Rev. Clin. Oncol. 2013, 10, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Kurman, R.J.; Shih, I.-M. Pathogenesis of ovarian cancer. Lessons from morphology and molecular biology and their clinical implications. Int. J. Gynecol. Pathol. 2008, 27, 151. [Google Scholar] [PubMed]
- Rosen, D.G.; Yang, G.; Liu, G.; Mercado-Uribe, I.; Chang, B.; Xiao, X.S.; Zheng, J.; Xue, F.X.; Liu, J. Ovarian cancer: Pathology, biology, and disease models. Front. Biosci. 2009, 14, 2089–2102. [Google Scholar] [CrossRef] [PubMed]
- Ducie, J.; Dao, F.; Considine, M.; Olvera, N.; Shaw, P.A.; Kurman, R.J.; Shih, I.-M.; Soslow, R.A.; Cope, L.; Levine, D.A. Molecular analysis of high-grade serous ovarian carcinoma with and without associated serous tubal intra-epithelial carcinoma. Nat. Commun. 2017, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Matulonis, U.A.; Sood, A.K.; Fallowfield, L.; Howitt, B.E.; Sehouli, J.; Karlan, B.Y. Ovarian cancer. Nat. Rev. Dis. Primers. 2016, 2, 16061. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Ueda, Y.; Naka, T.; Enomoto, T. Therapeutic strategies in epithelial ovarian cancer. J. Exp. Clin. Cancer Res. 2012, 31, 14. [Google Scholar] [CrossRef]
- Djebali, S.; Davis, C.; Merkel, A.; Dobin, A.; Lassmann, T.; Mortazavi, A.; Tanzer, A.; Lagarde, J.; Lin, W.; Schlesinger, F.; et al. Landscape of transcription in human cells. Nature 2012, 489, 101–108. [Google Scholar] [CrossRef]
- Palazzo, A.F.; Lee, E.S. Non-coding RNA: What is functional and what is junk? Front. Genet. 2015, 6, 2. [Google Scholar] [CrossRef]
- Abildgaard, C.; Do Canto, L.M.; Steffensen, K.D.; Rogatto, S.R. Long non-coding RNAs involved in resistance to chemotherapy in ovarian cancer. Front. Oncol. 2019, 9, 1549. [Google Scholar] [CrossRef]
- Iyer, M.K.; Niknafs, Y.S.; Malik, R.; Singhal, U.; Sahu, A.; Hosono, Y.; Barrette, T.R.; Prensner, J.R.; Evans, J.R.; Zhao, S. The landscape of long noncoding RNAs in the human transcriptome. Nat. Genet. 2015, 47, 199. [Google Scholar] [CrossRef]
- Goff, L.A.; Rinn, J.L. Linking RNA biology to lncRNAs. Genome. Res. 2015, 25, 1456–1465. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.M.; Anderson, K.M.; Chang, C.-L.; Makarewich, C.A.; Nelson, B.R.; McAnally, J.R.; Kasaragod, P.; Shelton, J.M.; Liou, J.; Bassel-Duby, R. A micropeptide encoded by a putative long noncoding RNA regulates muscle performance. Cell 2015, 160, 595–606. [Google Scholar] [CrossRef] [PubMed]
- Marchese, F.P.; Raimondi, I.; Huarte, M. The multidimensional mechanisms of long noncoding RNA function. Genome. Biol. 2017, 18, 206. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Li, X.; Zhi, H.; Zhang, Y.; Wang, P.; Wang, Y.; Shang, S.; Fang, Y.; Shen, W.; Ning, S. Comprehensive characterization of somatic mutations impacting lncRNA expression for pan-cancer. Mol. Ther. Nucleic Acids 2019, 18, 66–79. [Google Scholar] [CrossRef]
- Oncul, S.; Amero, P.; Rodriguez-Aguayo, C.; Calin, G.A.; Sood, A.K.; Lopez-Berestein, G. Long non-coding RNAs in ovarian cancer: Expression profile and functional spectrum. RNA Biol. 2019, 1–12. [Google Scholar] [CrossRef]
- Moran, V.A.; Perera, R.J.; Khalil, A.M. Emerging functional and mechanistic paradigms of mammalian long non-coding RNAs. Nucleic Acids Res. 2012, 40, 6391–6400. [Google Scholar] [CrossRef]
- Khachane, A.N.; Harrison, P.M. Mining mammalian transcript data for functional long non-coding RNAs. PLoS ONE 2010, 5, e10316. [Google Scholar] [CrossRef]
- Yu, A.D.; Wang, Z.; Morris, K.V. Long noncoding RNAs: A potent source of regulation in immunity and disease. Immunol. Cell Biol. 2015, 93, 277–283. [Google Scholar] [CrossRef]
- Guttman, M.; Rinn, J.L. Modular regulatory principles of large non-coding RNAs. Nature 2012, 482, 339–346. [Google Scholar] [CrossRef]
- Rinn, J.L.; Chang, H.Y. Genome regulation by long noncoding RNAs. Annu. Rev. Biochem. 2012, 81, 145–166. [Google Scholar] [CrossRef]
- Gudenas, B.L.; Wang, L. Prediction of LncRNA Subcellular Localization with Deep Learning from Sequence Features. Sci. Rep. 2018, 8, 16385. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.M.; Guttman, M.; Huarte, M.; Garber, M.; Raj, A.; Morales, D.R.; Thomas, K.; Presser, A.; Bernstein, B.E.; Van Oudenaarden, A. Many human large intergenic noncoding RNAs associate with chromatin-modifying complexes and affect gene expression. Proc. Natl. Acad. Sci. USA 2009, 106, 11667–11672. [Google Scholar] [CrossRef] [PubMed]
- Carlevaro-Fita, J.; Johnson, R. Global positioning system: Understanding long noncoding RNAs through subcellular localization. Mol. Cell 2019, 73, 869–883. [Google Scholar] [CrossRef] [PubMed]
- Bouvrette, L.P.B.; Cody, N.A.; Bergalet, J.; Lefebvre, F.A.; Diot, C.; Wang, X.; Blanchette, M.; Lécuyer, E. CeFra-seq reveals broad asymmetric mRNA and noncoding RNA distribution profiles in Drosophila and human cells. RNA 2018, 24, 98–113. [Google Scholar] [CrossRef] [PubMed]
- Cabili, M.N.; Dunagin, M.C.; McClanahan, P.D.; Biaesch, A.; Padovan-Merhar, O.; Regev, A.; Rinn, J.L.; Raj, A. Localization and abundance analysis of human lncRNAs at single-cell and single-molecule resolution. Genome Biol. 2015, 16, 20. [Google Scholar] [CrossRef]
- Dong, Y.; Yoshitomi, T.; Hu, J.-F.; Cui, J. Long noncoding RNAs coordinate functions between mitochondria and the nucleus. Epigenet. Chromatin. 2017, 10, 41. [Google Scholar] [CrossRef]
- Zeng, C.; Fukunaga, T.; Hamada, M. Identification and analysis of ribosome-associated lncRNAs using ribosome profiling data. BMC Genom. 2018, 19, 414. [Google Scholar] [CrossRef]
- Lin, A.; Hu, Q.; Li, C.; Xing, Z.; Ma, G.; Wang, C.; Li, J.; Ye, Y.; Yao, J.; Liang, K. The LINK-A lncRNA interacts with PtdIns (3, 4, 5) P 3 to hyperactivate AKT and confer resistance to AKT inhibitors. Nat. Cell Biol. 2017, 19, 238–251. [Google Scholar] [CrossRef]
- Gezer, U.; Özgür, E.; Cetinkaya, M.; Isin, M.; Dalay, N. Long non-coding RNAs with low expression levels in cells are enriched in secreted exosomes. Cell Biol. Int. 2014, 38, 1076–1079. [Google Scholar] [CrossRef]
- Zhu, S.; Li, W.; Liu, J.; Chen, C.-H.; Liao, Q.; Xu, P.; Xu, H.; Xiao, T.; Cao, Z.; Peng, J. Genome-scale deletion screening of human long non-coding RNAs using a paired-guide RNA CRISPR–Cas9 library. Nat. Biotechnol. 2016, 34, 1279. [Google Scholar] [CrossRef]
- Li, D.; Zhang, J.; Wang, M.; Li, X.; Gong, H.; Tang, H.; Chen, L.; Wan, L.; Liu, Q. Activity dependent LoNA regulates translation by coordinating rRNA transcription and methylation. Nat. Commun. 2018, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.C.; Chang, H.Y. Molecular mechanisms of long noncoding RNAs. Mol. Cell 2011, 43, 904–914. [Google Scholar] [CrossRef] [PubMed]
- Balas, M.M.; Johnson, A.M. Exploring the mechanisms behind long noncoding RNAs and cancer. Noncoding RNA Res. 2018, 3, 108–117. [Google Scholar] [CrossRef]
- Gupta, R.A.; Shah, N.; Wang, K.C.; Kim, J.; Horlings, H.M.; Wong, D.J.; Tsai, M.-C.; Hung, T.; Argani, P.; Rinn, J.L. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature 2010, 464, 1071–1076. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Fullwood, M.J. Roles, Functions, and Mechanisms of Long Non-coding RNAs in Cancer. Genom. Proteom. Bioinf. 2016, 14, 42–54. [Google Scholar] [CrossRef]
- Yamamura, S.; Imai-Sumida, M.; Tanaka, Y.; Dahiya, R. Interaction and cross-talk between non-coding RNAs. Cell. Mol. Life Sci. 2018, 75, 467–484. [Google Scholar] [CrossRef]
- Thomson, D.W.; Dinger, M.E. Endogenous microRNA sponges: Evidence and controversy. Nat. Rev. Genet. 2016, 17, 272. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, W.; Zhu, W.; Dong, J.; Cheng, Y.; Yin, Z.; Shen, F. Mechanisms and Functions of Long Non-Coding RNAs at Multiple Regulatory Levels. Int. J. Mol. Sci. 2019, 20, 5573. [Google Scholar] [CrossRef]
- Prestayko, A.W.; D’aoust, J.; Issell, B.; Crooke, S. Cisplatin (cis-diamminedichloroplatinum II). Cancer Treat. Rev. 1979, 6, 17–39. [Google Scholar] [CrossRef]
- Galluzzi, L.; Senovilla, L.; Vitale, I.; Michels, J.; Martins, I.; Kepp, O.; Castedo, M.; Kroemer, G. Molecular mechanisms of cisplatin resistance. Oncogene 2012, 31, 1869–1883. [Google Scholar] [CrossRef]
- Hu, Y.; Zhu, Q.-N.; Deng, J.-L.; Li, Z.-X.; Wang, G.; Zhu, Y.-S. Emerging role of long non-coding RNAs in cisplatin resistance. OncoTargets Ther. 2018, 11, 3185. [Google Scholar] [CrossRef] [PubMed]
- Mylavarapu, S.; Das, A.; Roy, M. Role of BRCA mutations in the modulation of response to platinum therapy. Front. Oncol. 2018, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Fink, D.; Nebel, S.; Aebi, S.; Zheng, H.; Cenni, B.; Nehmé, A.; Christen, R.D.; Howell, S.B. The role of DNA mismatch repair in platinum drug resistance. Cancer Res. 1996, 56, 4881–4886. [Google Scholar] [PubMed]
- Asada, N.; Tsuchiya, H.; Tomita, K. De novo deletions of p53 gene and wild-type p53 correlate with acquired cisplatin-resistance in human osteosarcoma OST cell line. Anticancer Res. 1999, 19, 5131–5137. [Google Scholar]
- Leong, C.-O.; Vidnovic, N.; DeYoung, M.P.; Sgroi, D.; Ellisen, L.W. The p63/p73 network mediates chemosensitivity to cisplatin in a biologically defined subset of primary breast cancers. J. Clin. Investig. 2007, 117, 1370–1380. [Google Scholar] [CrossRef]
- Liu, R.; Zeng, Y.; Zhou, C.-F.; Wang, Y.; Li, X.; Liu, Z.-Q.; Chen, X.-P.; Zhang, W.; Zhou, H.-H. Long noncoding RNA expression signature to predict platinum-based chemotherapeutic sensitivity of ovarian cancer patients. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Wang, A.; Zhang, Y.; Xu, X.; Zhang, X. Knockdown of MALAT1 enhances chemosensitivity of ovarian cancer cells to cisplatin through inhibiting the Notch1 signaling pathway. Exp. Cell Res. 2018, 366, 161–171. [Google Scholar] [CrossRef]
- Long, X.; Song, K.; Hu, H.; Tian, Q.; Wang, W.; Dong, Q.; Yin, X.; Di, W. Long non-coding RNA GAS5 inhibits DDP-resistance and tumor progression of epithelial ovarian cancer via GAS5-E2F4-PARP1-MAPK axis. J. Exp. Clin. 2019, 38, 345. [Google Scholar] [CrossRef]
- Jiang, P.; Wu, X.; Wang, X.; Huang, W.; Feng, Q. NEAT1 upregulates EGCG-induced CTR1 to enhance cisplatin sensitivity in lung cancer cells. Oncotarget 2016, 7, 43337–43351. [Google Scholar] [CrossRef]
- Zheng, Z.-G.; Xu, H.; Suo, S.-S.; Xu, X.-L.; Ni, M.-W.; Gu, L.-H.; Chen, W.; Wang, L.-Y.; Zhao, Y.; Tian, B. The essential role of H19 contributing to cisplatin resistance by regulating glutathione metabolism in high-grade serous ovarian cancer. Sci. Rep. 2016, 6, 26093. [Google Scholar] [CrossRef]
- Wu, Y.; Zhou, Y.; He, J.; Sun, H.; Jin, Z. Long non-coding RNA H19 mediates ovarian cancer cell cisplatin-resistance and migration during EMT. Int. J. Clin. Exp. Pathol. 2019, 12, 2506. [Google Scholar] [PubMed]
- Yu, Y.; Zhang, X.; Tian, H.; Zhang, Z.; Tian, Y. Knockdown of long non-coding RNA HOTAIR increases cisplatin sensitivity in ovarian cancer by inhibiting cisplatin-induced autophagy. J. Buon 2018, 23, 1396–1401. [Google Scholar] [PubMed]
- Sun, M.Y.; Zhu, J.Y.; Zhang, C.Y.; Zhang, M.; Song, Y.N.; Rahman, K.; Zhang, L.J.; Zhang, H. Autophagy regulated by lncRNA HOTAIR contributes to the cisplatin-induced resistance in endometrial cancer cells. Biotechnol. Lett. 2017, 39, 1477–1484. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ai, H.; Fan, X.; Chen, S.; Wang, Y.; Liu, L. Knockdown of long non-coding RNA HOTAIR reverses cisplatin resistance of ovarian cancer cells through inhibiting miR-138-5p-regulated EZH2 and SIRT1. Biol. Res. 2020, 53, 1–10. [Google Scholar] [CrossRef]
- Li, J.; Yang, S.; Su, N.; Wang, Y.; Yu, J.; Qiu, H.; He, X. Overexpression of long non-coding RNA HOTAIR leads to chemoresistance by activating the Wnt/β-catenin pathway in human ovarian cancer. Tumor Biol. 2016, 37, 2057–2065. [Google Scholar] [CrossRef]
- Özeş, A.R.; Miller, D.F.; Özeş, O.N.; Fang, F.; Liu, Y.; Matei, D.; Huang, T.; Nephew, K.P. NF-κB-HOTAIR axis links DNA damage response, chemoresistance and cellular senescence in ovarian cancer. Oncogene 2016, 35, 5350–5361. [Google Scholar] [CrossRef]
- Teschendorff, A.E.; Lee, S.H.; Jones, A.; Fiegl, H.; Kalwa, M.; Wagner, W.; Chindera, K.; Evans, I.; Dubeau, L.; Orjalo, A.; et al. HOTAIR and its surrogate DNA methylation signature indicate carboplatin resistance in ovarian cancer. Genome Med. 2015, 7, 108. [Google Scholar] [CrossRef]
- Wang, H.; Fang, L.; Jiang, J.; Kuang, Y.; Wang, B.; Shang, X.; Han, P.; Li, Y.; Liu, M.; Zhang, Z. The cisplatin-induced lncRNA PANDAR dictates the chemoresistance of ovarian cancer via regulating SFRS2-mediated p53 phosphorylation. Cell Death Dis. 2018, 9, 1–15. [Google Scholar] [CrossRef]
- Zhao, L.; Yang, J.; Liu, X.; Fu, X.; Ding, Y.; Wang, C. Overexpression of Long Noncoding RNA E2F-Mediated Cell Proliferation Enhancing Long Noncoding RNA Is Involved in the Development of Chemoresistance of Cancer Cells to Carboplatin in Ovarian Endometrioid Adenocarcinoma. Cancer Biother. Radiopharm. 2019, 34, 566–571. [Google Scholar] [CrossRef]
- Liu, E.; Liu, Z.; Zhou, Y. Carboplatin-docetaxel-induced activity against ovarian cancer is dependent on up-regulated lncRNA PVT1. Int. J. Clin. Exp. Pathol. 2015, 8, 3803. [Google Scholar]
- Wang, D.-Y.; Li, N.; Cui, Y.-L. Long Non-coding RNA CCAT1 Sponges miR-454 to Promote Chemoresistance of Ovarian Cancer Cells to Cisplatin by Regulation of Surviving. J. Korean Cancer Assoc. 2020, 52, 798–814. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Li, H. Knockdown of long non-coding RNA LINC00152 increases cisplatin sensitivity in ovarian cancer cells. Exp. Ther. Med. 2019, 18, 4510–4516. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Niu, H.; Qin, Q.; Yang, S.; Wang, Q.; Yu, C.; Wei, Z.; Jin, Z.; Wang, X.; Yang, A. lncRNA UCA1 mediates resistance to cisplatin by regulating the miR-143/FOSL2-signaling pathway in ovarian cancer. Mol. Ther. Nucleic Acids 2019, 17, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.-F.; Wu, J.; Luo, J.-H.; Li, K.-S.; Wang, F.; Huang, W.; Wu, Y.; Gao, S.-P.; Zhang, X.-M.; Zhang, P.-N. SNHG22 overexpression indicates poor prognosis and induces chemotherapy resistance via the miR-2467/Gal-1 signaling pathway in epithelial ovarian carcinoma. Aging 2019, 11, 8204. [Google Scholar] [CrossRef]
- Guo, J.; Pan, H. Long Noncoding RNA LINC01125 Enhances Cisplatin Sensitivity of Ovarian Cancer via miR-1972. Med Sci. Monit. Int. Med. J. Exp. Clin. Res. 2019, 25, 9844. [Google Scholar] [CrossRef]
- Yan, H.; Xia, J.; Feng, F. Long non-coding RNA ENST00000457645 reverses cisplatin resistance in CP70 ovarian cancer cells. Genet. Mol. Res. 2017, 16, gmr16019411. [Google Scholar] [CrossRef]
- Wu, D.I.; Wang, T.; Ren, C.; Liu, L.; Kong, D.; Jin, X.; Li, X.; Zhang, G. Downregulation of BC200 in ovarian cancer contributes to cancer cell proliferation and chemoresistance to carboplatin. Oncol. Lett. 2016, 11, 1189–1194. [Google Scholar] [CrossRef]
- Shen, F.; Feng, L.; Zhou, J.; Zhang, H.; Xu, Y.; Jiang, R.; Zhang, H.; Chen, Y. Overexpression of CASC11 in ovarian squamous cell carcinoma mediates the development of cancer cell resistance to chemotherapy. Gene 2019, 710, 363–366. [Google Scholar] [CrossRef]
- Qu, C.; Dai, C.; Guo, Y.; Qin, R.; Liu, J. Long noncoding RNA SNHG15 serves as an oncogene and predicts poor prognosis in epithelial ovarian cancer. OncoTargets Ther. 2019, 12, 101. [Google Scholar] [CrossRef]
- Vera, O.; Rodriguez-Antolin, C.; De Castro, J.; Karreth, F.A.; Sellers, T.A.; Ibanez de Caceres, I. An epigenomic approach to identifying differential overlapping and cis-acting lncRNAs in cisplatin-resistant cancer cells. Epigenetics 2018, 13, 251–263. [Google Scholar] [CrossRef]
- Ishida, S.; Lee, J.; Thiele, D.J.; Herskowitz, I. Uptake of the anticancer drug cisplatin mediated by the copper transporter Ctr1 in yeast and mammals. Proc. Natl. Acad. Sci. USA 2002, 99, 14298–14302. [Google Scholar] [CrossRef] [PubMed]
- Katano, K.; Kondo, A.; Safaei, R.; Holzer, A.; Samimi, G.; Mishima, M.; Kuo, Y.-M.; Rochdi, M.; Howell, S.B. Acquisition of resistance to cisplatin is accompanied by changes in the cellular pharmacology of copper. Cancer Res. 2002, 62, 6559–6565. [Google Scholar] [PubMed]
- Askarian-Amiri, M.E.; Crawford, J.; French, J.D.; Smart, C.E.; Smith, M.A.; Clark, M.B.; Ru, K.; Mercer, T.R.; Thompson, E.R.; Lakhani, S.R. SNORD-host RNA Zfas1 is a regulator of mammary development and a potential marker for breast cancer. RNA 2011, 17, 878–891. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.-H.; Kuo, C.; Kuo, M.T.; Chen, H.H. Modulating chemosensitivity of tumors to platinum-based antitumor drugs by transcriptional regulation of copper homeostasis. Int. J. Mol. Sci. 2018, 19, 1486. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, B.A.; Kelly, C.M.; Kim, J.; Hornsby, S.M.; Azizkhan-Clifford, J. Phosphorylation of Sp1 in response to DNA damage by ataxia telangiectasia-mutated kinase. Mol. Cancer Res. 2007, 5, 1319–1330. [Google Scholar] [CrossRef]
- Ji, P.; Diederichs, S.; Wang, W.; Böing, S.; Metzger, R.; Schneider, P.M.; Tidow, N.; Brandt, B.; Buerger, H.; Bulk, E. MALAT-1, a novel noncoding RNA, and thymosin β 4 predict metastasis and survival in early-stage non-small cell lung cancer. Oncogene 2003, 22, 8031–8041. [Google Scholar] [CrossRef]
- Shi, X.; Sun, M.; Liu, H.; Yao, Y.; Song, Y. Long non-coding RNAs: A new frontier in the study of human diseases. Cancer Lett. 2013, 339, 159–166. [Google Scholar] [CrossRef]
- Siddik, Z.H. Cisplatin: Mode of cytotoxic action and molecular basis of resistance. Oncogene 2003, 22, 7265–7279. [Google Scholar] [CrossRef]
- Dabrowiak, J.C.; Goodisman, J.; Souid, A.-K. Kinetic study of the reaction of cisplatin with thiols. Drug Metab. Dispos. 2002, 30, 1378–1384. [Google Scholar] [CrossRef] [PubMed]
- Jamali, B.; Nakhjavani, M.; Hosseinzadeh, L.; Amidi, S.; Nikounezhad, N.; Shirazi, F.H. Intracellular GSH alterations and its relationship to level of resistance following exposure to cisplatin in cancer cells. Iran. J. Pharm. Sci. 2015, 14, 513. [Google Scholar]
- Cao, T.; Jiang, Y.; Wang, Z.; Zhang, N.; Al-Hendy, A.; Mamillapalli, R.; Kallen, A.N.; Kodaman, P.; Taylor, H.S.; Li, D. H19 lncRNA identified as a master regulator of genes that drive uterine leiomyomas. Oncogene 2019, 38, 5356–5366. [Google Scholar] [CrossRef]
- Wang, Q.; Cheng, N.; Li, X.; Pan, H.; Li, C.; Ren, S.; Su, C.; Cai, W.; Zhao, C.; Zhang, L. Correlation of long non-coding RNA H19 expression with cisplatin-resistance and clinical outcome in lung adenocarcinoma. Oncotarget 2017, 8, 2558. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Gan, Y.; Peng, D.; Jiang, X.; Kitazawa, R.; Xiang, Y.; Dai, Y.; Tang, Y.; Yang, J. Long non-coding RNA H19 promotes TDRG1 expression and cisplatin resistance by sequestering miRNA-106b-5p in seminoma. Cancer Med. 2018, 7, 6247–6257. [Google Scholar] [CrossRef] [PubMed]
- Meijer, C.; Mulder, N.H.; Timmer-Bosscha, H.; Sluiter, W.J.; Meersma, G.J.; De Vries, E.G. Relationship of cellular glutathione to the cytotoxicity and resistance of seven platinum compounds. Cancer Res. 1992, 52, 6885–6889. [Google Scholar]
- Klionsky, D.J. Autophagy: From phenomenology to molecular understanding in less than a decade. Nat. Rev. Mol. Cell Biol. 2007, 8, 931–937. [Google Scholar] [CrossRef]
- Ren, J.-H.; He, W.-S.; Nong, L.; Zhu, Q.-Y.; Hu, K.; Zhang, R.-G.; Huang, L.-L.; Zhu, F.; Wu, G. Acquired cisplatin resistance in human lung adenocarcinoma cells is associated with enhanced autophagy. Cancer Biother. Radiopharm. 2010, 25, 75–80. [Google Scholar] [CrossRef]
- Hu, Z.; Cai, M.; Zhang, Y.; Tao, L.; Guo, R. miR-29c-3p inhibits autophagy and cisplatin resistance in ovarian cancer by regulating FOXP1/ATG14 pathway. Cell Cycle 2020, 19, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Satoo, K.; Suzuki, H.; Fujioka, Y.; Ohsumi, Y.; Inagaki, F.; Noda, N.N. Atg7 Activates an Autophagy-Essential Ubiquitin-like Protein Atg8 through Multi-Step Recognition. J. Mol. Biol. 2018, 430, 249–257. [Google Scholar] [CrossRef]
- Brown, C.J.; Ballabio, A.; Rupert, J.L.; Lafreniere, R.G.; Grompe, M.; Tonlorenzi, R.; Willard, H.F. A gene from the region of the human X inactivation centre is expressed exclusively from the inactive X chromosome. Nature 1991, 349, 38–44. [Google Scholar] [CrossRef]
- Sun, W.; Zu, Y.; Fu, X.; Deng, Y. Knockdown of lncRNA-XIST enhances the chemosensitivity of NSCLC cells via suppression of autophagy. Oncol. Rep. 2017, 38, 3347–3354. [Google Scholar] [CrossRef]
- Dongre, A.; Weinberg, R.A. New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 2019, 20, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Settleman, J. EMT, cancer stem cells and drug resistance: An emerging axis of evil in the war on cancer. Oncogene 2010, 29, 4741–4751. [Google Scholar] [CrossRef]
- Cano, A.; Pérez-Moreno, M.A.; Rodrigo, I.; Locascio, A.; Blanco, M.J.; Del Barrio, M.G.; Portillo, F.; Nieto, M.A. The transcription factor snail controls epithelial–mesenchymal transitions by repressing E-cadherin expression. Nat. Cell Biol. 2000, 2, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Herranz, N.; Pasini, D.; Díaz, V.M.; Francí, C.; Gutierrez, A.; Dave, N.; Escrivà, M.; Hernandez-Muñoz, I.; Di Croce, L.; Helin, K. Polycomb complex 2 is required for E-cadherin repression by the Snail1 transcription factor. Mol. Cell. Biol. 2008, 28, 4772–4781. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Shi, X.; Peng, Y.; Wu, M.; Zhang, P.; Xie, R.; Wu, Y.; Yan, Q.; Liu, S.; Wang, J. HIF-1α Promotes Epithelial-Mesenchymal Transition and Metastasis through Direct Regulation of ZEB1 in Colorectal Cancer. PLoS ONE 2015, 10, e0129603. [Google Scholar] [CrossRef]
- Jackson, S.P.; Bartek, J. The DNA-damage response in human biology and disease. Nature 2009, 461, 1071–1078. [Google Scholar] [CrossRef]
- Janssens, S.; Tinel, A.; Lippens, S.; Tschopp, J. PIDD mediates NF-κB activation in response to DNA damage. Cell 2005, 123, 1079–1092. [Google Scholar] [CrossRef]
- Righetti, S.C.; Della Torre, G.; Pilotti, S.; Ménard, S.; Ottone, F.; Colnaghi, M.I.; Pierotti, M.A.; Lavarino, C.; Cornarotti, M.; Oriana, S. A comparative study of p53 gene mutations, protein accumulation, and response to cisplatin-based chemotherapy in advanced ovarian carcinoma. Cancer Res. 1996, 56, 689–693. [Google Scholar]
- Schneider, C.; King, R.M.; Philipson, L. Genes specifically expressed at growth arrest of mammalian cells. Cell 1988, 54, 787–793. [Google Scholar] [CrossRef]
- Dong, S.; Qu, X.; Li, W.; Zhong, X.; Li, P.; Yang, S.; Chen, X.; Shao, M.; Zhang, L. The long non-coding RNA, GAS5, enhances gefitinib-induced cell death in innate EGFR tyrosine kinase inhibitor-resistant lung adenocarcinoma cells with wide-type EGFR via downregulation of the IGF-1R expression. J. Hematol. Oncol. 2015, 8, 43. [Google Scholar] [CrossRef]
- Yang, W.; Hong, L.; Xu, X.; Wang, Q.; Huang, J.; Jiang, L. LncRNA GAS5 suppresses the tumorigenesis of cervical cancer by downregulating miR-196a and miR-205. Tumor Biol. 2017, 39. [Google Scholar] [CrossRef] [PubMed]
- Tao, R.; Hu, S.; Wang, S.; Zhou, X.; Zhang, Q.; Wang, C.; Zhao, X.; Zhou, W.; Zhang, S.; Li, C. Association between indel polymorphism in the promoter region of lncRNA GAS5 and the risk of hepatocellular carcinoma. Carcinogenesis 2015, 36, 1136–1143. [Google Scholar] [CrossRef]
- Pickard, M.R.; Williams, G.T. Regulation of apoptosis by long non-coding RNA GAS5 in breast cancer cells: Implications for chemotherapy. Breast Cancer Res. Tr. 2014, 145, 359–370. [Google Scholar] [CrossRef]
- Jain, P.G.; Patel, B.D. Medicinal chemistry approaches of poly ADP-Ribose polymerase 1 (PARP1) inhibitors as anticancer agents-A recent update. Eur. J. Med. Chem. 2019, 165, 198–215. [Google Scholar] [CrossRef]
- Fulda, S.; Debatin, K.-M. Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene 2006, 25, 4798–4811. [Google Scholar] [CrossRef]
- Youle, R.J.; Strasser, A. The BCL-2 protein family: Opposing activities that mediate cell death. Nat. Rev. Mol. Cell Biol. 2008, 9, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Hung, T.; Wang, Y.; Lin, M.F.; Koegel, A.K.; Kotake, Y.; Grant, G.D.; Horlings, H.M.; Shah, N.; Umbricht, C.; Wang, P. Extensive and coordinated transcription of noncoding RNAs within cell-cycle promoters. Nat. Genet. 2011, 43, 621. [Google Scholar] [CrossRef] [PubMed]
- Kędzierska, H.; Piekiełko-Witkowska, A. Splicing factors of SR and hnRNP families as regulators of apoptosis in cancer. Cancer Lett. 2017, 396, 53–65. [Google Scholar] [CrossRef]
- Xiao, R.; Sun, Y.; Ding, J.-H.; Lin, S.; Rose, D.W.; Rosenfeld, M.G.; Fu, X.-D.; Li, X. Splicing regulator SC35 is essential for genomic stability and cell proliferation during mammalian organogenesis. Mol. Cell. Biol. 2007, 27, 5393–5402. [Google Scholar] [CrossRef]
- Park, S.M.; Choi, E.Y.; Bae, D.H.; Sohn, H.A.; Kim, S.Y.; Kim, Y.J. The LncRNA EPEL Promotes Lung Cancer Cell Proliferation Through E2F Target Activation. Cell. Physiol. Biochem. 2018, 45, 1270–1283. [Google Scholar] [CrossRef]
- Bao, W.; Fu, H.-J.; Jia, L.-T.; Zhang, Y.; Li, W.; Jin, B.-Q.; Yao, L.-B.; Chen, S.-Y.; Yang, A.-G. HER2-mediated upregulation of MMP-1 is involved in gastric cancer cell invasion. Arch. Biochem. Biophys. 2010, 499, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Würtz, S.Ø.; Møller, S.; Mouridsen, H.; Hertel, P.B.; Friis, E.; Brünner, N. Plasma and serum levels of tissue inhibitor of metalloproteinases-1 are associated with prognosis in node-negative breast cancer: A prospective study. Mol. Cell. Proteom. 2008, 7, 424–430. [Google Scholar] [CrossRef]
- Nissan, A.; Stojadinovic, A.; Mitrani-Rosenbaum, S.; Halle, D.; Grinbaum, R.; Roistacher, M.; Bochem, A.; Dayanc, B.E.; Ritter, G.; Gomceli, I. Colon cancer associated transcript-1: A novel RNA expressed in malignant and pre-malignant human tissues. Int. J. Cancer. 2012, 130, 1598–1606. [Google Scholar] [CrossRef]
- Deng, L.; Yang, S.-B.; Xu, F.-F.; Zhang, J.-H. Long noncoding RNA CCAT1 promotes hepatocellular carcinoma progression by functioning as let-7 sponge. J. Exp. Clin. 2015, 34, 18. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Chu, B.; Zhang, Y.; Weng, M.; Qin, Y.; Gong, W.; Quan, Z. Long non-coding RNA CCAT1 promotes gallbladder cancer development via negative modulation of miRNA-218-5p. Cell Death Dis. 2015, 6, e1583. [Google Scholar] [CrossRef] [PubMed]
- Liu, E.; Liu, Z.; Zhou, Y.; Mi, R.; Wang, D. Overexpression of long non-coding RNA PVT1 in ovarian cancer cells promotes cisplatin resistance by regulating apoptotic pathways. Int. J. Clin. Exp. Med. 2015, 8, 20565–20572. [Google Scholar]
- Xiao, J.-N.; Yan, T.-H.; Yu, R.-M.; Gao, Y.; Zeng, W.-L.; Lu, S.-W.; Que, H.-X.; Liu, Z.-P.; Jiang, J.-H. Long non-coding RNA UCA1 regulates the expression of Snail2 by miR-203 to promote hepatocellular carcinoma progression. J. Cancer Res. Clin. 2017, 143, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Bian, Z.; Jin, L.; Zhang, J.; Yin, Y.; Quan, C.; Hu, Y.; Feng, Y.; Liu, H.; Fei, B.; Mao, Y. LncRNA—UCA1 enhances cell proliferation and 5-fluorouracil resistance in colorectal cancer by inhibiting miR-204-5p. Sci. Rep. 2016, 6, 23892. [Google Scholar] [CrossRef]
- Zhou, Y.; Meng, X.; Chen, S.; Li, W.; Li, D.; Singer, R.; Gu, W. IMP1 regulates UCA1-mediated cell invasion through facilitating UCA1 decay and decreasing the sponge effect of UCA1 for miR-122-5p. Breast Cancer Res. 2018, 20, 32. [Google Scholar] [CrossRef]
- Zhang, L.; Cao, X.; Zhang, L.; Zhang, X.; Sheng, H.; Tao, K. UCA1 overexpression predicts clinical outcome of patients with ovarian cancer receiving adjuvant chemotherapy. Cancer Chemoth. Pharm. 2016, 77, 629–634. [Google Scholar] [CrossRef]
- Wang, X.-S.; Zhang, Z.; Wang, H.-C.; Cai, J.-L.; Xu, Q.-W.; Li, M.-Q.; Chen, Y.-C.; Qian, X.-P.; Lu, T.-J.; Yu, L.-Z. Rapid identification of UCA1 as a very sensitive and specific unique marker for human bladder carcinoma. Clin. Cancer Res. 2006, 12, 4851–4858. [Google Scholar] [CrossRef] [PubMed]
- Tulchinsky, E. Fos family members: Regulation, structure and role in oncogenic transformation. Histol. Histopathol. 2000, 15, 921–928. [Google Scholar]
- Li, S.; Fang, X.-D.; Wang, X.-Y.; Fei, B.-Y. Fos-like antigen 2 (FOSL2) promotes metastasis in colon cancer. Exp. Cell Res. 2018, 373, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Hu, W.; Fu, W.; Dai, L.; Jiang, Z.; Zhong, S.; Deng, B.; Zhao, J. HGF/MET Regulated Epithelial-Mesenchymal Transitions and Metastasis by FOSL2 in Non-Small Cell Lung Cancer. OncoTargets Ther. 2019, 12, 9227. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Qi, S.; Xie, C.; Li, C.; Wang, P.; Liu, D. Upregulation of long non-coding RNA XIST has anticancer effects on epithelial ovarian cancer cells through inverse downregulation of hsa-miR-214-3p. Gynecol. Oncol. 2018, 29, e99. [Google Scholar] [CrossRef]
- Samson, J.; Cronin, S.; Dean, K. BC200 (BCYRN1)–The shortest, long, non-coding RNA associated with cancer. Noncoding RNA Res. 2018, 3, 131–143. [Google Scholar] [CrossRef]
- Rao, S.; Orr, G.A.; Chaudhary, A.G.; Kingston, D.G.; Horwitz, S.B. Characterization of the Taxol Binding Site on the Microtubule. 2-(m-Azidobenzoyl)taxol Photolabels a Peptide (Amino Acids 217-231) of Beta-Tubulin. J. Biol. Chem. 1995, 270, 20235–20238. [Google Scholar] [CrossRef]
- Rowinsky, E.K.; Donehower, R.C. Paclitaxel (taxol). N. Engl. J. Med. 1995, 332, 1004–1014. [Google Scholar] [CrossRef]
- Ranganathan, S.; Benetatos, C.; Colarusso, P.; Dexter, D.; Hudes, G. Altered β-tubulin isotype expression in paclitaxel-resistant human prostate carcinoma cells. Br. J. Cancer 1998, 77, 562–566. [Google Scholar] [CrossRef]
- Rao, R.; Markovic, S.N.; Anderson, P. Aerosol therapy for malignancy involving the lungs. Curr. Cancer Drug Targets 2003, 3, 239–250. [Google Scholar] [CrossRef]
- Wang, J.; Ye, C.; Liu, J.; Hu, Y. UCA1 confers paclitaxel resistance to ovarian cancer through miR-129/ABCB1 axis. Biochem. Biophys. Res. Commun. 2018, 501, 1034–1040. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, X.; Dang, Y.; Zhu, X.; Zhang, Y.; Cai, B.; Zheng, L. Long non-coding RNA UCA1 promotes the progression of paclitaxel resistance in ovarian cancer by regulating the miR-654-5p/SIK2 axis. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 591–603. [Google Scholar]
- Shi, C.; Wang, M. LINC01118 modulates paclitaxel resistance of epithelial ovarian cancer by regulating miR-134/ABCC1. Med. Sci. Monit. 2018, 24, 8831. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Lv, W.; Zhang, Y. LncRNA NEAT1 contributes to paclitaxel resistance of ovarian cancer cells by regulating ZEB1 expression via miR-194. OncoTargets Ther. 2017, 10, 5377. [Google Scholar] [CrossRef] [PubMed]
- Dong, P.; Xiong, Y.; Yue, J.; Xu, D.; Ihira, K.; Konno, Y.; Kobayashi, N.; Todo, Y.; Watari, H. Long noncoding RNA NEAT1 drives aggressive endometrial cancer progression via miR-361-regulated networks involving STAT3 and tumor microenvironment-related genes. J. Exp. Clin. Cancer Res. 2019, 38, 295. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Shen, L.; Lin, Q.; Dong, C.; Maswela, B.; Illahi, G.S.; Wu, X. SNHG5 enhances Paclitaxel sensitivity of ovarian cancer cells through sponging miR-23a. Biomed. Pharmacother. 2020, 123, 109711. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, A.; Zhang, Z. LncRNA SDHAP1 confers paclitaxel resistance of ovarian cancer by regulating EIF4G2 expression via miR-4465. J. Biochem. 2020, 168. [Google Scholar] [CrossRef]
- Guo, J.-L.; Tang, T.; Li, J.-H.; Yang, Y.-H.; Zhang, L.; Quan, Y. LncRNA HEIH Enhances Paclitaxel-Tolerance of Endometrial Cancer Cells via Activation of MAPK Signaling Pathway. Pathol. Oncol. Res. 2019, 1–10. [Google Scholar] [CrossRef]
- Liu, S.; Zou, B.; Tian, T.; Luo, X.; Mao, B.; Zhang, X.; Lei, H. Overexpression of the lncRNA FER1L4 inhibits paclitaxel tolerance of ovarian cancer cells via the regulation of the MAPK signaling pathway. J. Cell. Biochem. 2019, 120, 7581–7589. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, M.; Shi, C.; Shi, F.; Pei, C. Long non-coding RNA Linc00312 modulates the sensitivity of ovarian cancer to cisplatin via the Bcl-2/Caspase-3 signaling pathway. Biosci. Trends 2018, 12, 309–316. [Google Scholar] [CrossRef]
- Shang, C.; Ao, C.N.; Cheong, C.C.; Meng, L. Long non-coding RNA CDKN2B antisense RNA 1 gene contributes to paclitaxel resistance in endometrial carcinoma. Front. Oncol. 2019, 9, 27. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Liu, S.; Fu, C.; Wang, X.; Zhang, M.; Liu, G.; Dai, C.; Gong, Z.; Xu, H.; Fu, Z. LncRNA KB-1471A8. 2 Overexpression Suppresses Cell Proliferation and Migration and Antagonizes the Paclitaxel Resistance of Ovarian Cancer Cells. Cancer Biother. Radiopharm. 2019, 34, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wu, J.; Fu, C.; Teng, F.; Liu, S.; Dai, C.; Shen, R.; Jia, X. Multidrug resistant lncRNA profile in chemotherapeutic sensitive and resistant ovarian cancer cells. J. Cell. Physiol. 2018, 233, 5034–5043. [Google Scholar] [CrossRef]
- Yu, X.; Li, Z.; Zheng, H.; Chan, M.T.; Wu, W.K.K. NEAT 1: A novel cancer-related long non-coding RNA. Cell Prolif. 2017, 50, e12329. [Google Scholar] [CrossRef] [PubMed]
- McCain, J. The MAPK (ERK) pathway: Investigational combinations for the treatment of BRAF-mutated metastatic melanoma. Pharm. Ther. 2013, 38, 96. [Google Scholar]
- Yang, F.; Xu, D.; Yuan, S.X.; Zhu, N.; Zhou, W.P.; Yang, G.S.; Wang, Y.Z.; Shang, J.L.; Gao, C.f.; Zhang, F.R.; et al. Long noncoding RNA high expression in hepatocellular carcinoma facilitates tumor growth through enhancer of zeste homolog 2 in humans. Hepatology 2011, 54, 1679–1689. [Google Scholar] [CrossRef] [PubMed]
- Ming, M.; He, Y.-Y. PTEN in DNA damage repair. Cancer Lett. 2012, 319, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.X.; Chen, C.P.; Zhang, N.; Wang, T.X. Low-expression of lncRNA FER1L4 might be a prognostic marker in osteosarcoma. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 2310–2314. [Google Scholar]
- Ma, W.; Zhang, C.Q.; Li, H.L.; Gu, J.; Miao, G.Y.; Cai, H.Y.; Wang, J.K.; Zhang, L.J.; Song, Y.M.; Tian, Y.H.; et al. LncRNA FER1L4 suppressed cancer cell growth and invasion in esophageal squamous cell carcinoma. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 2638–2645. [Google Scholar]
- Qiao, Q.; Li, H. LncRNA FER1L4 suppresses cancer cell proliferation and cycle by regulating PTEN expression in endometrial carcinoma. Biochem. Biophys. Res. Commun. 2016, 478, 507–512. [Google Scholar] [CrossRef]
- Wang, X.; Dong, K.; Jin, Q.; Ma, Y.; Yin, S.; Wang, S. Upregulation of lncRNA FER1L4 suppresses the proliferation and migration of the hepatocellular carcinoma via regulating PI3K/AKT signal pathway. J. Cell. Biochem. 2019, 120, 6781–6788. [Google Scholar] [CrossRef]
- Xia, T.; Liao, Q.; Jiang, X.; Shao, Y.; Xiao, B.; Xi, Y.; Guo, J. Long noncoding RNA associated-competing endogenous RNAs in gastric cancer. Sci. Rep. 2014, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Li, T.; Han, X.; Yuan, H. Knockdown of LncRNA ANRIL suppresses cell proliferation, metastasis, and invasion via regulating miR-122-5p expression in hepatocellular carcinoma. J. Cancer Res. Clin. Oncol. 2018, 144, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-J.; Wang, D.-D.; Du, C.-X.; Wang, Y. Long noncoding RNA ANRIL promotes cervical cancer development by acting as a sponge of miR-186. Oncol. Res. 2018, 26, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.-T.; Xu, J.-H.; Zheng, Z.-R.; Zhao, Q.-Q.; Zeng, X.-S.; Cheng, S.-X.; Liang, Y.-H.; Hu, Q.-F. Long non-coding RNA ANRIL promotes carcinogenesis via sponging miR-199a in triple-negative breast cancer. Biomed. Pharmacother. 2017, 96, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Lu, Y.-L.; Yang, Y.; Hu, L.-B.; Bai, Y.; Li, R.-Q.; Zhang, G.-Y.; Li, J.; Bi, C.-W.; Yang, L.-B. Overexpression of lncRNA ANRIL promoted the proliferation and migration of prostate cancer cells via regulating let-7a/TGF-β1/Smad signaling pathway. Cancer Biomark. 2018, 21, 613–620. [Google Scholar] [CrossRef]
- Zhong, L.; Sun, S.; Shi, J.; Cao, F.; Han, X.; Chen, Z. MicroRNA-125a-5p plays a role as a tumor suppressor in lung carcinoma cells by directly targeting STAT3. Tumor Biol. 2017, 39. [Google Scholar] [CrossRef]
- Tong, Z.; Liu, N.; Lin, L.; Guo, X.; Yang, D.; Zhang, Q. miR-125a-5p inhibits cell proliferation and induces apoptosis in colon cancer via targeting BCL2, BCL2L12 and MCL1. Biomed. Pharmacother. 2015, 75, 129–136. [Google Scholar] [CrossRef]
- Pacilli, A.; Ceccarelli, C.; Treré, D.; Montanaro, L. SnoRNA U50 Levels Are Regulated by Cell Proliferation and rRNA Transcription. Int. J. Mol. Sci. 2013, 14, 14923–14935. [Google Scholar] [CrossRef]
- Zhuang, R.-J.; Bai, X.-X.; Liu, W. MicroRNA-23a depletion promotes apoptosis of ovarian cancer stem cell and inhibits cell migration by targeting DLG2. Cancer Biol. Ther. 2019, 20, 897–911. [Google Scholar] [CrossRef]
- Xu, F.; Li, Q.; Wang, Z.; Cao, X. Sinomenine inhibits proliferation, migration, invasion and promotes apoptosis of prostate cancer cells by regulation of miR-23a. Biomed. Pharmacother. 2019, 112, 108592. [Google Scholar] [CrossRef] [PubMed]
- Majem, B.; Parrilla, A.; Jiménez, C.; Suárez-Cabrera, L.; Barber, M.; Marín, A.; Castellví, J.; Tamayo, G.; Moreno-Bueno, G.; Ponce, J. MicroRNA-654-5p suppresses ovarian cancer development impacting on MYC, WNT and AKT pathways. Oncogene 2019, 38, 6035–6050. [Google Scholar] [CrossRef] [PubMed]
- Du, W.-Q.; Zheng, J.-N.; Pei, D.-S. The diverse oncogenic and tumor suppressor roles of salt-inducible kinase (SIK) in cancer. Expert Opin. Ther. Targets 2016, 20, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Miranda, F.; Mannion, D.; Liu, S.; Zheng, Y.; Mangala, L.S.; Redondo, C.; Herrero-Gonzalez, S.; Xu, R.; Taylor, C.; Chedom, D.F. Salt-inducible kinase 2 couples ovarian cancer cell metabolism with survival at the adipocyte-rich metastatic niche. Cancer Cell 2016, 30, 273–289. [Google Scholar] [CrossRef]
- Wang, L.; Hu, Y.; Xiang, X.; Qu, K.; Teng, Y. Identification of long non-coding RNA signature for paclitaxel-resistant patients with advanced ovarian cancer. Oncotarget 2017, 8, 64191. [Google Scholar] [CrossRef]
- Huang, K.-C.; Rao, P.H.; Lau, C.C.; Heard, E.; Ng, S.-K.; Brown, C.; Mok, S.C.; Berkowitz, R.S.; Ng, S.-W. Relationship of XIST Expression and Responses of Ovarian Cancer to Chemotherapy 1. Mol. Cancer Ther. 2002, 1, 769–776. [Google Scholar]
- Arun, G.; Diermeier, S.D.; Spector, D.L. Therapeutic targeting of long non-coding RNAs in cancer. Trends Mol. Med. 2018, 24, 257–277. [Google Scholar] [CrossRef]
- Hannon, G.J.; Rossi, J.J. Unlocking the potential of the human genome with RNA interference. Nature 2004, 431, 371–378. [Google Scholar] [CrossRef]
- Morrissey, D.V.; Lockridge, J.A.; Shaw, L.; Blanchard, K.; Jensen, K.; Breen, W.; Hartsough, K.; Machemer, L.; Radka, S.; Jadhav, V.; et al. Potent and persistent in vivo anti-HBV activity of chemically modified siRNAs. Nat. Biotechnol. 2005, 23, 1002–1007. [Google Scholar] [CrossRef]
- Yoo, B.H.; Bochkareva, E.; Bochkarev, A.; Mou, T.C.; Gray, D.M. 2’-O-methyl-modified phosphorothioate antisense oligonucleotides have reduced non-specific effects in vitro. Nucleic Acids Res. 2004, 32, 2008–2016. [Google Scholar] [CrossRef]
- Crooke, S.T. Molecular Mechanisms of Antisense Oligonucleotides. Nucleic Acid Ther. 2017, 27, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Bennett, C.F.; Baker, B.F.; Pham, N.; Swayze, E.; Geary, R.S. Pharmacology of antisense drugs. Annu. Rev. Pharmacol. Toxicol. 2017, 57, 81–105. [Google Scholar] [CrossRef] [PubMed]
- Pickar-Oliver, A.; Gersbach, C.A. The next generation of CRISPR–Cas technologies and applications. Nat. Rev. Mol. Cell Biol. 2019, 20, 490–507. [Google Scholar] [CrossRef]
- Gilbert, L.A.; Horlbeck, M.A.; Adamson, B.; Villalta, J.E.; Chen, Y.; Whitehead, E.H.; Guimaraes, C.; Panning, B.; Ploegh, H.L.; Bassik, M.C.; et al. Genome-Scale CRISPR-Mediated Control of Gene Repression and Activation. Cell 2014, 159, 647–661. [Google Scholar] [CrossRef] [PubMed]
- Thakore, P.I.; D’ippolito, A.M.; Song, L.; Safi, A.; Shivakumar, N.K.; Kabadi, A.M.; Reddy, T.E.; Crawford, G.E.; Gersbach, C.A. Highly specific epigenome editing by CRISPR-Cas9 repressors for silencing of distal regulatory elements. Nat. Methods 2015, 12, 1143. [Google Scholar] [CrossRef]
- Abudayyeh, O.O.; Gootenberg, J.S.; Essletzbichler, P.; Han, S.; Joung, J.; Belanto, J.J.; Verdine, V.; Cox, D.B.T.; Kellner, M.J.; Regev, A.; et al. RNA targeting with CRISPR-Cas13. Nature 2017, 550, 280–284. [Google Scholar] [CrossRef]
- Wilusz, J.E.; JnBaptiste, C.K.; Lu, L.Y.; Kuhn, C.-D.; Joshua-Tor, L.; Sharp, P.A. A triple helix stabilizes the 3′ ends of long noncoding RNAs that lack poly (A) tails. Genes Dev. 2012, 26, 2392–2407. [Google Scholar] [CrossRef]
- Katayama, S.; Tomaru, Y.; Kasukawa, T.; Waki, K.; Nakanishi, M.; Nakamura, M.; Nishida, H.; Yap, C.; Suzuki, M.; Kawai, J. Antisense transcription in the mammalian transcriptome. Science 2005, 309, 1564–1566. [Google Scholar]
- Modarresi, F.; Faghihi, M.A.; Lopez-Toledano, M.A.; Fatemi, R.P.; Magistri, M.; Brothers, S.P.; Van Der Brug, M.P.; Wahlestedt, C. Inhibition of natural antisense transcripts in vivo results in gene-specific transcriptional upregulation. Nat. Biotechnol. 2012, 30, 453. [Google Scholar] [CrossRef]
- Yap, K.L.; Li, S.; Muñoz-Cabello, A.M.; Raguz, S.; Zeng, L.; Mujtaba, S.; Gil, J.; Walsh, M.J.; Zhou, M.-M. Molecular interplay of the noncoding RNA ANRIL and methylated histone H3 lysine 27 by polycomb CBX7 in transcriptional silencing of INK4a. Mol. Cell 2010, 38, 662–674. [Google Scholar] [CrossRef]
- Dimitrova, N.; Zamudio, J.R.; Jong, R.M.; Soukup, D.; Resnick, R.; Sarma, K.; Ward, A.J.; Raj, A.; Lee, J.T.; Sharp, P.A. LincRNA-p21 activates p21 in cis to promote Polycomb target gene expression and to enforce the G1/S checkpoint. Mol. Cell 2014, 54, 777–790. [Google Scholar] [CrossRef] [PubMed]
- Redis, R.S.; Vela, L.E.; Lu, W.; Ferreira de Oliveira, J.; Ivan, C.; Rodriguez-Aguayo, C.; Adamoski, D.; Pasculli, B.; Taguchi, A.; Chen, Y.; et al. Allele-specific reprogramming of cancer metabolism by the long non-coding RNA ccat2. Mol. Cell 2016, 61, 520–534. [Google Scholar] [CrossRef]
- Pichler, M.; Rodriguez-Aguayo, C.; Nam, S.Y.; Dragomir, M.P.; Bayraktar, R.; Anfossi, S.; Knutsen, E.; Ivan, C.; Fuentes-Mattei, E.; Lee, S.K.; et al. Therapeutic potential of flanc, a novel primate-specific long non-coding RNA in colorectal cancer. Gut 2020. [Google Scholar] [CrossRef] [PubMed]
- Rigoutsos, I.; Lee, S.K.; Nam, S.Y.; Anfossi, S.; Pasculli, B.; Pichler, M.; Jing, Y.; Rodriguez-Aguayo, C.; Telonis, A.G.; Rossi, S.; et al. N-blr, a primate-specific non-coding transcript leads to colorectal cancer invasion and migration. Genome Biol. 2017, 18, 98. [Google Scholar] [CrossRef] [PubMed]
- Salamini-Montemurri, M.; Lamas-Maceiras, M.; Barreiro-Alonso, A.; Vizoso-Vázquez, Á.; Rodríguez-Belmonte, E.; Quindós-Varela, M.; Cerdán, M.E. The challenges and opportunities of lncRNAs in ovarian cancer research and clinical use. Cancers 2020, 12, 1020. [Google Scholar] [CrossRef]
- Pei, C.L.; Fei, K.L.; Yuan, X.Y.; Gong, X.J. LncRNA dancr aggravates the progression of ovarian cancer by downregulating upf1. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 10657–10663. [Google Scholar]
- Yang, M.; Zhai, Z.; Guo, S.; Li, X.; Zhu, Y.; Wang, Y. Long non-coding RNA flj33360 participates in ovarian cancer progression by sponging mir-30b-3p. Onco Targets Ther. 2019, 12, 4469–4480. [Google Scholar] [CrossRef]
- Ning, L.; Hu, Y.C.; Wang, S.; Lang, J.H. Altered long noncoding rnas and survival outcomes in ovarian cancer: A systematic review and meta-analysis (prisma compliant). Medicine 2018, 97, e11481. [Google Scholar] [CrossRef]
- Luo, P.; Liu, X.F.; Wang, Y.C.; Li, N.D.; Liao, S.J.; Yu, M.X.; Liang, C.Z.; Tu, J.C. Prognostic value of abnormally expressed lncRNAs in ovarian carcinoma: A systematic review and meta-analysis. Oncotarget 2017, 8, 23927–23936. [Google Scholar] [CrossRef]
- Wang, X.; Han, L.; Zhou, L.; Wang, L.; Zhang, L.M. Prediction of candidate RNA signatures for recurrent ovarian cancer prognosis by the construction of an integrated competing endogenous RNA network. Oncol. Rep. 2018, 40, 2659–2673. [Google Scholar] [CrossRef]
- Lorenzi, L.; Avila Cobos, F.; Decock, A.; Everaert, C.; Helsmoortel, H.; Lefever, S.; Verboom, K.; Volders, P.J.; Speleman, F.; Vandesompele, J.; et al. Long noncoding RNA expression profiling in cancer: Challenges and opportunities. Genes Chromosomes Cancer 2019, 58, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Venter, J.C.; Adams, M.D.; Myers, E.W.; Li, P.W.; Mural, R.J.; Sutton, G.G.; Smith, H.O.; Yandell, M.; Evans, C.A.; Holt, R.A.; et al. The sequence of the human genome. Science 2001, 291, 1304–1351. [Google Scholar] [CrossRef] [PubMed]
- Everaert, C.; Luypaert, M.; Maag, J.L.V.; Cheng, Q.X.; Dinger, M.E.; Hellemans, J.; Mestdagh, P. Benchmarking of RNA-sequencing analysis workflows using whole-transcriptome rt-qpcr expression data. Sci. Rep. 2017, 7, 1559. [Google Scholar] [CrossRef] [PubMed]
- Assefa, A.T.; De Paepe, K.; Everaert, C.; Mestdagh, P.; Thas, O.; Vandesompele, J. Differential gene expression analysis tools exhibit substandard performance for long non-coding RNA-sequencing data. Genome Biol. 2018, 19, 96. [Google Scholar] [CrossRef]
- Mercer, T.R.; Gerhardt, D.J.; Dinger, M.E.; Crawford, J.; Trapnell, C.; Jeddeloh, J.A.; Mattick, J.S.; Rinn, J.L. Targeted RNA sequencing reveals the deep complexity of the human transcriptome. Nat. Biotechnol. 2011, 30, 99–104. [Google Scholar] [CrossRef]
- Clark, M.B.; Mercer, T.R.; Bussotti, G.; Leonardi, T.; Haynes, K.R.; Crawford, J.; Brunck, M.E.; Cao, K.A.; Thomas, G.P.; Chen, W.Y.; et al. Quantitative gene profiling of long noncoding RNAs with targeted RNA sequencing. Nat. Methods 2015, 12, 339–342. [Google Scholar] [CrossRef]
- Wong, B.C.; Chiu, R.W.; Tsui, N.B.; Chan, K.C.; Chan, L.W.; Lau, T.K.; Leung, T.N.; Lo, Y.M. Circulating placental RNA in maternal plasma is associated with a preponderance of 5’ mRNA fragments: Implications for noninvasive prenatal diagnosis and monitoring. Clin. Chem. 2005, 51, 1786–1795. [Google Scholar] [CrossRef]
- Luo, J.; Xiong, Y.; Fu, P.F.; Li, E.C.; Qu, L.; Fan, X.; Cai, Z.J.; Lin, A.F. Exosomal long non-coding RNAs: Biological properties and therapeutic potential in cancer treatment. J. Zhejiang Univ. Sci. B 2019, 20, 488–495. [Google Scholar] [CrossRef]
- Mohankumar, S.; Patel, T. Extracellular vesicle long noncoding RNA as potential biomarkers of liver cancer. Brief. Funct. Genom. 2016, 15, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Zhang, Y.; Zhang, X.; Yang, Y.; Zheng, X.; Li, X.; Liu, Y.; Zhang, Y. Exosomal long noncoding RNA hottip as potential novel diagnostic and prognostic biomarker test for gastric cancer. Mol. Cancer 2018, 17, 68. [Google Scholar] [CrossRef]
- Li, X.; Corbett, A.L.; Taatizadeh, E.; Tasnim, N.; Little, J.P.; Garnis, C.; Daugaard, M.; Guns, E.; Hoorfar, M.; Li, I.T.S. Challenges and opportunities in exosome research-perspectives from biology, engineering, and cancer therapy. APL Bioeng. 2019, 3, 011503. [Google Scholar] [CrossRef] [PubMed]
- Goudarzi, M.; Berg, K.; Pieper, L.M.; Schier, A.F. Individual long non-coding RNAs have no overt functions in zebrafish embryogenesis, viability and fertility. eLife 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Teng, H.; Yao, F.; Yap, S.; Sun, Y.; Ma, L. Challenges and strategies in ascribing functions to long noncoding RNAs. Cancers 2020, 12, 1458. [Google Scholar] [CrossRef]
- Derrien, T.; Johnson, R.; Bussotti, G.; Tanzer, A.; Djebali, S.; Tilgner, H.; Guernec, G.; Martin, D.; Merkel, A.; Knowles, D.G.; et al. The gencode v7 catalog of human long noncoding RNAs: Analysis of their gene structure, evolution, and expression. Genome Res. 2012, 22, 1775–1789. [Google Scholar] [CrossRef] [PubMed]
- Washietl, S.; Kellis, M.; Garber, M. Evolutionary dynamics and tissue specificity of human long noncoding RNAs in six mammals. Genome Res. 2014, 24, 616–628. [Google Scholar] [CrossRef]
- Bajan, S.; Hutvagner, G. RNA-based therapeutics: From antisense oligonucleotides to miRNAs. Cells 2020, 9, 137. [Google Scholar] [CrossRef]
- Landen, C.N., Jr.; Chavez-Reyes, A.; Bucana, C.; Schmandt, R.; Deavers, M.T.; Lopez-Berestein, G.; Sood, A.K. Therapeutic epha2 gene targeting in vivo using neutral liposomal small interfering RNA delivery. Cancer Res. 2005, 65, 6910–6918. [Google Scholar] [CrossRef]
- Hanna, N.; Ohana, P.; Konikoff, F.M.; Leichtmann, G.; Hubert, A.; Appelbaum, L.; Kopelman, Y.; Czerniak, A.; Hochberg, A. Phase 1/2a, dose-escalation, safety, pharmacokinetic and preliminary efficacy study of intratumoral administration of bc-819 in patients with unresectable pancreatic cancer. Cancer Gene Ther. 2012, 19, 374–381. [Google Scholar] [CrossRef]
- Lavie, O.; Edelman, D.; Levy, T.; Fishman, A.; Hubert, A.; Segev, Y.; Raveh, E.; Gilon, M.; Hochberg, A. A phase 1/2a, dose-escalation, safety, pharmacokinetic, and preliminary efficacy study of intraperitoneal administration of bc-819 (h19-dta) in subjects with recurrent ovarian/peritoneal cancer. Arch. Gynecol. Obstet. 2017, 295, 751–761. [Google Scholar] [CrossRef]

| Name of lncRNA | Classification | Type of Cancer | Role in Platinum Resistance | Mechanism | Reference |
|---|---|---|---|---|---|
| ZFAS1 (Zinc finger antisense 1) | Antisense | Ovarian cancer | ↑ Cisplatin resistance | Regulating CTR1 expression via regulating miR-150-5p/SP1 axis | [46] |
| MALAT1 (metastasis-associated lung adenocarcinoma transcript 1) | Intergenic | Ovarian cancer | ↑ Cisplatin resistance | Regulating Notch1/ABCC1/MRP1 signaling pathway | [47] |
| GAS5 (growth arrest-specific transcript 5) | Antisense | Ovarian cancer | ↓ Cisplatin resistance | ↓ PARP1 and ↓ phosphorylation of ERK, JNK, and MAPK | [48] |
| NEAT1 (nuclear-enriched abundant transcript 1) | Intergenic | Lung cancer | ↓ Cisplatin resistance | Upregulating EGCG-induced CTR1 expression | [49] |
| H19 (imprinted maternally expressed transcript) | Intergenic | Ovarian cancer | ↑ Cisplatin resistance | ↑ Expression of NRF2-targeted proteins and ↑ GSH activity → ↓ cisplatin-induced oxidative cytotoxicity | [50] |
| H19 | Intergenic | Ovarian cancer | ↑ Cisplatin resistance | ↑ TWIST, SLUG, and SNAIL → ↑ EMT | [51] |
| HOTAIR (HOX transcript antisense RNA) | Antisense | Ovarian cancer | ↑ Cisplatin resistance | Activating ATG7 and enhancing autophagy | [52] |
| Endometrial cancer | ↑ Cisplatin resistance | Activating beclin-1 and enhancing autophagy | [53] | ||
| Ovarian cancer | ↑ Cisplatin resistance | Regulating miR-138-5p/EZH2/SIRT1 axis | [54] | ||
| Ovarian cancer | ↑ Cisplatin resistance | Activating Wnt/β-catenin pathway → ↑ EMT | [55] | ||
| Ovarian cancer | ↑ Cisplatin resistance | HOTAIR ↑ NF-κB, IL-6, and CHK1-p53-p21 → repair of damaged DNA | [56] | ||
| Ovarian cancer | ↑ Carboplatin resistance | Unknown | [57] | ||
| PANDAR (promoter of CDKN1A antisense DNA damage activated RNA) | Antisense | Ovarian cancer | ↑ Cisplatin resistance | Modulating PANDAR/SRFS2/p53 axis → ↓ p53-induced apoptosis | [58] |
| EPEL (E2F-mediated proliferation enhancing lncRNA) | Intergenic | Endometroid cancer | ↑ Carboplatin resistance | ↓ Expression of p53 → ↓ apoptosis. | [59] |
| PVT1 (plasmacytoma variant translocation 1) | Intergenic | Ovarian cancer | ↓ Carboplatin and docetaxel resistance | ↑ p53 and TIMP1 → ↑ apoptosis and ↓ tumor invasion, respectively. | [60] |
| CCAT1 (colon cancer-associated transcript 1) | Antisense | Ovarian cancer | ↑ Cisplatin resistance | Modulating miR-454/survivin pathway | [61] |
| LINC00152 (long intergenic non-coding RNA 152) | Intergenic | Ovarian cancer | ↑ Cisplatin resistance | ↑ MDR1 and MRP1 and ↓ apoptosis | [62] |
| UCA1 (urothelial carcinoma associated 1) | Intergenic | Ovarian cancer | ↑ Cisplatin resistance | Regulating miR-143/FOSL2 axis | [63] |
| SNHG22 (small nucleolar RNA host gene 22) | Antisense | Ovarian cancer | ↑ Cisplatin and paclitaxel resistance | Regulating miR-2467/Gal-1 signaling cascade | [64] |
| LINC01125 | Sense-overlapping | Ovarian cancer | ↓ Cisplatin and paclitaxel resistance | Regulating miR-1972/apoptosis pathway | [65] |
| ENST00000457645 | Intergenic | Ovarian cancer | ↓ Cisplatin resistance | Unknown | [66] |
| BCYRN1 (brain cytoplasmic RNA1) | Intergenic | Ovarian cancer | ↓ Carboplatin resistance | Unknown | [67] |
| CASC11 (cancer susceptibility 11) | Intergenic | Ovarian cancer | ↑ Cisplatin and carboplatin resistance | Unknown | [68] |
| SNHG15 (small nucleolar RNA host gene 15) | Intergenic | Ovarian cancer | ↑ Cisplatin resistance | Unknown | [69] |
| CRNDE (colorectal neoplasia differentially expressed) | Intergenic | Ovarian cancer | Upregulated in cisplatin resistance | Unknown | [70] |
| RP11-1A16.1 | Intergenic | Ovarian cancer | Downregulated in cisplatin resistance | Unknown | [70] |
| AC000035.3 | Intergenic | Ovarian cancer | Downregulated in cisplatin resistance | Unknown | [70] |
| AC003986.7 | Antisense | Ovarian cancer | Upregulated in cisplatin resistance | Unknown | [70] |
| RP11-6N17.4 | Bidirectional | Ovarian cancer | Upregulated in cisplatin resistance | Unknown | [70] |
| PLAC2 | Intronic | Ovarian cancer | Upregulated in cisplatin resistance | Unknown | [70] |
| CTD-2026G22.1 | Intergenic | Ovarian cancer | Downregulated in cisplatin resistance | Unknown | [70] |
| RP11-1A16.1 | Intergenic | Ovarian cancer | Downregulated in cisplatin resistance | Unknown | [70] |
| Name of lncRNA | Classification | Type of Cancer | Role in Drug Resistance | Mechanism | Reference |
|---|---|---|---|---|---|
| UCA1 (urothelial carcinoma associated 1) | Intergenic | Ovarian cancer | ↑ PTX resistance | Sponging miR-129 → ↑ ABCB1-induced drug efflux Regulating miR-654-5p/SIK2 axis | [131] [132] |
| LINC01118 | Intergenic | Ovarian cancer | ↑ PTX resistance | Regulating miR-134/ABCC1 axis | [133] |
| NEAT1 (nuclear paraspeckle assembly transcript 1) | Intergenic | Ovarian cancer | ↑ PTX resistance | Sponging miR-194 → ↑ ZEB1 expression → ↑ EMT | [134] |
| NEAT1 (nuclear paraspeckle assembly transcript 1) | Intergenic | Endometrial cancer | ↑ PTX resistance | Sponging miR-194 → ↑ STAT3 expression | [135] |
| SNHG5 (small nucleolar RNA host gene 5) | Intergenic | Ovarian cancer | ↓ PTX resistance | Sponging oncogenic miR-23a | [136] |
| SDHAP1 (succinate dehydrogenase complex, subunit A, flavoprotein pseudogene 1) | Pseudogene | Ovarian cancer | ↑ PTX resistance | Regulating miR-4465/EIF4G2 axis | [137] |
| HEIH (highly expressed in hepatocellular carcinoma) | Intergenic | Endometrial cancer | ↑ PTX resistance | Activating MAPK pathway | [138] |
| FER1L4 (fer-1 like family member 4) | Pseudogene | Ovarian cancer | ↓ PTX resistance | ↓ Phosphorylation of MAPK | [139] |
| LINC00312 (long intergenic non-protein coding RNA 312) | Intergenic | Ovarian cancer | ↓ PTX resistance | Blocking BCL-2 expression and activating Bax/Caspas3 apoptotic pathways | [140]. |
| ANRIL (antisense non-coding RNA in the INK4 locus) | Antisense | Endometrial cancer | ↑ PTX resistance | Regulating both miR-125a-5p/Bcl2 and miR-125a-5p/MRP4 axes | [141] |
| KB-1471A8.2 | Antisense | Ovarian cancer | ↓ PTX resistance | Blocking S-phase entry and CDK4 | [142] |
| CTD-2589M5.4 | Intergenic | Ovarian cancer | Associated with multidrug resistance | Unknown but may be related to ABCB1, ABCB4, ABCC3, and ABCG2 | [143] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elsayed, A.M.; Amero, P.; Salama, S.A.; Abdelaziz, A.H.; Lopez-Berestein, G.; Rodriguez-Aguayo, C. Back to the Future: Rethinking the Great Potential of lncRNAS for Optimizing Chemotherapeutic Response in Ovarian Cancer. Cancers 2020, 12, 2406. https://doi.org/10.3390/cancers12092406
Elsayed AM, Amero P, Salama SA, Abdelaziz AH, Lopez-Berestein G, Rodriguez-Aguayo C. Back to the Future: Rethinking the Great Potential of lncRNAS for Optimizing Chemotherapeutic Response in Ovarian Cancer. Cancers. 2020; 12(9):2406. https://doi.org/10.3390/cancers12092406
Chicago/Turabian StyleElsayed, Abdelrahman M., Paola Amero, Salama A. Salama, Abdelaziz H. Abdelaziz, Gabriel Lopez-Berestein, and Cristian Rodriguez-Aguayo. 2020. "Back to the Future: Rethinking the Great Potential of lncRNAS for Optimizing Chemotherapeutic Response in Ovarian Cancer" Cancers 12, no. 9: 2406. https://doi.org/10.3390/cancers12092406
APA StyleElsayed, A. M., Amero, P., Salama, S. A., Abdelaziz, A. H., Lopez-Berestein, G., & Rodriguez-Aguayo, C. (2020). Back to the Future: Rethinking the Great Potential of lncRNAS for Optimizing Chemotherapeutic Response in Ovarian Cancer. Cancers, 12(9), 2406. https://doi.org/10.3390/cancers12092406








