Plasma-Derived Extracellular Vesicles Convey Protein Signatures That Reflect Pathophysiology in Lung and Pancreatic Adenocarcinomas
Abstract
1. Introduction
2. Results
2.1. Proteomic Characterization Extracellular Vesicles from LUAD and PDAC Cell Lines
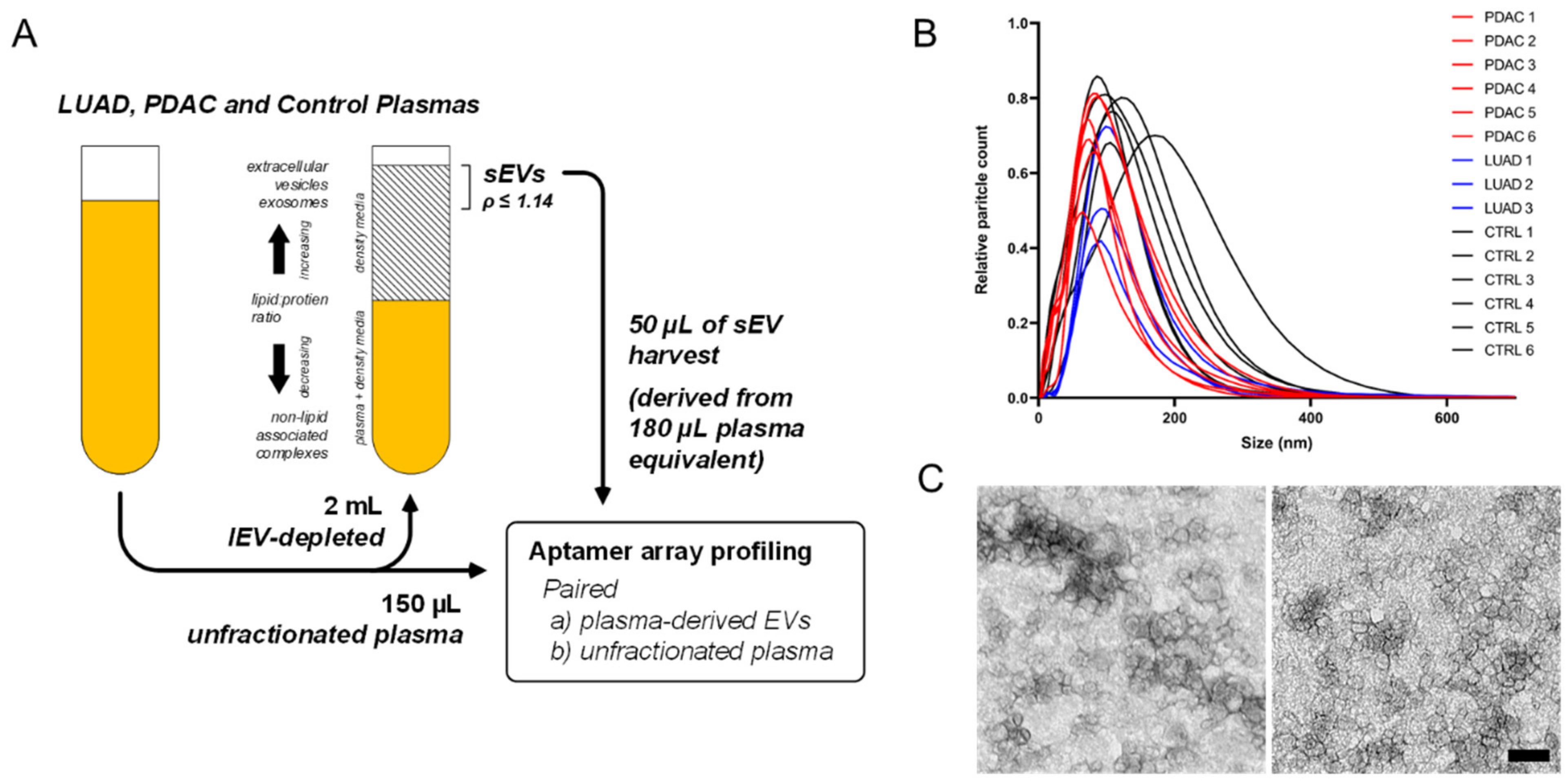
2.2. Isolation of Extracellular Vesicles from LUAD and PDAC Patient Plasmas
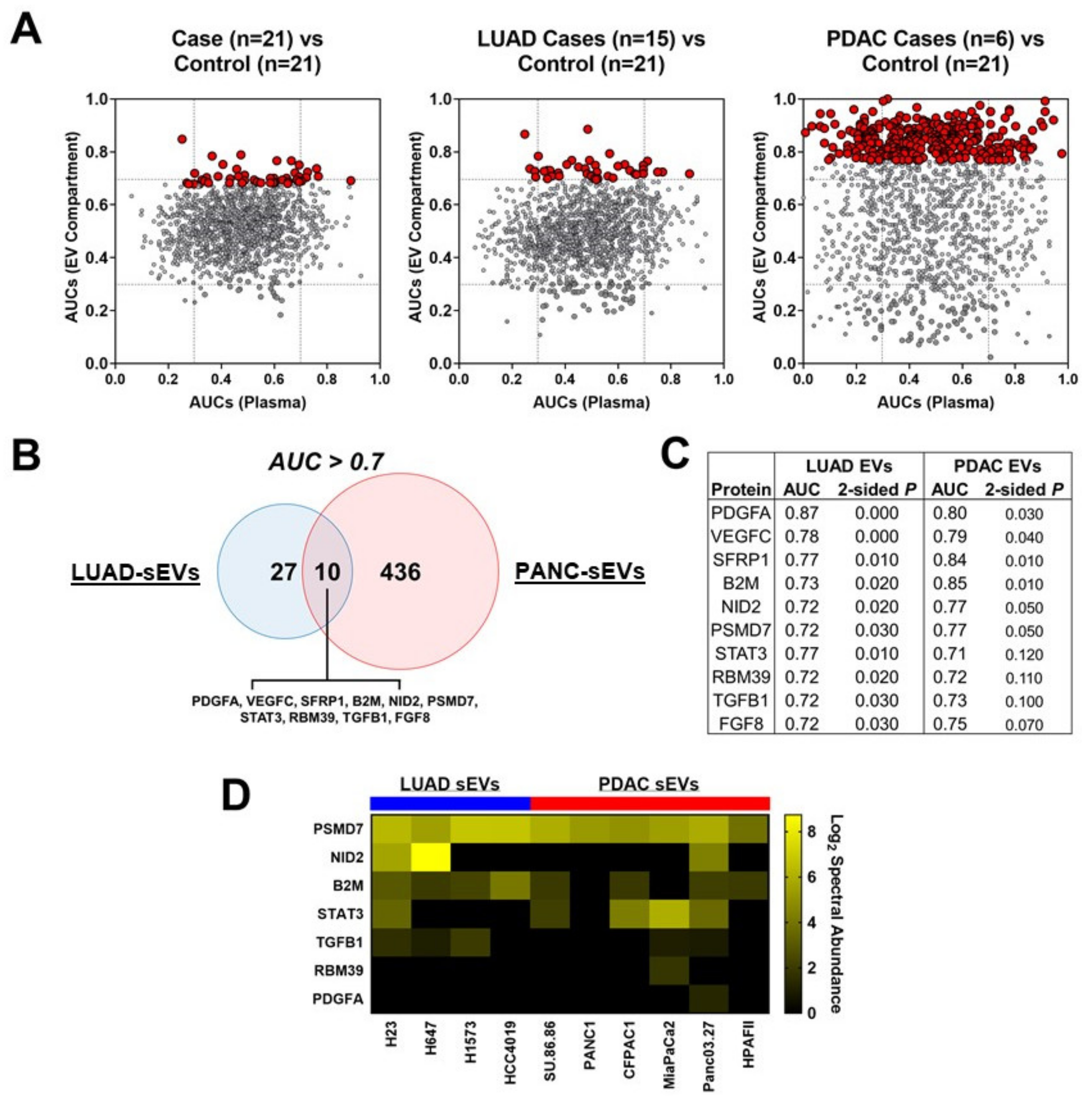
2.3. Profiling of Extracellular Vesicles from LUAD and PDAC Patient Plasmas
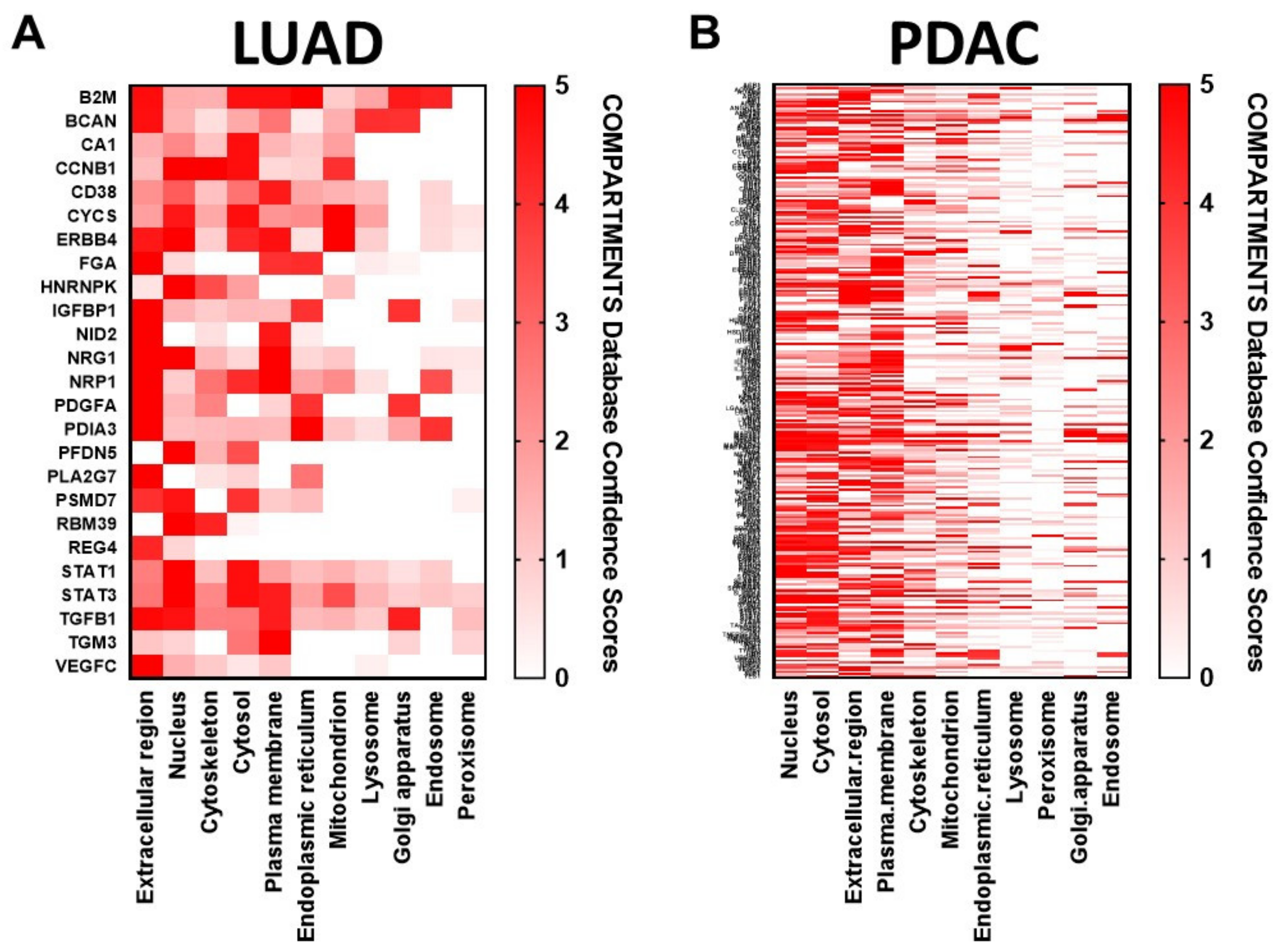
2.4. Extracellular Vesicle Associated Protein Cargoes Reflect Cancer Pathophysiology and Present a Diverse Intercellular Milieu
3. Discussion
4. Materials and Methods
4.1. Blood Samples
4.2. Exosome Isolation
4.3. Transmission Electron Microscopy
4.4. Particle Size Distribution and Quantification
4.5. Protein Quantitation, SDS-PAGE and Western Blot Assay
4.6. Aptamer Array
4.7. Mass Spectrometry Analysis
4.8. Statistical and Bioinformatics Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Pathway ID | Pathway Description | Observed Gene Count | False Discovery Rate |
|---|---|---|---|
| A. GO enrichment, Biological Process: LUAD and PDAC Cell Line sEVs. | |||
| GO:0016192 | vesicle-mediated transport | 476 | 6.28 × 10−84 |
| GO:0051234 | establishment of localization | 790 | 6.73 × 10−73 |
| GO:0006810 | transport | 769 | 1.52 × 10−70 |
| GO:0051179 | localization | 896 | 2.09 × 10−70 |
| GO:0009987 | cellular process | 1735 | 5.45 × 10−70 |
| GO:0071840 | cellular component organization or biogenesis | 903 | 9.03 × 10−69 |
| GO:0016043 | cellular component organization | 871 | 1.01 × 10−64 |
| GO:0006887 | exocytosis | 272 | 5.14 × 10−61 |
| GO:0045055 | regulated exocytosis | 251 | 3.56 × 10−58 |
| GO:0032940 | secretion by cell | 293 | 2.31 × 10−55 |
| B. GO enrichment, Molecular Function: LUAD and PDAC Cell Line sEVs. | |||
| GO:0005515 | protein binding | 1092 | 5.70 × 10−88 |
| GO:0005488 | binding | 1517 | 5.88 × 10−65 |
| GO:1901265 | nucleoside phosphate binding | 464 | 6.80 × 10−55 |
| GO:0000166 | nucleotide binding | 463 | 1.08 × 10−54 |
| GO:0043168 | anion binding | 539 | 3.26 × 10−53 |
| GO:0097367 | carbohydrate derivative binding | 467 | 5.88 × 10−53 |
| GO:0036094 | small molecule binding | 503 | 1.40 × 10−51 |
| GO:0017076 | purine nucleotide binding | 420 | 5.50 × 10−51 |
| GO:0035639 | purine ribonucleoside triphosphate binding | 409 | 1.11 × 10−50 |
| GO:0032555 | purine ribonucleotide binding | 415 | 7.85 × 10−50 |
| C. GO enrichment, Cellular Component: LUAD and PDAC Cell Line sEVs. | |||
| GO:0005829 | cytosol | 1058 | 2.42 × 10−154 |
| GO:0005737 | cytoplasm | 1639 | 1.56 × 10−151 |
| GO:0044444 | cytoplasmic part | 1489 | 7.89 × 10−150 |
| GO:0044424 | intracellular part | 1759 | 3.34 × 10−110 |
| GO:0005622 | intracellular | 1772 | 2.75 × 10−107 |
| GO:0044422 | organelle part | 1334 | 1.14 × 10−87 |
| GO:0044446 | intracellular organelle part | 1308 | 8.44 × 10−86 |
| GO:0032991 | protein−containing complex | 864 | 3.87 × 10−78 |
| GO:0044464 | cell part | 1836 | 7.39 × 10−76 |
| GO:0031982 | vesicle | 542 | 1.65 × 10−74 |
| A. Top-ranked statistically enriched Upstream Regulators. | |||
|---|---|---|---|
| Upstream Regulator | Molecule Type | p-Value | Mechanistic Network |
| TP53 | transcription regulator | 1.69 × 10−77 | 1105 (22) |
| MYC | transcription regulator | 1.13 × 10−70 | 1312 (20) |
| MAPT | other | 1.26 × 10−63 | 1145 (18) |
| TGFB1 | growth factor | 5.91 × 10−56 | 1169 (20) |
| MYCN | transcription regulator | 1.07 × 10−50 | 1063 (18) |
| KRAS | enzyme | 2.93 × 10−49 | 1119 (19) |
| B. Top ten statistically enriched IPA Diseases and Functions annotations. | |||
| Category | Diseases or Functions Annotation | p-Value | #Molecules |
| Cancer, Organismal Injury & Abnormalities | Nonhematologic malignant neoplasm | 2.76 × 10−133 | 2431 |
| Cancer, Organismal Injury & Abnormalities | Non-hematological solid tumor | 4.36 × 10−133 | 2436 |
| Cancer, Organismal Injury & Abnormalities | Epithelial neoplasm | 2.79 × 10−131 | 2410 |
| Cancer, Organismal Injury & Abnormalities | Carcinoma | 4.72 × 10−131 | 2407 |
| Cancer, Organismal Injury & Abnormalities | Non−melanoma solid tumor | 4.72 × 10−131 | 2421 |
| Cancer, Organismal Injury & Abnormalities | Tumorigenesis of tissue | 4.25 × 10−130 | 2414 |
| Cancer, Organismal Injury & Abnormalities | Cancer | 3.96 × 10−123 | 2457 |
| Cancer, Organismal Injury & Abnormalities | Malignant solid tumor | 2.43 × 10−120 | 2445 |
| Cancer, Organismal Injury & Abnormalities | Extracranial solid tumor | 9.08 × 10−119 | 2442 |
| Cancer, Organismal Injury & Abnormalities | Solid tumor | 1.03 × 10−118 | 2452 |
| C. Top ten statistically enriched IPA Cancer Signaling Canonical Pathways. | |||
| Ingenuity Canonical Pathway | p-Value | Ratio | #Molecules |
| Breast Cancer Regulation by Stathmin1 | 5.01 × 10−32 | 0.440 | 88/200 |
| ERK/MAPK Signaling | 5.01 × 10−22 | 0.378 | 73/193 |
| Role of Tissue Factor in Cancer | 6.31 × 10−19 | 0.436 | 51/117 |
| FAK Signaling | 1.26 × 10−18 | 0.474 | 45/95 |
| PI3K/AKT Signaling | 2.00 × 10−17 | 0.356 | 62/174 |
| Molecular Mechanisms of Cancer | 1.58 × 10−16 | 0.261 | 102/391 |
| Telomerase Signaling | 3.16 × 10−13 | 0.383 | 41/107 |
| Glioma Invasiveness Signaling | 3.98 × 10−13 | 0.446 | 37/74 |
| BAG2 Signaling Pathway | 1.58 × 10−12 | 0.558 | 24/43 |
| PTEN Signaling | 3.98 × 10−11 | 0.333 | 42/126 |
| Pathway ID | Pathway Description | Observed Gene Count | False Discovery Rate |
|---|---|---|---|
| A. GO enrichment analysis, Biological Process: ≥1.5-fold higher log2(SA) in LUAD vs. PDAC sEVs. | |||
| GO:0016192 | vesicle-mediated transport | 57 | 2.74 × 10−18 |
| GO:0002443 | leukocyte mediated immunity | 35 | 2.79 × 10−16 |
| GO:0045055 | regulated exocytosis | 36 | 3.43 × 10−16 |
| GO:0043312 | neutrophil degranulation | 31 | 4.72 × 10−16 |
| GO:0002252 | immune effector process | 40 | 6.09 × 10−16 |
| GO:0032940 | secretion by cell | 40 | 1.07 × 10−15 |
| GO:0002366 | leukocyte activation involved in immune response | 33 | 1.30 × 10−15 |
| GO:0002274 | myeloid leukocyte activation | 32 | 1.40 × 10−15 |
| GO:0045321 | leukocyte activation | 36 | 1.36 × 10−13 |
| GO:0001775 | cell activation | 38 | 2.21 × 10−13 |
| B. GO enrichment analysis, Molecular Function: ≥ 1.5-fold higher log2(SA) in LUAD vs. PDAC sEVs. | |||
| GO:0004298 | threonine-type endopeptidase activity | 15 | 3.10 × 10−20 |
| GO:0036094 | small molecule binding | 64 | 7.39 × 10−17 |
| GO:1901265 | nucleoside phosphate binding | 59 | 7.39 × 10−17 |
| GO:0003924 | GTPase activity | 25 | 1.80 × 10−16 |
| GO:0000166 | nucleotide binding | 58 | 2.40 × 10−16 |
| GO:0005525 | GTP binding | 26 | 3.67 × 10−15 |
| GO:0003824 | catalytic activity | 94 | 4.39 × 10−15 |
| GO:0097367 | carbohydrate derivative binding | 56 | 8.68 × 10−15 |
| GO:0016787 | hydrolase activity | 59 | 2.24 × 10−14 |
| GO:0032553 | ribonucleotide binding | 51 | 3.56 × 10−14 |
| C. GO enrichment analysis, Cellular Component: ≥1.5-fold higher log2(SA) in LUAD vs. PDAC sEVs. | |||
| GO:0005839 | proteasome core complex | 15 | 2.68 × 10−20 |
| GO:0005829 | cytosol | 93 | 1.48 × 10−17 |
| GO:0030141 | secretory granule | 39 | 3.32 × 10−17 |
| GO:0099503 | secretory vesicle | 41 | 4.69 × 10−17 |
| GO:0044433 | cytoplasmic vesicle part | 49 | 1.04 × 10−16 |
| GO:0000502 | proteasome complex | 16 | 1.15 × 10−16 |
| GO:0031410 | cytoplasmic vesicle | 60 | 1.18 × 10−16 |
| GO:0031982 | vesicle | 61 | 1.18 × 10−16 |
| GO:0044444 | cytoplasmic part | 126 | 1.92 × 10−16 |
| GO:0060205 | cytoplasmic vesicle lumen | 24 | 3.50 × 10−14 |
| Pathway ID | Pathway Description | Observed Gene Count | False Discovery Rate |
|---|---|---|---|
| A. GO enrichment analysis, Biological Process: ≥1.5-fold higher log2(SA) in PDAC vs. LUAD sEVs. | |||
| GO:0006401 | RNA catabolic process | 17 | 1.72 × 10−10 |
| GO:0006412 | translation | 20 | 1.72 × 10−10 |
| GO:0043604 | amide biosynthetic process | 22 | 1.79 × 10−10 |
| GO:0006413 | translational initiation | 14 | 1.82 × 10−10 |
| GO:0000184 | nuclear−transcribed mRNA catabolic process, nonsense-mediated decay | 13 | 3.02 × 10−10 |
| GO:0000956 | nuclear-transcribed mRNA catabolic process | 15 | 3.35 × 10−10 |
| GO:0006614 | SRP-dependent cotranslational protein targeting to membrane | 12 | 3.35 × 10−10 |
| GO:0051641 | cellular localization | 42 | 3.35 × 10−10 |
| GO:0006518 | peptide metabolic process | 21 | 4.88 × 10−10 |
| GO:0046907 | intracellular transport | 33 | 5.36 × 10−10 |
| B. GO enrichment analysis, Molecular Function: ≥1.5-fold higher log2(SA) in PDAC vs. LUAD sEVs. | |||
| GO:0031683 | G-protein beta/gamma-subunit complex binding | 8 | 1.23 × 10−9 |
| GO:0003735 | structural constituent of ribosome | 11 | 4.10 × 10−7 |
| GO:0005198 | structural molecule activity | 20 | 4.80 × 10−7 |
| GO:0061608 | nuclear import signal receptor activity | 5 | 1.89 × 10−6 |
| GO:0005048 | signal sequence binding | 7 | 2.27 × 10−6 |
| GO:0044877 | protein−containing complex binding | 22 | 3.48 × 10−6 |
| GO:0003723 | RNA binding | 20 | 7.75 × 10−6 |
| GO:0017076 | purine nucleotide binding | 30 | 1.16 × 10−5 |
| GO:0031821 | G protein-coupled serotonin receptor binding | 4 | 1.51 × 10−5 |
| GO:0035639 | purine ribonucleoside triphosphate binding | 29 | 1.51 × 10−5 |
| C. GO enrichment analysis, Cellular Component: ≥1.5-fold higher log2(SA) in PDAC vs. LUAD sEVs. | |||
| GO:0005829 | cytosol | 75 | 7.57 × 10−17 |
| GO:0032991 | protein-containing complex | 72 | 7.86 × 10−16 |
| GO:0044444 | cytoplasmic part | 95 | 7.79 × 10−13 |
| GO:0005737 | cytoplasm | 102 | 7.82 × 10−12 |
| GO:0005622 | intracellular | 111 | 3.33 × 10−10 |
| GO:0044424 | intracellular part | 110 | 3.78 × 10−10 |
| GO:0005834 | heterotrimeric G-protein complex | 8 | 2.04 × 10−9 |
| GO:1990904 | ribonucleoprotein complex | 22 | 3.95 × 10−8 |
| GO:0044464 | cell part | 113 | 6.82 × 10−7 |
| GO:0019897 | extrinsic component of plasma membrane | 10 | 8.30 × 10−7 |
| Feature | Cases, LUAD & PDAC | LUAD Cases | PDAC Cases | Controls | ||||
|---|---|---|---|---|---|---|---|---|
| N | % | N | % | N | % | N | % | |
| N | 21 | 15 | 6 | 21 | ||||
| Sex | ||||||||
| Male | 9 | 42.9% | 5 | 33.3% | 4 | 66.7% | 9 | 42.9% |
| Female | 12 | 57.1% | 10 | 66.7% | 2 | 33.3% | 12 | 57.1% |
| Age, y | 69 ± 12 | 70 ± 12 | 65 ± 3 | 65 ± 4 | ||||
| Smoking | ||||||||
| Never | 5 | 33.3% | - | - | ||||
| Former | 6 | 40.0% | - | - | ||||
| Current | 4 | 26.7% | - | - | ||||
| Stage | ||||||||
| I | 8 | 38.1% | 8 | 53.3% | - | |||
| II | 7 | 33.3% | 7 | 46.7% | - | |||
| IV | 6 | 28.6% | 6 | 100% | - | |||
| Upstream Regulator | Molecule Type | p-Value | Mechanistic Network |
|---|---|---|---|
| A. Lung adenocarcinoma (LUAD) plasma-derived sEVs | |||
| AHR | ligand-dependent nuclear receptor | 1.22 × 10−7 | |
| Vegf | group | 1.14 × 10−6 | |
| EGF | growth factor | 1.38 × 10−6 | 13 (7) |
| IL6 | cytokine | 1.58 × 10−6 | 13 (10) |
| IRF1 | transcription regulator | 1.93 × 10−6 | |
| STAT3 | transcription regulator | 3.45 × 10−6 | 13 (8) |
| TAF4 | transcription regulator | 3.91 × 10−6 | |
| TP73 | transcription regulator | 5.19 × 10−6 | |
| B. Pancreatic ductal adenocarcinoma (PDAC) plasma-derived sEVs | |||
| TNF | cytokine | 2.45 × 10−29 | 134 (13) |
| APP | other | 3.50 × 10−25 | 154 (18) |
| INFG | cytokine | 5.92 × 10−24 | 143 (14) |
| TGFB1 | growth factor | 9.62 × 10−23 | 156 (19) |
| IL1B | cytokine | 2.97 × 10−20 | 134 (13) |
| KRAS | enzyme | 1.63 × 10−19 | 162 (22) |
| HGF | growth factor | 2.26 × 10−18 | 153 (19) |
| TP53 | transcription regulator | 2.96 × 10−18 | 133 (16) |
References
- Tkach, M.; Kowal, J.; Théry, C. Why the need and how to approach the functional diversity of extracellular vesicles. Phil. Trans. R. Soc. B 2018, 373, e20160479. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, e1535750. [Google Scholar] [CrossRef] [PubMed]
- Witwer, K.W.; Théry, C. Extracellular vesicles or exosomes? On primacy, precision and popularity influencing a choice of nomenclature. J. Extracell. Vesicles 2019, 8, e1648167. [Google Scholar] [CrossRef] [PubMed]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef]
- Jeppesen, D.K.; Fenix, A.M.; Franklin, J.L.; Higginbotham, J.N.; Zhang, Q.; Zimmerman, L.J.; Liebler, D.C.; Ping, J.; Liu, Q.; Evans, R.; et al. Reassessment of Exosome Composition. Cell 2019, 177, 428–445. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, M.; Martin-Jaular, L.; Lavieu, G.; Théry, C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat. Cell Biol. 2019, 21, 9–17. [Google Scholar] [CrossRef]
- Daßler-Plenker, J.; Küttner, V.; Egeblad, M. Communication in tiny packages: Exosomes as means of tumor-stroma communication. Biochim. Biophys. Acta BBA-Rev. Cancer 2020, 1873, e188340. [Google Scholar] [CrossRef]
- Tkach, M.; Théry, C. Communication by Extracellular Vesicles: Where We Are and Where We Need to Go. Cell 2016, 164, 1226–1232. [Google Scholar] [CrossRef]
- Becker, A.; Thakur, B.K.; Weiss, J.M.; Kim, H.S.; Peinado, H.; Lyden, D. Extracellular Vesicles in Cancer: Cell-to-Cell Mediators of Metastasis. Cancer Cell 2016, 30, 836–848. [Google Scholar] [CrossRef]
- Capello, M.; Vykoukal, J.V.; Katayama, H.; Bantis, L.E.; Wang, H.; Kundnani, D.L.; Aguilar-Bonavides, C.; Aguilar, M.; Tripathi, S.C.; Dhillon, D.S.; et al. Exosomes harbor B cell targets in pancreatic adenocarcinoma and exert decoy function against complement-mediated cytotoxicity. Nat. Commun. 2019, 10, e254. [Google Scholar] [CrossRef]
- Murillo, O.D.; Thistlethwaite, W.; Rozowsky, J.; Subramanian, S.L.; Lucero, R.; Shah, N.; Jackson, A.R.; Srinivasan, S.; Chung, A.; Laurent, C.D.; et al. exRNA Atlas Analysis Reveals Distinct Extracellular RNA Cargo Types and Their Carriers Present across Human Biofluids. Cell 2019, 177, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Rontogianni, S.; Synadaki, E.; Li, B.; Liefaard, M.C.; Lips, E.H.; Wesseling, J.; Wu, W.; Altelaar, M. Proteomic profiling of extracellular vesicles allows for human breast cancer subtyping. Commun. Biol. 2019, 2, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Jansen, F.H.; Krijgsveld, J.; van Rijswijk, A.; van den Bemd, G.-J.; van den Berg, M.S.; van Weerden, W.M.; Willemsen, R.; Dekker, L.J.; Luider, T.M.; Jenster, G. Exosomal Secretion of Cytoplasmic Prostate Cancer Xenograft-derived Proteins. Mol. Cell. Proteom. 2009, 8, 1192–1205. [Google Scholar] [CrossRef] [PubMed]
- Minciacchi, V.R.; Zijlstra, A.; Rubin, M.A.; Vizio, D.D. Extracellular vesicles for liquid biopsy in prostate cancer: Where are we and where are we headed? Prostate Cancer Prostatic Dis. 2017, 20, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Willms, E.; Cabañas, C.; Mäger, I.; Wood, M.J.A.; Vader, P. Extracellular Vesicle Heterogeneity: Subpopulations, Isolation Techniques and Diverse Functions in Cancer Progression. Front. Immunol. 2018, 9, e738. [Google Scholar] [CrossRef]
- Kosaka, N.; Yoshioka, Y.; Fujita, Y.; Ochiya, T. Versatile roles of extracellular vesicles in cancer. J. Clin. Investig. 2016, 126, 1163–1172. [Google Scholar] [CrossRef]
- Chevillet, J.R.; Kang, Q.; Ruf, I.K.; Briggs, H.A.; Vojtech, L.N.; Hughes, S.M.; Cheng, H.H.; Arroyo, J.D.; Meredith, E.K.; Gallichotte, E.N.; et al. Quantitative and stoichiometric analysis of the microRNA content of exosomes. Proc. Natl. Acad. Sci. USA 2014, 111, 14888–14893. [Google Scholar] [CrossRef]
- Castillo, J.; Bernard, V.; San Lucas, F.A.; Allenson, K.; Capello, M.; Kim, D.U.; Gascoyne, P.; Mulu, F.C.; Stephens, B.M.; Huang, J.; et al. Surfaceome profiling enables isolation of cancer-specific exosomal cargo in liquid biopsies from pancreatic cancer patients. Ann. Oncol. 2018, 29, 223–229. [Google Scholar] [CrossRef]
- Carvalho, A.S.; Baeta, H.; Silva, B.C.; Moraes, M.C.S.; Bodo, C.; Beck, H.C.; Rodriguez, M.S.; Saraswat, M.; Pandey, A.; Matthiesen, R. Extra-cellular vesicles carry proteome of cancer hallmarks. Front. Biosci. Landmark Ed. 2020, 25, 398–436. [Google Scholar]
- Chen, R.; Xu, X.; Qian, Z.; Zhang, C.; Niu, Y.; Wang, Z.; Sun, J.; Zhang, X.; Yu, Y. The biological functions and clinical applications of exosomes in lung cancer. Cell. Mol. Life Sci. 2019, 76, 4613–4633. [Google Scholar] [CrossRef]
- Sandfeld-Paulsen, B.; Aggerholm-Pedersen, N.; Bæk, R.; Jakobsen, K.R.; Meldgaard, P.; Folkersen, B.H.; Rasmussen, T.R.; Varming, K.; Jørgensen, M.M.; Sorensen, B.S. Exosomal proteins as prognostic biomarkers in non-small cell lung cancer. Mol. Oncol. 2016, 10, 1595–1602. [Google Scholar] [CrossRef] [PubMed]
- Vykoukal, J.; Sun, N.; Aguilar-Bonavides, C.; Katayama, H.; Tanaka, I.; Fahrmann, J.F.; Capello, M.; Fujimoto, J.; Aguilar, M.; Wistuba, I.I.; et al. Plasma-derived extracellular vesicle proteins as a source of biomarkers for lung adenocarcinoma. Oncotarget 2017, 8, 95466–95480. [Google Scholar] [CrossRef] [PubMed]
- Webber, J.; Stone, T.C.; Katilius, E.; Smith, B.C.; Gordon, B.; Mason, M.D.; Tabi, Z.; Brewis, I.A.; Clayton, A. Proteomics Analysis of Cancer Exosomes Using a Novel Modified Aptamer-based Array (SOMAscan) Platform. Mol. Cell. Proteom. 2014, 13, 1050–1064. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Amigorena, S.; Raposo, G.; Clayton, A. Isolation and Characterization of Exosomes from Cell Culture Supernatants and Biological Fluids. In Current Protocols in Cell Biology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2001; ISBN 978-0-471-14303-1. [Google Scholar]
- Szklarczyk, D.; Gable, A.L.; Lyon, D.; Junge, A.; Wyder, S.; Huerta-Cepas, J.; Simonovic, M.; Doncheva, N.T.; Morris, J.H.; Bork, P.; et al. STRING v11: Protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019, 47, D607–D613. [Google Scholar] [CrossRef]
- Gold, L.; Ayers, D.; Bertino, J.; Bock, C.; Bock, A.; Brody, E.N.; Carter, J.; Dalby, A.B.; Eaton, B.E.; Fitzwater, T.; et al. Aptamer-Based Multiplexed Proteomic Technology for Biomarker Discovery. PLoS ONE 2010, 5, e15004. [Google Scholar] [CrossRef]
- Welton, J.L.; Brennan, P.; Gurney, M.; Webber, J.P.; Spary, L.K.; Carton, D.G.; Falcón-Pérez, J.M.; Walton, S.P.; Mason, M.D.; Tabi, Z.; et al. Proteomics analysis of vesicles isolated from plasma and urine of prostate cancer patients using a multiplex, aptamer-based protein array. J. Extracell. Vesicles 2016, 5, e31209. [Google Scholar] [CrossRef]
- Binder, J.X.; Pletscher-Frankild, S.; Tsafou, K.; Stolte, C.; O’Donoghue, S.I.; Schneider, R.; Jensen, L.J. COMPARTMENTS: Unification and visualization of protein subcellular localization evidence. Database 2014, 2014. [Google Scholar] [CrossRef]
- Jiao, Y.-J.; Jin, D.-D.; Jiang, F.; Liu, J.-X.; Qu, L.-S.; Ni, W.-K.; Liu, Z.-X.; Lu, C.-H.; Ni, R.-Z.; Zhu, J.; et al. Characterization and proteomic profiling of pancreatic cancer-derived serum exosomes. J. Cell. Biochem. 2019, 120, 988–999. [Google Scholar] [CrossRef]
- Wang, N.; Song, X.; Liu, L.; Niu, L.; Wang, X.; Song, X.; Xie, L. Circulating exosomes contain protein biomarkers of metastatic non-small-cell lung cancer. Cancer Sci. 2018, 109, 1701–1709. [Google Scholar] [CrossRef]
- Safi, A.; Sadmi, M.; Martinet, N.; Menard, O.; Vaillant, P.; Gallati, H.; Hosang, M.; Martinet, Y. Presence of Elevated Levels of Platelet-Derived Growth Factor (PDGF) in Lung Adenocarcinoma Pleural Effusions. Chest 1992, 102, 204–207. [Google Scholar] [CrossRef][Green Version]
- Lunardi, S.; Muschel, R.J.; Brunner, T.B. The stromal compartments in pancreatic cancer: Are there any therapeutic targets? Cancer Lett. 2014, 343, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Yang, J.; Su, H.-Y.; Waldron, R.T.; Zhi, M.; Li, L.; Xia, Q.; Pandol, S.J.; Lugea, A. Yes-Associated Protein 1 Plays Major Roles in Pancreatic Stellate Cell Activation and Fibroinflammatory Responses. Front. Physiol. 2019, 10, e1467. [Google Scholar] [CrossRef] [PubMed]
- Karaman, S.; Detmar, M. Mechanisms of lymphatic metastasis. J. Clin. Investig. 2014, 124, 922–928. [Google Scholar] [CrossRef] [PubMed]
- Stacker, S.A.; Williams, S.P.; Karnezis, T.; Shayan, R.; Fox, S.B.; Achen, M.G. Lymphangiogenesis and lymphatic vessel remodelling in cancer. Nat. Rev. Cancer 2014, 14, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Dieterich, L.C.; Ikenberg, K.; Bachmann, S.B.; Mangana, J.; Proulx, S.T.; Amann, V.C.; Levesque, M.P.; Dummer, R.; Baluk, P.; et al. Unexpected contribution of lymphatic vessels to promotion of distant metastatic tumor spread. Sci. Adv. 2018, 4, eaat4758. [Google Scholar] [CrossRef] [PubMed]
- Popeda, M.; Stokowy, T.; Bednarz-Knoll, N.; Jurek, A.; Niemira, M.; Bielska, A.; Kretowski, A.; Kalinowski, L.; Szade, J.; Markiewicz, A.; et al. NF-kappa B Signaling-Related Signatures Are Connected with the Mesenchymal Phenotype of Circulating Tumor Cells in Non-Metastatic Breast Cancer. Cancers 2019, 11, e1961. [Google Scholar] [CrossRef]
- Shi, K.; Zhang, J.; Zhao, R.; Yang, L.; Guo, D. PSMD7 downregulation induces apoptosis and suppresses tumorigenesis of esophageal squamous cell carcinoma via the mTOR/p70S6K pathway. FEBS Open Bio. 2018, 8, 533–543. [Google Scholar] [CrossRef]
- Hegde, A.N.; Upadhya, S.C. The ubiquitin–proteasome pathway in health and disease of the nervous system. Trends Neurosci. 2007, 30, 587–595. [Google Scholar] [CrossRef]
- Böse, K.; Nischt, R.; Page, A.; Bader, B.L.; Paulsson, M.; Smyth, N. Loss of Nidogen-1 and -2 Results in Syndactyly and Changes in Limb Development. J. Biol. Chem. 2006, 281, 39620–39629. [Google Scholar] [CrossRef] [PubMed]
- Klinke, D.J.; Torang, A. An unsupervised feature extraction and selection strategy for identifying epithelial-mesenchymal transition state metrics in breast cancer and melanoma. BioRxiv 2019. [Google Scholar] [CrossRef]
- Chai, A.W.Y.; Cheung, A.K.L.; Dai, W.; Ko, J.M.Y.; Lee, N.P.Y.; Chan, K.T.; Law, S.Y.-K.; Lung, M.L. Elevated levels of serum nidogen-2 in esophageal squamous cell carcinoma. Cancer Biomark. 2018, 21, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Chai, A.W.Y.; Cheung, A.K.L.; Dai, W.; Ko, J.M.Y.; Ip, J.C.Y.; Chan, K.W.; Kwong, D.L.-W.; Ng, W.T.; Lee, A.W.M.; Ngan, R.K.C.; et al. Metastasis-suppressing NID2, an epigenetically-silenced gene, in the pathogenesis of nasopharyngeal carcinoma and esophageal squamous cell carcinoma. Oncotarget 2016, 7, 78859–78871. [Google Scholar] [CrossRef] [PubMed]
- Baharudin, R.; Tieng, F.Y.F.; Lee, L.-H.; Ab Mutalib, N.S. Epigenetics of SFRP1: The Dual Roles in Human Cancers. Cancers 2020, 12, e445. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.-J.; Wang, Z.-W.; Chang, Y.-W.; Lee, K.-C.; Lin, W.-H.; Lee, J.-L. SFRPs Are Biphasic Modulators of Wnt-Signaling-Elicited Cancer Stem Cell Properties beyond Extracellular Control. Cell Rep. 2019, 28, 1511–1525. [Google Scholar] [CrossRef] [PubMed]
- Sade-Feldman, M.; Jiao, Y.J.; Chen, J.H.; Rooney, M.S.; Barzily-Rokni, M.; Eliane, J.-P.; Bjorgaard, S.L.; Hammond, M.R.; Vitzthum, H.; Blackmon, S.M.; et al. Resistance to checkpoint blockade therapy through inactivation of antigen presentation. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.; Morelli, A.E. Extracellular vesicle-mediated MHC cross-dressing in immune homeostasis, transplantation, infectious diseases and cancer. Semin. Immunopathol. 2018, 40, 477–490. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.; Gimenez-Xavier, P.; Pros, E.; Pajares, M.J.; Moro, M.; Gomez, A.; Navarro, A.; Condom, E.; Moran, S.; Gomez-Lopez, G.; et al. Genomic Profiling of Patient-Derived Xenografts for Lung Cancer Identifies B2M Inactivation Impairing Immunorecognition. Clin. Cancer Res. 2017, 23, 3203–3213. [Google Scholar] [CrossRef]
- Cohen, J.D.; Li, L.; Wang, Y.; Thoburn, C.; Afsari, B.; Danilova, L.; Douville, C.; Javed, A.A.; Wong, F.; Mattox, A.; et al. Detection and localization of surgically resectable cancers with a multi-analyte blood test. Science 2018, 359, 926–930. [Google Scholar] [CrossRef]
- Kelley, S.O.; Pantel, K. A New Era in Liquid Biopsy: From Genotype to Phenotype. Clin. Chem. 2020, 66, 89–96. [Google Scholar] [CrossRef]
- Fahrmann, J.F.; Bantis, L.E.; Capello, M.; Scelo, G.; Dennison, J.B.; Patel, N.; Murage, E.; Vykoukal, J.; Kundnani, D.L.; Foretova, L.; et al. A Plasma-Derived Protein-Metabolite Multiplexed Panel for Early-Stage Pancreatic Cancer. JNCI J. Natl. Cancer Inst. 2019, 111, 372–379. [Google Scholar] [CrossRef]
- Elazezy, M.; Joosse, S.A. Techniques of using circulating tumor DNA as a liquid biopsy component in cancer management. Comput. Struct. Biotechnol. J. 2018, 16, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Capello, M.; Bantis, L.E.; Scelo, G.; Zhao, Y.; Li, P.; Dhillon, D.S.; Patel, N.J.; Kundnani, D.L.; Wang, H.; Abbruzzese, J.L.; et al. Sequential Validation of Blood-Based Protein Biomarker Candidates for Early-Stage Pancreatic Cancer. JNCI J. Natl. Cancer Inst. 2017, 109. [Google Scholar] [CrossRef] [PubMed]
- Guida, F.; Sun, N.; Bantis, L.E.; Muller, D.C.; Li, P.; Taguchi, A.; Dhillon, D.; Kundnani, D.L.; Patel, N.J.; Yan, Q.; et al. Assessment of Lung Cancer Risk on the Basis of a Biomarker Panel of Circulating Proteins. JAMA Oncol. 2018, 4, e182078. [Google Scholar] [PubMed]
- Wang, H.; Hanash, S. Intact-Protein Analysis System for Discovery of Serum-Based Disease Biomarkers. In Serum/Plasma Proteomics; Simpson, R.J., Greening, D.W., Eds.; Humana Press: Totowa, NJ, USA, 2011; Volume 728, pp. 69–85. ISBN 978-1-61779-067-6. [Google Scholar]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fahrmann, J.F.; Mao, X.; Irajizad, E.; Katayama, H.; Capello, M.; Tanaka, I.; Kato, T.; Wistuba, I.I.; Maitra, A.; Ostrin, E.J.; et al. Plasma-Derived Extracellular Vesicles Convey Protein Signatures That Reflect Pathophysiology in Lung and Pancreatic Adenocarcinomas. Cancers 2020, 12, 1147. https://doi.org/10.3390/cancers12051147
Fahrmann JF, Mao X, Irajizad E, Katayama H, Capello M, Tanaka I, Kato T, Wistuba II, Maitra A, Ostrin EJ, et al. Plasma-Derived Extracellular Vesicles Convey Protein Signatures That Reflect Pathophysiology in Lung and Pancreatic Adenocarcinomas. Cancers. 2020; 12(5):1147. https://doi.org/10.3390/cancers12051147
Chicago/Turabian StyleFahrmann, Johannes F., Xiangying Mao, Ehsan Irajizad, Hiroyuki Katayama, Michela Capello, Ichidai Tanaka, Taketo Kato, Ignacio I. Wistuba, Anirban Maitra, Edwin J. Ostrin, and et al. 2020. "Plasma-Derived Extracellular Vesicles Convey Protein Signatures That Reflect Pathophysiology in Lung and Pancreatic Adenocarcinomas" Cancers 12, no. 5: 1147. https://doi.org/10.3390/cancers12051147
APA StyleFahrmann, J. F., Mao, X., Irajizad, E., Katayama, H., Capello, M., Tanaka, I., Kato, T., Wistuba, I. I., Maitra, A., Ostrin, E. J., Hanash, S. M., & Vykoukal, J. (2020). Plasma-Derived Extracellular Vesicles Convey Protein Signatures That Reflect Pathophysiology in Lung and Pancreatic Adenocarcinomas. Cancers, 12(5), 1147. https://doi.org/10.3390/cancers12051147







