Scientific Rationale for Combined Immunotherapy with PD-1/PD-L1 Antibodies and VEGF Inhibitors in Advanced Hepatocellular Carcinoma
Abstract
1. Introduction
2. The Rationale Underlying the Combination of PD-1/PD-L1 and VEGF Inhibitors
3. Classification of the Tumor Microenvironment and Determination of Immunotherapeutic Strategies
4. The Results of a Phase Ib Trial of the Combination of Atezolizumab and Bevacizumab (Clinical Trials.Gov Identifier NCT02715531)
4.1. The Use of the Combination of Atezolizumab (a PD-L1 Antibody) and Bevacizumab (a VEGF Antibody) in Unresectable Hepatocellular Carcinoma (Arm A)
4.2. Randomized Controlled Arm Comparing the Combination of Atezolizumab Plus Bevacizumab Versus Atezolizumab Alone (Arm F)
5. Results of Phase Ib Studies of Other Combinations of PD-1/PD-L1 Antibodies and VEGF Inhibitors
6. Conclusions
Funding
Conflicts of Interest
References
- Cheng, A.L.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Zhu, A.X.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; et al. IMbrave150: Efficacy and safety results from a ph III study evaluating atezolizumab (atezo) + bevacizumab (bev) vs sorafenib (Sor) as first treatment (tx) for patients (pts) with unresectable hepatocellular carcinoma (HCC). Ann. Oncol. 2019, 30 (Suppl. 9), ix186–ix187. [Google Scholar] [CrossRef]
- Fukumura, D.; Kloepper, J.; Amoozgar, Z.; Duda, D.G.; Jain, R.K. Enhancing cancer immunotherapy using antiangiogenics: Opportunities and challenges. Nat. Rev. Clin. Oncol. 2018, 15, 325–340. [Google Scholar] [CrossRef]
- Chouaib, S.; Messai, Y.; Couve, S.; Escudier, B.; Hasmim, M.; Noman, M.Z. Hypoxia promotes tumor growth in linking angiogenesis to immune escape. Front. Immunol. 2012, 3, 21. [Google Scholar] [CrossRef] [PubMed]
- Voron, T.; Marcheteau, E.; Pernot, S.; Colussi, O.; Tartour, E.; Taieb, J.; Terme, M. Control of the immune response by pro-angiogenic factors. Front. Oncol. 2014, 4, 70. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.S.; Mellman, I. Oncology meets immunology: The cancer-immunity cycle. Immunity 2013, 39, 1–10. [Google Scholar] [CrossRef]
- Hegde, P.S.; Wallin, J.J.; Mancao, C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immunotherapeutics. Semin. Cancer Biol. 2018, 52, 117–124. [Google Scholar] [CrossRef]
- Ferrara, N.; Hillan, K.J.; Gerber, H.P.; Novotny, W. Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat. Rev. Drug Discov. 2004, 3, 391–400. [Google Scholar] [CrossRef]
- Gabrilovich, D.I.; Chen, H.L.; Girgis, K.R.; Cunningham, H.T.; Meny, G.M.; Nadaf, S.; Kavanaugh, D.; Carbone, D.P. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat. Med. 1996, 2, 1096–1103. [Google Scholar] [CrossRef]
- Gabrilovich, D.; Ishida, T.; Oyama, T.; Ran, S.; Kravtsov, V.; Nadaf, S.; Carbone, D.P. Vascular endothelial growth factor inhibits the development of dendritic cells and dramatically affects the differentiation of multiple hematopoietic lineages in vivo. Blood 1998, 92, 4150–4166. [Google Scholar] [CrossRef]
- Elovic, A.E.; Ohyama, H.; Sauty, A.; McBride, J.; Tsuji, T.; Nagai, M.; Weller, P.F.; Wong, D.T. IL-4-dependent regulation of TGF-alpha and TGF-beta1 expression in human eosinophils. J. Immunol. 1998, 160, 6121–6127. [Google Scholar]
- Guermonprez, P.; Valladeau, J.; Zitvogel, L.; Thery, C.; Amigorena, S. Antigen presentation and T cell stimulation by dendritic cells. Annu. Rev. Immunol. 2002, 20, 621–667. [Google Scholar] [CrossRef] [PubMed]
- Villadangos, J.A.; Schnorrer, P. Intrinsic and cooperative antigen-presenting functions of dendritic-cell subsets in vivo. Nat. Rev. Immunol. 2007, 7, 543–555. [Google Scholar] [CrossRef] [PubMed]
- Griffioen, A.W.; Damen, C.A.; Blijham, G.H.; Groenewegen, G. Tumor angiogenesis is accompanied by a decreased inflammatory response of tumor-associated endothelium. Blood 1996, 88, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Griffioen, A.W.; Damen, C.A.; Martinotti, S.; Blijham, G.H.; Groenewegen, G. Endothelial intercellular adhesion molecule-1 expression is suppressed in human malignancies: The role of angiogenic factors. Cancer Res. 1996, 56, 1111–1117. [Google Scholar] [PubMed]
- Goel, S.; Duda, D.G.; Xu, L.; Munn, L.L.; Boucher, Y.; Fukumura, D.; Jain, R.K. Normalization of the vasculature for treatment of cancer and other diseases. Physiol. Rev. 2011, 91, 1071–1121. [Google Scholar] [CrossRef] [PubMed]
- Motz, G.T.; Santoro, S.P.; Wang, L.P.; Garrabrant, T.; Lastra, R.R.; Hagemann, I.S.; Lal, P.; Feldman, M.D.; Benencia, F.; Coukos, G. Tumor endothelium FasL establishes a selective immune barrier promoting tolerance in tumors. Nat. Med. 2014, 20, 607–615. [Google Scholar] [CrossRef]
- Hodi, F.S.; Lawrence, D.; Lezcano, C.; Wu, X.; Zhou, J.; Sasada, T.; Zeng, W.; Giobbie-Hurder, A.; Atkins, M.B.; Ibrahim, N.; et al. Bevacizumab plus ipilimumab in patients with metastatic melanoma. Cancer Immunol. Res. 2014, 2, 632–642. [Google Scholar] [CrossRef]
- Wallin, J.J.; Bendell, J.C.; Funke, R.; Sznol, M.; Korski, K.; Jones, S.; Hernandez, G.; Mier, J.; He, X.; Hodi, F.S.; et al. Atezolizumab in combination with bevacizumab enhances antigen-specific T-cell migration in metastatic renal cell carcinoma. Nat. Commun. 2016, 7, 12624. [Google Scholar] [CrossRef]
- Gabrilovich, D.I.; Nagaraj, S. Myeloid-derived suppressor cells as regulators of the immune system. Nat. Rev. Immunol. 2009, 9, 162–174. [Google Scholar] [CrossRef]
- Huang, C.; Li, J.; Song, L.; Zhang, D.; Tong, Q.; Ding, M.; Bowman, L.; Aziz, R.; Stoner, G.D. Black raspberry extracts inhibit benzo(a)pyrene diol-epoxide-induced activator protein 1 activation and VEGF transcription by targeting the phosphotidylinositol 3-kinase/Akt pathway. Cancer Res. 2006, 66, 581–587. [Google Scholar] [CrossRef]
- Ko, S.Y.; Guo, H.; Barengo, N.; Naora, H. Inhibition of ovarian cancer growth by a tumor-targeting peptide that binds eukaryotic translation initiation factor 4E. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2009, 15, 4336–4347. [Google Scholar] [CrossRef] [PubMed]
- Kusmartsev, S.; Eruslanov, E.; Kubler, H.; Tseng, T.; Sakai, Y.; Su, Z.; Kaliberov, S.; Heiser, A.; Rosser, C.; Dahm, P.; et al. Oxidative stress regulates expression of VEGFR1 in myeloid cells: Link to tumor-induced immune suppression in renal cell carcinoma. J. Immunol. 2008, 181, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zeng, Q.; Wu, M.X. Improved efficacy of dendritic cell-based immunotherapy by cutaneous laser illumination. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 2240–2249. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.; Chen, L. Inhibitory B7-family molecules in the tumour microenvironment. Nat. Rev. Immunol. 2008, 8, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Tiegs, G.; Lohse, A.W. Immune tolerance: What is unique about the liver. J Autoimmun. 2010, 34, 1–6. [Google Scholar] [CrossRef]
- Oyama, T.; Ran, S.; Ishida, T.; Nadaf, S.; Kerr, L.; Carbone, D.P.; Gabrilovich, D.I. Vascular endothelial growth factor affects dendritic cell maturation through the inhibition of nuclear factor-kappa B activation in hemopoietic progenitor cells. J. Immunol. 1998, 160, 1224–1232. [Google Scholar]
- Kato, Y.; Tabata, K.; Kimura, T.; Yachie-Kinoshita, A.; Ozawa, Y.; Yamada, K.; Ito, J.; Tachino, S.; Hori, Y.; Matsuki, M.; et al. Lenvatinib plus anti-PD-1 antibody combination treatment activates CD8+ T cells through reduction of tumor-associated macrophage and activation of the interferon pathway. PLoS ONE 2019, 14, e0212513. [Google Scholar] [CrossRef]
- Kudo, M. Combination Cancer Immunotherapy in Hepatocellular Carcinoma. Liver Cancer 2018, 7, 20–27. [Google Scholar] [CrossRef]
- Kudo, M. Targeted and immune therapies for hepatocellular carcinoma: Predictions for 2019 and beyond. World J. Gastroenterol. 2019, 25, 789–807. [Google Scholar] [CrossRef]
- Kudo, M. Combination Cancer Immunotherapy with Molecular Targeted Agents/Anti-CTLA-4 Antibody for Hepatocellular Carcinoma. Liver Cancer 2019, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Teng, M.W.; Ngiow, S.F.; Ribas, A.; Smyth, M.J. Classifying Cancers Based on T-cell Infiltration and PD-L1. Cancer Res. 2015, 75, 2139–2145. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.H.; Lee, M.S.; Lee, K.H.; Numata, K.; Stein, S.; Verret, W.; Hack, S.; Spahn, J.; Liu, B.; Huang, C.; et al. Randomised efficacy and safety results for atezolizumab + bevacizumab in patients with previously untreated, unresectable hepatocellular carcinoma. Ann. Oncol. 2019, 30 (Suppl. 9), ix187. [Google Scholar] [CrossRef]
- Yau, T.; Park, J.W.; Finn, R.S.; Cheng, A.L.; Mathurin, P.; Edeline, J.; Kudo, M.; Han, K.H.; Harding, J.J.; Merle, P.; et al. CheckMate 459: A randomized, multi-center phase III study of nivolumab vs sorafenib as first-line treatment in patients with advanced hepatocellular carcinoma. Ann. Oncol. 2019, 30 (Suppl. 5), v874–v875. [Google Scholar] [CrossRef]
- Zhu, A.X.; Finn, R.S.; Edeline, J.; Cattan, S.; Ogasawara, S.; Palmer, D.; Verslype, C.; Zagonel, V.; Fartoux, L.; Vogel, A.; et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): A non-randomised, open-label phase 2 trial. Lancet Oncol. 2018, 19, 940–952. [Google Scholar] [CrossRef]
- Finn, R.S.; Ryoo, B.Y.; Merle, P.; Kudo, M.; Bouattour, M.; Lim, H.Y.; Breder, V.; Edeline, J.; Chao, Y.; Ogasawara, S.; et al. Pembrolizumab As Second-Line Therapy in Patients With Advanced Hepatocellular Carcinoma in KEYNOTE-240: A Randomized, Double-Blind, Phase III Trial. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2020, 38, 193–202. [Google Scholar] [CrossRef]
- Kudo, M. Pembrolizumab for the Treatment of Hepatocellular Carcinoma. Liver Cancer 2019, 8, 143–154. [Google Scholar] [CrossRef]
- Kudo, M. Immune checkpoint blockade in hepatocellular carcinoma: 2017 update. Liver Cancer 2017, 6, 1–12. [Google Scholar] [CrossRef]
- El-Khoueiry, A.B.; Sangro, B.; Yau, T.; Crocenzi, T.S.; Kudo, M.; Hsu, C.; Kim, T.Y.; Choo, S.P.; Trojan, J.; Welling, T.H.R.; et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 2017, 389, 2492–2502. [Google Scholar] [CrossRef]
- Llovet, J.M.; Kudo, M.; Cheng, A.L.; Finn, R.S.; Galle, P.R.; Kaneko, S.; Meyer, T.; Qin, S.; dutcus, C.E.; Chen, E.; et al. First-Line Combination Therapy With Lenvatinib Plus Pembrolizumab for Patients with Advanced Hepatocellular Carcinoma: Phase 3 LEAP-002 Study. In Proceedings of the ILCA 13th Annual Conference, Chicago, IL, USA, 20–22 September 2019. [Google Scholar]
- Kudo, M. Immuno-Oncology Therapy for Hepatocellular Carcinoma: Current Status and Ongoing Trials. Liver Cancer 2019, 8, 221–238. [Google Scholar] [CrossRef]
- Llovet, J.M.; Finn, R.S.; Ikeda, M.; Sung, M.W.; Baron, A.D.; Kudo, M.; Okusaka, T.; Kobayashi, M.; Kumada, H.; Kaneko, S.; et al. A phase 1b trial of lenvatinib plus pembrolizumab in unresectable hepatocellular carcinoma: Upated results. Ann. Oncol. 2019, 30 (Suppl. 5), v253–v324. [Google Scholar] [CrossRef]
- Kudo, M.; Ikeda, K.; Motomura, K.; Okusaka, T.; Kato, N.; Dutcus, C.E.; Hisai, T.; Suzuki, M.; Ikezawa, H.; Iwata, T.; et al. A Phase 1b Study of Lenvatinib Plus Nivolumab in Patients With Unresectable Hepatocellular Carcinoma. In Proceedings of the ASCO-GI, San Francisco, CA, USA, 23–25 January 2020. [Google Scholar]
- Xu, J.M.; Zhang, Y.; Jia, R.; Wang, Y.; Liu, R.; Zhang, G.; Zhao, C.; Zhang, Y.; Zhou, J.; Wang, Q. Anti-programmed death-1 antibody SHR-1210 (S) combined with apatinib (A) for advanced hepatocellular carcinoma (HCC), gastric cancer (GC) or esophagogastric junction (EGJ) cancer refractory to standard therapy: A phase 1 trial. J. Clin. Oncol. 2018, 36, 4075. [Google Scholar] [CrossRef]
- Kudo, M.; Motomura, K.; Wada, Y.; Inaba, Y.; Sakamoto, Y.; Kurosaki, M.; Umeyama, Y.; Kamei, Y.; Yoshimitsu, J.; Fujii, Y.; et al. First-line avelumab + axitinib in patients with advanced hepatocellular carcinoma: Results from a phase 1b trial (VEGF Liver 100). J. Clin. Oncol. 2019, 37 (Suppl. 15). [Google Scholar] [CrossRef]
- Cheng, A.L.; Hsu, C.; Chan, S.L.; Choo, S.P.; Kudo, M. Challenges of combination therapy with immune checkpoint inhibitors for hepatocellular carcinoma. J. Hepatol. 2020, 72, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.L.; Kang, Y.K.; Lin, D.Y.; Park, J.W.; Kudo, M.; Qin, S.; Chung, H.C.; Song, X.; Xu, J.; Poggi, G.; et al. Sunitinib versus sorafenib in advanced hepatocellular cancer: results of a randomized phase III trial. J. Clin. Oncol. 2013, 31, 4067–4075. [Google Scholar] [CrossRef]
- Zhu, A.X.; Rosmorduc, O.; Evans, T.R.; Ross, P.J.; Santoro, A.; Carrilho, F.J.; Bruix, J.; Qin, S.; Thuluvath, P.J.; Llovet, J.M.; et al. SEARCH: a phase III, randomized, double-blind, placebo-controlled trial of sorafenib plus erlotinib in patients with advanced hepatocellular carcinoma. J. Clin. Oncol. 2015, 33, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.J.; Qin, S.; Park, J.W.; Poon, R.T.; Raoul, J.L.; Philip, P.A.; Hsu, C.H.; Hu, T.H.; Heo, J.; Xu, J.; et al. Brivanib versus sorafenib as first-line therapy in patients with unresectable, advanced hepatocellular carcinoma: results from the randomized phase III BRISK-FL study. J. Clin. Oncol. 2013, 31, 3517–3524. [Google Scholar] [CrossRef]
- Cainap, C.; Qin, S.; Huang, W.T.; Chung, I.J.; Pan, H.; Cheng, Y.; Kudo, M.; Kang, Y.K.; Chen, P.J.; Toh, H.C.; et al. Linifanib versus Sorafenib in patients with advanced hepatocellular carcinoma: results of a randomized phase III trial. J. Clin. Oncol. 2015, 33, 172–179. [Google Scholar] [CrossRef]
- Kudo, M.; Ueshima, K.; Yokosuka, O.; Ogasawara, S.; Obi, S.; Izumi, N.; Aikata, H.; Nagano, H.; Hatano, E.; Sasaki, Y.; et al. Sorafenib plus low-dose cisplatin and fluorouracil hepatic arterial infusion chemotherapy versus sorafenib alone in patients with advanced hepatocellular carcinoma (SILIUS): a randomised, open label, phase 3 trial. Lancet Gastroenterol. Hepatol. 2018, 3, 424–432. [Google Scholar] [CrossRef]
- Vilgrain, V.; Pereira, H.; Assenat, E.; Guiu, B.; Ilonca, A.D.; Pageaux, G.P.; Sibert, A.; Bouattour, M.; Lebtahi, R.; Allaham, W.; et al. Efficacy and safety of selective internal radiotherapy with yttrium-90 resin microspheres compared with sorafenib in locally advanced and inoperable hepatocellular carcinoma (SARAH): an open-label randomised controlled phase 3 trial. Lancet Oncol. 2017, 18, 1624–1636. [Google Scholar] [CrossRef]
- Chow, P.K.H.; Gandhi, M.; Tan, S.B.; Khin, M.W.; Khasbazar, A.; Ong, J.; Choo, S.P.; Cheow, P.C.; Chotipanich, C.; Lim, K.; et al. SIRveNIB: Selective Internal Radiation Therapy Versus Sorafenib in Asia-Pacific Patients With Hepatocellular Carcinoma. J. Clin. Oncol. 2018, 36, 1913–1921. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Ricke, J.; Klumpen, H.J.; Amthauer, H.; Bargellini, I.; Bartenstein, P.; de Toni, E.N.; Gasbarrini, A.; Pech, M.; Peck-Radosavljevic, M.; Popovic, P.; et al. Impact of combined selective internal radiation therapy and sorafenib on survival in advanced hepatocellular carcinoma. J. Hepatol. 2019, 71, 1164–1174. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Decaens, T.; Raoul, J.L.; Boucher, E.; Kudo, M.; Chang, C.; Kang, Y.K.; Assenat, E.; Lim, H.Y.; Boige, V.; et al. Brivanib in patients with advanced hepatocellular carcinoma who were intolerant to sorafenib or for whom sorafenib failed: results from the randomized phase III BRISK-PS study. J. Clin. Oncol. 2013, 31, 3509–3516. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.X.; Kudo, M.; Assenat, E.; Cattan, S.; Kang, Y.K.; Lim, H.Y.; Poon, R.T.; Blanc, J.F.; Vogel, A.; Chen, C.L.; et al. Effect of everolimus on survival in advanced hepatocellular carcinoma after failure of sorafenib: the EVOLVE-1 randomized clinical trial. JAMA 2014, 312, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.X.; Park, J.O.; Ryoo, B.Y.; Yen, C.J.; Poon, R.; Pastorelli, D.; Blanc, J.F.; Chung, H.C.; Baron, A.D.; Pfiffer, T.E.; et al. Ramucirumab versus placebo as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib (REACH): a randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol. 2015, 16, 859–870. [Google Scholar] [CrossRef]
- Kudo, M.; Moriguchi, M.; Numata, K.; Hidaka, H.; Tanaka, H.; Ikeda, M.; Kawazoe, S.; Ohkawa, S.; Sato, Y.; Kaneko, S.; et al. S-1 versus placebo in patients with sorafenib-refractory advanced hepatocellular carcinoma (S-CUBE): a randomised, double-blind, multicentre, phase 3 trial. Lancet Gastroenterol. Hepatol. 2017, 2, 407–417. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Qin, S.; Ryoo, B.Y.; Lu, S.N.; Yen, C.J.; Feng, Y.H.; Lim, H.Y.; Izzo, F.; Colombo, M.; Sarker, D.; et al. Phase III randomized study of second line ADI-PEG 20 plus best supportive care versus placebo plus best supportive care in patients with advanced hepatocellular carcinoma. Ann. Oncol. 2018, 29, 1402–1408. [Google Scholar] [CrossRef]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Rimassa, L.; Assenat, E.; Peck-Radosavljevic, M.; Pracht, M.; Zagonel, V.; Mathurin, P.; Rota Caremoli, E.; Porta, C.; Daniele, B.; Bolondi, L.; et al. Tivantinib for second-line treatment of MET-high, advanced hepatocellular carcinoma (METIV-HCC): a final analysis of a phase 3, randomised, placebo-controlled study. Lancet Oncol. 2018, 19, 682–693. [Google Scholar] [CrossRef]
- Merle, P.; Blanc, J.F.; Phelip, J.M.; Pelletier, G.; Bronowicki, J.P.; Touchefeu, Y.; Pageaux, G.; Gerolami, R.; Habersetzer, F.; Nguyen-Khac, E.; et al. Doxorubicin-loaded nanoparticles for patients with advanced hepatocellular carcinoma after sorafenib treatment failure (RELIVE): a phase 3 randomised controlled trial. Lancet Gastroenterol. Hepatol. 2019, 4, 454–465. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Meyer, T.; Cheng, A.L.; El-Khoueiry, A.B.; Rimassa, L.; Ryoo, B.Y.; Cicin, I.; Merle, P.; Chen, Y.; Park, J.W.; et al. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N. Engl. J. Med. 2018, 379, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.X.; Kang, Y.K.; Yen, C.J.; Finn, R.S.; Galle, P.R.; Llovet, J.M.; Assenat, E.; Brandi, G.; Pracht, M.; Lim, H.Y.; et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased alpha-fetoprotein concentrations (REACH-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019, 20, 282–296. [Google Scholar] [CrossRef]
- Kudo, M. Scientific rationale for combination immunotherapy of hepatocellular carcinoma with anti-PD-1/PD-L1 and anti-CTLA-4 antibodies. Liver Cancer 2019, 8, 413–426. [Google Scholar] [CrossRef]
- Bouattour, M.; Mehta, N.; He, A.R.; Cohen, E.I.; Nault, J.C. Systemic Treatment for Advanced Hepatocellular Carcinoma. Liver Cancer 2019, 8, 341–358. [Google Scholar] [CrossRef] [PubMed]
- Rimassa, L.; Pressiani, T.; Merle, P. Systemic Treatment Options in Hepatocellular Carcinoma. Liver Cancer 2019, 8, 427–446. [Google Scholar] [CrossRef]
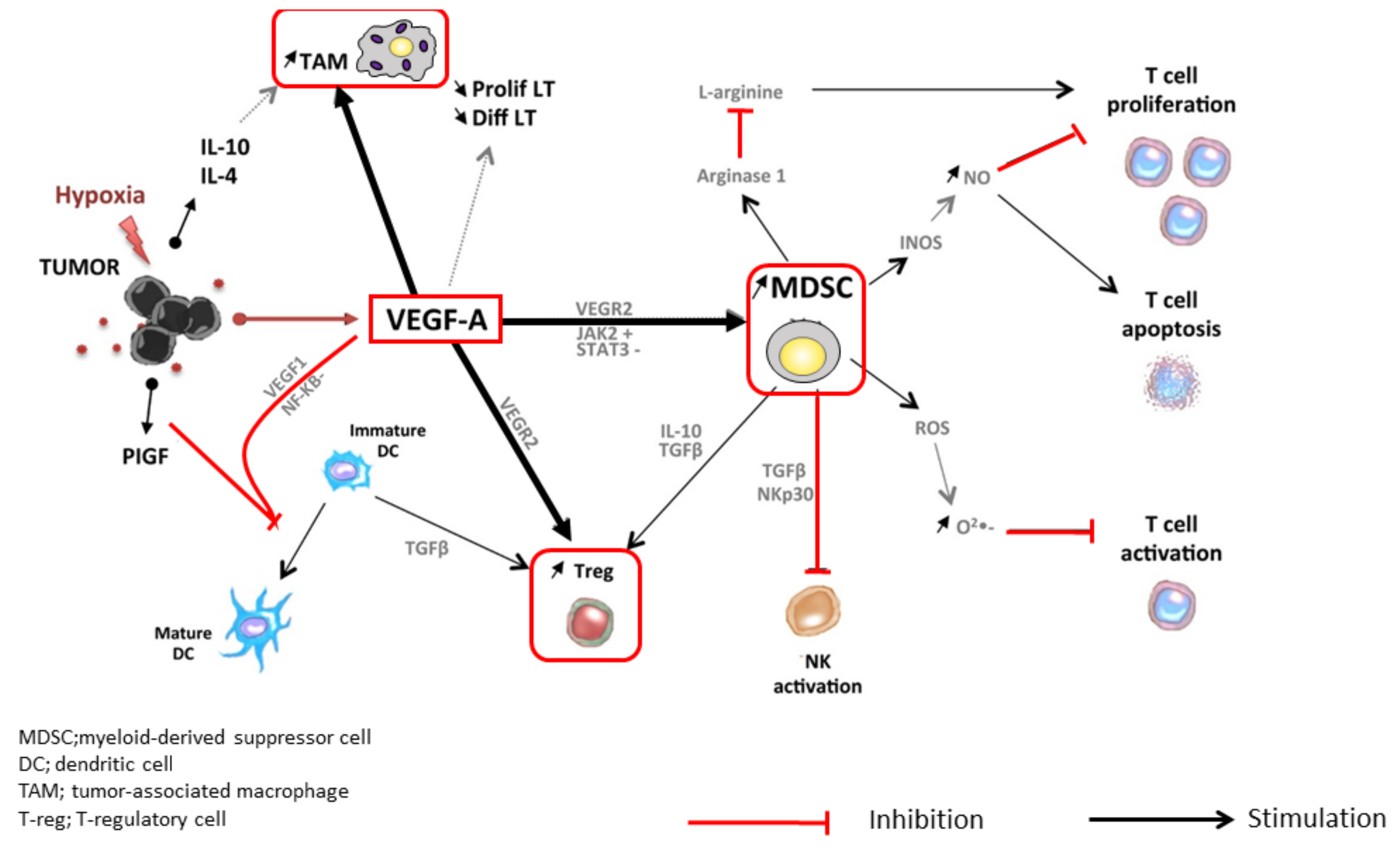
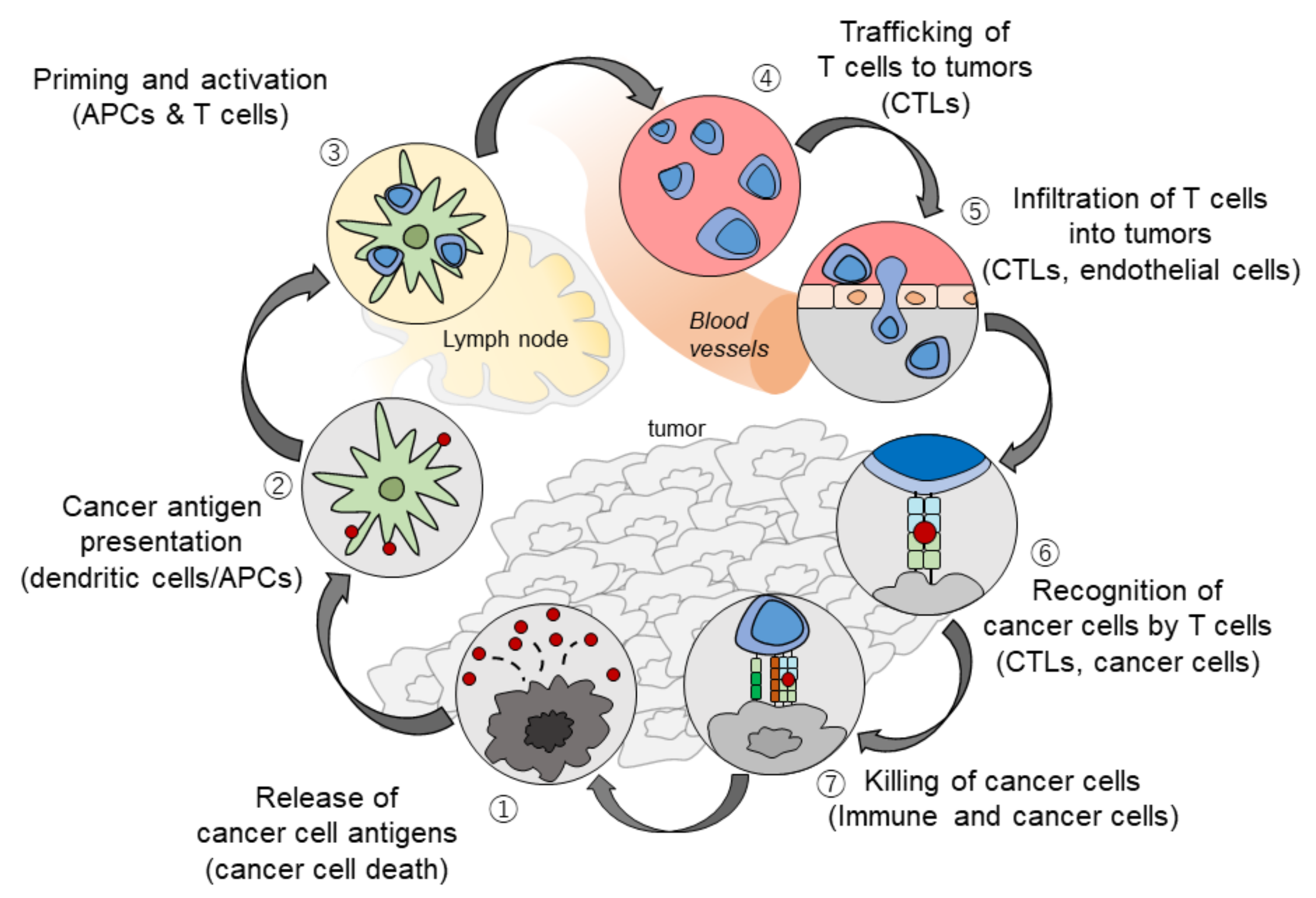

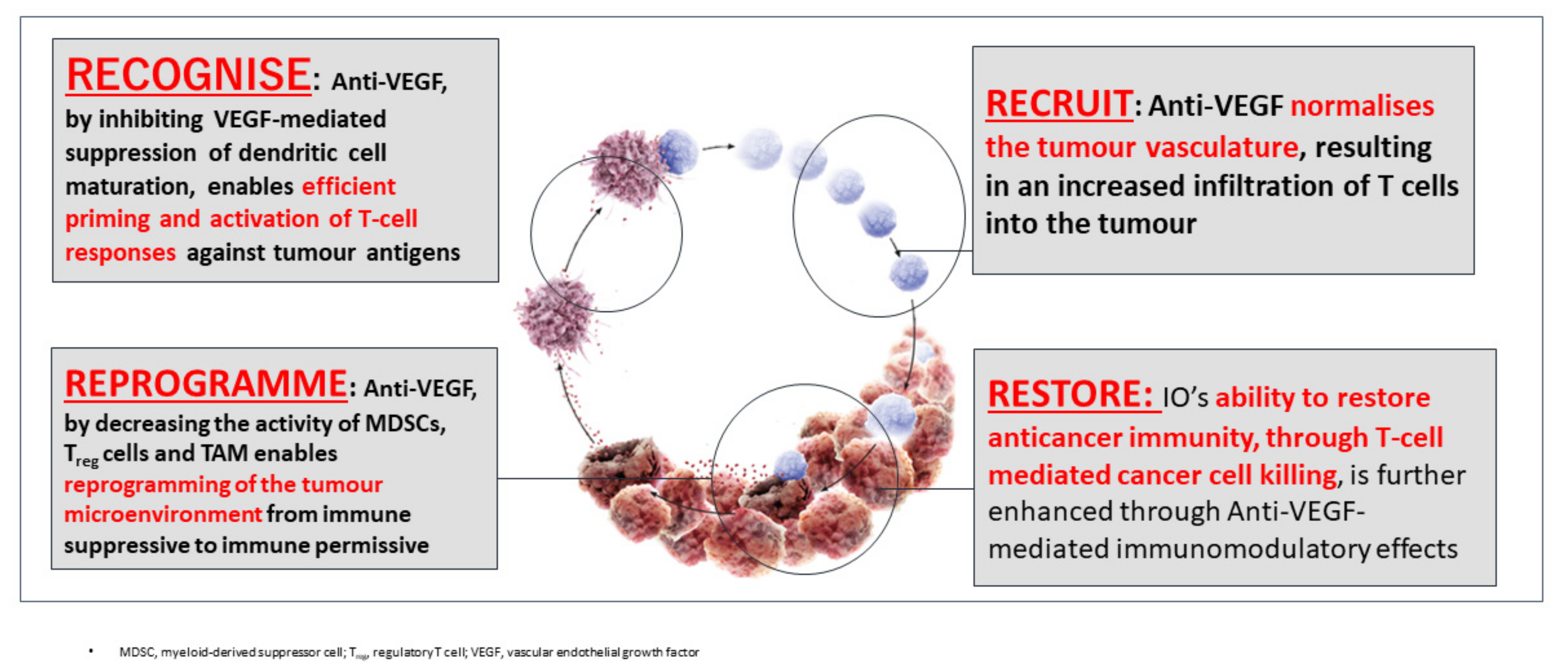
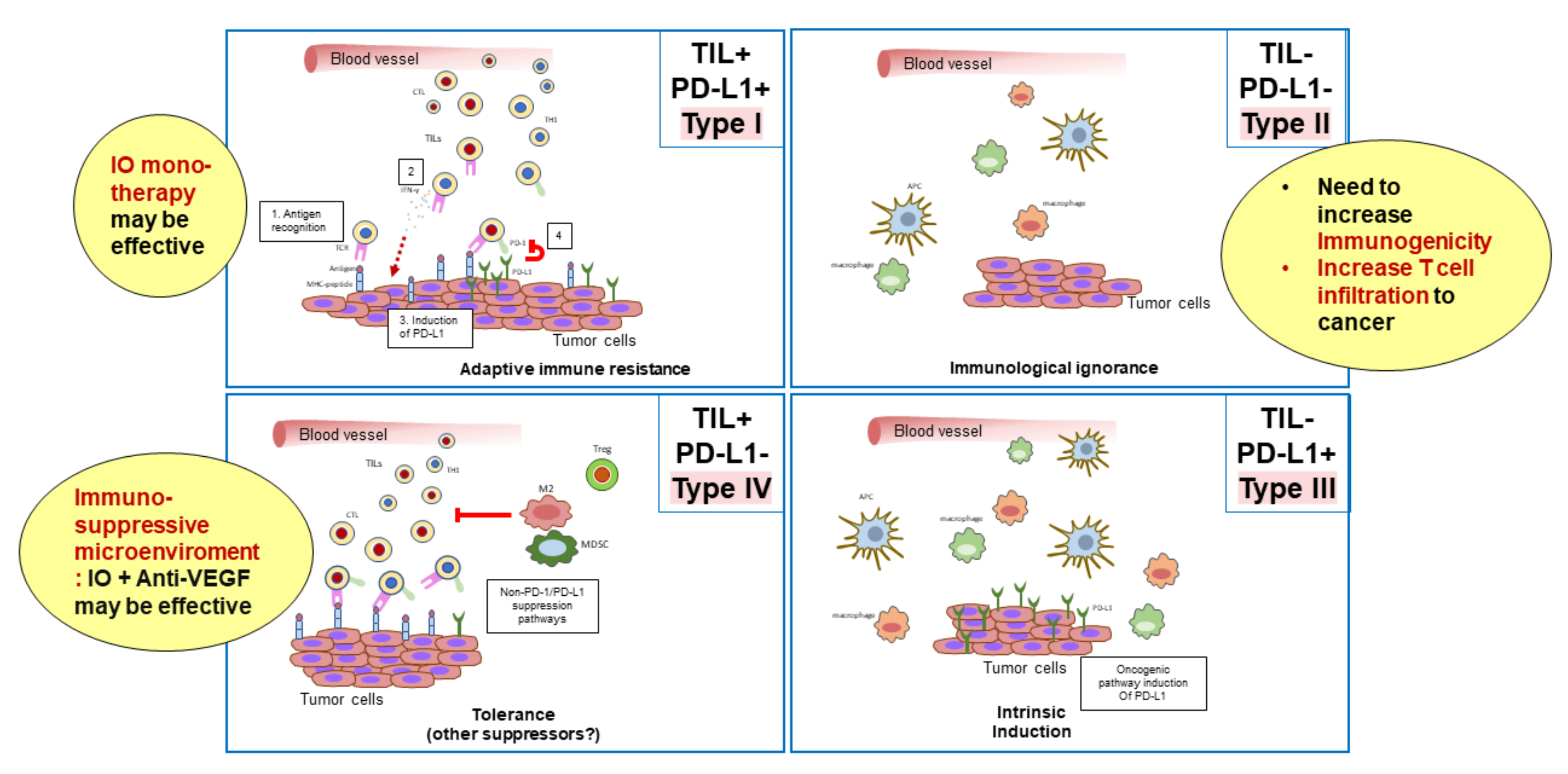
| Efficacy | Anti-PD-1 Monotherapy (Phase 3 Trial) | Anti-PD-1/PD-L1 plus TKI/Anti-VEGF (Phase 1b Trial) | |||||
|---|---|---|---|---|---|---|---|
| Nivolumab [34] (n = 214) | Pembrolizumab [36] (n = 278) | Atezolizumab + bevacizumab [33] (n = 104) | Pembrolizumab + Lenvatinib [42] (n = 67) | Camrelizumab + apatinib [44] (n = 18) | Avelumab + axitinib [45] (n = 22) | Nivolumab + Lenvatinib [43] (n = 24) | |
| ORR (95% CI) | 15% | 18.3% (14.0–23.4) | 36% (26–46) | 40.3% (28.5–53.0) | 38.9% | 13.6% (2.9–34.9) | 54.2% (32.8–74.4) |
| DCR (95% CI) | 55% | 62.2% | 71% | 85.1% (74.3–92.6) | 83.3% | 68.2% (45.1–86.1) | 91.7% (73.0–99.0) |
| PFS, months (95% CI) | 3.7 (3.1–3.9) | 3.0 (2.8–4.1) | 7.4 (5.6–10.7) | 9.7 (5.3–13.8) | 7.2 (2.6–NE) | 5.5 (1.9–7.4) | 7.4 (3.7–NE) |
| OS, months (95% CI) | 16.4 (13.9–18.4) | 13.9 (11.6–16.0) | 17.1 (13.8–NE) | 20.4 (11.0–NE) | NR | 12.7 (8.0–NE) | NR |
| DOR, months (M) | 23.3 (3.1–34.5+) | 13.8 (1.5–23.6) | NE (11.7–NE) | 11.0 (5.6–11.0) | NA | 5.5 (3.7–7.3) | NA |
| Target Population | Design | Trial Name | Result | Presentation | Publication | 1st Author | |
|---|---|---|---|---|---|---|---|
| Advanced | First line | 1. Sorafenib vs. Sunitinib | SUN1170 | Negative | ASCO 2011 | JCO 2013 | Cheng AL [47] |
| 2. Sorafenib ± Erlotinib | SEARCH | Negative | ESMO 2012 | JCO 2015 | Zhu AX [48] | ||
| 3. Sorafenib vs. Brivanib | BRISK-FL | Negative | AASLD 2012 | JCO 2013 | Johnson PJ [49] | ||
| 4. Sorafenib vs. Linifanib | LiGHT | Negative | ASCO-GI 2013 | JCO 2015 | Cainap C [50] | ||
| 5. Sorafenib ± Doxorubicin | CALGB 80802 | Negative | ASCO-GI 2016 | ||||
| 6. Sorafenib ±- HAIC | SILIUS | Negative | EASL 2016 | Lancet GH 2018 | Kudo M [51] | ||
| 7. Sorafenib ± Y90 | SARAH | Negative | EASL 2017 | Lancet-O 2017 | Vilgrain V [52] | ||
| 8. Sorafenib ± Y90 | SIRveNIB | Negative | ASCO 2017 | JCO 2018 | Chow PKH [53] | ||
| 9. Sorafenib vs. Lenvatinib | REFLECT | Positive | ASCO 2017 | Lancet 2018 | Kudo M [54] | ||
| 10. Sorafenib vs. Nivolumab | CheckMate-459 | Negative | ESMO 2019 | Yau T [34] | |||
| 11. Sorafenib ± Y90 | SORAMIC | Negative | EASL 2018 | J Hepatol 2019 | Ricke J [55] | ||
| 12. Sorafenib vs. Atezolizumab + Bevacizumab | IMbrave150 | Positive | ESMO-Asia 2019 | Cheng AL [1] | |||
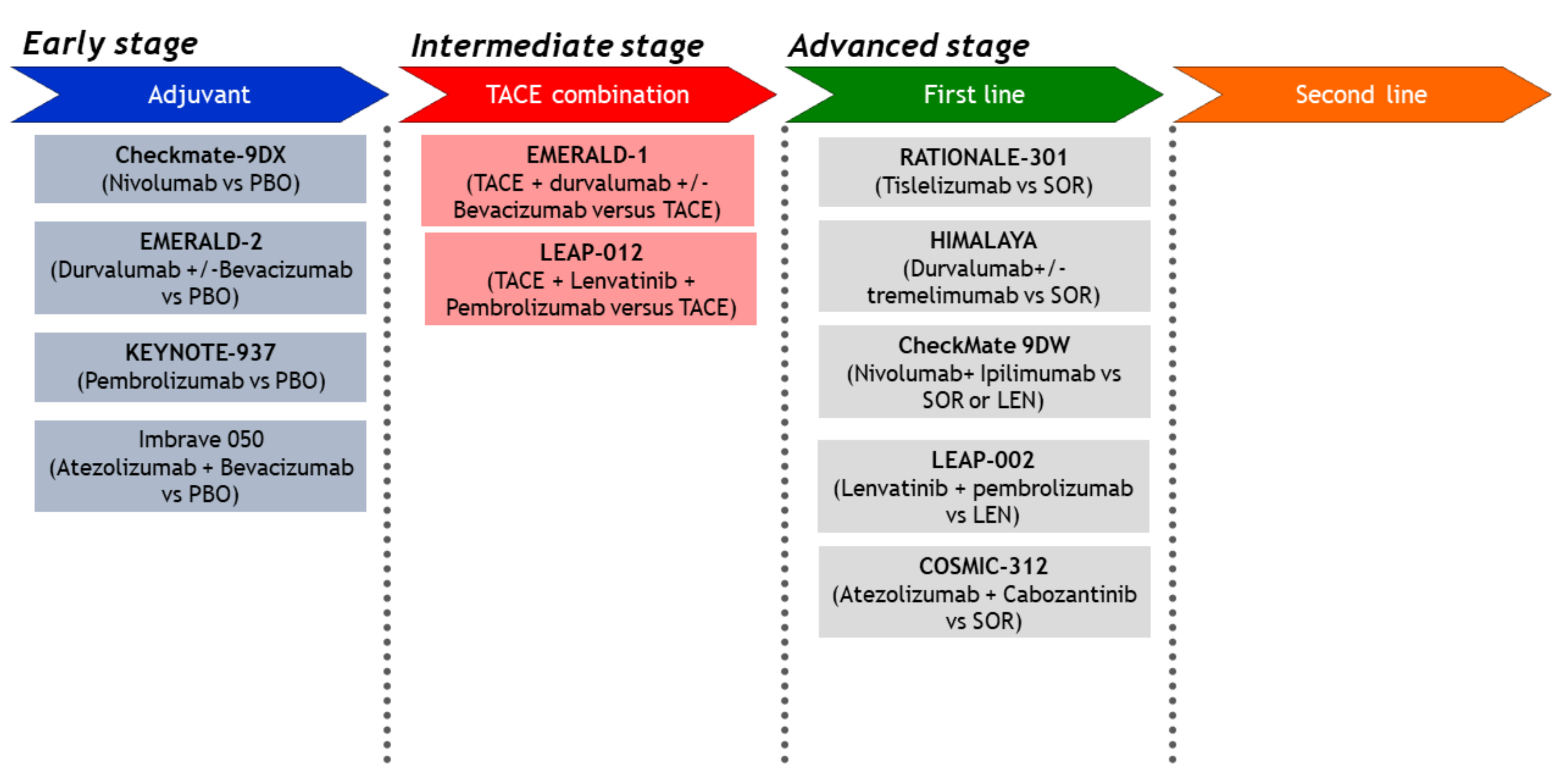
| 13. Sorafenib vs. Durvalumab + Tremelimumab vs. Durva | HIMALAYA | Ongoing | |||||
| 14. Sorafenib vs. Tislelizumab | Rationale301 | Ongoing | |||||
| 15. Lenvatinib ± Pembrolizumab | LEAP002 | Ongoing | |||||
| 16. Lenvatinib or Sorafenib vs. Nivolumab + Ipilimumab | CheckMate 9DW | Ongoing | |||||
| 17. Sorafenib vs. Atezolizumab + Cabozantinib | COSMIC-312 | Ongoing | |||||
| Second line | 1. Brivanib vs. Placebo | BRISK-PS | Negative | EASL 2012 | JCO 2013 | Llovet JM [56] | |
| 2. Everolimus vs. Placebo | EVOLVE-1 | Negative | ASCO-GI 2014 | JAMA 2014 | Zhu AX [57] | ||
| 3. Ramucirumab vs. Placebo | REACH | Negative | ESMO 2014 | Lancet-O 2015 | Zhu AX [58] | ||
| 4. S-1 vs. Placebo | S-CUBE | Negative | ASCO 2015 | Lancet GH 2017 | Kudo M [59] | ||
| 5. ADI-PEG 20 vs. Placebo | NA | Negative | ASCO 2016 | Ann Oncol 2018 | Abou-Alfa GK [60] | ||
| 6. Regorafenib vs. Placebo | RESORCE | Positive | WCGC 2016 | Lancet 2017 | Bruix J [61] | ||
| 7. Tivantinib vs. Placebo | METIV-HCC | Negative | ASCO 2017 | Lancet-O 2018 | Rimassa L [62] | ||
| 8. Tivantinib vs. Placebo | JET-HCC | Negative | ESMO 2017 | ||||
| 9. DT# vs. Placebo | ReLive | Negative | ILCA 2017 | Lancet Gastroenterol Hepatol | Merle P [63] | ||
| 10. Cabozantinib vs. Placebo | CELESTIAL | Positive | ASCO-GI 2018 | NEJM 2018 | Abou-Alfa G [64] | ||
| 11. Ramucirumab vs. Placebo | REACH-2 | Positive | ASCO 2018 | Lancet-O 2019 | Zhu AX [65] | ||
| 12. Pembrolizumab vs. Placebo | KEYNOTE-240 | Negative | ASCO 2019 | JCO 2020 | Finn RS [36] | ||
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kudo, M. Scientific Rationale for Combined Immunotherapy with PD-1/PD-L1 Antibodies and VEGF Inhibitors in Advanced Hepatocellular Carcinoma. Cancers 2020, 12, 1089. https://doi.org/10.3390/cancers12051089
Kudo M. Scientific Rationale for Combined Immunotherapy with PD-1/PD-L1 Antibodies and VEGF Inhibitors in Advanced Hepatocellular Carcinoma. Cancers. 2020; 12(5):1089. https://doi.org/10.3390/cancers12051089
Chicago/Turabian StyleKudo, Masatoshi. 2020. "Scientific Rationale for Combined Immunotherapy with PD-1/PD-L1 Antibodies and VEGF Inhibitors in Advanced Hepatocellular Carcinoma" Cancers 12, no. 5: 1089. https://doi.org/10.3390/cancers12051089
APA StyleKudo, M. (2020). Scientific Rationale for Combined Immunotherapy with PD-1/PD-L1 Antibodies and VEGF Inhibitors in Advanced Hepatocellular Carcinoma. Cancers, 12(5), 1089. https://doi.org/10.3390/cancers12051089





