Treatment of Combined Hepatocellular and Cholangiocarcinoma
Abstract
1. Introduction
2. Diagnosis
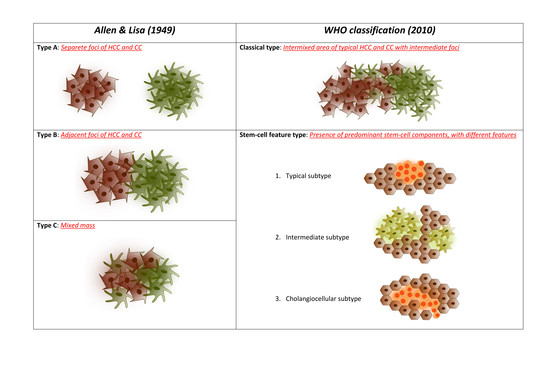
3. Pathological Classification and Molecular Alterations
4. Staging Systems
5. Surgical Treatments
5.1. Resection
5.2. Transplantation
6. Locoregional Treatments
7. Chemotherapy
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jarnagin, W.R.; Weber, S.; Tickoo, S.K.; Koea, J.B.; Obiekwe, S.; Fong, Y.; DeMatteo, R.P.; Blumgart, L.H.; Klimstra, D. Combined hepatocellular and cholangiocarcinoma: Demographic, clinical, and prognostic factors. Cancer 2002, 94, 2040–2046. [Google Scholar] [CrossRef]
- Ng, I.O.; Shek, T.W.; Nicholls, J.; Ma, L.T. Combined hepatocellular-cholangiocarcinoma: A clinicopathological study. J. Gastroenterol. Hepatol. 1998, 13, 34–40. [Google Scholar] [CrossRef]
- Goodman, Z.D.; Ishak, K.G.; Langloss, J.M.; Sesterhenn, I.A.; Rabin, L. Combined hepatocellular-cholangiocarcinoma: A histologic and immunohistochemical study. Cancer 1985, 55, 124–135. [Google Scholar] [CrossRef]
- Taguchi, J.; Nakashima, O.; Tanaka, M.; Hisaka, T.; Takazawa, T.; Kojiro, M. A clinicopathological study on combined hepatocellular and cholangiocarcinoma. J. Gastroenterol. Hepatol. 1996, 11, 758–764. [Google Scholar] [CrossRef]
- Heimbach, J.K.; Kulik, L.M.; Finn, R.S.; Sirlin, C.B.; Abecassis, M.M.; Roberts, L.R.; Zhu, A.X.; Murad, M.H.; Marrero, J.A. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 2018, 67, 358–380. [Google Scholar] [CrossRef]
- Galle, P.R.; Forner, A.; Llovet, J.M.; Mazzaferro, V.; Piscaglia, F.; Raoul, J.-L.; Schirmacher, P.; Vilgrain, V. EASL clinical practice guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef]
- Omata, M.; Cheng, A.-L.; Kokudo, N.; Kudo, M.; Lee, J.M.; Jia, J.; Tateishi, R.; Han, K.-H.; Chawla, Y.K.; Shiina, S.; et al. Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: A 2017 update. Hepatol. Int. 2017, 11, 317–370. [Google Scholar] [CrossRef]
- Kono, Y.; Lyshchik, A.; Cosgrove, D.; Dietrich, C.F.; Jang, H.-J.; Kim, T.K.; Piscaglia, F.; Willmann, J.K.; Wilson, S.R.; Santillan, C.; et al. Contrast enhanced ultrasound (CEUS) liver imaging reporting and data system (LI-RADS(R)): The official version by the American college of radiology (ACR). Ultraschall Med. 2017, 38, 85–86. [Google Scholar] [CrossRef]
- Elsayes, K.M.; Hooker, J.C.; Agrons, M.M.; Kielar, A.Z.; Tang, A.; Fowler, K.J.; Chernyak, V.; Bashir, M.R.; Kono, Y.; Do, R.K.; et al. 2017 version of LI-RADS for CT and MR imaging: An update. Radiographics 2017, 37, 1994–2017. [Google Scholar] [CrossRef]
- Sagrini, E.; Iavarone, M.; Stefanini, F.; Tovoli, F.; Vavassori, S.; Maggioni, M.; Renzulli, M.; Salvatore, V.; Stefanescu, H.; Colombo, M.; et al. Imaging of combined hepatocellular-cholangiocarcinoma in cirrhosis and risk of false diagnosis of hepatocellular carcinoma. United Eur. Gastroenterol. J. 2019, 7, 69–77. [Google Scholar] [CrossRef]
- Bosman, F.T.; Carneiro, F.; Hruban, R.H.; Theise, N.D. WHO Classification of Tumours of the Digestive System; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Wells, H. Primary carcinoma of the liver. Am. J. Sci. 1903, 126, 403–417. [Google Scholar] [CrossRef]
- Allen, R.A.; Lisa, J.R. Combined liver cell and bile duct carcinoma. Am. J. Pathol. 1949, 25, 647–655. [Google Scholar]
- Liver Cancer Study Group of Japan. The general rules for the clinical and pathological study of primary liver cancer. Jpn. J. Surg. 1989, 19, 98–129. [Google Scholar] [CrossRef]
- Roskams, T.A.; Libbrecht, L.; Desmet, V.J. Progenitor cells in diseased human liver. Semin. Liver Dis. 2003, 23, 385–396. [Google Scholar]
- Yeh, M.M. Pathology of combined hepatocellular-cholangiocarcinoma. J. Gastroenterol. Hepatol. 2010, 25, 1485–1492. [Google Scholar] [CrossRef]
- Yano, H.; Iemura, A.; Haramaki, M.; Momosaki, S.; Ogasawara, S.; Higaki, K.; Kojiro, M. A human combined hepatocellular and cholangiocarcinoma cell line (KMCH-2) that shows the features of hepatocellular carcinoma or cholangiocarcinoma under different growth conditions. J. Hepatol. 1996, 24, 413–422. [Google Scholar] [CrossRef]
- Fujii, H.; Zhu, X.G.; Matsumoto, T.; Inagaki, M.; Tokusashi, Y.; Miyokawa, N.; Fukusato, T.; Uekusa, T.; Takagaki, T.; Kadowaki, N.; et al. Genetic classification of combined hepatocellular-cholangiocarcinoma. Hum. Pathol. 2000, 31, 1011–1017. [Google Scholar] [CrossRef]
- Woo, H.G.; Lee, J.-H.; Yoon, J.-H.; Kim, C.Y.; Lee, H.-S.; Jang, J.J.; Yi, N.-J.; Suh, K.-S.; Lee, K.U.; Park, E.S.; et al. Identification of a cholangiocarcinoma-like gene expression trait in hepatocellular carcinoma. Cancer Res. 2010, 70, 3034–3041. [Google Scholar] [CrossRef]
- Xue, R.; Chen, L.; Zhang, C.; Fujita, M.; Li, R.; Yan, S.-M.; Ong, C.K.; Liao, X.; Gao, Q.; Sasagawa, S.; et al. Genomic and transcriptomic profiling of combined hepatocellular and intrahepatic cholangiocarcinoma reveals distinct molecular subtypes. Cancer Cell 2019, 35, 932–947. [Google Scholar] [CrossRef]
- Llovet, J.M.; Bru, C.; Bruix, J. Prognosis of hepatocellular carcinoma: The BCLC staging classification. Semin. Liver Dis. 1999, 19, 329–338. [Google Scholar] [CrossRef]
- Yau, T.; Tang, V.Y.F.; Yao, T.-J.; Fan, S.-T.; Lo, C.-M.; Poon, R.T.P. Development of Hong Kong liver cancer staging system with treatment stratification for patients with hepatocellular carcinoma. Gastroenterology 2014, 146, 1691–1700. [Google Scholar]
- Farges, O.; Fuks, D.; Le Treut, Y.-P.; Azoulay, D.; Laurent, A.; Bachellier, P.; Nuzzo, G.; Belghiti, J.; Pruvot, F.R.; Regimbeau, J.M. AJCC 7th edition of TNM staging accurately discriminates outcomes of patients with resectable intrahepatic cholangiocarcinoma: By the AFC-IHCC-2009 study group. Cancer 2011, 117, 2170–2177. [Google Scholar] [CrossRef]
- Nathan, H.; Aloia, T.A.; Vauthey, J.-N.; Abdalla, E.K.; Zhu, A.X.; Schulick, R.D.; Choti, M.A.; Pawlik, T.M. A proposed staging system for intrahepatic cholangiocarcinoma. Ann. Surg. Oncol. 2009, 16, 14–22. [Google Scholar] [CrossRef]
- Yamasaki, S. Intrahepatic cholangiocarcinoma: Macroscopic type and stage classification. J. Hepatobiliary Pancreat. Surg. 2003, 10, 288–291. [Google Scholar] [CrossRef]
- Tian, M.-X.; He, W.-J.; Liu, W.-R.; Yin, J.-C.; Jin, L.; Tang, Z.; Jiang, X.-F.; Wang, H.; Zhou, P.-Y.; Tao, C.-Y.; et al. A novel risk prediction model for patients with combined hepatocellular-cholangiocarcinoma. J. Cancer 2018, 9, 1025–1032. [Google Scholar] [CrossRef]
- Nakamura, S.; Suzuki, S.; Sakaguchi, T.; Serizawa, A.; Konno, H.; Baba, S.; Baba, S.; Muro, H. Surgical treatment of patients with mixed hepatocellular carcinoma and cholangiocarcinoma. Cancer 1996, 78, 1671–1676. [Google Scholar] [CrossRef]
- Koh, K.C.; Lee, H.; Choi, M.S.; Lee, J.H.; Paik, S.W.; Yoo, B.C.; Rhee, J.C.; Cho, J.W.; Park, C.K.; Kim, H.J. Clinicopathologic features and prognosis of combined hepatocellular cholangiocarcinoma. Am. J. Surg. 2005, 189, 120–125. [Google Scholar]
- Lee, W.-S.; Lee, K.-W.; Heo, J.-S.; Kim, S.-J.; Choi, S.-H.; Kim, Y.-I.; Joh, J.-W. Comparison of combined hepatocellular and cholangiocarcinoma with hepatocellular carcinoma and intrahepatic cholangiocarcinoma. Surg. Today 2006, 36, 892–897. [Google Scholar] [CrossRef]
- Zuo, H.-Q.; Yan, L.-N.; Zeng, Y.; Yang, J.-Y.; Luo, H.-Z.; Liu, J.-W.; Zhou, L.-X. Clinicopathological characteristics of 15 patients with combined hepatocellular carcinoma and cholangiocarcinoma. Hepatobiliary Pancreat. Dis. Int. 2007, 6, 161–165. [Google Scholar]
- Kim, K.H.; Lee, S.G.; Park, E.H.; Hwang, S.; Ahn, C.S.; Moon, D.B.; Ha, T.Y.; Song, G.W.; Jung, D.H.; Kim, K.M.; et al. Surgical treatments and prognoses of patients with combined hepatocellular carcinoma and cholangiocarcinoma. Ann. Surg. Oncol. 2009, 16, 623–629. [Google Scholar] [CrossRef]
- Panjala, C.; Senecal, D.L.; Bridges, M.D.; Kim, G.P.; Nakhleh, R.E.; Nguyen, J.H.H.; Harnois, D.M. The diagnostic conundrum and liver transplantation outcome for combined hepatocellular-cholangiocarcinoma. Am. J. Transpl. 2010, 10, 1263–1267. [Google Scholar] [CrossRef] [PubMed]
- Sapisochin, G.; Fidelman, N.; Roberts, J.P.; Yao, F.Y. Mixed hepatocellular cholangiocarcinoma and intrahepatic cholangiocarcinoma in patients undergoing transplantation for hepatocellular carcinoma. Liver Transpl. 2011, 17, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Chung, G.E.; Yu, S.J.; Hwang, S.Y.; Kim, J.S.; Kim, H.Y.; Yoon, J.-H.; Lee, H.-S.; Yi, N.-J.; Suh, K.-S.; et al. Long-term prognosis of combined hepatocellular and cholangiocarcinoma after curative resection comparison with hepatocellular carcinoma and cholangiocarcinoma. J. Clin. Gastroenterol. 2011, 45, 69–75. [Google Scholar] [PubMed]
- Yin, X.; Zhang, B.H.; Qiu, S.J.; Ren, Z.G.; Zhou, J.; Chen, X.H.; Zhou, Y.; Fan, J. Combined hepatocellular carcinoma and cholangiocarcinoma: Clinical features, treatment modalities, and prognosis. Ann. Surg. Oncol. 2012, 19, 2869–2876. [Google Scholar] [CrossRef]
- Groeschl, R.T.; Turaga, K.K.; Gamblin, T.C. Transplantation versus resection for patients with combined hepatocellular carcinoma-cholangiocarcinoma. J. Surg. Oncol. 2013, 107, 608–612. [Google Scholar] [CrossRef]
- Song, S.; Moon, H.H.; Lee, S.; Kim, T.S.; Shin, M.; Kim, J.M.; Park, J.B.; Kwon, C.H.D.; Kim, S.J.; Lee, S.K.; et al. Comparison between resection and transplantation in combined hepatocellular and cholangiocarcinoma. Transpl. Proc. 2013, 45, 3041–3046. [Google Scholar] [CrossRef]
- Garancini, M.; Goffredo, P.; Pagni, F.; Romano, F.; Roman, S.; Sosa, J.A.; Giardini, V. Combined hepatocellular-cholangiocarcinoma: A population-level analysis of an uncommon primary liver tumor. Liver Transpl. 2014, 20, 952–959. [Google Scholar] [CrossRef]
- Wu, D.; Shen, Z.Y.; Zhang, Y.M.; Wang, J.; Zheng, H.; Deng, Y.L.; Pan, C. Effect of liver transplantation in combined hepatocellular and cholangiocellular carcinoma: A case series. BMC Cancer 2015, 15, 232. [Google Scholar] [CrossRef]
- Jung, D.H.; Hwang, S.; Song, G.W.; Ahn, C.S.; Moon, D.B.; Kim, K.H.; Ha, T.Y.; Park, G.C.; Hong, S.M.; Kim, W.J.; et al. Longterm prognosis of combined hepatocellular carcinoma-cholangiocarcinoma following liver transplantation and resection. Liver Transpl. 2017, 23, 330–341. [Google Scholar] [CrossRef]
- Yoon, Y.-I.; Hwang, S.; Lee, Y.-J.; Kim, K.-H.; Ahn, C.-S.; Moon, D.-B.; Ha, T.-Y.; Song, G.-W.; Jung, D.-H.; Lee, J.-W.; et al. Postresection outcomes of combined hepatocellular carcinoma-cholangiocarcinoma, hepatocellular carcinoma and intrahepatic cholangiocarcinoma. J. Gastrointest. Surg. 2016, 20, 411–420. [Google Scholar] [CrossRef]
- Vilchez, V.; Shah, M.B.; Daily, M.F.; Pena, L.; Tzeng, C.W.D.; Davenport, D.; Hosein, P.J.; Gedaly, R.; Maynard, E. Long-term outcome of patients undergoing liver transplantation for mixed hepatocellular carcinoma and cholangiocarcinoma: An analysis of the UNOS database. HPB 2016, 18, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.W.; Chok, K.S.H. Importance of surgical margin in the outcomes of hepatocholangiocarcinoma. World J. Hepatol. 2017, 9, 635. [Google Scholar] [CrossRef]
- Sasaki, A.; Kawano, K.; Aramaki, M.; Ohno, T.; Tahara, K.; Takeuchi, Y.; Yoshida, T.; Kitano, S. Clinicopathologic study of mixed hepatocellular and cholangiocellular carcinoma: Modes of spreading and choice of surgical treatment by reference to macroscopic type. J. Surg. Oncol. 2001, 76, 37–46. [Google Scholar] [PubMed]
- Ercolani, G.; Grazi, G.L.; Ravaioli, M.; Grigioni, W.F.; Cescon, M.; Gardini, A.; Del Gaudio, M.; Cavallari, A. The role of lymphadenectomy for liver tumors: Further considerations on the appropriateness of treatment strategy. Ann. Surg. 2004, 239, 202–209. [Google Scholar] [PubMed]
- Tao, C.Y.; Liu, W.R.; Jin, L.; Tang, Z.; Tian, M.X.; Jiang, X.F.; Wang, H.; Zhou, P.Y.; Fang, Y.; Ding, Z.B.; et al. Surgical treatment of combined hepatocellular-cholangiocarcinoma is as effective in elderly patients as it is in younger patients: A propensity score matching analysis. J. Cancer 2018, 9, 1106–1112. [Google Scholar]
- Kassahun, W.T.; Hauss, J. Management of combined hepatocellular and cholangiocarcinoma. Int. J. Clin. Pract. 2008, 62, 1271–1278. [Google Scholar] [CrossRef]
- Tian, M.-X.; Luo, L.-P.; Liu, W.-R.; Deng, W.; Yin, J.-C.; Jin, L.; Jiang, X.-F.; Zhou, Y.-F.; Qu, W.-F.; Tang, Z.; et al. Development and validation of a prognostic score predicting recurrence in resected combined hepatocellular cholangiocarcinoma. Cancer Manag. Res. 2019, 11, 5187–5195. [Google Scholar] [CrossRef]
- Bruix, J.; Takayama, T.; Mazzaferro, V.; Chau, G.-Y.; Yang, J.; Kudo, M.; Cai, J.; Poon, R.T.; Han, K.-H.; Tak, W.Y.; et al. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): A phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2015, 16, 1344–1354. [Google Scholar]
- Primrose, J.N.; Fox, R.P.; Palmer, D.H.; Malik, H.Z.; Prasad, R.; Mirza, D.; Anthony, A.; Corrie, P.; Falk, S.; Finch-Jones, M.; et al. Capecitabine compared with observation in resected biliary tract cancer (BILCAP): A randomised, controlled, multicentre, phase 3 study. Lancet Oncol. 2019, 20, 663–673. [Google Scholar]
- Hashimoto, K.; Miller, C.M. Liver transplantation for intrahepatic cholangiocarcinoma. J. Hepatobiliary Pancreat. Sci. 2015, 22, 138–143. [Google Scholar] [CrossRef]
- Gupta, R.; Togashi, J.; Akamatsu, N.; Sakamoto, Y.; Kokudo, N. Impact of incidental/misdiagnosed intrahepatic cholangiocarcinoma and combined hepatocellular cholangiocarcinoma on the outcomes of liver transplantation: An institutional case series and literature review. Surg. Today 2017, 47, 908–917. [Google Scholar] [CrossRef]
- Chan, A.C.; Lo, C.M.; Ng, I.O.; Fan, S.T. Case report liver transplantation for combined hepatocellular cholangiocarcinoma case reports. Asian J. Surg. 2007, 30, 143–146. [Google Scholar] [CrossRef]
- Maganty, K.; Levi, D.; Moon, J.; Bejarano, P.A.; Arosemena, L.; Tzakis, A.; Martin, P. Combined hepatocellular carcinoma and intrahepatic cholangiocarcinoma: Outcome after liver transplantation. Dig. Dis. Sci. 2010, 55, 3597–3601. [Google Scholar] [CrossRef]
- Magistri, P.; Tarantino, G.; Serra, V.; Guidetti, C.; Ballarin, R.; Di, F. Liver transplantation and combined hepatocellular-cholangiocarcinoma: Feasibility and outcomes. Dig. Liver Dis. 2017, 49, 467–470. [Google Scholar] [CrossRef]
- Li, D.; Si, X.; Wang, S.; Zhou, Y. Long-term outcomes of combined hepatocellular-cholangiocarcinoma after hepatectomy or liver transplantation: A systematic review and meta-analysis. Hepatobiliary Pancreat. Dis. Int. 2019, 18, 12–18. [Google Scholar] [CrossRef]
- Kim, J.H.; Yoon, H.K.; Ko, G.Y.; Gwon, D.; Jang, C.S.; Song, H.Y.; Shin, J.H.; Sung, K.B. Nonresectable combined hepatocellular carcinoma and cholangiocarcinoma: Analysis of the response and prognostic factors after transcatheter arterial chemoembolization. Radiology 2010, 255, 270–277. [Google Scholar] [CrossRef]
- Fowler, K.; Saad, N.E.; Brunt, E.; Doyle, M.B.M.; Amin, M.; Vachharajani, N.; Tan, B.; Chapman, W.C. Biphenotypic primary liver carcinomas: Assessing outcomes of hepatic directed therapy. Ann. Surg. Oncol. 2015, 22, 4130–4137. [Google Scholar] [CrossRef]
- Na, S.K.; Choi, G.H.; Lee, H.C.; Shin, Y.M.; An, J.; Lee, D.; Shim, J.H.; Kim, K.M.; Lim, Y.S.; Chung, Y.H.; et al. The effectiveness of transarterial chemoembolization in recurrent hepatocellular-cholangiocarcinoma after resection. PLoS ONE 2018, 13, e0198138. [Google Scholar] [CrossRef]
- Rogers, J.E.; Bolonesi, R.M.; Rashid, A.; Elsayes, K.M.; Elbanan, M.G.; Law, L.; Kaseb, A.; Shroff, R.T. Systemic therapy for unresectable, mixed hepatocellular-cholangiocarcinoma: Treatment of a rare malignancy. J. Gastrointest. Oncol. 2017, 8, 347–351. [Google Scholar] [CrossRef]
- Kobayashi, S.; Terashima, T.; Shiba, S.; Yoshida, Y.; Yamada, I.; Iwadou, S.; Horiguchi, S.; Takahashi, H.; Suzuki, E.; Moriguchi, M.; et al. Multicenter retrospective analysis of systemic chemotherapy for unresectable combined hepatocellular and cholangiocarcinoma. Cancer Sci. 2018, 109, 2549–2557. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.-F.; De Oliveira, A.C.; Santoro, A.; Raoul, J.-L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Cheng, A.-L.; Kang, Y.-K.; Chen, Z.; Tsao, C.-J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.-S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: A phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Tovoli, F.; Ielasi, L.; Casadei-Gardini, A.; Granito, A.; Foschi, F.G.; Rovesti, G.; Negrini, G.; Orsi, G.; Renzulli, M.; Piscaglia, F. Management of adverse events with tailored sorafenib dosing prolongs survival of hepatocellular carcinoma patients. J. Hepatol. 2019, 71, 1175–1183. [Google Scholar] [CrossRef]
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N. Engl. J. Med. 2010, 362, 1273–1281. [Google Scholar] [CrossRef]
- Zaanan, A.; Williet, N.; Hebbar, M.; Dabakuyo, T.S.; Fartoux, L.; Mansourbakht, T.; Dubreuil, O.; Rosmorduc, O.; Cattan, S.; Bonnetain, F.; et al. Gemcitabine plus oxaliplatin in advanced hepatocellular carcinoma: A large multicenter AGEO study. J. Hepatol. 2013, 58, 81–88. [Google Scholar] [CrossRef]
- Zhu, A.X.; Blaszkowsky, L.S.; Ryan, D.P.; Clark, J.W.; Muzikansky, A.; Horgan, K.; Sheehan, S.; Hale, K.E.; Enzinger, P.C.; Bhargava, P.; et al. Phase II study of gemcitabine and oxaliplatin in combination with bevacizumab in patients with advanced hepatocellular carcinoma. J. Clin. Oncol. 2006, 24, 1898–1903. [Google Scholar] [CrossRef]
- Zhu, A.X.; Meyerhardt, J.A.; Blaszkowsky, L.S.; Kambadakone, A.R.; Muzikansky, A.; Zheng, H.; Clark, J.W.; Abrams, T.A.; Chan, J.A.; Enzinger, P.C.; et al. Efficacy and safety of gemcitabine, oxaliplatin, and bevacizumab in advanced biliary-tract cancers and correlation of changes in 18-fluorodeoxyglucose PET with clinical outcome: A phase 2 study. Lancet Oncol. 2010, 11, 48–54. [Google Scholar] [CrossRef]
- Hatano, H.; Kobayashi, S.; Nagano, H.; Tomokuni, A.; Tomimaru, Y.; Murakami, M.; Marubashi, S.; Eguchi, H.; Takeda, Y.; Tanemura, M.; et al. A case of successful multimodal treatment for combined hepatocellular and cholangiocarcinoma with portal venous tumor thrombus. Gan Kagaku Ryoho Cancer Chemother. 2009, 36, 2374–2376. [Google Scholar]
- Kitamura, T.; Okada, S.; Yamamoto, T. A case of combined hepatocellular and cholangiocarcinoma developed 6 years after sustained virological response of interferon for hepatitis C and beneficial effect of gemcitabine for lymph node metastases. Liver Cancer 2008, 14, 172–177. [Google Scholar]
- Shimizu, J.; Hayashi, S.; Dono, K.; Yasumoto, T.; Zenitani, M.; Munakata, K.; Watanabe, N.; Takamoto, K.; Kagawa, Y.; Hata, T.; et al. A case report of combined hepatocellular-cholangiocarcinoma whose lymph node recurrence effectively treated with UFT. Gan Kagaku Ryoho Cancer Chemother. 2009, 36, 2380–2382. [Google Scholar]
- Kim, G.M.; Jeung, H.-C.; Kim, D.; Kim, J.H.; Yoon, S.H.; Jung, E.S.; Shin, S.J. A case of combined hepatocellular-cholangiocarcinoma with favorable response to systemic chemotherapy. Cancer Res. Treat. 2010, 42, 235–238. [Google Scholar] [CrossRef]
- Chi, M.; Mikhitarian, K.; Shi, C.; Goff, L.W. Management of combined hepatocellular-cholangiocarcinoma: A case report and literature review. Gastrointest. Cancer Res. 2012, 5, 199–202. [Google Scholar]
- Salimon, M.; Prieux-Klotz, C.; Tougeron, D.; Hautefeuille, V.; Caulet, M.; Gournay, J.; Matysiak-Budnik, T.; Bennouna, J.; Tiako Meyo, M.; Lecomte, T.; et al. Gemcitabine plus platinum-based chemotherapy for first-line treatment of hepatocholangiocarcinoma: An AGEO French multicentre retrospective study. Br. J. Cancer 2018, 118, 325–330. [Google Scholar] [CrossRef]
- Trikalinos, N.A.; Zhou, A.; Doyle, M.B.M.; Fowler, K.J.; Morton, A.; Vachharajani, N.; Amin, M.; Keller, J.W.; Chapman, W.C.; Brunt, E.M.; et al. Systemic therapy for combined hepatocellular-cholangiocarcinoma: A single-institution experience. J. Natl. Compr. Cancer Netw. 2018, 16, 1193–1199. [Google Scholar] [CrossRef]
- Tovoli, F.; Negrini, G.; Benevento, F.; Faggiano, C.; Goio, E.; Granito, A. Systemic treatments for hepatocellular carcinoma: Challenges and future perspectives. Hepatic Oncol. 2018, 5, HEP01. [Google Scholar] [CrossRef]
- Seehawer, M.; D’Artista, L.; Zender, L. The worst from both worlds: cHCC-ICC. Cancer Cell 2019, 35, 823–824. [Google Scholar] [CrossRef]
- Rickman, D.S.; Beltran, H.; Demichelis, F.; Rubin, M.A. Biology and evolution of poorly differentiated neuroendocrine tumors. Nat. Med. 2017, 23, 1–10. [Google Scholar] [CrossRef]
- Tovoli, F.; Casadei-Gardini, A.; Benevento, F.; Piscaglia, F. Immunotherapy for hepatocellular carcinoma: A review of potential new drugs based on ongoing clinical studies as of 2019. Dig. Liver Dis. 2019, 51, 1067–1073. [Google Scholar] [CrossRef]
- Chen, W.-X.; Li, G.-X.; Hu, Z.-N.; Zhu, P.; Zhang, B.-X.; Ding, Z.-Y. Significant response to anti-PD-1 based immunotherapy plus lenvatinib for recurrent intrahepatic cholangiocarcinoma with bone metastasis: A case report and literature review. Medicine 2019, 98, e17832. [Google Scholar] [CrossRef]
- Cheng, A.-L.; Qin, S.; Ikeda, M.; Galle, P.; Ducreux, M.; Zhu, A.; Kim, T.-Y.; Kudo, M.; Breder, V.; Merle, P.; et al. LBA3IMbrave150: Efficacy and safety results from a ph III study evaluating atezolizumab (atezo) + bevacizumab (bev) vs sorafenib (Sor) as first treatment (tx) for patients (pts) with unresectable hepatocellular carcinoma (HCC). Ann. Oncol. 2019, 30, ix186–ix187. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Macarulla Mercade, T.; Javle, M.; Kelley, R.K.; Lubner, S.; Adeva, J.; Cleary, J.M.; Catenacci, D.V.; Borad, M.J.; Bridgewater, J.A.; et al. LBA10_PRClarIDHy: A global, phase III, randomized, double-blind study of ivosidenib (IVO) vs placebo in patients with advanced cholangiocarcinoma (CC) with an isocitrate dehydrogenase 1 (IDH1) mutation. Ann. Oncol. 2019, 30, v872–v873. [Google Scholar] [CrossRef]
| Authors | Reference | Study Design | Patients | Surgical Treatment | Median OS (Months) | 5-year OS (%) | Median DFS (Months) | 5-year DFS (%) | Total Recurrence (%) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Nakamura et al., 1996 | [27] | Case series | 6 | Limited resection | 4 (66.7%) | 52.5 | 60.0 | n/a | n/a | 4 (66.7) |
| Major resection | 2 (33.3%) | |||||||||
| Koh et al., 2005 | [28] | Retrospective | 24 | Hepatic resection | 37 | n/a | n/a | n/a | 14 (58.3) | |
| Lee et al., 2006 | [29] | Retrospective | 33 | Limited resection | 10 (30.3%) | 47.3 | n/a | 23.4 | n/a | 16 (48.5) |
| Major resection | 22 (66.7%) | |||||||||
| Liver transplantation | 1 (3%) | |||||||||
| Zuo et al., 2007 | [30] | Retrospective | 15 | Limited resection | 7 (46.7%) | n/a | 8.0 | n/a | n/a | n/a |
| Major resection | 8 (53.3%) | |||||||||
| Kim et al., 2009 | [31] | Retrospective | 29 | Limited resection | 12 (41.4 %) | 28.8 | n/a | n/a | n/a | 18 (62.1) |
| Major resection | 13 (44.8%) | |||||||||
| Liver transplantation | 4 (13.8%) | |||||||||
| Panjala et al., 2010 | [32] | Case series | 12 | Liver transplantation | 43.2 | 16.0 | 17.7 | n/a | 7 (58) | |
| Sapisochin et al., 2011 | [33] | Retrospective | 14 | Liver transplantation | 19.5 | n/a | 8 | n/a | 8 (57) | |
| Lee et al., 2011 | [34] | Retrospective | 30 | Limited resection | 24 (80%) | 18.3 | n/a | 5.4 | n/a | 26 (86.6) |
| Major resection | 6 (20%) | |||||||||
| Yin et al., 2012 | [35] | Retrospective | 113 | Hepatic resection | 103 (91.2%) | 16.5 | 36.4 | 8 | n/a | 67 (65) |
| Other | 10 (8.8%) | / | / | / | / | |||||
| Groeschl et al., 2013 | [36] | Retrospective | 54 | Hepatic resection | 35 (65%) | 36 | 28.0 | n/a | n/a | n/a |
| Liver transplantation | 19 (35%) | |||||||||
| Song et al., 2013 | [37] | Retrospective | 76 | Hepatic resection | 68 (89.5%) | n/a | 42.1 | n/a | 26.2 | 50 (73.5) |
| Liver transplantation | 8 (10.5%) | 50.0 | 37.5 | |||||||
| Garancini et al., 2014 | [38] | Retrospective | 465 | Limited resection | 35 (7.5%) | n/a | 10.5 | n/a | 17.8 | n/a |
| Major resection | 47 (10.1%) | |||||||||
| Liver transplantation | 61 (13.1%) | |||||||||
| Other | 322 (69.2%) | |||||||||
| Wu et al., 2015 | [39] | Retrospective | 21 | Liver transplantation | 23 | 39.0 | n/a | 30.0 | 8 (38.1) | |
| Jung et al., 2017 | [40] | Retrospective | 132 | Hepatic resection | 100 (75.7%) | n/a | 63.0 | n/a | n/a | 42 (42) |
| Liver transplantation | 32 (24.3%) | 66.0 | 12 (38) | |||||||
| Yoon et al., 2016 | [41] | Retrospective | 53 | Limited resection | 31 (58.5%) | ~24 | 30.5 | ~7 | n/a | 39 (75) |
| Major resection | 22 (41.5%) | |||||||||
| Vilchez et al., 2016 | [42] | Retrospective | 94 | Liver transplantation | 29 | 40.0 | n/a | n/a | n/a | |
| Ma et al., 2017 | [43] | Retrospective | 42 | Limited resection | 13 (30.9%) | 32 | 35.4 | 9 | 23.6 | 33 (78.6) |
| Major resection | 29 (69.1%) | |||||||||
| Authors | Reference | Study Design | Patients | Treatment | Median OS (Months) | 3-year OS (%) | Median PFS (Months) | 5-year PFS (%) | Tumour Progression (%) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Kim et al., 2010 | [57] | Retrospective | 50 | TACE | 12.3 | 16.0 | n/a | n/a | 15 (30) | |
| Fowler et al., 2015 | [58] | Retrospective | 79 | Locoregional | TACE (6) | 8.3 | n/a | 16 | n/a | 2 (30) |
| TARE (6) | 3 (50) | |||||||||
| HAI pump (6) | 0 | |||||||||
| Surgery | 33 | Not reported | Not reported | 15 (44) | ||||||
| Chemotherapy | 28 | Not reported | Not reported | Not reported | ||||||
| Na et al., 2018 | [59] | Retrospective | 42 | TACE | 32.6 | n/a | 3.4 | n/a | 37 (88.1) | |
| Authors | Reference | Study Design | N° Patient(s) | Regimen (1st Line) | Median OS (Months) | Median PFS (Months) | |
|---|---|---|---|---|---|---|---|
| Hatano et al., 2009 | [69] | Case report | 1 | S-1 | n/a | n/a | |
| Kitamura et al., 2008 | [70] | Case report | 1 | 5-FU + CDDP | 6 | / | |
| Shimizu et al., 2009 | [71] | Case report | 1 | UFT | 14 | / | |
| Kim et al., 2010 | [72] | Case report | 1 | Sorafenib | / | 2 | |
| Chi et al., 2012 | [73] | Case report | 1 | GEM + CDDP | / | 12 | |
| Rogers et al., 2017 | [60] | Case series | 7 | GEM | 1 (14.3%) | / | 3.6 |
| GEM + Beva | 2 (28.6%) | / | 4.8 | ||||
| GEM + CDDP | 1 (14.3%) | / | 17.0 | ||||
| Sorafenib | 3 (42.8%) | / | 2.7 | ||||
| Salimon et al., 2018 | [74] | Retrospective | 30 | GEM + OX | 18 (60%) | 16.2 | 9.0 |
| GEM + OX + Beva | 9 (30%) | ||||||
| GEM + CDDP | 3 (10%) | ||||||
| GEM + OX | 18 (60%) | ||||||
| Kobayashi et al., 2018 | [61] | Retrospective | 36 | GEM + CDDP | 12 (33.3%) | 10.2 | 3.0 |
| 5-FU + CDDP | 11 (30.5%) | 11.9 | 3.8 | ||||
| Sorafenib | 5 (13.8%) | 3.5 | 1.6 | ||||
| Other | 8 (22.2%) | 8.1 | 2.8 | ||||
| Trikalinos et al., 2018 | [75] | Retrospective | 68 | GEM + platinum drug (CDDP or OX) | 41 (60.3%) | 11.5 | 8.0 |
| GEM ± 5-FU | 16 (23.5%) | 11.7 | 6.6 | ||||
| Sorafenib | 7 (10.3%) | 9.6 | 4.8 | ||||
| Other | 4 (5.9%) | n/a | n/a | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leoni, S.; Sansone, V.; De Lorenzo, S.; Ielasi, L.; Tovoli, F.; Renzulli, M.; Golfieri, R.; Spinelli, D.; Piscaglia, F. Treatment of Combined Hepatocellular and Cholangiocarcinoma. Cancers 2020, 12, 794. https://doi.org/10.3390/cancers12040794
Leoni S, Sansone V, De Lorenzo S, Ielasi L, Tovoli F, Renzulli M, Golfieri R, Spinelli D, Piscaglia F. Treatment of Combined Hepatocellular and Cholangiocarcinoma. Cancers. 2020; 12(4):794. https://doi.org/10.3390/cancers12040794
Chicago/Turabian StyleLeoni, Simona, Vito Sansone, Stefania De Lorenzo, Luca Ielasi, Francesco Tovoli, Matteo Renzulli, Rita Golfieri, Daniele Spinelli, and Fabio Piscaglia. 2020. "Treatment of Combined Hepatocellular and Cholangiocarcinoma" Cancers 12, no. 4: 794. https://doi.org/10.3390/cancers12040794
APA StyleLeoni, S., Sansone, V., De Lorenzo, S., Ielasi, L., Tovoli, F., Renzulli, M., Golfieri, R., Spinelli, D., & Piscaglia, F. (2020). Treatment of Combined Hepatocellular and Cholangiocarcinoma. Cancers, 12(4), 794. https://doi.org/10.3390/cancers12040794