Elevated Tumor Lactate and Efflux in High-grade Prostate Cancer demonstrated by Hyperpolarized 13C Magnetic Resonance Spectroscopy of Prostate Tissue Slice Cultures
Abstract
1. Introduction
2. Results
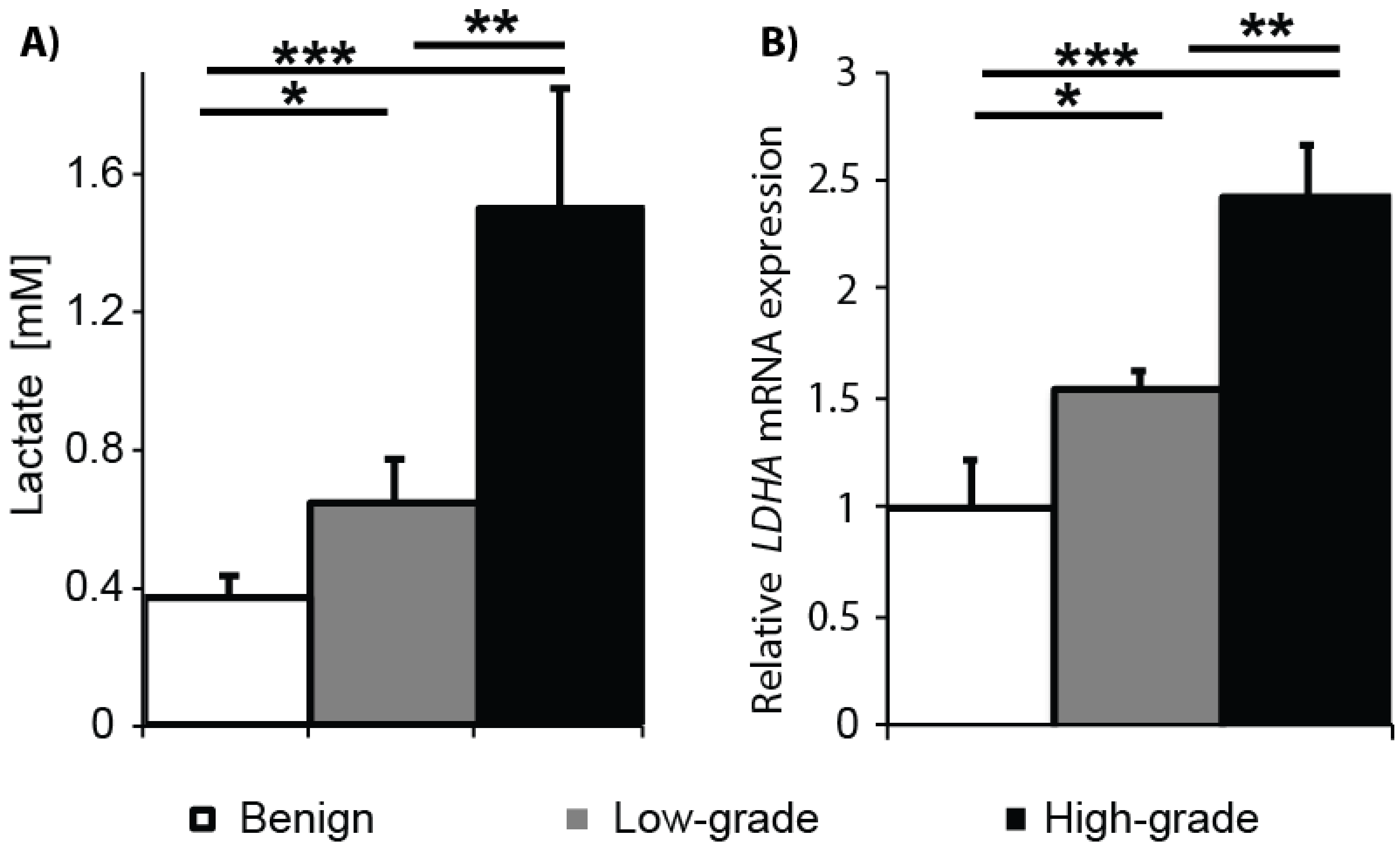
2.1. Gleason Grade-Dependent Increase in Lactate Concentration and LDHA Expression in Snap-Frozen Patient Biopsies
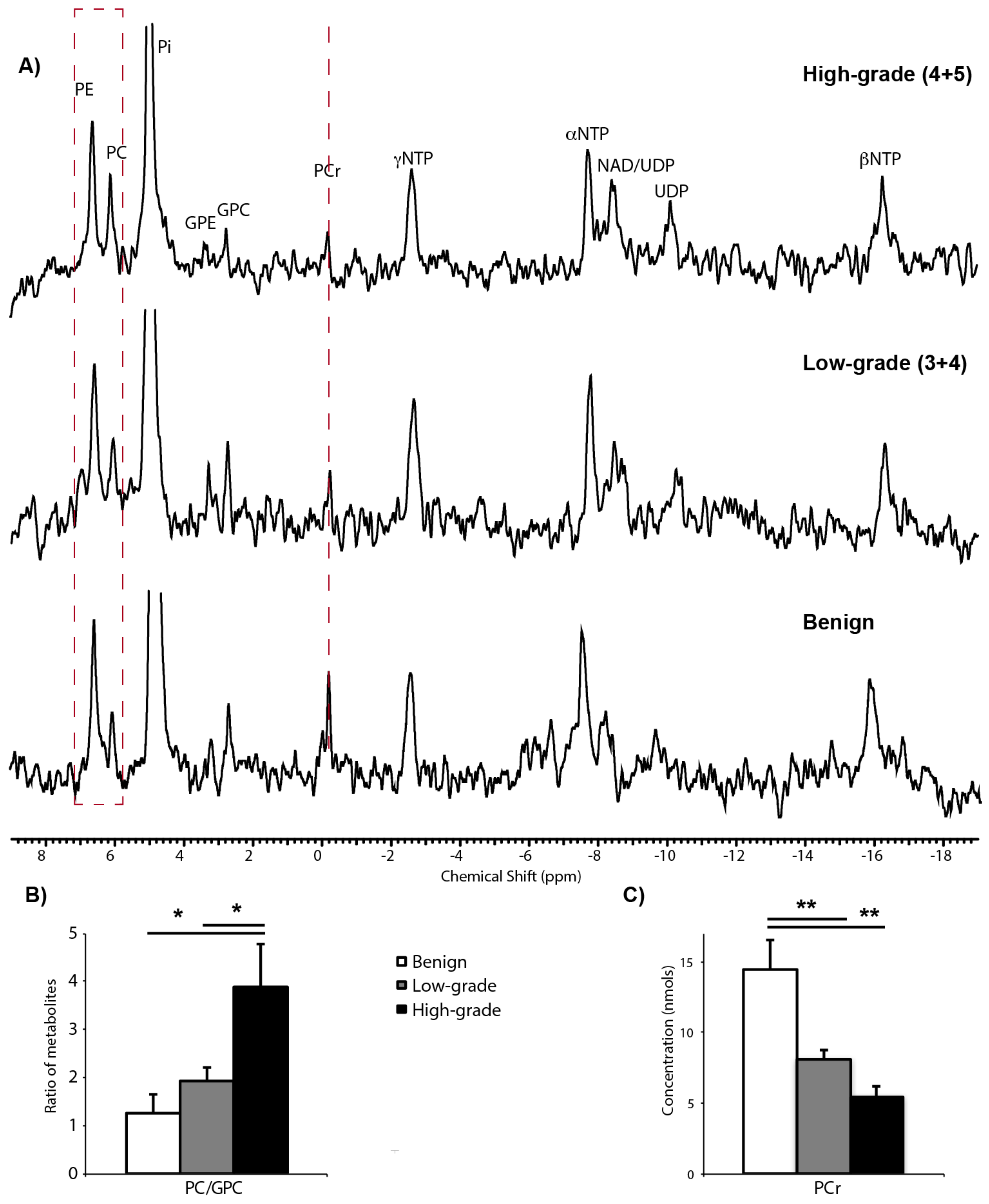
2.2. 31P Spectroscopy of TSCs: Tissue Viability and Grade-Dependent 31P Spectral Changes
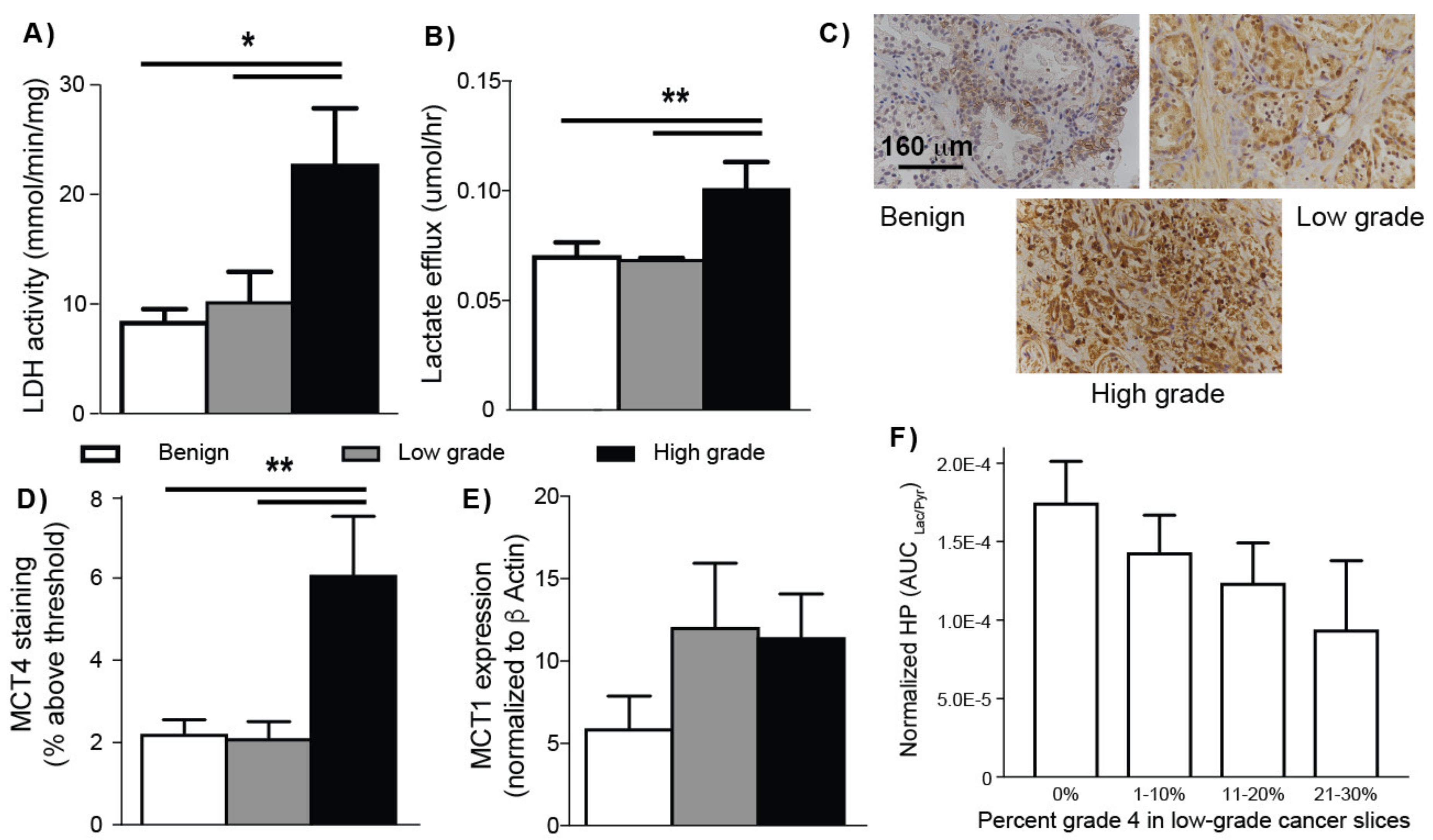
2.3. HP 13C MRS of TSCs: Gleason Grade-Dependent Increases in HP 13C Lactate Signal and Efflux
3. Discussion
4. Materials and Methods
4.1. Quantitative HR-MAS NMR Spectroscopy of Prostate Biopsies
4.2. Prostate Tissue Slice Acquisition and Culture
4.3. 3D Tissue Culture NMR-Compatible Bioreactor
4.4. NMR Spectroscopy Measurements of Prostate TSCs
4.5. Measurement of TSC Lactate Efflux Rate
4.6. Immunohistochemistry (IHC)
4.7. Image Analysis of MCT4 IHC
4.8. Biochemical and Molecular Assays
4.9. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kelloff, G.J.; Choyke, P.; Coffey, D.S. Prostate cancer imaging working group challenges in clinical prostate cancer: Role of imaging. AJR Am. J. Roentgenol. 2009, 192, 1455–1470. [Google Scholar] [CrossRef]
- Key Statistics for Prostate Cancer. Prostate Cancer Facts. Available online: https://www.cancer.org/cancer/prostate-cancer/about/key-statistics.html (accessed on 11 December 2019).
- Carroll, P.R.; Whitson, J.M.; Cooperberg, M.R. Serum prostate-specific antigen for the early detection of prostate cancer: Always, never, or only sometimes? J. Clin. Oncol. 2016, 29, 345–347. [Google Scholar] [CrossRef]
- Wilt, T.J.; MacDonald, R.; Rutks, I.; Shamliyan, T.A.; Taylor, B.C.; Kane, R.L. Systematic review: Comparative effectiveness and harms of treatments for clinically localized prostate cancer. Ann. Intern. Med. 2008, 148, 435–448. [Google Scholar] [CrossRef]
- Ganz, P.A.; Barry, J.M.; Burke, W.; Col, N.F.; Corso, P.S.; Dodson, E.; Hammond, M.E.; Kogan, B.A.; Lynch, C.F.; Newcomer, L.; et al. National Institutes of health state-of-the-science conference: Role of active surveillance in the management of men with localized prostate cancer. Ann. Intern. Med. 2012, 156, 591–595. [Google Scholar] [CrossRef] [PubMed]
- Moyer, V.A. Screening for prostate cancer: U.S. preventive services task force recommendation statement. Ann. Intern. Med. 2012, 157, 120–134. [Google Scholar] [CrossRef] [PubMed]
- Esserman, L.J.; Thompson, I.M.; Reid, B.; Nelson, P.; Ransohoff, D.F.; Welch, H.G.; Hwang, S.; Berry, D.A.; Kinzler, K.W.; Black, W.C.; et al. Addressing overdiagnosis and overtreatment in cancer: A prescription for change. Lancet Oncol. 2014, 15, e234–e242. [Google Scholar] [CrossRef]
- Loeb, S.; Bjurlin, M.A.; Nicholson, J.; Tammela, T.L.; Penson, D.F.; Carter, H.B.; Carroll, P.; Etzioni, R. Overdiagnosis and overtreatment of prostate cancer. Eur. Urol. 2014, 65, 1046–1055. [Google Scholar] [CrossRef]
- Heijnsdijk, E.A.M.; der Kinderen, A.; Wever, E.M.; Draisma, G.; Roobol, M.J.; de Koning, H.J. Overdetection, overtreatment and costs in prostate-specific antigen screening for prostate cancer. Br. J. Cancer 2009, 101, 1833–1838. [Google Scholar] [CrossRef]
- Klotz, L. Prostate cancer overdiagnosis and overtreatment. Curr. Opin. Endocrinol. Diabetes Obes. 2013, 20, 204–209. [Google Scholar] [CrossRef]
- Vickers, A.J.; Roobol, M.J.; Lilja, H. Screening for prostate cancer: Early detection or overdetection? Annu. Rev. Med. 2012, 63, 161–170. [Google Scholar] [CrossRef]
- Cooperberg, M.R.; Carroll, P.R. Trends in management for patients with localized prostate cancer, 1990–2013. JAMA 2015, 314, 80–82. [Google Scholar] [CrossRef] [PubMed]
- Cooperberg, M.R. Long-Term active surveillance for prostate cancer: Answers and questions. J. Clin. Oncol. 2015, 33, 238–240. [Google Scholar] [CrossRef] [PubMed]
- Cooperberg, M.R.; Broering, J.M.; Carroll, P.R. Risk assessment for prostate cancer metastasis and mortality at the time of diagnosis. J. Natl. Cancer Inst. 2009, 101, 878–887. [Google Scholar] [CrossRef]
- Cooperberg, M.R.; Broering, J.M.; Carroll, P.R. Time trends and local variation in primary treatment of localized prostate cancer. J. Clin. Oncol. 2010, 28, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Muller, B.G.; Fütterer, J.J.; Gupta, R.T.; Katz, A.; Kirkham, A.; Kurhanewicz, J.; Moul, J.W.; Pinto, P.A.; Rastinehad, A.R.; Robertson, C.; et al. The role of magnetic resonance imaging (MRI) in focal therapy for prostate cancer: Recommendations from a consensus panel. BJU Int. 2014, 113, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Costello, L.C.; Franklin, R.B. The genetic/metabolic transformation concept of carcinogenesis. Cancer Metastasis Rev. 2012, 31, 123–130. [Google Scholar] [CrossRef]
- Costello, L.C.; Franklin, R.B. The intermediary metabolism of the prostate: A key to understanding the pathogenesis and progression of prostate malignancy. Oncology 2000, 59, 269–282. [Google Scholar] [CrossRef]
- Keshari, K.R.; Sriram, R.; Van Criekinge, M.; Wilson, D.M.; Wang, Z.J.; Vigneron, D.B.; Peehl, D.M.; Kurhanewicz, J. Metabolic reprogramming and validation of hyperpolarized 13C lactate as a prostate cancer biomarker using a human prostate tissue slice culture bioreactor. Prostate 2013, 73, 1171–1181. [Google Scholar] [CrossRef]
- Kurhanewicz, J.; Vigneron, D.B. Magnetic Resonance Spectroscopy of Prostate Cancer; John Wiley & Sons Ltd.: Chichester, UK, 2007; Volume 64, pp. 923–944. [Google Scholar]
- Ardenkjaer-Larsen, J.H.; Fridlund, B.; Gram, A.; Hansson, G.; Hansson, L.; Lerche, M.H.; Servin, R.; Thaning, M.; Golman, K. Increase in signal-to-noise ratio of >10,000 times in liquid-state NMR. Proc. Natl. Acad. Sci. USA 2003, 100, 10158–10163. [Google Scholar] [CrossRef]
- Farsad, M.; Schiavina, R.; Franceschelli, A.; Sanguedolce, F.; Castellucci, P.; Bertaccini, A.; Brunocilla, E.; Manferrari, F.; Concetti, S.; Garofalo, M.; et al. Positron-Emission tomography in imaging and staging prostate cancer. Cancer Biomark 2008, 4, 277–284. [Google Scholar] [CrossRef]
- Beauregard, J.-M.; Williams, S.G.; DeGrado, T.R.; Roselt, P.; Hicks, R.J. Pilot comparison of 18F-fluorocholine and 18F-fluorodeoxyglucose PET/CT with conventional imaging in prostate cancer. J. Med. Imaging Radiat. Oncol. 2010, 54, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Albers, M.J.; Bok, R.; Chen, A.P.; Cunningham, C.H.; Zierhut, M.L.; Zhang, V.Y.; Kohler, S.J.; Tropp, J.; Hurd, R.E.; Yen, Y.-F.; et al. Hyperpolarized 13C lactate, pyruvate, and alanine: Noninvasive biomarkers for prostate cancer detection and grading. Cancer Res. 2008, 68, 8607–8615. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-Y.; Larson, P.E.Z.; Bok, R.A.; von Morze, C.; Sriram, R.; Delos Santos, R.; DeLos Santos, J.; Gordon, J.W.; Bahrami, N.; Ferrone, M.; et al. Assessing prostate cancer aggressiveness with hyperpolarized dual-agent 3D dynamic imaging of metabolism and perfusion. Cancer Res. 2017, 77, 3207–3216. [Google Scholar] [CrossRef] [PubMed]
- Priolo, C.; Pyne, S.; Rose, J.; Regan, E.R.; Zadra, G.; Photopoulos, C.; Cacciatore, S.; Schultz, D.; Scaglia, N.; McDunn, J.; et al. AKT1 and MYC induce distinctive metabolic fingerprints in human prostate cancer. Cancer Res. 2014, 74, 7198–7204. [Google Scholar] [CrossRef] [PubMed]
- Halestrap, A.P.; Price, N.T. The proton-linked monocarboxylate transporter (MCT) family: Structure, function and regulation. Biochem. J. 1999, 343 Pt 2, 281–299. [Google Scholar] [CrossRef]
- Baek, G.; Tse, Y.F.; Hu, Z.; Cox, D.; Buboltz, N.; McCue, P.; Yeo, C.J.; White, M.A.; DeBerardinis, R.J.; Knudsen, E.S.; et al. MCT4 Defines a glycolytic subtype of pancreatic cancer with poor prognosis and unique metabolic dependencies. Cell Rep. 2014, 9, 2233–2249. [Google Scholar] [CrossRef]
- Hao, J.; Chen, H.; Madigan, M.C.; Cozzi, P.J.; Beretov, J.; Xiao, W.; Delprado, W.J.; Russell, P.J.; Li, Y. Co-Expression of CD147 (EMMPRIN), CD44v3-10, MDR1 and monocarboxylate transporters is associated with prostate cancer drug resistance and progression. Br. J. Cancer 2010, 103, 1008–1018. [Google Scholar] [CrossRef]
- Pértega-Gomes, N.; Vizcaíno, J.R.; Miranda-Gonçalves, V.; Pinheiro, C.; Silva, J.; Pereira, H.; Monteiro, P.; Henrique, R.M.; Reis, R.M.; Lopes, C.; et al. Monocarboxylate transporter 4 (MCT4) and CD147 overexpression is associated with poor prognosis in prostate cancer. BMC Cancer 2011, 11, 312. [Google Scholar] [CrossRef]
- Maund, S.L.; Nolley, R.; Peehl, D.M. Optimization and comprehensive characterization of a faithful tissue culture model of the benign and malignant human prostate. Lab. Investig. 2013, 94, 208–221. [Google Scholar] [CrossRef]
- Ugurbil, K.; Rottenberg, H.; Glynn, P.; Shulman, R.G. 31P nuclear magnetic resonance studies of bioenergetics and glycolysis in anaerobic Escherichia coli cells. Proc. Natl. Acad. Sci. USA 1978, 75, 2244–2248. [Google Scholar] [CrossRef]
- Kurhanewicz, J.; Thomas, A.; Jajodia, P.; Weiner, M.W.; James, T.L.; Vigneron, D.B.; Narayan, P. 31P spectroscopy of the human prostate glandin vivo using a transrectal probe. Magn. Reson. Med. 1991, 22, 404–413. [Google Scholar] [CrossRef] [PubMed]
- Sriram, R.; Van Criekinge, M.; DeLos Santos, J.; Keshari, K.R.; Peehl, D.M.; Wang, Z.J. Non-Invasive differentiation of benign renal tumors from clear cell renal cell carcinomas using clinically translatable hyperpolarized 13C pyruvate magnetic resonance. Tomography 2016, 2, 35–42. [Google Scholar]
- Sriram, R.; Van Criekinge, M.; Hansen, A.; Wang, Z.J.; Vigneron, D.B.; Wilson, D.M.; Keshari, K.R.; Kurhanewicz, J. Real-Time measurement of hyperpolarized lactate production and efflux as a biomarker of tumor aggressiveness in an MR compatible 3D cell culture bioreactor. NMR Biomed. 2015, 28, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Keshari, K.R.; Sriram, R.; Koelsch, B.L.; Van Criekinge, M.; Wilson, D.M.; Kurhanewicz, J.; Wang, Z.J. Hyperpolarized 13C-pyruvate magnetic resonance reveals rapid lactate export in metastatic renal cell carcinomas. Cancer Res. 2013, 73, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Dimmer, K.S.; Friedrich, B.; Lang, F.; Deitmer, J.W.; Bröer, S. The low-affinity monocarboxylate transporter MCT4 is adapted to the export of lactate in highly glycolytic cells. Biochem. J. 2000, 350 Pt 1, 219–227. [Google Scholar] [CrossRef]
- Contreras-Baeza, Y.; Sandoval, P.Y.; Alarcón, R.; Galaz, A.; Cortés-Molina, F.; Alegría, K.; Baeza-Lehnert, F.; Arce-Molina, R.; Guequén, A.; Flores, C.A.; et al. Monocarboxylate transporter 4 (MCT4) is a high affinity transporter capable of exporting lactate in high-lactate microenvironments. J. Biol. Chem. 2019, 294, 20135–20147. [Google Scholar] [CrossRef]
- Ullah, M.S.; Davies, A.J.; Halestrap, A.P. The plasma membrane lactate transporter MCT4, but not MCT1, is up-regulated by hypoxia through a HIF-1alpha-dependent mechanism. J. Biol. Chem. 2006, 281, 9030–9037. [Google Scholar] [CrossRef]
- Tessem, M.-B.; Swanson, M.G.; Keshari, K.R.; Albers, M.J.; Joun, D.; Tabatabai, Z.L.; Simko, J.P.; Shinohara, K.; Nelson, S.J.; Vigneron, D.B.; et al. Evaluation of lactate and alanine as metabolic biomarkers of prostate cancer using 1H HR-MAS spectroscopy of biopsy tissues. Magn. Reson. Med. 2008, 60, 510–516. [Google Scholar] [CrossRef]
- Liu, J.; Chen, G.; Liu, Z.; Liu, S.; Cai, Z.; You, P.; Ke, Y.; Lai, L.; Huang, Y.; Gao, H.; et al. Aberrant FGFR tyrosine kinase signaling enhances the warburg effect by reprogramming LDH isoform expression and activity in prostate cancer. Cancer Res. 2018, 78, 4459–4470. [Google Scholar] [CrossRef]
- Bok, R.; Lee, J.; Sriram, R.; Keshari, K.; Sukumar, S.; Daneshmandi, S.; Korenchan, D.E.; Flavell, R.R.; Vigneron, D.B.; Kurhanewicz, J.; et al. The role of lactate metabolism in prostate cancer progression and metastases revealed by dual-agent hyperpolarized 13C MRSI. Cancers 2019, 11, 257. [Google Scholar] [CrossRef]
- Bovenzi, C.D.; Hamilton, J.; Tassone, P.; Johnson, J.; Cognetti, D.M.; Luginbuhl, A.; Keane, W.M.; Zhan, T.; Tuluc, M.; Bar-Ad, V.; et al. Prognostic indications of Elevated MCT4 and CD147 across cancer types: A meta-analysis. BioMed Res. Int. 2015, 2015, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.Y.C.; Xue, H.; Wu, R.; Fazli, L.; Lin, D.; Collins, C.C.; Gleave, M.E.; Gout, P.W.; Wang, Y. The MCT4 gene: A novel, potential target for therapy of advanced prostate cancer. Clin. Cancer Res. 2016, 22, 2721–2733. [Google Scholar] [CrossRef] [PubMed]
- Halestrap, A.P. The monocarboxylate transporter family—Structure and functional characterization. IUBMB Life 2011, 64, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Halestrap, A.P. The SLC16 gene family—Structure, role and regulation in health and disease. Mol. Asp. Med. 2013, 34, 337–349. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.S.; Graham, N.A.; Gu, W.; Espindola Camacho, C.; Mah, V.; Maresh, E.L.; Alavi, M.; Bagryanova, L.; Krotee, P.A.L.; Gardner, B.K.; et al. MCT1 modulates cancer cell pyruvate export and growth of tumors that co-express MCT1 and MCT4. Cell Rep. 2016, 14, 1590–1601. [Google Scholar] [CrossRef] [PubMed]
- Polanski, R.; Hodgkinson, C.L.; Fusi, A.; Nonaka, D.; Priest, L.; Kelly, P.; Trapani, F.; Bishop, P.W.; White, A.; Critchlow, S.E.; et al. Activity of the monocarboxylate transporter 1 inhibitor AZD3965 in small cell lung cancer. Clin. Cancer Res. 2014, 20, 926–937. [Google Scholar] [CrossRef]
- Sriram, R.; Gordon, J.; Baligand, C.; Ahamed, F.; DeLos Santos, J.; Qin, H.; Bok, R.A.; Vigneron, D.B.; Kurhanewicz, J.; Larson, P.E.Z.; et al. Non-Invasive assessment of lactate production and compartmentalization in renal cell carcinomas using hyperpolarized 13C pyruvate MRI. Cancers 2018, 10, 313. [Google Scholar] [CrossRef]
- Servoll, E.; Sæter, T.; Vlatkovic, L.; Lund, T.; Nesland, J.; Waaler, G.; Axcrona, K.; Beisland, H.O. Impact of a tertiary Gleason pattern 4 or 5 on clinical failure and mortality after radical prostatectomy for clinically localised prostate cancer. BJU Int. 2011, 109, 1489–1494. [Google Scholar] [CrossRef]
- Pierorazio, P.M.; Walsh, P.C.; Partin, A.W.; Epstein, J.I. Prognostic Gleason grade grouping: Data based on the modified Gleason scoring system. BJU Int. 2013, 111, 753–760. [Google Scholar] [CrossRef]
- Keshari, K.R.; Tsachres, H.; Iman, R.; Delos Santos, L.; Tabatabai, Z.L.; Shinohara, K.; Vigneron, D.B.; Kurhanewicz, J. Correlation of phospholipid metabolites with prostate cancer pathologic grade, proliferative status and surgical stage—Impact of tissue environment. NMR Biomed. 2011, 24, 691–699. [Google Scholar] [CrossRef]
- Kurhanewicz, J.; Swanson, M.G.; Vigneron, D.B. Progress of nuclear magnetic resonance spectroscopy in the study of prostate diseases. In Nuclear Magnetic Resonance Spectroscopy in the Study of Neoplastic Tissue; Tosi, R., Tugnoli, V., Eds.; Nova Science Publishers: Hauppauge, NY, USA, 2005. [Google Scholar]
- Nelson, S.J.; Kurhanewicz, J.; Vigneron, D.B.; Larson, P.E.Z.; Harzstark, A.L.; Ferrone, M.; Van Criekinge, M.; Chang, J.W.; Bok, R.; Park, I.; et al. Metabolic imaging of patients with prostate cancer using hyperpolarized [1-13C]Pyruvate. Sci. Transl. Med. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Schilling, F.; Düwel, S.; Köllisch, U.; Durst, M.; Schulte, R.F.; Glaser, S.J.; Haase, A.; Otto, A.M.; Menzel, M.I. Diffusion of hyperpolarized (13) C-metabolites in tumor cell spheroids using real-time NMR spectroscopy. NMR Biomed. 2013, 26, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Søgaard, L.V.; Schilling, F.; Janich, M.A.; Menzel, M.I.; Ardenkjaer-Larsen, J.H. In Vivo measurement of apparent diffusion coefficients of hyperpolarized 13C-labeled metabolites. NMR Biomed. 2014, 27, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Koelsch, B.L.; Keshari, K.R.; Peeters, T.H.; Larson, P.E.Z.; Wilson, D.M.; Kurhanewicz, J. Diffusion MR of hyperpolarized 13C molecules in solution. Analyst 2013, 138, 1011–1014. [Google Scholar] [CrossRef] [PubMed]
- Koelsch, B.L.; Reed, G.D.; Keshari, K.R.; Chaumeil, M.M.; Bok, R.; Ronen, S.M.; Vigneron, D.B.; Kurhanewicz, J.; Larson, P.E.Z. Rapid In Vivo apparent diffusion coefficient mapping of hyperpolarized (13) C metabolites. Magn. Reson. Med. 2015, 74, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Feuerecker, B.; Durst, M.; Michalik, M.; Schneider, G.; Saur, D.; Menzel, M.; Schwaiger, M.; Schilling, F. Hyperpolarized 13C diffusion MRS of co-polarized Pyruvate and Fumarate to measure lactate export and necrosis. J. Cancer 2017, 8, 3078–3085. [Google Scholar] [CrossRef] [PubMed]
- Albers, M.J.; Butler, T.N.; Rahwa, I.; Bao, N.; Keshari, K.R.; Swanson, M.G.; Kurhanewicz, J. Evaluation of the ERETIC method as an improved quantitative reference for 1H HR-MAS spectroscopy of prostate tissue. Magn. Reson. Med. 2009, 61, 525–532. [Google Scholar] [CrossRef]
- Zhao, H.; Nolley, R.; Chen, Z.; Peehl, D.M. Tissue slice grafts: An In Vivo model of human prostate androgen signaling. Am. J. Pathol. 2010, 177, 229–239. [Google Scholar] [CrossRef]
- Keshari, K.R.; Wilson, D.M.; Van Criekinge, M.; Sriram, R.; Koelsch, B.L.; Wang, Z.J.; VanBrocklin, H.F.; Peehl, D.M.; O’Brien, T.; Sampath, D.; et al. Metabolic response of prostate cancer to nicotinamide phophoribosyltransferase inhibition in a hyperpolarized MR/PET compatible bioreactor. Prostate 2015, 75, 1601–1609. [Google Scholar] [CrossRef][Green Version]
- Hill, D.K.; Orton, M.R.; Mariotti, E.; Boult, J.K.R.; Panek, R.; Jafar, M.; Parkes, H.G.; Jamin, Y.; Miniotis, M.F.; Al-Saffar, N.M.S.; et al. Model free approach to kinetic analysis of real-time hyperpolarized 13C magnetic resonance spectroscopy data. PLoS ONE 2013, 8, e71996. [Google Scholar] [CrossRef]
- Larson, P.E.Z.; Chen, H.-Y.; Gordon, J.W.; Korn, N.; Maidens, J.; Arcak, M.; Tang, S.; Criekinge, M.; Carvajal, L.; Mammoli, D.; et al. Investigation of analysis methods for hyperpolarized 13C-pyruvate metabolic MRI in prostate cancer patients. NMR Biomed. 2018, 31, e3997. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.I.; Egevad, L.; Amin, M.B.; Delahunt, B.; Srigley, J.R.; Humphrey, P.A. The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason grading of prostatic carcinoma. Am. J. Surg. Pathol. 2015, 1, 244–252. [Google Scholar] [CrossRef] [PubMed]

| ID | Age | Pre-op PSA ng/mL | Stage | Gleason Score | % Cancer Cells | % Epithelial Cells | % Stroma |
|---|---|---|---|---|---|---|---|
| 1 | 55 | 5.2 | pT2c pN0 | 3 + 3 | 7 | 37 | 57 |
| 2 | 53 | 5 | pT2a | 3 + 4 | 53 | 21 | 25 |
| 3 | 63 | 6.3 | pT2c pN0 | 3 + 4 | 19 | 27 | 54 |
| 4 | 56 | 7.6 | pT2a | N | -- | 40 | 60 |
| 5 | 50 | 4.5 | pT2c | N | -- | 40 | 60 |
| 6 | 65 | 7.1 | pT2c | N | -- | 46 | 53 |
| 7 | 54 | 6.9 | pT2c | 3 + 4 | 84 | 2 | 15 |
| 8 | 67 | 3.1 | pT2b | N | -- | 36 | 65 |
| 9 | 60 | 6.6 | pT2c | N | -- | 45 | 53 |
| 10 | 71 | 6.9 | pT3b | 3 + 4 | 27 | 36 | 38 |
| 11 | 71 | 6.9 | pT3b | 4 + 5 | 33 | 36 | 31 |
| 12 | 55 | 5.4 | pT3a | 4 + 5 | 31 | 29 | 41 |
| 13 | 55 | 5.4 | pT3a | N | -- | 37 | 63 |
| 14 | 61 | 6.8 | pT2c | 3 + 4 | 31 | 33 | 36 |
| 15 | 54 | 4.7 | pT2c | 3 + 4 | 49 | 33 | 19 |
| 16 | 63 | 4.1 | pT3a | 3 + 4 | 44 | 16 | 41 |
| 17 | 71 | 5 | pT3b pN0 | 3 + 4 | 11 | 48 | 43 |
| 18 | 52 | 15.8 | pT3b | 3 + 3 | 26 | 43 | 31 |
| 19 | 52 | 6.3 | pT3a | 3 + 4 | 20 | 20 | 60 |
| 20 | 71 | 8.1 | pT3a | 3 + 4 | 48 | 16 | 36 |
| 21 | 51 | 11.1 | pT3b pN0 | 3 + 3 | 6 | 19 | 74 |
| 22 | 67 | 4.2 | pT2c | N | -- | 28 | 72 |
| 23 | 67 | 3.5 | pT2c pN0 | 3 + 4 | 16 | 32 | 52 |
| 24 | 72 | 5.8 | pT3a | N | -- | 20 | 80 |
| 25 | 69 | 5 | pT2c | N | -- | 44 | 56 |
| 26 | 61 | 7.87 | pT2c | 3 + 3 | 42 | 27 | 32 |
| 27 | 64 | 24.37 | pT3a | 4 + 5 | 40 | 18 | 43 |
| 30 | 65 | 2.11 | pT3a pN0 | 3 + 4 | 2 | 41 | 58 |
| 29 | 65 | 2.56 | pT3a | N | -- | 40 | 61 |
| 31 | 56 | 7.5 | pT2c | N | -- | 34 | 67 |
| 32 | 56 | 4.5 | pT2c | N | -- | 47 | 53 |
| 33 | 58 | 13.9 | pT3a pN0 | 3 + 3 | 1 | 34 | 65 |
| 34 | 66 | 20.6 | pT3b | 4 + 5 | 31 | 8 | 61 |
| 35 | 69 | 13.8 | pT3b NX | 4 + 4 | 8 | 23 | 68 |
| 36 | 71 | 7.2 | pT3a | 3 + 4 | 35 | 25 | 40 |
| 37 | 61 | 5.6 | pT2c | 4 + 3 | 48 | 3 | 51 |
| 38 | 68 | 8.4 | pT3a pN0 | 3 + 4 | 18 | 33 | 49 |
| 39 | 71 | 5.9 | pT3a pN0 | 3 + 4 | 11 | 35 | 54 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sriram, R.; Van Criekinge, M.; DeLos Santos, J.; Ahamed, F.; Qin, H.; Nolley, R.; DeLos Santos, R.; Tabatabai, Z.L.; Bok, R.A.; Keshari, K.R.; et al. Elevated Tumor Lactate and Efflux in High-grade Prostate Cancer demonstrated by Hyperpolarized 13C Magnetic Resonance Spectroscopy of Prostate Tissue Slice Cultures. Cancers 2020, 12, 537. https://doi.org/10.3390/cancers12030537
Sriram R, Van Criekinge M, DeLos Santos J, Ahamed F, Qin H, Nolley R, DeLos Santos R, Tabatabai ZL, Bok RA, Keshari KR, et al. Elevated Tumor Lactate and Efflux in High-grade Prostate Cancer demonstrated by Hyperpolarized 13C Magnetic Resonance Spectroscopy of Prostate Tissue Slice Cultures. Cancers. 2020; 12(3):537. https://doi.org/10.3390/cancers12030537
Chicago/Turabian StyleSriram, Renuka, Mark Van Criekinge, Justin DeLos Santos, Fayyaz Ahamed, Hecong Qin, Rosalie Nolley, Romelyn DeLos Santos, Z. Laura Tabatabai, Robert A. Bok, Kayvan R. Keshari, and et al. 2020. "Elevated Tumor Lactate and Efflux in High-grade Prostate Cancer demonstrated by Hyperpolarized 13C Magnetic Resonance Spectroscopy of Prostate Tissue Slice Cultures" Cancers 12, no. 3: 537. https://doi.org/10.3390/cancers12030537
APA StyleSriram, R., Van Criekinge, M., DeLos Santos, J., Ahamed, F., Qin, H., Nolley, R., DeLos Santos, R., Tabatabai, Z. L., Bok, R. A., Keshari, K. R., Vigneron, D. B., Peehl, D. M., & Kurhanewicz, J. (2020). Elevated Tumor Lactate and Efflux in High-grade Prostate Cancer demonstrated by Hyperpolarized 13C Magnetic Resonance Spectroscopy of Prostate Tissue Slice Cultures. Cancers, 12(3), 537. https://doi.org/10.3390/cancers12030537





