Cancer Stem Cell Marker DCLK1 Correlates with Tumorigenic Immune Infiltrates in the Colon and Gastric Adenocarcinoma Microenvironments
Abstract
1. Introduction
2. Results
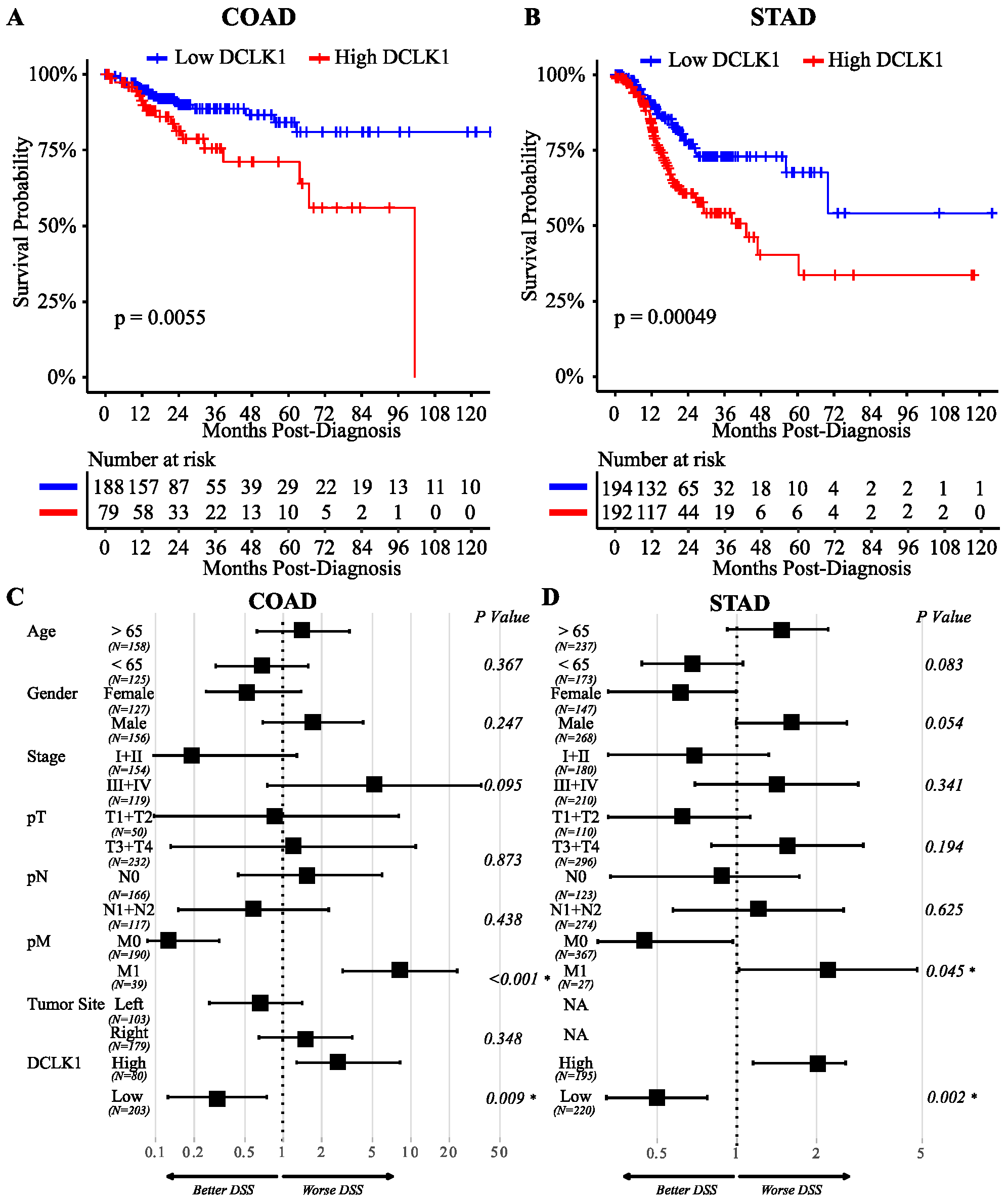
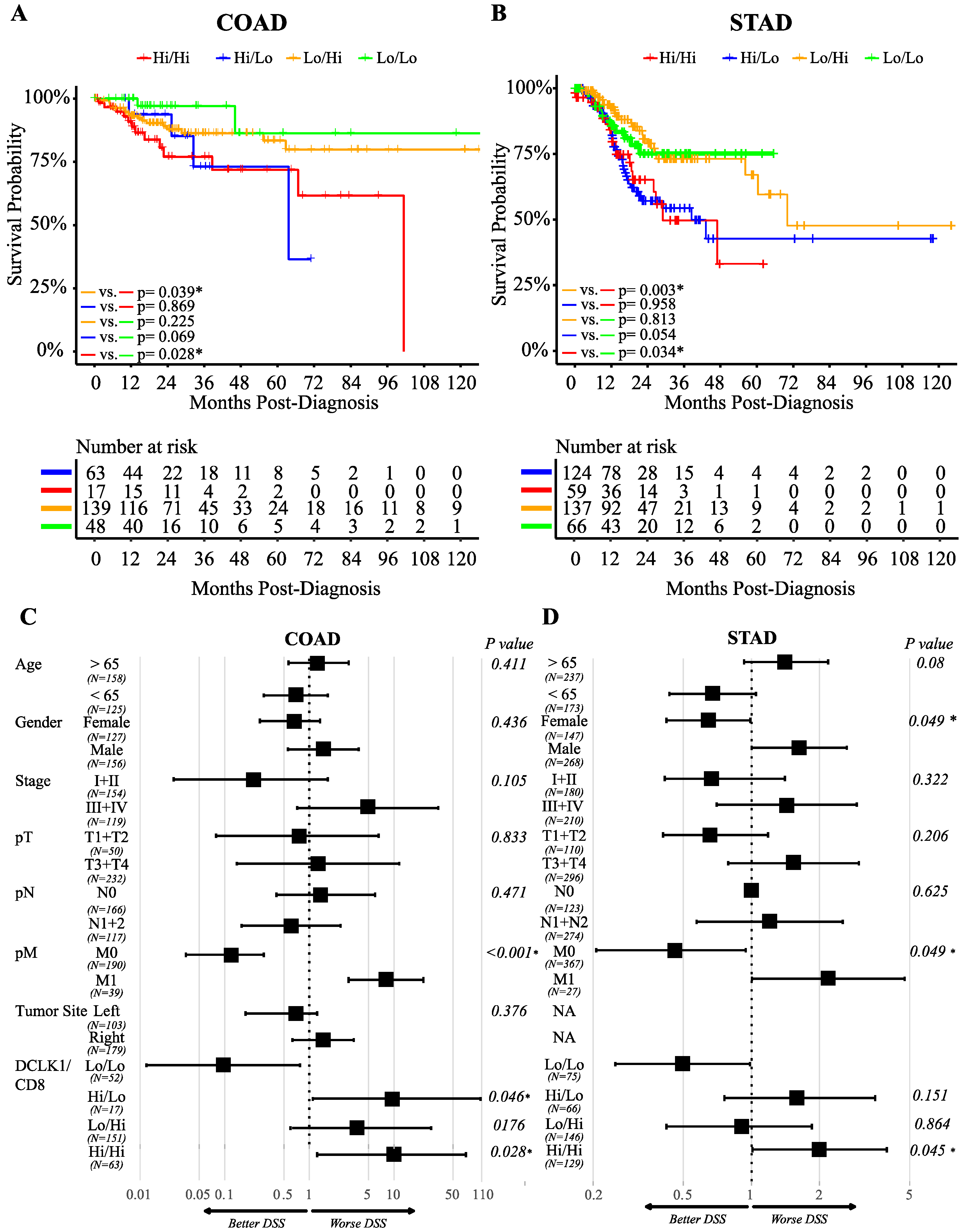
2.1. High Expression of DCLK1 is an Independent Prognostic Factor in Colon and Gastric Adenocarcinoma
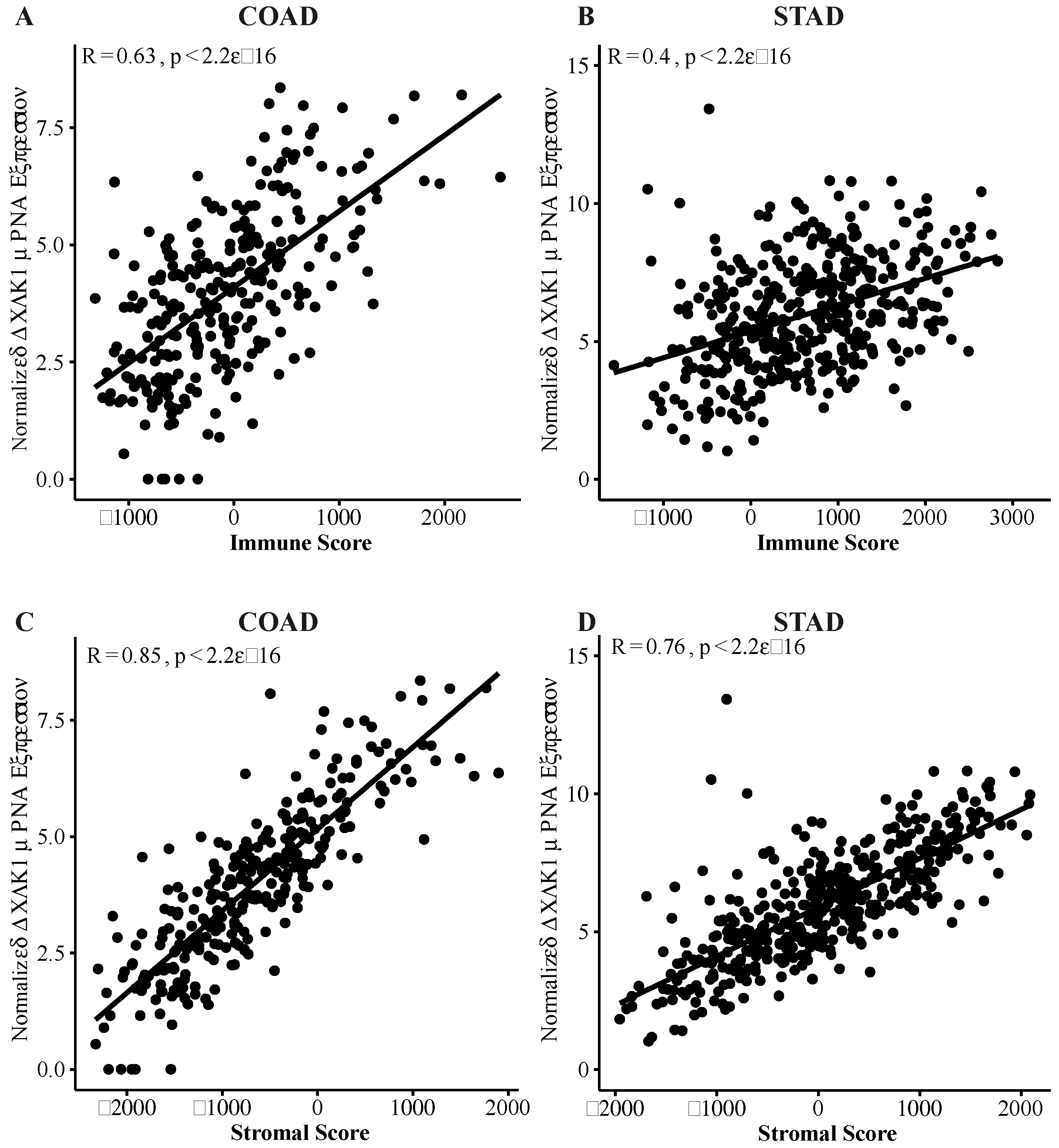
2.2. DCLK1 Expression Levels Significantly Correlate with TME Immune and Stromal Scores
2.3. DCLK1 Expression Level is Correlated with Various Immune Cell Subtypes in Both Colon and Stomach Cancer
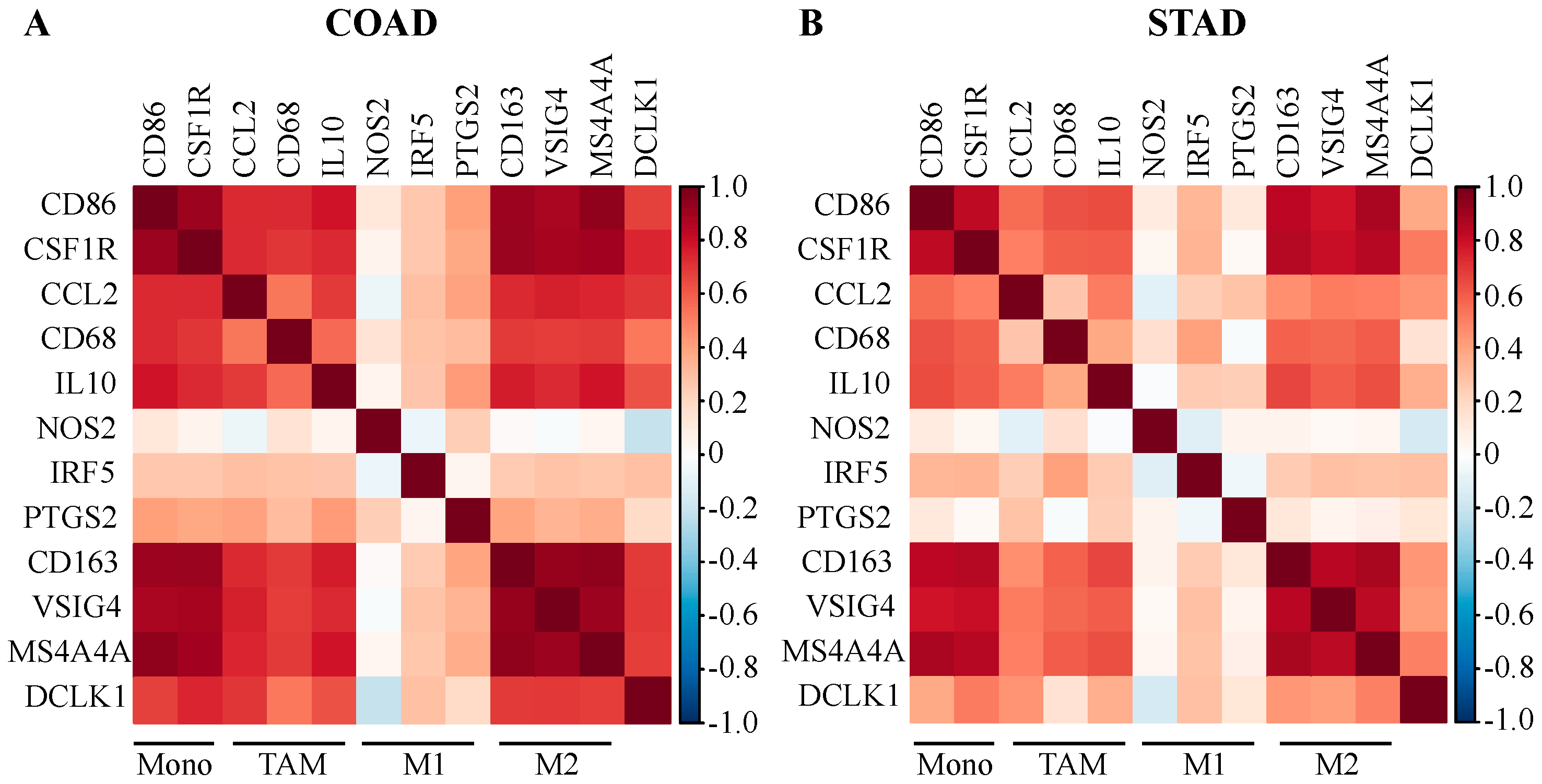
2.4. DCLK1 Expression Level is Associated with TAM and M2 Macrophage Markers
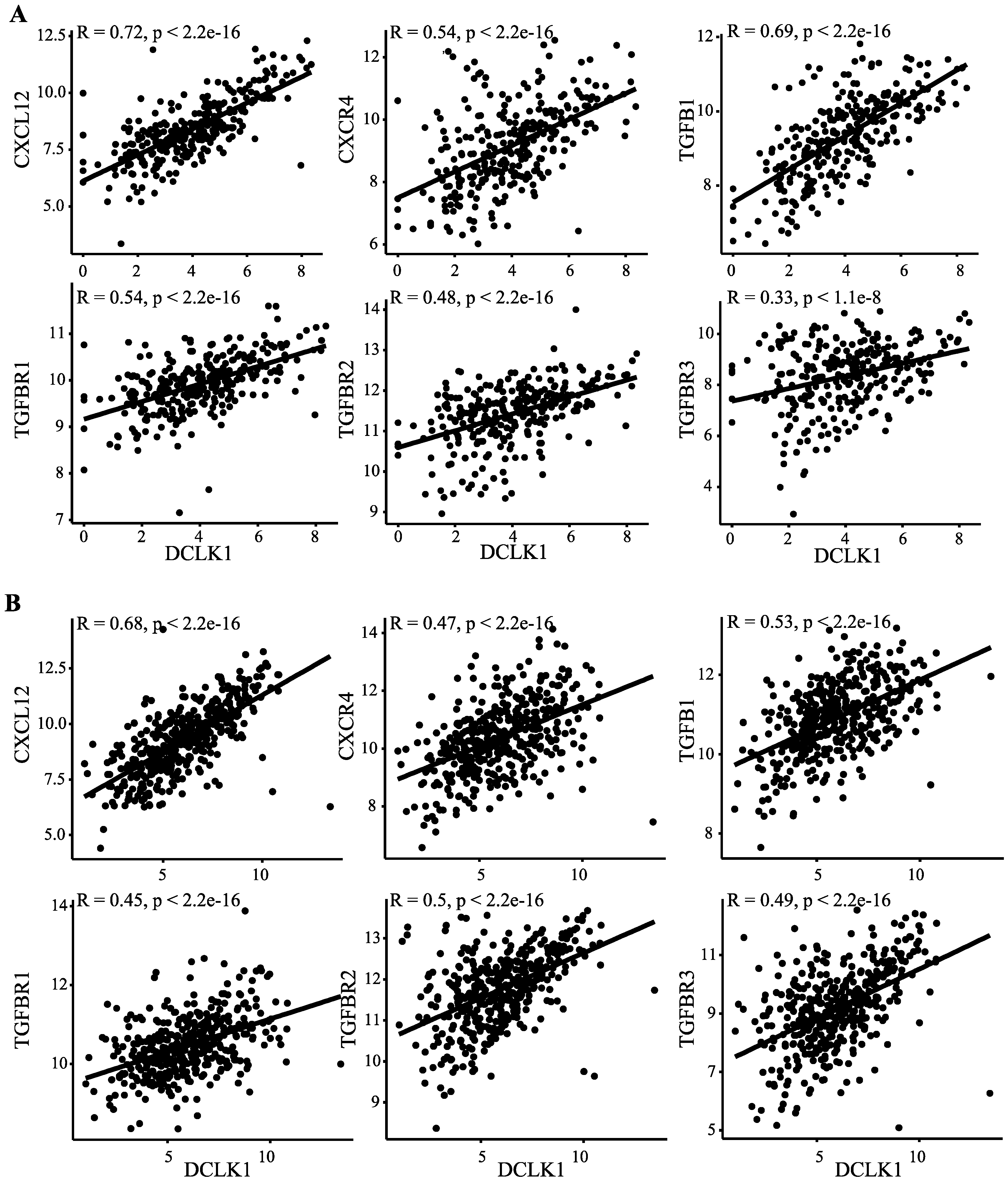
2.5. DCLK1 Correlates with Evasion of Antitumor Immunity, TAM Activation, and Inhibition of CD8+ T Cells
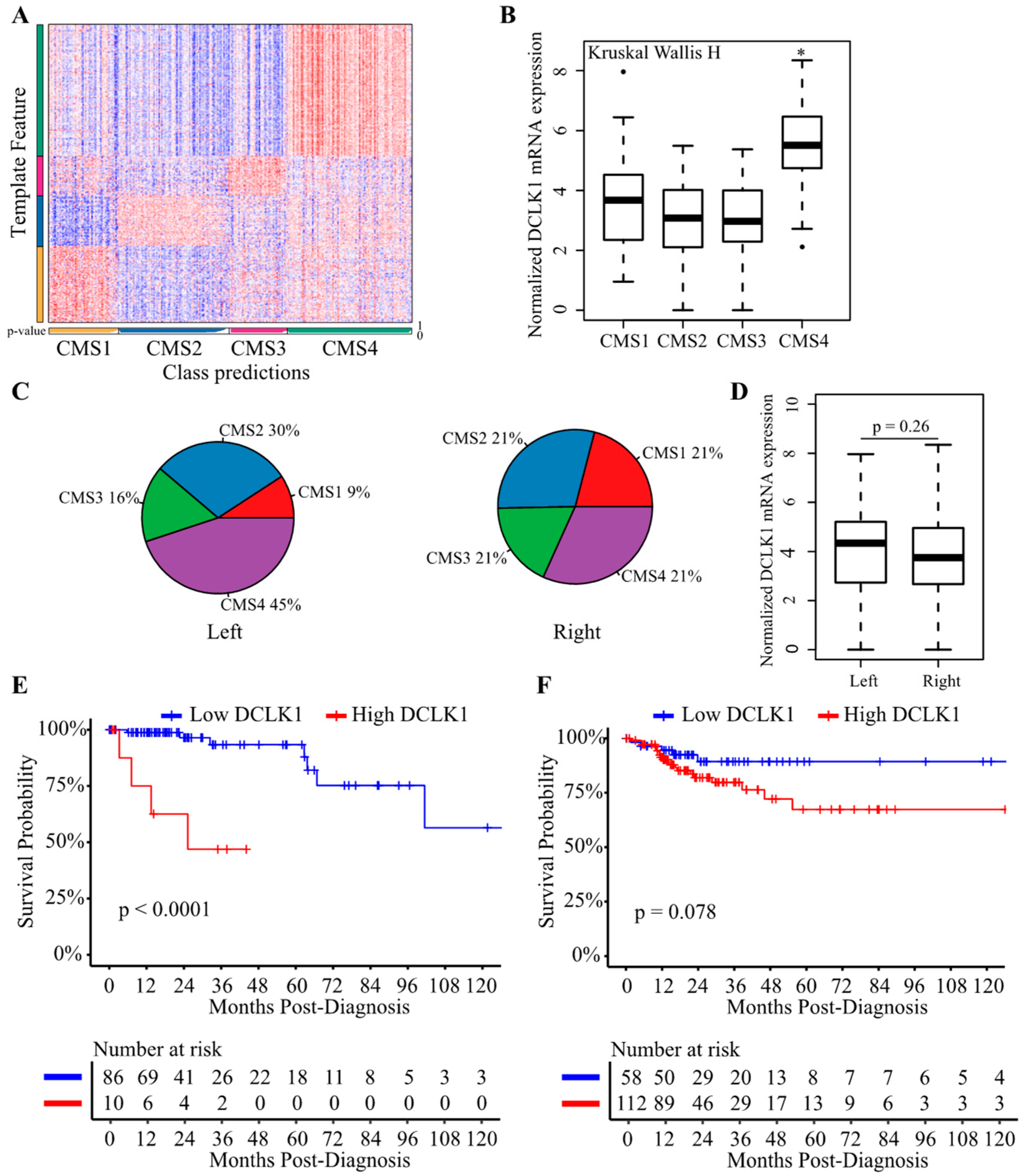
2.6. DCLK1 Predicts the Survival of Patients with Left-Sided Colon Cancer
3. Discussion
4. Materials and Methods
4.1. TCGA Colon and Stomach Cancer Dataset
4.2. Clinical Patient Characteristics
4.3. Estimation of Tumor Cellular Components and CRC Subtypes
4.4. Visualization of RNA Sequencing Dataset
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef]
- Jones, S.; Chen, W.D.; Parmigiani, G.; Diehl, F.; Beerenwinkel, N.; Antal, T.; Traulsen, A.; Nowak, M.A.; Siegel, C.; Velculescu, V.E.; et al. Comparative lesion sequencing provides insights into tumor evolution. Proc. Natl. Acad. Sci. USA 2008, 105, 4283–4288. [Google Scholar] [CrossRef] [PubMed]
- Maconi, G.; Manes, G.; Porro, G.B. Role of symptoms in diagnosis and outcome of gastric cancer. World J. Gastroenterol. 2008, 14, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Grady, W.M.; Pritchard, C.C. Molecular alterations and biomarkers in colorectal cancer. Toxicol. Pathol. 2014, 42, 124–139. [Google Scholar] [CrossRef] [PubMed]
- Tan, P.; Yeoh, K.G. Genetics and molecular pathogenesis of gastric adenocarcinoma. Gastroenterology 2015, 149, 1153–1162.e3. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.G.; Takeshima, H.; Niwa, T.; Rehnberg, E.; Shigematsu, Y.; Yoda, Y.; Yamashita, S.; Kushima, R.; Maekita, T.; Ichinose, M.; et al. Comprehensive DNA methylation and extensive mutation analyses reveal an association between the CpG island methylator phenotype and oncogenic mutations in gastric cancers. Cancer Lett. 2013, 330, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Herrinton, L.J.; Liu, L.; Levin, T.R.; Allison, J.E.; Lewis, J.D.; Velayos, F. Incidence and mortality of colorectal adenocarcinoma in persons with inflammatory bowel disease from 1998 to 2010. Gastroenterology 2012, 143, 382–389. [Google Scholar] [CrossRef]
- Kamangar, F.; Dawsey, S.M.; Blaser, M.J.; Perez-Perez, G.I.; Pietinen, P.; Newschaffer, C.J.; Abnet, C.C.; Albanes, D.; Virtamo, J.; Taylor, P.R. Opposing risks of gastric cardia and noncardia gastric adenocarcinomas associated with Helicobacter pylori seropositivity. J. Natl. Cancer Inst. 2006, 98, 1445–1452. [Google Scholar] [CrossRef]
- Zhao, J.; Liang, Q.; Cheung, K.F.; Kang, W.; Lung, R.W.; Tong, J.H.; To, K.F.; Sung, J.J.; Yu, J. Genome-wide identification of Epstein-Barr virus-driven promoter methylation profiles of human genes in gastric cancer cells. Cancer 2013, 119, 304–312. [Google Scholar] [CrossRef]
- Li, X.B.; Yang, G.; Zhu, L.; Tang, Y.L.; Zhang, C.; Ju, Z.; Yang, X.; Teng, Y. Gastric LGR5+ stem cells are the cellular origin of invasive intestinal-type gastric cancer in mice. Cell Res. 2016, 26, 838–849. [Google Scholar] [CrossRef] [PubMed]
- Barker, N.; Tan, S.; Clevers, H. LGR proteins in epithelial stem cell biology. Development 2013, 140, 2484–2494. [Google Scholar] [CrossRef] [PubMed]
- Morgan, R.G.; Mortensson, E.; Williams, A.C. Targeting LGR5 in colorectal cancer: Therapeutic gold or too plastic? Br. J. Cancer 2018, 118, 1410–1418. [Google Scholar] [CrossRef]
- Westphalen, C.B.; Asfaha, S.; Hayakawa, Y.; Takemoto, Y.; Lukin, D.J.; Nuber, A.H.; Brandtner, A.; Setlik, W.; Remotti, H.; Muley, A.; et al. Long-lived intestinal tuft cells serve as colon cancer-initiating cells. J. Clin. Investig. 2014, 124, 1283–1295. [Google Scholar] [CrossRef] [PubMed]
- Coutzac, C.; Pernot, S.; Chaput, N.; Zaanan, A. Immunotherapy in advanced gastric cancer, is it the future? Crit. Rev. Oncol. Hematol. 2019, 133, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, K.; Stadler, Z.K.; Cercek, A.; Mendelsohn, R.B.; Shia, J.; Segal, N.H.; Diaz, L.A., Jr. Immunotherapy in colorectal cancer: Rationale, challenges and potential. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 361–375. [Google Scholar] [CrossRef] [PubMed]
- Procaccio, L.; Schirripa, M.; Fassan, M.; Vecchione, L.; Bergamo, F.; Prete, A.A.; Intini, R.; Manai, C.; Dadduzio, V.; Boscolo, A.; et al. Immunotherapy in gastrointestinal cancers. Biomed. Res. Int. 2017, 2017, 4346576. [Google Scholar] [CrossRef]
- Barbee, M.S.; Ogunniyi, A.; Horvat, T.Z.; Dang, T.O. Current status and future directions of the immune checkpoint inhibitors ipilimumab, pembrolizumab, and nivolumab in oncology. Ann. Pharmacother. 2015, 49, 907–937. [Google Scholar] [CrossRef]
- Garon, E.B.; Rizvi, N.A.; Hui, R.; Leighl, N.; Balmanoukian, A.S.; Eder, J.P.; Patnaik, A.; Aggarwal, C.; Gubens, M.; Horn, L.; et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N. Engl. J. Med. 2015, 372, 2018–2028. [Google Scholar] [CrossRef]
- Muro, K.; Chung, H.C.; Shankaran, V.; Geva, R.; Catenacci, D.; Gupta, S.; Eder, J.P.; Golan, T.; Le, D.T.; Burtness, B.; et al. Pembrolizumab for patients with PD-L1-positive advanced gastric cancer (KEYNOTE-012): A multicentre, open-label, phase 1b trial. Lancet Oncol. 2016, 17, 717–726. [Google Scholar] [CrossRef]
- Le, D.T.; Uram, J.N.; Wang, H.; Bartlett, B.R.; Kemberling, H.; Eyring, A.D.; Skora, A.D.; Luber, B.S.; Azad, N.S.; Laheru, D.; et al. PD-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med. 2015, 372, 2509–2520. [Google Scholar] [CrossRef] [PubMed]
- Overman, M.J.; McDermott, R.; Leach, J.L.; Lonardi, S.; Lenz, H.-J.; Morse, M.A.; Desai, J.; Hill, A.; Axelson, M.; Moss, R.A.; et al. Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): An open-label, multicentre, phase 2 study. Lancet Oncol. 2017, 18, 1182–1191. [Google Scholar] [CrossRef]
- Anderson, K.G.; Stromnes, I.M.; Greenberg, P.D. Obstacles posed by the tumor microenvironment to T cell activity: A case for synergistic therapies. Cancer Cell 2017, 31, 311–325. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.S.; Mellman, I. Elements of cancer immunity and the cancer-immune set point. Nature 2017, 541, 321–330. [Google Scholar] [CrossRef]
- Joyce, J.A.; Fearon, D.T. T cell exclusion, immune privilege, and the tumor microenvironment. Science 2015, 348, 74–80. [Google Scholar] [CrossRef]
- Oh, S.A.; Li, M.O. TGF-β: Guardian of T cell function. J. Immunol. 2013, 191, 3973–3979. [Google Scholar] [CrossRef]
- Gocheva, V.; Wang, H.W.; Gadea, B.B.; Shree, T.; Hunter, K.E.; Garfall, A.L.; Berman, T.; Joyce, J.A. IL-4 induces cathepsin protease activity in tumor-associated macrophages to promote cancer growth and invasion. Genes Dev. 2010, 24, 241–255. [Google Scholar] [CrossRef]
- Coussens, L.M.; Zitvogel, L.; Palucka, A.K. Neutralizing tumor-promoting chronic inflammation: A magic bullet? Science 2013, 339, 286–291. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, H.; Shen, Z.; Lin, C.; Wang, X.; Qin, J.; Qin, X.; Xu, J.; Sun, Y. Tumor-infiltrating neutrophils is prognostic and predictive for postoperative adjuvant chemotherapy benefit in patients with gastric cancer. Ann. Surg. 2018, 267, 311–318. [Google Scholar] [CrossRef]
- Waniczek, D.; Lorenc, Z.; Snietura, M.; Wesecki, M.; Kopec, A.; Muc-Wierzgon, M. Tumor-associated macrophages and regulatory T cells infiltration and the clinical outcome in colorectal cancer. Arch. Immunol. Ther. Exp. 2017, 65, 445–454. [Google Scholar] [CrossRef]
- Noy, R.; Pollard, J.W. Tumor-associated macrophages: From mechanisms to therapy. Immunity 2014, 41, 49–61. [Google Scholar] [CrossRef] [PubMed]
- May, R.; Riehl, T.E.; Hunt, C.; Sureban, S.M.; Anant, S.; Houchen, C.W. Identification of a novel putative gastrointestinal stem cell and adenoma stem cell marker, doublecortin and CaM kinase-like-1, following radiation injury and in adenomatous polyposis coli/multiple intestinal neoplasia mice. Stem Cells 2008, 26, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Westphalen, C.B.; Takemoto, Y.; Tanaka, T.; Macchini, M.; Jiang, Z.; Renz, B.W.; Chen, X.; Ormanns, S.; Nagar, K.; Tailor, Y.; et al. Dclk1 defines quiescent pancreatic progenitors that promote injury-induced regeneration and tumorigenesis. Cell Stem Cell 2016, 18, 441–455. [Google Scholar] [CrossRef] [PubMed]
- Weygant, N.; Ge, Y.; Qu, D.; Kaddis, J.S.; Berry, W.L.; May, R.; Chandrakesan, P.; Bannerman-Menson, E.; Vega, K.J.; Tomasek, J.J.; et al. Survival of patients with gastrointestinal cancers can be predicted by a surrogate microRNA signature for cancer stem-like cells marked by DCLK1 kinase. Cancer Res. 2016, 76, 4090–4099. [Google Scholar] [CrossRef]
- Ge, Y.; Weygant, N.; Qu, D.; May, R.; Berry, W.L.; Yao, J.; Chandrakesan, P.; Zheng, W.; Zhao, L.; Zhao, K.L.; et al. Alternative splice variants of DCLK1 mark cancer stem cells, promote self-renewal and drug-resistance, and can be targeted to inhibit tumorigenesis in kidney cancer. Int. J. Cancer 2018, 143, 1162–1175. [Google Scholar] [CrossRef]
- Sureban, S.M.; May, R.; Ramalingam, S.; Subramaniam, D.; Natarajan, G.; Anant, S.; Houchen, C.W. Selective blockade of DCAMKL-1 results in tumor growth arrest by a Let-7a microRNA-dependent mechanism. Gastroenterology 2009, 137, 649–659.e2. [Google Scholar] [CrossRef]
- Weygant, N.; Qu, D.; May, R.; Tierney, R.M.; Berry, W.L.; Zhao, L.; Agarwal, S.; Chandrakesan, P.; Chinthalapally, H.R.; Murphy, N.T.; et al. DCLK1 is a broadly dysregulated target against epithelial-mesenchymal transition, focal adhesion, and stemness in clear cell renal carcinoma. Oncotarget 2015, 6, 2193–2205. [Google Scholar] [CrossRef]
- Sureban, S.M.; May, R.; Lightfoot, S.A.; Hoskins, A.B.; Lerner, M.; Brackett, D.J.; Postier, R.G.; Ramanujam, R.; Mohammed, A.; Rao, C.V.; et al. DCAMKL-1 regulates epithelial-mesenchymal transition in human pancreatic cells through a miR-200a-dependent mechanism. Cancer Res. 2011, 71, 2328–2338. [Google Scholar] [CrossRef]
- Gao, T.; Wang, M.; Xu, L.; Wen, T.; Liu, J.; An, G. DCLK1 is up-regulated and associated with metastasis and prognosis in colorectal cancer. J. Cancer Res. Clin. Oncol. 2016, 142, 2131–2140. [Google Scholar] [CrossRef]
- Li, J.; Wang, Y.; Ge, J.; Li, W.; Yin, L.; Zhao, Z.; Liu, S.; Qin, H.; Yang, J.; Wang, L.; et al. Doublecortin-like kinase 1 (DCLK1) regulates B cell-specific moloney murine leukemia virus insertion site 1 (Bmi-1) and is associated with metastasis and prognosis in pancreatic cancer. Cell Physiol. Biochem. 2018, 51, 262–277. [Google Scholar] [CrossRef]
- Qu, D.; Weygant, N.; Yao, J.; Chandrakesan, P.; Berry, W.L.; May, R.; Pitts, K.; Husain, S.; Lightfoot, S.; Li, M.; et al. Overexpression of DCLK1-AL increases tumor cell invasion, drug resistance, and KRAS activation and can be targeted to inhibit tumorigenesis in pancreatic cancer. J. Oncol. 2019, 2019, 6402925. [Google Scholar] [CrossRef] [PubMed]
- Chandrakesan, P.; Panneerselvam, J.; Qu, D.; Weygant, N.; May, R.; Bronze, M.S.; Houchen, C.W. Regulatory roles of DCLK1 in epithelial mesenchymal transition and cancer stem cells. J. Carcinog. Mutagen. 2016, 7, 257. [Google Scholar] [CrossRef] [PubMed]
- Chandrakesan, P.; Weygant, N.; May, R.; Qu, D.; Chinthalapally, H.R.; Sureban, S.M.; Ali, N.; Lightfoot, S.A.; Umar, S.; Houchen, C.W. DCLK1 facilitates intestinal tumor growth via enhancing pluripotency and epithelial mesenchymal transition. Oncotarget 2014, 5, 9269–9280. [Google Scholar] [CrossRef]
- Bailey, J.M.; Alsina, J.; Rasheed, Z.A.; McAllister, F.M.; Fu, Y.Y.; Plentz, R.; Zhang, H.; Pasricha, P.J.; Bardeesy, N.; Matsui, W.; et al. DCLK1 marks a morphologically distinct subpopulation of cells with stem cell properties in preinvasive pancreatic cancer. Gastroenterology 2014, 146, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, Y.; Seno, H.; Fukuoka, A.; Ueo, T.; Yamaga, Y.; Maruno, T.; Nakanishi, N.; Kanda, K.; Komekado, H.; Kawada, M.; et al. DCLK1 distinguishes between tumor and normal stem cells in the intestine. Nat. Genet. 2013, 45, 98–103. [Google Scholar] [CrossRef]
- Howitt, M.R.; Lavoie, S.; Michaud, M.; Blum, A.M.; Tran, S.V.; Weinstock, J.V.; Gallini, C.A.; Redding, K.; Margolskee, R.F.; Osborne, L.C.; et al. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 2016, 351, 1329–1333. [Google Scholar] [CrossRef]
- Von Moltke, J.; Ji, M.; Liang, H.E.; Locksley, R.M. Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature 2016, 529, 221–225. [Google Scholar] [CrossRef]
- Gerbe, F.; Sidot, E.; Smyth, D.J.; Ohmoto, M.; Matsumoto, I.; Dardalhon, V.; Cesses, P.; Garnier, L.; Pouzolles, M.; Brulin, B.; et al. Intestinal epithelial tuft cells initiate type 2 mucosal immunity to helminth parasites. Nature 2016, 529, 226–230. [Google Scholar] [CrossRef]
- Schneider, C.; O’Leary, C.E.; Locksley, R.M. Regulation of immune responses by tuft cells. Nat. Rev. Immunol. 2019, 19, 584–593. [Google Scholar] [CrossRef]
- Schneider, C.; O’Leary, C.E.; von Moltke, J.; Liang, H.E.; Ang, Q.Y.; Turnbaugh, P.J.; Radhakrishnan, S.; Pellizzon, M.; Ma, A.; Locksley, R.M. A metabolite-triggered tuft cell-ILC2 circuit drives small intestinal remodeling. Cell 2018, 174, 271–284.e214. [Google Scholar] [CrossRef]
- Lei, W.; Ren, W.; Ohmoto, M.; Urban, J.F., Jr.; Matsumoto, I.; Margolskee, R.F.; Jiang, P. Activation of intestinal tuft cell-expressed SUCNR1 triggers type 2 immunity in the mouse small intestine. Proc. Natl. Acad. Sci. USA 2018, 115, 5552–5557. [Google Scholar] [CrossRef] [PubMed]
- Nadjsombati, M.S.; McGinty, J.W.; Lyons-Cohen, M.R.; Jaffe, J.B.; DiPeso, L.; Schneider, C.; Miller, C.N.; Pollack, J.L.; Nagana Gowda, G.A.; Fontana, M.F.; et al. Detection of succinate by intestinal tuft cells triggers a type 2 innate immune circuit. Immunity 2018, 49, 33–41.e37. [Google Scholar] [CrossRef] [PubMed]
- Ting, H.A.; von Moltke, J. The immune function of tuft cells at gut mucosal surfaces and beyond. J. Immunol. 2019, 202, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Middelhoff, M.; Westphalen, C.B.; Hayakawa, Y.; Yan, K.S.; Gershon, M.D.; Wang, T.C.; Quante, M. DCLK1-expressing tuft cells: Critical modulators of the intestinal niche? Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 313, G285–G299. [Google Scholar] [CrossRef]
- Luo, X.C.; Chen, Z.H.; Xue, J.B.; Zhao, D.X.; Lu, C.; Li, Y.H.; Li, S.M.; Du, Y.W.; Liu, Q.; Wang, P.; et al. Infection by the parasitic helminth Trichinella spiralis activates a TAS2R-mediated signaling pathway in intestinal tuft cells. Proc. Natl. Acad. Sci. USA 2019, 116, 5564–5569. [Google Scholar] [CrossRef]
- The Cancer Genome Atlas Network. Comprehensive molecular characterization of human colon and rectal cancer. Nature 2012, 487, 330–337. [Google Scholar] [CrossRef]
- The Cancer Genome Atlas Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014, 513, 202–209. [Google Scholar] [CrossRef]
- Dai, T.; Hu, Y.; Lv, F.; Ozawa, T.; Sun, X.; Huang, J.; Han, X.; Kishi, H.; Muraguchi, A.; Jin, A. Analysis of the clinical significance of DCLK1+ colorectal cancer using novel monoclonal antibodies against DCLK1. OncoTargets Ther. 2018, 11, 5047–5057. [Google Scholar] [CrossRef]
- Meng, Q.B.; Yu, J.C.; Kang, W.M.; Ma, Z.Q.; Zhou, W.X.; Li, J.; Zhou, L.; Cao, Z.J.; Tian, S.B. Expression of doublecortin-like kinase 1 in human gastric cancer and its correlation with prognosis. Zhongguo Yi Xue Ke Xue Yuan Xue Bao Acta Acad. Med. Sin. 2013, 35, 639–644. [Google Scholar] [CrossRef]
- Liu, W.; Ye, H.; Liu, Y.F.; Xu, C.Q.; Zhong, Y.X.; Tian, T.; Ma, S.W.; Tao, H.; Li, L.; Xue, L.C.; et al. Transcriptome-derived stromal and immune scores infer clinical outcomes of patients with cancer. Oncol. Lett. 2018, 15, 4351–4357. [Google Scholar] [CrossRef]
- Arnold, T.; Betsholtz, C. The importance of microglia in the development of the vasculature in the central nervous system. Vasc. Cell 2013, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Savage, N.D.; de Boer, T.; Walburg, K.V.; Joosten, S.A.; van Meijgaarden, K.; Geluk, A.; Ottenhoff, T.H. Human anti-inflammatory macrophages induce FOXP3+ GITR+ CD25+ regulatory T cells, which suppress via membrane-bound TGFβ-1. J. Immunol. 2008, 181, 2220–2226. [Google Scholar] [CrossRef] [PubMed]
- Guinney, J.; Dienstmann, R.; Wang, X.; de Reynies, A.; Schlicker, A.; Soneson, C.; Marisa, L.; Roepman, P.; Nyamundanda, G.; Angelino, P.; et al. The consensus molecular subtypes of colorectal cancer. Nat. Med. 2015, 21, 1350–1356. [Google Scholar] [CrossRef] [PubMed]
- Eide, P.W.; Bruun, J.; Lothe, R.A.; Sveen, A. CMScaller: An R package for consensus molecular subtyping of colorectal cancer pre-clinical models. Sci. Rep. 2017, 7, 16618. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yang, L.; Zhou, M.; Shen, L.; Zhang, J.; Deng, W.; Liang, L.; Hu, R.; Yang, W.; Yao, Y.; et al. Disparities in survival for right-sided vs. left-sided colon cancers in young patients: A study based on the Surveillance, Epidemiology, and End Results database (1990–2014). Cancer Manag. Res. 2018, 10, 1735–1747. [Google Scholar] [CrossRef]
- Franke, A.J.; Skelton, W.P.; Starr, J.S.; Parekh, H.; Lee, J.J.; Overman, M.J.; Allegra, C.; George, T.J. Immunotherapy for colorectal cancer: A review of current and novel therapeutic approaches. J. Natl. Cancer Inst. 2019, 111, 1131–1141. [Google Scholar] [CrossRef]
- Hu, B.; El Hajj, N.; Sittler, S.; Lammert, N.; Barnes, R.; Meloni-Ehrig, A. Gastric cancer: Classification, histology and application of molecular pathology. J. Gastrointest. Oncol. 2012, 3, 251–261. [Google Scholar] [CrossRef]
- Ajani, J.A.; Lee, J.; Sano, T.; Janjigian, Y.Y.; Fan, D.; Song, S. Gastric adenocarcinoma. Nat. Rev. Dis. Primers 2017, 3, 17036. [Google Scholar] [CrossRef]
- Zhou, B.; Sun, C.; Hu, X.; Zhan, H.; Zou, H.; Feng, Y.; Qiu, F.; Zhang, S.; Wu, L.; Zhang, B. MicroRNA-195 suppresses the progression of pancreatic cancer by targeting DCLK1. Cell Physiol. Biochem. 2017, 44, 1867–1881. [Google Scholar] [CrossRef]
- Whorton, J.; Sureban, S.M.; May, R.; Qu, D.; Lightfoot, S.A.; Madhoun, M.; Johnson, M.; Tierney, W.M.; Maple, J.T.; Vega, K.J.; et al. DCLK1 is detectable in plasma of patients with Barrett’s esophagus and esophageal adenocarcinoma. Dig. Dis. Sci. 2015, 60, 509–513. [Google Scholar] [CrossRef]
- Lv, Y.; Song, G.; Wang, R.; Di, L.; Wang, J. Doublecortin-like kinase 1 is a novel biomarker for prognosis and regulates growth and metastasis in basal-like breast cancer. Biomed. Pharmacother. 2017, 88, 1198–1205. [Google Scholar] [CrossRef]
- Kalantari, E.; Asadi Lari, M.H.; Roudi, R.; Korourian, A.; Madjd, Z. LGR5High/DCLK1High phenotype is more common in early stage and intestinal subtypes of gastric carcinomas. Cancer Biomark. 2017, 20, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Sato, E.; Olson, S.H.; Ahn, J.; Bundy, B.; Nishikawa, H.; Qian, F.; Jungbluth, A.A.; Frosina, D.; Gnjatic, S.; Ambrosone, C.; et al. Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 18538–18543. [Google Scholar] [CrossRef] [PubMed]
- Szabo, S.J.; Kim, S.T.; Costa, G.L.; Zhang, X.; Fathman, C.G.; Glimcher, L.H. A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell 2000, 100, 655–669. [Google Scholar] [CrossRef]
- Kanhere, A.; Hertweck, A.; Bhatia, U.; Gokmen, M.R.; Perucha, E.; Jackson, I.; Lord, G.M.; Jenner, R.G. T-bet and GATA3 orchestrate Th1 and Th2 differentiation through lineage-specific targeting of distal regulatory elements. Nat. Commun. 2012, 3, 1268. [Google Scholar] [CrossRef] [PubMed]
- Hertweck, A.; Evans, C.M.; Eskandarpour, M.; Lau, J.C.; Oleinika, K.; Jackson, I.; Kelly, A.; Ambrose, J.; Adamson, P.; Cousins, D.J.; et al. T-bet activates Th1 genes through mediator and the super elongation complex. Cell Rep. 2016, 15, 2756–2770. [Google Scholar] [CrossRef]
- Nishikomori, R.; Usui, T.; Wu, C.Y.; Morinobu, A.; O’Shea, J.J.; Strober, W. Activated STAT4 has an essential role in Th1 differentiation and proliferation that is independent of its role in the maintenance of IL-12Rβ2 chain expression and signaling. J. Immunol. 2002, 169, 4388–4398. [Google Scholar] [CrossRef]
- Thieu, V.T.; Yu, Q.; Chang, H.C.; Yeh, N.; Nguyen, E.T.; Sehra, S.; Kaplan, M.H. Signal transducer and activator of transcription 4 is required for the transcription factor T-bet to promote T helper 1 cell-fate determination. Immunity 2008, 29, 679–690. [Google Scholar] [CrossRef]
- Mandai, M.; Hamanishi, J.; Abiko, K.; Matsumura, N.; Baba, T.; Konishi, I. Dual faces of IFNγ in cancer progression: A role of PD-L1 induction in the determination of pro- and antitumor immunity. Clin. Cancer Res. 2016, 22, 2329–2334. [Google Scholar] [CrossRef]
- Decker, T.; Lohmann-Matthes, M.L.; Gifford, G.E. Cell-associated tumor necrosis factor (TNF) as a killing mechanism of activated cytotoxic macrophages. J. Immunol. 1987, 138, 957–962. [Google Scholar]
- Pylayeva-Gupta, Y.; Das, S.; Handler, J.S.; Hajdu, C.H.; Coffre, M.; Koralov, S.B.; Bar-Sagi, D. IL35-producing B cells promote the development of pancreatic neoplasia. Cancer Discov. 2016, 6, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Sarvaria, A.; Madrigal, J.A.; Saudemont, A. B cell regulation in cancer and anti-tumor immunity. Cell Mol. Immunol. 2017, 14, 662–674. [Google Scholar] [CrossRef] [PubMed]
- Wesa, A.K.; Galy, A. IL-1β induces dendritic cells to produce IL-12. Int. Immunol. 2001, 13, 1053–1061. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A. Interleukin-1 in the pathogenesis and treatment of inflammatory diseases. Blood 2011, 117, 3720–3732. [Google Scholar] [CrossRef]
- Wesa, A.; Galy, A. Increased production of pro-inflammatory cytokines and enhanced T cell responses after activation of human dendritic cells with IL-1 and CD40 ligand. BMC Immunol. 2002, 3, 14. [Google Scholar] [CrossRef]
- Myers, L.M.; Tal, M.C.; Torrez Dulgeroff, L.B.; Carmody, A.B.; Messer, R.J.; Gulati, G.; Yiu, Y.Y.; Staron, M.M.; Angel, C.L.; Sinha, R.; et al. A functional subset of CD8+ T cells during chronic exhaustion is defined by SIRPα expression. Nat. Commun. 2019, 10, 794. [Google Scholar] [CrossRef]
- Blackburn, S.D.; Shin, H.; Haining, W.N.; Zou, T.; Workman, C.J.; Polley, A.; Betts, M.R.; Freeman, G.J.; Vignali, D.A.; Wherry, E.J. Coregulation of CD8+ T cell exhaustion by multiple inhibitory receptors during chronic viral infection. Nat. Immunol. 2009, 10, 29–37. [Google Scholar] [CrossRef]
- Barber, D.L.; Wherry, E.J.; Masopust, D.; Zhu, B.; Allison, J.P.; Sharpe, A.H.; Freeman, G.J.; Ahmed, R. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 2006, 439, 682–687. [Google Scholar] [CrossRef]
- Bae, E.A.; Seo, H.; Kim, B.S.; Choi, J.; Jeon, I.; Shin, K.S.; Koh, C.H.; Song, B.; Kim, I.K.; Min, B.S.; et al. Activation of NKT cells in an anti-PD-1-resistant tumor model enhances antitumor immunity by reinvigorating exhausted CD8 T cells. Cancer Res. 2018, 78, 5315–5326. [Google Scholar] [CrossRef]
- Chakraborty, H.; Hossain, A. R package to estimate intracluster correlation coefficient with confidence interval for binary data. Comput. Methods Programs Biomed. 2018, 155, 85–92. [Google Scholar] [CrossRef]
- Yoshihara, K.; Shahmoradgoli, M.; Martinez, E.; Vegesna, R.; Kim, H.; Torres-Garcia, W.; Trevino, V.; Shen, H.; Laird, P.W.; Levine, D.A.; et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat. Commun. 2013, 4, 2612. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Cheng, W.; Ren, X.; Wang, Z.; Liu, X.; Li, G.; Han, S.; Jiang, T.; Wu, A. Tumor purity as an underlying key factor in glioma. Clin. Cancer Res. 2017, 23, 6279–6291. [Google Scholar] [CrossRef] [PubMed]
- Finotello, F.; Mayer, C.; Plattner, C.; Laschober, G.; Rieder, D.; Hackl, H.; Krogsdam, A.; Loncova, Z.; Posch, W.; Wilflingseder, D.; et al. Molecular and pharmacological modulators of the tumor immune contexture revealed by deconvolution of RNA-seq data. Genome Med. 2019, 11, 34. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.H.; Zhou, H.; Cooper, L.; Huang, J.L.; Zhu, S.B.; Zhao, X.X.; Ding, H.; Pan, Y.L.; Rong, L. LAYN is a prognostic biomarker and correlated with immune infiltrates in gastric and colon cancers. Front. Immunol. 2019, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Lausen, B.; Lerche, R.; Schumacher, M. Maximally selected rank statistics for dose-response problems. Biom. J. 2002, 44, 131–147. [Google Scholar] [CrossRef]
- Hillengass, J.; Ritsch, J.; Merz, M.; Wagner, B.; Kunz, C.; Hielscher, T.; Laue, H.; Bauerle, T.; Zechmann, C.M.; Ho, A.D.; et al. Increased microcirculation detected by dynamic contrast-enhanced magnetic resonance imaging is of prognostic significance in asymptomatic myeloma. Br. J. Haematol. 2016, 174, 127–135. [Google Scholar] [CrossRef]

| Description | Gene Markers | COAD | STAD | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Cor | 95%CI | p | Cor | 95%CI | p | ||||
| CD8+ T cell | CD8A | 0.412 | 0.311 | 0.503 | *** | 0.328 | 0.239 | 0.411 | *** |
| CD8B | 0.253 | 0.141 | 0.358 | *** | 0.187 | 0.092 | 0.278 | 0.0001 | |
| T cell (general) | CD3D | 0.415 | 0.315 | 0.507 | *** | 0.258 | 0.166 | 0.346 | *** |
| CD3E | 0.512 | 0.421 | 0.593 | *** | 0.303 | 0.213 | 0.388 | *** | |
| CD2 | 0.476 | 0.381 | 0.561 | *** | 0.320 | 0.231 | 0.404 | *** | |
| B cell | CD19 | 0.477 | 0.382 | 0.562 | *** | 0.372 | 0.286 | 0.452 | *** |
| CD79A | 0.568 | 0.484 | 0.642 | *** | 0.396 | 0.312 | 0.474 | *** | |
| IL35 | 0.559 | 0.474 | 0.634 | *** | 0.355 | 0.268 | 0.436 | *** | |
| Monocyte | CD86 | 0.680 | 0.612 | 0.738 | *** | 0.379 | 0.293 | 0.458 | *** |
| CD115(CSF1R) | 0.748 | 0.692 | 0.795 | *** | 0.522 | 0.448 | 0.589 | *** | |
| CCL2 | 0.704 | 0.640 | 0.758 | *** | 0.445 | 0.364 | 0.519 | *** | |
| CD68 | 0.525 | 0.436 | 0.604 | *** | 0.160 | 0.064 | 0.252 | 0.001 | |
| IL10 | 0.633 | 0.558 | 0.698 | *** | 0.363 | 0.276 | 0.444 | *** | |
| M1 Macrophage | INOS(NOS2) | −0.207 | −0.315 | −0.093 | 0.0004 | −0.147 | −0.240 | −0.052 | 0.003 |
| IRF5 | 0.292 | 0.182 | 0.395 | *** | 0.297 | 0.207 | 0.382 | *** | |
| COX2(PTGS2) | 0.186 | 0.071 | 0.295 | 0.001 | 0.135 | 0.039 | 0.228 | 0.006 | |
| M2 Macrophage | CD163 | 0.692 | 0.627 | 0.748 | *** | 0.437 | 0.356 | 0.512 | *** |
| VSIG4 | 0.695 | 0.630 | 0.751 | *** | 0.416 | 0.333 | 0.492 | *** | |
| MS4A4A | 0.690 | 0.624 | 0.746 | *** | 0.502 | 0.426 | 0.570 | *** | |
| Neutrophil | CD66b(CEACAM8) | −0.129 | −0.242 | −0.013 | 0.029 | −0.008 | −0.104 | 0.089 | 0.875 |
| CD11b(ITGAM) | 0.725 | 0.665 | 0.776 | *** | 0.475 | 0.397 | 0.546 | *** | |
| CCR7 | 0.585 | 0.503 | 0.657 | *** | 0.473 | 0.395 | 0.545 | *** | |
| Natural killer cell | KIR2DL1 | 0.196 | 0.082 | 0.305 | 0.0008 | 0.181 | 0.086 | 0.272 | 0.0002 |
| KIR2DL3 | 0.275 | 0.164 | 0.379 | *** | 0.105 | 0.009 | 0.199 | 0.032 | |
| KIR2DL4 | 0.105 | −0.011 | 0.218 | 0.076 | −0.069 | −0.164 | 0.027 | 0.160 | |
| KIR3DL1 | 0.210 | 0.097 | 0.319 | 0.0003 | 0.181 | 0.086 | 0.273 | 0.0002 | |
| KIR3DL2 | 0.281 | 0.171 | 0.385 | *** | 0.138 | 0.042 | 0.231 | 0.032 | |
| KIR3DL3 | 0.040 | −0.076 | 0.155 | 0.500 | −0.051 | −0.146 | 0.046 | 0.302 | |
| KIR2DS4 | 0.187 | 0.073 | 0.297 | 0.0014 | 0.076 | −0.021 | 0.171 | 0.124 | |
| Dendritic cell | HLA-DPB1 | 0.648 | 0.576 | 0.711 | *** | 0.311 | 0.222 | 0.396 | *** |
| HLA-DQB1 | 0.386 | 0.283 | 0.480 | *** | 0.142 | 0.047 | 0.235 | 0.004 | |
| HLA-DRA | 0.506 | 0.414 | 0.587 | *** | 0.211 | 0.117 | 0.301 | *** | |
| HLA-DPA1 | 0.583 | 0.501 | 0.655 | *** | 0.268 | 0.176 | 0.355 | *** | |
| BDCA-1(CD1C) | 0.668 | 0.598 | 0.728 | *** | 0.567 | 0.497 | 0.629 | *** | |
| Th1 | BDCA-1(NRP1) | 0.760 | 0.707 | 0.805 | *** | 0.635 | 0.574 | 0.689 | *** |
| CD11c(ITGAX) | 0.635 | 0.560 | 0.699 | *** | 0.405 | 0.321 | 0.482 | *** | |
| CD11b (ITGAM) | 0.725 | 0.655 | 0.776 | *** | 0.475 | 0.397 | 0.546 | *** | |
| IL12A | 0.046 | −0.07 | 0.161 | 0.437 | 0.192 | 0.098 | 0.283 | *** | |
| IL1B | −0.228 | −0.318 | −0.135 | *** | 0.068 | −0.048 | 0.183 | 0.246 | |
| T-bet (TBX21) | 0.483 | 0.388 | 0.567 | *** | 0.301 | 0.211 | 0.386 | *** | |
| STAT4 | 0.530 | 0.442 | 0.609 | *** | 0.396 | 0.311 | 0.474 | *** | |
| STAT1 | 0.374 | 0.270 | 0.470 | *** | 0.004 | −0.092 | 0.100 | 0.933 | |
| IFN-gamma (IFNG) | 0.158 | 0.043 | 0.269 | 0.007 | −0.042 | −0.137 | 0.055 | 0.398 | |
| TNF-alpha (TNF) | 0.339 | 0.232 | 0.438 | *** | 0.011 | −0.085 | 0.107 | 0.820 | |
| Th2 | GATA3 | 0.628 | 0.552 | 0.693 | *** | 0.424 | 0.342 | 0.500 | *** |
| STAT6 | −0.010 | −0.126 | 0.106 | 0.860 | 0.163 | 0.067 | 0.255 | 0.0009 | |
| STAT5A | 0.393 | 0.290 | 0.487 | *** | 0.421 | 0.339 | 0.497 | *** | |
| IL13 | 0.259 | 0.147 | 0.364 | *** | 0.146 | 0.050 | 0.239 | 0.003 | |
| Tfh | BCL6 | 0.536 | 0.448 | 0.614 | *** | 0.456 | 0.376 | 0.529 | *** |
| Th17 | IL21 | 0.319 | 0.211 | 0.419 | *** | 0.121 | 0.025 | 0.215 | 0.013 |
| STAT3 | 0.339 | 0.232 | 0.438 | *** | 0.321 | 0.232 | 0.405 | *** | |
| Treg | IL17A | −0.208 | −0.316 | −0.094 | 0.0003 | −0.308 | −0.393 | −0.218 | *** |
| FOXP3 | 0.615 | 0.537 | 0.682 | *** | 0.241 | 0.148 | 0.329 | *** | |
| T cell exhaustion | CCR8 | 0.628 | 0.552 | 0.693 | *** | 0.353 | 0.266 | 0.435 | *** |
| STAT5B | 0.460 | 0.363 | 0.547 | *** | 0.635 | 0.573 | 0.689 | *** | |
| TGFbeta (TGFB1) | 0.692 | 0.626 | 0.748 | *** | 0.530 | 0.457 | 0.596 | *** | |
| CD25 (IL2RA) | 0.575 | 0.491 | 0.648 | *** | 0.292 | 0.201 | 0.377 | *** | |
| CD4 | 0.708 | 0.646 | 0.762 | *** | 0.424 | 0.342 | 0.500 | *** | |
| PD-1(PDCD1) | 0.435 | 0.336 | 0.524 | *** | 0.201 | 0.107 | 0.291 | *** | |
| CTLA4 | 0.478 | 0.383 | 0.563 | *** | 0.106 | 0.010 | 0.200 | 0.031 | |
| LAG3 | 0.341 | 0.234 | 0.439 | *** | 0.140 | 0.045 | 0.233 | 0.004 | |
| TIM-3(HAVCR2) | 0.666 | 0.596 | 0.726 | *** | 0.362 | 0.275 | 0.443 | *** | |
| BTLA | 0.556 | 0.471 | 0.632 | *** | 0.436 | 0.354 | 0.511 | *** | |
| TIGIT | 0.541 | 0.453 | 0.618 | *** | 0.344 | 0.256 | 0.426 | *** | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, X.; Qu, D.; Weygant, N.; Peng, J.; Houchen, C.W. Cancer Stem Cell Marker DCLK1 Correlates with Tumorigenic Immune Infiltrates in the Colon and Gastric Adenocarcinoma Microenvironments. Cancers 2020, 12, 274. https://doi.org/10.3390/cancers12020274
Wu X, Qu D, Weygant N, Peng J, Houchen CW. Cancer Stem Cell Marker DCLK1 Correlates with Tumorigenic Immune Infiltrates in the Colon and Gastric Adenocarcinoma Microenvironments. Cancers. 2020; 12(2):274. https://doi.org/10.3390/cancers12020274
Chicago/Turabian StyleWu, Xiangyan, Dongfeng Qu, Nathaniel Weygant, Jun Peng, and Courtney W. Houchen. 2020. "Cancer Stem Cell Marker DCLK1 Correlates with Tumorigenic Immune Infiltrates in the Colon and Gastric Adenocarcinoma Microenvironments" Cancers 12, no. 2: 274. https://doi.org/10.3390/cancers12020274
APA StyleWu, X., Qu, D., Weygant, N., Peng, J., & Houchen, C. W. (2020). Cancer Stem Cell Marker DCLK1 Correlates with Tumorigenic Immune Infiltrates in the Colon and Gastric Adenocarcinoma Microenvironments. Cancers, 12(2), 274. https://doi.org/10.3390/cancers12020274





