Assessing Prognostic and Predictive Biomarkers of Regorafenib Response in Patients with Advanced Soft Tissue Sarcoma: REGOSARC Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Translational Research
2.3. Statistical Analysis
3. Results
3.1. Patient Disposition and Disease Characteristics
3.2. Biomarker Distribution
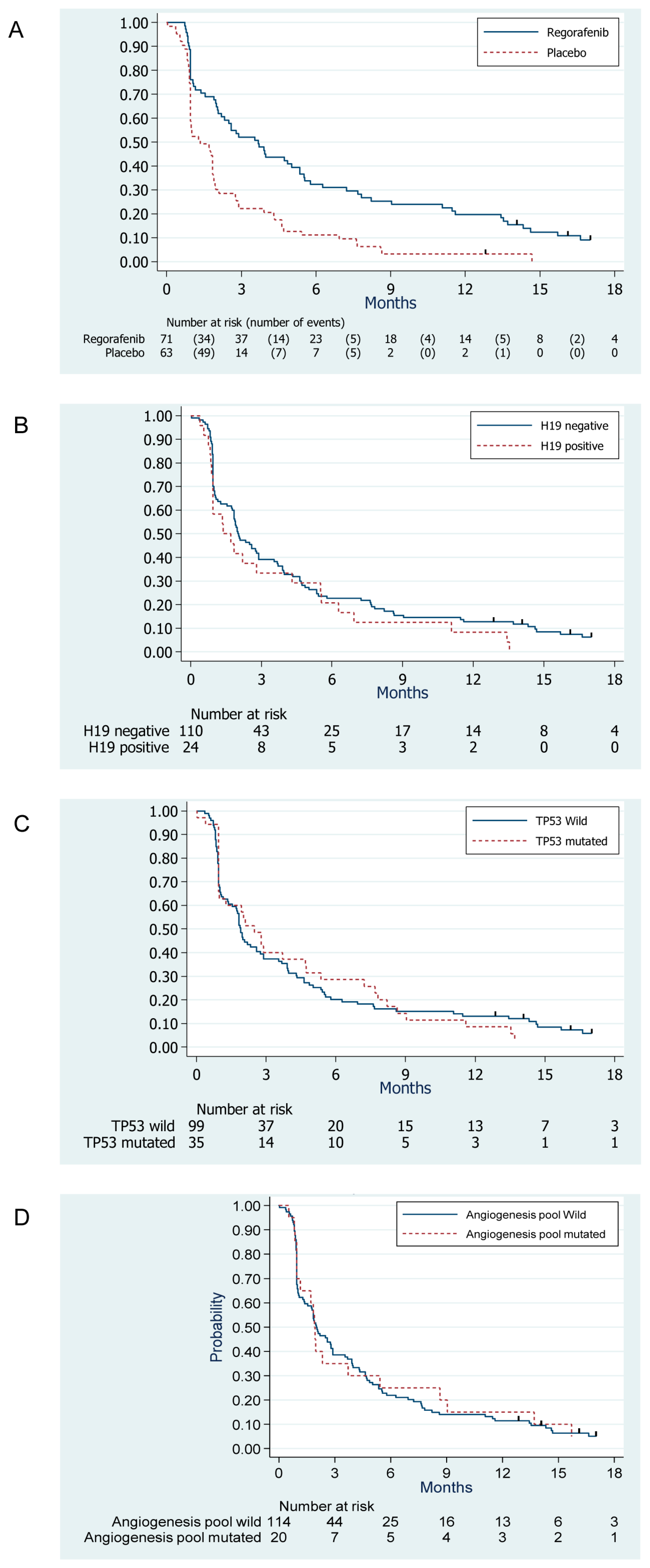
3.3. Survival Outcomes
3.4. Evaluation of the Prognostic Value of Biomarkers
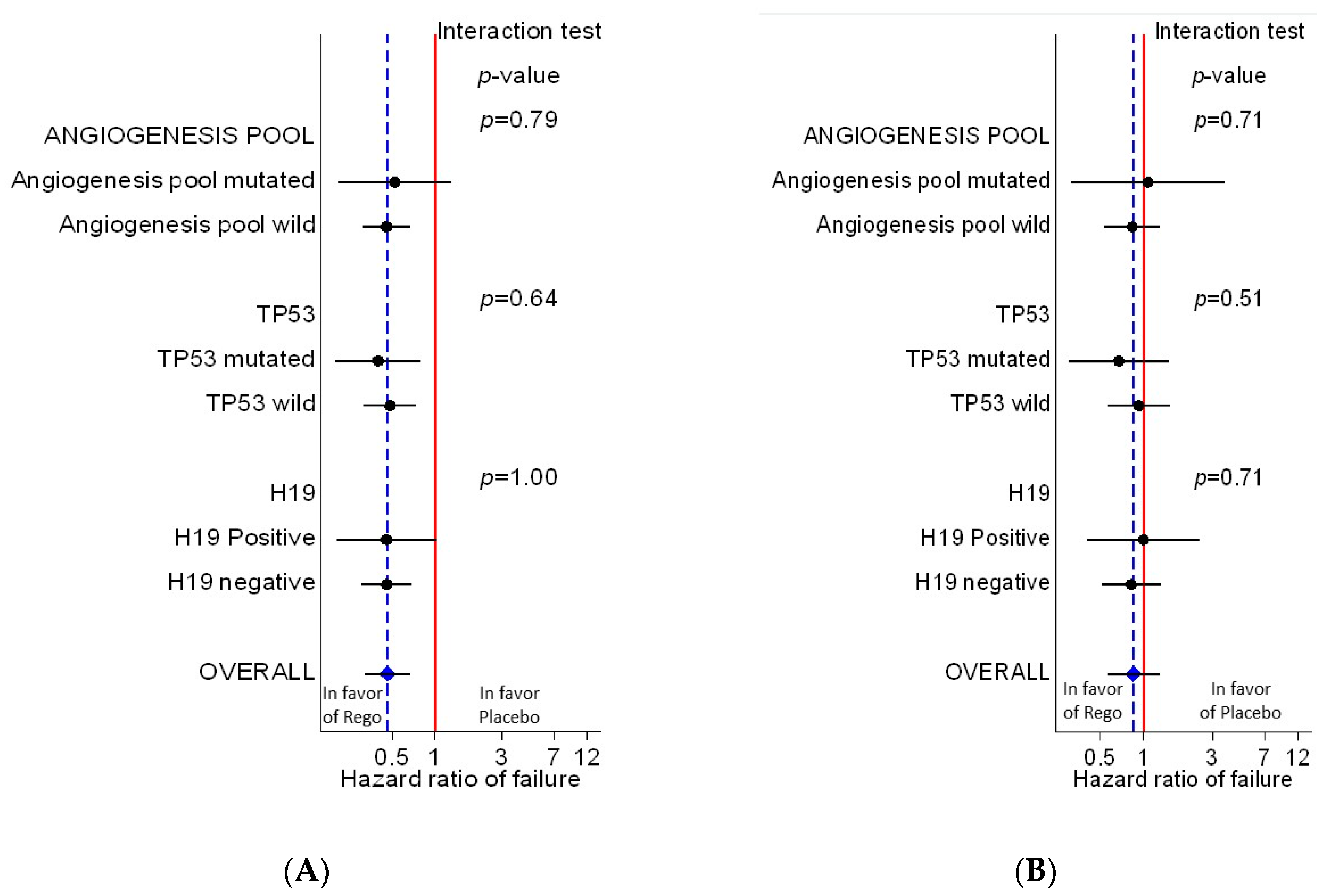
3.5. Evaluation of the Predictive Value of Biomarkers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Ferreira, A.L.D.A.; Matsubara, L.S. Anthracycline-Induced Cardiotoxicity. Cardiovasc. Hematol. Agents Med. Chem. 2008, 6, 278–281. [Google Scholar] [CrossRef] [PubMed]
- Van Oosterom, A.; Mouridsen, H.; Nielsen, O.; Dombernowsky, P.; Krzemieniecki, K.; Judson, I.; Svancarova, L.; Spooner, D.; Hermans, C.; Van Glabbeke, M.; et al. Results of randomised studies of the EORTC Soft Tissue and Bone Sarcoma Group (STBSG) with two different ifosfamide regimens in first- and second-line chemotherapy in advanced soft tissue sarcoma patients. Eur. J. Cancer 2002, 38, 2397–2406. [Google Scholar] [CrossRef]
- Van Der Graaf, W.T.A.; Blay, J.-Y.; Chawla, S.P.; Kim, D.-W.; Bui-Nguyen, B.; Casali, P.G.; Schöffski, P.; Aglietta, M.; Staddon, A.; Beppu, Y.; et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2012, 379, 1879–1886. [Google Scholar] [CrossRef]
- Takigawa, H.; Kitadai, Y.; Shinagawa, K.; Yuge, R.; Higashi, Y.; Tanaka, S.; Yasui, W.; Chayama, K. Multikinase inhibitor regorafenib inhibits the growth and metastasis of colon cancer with abundant stroma. Cancer Sci. 2016, 107, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Grothey, A.; Van Cutsem, E.; Sobrero, A.; Siena, S.; Falcone, A.; Ychou, M.; Humblet, Y.; Bouché, O.; Mineur, L.; Barone, C.A.; et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 2013, 381, 303–312. [Google Scholar] [CrossRef]
- Li, M.; Chen, H.; Zhao, Y.; Gao, S.; Cheng, C. H19 Functions as a ceRNA in Promoting Metastasis Through Decreasing miR-200s Activity in Osteosarcoma. DNA Cell Biol. 2016, 35, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Mir, O.; Brodowicz, T.; Italiano, A.; Wallet, J.; Blay, J.-Y.; Bertucci, F.; Chevreau, C.; Piperno-Neumann, S.; Bompas, E.; Salas, S.; et al. Safety and efficacy of regorafenib in patients with advanced soft tissue sarcoma (REGOSARC): A randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Oncol. 2016, 17, 1732–1742. [Google Scholar] [CrossRef]
- Brodowicz, T.; Liegl-Atzwanger, B.; Tresch, E.; Taïeb, S.; Kramar, A.; Grünwald, V.; Vanseymortier, M.; Clisant, S.; Blay, J.-Y.; Le Cesne, A.; et al. Study protocol of REGOSARC trial: Activity and safety of regorafenib in advanced soft tissue sarcoma: A multinational, randomized, placebo-controlled, phase II trial. BMC Cancer 2015, 15, 127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ballinger, M.L.; Goode, D.L.; Ray-Coquard, I.; James, P.A.; Mitchell, G.; Niedermayr, E.; Puri, A.; Schiffman, J.D.; Dite, G.S.; Cipponi, A.; et al. Monogenic and polygenic determinants of sarcoma risk: An international genetic study. Lancet Oncol. 2016, 17, 1261–1271. [Google Scholar] [CrossRef]
- Mitchell, G.; Ballinger, M.; Wong, S.Q.; Hewitt, C.; James, P.; Young, M.-A.; Cipponi, A.; Pang, T.; Goode, D.L.; Dobrovic, A.; et al. High Frequency of Germline TP53 Mutations in a Prospective Adult-Onset Sarcoma Cohort. PLoS ONE 2013, 8, e69026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duffaud, F.; Mir, O.; Boudou-Rouquette, P.; Piperno-Neumann, S.; Penel, N.; Bompas, E.; Delcambre, C.; Kalbacher, E.; Italiano, A.; Collard, O.; et al. Efficacy and safety of regorafenib in adult patients with metastatic osteosarcoma: A non-comparative, randomised, double-blind, placebo-controlled, phase 2 study. Lancet Oncol. 2019, 20, 120–133. [Google Scholar] [CrossRef]
- Demetri, G.D.; Reichardt, P.; Kang, Y.-K.; Blay, J.-Y.; Rutkowski, P.; Gelderblom, H.; Hohenberger, P.; Leahy, M.; Von Mehren, M.; Joensuu, H.; et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 2013, 381, 295–302. [Google Scholar] [CrossRef] [Green Version]
| Characteristics | Regorafenib n = 71 | Placebo n = 63 | Total n = 134 | ||||
|---|---|---|---|---|---|---|---|
| Sex | Male | 32 | 45% | 31 | 49% | 63 | 47% |
| Female | 39 | 55% | 32 | 51% | 71 | 53% | |
| Country | France | 64 | 90% | 58 | 92% | 122 | 91% |
| Austria | 7 | 10% | 5 | 8% | 12 | 9% | |
| ECOG PS | 0 | 33 | 46% | 29 | 46% | 62 | 46% |
| 1 | 38 | 54% | 33 | 52% | 71 | 53% | |
| 2 | 0 | 0% | 1 | 2% | 1 | 1% | |
| Age | Median (range) | 59 | (21–81) | 59 | (22–80) | 59 | (21–81) |
| Histology | Liposarcoma (Cohort A) | 18 | 25% | 20 | 32% | 38 | 28% |
| Leiomyosarcoma (Cohort B) | 23 | 32% | 22 | 35% | 45 | 34% | |
| Synovial sarcoma (Cohort C) | 10 | 14% | 7 | 11% | 17 | 13% | |
| Other sarcomas (Cohort D) * | 20 | 28% | 14 | 22% | 34 | 25% | |
| Tumor grade (FNCLCC) | Grade 1 | 7 | 10% | 4 | 6% | 11 | 8% |
| Grade 2 | 18 | 25% | 35 | 56% | 53 | 40% | |
| Grade 3 | 34 | 48% | 17 | 27% | 51 | 38% | |
| Not documented | 12 | 17% | 7 | 11% | 19 | 14% | |
| Primary site | Retroperitoneal | 24 | 34% | 24 | 38% | 48 | 36% |
| Lower limb | 19 | 27% | 18 | 29% | 37 | 28% | |
| Uterus | 10 | 14% | 7 | 11% | 17 | 13% | |
| Trunk | 8 | 11% | 3 | 5% | 11 | 8% | |
| Viscera | 3 | 4% | 6 | 10% | 9 | 7% | |
| Upper limb | 1 | 1% | 3 | 5% | 4 | 3% | |
| Head and neck | 4 | 6% | 0 | 0% | 4 | 3% | |
| Other | 2 | 3% | 2 | 3% | 4 | 3% | |
| Staging | Metastatic disease | 69 | 97% | 61 | 97% | 130 | 97% |
| Metastatic sites ** | |||||||
| Lung | 46 | 65% | 48 | 76% | 94 | 70% | |
| Other | 30 | 42% | 31 | 49% | 61 | 46% | |
| Peritoneal | 24 | 34% | 18 | 29% | 42 | 31% | |
| Liver | 21 | 30% | 20 | 32% | 41 | 31% | |
| Lymph nodes | 19 | 27% | 12 | 19% | 31 | 23% | |
| Bones | 11 | 15% | 13 | 21% | 24 | 18% | |
| Cutaneous | 6 | 8% | 6 | 10% | 12 | 9% | |
| Pleural | 5 | 7% | 5 | 8% | 10 | 7% | |
| Prior treatments | Prior surgery | 68 | 96% | 59 | 94% | 127 | 95% |
| Prior radiotherapy | 35 | 49% | 38 | 60% | 73 | 54% | |
| Prior chemotherapy | 71 | 100% | 63 | 100% | 134 | 100% | |
| Number of prior chemotherapy lines (neoadjuvant, adjuvant and advanced) | Median (range) | 2 | (1–4) | 2 | (1–9) | 2 | (1–9) |
| 1 | 24 | 34% | 19 | 30% | 43 | 32% | |
| 2 | 25 | 35% | 24 | 38% | 49 | 37% | |
| 3 | 20 | 28% | 14 | 22% | 34 | 25% | |
| 4 | 2 | 3% | 5 | 8% | 7 | 5% | |
| 9 | 0 | 0% | 1 | 2% | 1 | 1% | |
| Prior treatments (≥10% of patients) *** | Doxorubicin | 71 | 100% | 60 | 95% | 131 | 98% |
| Ifosfamide | 41 | 58% | 37 | 59% | 78 | 58% | |
| Trabectedin | 30 | 42% | 23 | 37% | 53 | 40% | |
| Gemcitabine | 9 | 13% | 18 | 29% | 27 | 20% | |
| Dacarbazine | 6 | 8% | 14 | 22% | 20 | 15% | |
| Gene | Assessment Technique | Overall N = 134 | Lipo-Sarcoma N = 38 | Leiomyo-Sarcoma N = 45 | Synovial Sarcoma N = 17 | Others N = 34 | p |
|---|---|---|---|---|---|---|---|
| TP53 | HotSpot + FCS (1) | 35 (26%) | 5 | 16 | 1 | 13 | 0.009 |
| Angiogenesis pool (2) | 20 (15%) | 7 | 5 | 1 | 7 | 0.42 | |
| H19 | RNA in situ hybridization on TMA | 24 (18%) | 9 | 5 | 7 | 3 | 0.02 |
| Outcome | Prognostic Value (1) | Predictive Value (2) | |||
|---|---|---|---|---|---|
| Gene | HR (Abnormal vs. normal) | P | HR (Rego. vs. plac.) if abnormal | HR (Rego. vs. plac.)if normal | p interaction test |
| Progression-free survival | |||||
| H19 | 1.41 (0.90–2.20) | 0.14 | 0.46 (0.20–1.03) | 0.46 (0.30–0.68) | 1.00 |
| TP53 | 1.08 (0.72–1.60) | 0.72 | 0.40 (0.20–0.80) | 0.48 (0.32–0.74) | 0.64 |
| Angiogenesis pool | 0.96 (0.59–1.57) | 0.88 | 0.52 (0.21–1.31) | 0.45 (0.31–0.67) | 0.79 |
| Overall survival | |||||
| H19 | 1.60 (0.96–2.68) | 0.07 | 1.00 (0.40–2.53) | 0.82 (0.51–1.32) | 0.71 |
| TP53 | 1.03 (0.64–1.65) | 0.89 | 0.67 (0.30–1.49) | 0.92 (0.56–1.52) | 0.51 |
| Angiogenesis pool | 0.73 (0.39–1.38) | 0.33 | 0.83 (0.53–1.31) | 1.07 (0.31–3.66) | 0.71 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brodowicz, T.; Liegl-Atzwanger, B.; Penel, N.; Mir, O.; Blay, J.-Y.; Kashofer, K.; Le Cesne, A.; Decoupigny, E.; Wallet, J.; Hamacher, R.; et al. Assessing Prognostic and Predictive Biomarkers of Regorafenib Response in Patients with Advanced Soft Tissue Sarcoma: REGOSARC Study. Cancers 2020, 12, 3746. https://doi.org/10.3390/cancers12123746
Brodowicz T, Liegl-Atzwanger B, Penel N, Mir O, Blay J-Y, Kashofer K, Le Cesne A, Decoupigny E, Wallet J, Hamacher R, et al. Assessing Prognostic and Predictive Biomarkers of Regorafenib Response in Patients with Advanced Soft Tissue Sarcoma: REGOSARC Study. Cancers. 2020; 12(12):3746. https://doi.org/10.3390/cancers12123746
Chicago/Turabian StyleBrodowicz, Thomas, Bernadette Liegl-Atzwanger, Nicolas Penel, Olivier Mir, Jean-Yves Blay, Karl Kashofer, Axel Le Cesne, Emilie Decoupigny, Jennifer Wallet, Rainer Hamacher, and et al. 2020. "Assessing Prognostic and Predictive Biomarkers of Regorafenib Response in Patients with Advanced Soft Tissue Sarcoma: REGOSARC Study" Cancers 12, no. 12: 3746. https://doi.org/10.3390/cancers12123746






