Impact of BRCA Mutation Status on Tumor Infiltrating Lymphocytes (TILs), Response to Treatment, and Prognosis in Breast Cancer Patients Treated with Neoadjuvant Chemotherapy
Abstract
:Simple Summary
Abstract
1. Introduction
2. Results
2.1. Study Population and Tumors Characteristics
2.2. Response to Treatment and Post-NAC Immune Infiltration
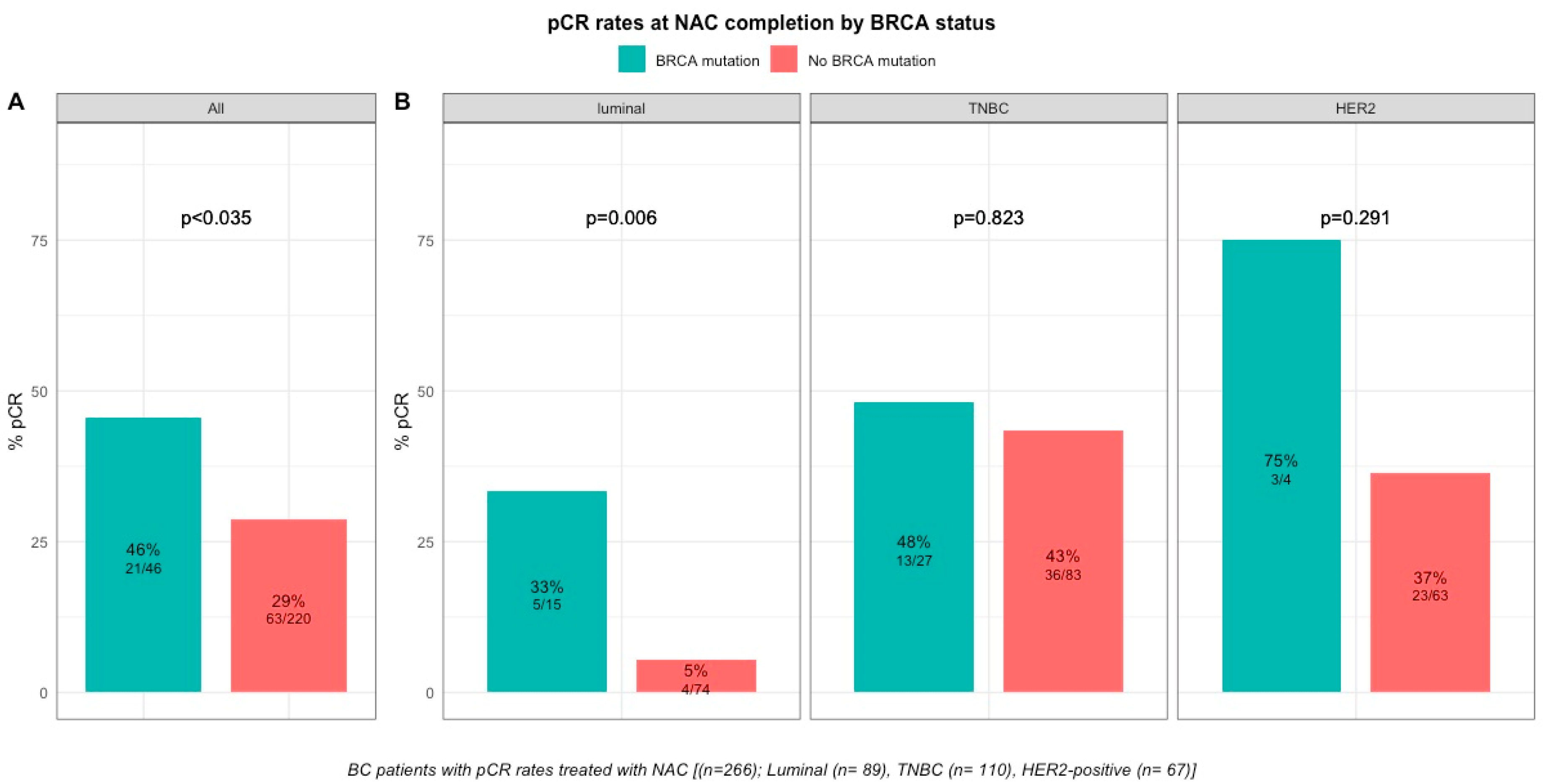
2.2.1. Response to Treatment
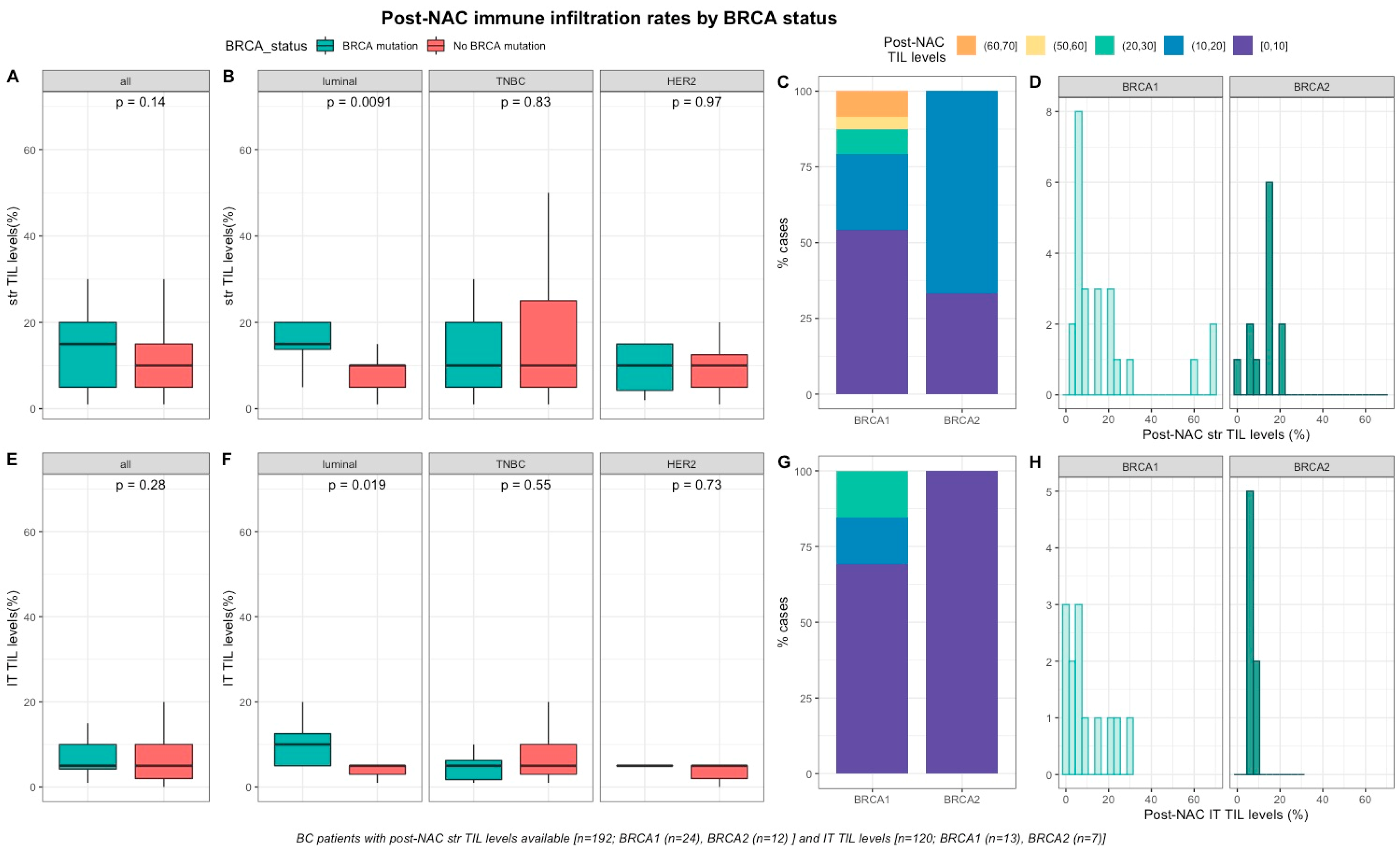
2.2.2. Post-NAC Immune Infiltration by BRCA Status
2.2.3. Survival Analysis
3. Discussion
| Study | Setting/Design | Control Group | Number of Patients (n) | TNBC (n) | HER2-Positive (n) | Luminal (n) | BRCA1 | BRCA2 | BRCA 1 and 2 | Chemotherapy Regimen | sTILS Evaluation | pCR in BRCA-Carriers vs. Non-Carriers | Survival Analyses | Comments |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Byrski (2014) [26] {BCRT | Neoadjuvant epidemiologic prospective cohort | No | 10 | 10 | 0 | 10 | 0 | 0 | 0 | Cis | No | 90% | No | 90% (9/10) in BRCA1-mutated BC patients achieved a pCR after NAC with cisplatin chemotherapy |
| Byrski (2015) [27] HCCP | Neoadjuvant epidemiologic prospective cohort | No | 107 | 82 | 2 | NA | 107 | 0 | 0 | Cis | No | 61% | No | 61% (65/107) in BRCA1-mutated BC patients achieved pCR after NAC with cisplatin chemotherapy. In this study of BRCA1-mutation carriers, a pCR was also achieved in 56% of 16 patients with ER-positive BC. No survival analysis were provided in the current study. |
| Hanhnen (2017) [28] JAMA Oncology | Neoadjuvant secondary analysis of the GeparSixto randomized clinical trial | Yes | 291 | 291 | 0 | 0 | 50 | 0 | P + Dox + Bev ± Cb | No | 66.7% vs. 36.4% | Yes | Patients with BRCA-mutation did not derive a pCR benefit from the addition of carboplatine (65.4% vs. 66.7%) compared to non-BRCA carriers (55% vs. 36.4%). No significant difference in overall prognosis observed in the BRCA-mutated subgroup. | |
| Sharma (2017) [39] CCR | Neoadjuvant prospective, multicenter, non-randomized trial | Yes | 190 | 190 | 0 | 0 | 30 | 0 | Cb + D | No | 59% vs. 56% | No | No significative difference in pCR between BRCA-carriers and WT TNBC (59% and 56%, respectively (p = 0.83)). The Cb-D regimen was well tolerated and yielded high pCR rates in both BRCA associated and WT TNBC. These results are comparable to pCR of previous studies (who investigated pCR after NAC with addition of Cb to AT regimen in TNBC cohort). | |
| Poggio (2018) [20] Annals of Oncology | Neoadjuvant meta-analysis of nine randomized controlled trials | No | 96 | 96 | 0 | 0 | 96 | 0 | P + Dox + Bev ± Cb P + AC ± Cb | No | 54.3% | No | Among 96 BRCA-mutated patients included in 2 controlled trials, the addition of carboplatin was not associated with increased pCR rate (OR 1.17, 95% CI 0.51–2.67, p = 0.711). No survival analyses were available according to BRCA status. | |
| Telli (2019) [25] CCR | Five randomized controlled trials | Yes | 161 | 161 | 0 | 0 | 34 | 0 | Cb + Gem + Iniparib; Cis; Cis + Bev; Cb + Eribulin; Cb + nab-P ± Vorinostat | Yes | No | No | pCR was achieved in 51 (31.7%) patients. In patients with TNBC treated with neoadjuvant platinum-based therapy, iTIL and sTIL densities were not significantly associated with BRCA1/2-mutated tumor status (p = 0.312 and p = 0.391). In multivariate analyses, sTIL density (OR 1.23, 95% CI 0.94–1.61, p = 0.139) was not associated with pCR, but was associated with RCB 0/I status (OR 1.62, 95% CI 1.20–2.28, p = 0.001). | |
| Sønderstrup (2019) [23] Acta Oncologica | Epidemiologic prospective mulitcentric cohort (nationwide) | No | 411 | NA | 24 | NA | 243 | 168 | 0 | NA | Yes | No | Yes | High sTILs (defined as TILs > 60%) were observed in 36% in BRCA1- and 15% in BRCA2-mutated tumors (p < 0.0001). Significant association with survival (OS and DFS) was observed in BRCA1 subgroup. sTILs are an important prognostic factor in BRCA BC and increasing sTILs is associated with a better prognosis. |
| Byrski (2009) [17] JCO | Neoadjuvant Epidemiologic epidemiologic retrospective cohort | No | 102 | NA | 6 | NA | 102 | 0 | 0 | CMF; AT; AC FAC or Cis | No | 23.5% | No | pCR was achieved in 23.5% of 102 patients with a BRCA1 mutation who received NAC. Especially, a complete pCR was observed in 8% (2/25) with AT- regimen (standard of care) compared to 83% (10/12) with cisplatin. |
| Chappuis (2002) [29] JMG | Neoadjuvant Retrospective retrospective multicentric clinical trial | Yes | 38 | NA | NA | NA | 7 | 4 | 0 | FAC; AC; CEF AC + CMF AC + D | No | 44% vs. 4% | No | pCR was achieved in 44% (4/11) of the BRCA-carriers and 4%(1/27) of the non-carriers (p = 0.009). No survival analysis were experienced in this study. |
| Arun (2011) [30] JCO | Neoadjuvant Epidemiologic epidemiologic retrospective cohort | Yes | 317 | 77 | 60 | NA | 57 | 23 | 0 | A-single agent; AT or T-single-agent | No | 46% vs. 22% | Yes | pCR was achieved in 46% of BRCA1-carriers and 13% of BRCA2-carriers and 22% of BRCA non-carriers (<0.001). In the multivariate logistic model, BRCA1 status (OR = 1.96, p = 0.03) remained as independant significant predictors of a pCR. No significant difference in overall prognosis. |
| Wang (2014) [40] Annals of Oncology | Neoadjuvant Epidemiologic retrospective cohort | Yes | 652 | 652 | 0 | 0 | 52 | NA | 0 | A-single agent; AT or T-single-agent | No | 53.8% vs. 29.7% | Yes | The pCR rate was 31.6% in the 652 patients who received NAC. BRCA1 carriers had a significantly higher pCR rate than non-carriers (BRCA1 carriers versus non-carriers, 53.8% versus 29.7%, p < 0.001). Among women treated with anthracycline with or without taxane regimens, the pCR rate was 57.1% for BRCA1 carriers, 29.0% for non-carriers (p < 0.001). The RFS was similar according to BRCA status. |
| Paluch-Shimon(2016) [31] BCRT | Neoadjuvant epidemiologic retrospective cohort | Yes | 80 | 80 | 0 | 0 | 34 | 0 | 0 | AT | No | 68% vs. 37% | Yes | The BRCA1-carriers had pCR rate of 68% compared with 37% among non-carriers, p = 0.01. Yet this did not translate into superior survival for BRCA1 carriers compared with non-carriers. |
| Bignon (2017) [41] Breast | Neoadjuvant epidemiologic retrospective cohort | No | 53 | 53 | 0 | 0 | 46 | 6 | 1 | A-single agent or AT | No | 66% | Yes | The pCR rate was 38.3% [95% CI, 26%–55%] among BRCA1 mutation carriers, and 66% among the 6 BRCA2 mutation carriers. 15 relapses and 6 s cancers were recorded during the follow-up period. 11 deaths occurred, all of which were in the non-pCR group. DFS (p < 0.01) and OS (p < 0.01) were significantly better in the pCR group than the non-pCR group. |
| Wunderle (2018) [18] BCRT | Neoadjuvant Epidemiologic retrospective cohort | Yes | 355 | 138 | 58 | 159 | 43 | 16 | 0 | AT; Cb | No | 54.3% vs. 12.6% | Yes | pCR was observed in 54.3% of BRCA1/2 mutation carriers, but only in 12.6% of non-carriers. The adjusted odds ratio was 2.48 (95% CI 1.26–4.91) for BRCA1/2 carriers versus non-carriers. No difference in overall survival was observed. |
| Saether (2018) [32] HCCP | Neoadjuvant Epidemiologic retrospective cohort | No | 12 | NA | NA | NA | 12 | 0 | 0 | Cis + Dox or Cb + D | No | 83% | No | 11 patients received a combination of cisplatin and doxorubicin, and 1 patient received carboplatin and docetaxel. 83% (10/12) of the BRCA1-carriers achieved pCR. This results were comparable to existing results found in similar studies. No information about BC subtype among the study population and the toxicity of the chemotherapy was not evaluated. |
| Sella (2018) [19] Breast | Neoadjuvant Epidemiologic retrospective cohort | Yes | 43 | 43 | 0 | 0 | 14 | 0 | 0 | AT ± Cb | No | 67% vs. 38% | No | pCR was achieved in 38% in BRCA WT compared to 67% in BRCA-associated TNBC (p = 0.232). No benefit from the addition of carboplatine in BRCA-carriers (64.3% vs. 67%) compared to non-BRCA carriers (44.8% vs. 38%) when compared to historic institutional rates with AT. |
| Solinas (2019) [24] Cancer Letters | Epidemiologic retrospective cohort | Yes | 85 | 85 | 0 | 0 | 38 | 6 | 0 | NA | Yes | No | Yes | The BRCA-mutated tumors had a significantly higher incidence of TIL-positive levels compared to WT (44% and 41%, respectively p = 0.037). No significant difference between BRCA-mutated and WT groups neither in TIL subpopulation nor their location. No difference in I-DFS and OS after stratification on TIL infiltration levels. |
| Our study (2020) | Epidemiologic retrospective cohort | Yes | 267 | 110 | 67 | 90 | 31 | 14 | 1 | A-single agent; AT or T-single-agent | Yes | 45.7% vs 28% | Yes | Among the whole population, 84 tumors achieved a pCR (31.5%). After stratification by BC subtype, pCR rates were significantly higher in luminal BRCA-mutated BCs when compared with WT tumors (33.3% vs. 5.4%, p = 0.006).Pre and post-NAC str or IT TILs were not significantly different between BRCA-carriers and non-carriers in whole population. In the luminal BC, both str and IT post-NAC TIL levels were significantly higher in BRCA-mutated tumors when compared with WT tumors but was no longer significant after multivariate analysis. No difference in RFS or OS between BRCA-mutated and BRCA-WT patients. |
4. Materials and Methods
4.1. Patients and Tumors
4.2. Tumors Samples
4.3. TIL Levels, Pathological Complete Response and Pathological Review
4.4. BRCA Status
4.5. Survival Endpoints
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rastogi, P.; Anderson, S.J.; Bear, H.D.; Geyer, C.E.; Kahlenberg, M.S.; Robidoux, A.; Margolese, R.G.; Hoehn, J.L.; Vogel, V.G.; Dakhil, S.R.; et al. Preoperative chemotherapy: Updates of National Surgical Adjuvant Breast and Bowel Project Protocols B-18 and B-27. J. Clin. Oncol. 2008, 26, 778–785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reyal, F.; Hamy, A.S.; Piccart, M.J. Neoadjuvant treatment: The future of patients with breast cancer. ESMO Open 2018, 3, e000371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luangdilok, S.; Samarnthai, N.; Korphaisarn, K. Association between Pathological Complete Response and Outcome Following Neoadjuvant Chemotherapy in Locally Advanced Breast Cancer Patients. J. Breast Cancer 2014, 17, 376–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, K.-L.; Mashl, R.J.; Wu, Y.; Ritter, D.I.; Wang, J.; Oh, C.; Paczkowska, M.; Reynolds, S.; Wyczalkowski, M.A.; Oak, N.; et al. Pathogenic Germline Variants in 10,389 Adult Cancers. Cell 2018, 173, 355–370.e14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuchenbaecker, K.B.; Hopper, J.L.; Barnes, D.R.; Phillips, K.-A.; Mooij, T.M.; Roos-Blom, M.-J.; Jervis, S.; van Leeuwen, F.E.; Milne, R.L.; Andrieu, N.; et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 2017, 317, 2402–2416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, E.N.; Brianese, R.C.; de Almeida, R.V.B.; Drummond, R.D.; de Souza, J.E.; da Silva, I.T.; de Souza, S.J.; Carraro, D.M. Influence of BRCA1 Germline Mutations in the Somatic Mutational Burden of Triple-Negative Breast Cancer. Transl. Oncol. 2019, 12, 1453–1460. [Google Scholar] [CrossRef]
- Eisinger, F.; Jacquemier, J.; Charpin, C.; Stoppa-Lyonnet, D.; Bressac-de Paillerets, B.; Peyrat, J.P.; Longy, M.; Guinebretière, J.M.; Sauvan, R.; Noguchi, T.; et al. Mutations at BRCA1: The medullary breast carcinoma revisited. Cancer Res. 1998, 58, 1588–1592. [Google Scholar]
- Phillips, K.A. Immunophenotypic and pathologic differences between BRCA1 and BRCA2 hereditary breast cancers. J. Clin. Oncol. 2000, 18, 107S–112S. [Google Scholar]
- Mavaddat, N.; Barrowdale, D.; Andrulis, I.L.; Domchek, S.M.; Eccles, D.; Nevanlinna, H.; Ramus, S.J.; Spurdle, A.; Robson, M.; Sherman, M.; et al. Pathology of breast and ovarian cancers among BRCA1 and BRCA2 mutation carriers: Results from the Consortium of Investigators of Modifiers of BRCA1/2 (CIMBA). Cancer Epidemiol. Biomark. Prev. 2012, 21, 134–147. [Google Scholar] [CrossRef] [Green Version]
- Lakhani, S.R.; Van De Vijver, M.J.; Jacquemier, J.; Anderson, T.J.; Osin, P.P.; McGuffog, L.; Easton, D.F. The pathology of familial breast cancer: Predictive value of immunohistochemical markers estrogen receptor, progesterone receptor, HER-2, and p53 in patients with mutations in BRCA1 and BRCA2. J. Clin. Oncol. 2002, 20, 2310–2318. [Google Scholar] [CrossRef] [Green Version]
- Armes, J.E.; Trute, L.; White, D.; Southey, M.C.; Hammet, F.; Tesoriero, A.; Hutchins, A.M.; Dite, G.S.; McCredie, M.R.; Giles, G.G.; et al. Distinct molecular pathogeneses of early-onset breast cancers in BRCA1 and BRCA2 mutation carriers: A population-based study. Cancer Res. 1999, 59, 2011–2017. [Google Scholar] [PubMed]
- Palacios, J.; Honrado, E.; Osorio, A.; Cazorla, A.; Sarrió, D.; Barroso, A.; Rodríguez, S.; Cigudosa, J.C.; Diez, O.; Alonso, C.; et al. Phenotypic characterization of BRCA1 and BRCA2 tumors based in a tissue microarray study with 37 immunohistochemical markers. Breast Cancer Res. Treat. 2005, 90, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Lakhani, S.R.; Reis-Filho, J.S.; Fulford, L.; Penault-Llorca, F.; van der Vijver, M.; Parry, S.; Bishop, T.; Benitez, J.; Rivas, C.; Bignon, Y.-J.; et al. Prediction of BRCA1 status in patients with breast cancer using estrogen receptor and basal phenotype. Clin. Cancer Res. 2005, 11, 5175–5180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Denkert, C.; Liedtke, C.; Tutt, A.; von Minckwitz, G. Molecular alterations in triple-negative breast cancer-the road to new treatment strategies. Lancet Lond. Engl. 2017, 389, 2430–2442. [Google Scholar] [CrossRef] [Green Version]
- Mancini, P.; Angeloni, A.; Risi, E.; Orsi, E.; Mezi, S. Standard of Care and Promising New Agents for Triple Negative Metastatic Breast Cancer. Cancers 2014, 6, 2187–2223. [Google Scholar] [CrossRef]
- Godet, I.; Gilkes, D.M. BRCA1 and BRCA2 mutations and treatment strategies for breast cancer. Integr. Cancer Sci. Ther. 2017, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Byrski, T.; Gronwald, J.; Huzarski, T.; Grzybowska, E.; Budryk, M.; Stawicka, M.; Mierzwa, T.; Szwiec, M.; Wiśniowski, R.; Siolek, M.; et al. Pathologic Complete Response Rates in Young Women With BRCA1-Positive Breast Cancers After Neoadjuvant Chemotherapy. J. Clin. Oncol. 2009, 28, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Wunderle, M.; Gass, P.; Häberle, L.; Flesch, V.M.; Rauh, C.; Bani, M.R.; Hack, C.C.; Schrauder, M.G.; Jud, S.M.; Emons, J.; et al. BRCA mutations and their influence on pathological complete response and prognosis in a clinical cohort of neoadjuvantly treated breast cancer patients. Breast Cancer Res. Treat. 2018, 171, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Sella, T.; Gal Yam, E.N.; Levanon, K.; Rotenberg, T.S.; Gadot, M.; Kuchuk, I.; Molho, R.B.; Itai, A.; Modiano, T.M.; Gold, R.; et al. Evaluation of tolerability and efficacy of incorporating carboplatin in neoadjuvant anthracycline and taxane based therapy in a BRCA1 enriched triple-negative breast cancer cohort. Breast Edinb. Scotl. 2018, 40, 141–146. [Google Scholar] [CrossRef]
- Poggio, F.; Bruzzone, M.; Ceppi, M.; Pondé, N.F.; La Valle, G.; Del Mastro, L.; De Azambuja, E.; Lambertini, M. Platinum-based neoadjuvant chemotherapy in triple-negative breast cancer: A systematic review and meta-analysis. Ann. Oncol. 2018, 29, 1497–1508. [Google Scholar] [CrossRef]
- Solinas, C.; Ceppi, M.; Lambertini, M.; Scartozzi, M.; Buisseret, L.; Garaud, S.; Fumagalli, D.; de Azambuja, E.; Salgado, R.; Sotiriou, C.; et al. Tumor-infiltrating lymphocytes in patients with HER2-positive breast cancer treated with neoadjuvant chemotherapy plus trastuzumab, lapatinib or their combination: A meta-analysis of randomized controlled trials. Cancer Treat. Rev. 2017, 57, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Solinas, C.; Carbognin, L.; De Silva, P.; Criscitiello, C.; Lambertini, M. Tumor-infiltrating lymphocytes in breast cancer according to tumor subtype: Current state of the art. Breast Edinb. Scotl. 2017, 35, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Sønderstrup, I.M.H.; Jensen, M.B.; Ejlertsen, B.; Eriksen, J.O.; Gerdes, A.M.; Kruse, T.A.; Larsen, M.J.; Thomassen, M.; Laenkholm, A.V. Evaluation of tumor-infiltrating lymphocytes and association with prognosis in BRCA-mutated breast cancer. Acta Oncol. Stockh. Swed. 2019, 58, 363–370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solinas, C.; Marcoux, D.; Garaud, S.; Vitória, J.R.; Van den Eynden, G.; de Wind, A.; De Silva, P.; Boisson, A.; Craciun, L.; Larsimont, D.; et al. BRCA gene mutations do not shape the extent and organization of tumor infiltrating lymphocytes in triple negative breast cancer. Cancer Lett. 2019, 450, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Telli, M.L.; Chu, C.; Badve, S.S.; Vinayak, S.; Silver, D.P.; Isakoff, S.J.; Kaklamani, V.; Gradishar, W.; Stearns, V.; Connolly, R.M.; et al. Association of Tumor Infiltrating Lymphocytes with Homologous Recombination Deficiency and BRCA1/2 Status in Patients with Early Triple-Negative Breast Cancer: A Pooled Analysis. Clin. Cancer Res. 2019. [Google Scholar] [CrossRef]
- Byrski, T.; Huzarski, T.; Dent, R.; Marczyk, E.; Jasiowka, M.; Gronwald, J.; Jakubowicz, J.; Cybulski, C.; Wisniowski, R.; Godlewski, D.; et al. Pathologic complete response to neoadjuvant cisplatin in BRCA1-positive breast cancer patients. Breast Cancer Res. Treat. 2014, 147, 401–405. [Google Scholar] [CrossRef]
- Byrski, T.; Huzarski, T.; Dent, R.; Marczyk, E.; Jasiowka, M.; Gronwald, J.; Jakubowicz, J.; Cybulski, C.; Wisniowski, R.; Godlewski, D.; et al. Pathological complete response to neoadjuvant cisplatin in BRCA1-positive breast cancer patients. Hered. Cancer Clin. Pract. 2015, 13, A8. [Google Scholar] [CrossRef] [Green Version]
- Hahnen, E.; Lederer, B.; Hauke, J.; Loibl, S.; Kröber, S.; Schneeweiss, A.; Denkert, C.; Fasching, P.A.; Blohmer, J.U.; Jackisch, C.; et al. Germline Mutation Status, Pathological Complete Response, and Disease-Free Survival in Triple-Negative Breast Cancer. JAMA Oncol. 2017, 3, 1378–1385. [Google Scholar] [CrossRef]
- Chappuis, P.; Goffin, J.; Wong, N.; Perret, C.; Ghadirian, P.; Tonin, P.; Foulkes, W. A significant response to neoadjuvant chemotherapy in BRCA1/2 related breast cancer. J. Med. Genet. 2002, 39, 608–610. [Google Scholar] [CrossRef] [Green Version]
- Arun, B.; Bayraktar, S.; Liu, D.D.; Gutierrez Barrera, A.M.; Atchley, D.; Pusztai, L.; Litton, J.K.; Valero, V.; Meric-Bernstam, F.; Hortobagyi, G.N.; et al. Response to Neoadjuvant Systemic Therapy for Breast Cancer in BRCA Mutation Carriers and Noncarriers: A Single-Institution Experience. J. Clin. Oncol. 2011, 29, 3739–3746. [Google Scholar] [CrossRef] [Green Version]
- Paluch-Shimon, S.; Cardoso, F.; Sessa, C.; Balmana, J.; Cardoso, M.J.; Gilbert, F.; Senkus, E. Prevention and screening in BRCA mutation carriers and other breast/ovarian hereditary cancer syndromes: ESMO Clinical Practice Guidelines for cancer prevention and screening. Ann. Oncol. 2016, 27, v103–v110. [Google Scholar] [CrossRef] [PubMed]
- Sæther, N.H.; Skuja, E.; Irmejs, A.; Maksimenko, J.; Miklasevics, E.; Purkalne, G.; Gardovskis, J. Platinum-based neoadjuvant chemotherapy in BRCA1-positive breast cancer: A retrospective cohort analysis and literature review. Hered. Cancer Clin. Pract. 2018, 16. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.; Bertucci, A.; Bertucci, F. PARP Inhibitors in the Treatment of Early Breast Cancer: The Step Beyond? Cancers 2020, 12, 1378. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.R.; Dariush, A.; Provenzano, E.; Bardwell, H.; Abraham, J.E.; Iddawela, M.; Vallier, A.-L.; Hiller, L.; Dunn, J.A.; Bowden, S.J.; et al. Computational pathology of pre-treatment biopsies identifies lymphocyte density as a predictor of response to neoadjuvant chemotherapy in breast cancer. Breast Cancer Res. BCR 2016, 18, 21. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.R.; Dariush, A.; Thomas, J.; Provenzano, E.; Dunn, J.; Hiller, L.; Vallier, A.-L.; Abraham, J.; Piper, T.; Bartlett, J.M.S.; et al. Lymphocyte density determined by computational pathology validated as a predictor of response to neoadjuvant chemotherapy in breast cancer: Secondary analysis of the ARTemis trial. Ann. Oncol. 2017, 28, 1832–1835. [Google Scholar] [CrossRef] [PubMed]
- Hamy, A.-S.; Bonsang-Kitzis, H.; De Croze, D.; Laas, E.; Darrigues, L.; Topciu, L.; Menet, E.; Vincent-Salomon, A.; Lerebours, F.; Pierga, J.-Y.; et al. Interaction between Molecular Subtypes and Stromal Immune Infiltration before and after Treatment in Breast Cancer Patients Treated with Neoadjuvant Chemotherapy. Clin. Cancer Res. 2019, 25, 6731–6741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loi: LBA13Relationship between Tumor Infiltrating. Available online: https://scholar.google.com/scholar_lookup?hl=en&volume=28&publication_year=2017&pages=v605-v649&journal=Ann+Oncol&issue=suppl+5&author=S+Loi&author=S+Adams&author=P+Schmid&title=Relationship+between+tumor+infiltrating+lymphocyte+%28TIL%29+levels+and+response+to+pembrolizumab+%28pembro%29+in+metastatic+triple%E2%80%90negative+breast+cancer+%28mTNBC%29%3A+results+from+KEYNOTE%E2%80%90086 (accessed on 22 November 2020).
- Anurag, M.; Zhu, M.; Huang, C.; Vasaikar, S.; Wang, J.; Hoog, J.; Burugu, S.; Gao, D.; Suman, V.; Zhang, X.H.; et al. Immune checkpoint profiles in luminal B breast cancer (Alliance). J. Natl. Cancer Inst. 2020, 112, 737–746. [Google Scholar] [CrossRef]
- Sharma, P.; López-Tarruella, S.; García-Saenz, J.A.; Ward, C.; Connor, C.S.; Gómez, H.L.; Prat, A.; Moreno, F.; Jerez-Gilarranz, Y.; Barnadas, A.; et al. Efficacy of neoadjuvant carboplatin plus docetaxel in triple negative breast cancer: Combined analysis of two cohorts. Clin. Cancer Res. 2017, 23, 649–657. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Zhang, J.; Wang, Y.; Ouyang, T.; Li, J.; Wang, T.; Fan, Z.; Fan, T.; Lin, B.; Xie, Y. Prevalence of BRCA1 mutations and responses to neoadjuvant chemotherapy among BRCA1 carriers and non-carriers with triple-negative breast cancer. Ann. Oncol. 2015, 26, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Bignon, L.; Fricker, J.-P.; Nogues, C.; Mouret-Fourme, E.; Stoppa-Lyonnet, D.; Caron, O.; Lortholary, A.; Faivre, L.; Lasset, C.; Mari, V.; et al. Efficacy of anthracycline/taxane-based neo-adjuvant chemotherapy on triple-negative breast cancer in BRCA1/BRCA2 mutation carriers. Breast J. 2018, 24, 269–277. [Google Scholar] [CrossRef]
- Yadav, S.; Ladkany, R.; Yadav, D.; Alhalabi, O.; Khaddam, S.; Isaac, D.; Cardenas, P.Y.; Zakalik, D. Impact of BRCA Mutation Status on Survival of Women With Triple-negative Breast Cancer. Clin. Breast Cancer 2018, 18, e1229–e1235. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Gou, Q.; Wang, Q.; Zhong, X.; Zheng, H. The role of BRCA status on prognosis in patients with triple-negative breast cancer. Oncotarget 2017, 8, 87151–87162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Copson, E.R.; Maishman, T.C.; Tapper, W.J.; Cutress, R.I.; Greville-Heygate, S.; Altman, D.G.; Eccles, B.; Gerty, S.; Durcan, L.T.; Jones, L.; et al. Germline BRCA mutation and outcome in young-onset breast cancer (POSH): A prospective cohort study. Lancet Oncol. 2018, 19, 169–180. [Google Scholar] [CrossRef] [Green Version]
- Brekelmans, C.T.M.; Tilanus-Linthorst, M.M.A.; Seynaeve, C.; vd Ouweland, A.; Menke-Pluymers, M.B.E.; Bartels, C.C.M.; Kriege, M.; van Geel, A.N.; Burger, C.W.; Eggermont, A.M.M.; et al. Tumour characteristics, survival and prognostic factors of hereditary breast cancer from BRCA2-, BRCA1- and non-BRCA1/2 families as compared to sporadic breast cancer cases. Eur. J. Cancer Oxf. Engl. 1990 2007, 43, 867–876. [Google Scholar] [CrossRef] [PubMed]
- Templeton, A.J.; Gonzalez, L.D.; Vera-Badillo, F.E.; Tibau, A.; Goldstein, R.; Šeruga, B.; Srikanthan, A.; Pandiella, A.; Amir, E.; Ocana, A. Interaction between Hormonal Receptor Status, Age and Survival in Patients with BRCA1/2 Germline Mutations: A Systematic Review and Meta-Regression. PLoS ONE 2016, 11, e0154789. [Google Scholar] [CrossRef] [PubMed]
- Kurian, A.W.; Ward, K.C.; Howlader, N.; Deapen, D.; Hamilton, A.S.; Mariotto, A.; Miller, D.; Penberthy, L.S.; Katz, S.J. Genetic Testing and Results in a Population-Based Cohort of Breast Cancer Patients and Ovarian Cancer Patients. J. Clin. Oncol. 2019, 37, 1305–1315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riaz, N.; Blecua, P.; Lim, R.S.; Shen, R.; Higginson, D.S.; Weinhold, N.; Norton, L.; Weigelt, B.; Powell, S.N.; Reis-Filho, J.S. Pan-cancer analysis of bi-allelic alterations in homologous recombination DNA repair genes. Nat. Commun. 2017, 8, 857. [Google Scholar] [CrossRef]
- Recommendations for the immunohistochemistry of the hormonal receptors on paraffin sections in breast cancer. Update 1999. Group for Evaluation of Prognostic Factors using Immunohistochemistry in Breast Cancer (GEFPICS-FNCLCC). Ann. Pathol. 1999, 19, 336–343.
- Salgado, R.; Denkert, C.; Demaria, S.; Sirtaine, N.; Klauschen, F.; Pruneri, G.; Wienert, S.; Van den Eynden, G.; Baehner, F.L.; Penault-Llorca, F.; et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: Recommendations by an International TILs Working Group 2014. Ann. Oncol. 2015, 26, 259–271. [Google Scholar] [CrossRef]
- Dieci, M.V.; Radosevic-Robin, N.; Fineberg, S.; van den Eynden, G.; Ternes, N.; Penault-Llorca, F.; Pruneri, G.; D’Alfonso, T.M.; Demaria, S.; Castaneda, C.; et al. Update on tumor-infiltrating lymphocytes (TILs) in breast cancer, including recommendations to assess TILs in residual disease after neoadjuvant therapy and in carcinoma in situ: A report of the International Immuno-Oncology Biomarker Working Group on Breast Cancer. Semin. Cancer Biol. 2018, 52, 16–25. [Google Scholar] [CrossRef]
- Cardoso, F.; Kyriakides, S.; Ohno, S.; Penault-Llorca, F.; Poortmans, P.; Rubio, I.T.; Zackrisson, S.; Senkus, E. ESMO Guidelines Committee Early breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 1194–1220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cardoso, F.; van’t Veer, L.J.; Bogaerts, J.; Slaets, L.; Viale, G.; Delaloge, S.; Pierga, J.Y.; Brain, E.; Causeret, S.; DeLorenzi, M.; et al. 70-Gene Signature as an Aid to Treatment Decisions in Early-Stage Breast Cancer. N. Engl. J. Med. 2016, 375, 717–729. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Characteristics | Class | All | BRCA Mutation | BRCA Wild-Type | Not Screened | p |
|---|---|---|---|---|---|---|
| n = 1199 (100%) | n = 46 (3.8%) | n = 221 (18.4%) | n = 932 (77.7%) | |||
| Age (mean) | 48.6 | 39.5 | 41.7 | 50.6 | <0.01 | |
| Menopausal Status | pre | 747 (62.8) | 41 (89.1%) | 187 (85.0%) | 519 (56.2%) | <0.01 |
| post | 442 (37.2) | 5 (10.9%) | 33 (15.0%) | 404 (43.8%) | ||
| BMI (mean) | 24.7 | 22.8 | 23.6 | 25.1 | <0.01 | |
| BMI class | (15,19] | 72 (6.0) | 6 (13.3) | 17 (7.7) | 49 (5.3) | <0.01 |
| (19,25] | 664 (55.7) | 31 (68.9) | 147 (66.5) | 486 (52.4) | ||
| (25,30] | 299 (25.1) | 4 (8.9) | 43 (19.5) | 252 (27.2) | ||
| (30,50] | 158 (13.2) | 4 (8.9) | 14 (6.3) | 140 (15.1) | ||
| Family history of BC | no | 887 (74.2) | 12 (26.1%) | 104 (47.7%) | 771 (82.7%) | <0.01 |
| yes | 309 (25.8) | 34 (73.9%) | 114 (52.3%) | 161 (17.3%) | ||
| Clinical tumor size | T1 | 70 (5.8%) | 5 (10.9%) | 22 (10.0%) | 43 (4.6%) | <0.01 |
| T2 | 798 (66.6%) | 28 (60.9%) | 153 (69.2%) | 617 (66.3%) | ||
| T3 | 330 (27.5%) | 13 (28.3%) | 46 (20.8%) | 271 (29.1%) | ||
| Clinical | N0 | 525 (43.8%) | 17 (37.0%) | 93 (42.1%) | 415 (44.6%) | 0.51 |
| nodal status | N1-N2-N3 | 673 (56.2%) | 29 (63.1%) | 128 (57.9%) | 516 (55.4%) | |
| Histology | NST | 1062 (90%) | 43 (93.5%) | 213 (96.4%) | 806 (88.3%) | 0.03 |
| others | 118 (10%) | 3 (6.5%) | 8 (3.6%) | 108 (11.6%) | ||
| Grade | I-II | 479 (41.4%) | 10 (23.3%) | 76 (34.7%) | 393 (43.9%) | 0.01 |
| III | 678 (58.6%) | 33 (76.7%) | 143 (65.3%) | 502 (56.1%) | ||
| Mitotic Index (mean) | 25.1 | 30.8 | 25.6 | 24.6 | 0.25 | |
| Subtype | luminal | 528 (44.0%) | 15 (32.6%) | 75 (33.9%) | 438 (47.0%) | <0.01 |
| TNBC | 376 (31.4%) | 27 (58.7%) | 83 (37.6%) | 266 (28.5%) | ||
| HER2 | 295 (24.6%) | 4 (8.7%) | 63 (28.5%) | 228 (24.5%) | ||
| str TILs (mean) | 20.0 [10.0–30.0] | 20.0 [13.8–40.0] | 20.0 [10.0–40.0] | 15.0 [10.0–30.0] | 0.02 | |
| IT TILs (mean) | 5.0 [5.0–15.0] | 5.0 [5.0–11.2] | 7.5 [5.0–20.0] | 5.0 [3.0–15.0] | 0,47 | |
| NAC Regimen | AC | 235 (19.6%) | 4 (8.7%) | 25 (11.4%) | 206 (22.2%) | <0.01 |
| AC-Taxanes | 845 (70.7%) | 41 (89.1%) | 180 (81.8%) | 624 (67.1%) | ||
| Taxanes | 25 (2.1%) | 1 (2.2%) | 6 (2.7%) | 18 (1.9%) | ||
| Others | 91 (7.6%) | 0 (0.0%) | 9 (4.1%) | 82 (8.8%) | ||
| pCR class | No pCR | 911 (76.2) | 25 (54.3) | 157 (71.4) | 729 (78.4) | <0.001 |
| pCR | 285 (23.8) | 21 (45.7) | 63 (28.6) | 201 (21.6) | ||
| Nodal involvment | 0 | 682 (57.0) | 35 (76.1) | 141 (64.1) | 506 (54.4) | 0.003 |
| 1–3 | 341 (28.5) | 6 (13.0) | 58 (26.4) | 277 (29.8) | ||
| ≥4 | 174 (14.5) | 5 (10.9) | 21 (9.5) | 148 (15.9) | ||
| str TILs (mean) | 10.0 [5.0–15.0] | 15.0 [5.0–20.0] | 10.0 [5.0–15.0] | 10.0 [5.0–15.0] | 0.36 | |
| IT TILs (mean) | 5.0 [2.0–10.0] | 5.0 [4.3–10.0] | 5.0 [2.0–10.0] | 5.0 [2.0–10.0] | 0.57 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grandal, B.; Evrevin, C.; Laas, E.; Jardin, I.; Rozette, S.; Laot, L.; Dumas, E.; Coussy, F.; Pierga, J.-Y.; Brain, E.; et al. Impact of BRCA Mutation Status on Tumor Infiltrating Lymphocytes (TILs), Response to Treatment, and Prognosis in Breast Cancer Patients Treated with Neoadjuvant Chemotherapy. Cancers 2020, 12, 3681. https://doi.org/10.3390/cancers12123681
Grandal B, Evrevin C, Laas E, Jardin I, Rozette S, Laot L, Dumas E, Coussy F, Pierga J-Y, Brain E, et al. Impact of BRCA Mutation Status on Tumor Infiltrating Lymphocytes (TILs), Response to Treatment, and Prognosis in Breast Cancer Patients Treated with Neoadjuvant Chemotherapy. Cancers. 2020; 12(12):3681. https://doi.org/10.3390/cancers12123681
Chicago/Turabian StyleGrandal, Beatriz, Clémence Evrevin, Enora Laas, Isabelle Jardin, Sonia Rozette, Lucie Laot, Elise Dumas, Florence Coussy, Jean-Yves Pierga, Etienne Brain, and et al. 2020. "Impact of BRCA Mutation Status on Tumor Infiltrating Lymphocytes (TILs), Response to Treatment, and Prognosis in Breast Cancer Patients Treated with Neoadjuvant Chemotherapy" Cancers 12, no. 12: 3681. https://doi.org/10.3390/cancers12123681
APA StyleGrandal, B., Evrevin, C., Laas, E., Jardin, I., Rozette, S., Laot, L., Dumas, E., Coussy, F., Pierga, J.-Y., Brain, E., Saule, C., Stoppa-Lyonnet, D., Frank, S., Sénéchal, C., Lae, M., De Croze, D., Bataillon, G., Guerin, J., Reyal, F., & Hamy, A.-S. (2020). Impact of BRCA Mutation Status on Tumor Infiltrating Lymphocytes (TILs), Response to Treatment, and Prognosis in Breast Cancer Patients Treated with Neoadjuvant Chemotherapy. Cancers, 12(12), 3681. https://doi.org/10.3390/cancers12123681






