Women’s Intentions to Engage in Risk-Reducing Behaviours after Receiving Personal Ovarian Cancer Risk Information: An Experimental Survey Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Results
2.1. Sample Characteristics
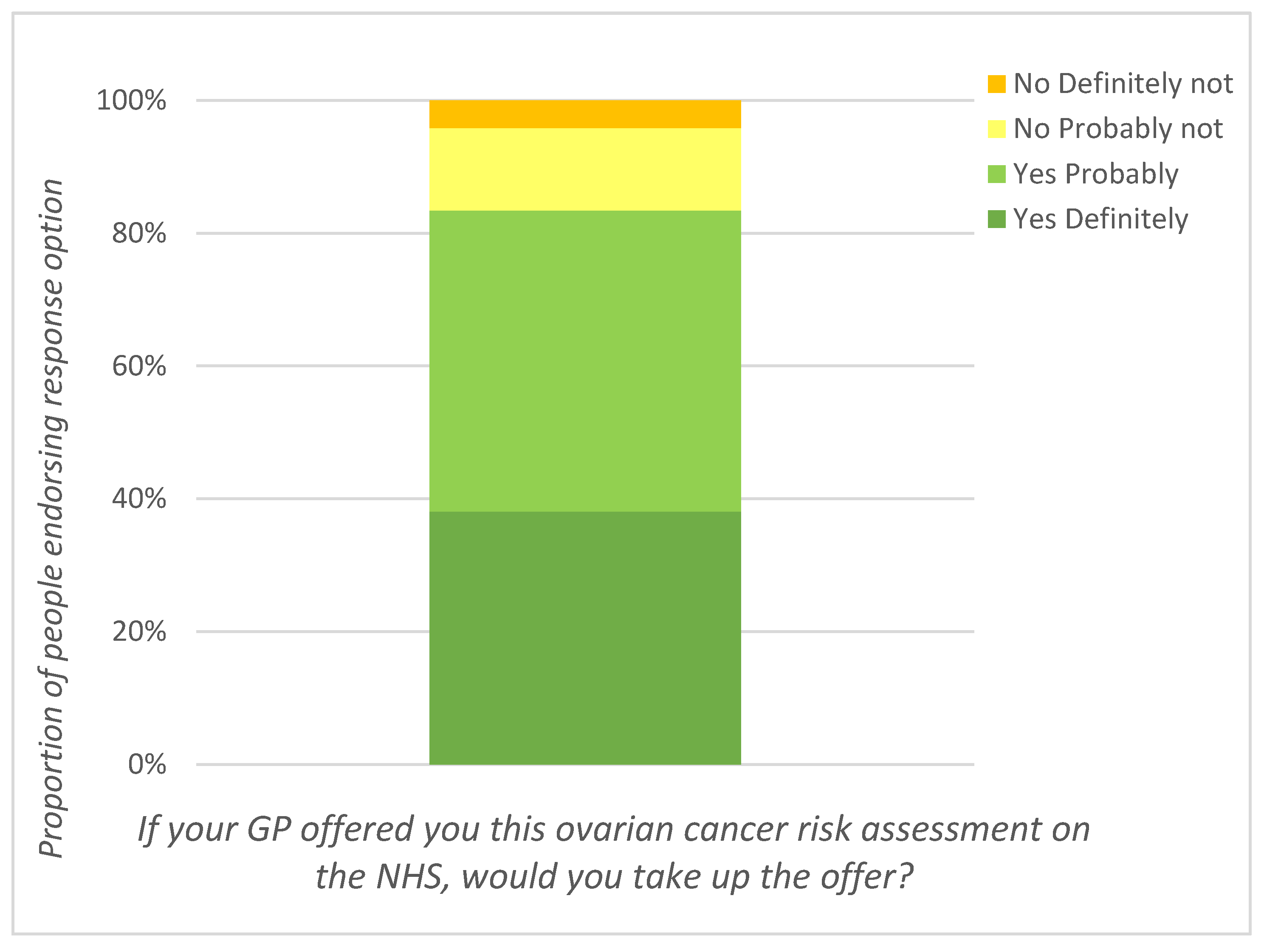
2.2. Interest in Ovarian Cancer Risk Assessment
2.3. Behavioural Response to Personalised Ovarian Cancer Risk Information
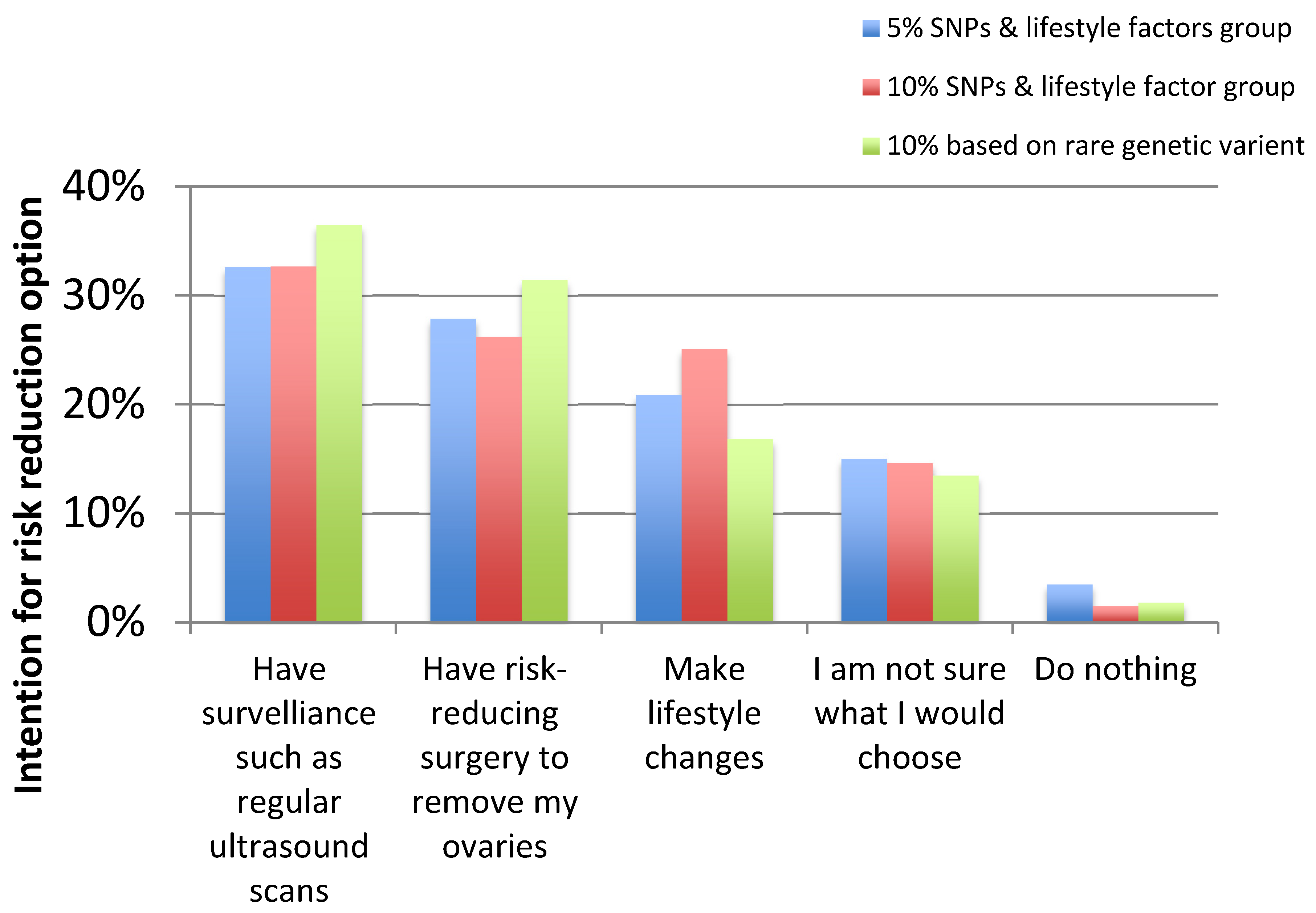
2.4. Differences by Experimental Condition
2.5. EPPM Variables
2.6. Intention to Have Risk-Reducing Surgery (RRSO) versus Other Risk-Management Options
3. Discussion
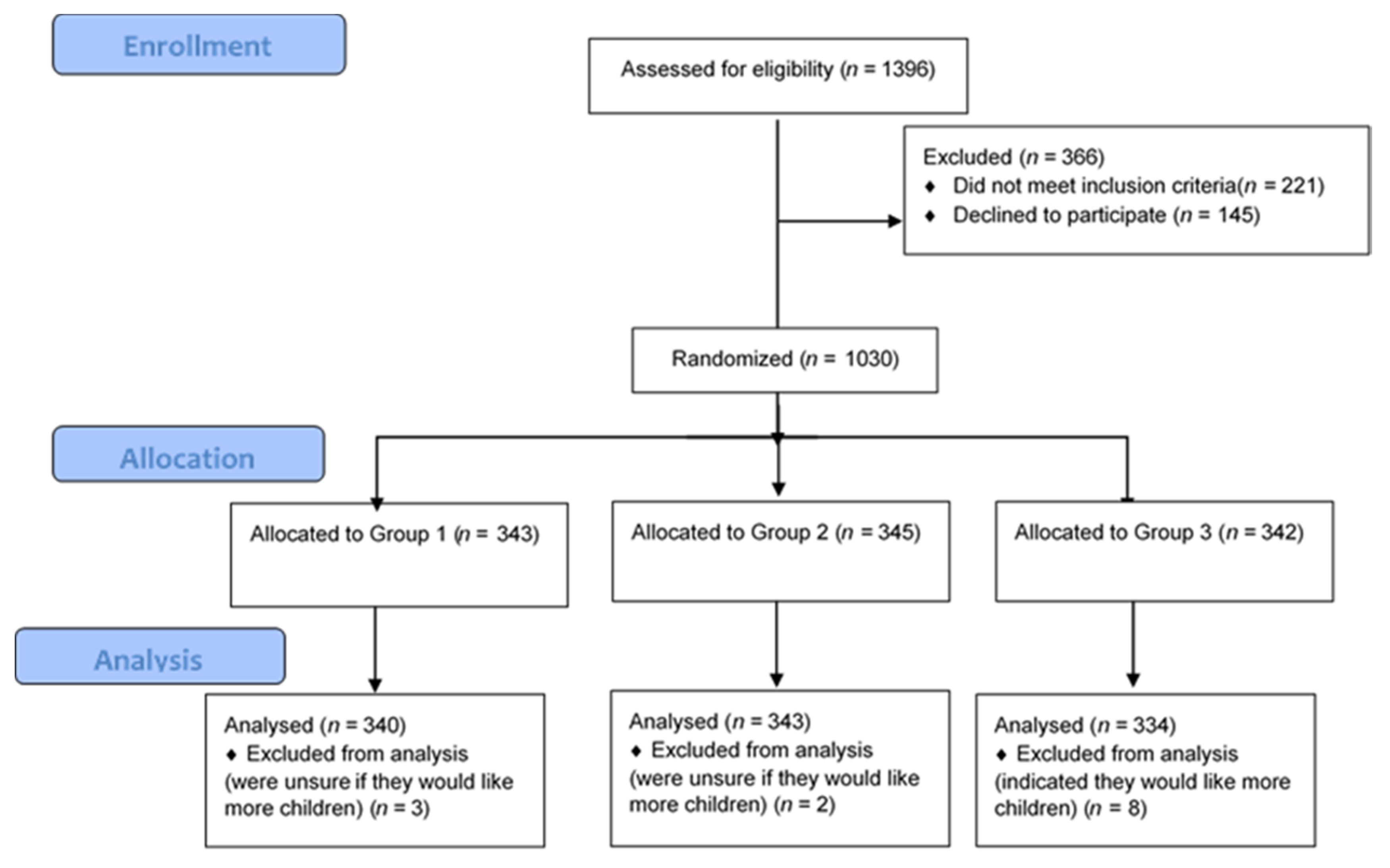
4. Materials and Methods
4.1. Overview
4.2. Inclusion Criteria
4.3. Measures
4.3.1. Interest in Ovarian Cancer Risk Assessment
4.3.2. Perceived Risk of Ovarian Cancer
4.3.3. Perceived Severity of Ovarian Cancer
4.3.4. Self-Efficacy for Risk-Reducing Surgery
4.3.5. Perceived Response-Efficacy of Risk-Reducing Surgery
4.3.6. Behavioural Intention
4.3.7. Demographic and Health Characteristic Measures
4.4. Data Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Ovarian cancer and risk information
Section 1: Hypothetical scenario
| Please imagine that you have gone to your GP, and they have offered you a new approach to assessing your risk of developing ovarian cancer in the future. When your GP offered this to you, they gave you some written information to help you decide whether or not you want to have the risk assessment done. This information is below. Please read the information, and then answer the question that follows. |
Assessing ovarian cancer risk
| Ovarian cancer is the sixth most common cancer among women in the UK: 2% of women will be diagnosed with ovarian cancer during their lifetime. Ovarian cancer is caused by many genetic and non-genetic factors. Currently, ovarian cancer is often detected at a late stage because symptoms are hard to spot. This means that it is often very hard to treat effectively. A new ovarian cancer risk assessment has been developed. This risk assessment combines lots of different types of information about you to estimate how likely you are to develop ovarian cancer in your lifetime. The types of information included in the risk assessment include lifestyle factors, rare genetic variants, and common genetic variants. Lifestyle factors: Lifestyle factors that may increase a woman’s risk of ovarian cancer include tobacco smoking and being overweight. Common genetic variants: Single nucleotide polymorphisms, frequently called SNPs (pronounced “snips”), are the most common type of genetic variation among people. SNPs occur normally throughout a person’s DNA. Most SNPs have no effect on health, but some are important to a person’s health. Some SNPs can influence a woman’s risk of developing ovarian cancer. Individually, each one of these SNPs only influences ovarian cancer risk by a tiny amount, but if a woman has a large number of these SNPs then her risk of ovarian cancer may be increased. Rare genetic variants: Some ovarian cancers are caused by a rare variant in a person’s DNA. These rare variants can have quite a strong effect on a woman’s risk of developing ovarian cancer. For example, variants in the BRCA2 gene can increase a woman’s lifetime risk of ovarian cancer up to between 10% and 20%. If you want to have this ovarian cancer risk assessment carried out, you will need to provide your GP with the information they request, including about your lifestyle. You will also need to have a blood test, so that the scientists can see whether you have any of the genetic variants that increase your risk. |
Appendix B
Generic Risk Scenarios
| Next, regardless of how you answered in the previous question, please imagine that you have had the ovarian cancer risk assessment done, and your GP has now given you the result from the assessment. Please read your hypothetical result below. |
Your personal ovarian cancer risk assessment: results [Group 1 only]
| Your result indicates that your lifetime risk of developing ovarian cancer is 5%. This is higher than the average risk for women, which is 2%. Your risk of ovarian cancer is higher than average because you have been found to have at least one lifestyle factor and several common genetic variants which are known to put women at increased risk of developing ovarian cancer. |
Your personal ovarian cancer risk assessment: results [Group 2 only]
| Your result indicates that your lifetime risk of developing ovarian cancer is 10%. This is higher than the average risk for women, which is 2%. Your risk of ovarian cancer is higher than average because you have been found to have at least one lifestyle factor and several common genetic variants which are known to put women at increased risk of developing ovarian cancer. |
Your personal ovarian cancer risk assessment: results [Group 3 only]
| Your result indicates that your lifetime risk of developing ovarian cancer is 10%. This is higher than the average risk for women, which is 2%. Your risk of ovarian cancer is higher than average because you have been found to have a rare genetic variant which is known to put women at increased risk of developing ovarian cancer. |
[All groups]
| There are several options for women who are at higher than average risk of ovarian cancer. Risk-reducing surgery involves removing the ovaries and fallopian tubes to prevent ovarian cancer from developing. However, removing the ovaries has downsides. For example, it causes a woman who has not yet been through her menopause to start her menopause (a natural process that usually happens in a woman’s early 50 s). Surveillance includes having a regular (e.g., annual) ultrasound of your ovaries to see if a tumour is present. This ultrasound is usually an internal (transvaginal) ultrasound. Effective screening has not been established. Lifestyle changes include maintaining a healthy weight and quitting smoking. These types of lifestyle changes may reduce a woman’s risk of developing ovarian cancer. |
Appendix C
Questionnaire
| Please carefully imagine what you would think and how you would feel if you had received this personal result from the ovarian cancer risk assessment. Now please answer the following questions. |
Intention for Genetic Screening
- No definitely not
- No probably not
- Yes probably
- Yes definitely
- (a)
- …Not at all likely
- (b)
- …Not very likely
- (c)
- …Quite likely
- (d)
- …Extremely likely
Behavioural outcome
- (a)
- …have risk-reducing surgery to remove my ovaries.
- (b)
- …have surveillance such as regular ultrasound scans.
- (c)
- …make lifestyle changes.
- (d)
- …do nothing.
- (e)
- …I am not sure what I would choose.
Perceived risk
- (a)
- …much lower than other women of my age
- (b)
- …lower than other women of my age
- (c)
- …the same as other women of my age
- (d)
- …higher than other women of my age
- (e)
- …much higher than other women of my age
Perceived Response efficacy
| How much do you agree or disagree with the following statement based on how you would feel if you had received this personal ovarian cancer risk result from the ovarian cancer risk assessment. |
- (a)
- …Strongly agree
- (b)
- …Agree
- (c)
- …Neither agree or disagree
- (d)
- …Disagree
- (e)
- …Strongly disagree
- (a)
- …Strongly agree
- (b)
- …Agree
- (c)
- …Neither agree or disagree
- (d)
- …Disagree
- (e)
- …Strongly disagree
Perceived Response efficacy
- (a)
- …Strongly agree
- (b)
- …Agree
- (c)
- …Neither agree or disagree
- (d)
- …Disagree
- (e)
- …Strongly disagree
Perceived severity
- (a)
- …Strongly agree
- (b)
- …Agree
- (c)
- …Neither agree or disagree
- (d)
- …Disagree
- (e)
- …Strongly disagree
Perceived severity
- (a)
- …Strongly agree
- (b)
- …Agree
- (c)
- …Neither agree or disagree
- (d)
- …Disagree
- (e)
- …Strongly disagree
Self-efficacy
- (a)
- …not at all confident
- (b)
- …somewhat confident
- (c)
- …fairly confident
- (d)
- …very confident
- (e)
- …extremely confident
- Yes
- No
- Not sure
- Yes
- No
- Not sure
- Premenopausal (before menopause; having regular periods)
- Perimenopause (menopause transition—changes in periods, but have not gone 12 months in a row without a period)
- Postmenopausal (After menopause; periods have stopped for at least 12 months)Don’t know
- Spontaneous (“natural”)
- Surgical (removal of both ovaries)
- Due to chemotherapy or radiation therapy
- Other
- n/a—haven’t yet reached menopause(If indicated they are still having periods don’t ask this question)
- I’m up to date with cervical screening
- I’m overdue for cervical screening
- I’ve never been for cervical screening
- I’m 65 or over so I’m not invited any more
- I’m up to date with breast screening
- I’m overdue for breast screening
- I’ve never been for breast screening
- I’m under 50 or over 70 so I’m not eligible for breast screening.
- White British
- White non-British
- Black
- Asian
- Mixed
- Other
- Do not wish to answer
- No formal qualifications
- GCSEs/O levels or equivalent
- A-Levels or equivalent
- Undergraduate degree or equivalent
- Postgraduate degree or equivalent
- Other (please state)
- Single
- In a relationship
- Living with a partner
- Married
- Separated/divorced/widowed
- 0
- 1
- 2
- 3
- 4
- 5 or more
- Yes
- No
- Not sure
References
- Cancer Research UK, Ovarian Cancer Statistics. 2016. Available online: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/ovarian-cancer (accessed on 25 June 2017).
- Hunn, J.; Rodriguez, G.C. Ovarian Cancer: Etiology, risk factors, and epidemiology. Clin. Obstet. Gynecol. 2012, 55, 3–23. [Google Scholar] [CrossRef] [PubMed]
- Levy-Lahad, E.; Gabai-Kapara, E.; Kaufman, B.; Catane, R.; Segev, S.; Renbaum, P.; Beller, U.; King, M.; Lahad, A. Identification of BRCA1/BRCA2 carriers by screening in the healthy population and its implications. J. Clin. Oncol. 2011, 29, 1513. [Google Scholar] [CrossRef]
- King, M.-C.; Levy-Lahad, E.; Lahad, A. Population-Based Screening for BRCA1 and BRCA2. JAMA 2014, 312, 1091–1092. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuchenbaecker, K.B.; Hopper, J.L.; Barnes, D.R.; Phillips, K.-A.; Mooij, T.M.; Roos-Blom, M.-J.; Jervis, S.E.; Van Leeuwen, F.; Milne, R.L.; Andrieu, N.; et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 2017, 317, 2402–2416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manchanda, R.; Burnell, M.; Gaba, F.; Desai, R.; Wardle, J.; Gessler, S.; Side, L.; Sanderson, S.; Loggenberg, K.; Brady, A.F.; et al. Randomised trial of population-based BRCA testing in Ashkenazi Jews: Long-term outcomes. BJOG: Int. J. Obstet. Gynaecol. 2019, 127, 364–375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manchanda, R.; Loggenberg, K.; Sanderson, S.; Burnell, M.; Wardle, J.; Gessler, S.; Side, L.; Balogun, N.; Desai, R.; Kumar, A.; et al. Population testing for cancer predisposing BRCA1/BRCA2 mutations in the Ashkenazi-Jewish community: A randomized controlled trial. J. Natl. Cancer Inst. 2014, 107, 379. [Google Scholar] [CrossRef] [Green Version]
- Manchanda, R.; Blyuss, O.; Gaba, F.; Gordeev, V.S.; Jacobs, C.; Burnell, M.; Gan, C.; Taylor, R.; Turnbull, C.; Legood, R.; et al. Current detection rates and time-to-detection of all identifiable BRCA carriers in the Greater London population. J. Med. Genet. 2018, 55, 538–545. [Google Scholar] [CrossRef]
- Jervis, S.; Song, H.; Lee, A.; Dicks, E.; Harrington, P.; Baynes, C.; Manchanda, R.; Easton, D.F.; Jacobs, I.; Pharoah, P.P.D.; et al. A risk prediction algorithm for ovarian cancer incorporating BRCA1, BRCA2, common alleles and other familial effects. J. Med. Genet. 2015, 52, 465–475. [Google Scholar] [CrossRef] [Green Version]
- Manchanda, R.; Gaba, F. Population Based Testing for Primary Prevention: A Systematic Review. Cancers 2018, 10, 424. [Google Scholar] [CrossRef] [Green Version]
- Evans, O.; Gaba, F.; Manchanda, R. Population-based genetic testing for Women’s cancer prevention. Best Pr. Res. Clin. Obstet. Gynaecol. 2020, 65, 139–153. [Google Scholar] [CrossRef]
- Gaba, F.; Blyuss, O.; Liu, X.; Goyal, S.; Lahoti, N.; Chandrasekaran, D.; Kurzer, M.; Kalsi, J.K.; Sanderson, S.C.; Lanceley, A.; et al. Population Study of Ovarian Cancer Risk Prediction for Targeted Screening and Prevention. Cancers 2020, 12, 1241. [Google Scholar] [CrossRef] [PubMed]
- Rahman, B.; Side, L.; Gibbon, S.; Meisel, S.F.; Fraser, L.; Gessler, S.; Wardle, J.; Lanceley, A. Moving towards population-based genetic risk prediction for ovarian cancer. BJOG: Int. J. Obstet. Gynaecol. 2017, 124, 855–858. [Google Scholar] [CrossRef] [Green Version]
- Manchanda, R.; Legood, R.; Antoniou, A.; Pearce, L.; Menon, U. Commentary on changing the risk threshold for surgical prevention of ovarian cancer. BJOG: Int. J. Obstet. Gynaecol. 2017, 125, 541–544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manchanda, R.; Menon, U. Setting the Threshold for Surgical Prevention in Women at Increased Risk of Ovarian Cancer. Int. J. Gynecol. Cancer 2018, 28, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Manchanda, R.; Legood, R.; Antoniou, A.C.; Gordeev, V.S.; Menon, U. Specifying the ovarian cancer risk threshold of ‘premenopausal risk-reducing salpingo-oophorectomy’ for ovarian cancer prevention: A cost-effectiveness analysis. J. Med. Genet. 2016, 53, 591–599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manchanda, R.; Legood, R.; Pearce, L.; Menon, U. Defining the risk threshold for risk reducing salpingo-oophorectomy for ovarian cancer prevention in low risk postmenopausal women. Gynecol. Oncol. 2015, 139, 487–494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manchanda, R.; Patel, S.; Gordeev, V.S.; Antoniou, A.C.; Smith, S.; Lee, A.; Hopper, J.L.; MacInnis, R.J.; Turnbull, C.G.N.; Ramus, S.J.; et al. Cost-effectiveness of Population-Based BRCA1, BRCA2, RAD51C, RAD51D, BRIP1, PALB2 Mutation Testing in Unselected General Population Women. J. Natl. Cancer Inst. 2018, 110, 714–725. [Google Scholar] [CrossRef]
- Chandrasekaran, D.; Manchanda, R. Germline and somatic genetic testing in ovarian cancer patients. BJOG: Int. J. Obstet. Gynaecol. 2018, 125, 1460. [Google Scholar] [CrossRef] [Green Version]
- Meisel, S.F.; for the PROMISE-2016 Study Team; Rahman, B.; Side, L.; Fraser, L.; Gessler, S.; Lanceley, A.; Wardle, J. Genetic testing and personalized ovarian cancer screening: A survey of public attitudes. BMC Women’s Health 2016, 16, 46. [Google Scholar] [CrossRef] [Green Version]
- Manchanda, R.; Burnell, M.; Loggenberg, K.; Desai, R.; Wardle, J.; Sanderson, S.C.; Gessler, S.; Side, L.; Balogun, N.; Kumar, A.; et al. Cluster-randomised non-inferiority trial comparing DVD-assisted and traditional genetic counselling in systematic population testing for BRCA1/2 mutations. J. Med. Genet. 2016, 53, 472–480. [Google Scholar] [CrossRef] [Green Version]
- Manchanda, R.; Legood, R.; Burnell, M.; McGuire, A.; Raikou, M.; Loggenberg, K.; Wardle, J.; Sanderson, S.; Gessler, S.; Side, L.; et al. Cost-effectiveness of Population Screening for BRCA Mutations in Ashkenazi Jewish Women Compared with Family History-Based Testing. J. Natl. Cancer Inst. 2015, 107, 380. [Google Scholar] [CrossRef] [PubMed]
- Manchanda, R.; Patel, S.; Antoniou, A.C.; Levy-Lahad, E.; Turnbull, C.; Evans, D.G.; Hopper, J.L.; MacInnis, R.J.; Menon, U.; Jacobs, I.; et al. Cost-effectiveness of population based BRCA testing with varying Ashkenazi Jewish ancestry. Am. J. Obstet. Gynecol. 2017, 217, 578.e1–578.e12. [Google Scholar] [CrossRef] [PubMed]
- Rebbeck, T.R.; Lynch, H.T.; Neuhausen, S.L.; Narod, S.A.; Veer, L.V.; Garber, J.E.; Evans, G.; Isaacs, C.; Daly, M.B.; Matloff, E.; et al. Prophylactic Oophorectomy in Carriers of BRCA1 or BRCA2 Mutations. N. Engl. J. Med. 2002, 346, 1616–1622. [Google Scholar] [CrossRef] [PubMed]
- Manchanda, R.; Burnell, M.; Abdelraheim, A.; Johnson, M.; Sharma, A.; Benjamin, E.; Brunell, C.; Saridogan, E.; Gessler, S.; Oram, D.; et al. Factors influencing uptake and timing of risk reducing salpingo-oophorectomy in women at risk of familial ovarian cancer: A competing risk time to event analysis. BJOG: Int. J. Obstet. Gynaecol. 2012, 119, 527–536. [Google Scholar] [CrossRef]
- Manchanda, R.; Abdelraheim, A.; Johnson, M.; Rosenthal, A.N.; Benjamin, E.; Brunell, C.; Burnell, M.; Side, L.; Gessler, S.; Saridogan, E.; et al. Outcome of risk-reducing salpingo-oophorectomy in BRCA carriers and women of unknown mutation status. BJOG: Int. J. Obstet. Gynaecol. 2011, 118, 814–824. [Google Scholar] [CrossRef]
- Yang, X.; Song, H.; Leslie, G.; Engel, C.; Hahnen, E.; Auber, B.; Horváth, J.; Kast, K.; Niederacher, D.; Turnbull, C.; et al. Ovarian and Breast Cancer Risks Associated with Pathogenic Variants in RAD51C and RAD51D. J. Natl. Cancer Inst. 2020. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Leslie, G.; Doroszuk, A.; Schneider, S.; Allen, J.; Decker, B.; Dunning, A.M.; Redman, J.; Scarth, J.; Plaskocinska, I.; et al. Cancer Risks Associated with Germline PALB2 Pathogenic Variants: An International Study of 524 Families. J. Clin. Oncol. 2020, 38, 674–685. [Google Scholar] [CrossRef]
- Ramus, S.J.; Song, H.; Dicks, E.; Tyrer, J.P.; Rosenthal, A.N.; Intermaggio, M.P.; Fraser, L.; Gentry-Maharaj, A.; Hayward, J.; Philpott, S.; et al. Germline Mutations in the BRIP1, BARD1, PALB2, and NBN Genes in Women with Ovarian Cancer. J. Natl. Cancer Inst. 2015, 107. [Google Scholar] [CrossRef]
- Jacobs, I.; Menon, U.; Ryan, A.; Gentry-Maharaj, A.; Burnell, M.; Kalsi, J.K.; Amso, N.N.; Apostolidou, S.; Benjamin, E.; Cruickshank, D.; et al. Ovarian cancer screening and mortality in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): A randomised controlled trial. Lancet 2016, 387, 945–956. [Google Scholar] [CrossRef] [Green Version]
- Rosenthal, A.N.; Fraser, L.S.; Philpott, S.; Manchanda, R.; Burnell, M.; Badman, P.; Hadwin, R.; Rizzuto, I.; Benjamin, E.C.; Singh, N.; et al. Evidence of Stage Shift in Women Diagnosed with Ovarian Cancer during Phase II of the United Kingdom Familial Ovarian Cancer Screening Study. J. Clin. Oncol. 2017, 35, 1411–1420. [Google Scholar] [CrossRef]
- Kauff, N.D.; Domchek, S.M.; Friebel, T.M.; Robson, M.E.; Lee, J.; Garber, J.E.; Isaacs, C.; Evans, D.G.; Lynch, H.; Eeles, R.A.; et al. Risk-Reducing Salpingo-Oophorectomy for the Prevention of BRCA1- and BRCA2-Associated Breast and Gynecologic Cancer: A Multicenter, Prospective Study. J. Clin. Oncol. 2008, 26, 1331–1337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hollands, G.J.; French, D.P.; Griffin, S.J.; Prevost, A.T.; Sutton, S.; King, S.; Marteau, T.M. The impact of communicating genetic risks of disease on risk-reducing health behaviour: Systematic review with meta-analysis. BMJ 2016, 352, i1102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hallowell, N.; kConFab Psychosocial Group on Behalf of the kConFab Investigators; Baylock, B.; Heiniger, L.; Butow, P.N.; Patel, D.; Meiser, B.; Saunders, C.M.; Price, M.A. Looking different, feeling different: Women’s reactions to risk-reducing breast and ovarian surgery. Fam. Cancer 2011, 11, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Meiser, B.; Butow, P.; Friedlander, M.; Barratt, A.; Schnieden, V.; Watson, M.; Brown, J.; Tucker, K. Psychological impact of genetic testing in women from high-risk breast cancer families. Eur. J. Cancer 2002, 38, 2025–2031. [Google Scholar] [CrossRef]
- Pruthi, S.; Gostout, B.S.; Lindor, N.M. Identification and Management of Women with BRCA Mutations or Hereditary Predisposition for Breast and Ovarian Cancer. Mayo Clinic Proc. 2010, 85, 1111–1120. [Google Scholar] [CrossRef] [Green Version]
- Rebbeck, T.R.; Kauff, N.D.; Domchek, S.M. Meta-analysis of Risk Reduction Estimates Associated with Risk-Reducing Salpingo-oophorectomy in BRCA1 or BRCA2 Mutation Carriers. J. Natl. Cancer Inst. 2009, 101, 80–87. [Google Scholar] [CrossRef]
- Marteau, T.M.; French, D.P.; Griffin, S.J.; Prevost, T.; Sutton, S.; Watkinson, C.; Attwood, S.; Hollands, G.J. Effects of communicating DNA-based disease risk estimates on risk-reducing behaviours. Cochrane Database Syst. Rev. 2010, CD007275. [Google Scholar] [CrossRef]
- Cameron, L.D.; Sherman, K.; Marteau, T.M.; Brown, P.M. Impact of genetic risk information and type of disease on perceived risk, anticipated affect, and expected consequences of genetic tests. Health Psychol. 2009, 28, 307–316. [Google Scholar] [CrossRef]
- French, D.P.; Southworth, J.; Howell, A.; Harvie, M.; Stavrinos, P.; Watterson, D.; Sampson, S.; Evans, D.G.; Donnelly, L.S. Psychological impact of providing women with personalised 10-year breast cancer risk estimates. Br. J. Cancer 2018, 118, 1648–1657. [Google Scholar] [CrossRef] [Green Version]
- Hart, M.R.; Biesecker, B.B.; Blout, C.L.; Christensen, K.D.; Amendola, L.M.; Bergstrom, K.L.; Biswas, S.; Bowling, K.M.; Brothers, K.B.; Conlin, L.K.; et al. Secondary findings from clinical genomic sequencing: Prevalence, patient perspectives, family history assessment, and health-care costs from a multisite study. Genet. Med. 2019, 21, 1100–1110. [Google Scholar] [CrossRef]
- Witte, K.; Allen, M. A Meta-Analysis of Fear Appeals: Implications for Effective Public Health Campaigns. Heal. Educ. Behav. 2000, 27, 591–615. [Google Scholar] [CrossRef] [PubMed]
- Meisel, S.F.; Side, L.; Fraser, L.; Gessler, S.; Wardle, J.; Lanceley, A. Population-Based, Risk-Stratified Genetic Testing for Ovarian Cancer Risk: A Focus Group Study. Public Health Genom. 2013, 16, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Waller, J.L.; Osborne, K.; Wardle, J.F.C. Enthusiasm for cancer screening in Great Britain: A general population survey. Br. J. Cancer 2015, 112, 562–566. [Google Scholar] [CrossRef] [PubMed]
- Mai, P.L.; Piedmonte, M.; Han, P.K.J.; Moser, R.P.; Walker, J.L.; Rodriguez, G.C.; Boggess, J.; Rutherford, T.J.; Zivanovic, O.; Cohn, D.E.; et al. Factors associated with deciding between risk-reducing salpingo-oophorectomy and ovarian cancer screening among high-risk women enrolled in GOG-0199: An NRG Oncology/Gynecologic Oncology Group study. Gynecol. Oncol. 2017, 145, 122–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Witte, K. Putting the fear back into fear appeals: The extended parallel process model. Commun. Monogr. 1992, 59, 329–349. [Google Scholar] [CrossRef]
- Claassen, L.; Henneman, L.; De Vet, R.; Knol, D.; Marteau, T.M.; Timmermans, D. Fatalistic responses to different types of genetic risk information: Exploring the role of Self-Malleability. Psychol. Health 2010, 25, 183–196. [Google Scholar] [CrossRef] [PubMed]
- Marteau, T.M. Genetic risk and behavioural change. BMJ 2001, 322, 1056–1059. [Google Scholar] [CrossRef] [Green Version]
- Marteau, T.M.; Weinman, J. Self-regulation and the behavioural response to DNA risk information: A theoretical analysis and framework for future research. Soc. Sci. Med. 2006, 62, 1360–1368. [Google Scholar] [CrossRef]
- Evans, R.E.; Beeken, R.J.; Steptoe, A.; Wardle, J. Cancer information and anxiety: Applying the Extended Parallel Process Model. J. Health Psychol. 2012, 17, 579–589. [Google Scholar] [CrossRef]
- Meisel, S.F.; Freeman, M.; Waller, J.; Fraser, L.; Gessler, S.; Jacobs, I.; Kalsi, J.K.; Manchanda, R.; Rahman, B.; Side, L.; et al. Impact of a decision aid about stratified ovarian cancer risk-management on women’s knowledge and intentions: A randomised online experimental survey study. BMC Public Health 2017, 17, 882. [Google Scholar] [CrossRef] [Green Version]
- Evans, O.; Manchanda, R. Population-based Genetic Testing for Precision Prevention. Cancer Prev. Res. 2020, 13, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Manchanda, R.; Lieberman, S.; Gaba, F.; Lahad, A.; Levy-Lahad, E. Population Screening for Inherited Predisposition to Breast and Ovarian Cancer. Annu. Rev. Genom. Hum. Genet. 2020, 21, 373–412. [Google Scholar] [CrossRef] [PubMed]
- Gurmankin, A.D.; Shea, J.; Williams, S.V.; Quistberg, D.A.; Armstrong, K. Measuring Perceptions of Breast Cancer Risk. Cancer Epidemiol. Biomarkers Prev. 2006, 15, 1893–1898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peipins, L.A.; Mccarty, F.; Hawkins, N.A.; Rodriguez, J.L.; Scholl, L.E.; Leadbetter, S. Cognitive and affective influences on perceived risk of ovarian cancer. Psycho-Oncology 2015, 24, 279–286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rawl, S.; Champion, V.; Menon, U.; Loehrer, P.J.; Vance, G.H.; Skinner, C.S. Validation of Scales to Measure Benefits of and Barriers to Colorectal Cancer Screening. J. Psychosoc. Oncol. 2001, 19, 47–63. [Google Scholar] [CrossRef]
| Variables | Group 1: 5% SNPs and Lifestyle (n = 340) | Group 2: 10% SNPs and Lifestyle (n = 343) | Group 3: 10% Rare Genetic Variant (n = 334) |
|---|---|---|---|
| Demographics | n (%) | ||
| Age Mean (SD) | 57.43 (8.32) | 57.37 (7.78) | 58.08 (8.19) |
| Age group | |||
| 45–50 | 89 (26.2) | 82 (23.9) | 73 (21.9) |
| 51–55 | 71 (20.9) | 66 (19.2) | 67 (20.1) |
| 56–60 | 59 (17.4) | 70 (20.4) | 65 (19.5) |
| 61–65 | 53 (15.6) | 70 (20.4) | 61 (18.3) |
| 66–70 | 42 (12.4) | 34 (9.9) | 38 (11.4) |
| 71–75 | 26 (7.6) | 21 (6.1) | 30 (9.0) |
| Ethnicity | |||
| White (Any background) | 327 (96.2) | 324 (94.5) | 321 (96.1) |
| Other ethnic group | 11 (3.2) | 17 (5.0) | 11 (3.3) |
| Educational Attainment | |||
| No Formal Qualification | 26 (7.6) | 23 (6.7) | 17 (5.1) |
| GCSE or equivalent | 115 (33.8) | 126 (36.7) | 111 (33.2) |
| A-Levels or equivalent | 75 (22.1) | 80 (23.3) | 87 (26.0) |
| Undergraduate degree/equivalent | 89 (26.2) | 77 (22.4) | 79 (23.7) |
| Postgraduate degree/equivalent | 31 (9.1) | 30 (8.7) | 31 (9.3) |
| Other | 4 (1.2) | 7 (2.0) | 9 (2.7) |
| Relationship Status | |||
| Married/Cohabiting/In a relationship | 245 (72.1) | 232 (67.6) | 233 (69.8) |
| Single/Separated/divorced/widowed | 93 (27.4) | 111 (32.4) | 99 (29.6) |
| Health Characteristics | |||
| Menopause status | |||
| Premenopausal | 41 (12.1) | 37 (10.8) | 43 (12.9) |
| During/post menopause | 283 (83.2) | 298 (86.9) | 285 (85.3) |
| Personal History of Cancer | |||
| Yes | 17 (5.0) | 16 (4.7) | 17 (5.1) |
| No/Not sure | 323 (95.0) | 327 (95.3) | 317 (94.9) |
| Family History of Cancer | |||
| Yes | 214 (62.4%) | 206 (59.7%) | 201 (58.8%) |
| No/Not sure | 129 (37.6%) | 139 (40.3%) | 141 (41.2%) |
| Cervical Screening | |||
| Regular | 192 (72.7) | 214 (76.7) | 179 (67.8) |
| Irregular | 72 (27.3) | 65 (23.3) | 85 (32.2) |
| Not eligible | 58 (17.1) | 44 (12.8) | 62 (18.6) |
| Breast Screening | |||
| Regular | 196 (81.0) | 201 (79.4) | 196 (79.7) |
| Irregular | 46 (19.0) | 52 (20.6) | 50 (20.3) |
| Not eligible | 72 (21.2) | 59 (17.2) | 61 (18.3) |
| Interest in ovarian cancer risk assessment | |||
| Yes Definitely | 139 (40.9) | 122 (35.6) | 125 (37.4) |
| Yes Probably | 151 (44.4) | 156 (45.5) | 153 (45.8) |
| No Probably not | 36 (10.6) | 46 (13.4) | 46 (13.8) |
| No definitely not | 14 (4.1) | 19 (5.5) | 10 (3.0) |
| Variable | Intention to Have Risk-Reducing Surgery | Odds Ratios (95% CI) | |
|---|---|---|---|
| Unadjusted | Adjusted | ||
| Demographic Factors | n (%) | ||
| Age | |||
| 45–50 | 74 (30.3) | Ref | Ref |
| 51–55 | 65 (31.9) | 1.07 (0.72–1.61) | 0.94 (0.51–1.74) |
| 56–60 | 60 (30.9) | 1.03 (0.68–1.55) | 1.00 (0.53–1.87) |
| 61–65 | 49 (26.6) | 0.83 (0.55–1.28) | 0.62 (0.32–1.19) |
| 66–70 | 27 (23.7) | 0.71 (0.43–1.19) | 0.48 (0.14–1.65) |
| 71–75 | 15 (19.5) | 0.56 (0.30–1.04) | 0.29 (0.06–1.50) |
| Ethnicity | |||
| White (Any background) | 278 (28.6) | Ref | Ref |
| Other ethnic group | 11 (28.2) | 0.98 (0.48–2.00) | 1.16 (0.38–3.50) |
| Educational Attainment | |||
| No formal qualifications | 16 (24.2) | Ref | |
| GCSE/O Levels | 111 (31.5) | 1.44 (0.79–2.64) | |
| A-Levels or Equivalent | 68 (28.1) | 1.22 (0.68–2.29) | |
| Undergraduate degree | 59 (24.1) | 0.99 (0.53–1.87) | |
| Postgraduate degree | 28 (30.4) | 1.37 (0.67–2.80) | |
| Other | 8 (40.0) | 2.08 (0.72–6.00) | |
| Relationship Status | |||
| Not married/in a relationship | 82 (27.1) | Ref | |
| Married/in a relationship | 207 (29.2) | 1.11 (0.82–1.50) | |
| Health history | |||
| Cervical Screening Attendance (n = 807) | |||
| Regular | 200 (34.2) | Ref | Ref |
| Irregular | 49 (22.1) | 0.59 (0.41–0.85) * | 0.67 (0.41–1.11) |
| Breast Screening Attendance (n = 741) | |||
| Regular | 205 (34.6) | Ref | Ref |
| Irregular | 35 (23.6) | 0.53 (0.36–0.80) * | 0.67 (0.38–1.17) |
| Menopause status | |||
| Pre-menopause | 35 (28.9) | Ref | Ref |
| Peri/Post-menopause | 243 (28.1) | 0.96 (0.63–1.46) | |
| Extended Parallel Processing Model Variables | |||
| Perceived Risk | 1.43 (1.19–1.71) ** | 1.14 (0.90–1.45) | |
| Perceived Severity | 1.42 (1.11–1.82) * | 1.08 (0.74–1.57) | |
| Self-Efficacy | 2.19 (1.94–2.47) ** | 1.90 (1.63–2.22) ** | |
| Perceived Response Efficacy | 3.15 (2.50–3.96) ** | 2.22 (1.64–3.00) ** | |
| Experimental condition | |||
| 5% SNPs & Lifestyle | 95 (32.8) | Ref | Ref |
| 10% SNPS & Lifestyle | 90 (31.0) | 0.92 (0.66–1.29) | 1.08 (0.67–1.73) |
| 10% rare genetic variant | 105 (36.2) | 1.18 (0.85–1.65) | 1.87 (1.17–3.00) ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallagher, A.; Waller, J.; Manchanda, R.; Jacobs, I.; Sanderson, S. Women’s Intentions to Engage in Risk-Reducing Behaviours after Receiving Personal Ovarian Cancer Risk Information: An Experimental Survey Study. Cancers 2020, 12, 3543. https://doi.org/10.3390/cancers12123543
Gallagher A, Waller J, Manchanda R, Jacobs I, Sanderson S. Women’s Intentions to Engage in Risk-Reducing Behaviours after Receiving Personal Ovarian Cancer Risk Information: An Experimental Survey Study. Cancers. 2020; 12(12):3543. https://doi.org/10.3390/cancers12123543
Chicago/Turabian StyleGallagher, Ailish, Jo Waller, Ranjit Manchanda, Ian Jacobs, and Saskia Sanderson. 2020. "Women’s Intentions to Engage in Risk-Reducing Behaviours after Receiving Personal Ovarian Cancer Risk Information: An Experimental Survey Study" Cancers 12, no. 12: 3543. https://doi.org/10.3390/cancers12123543
APA StyleGallagher, A., Waller, J., Manchanda, R., Jacobs, I., & Sanderson, S. (2020). Women’s Intentions to Engage in Risk-Reducing Behaviours after Receiving Personal Ovarian Cancer Risk Information: An Experimental Survey Study. Cancers, 12(12), 3543. https://doi.org/10.3390/cancers12123543






