Magnetic Particles for CTC Enrichment
Abstract
:Simple Summary
Abstract
1. Introduction
2. Immunomagnetic Particles for Rare Cell Enrichment
3. The Characteristics of an Ideal Magnetic Particle for Cell Separation
- the volume of a single magnetic nanoparticle (Vp);
- the magnetic susceptibility of the magnetic nanoparticle (Δχp);
- the magnetic field generated by the external magnetic flux source (B).
Superparamagnetic and Magnetic Susceptibility
4. Synthesis Methods of MNPs
4.1. Co-Precipitation
4.2. Thermal Decomposition
4.3. Solvothermal
4.4. Microemulsion
5. Methods to Bind Targeting Ligands on Magnetic Particles
5.1. Antibodies and Ligands Used for CTC Enrichment
5.2. Binding Antibodies to Magnetic Particles
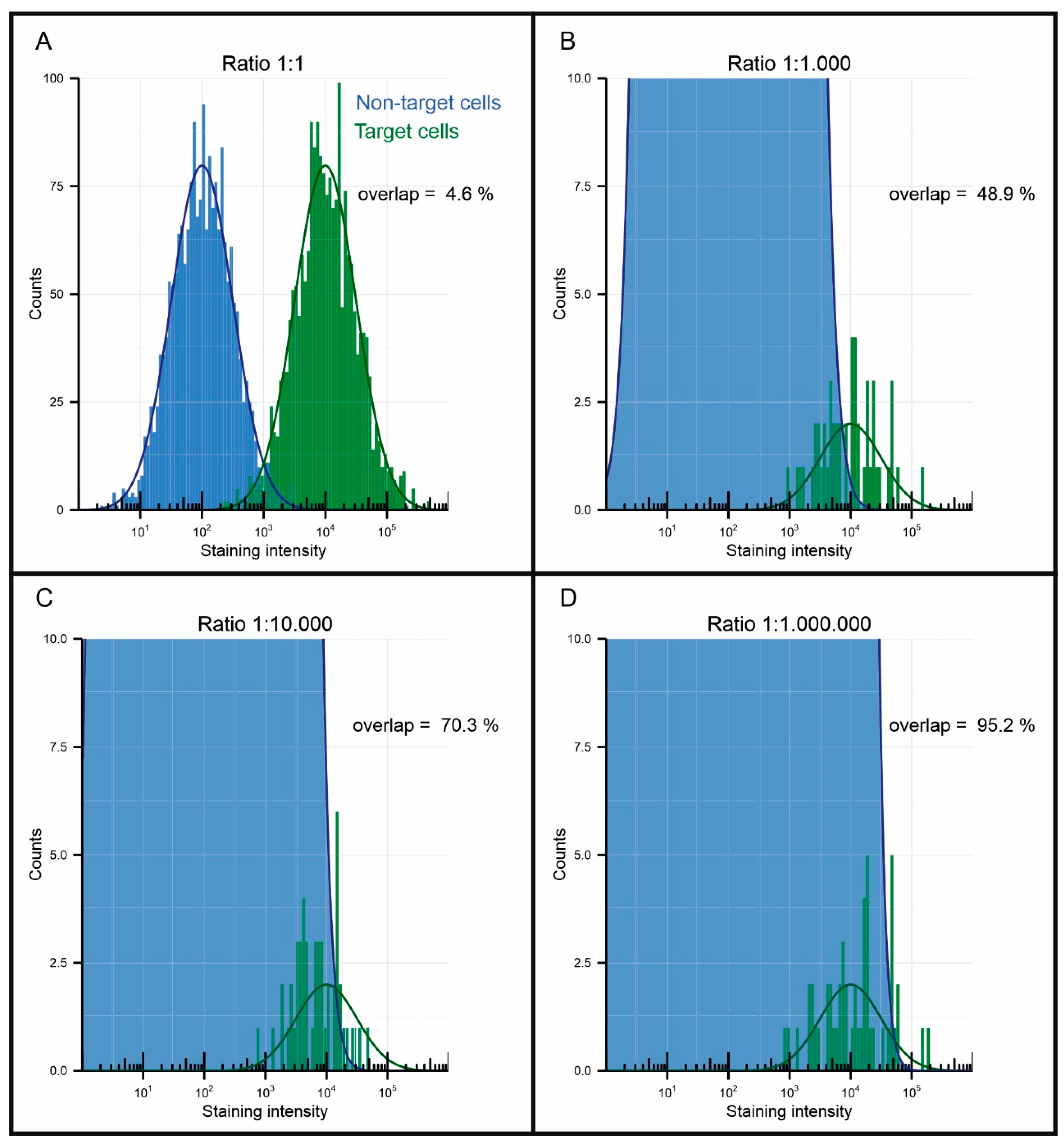
6. Characterization of Magnetic Particle Performance
Low Non-Specific Binding and No Agglomeration
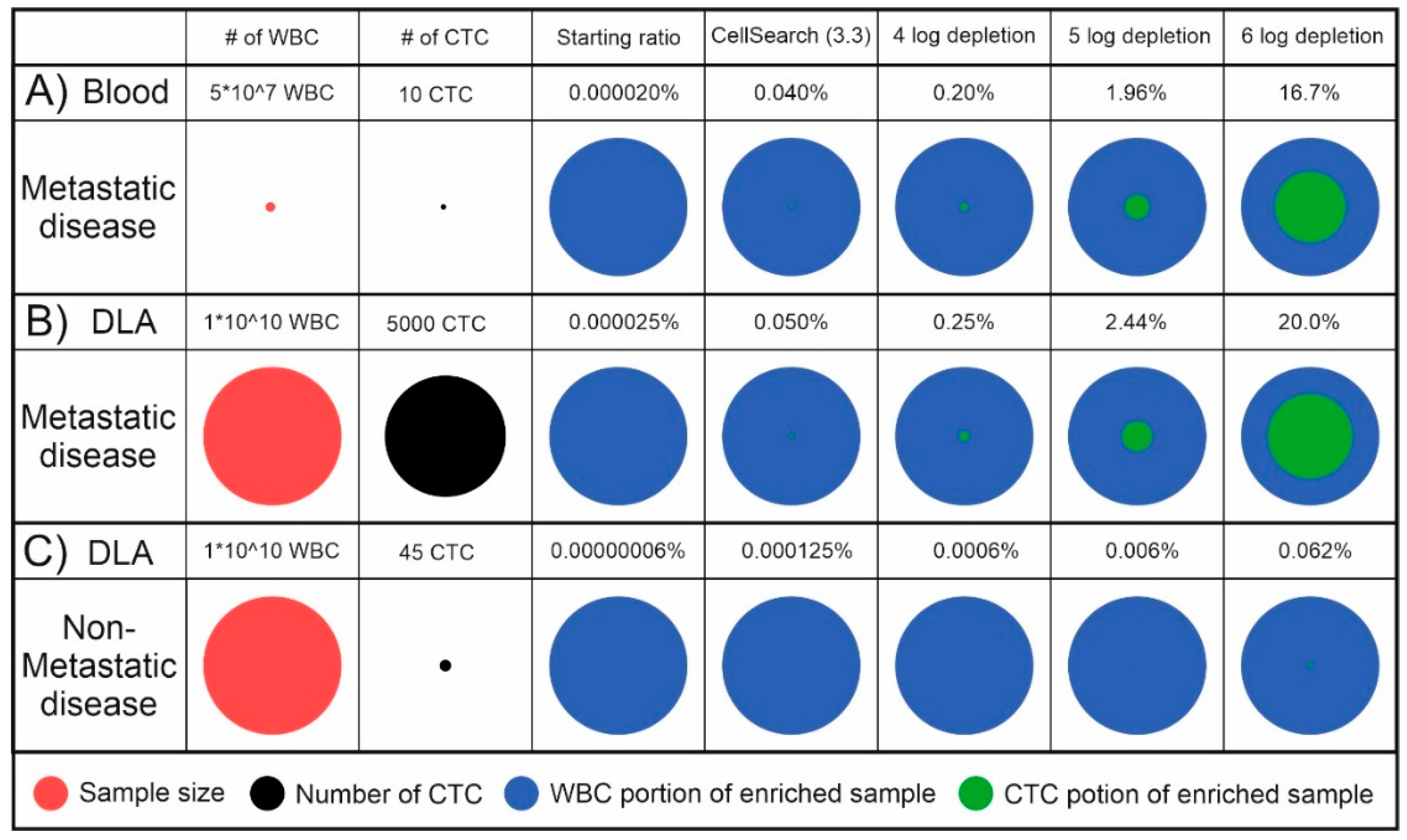
7. Separation of Magnetic Particles in Systems for CTC Enumeration
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dean, R.S.; Davis, C.W. Magnetic Separation of Ores; US Government Printing Office: Washington, DC, USA, 1941.
- Melville, D.; Paul, F.; Roath, S. Direct magnetic separation of red cells from whole blood. Nature 1975, 255, 706-706. [Google Scholar] [CrossRef] [PubMed]
- Molday, R.S.; Yen, S.P.S.; Rembaum, A. Application of magnetic microspheres in labelling and separation of cells. Nature 1977, 268, 437–438. [Google Scholar] [CrossRef]
- Coumans, F.; Terstappen, L. Detection and Characterization of Circulating Tumor Cells by the CellSearch Approach. In Whole Genome Amplification: Methods and Protocols; Kroneis, T., Ed.; Springer: New York, NY, USA, 2015; pp. 263–278. [Google Scholar]
- Masud, M.K.; Na, J.; Younus, M.; Hossain, M.S.A.; Bando, Y.; Shiddiky, M.J.A.; Yamauchi, Y. Superparamagnetic nanoarchitectures for disease-specific biomarker detection. Chem. Soc. Rev. 2019, 48, 5717–5751. [Google Scholar] [CrossRef] [PubMed]
- Majidi, S.; Zeinali Sehrig, F.; Farkhani, S.M.; Soleymani Goloujeh, M.; Akbarzadeh, A. Current methods for synthesis of magnetic nanoparticles. Artif. Cells Nanomed. Biotechnol. 2016, 44, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Carregal-Romero, S.; Casula, M.F.; Gutiérrez, L.; Morales, M.P.; Böhm, I.B.; Heverhagen, J.T.; Prosperi, D.; Parak, W.J. Biological applications of magnetic nanoparticles. Chem. Soc. Rev. 2012, 41, 4306–4334. [Google Scholar] [CrossRef] [PubMed]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic Iron Oxide Nanoparticles: Synthesis, Stabilization, Vectorization, Physicochemical Characterizations, and Biological Applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef]
- Hasany, S.; Ahmed, I.; Rajan, J.; Rehman, A. Systematic review of the preparation techniques of iron oxide magnetic nanoparticles. Nanosci. Nanotechnol. 2012, 2, 148–158. [Google Scholar] [CrossRef] [Green Version]
- van Dalum, G.; Stam, G.J.; Scholten, L.F.; Mastboom, W.J.; Vermes, I.; Tibbe, A.G.; De Groot, M.R.; Terstappen, L.W. Importance of circulating tumor cells in newly diagnosed colorectal cancer. Int. J. Oncol. 2015, 46, 1361–1368. [Google Scholar] [CrossRef] [Green Version]
- Janni, W.J.; Rack, B.; Terstappen, L.W.; Pierga, J.Y.; Taran, F.A.; Fehm, T.; Hall, C.; de Groot, M.R.; Bidard, F.C.; Friedl, T.W.; et al. Pooled Analysis of the Prognostic Relevance of Circulating Tumor Cells in Primary Breast Cancer. Clin. Cancer Res. 2016, 22, 2583–2593. [Google Scholar] [CrossRef] [Green Version]
- Tibbe, A.G.; Miller, M.C.; Terstappen, L.W. Statistical considerations for enumeration of circulating tumor cells. Cytometry 2007, 71, 154–162. [Google Scholar] [CrossRef] [Green Version]
- Rao, C.; Bui, T.; Connelly, M.; Doyle, G.; Karydis, I.; Middleton, M.R.; Clack, G.; Malone, M.; Coumans, F.A.; Terstappen, L.W. Circulating melanoma cells and survival in metastatic melanoma. Int. J. Oncol. 2011, 38, 755–760. [Google Scholar] [PubMed] [Green Version]
- Coumans, F.A.W.; Doggen, C.J.M.; Attard, G.; de Bono, J.S.; Terstappen, L.W.M.M. All circulating EpCAM+CK+CD45- objects predict overall survival in castration-resistant prostate cancer. Ann. Oncol. 2010, 21, 1851–1857. [Google Scholar] [CrossRef] [PubMed]
- Eifler, R.L.; Lind, J.; Falkenhagen, D.; Weber, V.; Fischer, M.B.; Zeillinger, R. Enrichment of circulating tumor cells from a large blood volume using leukapheresis and elutriation: Proof of concept. Cytom. B Clin. Cytom. 2011, 80, 100–111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fischer, J.C.; Niederacher, D.; Topp, S.A.; Honisch, E.; Schumacher, S.; Schmitz, N.; Zacarias Fohrding, L.; Vay, C.; Hoffmann, I.; Kasprowicz, N.S.; et al. Diagnostic leukapheresis enables reliable detection of circulating tumor cells of nonmetastatic cancer patients. Proc. Natl. Acad. Sci. USA 2013, 110, 16580–16585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coumans, F.A.W.; Siesling, S.; Terstappen, L.W.M.M. Detection of cancer before distant metastasis. BMC Cancer 2013, 13, 283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keeney, M.; Gratama, J.W.; Chin-Yee, I.H.; Sutherland, D.R. Isotype controls in the analysis of lymphocytes and CD34+ stem and progenitor cells by flow cytometry—time to let go? Cytometry 1998, 34, 280–283. [Google Scholar] [CrossRef]
- Chen, P.; Huang, Y.-Y.; Hoshino, K.; Zhang, X. Multiscale immunomagnetic enrichment of circulating tumor cells: From tubes to microchips. Lab A Chip 2014, 14, 446–458. [Google Scholar] [CrossRef]
- Hoshino, K.; Chen, P.; Huang, Y.-Y.; Zhang, X. Computational Analysis of Microfluidic Immunomagnetic Rare Cell Separation from a Particulate Blood Flow. Anal. Chem. 2012, 84, 4292–4299. [Google Scholar] [CrossRef] [Green Version]
- Lu, P.J.; Weitz, D.A. Colloidal Particles: Crystals, Glasses, and Gels. Annu. Rev. Condens. Matter Phys. 2013, 4, 217–233. [Google Scholar]
- Wang, H.; Zhao, X.; Han, X.; Tang, Z.; Song, F.; Zhang, S.; Zhu, Y.; Guo, W.; He, Z.; Guo, Q.; et al. Colloidal stability of Fe3O4 magnetic nanoparticles differentially impacted by dissolved organic matter and cations in synthetic and naturally-occurred environmental waters. Environ. Pollut. 2018, 241, 912–921. [Google Scholar] [CrossRef]
- Kang, J.H.; Um, E.; Diaz, A.; Driscoll, H.; Rodas, M.J.; Domansky, K.; Watters, A.L.; Super, M.; Stone, H.A.; Ingber, D.E. Optimization of Pathogen Capture in Flowing Fluids with Magnetic Nanoparticles. Small 2015, 11, 5657–5666. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Mendoza-Garcia, A.; Li, Q.; Sun, S. Organic Phase Syntheses of Magnetic Nanoparticles and Their Applications. Chem. Rev. 2016, 116, 10473–10512. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Kartikowati, C.W.; Horie, S.; Ogi, T.; Iwaki, T.; Okuyama, K. Correlation between particle size/domain structure and magnetic properties of highly crystalline Fe3O4 nanoparticles. Sci. Rep. 2017, 7, 9894. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.H.; Lee, N.; Kim, H.; An, K.; Park, Y.I.; Choi, Y.; Shin, K.; Lee, Y.; Kwon, S.G.; Na, H.B.; et al. Large-Scale Synthesis of Uniform and Extremely Small-Sized Iron Oxide Nanoparticles for High-Resolution T1 Magnetic Resonance Imaging Contrast Agents. J. Am. Chem. Soc. 2011, 133, 12624–12631. [Google Scholar] [CrossRef] [PubMed]
- Daou, T.J.; Grenèche, J.M.; Pourroy, G.; Buathong, S.; Derory, A.; Ulhaq-Bouillet, C.; Donnio, B.; Guillon, D.; Begin-Colin, S. Coupling Agent Effect on Magnetic Properties of Functionalized Magnetite-Based Nanoparticles. Chem. Mater. 2008, 20, 5869–5875. [Google Scholar] [CrossRef]
- McCloskey, K.E.; Chalmers, J.J.; Zborowski, M. Magnetic Cell Separation: Characterization of Magnetophoretic Mobility. Anal. Chem. 2003, 75, 6868–6874. [Google Scholar] [CrossRef]
- Allard, W.J.; Matera, J.; Miller, M.C.; Repollet, M.; Connelly, M.C.; Rao, C.; Tibbe, A.G.J.; Uhr, J.W.; Terstappen, L.W.M.M. Tumor Cells Circulate in the Peripheral Blood of All Major Carcinomas but not in Healthy Subjects or Patients With Nonmalignant Diseases. Clin. Cancer Res. 2004, 10, 6897. [Google Scholar] [CrossRef] [Green Version]
- Osman, O.; Toru, S.; Dumas-Bouchiat, F.; Dempsey, N.M.; Haddour, N.; Zanini, L.F.; Buret, F.; Reyne, G.; Frenea-Robin, M. Microfluidic immunomagnetic cell separation using integrated permanent micromagnets. Biomicrofluidics 2013, 7, 54115. [Google Scholar] [CrossRef] [Green Version]
- Sun, C.; Hassanisaber, H.; Yu, R.; Ma, S.; Verbridge, S.S.; Lu, C. Paramagnetic Structures within a Microfluidic Channel for Enhanced Immunomagnetic Isolation and Surface Patterning of Cells. Sci. Rep. 2016, 6, 29407. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.-Y.; Chen, P.; Wu, C.-H.; Hoshino, K.; Sokolov, K.; Lane, N.; Liu, H.; Huebschman, M.; Frenkel, E.; Zhang, J.X.J. Screening and Molecular Analysis of Single Circulating Tumor Cells Using Micromagnet Array. Sci. Rep. 2015, 5, 16047. [Google Scholar] [CrossRef]
- Miltenyi, S.; Müller, W.; Weichel, W.; Radbruch, A. High gradient magnetic cell separation with MACS. Cytometry 1990, 11, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Molday, R.S. Magnetic iron-Dextran Microspheres. U.S. Patent 4,452,773, 5 June 1984. [Google Scholar]
- Gijs, M.A.M. Magnetic bead handling on-chip: New opportunities for analytical applications. Microfluid. Nanofluidics 2004, 1, 22–40. [Google Scholar] [CrossRef] [Green Version]
- Hildonen, S.; Skarpen, E.; Halvorsen, T.G.; Reubsaet, L. Isolation and mass spectrometry analysis of urinary extraexosomal proteins. Sci. Rep. 2016, 6, 36331. [Google Scholar] [CrossRef] [PubMed]
- Unni, M.; Zhang, J.; George, T.J.; Segal, M.S.; Fan, Z.H.; Rinaldi, C. Engineering magnetic nanoparticles and their integration with microfluidics for cell isolation. J. Colloid Interface Sci. 2020, 564, 204–215. [Google Scholar] [CrossRef]
- Xu, H.; Aguilar, Z.P.; Yang, L.; Kuang, M.; Duan, H.; Xiong, Y.; Wei, H.; Wang, A. Antibody conjugated magnetic iron oxide nanoparticles for cancer cell separation in fresh whole blood. Biomaterials 2011, 32, 9758–9765. [Google Scholar] [CrossRef] [Green Version]
- Zhang, F.; Wu, L.; Nie, W.; Huang, L.; Zhang, J.; Li, F.; Xie, H.-Y. Biomimetic Microfluidic System for Fast and Specific Detection of Circulating Tumor Cells. Anal. Chem. 2019, 91, 15726–15731. [Google Scholar] [CrossRef]
- Kim, J.; Tran, V.T.; Oh, S.; Kim, C.-S.; Hong, J.C.; Kim, S.; Joo, Y.-S.; Mun, S.; Kim, M.-H.; Jung, J.-W.; et al. Scalable Solvothermal Synthesis of Superparamagnetic Fe3O4 Nanoclusters for Bioseparation and Theragnostic Probes. ACS Appl. Mater. Interfaces 2018, 10, 41935–41946. [Google Scholar] [CrossRef]
- Vidal-Vidal, J.; Rivas, J.; Lopez-Quintela, M.A. Synthesis of monodisperse maghemite nanoparticles by the microemulsion method. Colloids Surf. A Physicochem. Eng. Asp. 2006, 288, 44–51. [Google Scholar] [CrossRef]
- Massart, R. Preparation of aqueous magnetic liquids in alkaline and acidic media. IEEE Trans. Magn. 1981, 17, 1247–1248. [Google Scholar] [CrossRef]
- Molday, R.S.; Mackenzie, D. Immunospecific ferromagnetic iron-dextran reagents for the labeling and magnetic separation of cells. J. Immunol. Methods 1982, 52, 353–367. [Google Scholar] [CrossRef]
- Ugelstad, J.; Hansen, F.K. Kinetics and Mechanism of Emulsion Polymerization. Rubber Chem. Technol. 1976, 49, 536–609. [Google Scholar] [CrossRef]
- Charles, S.; Owen, J.S.S.; D’Angelo, L.; Liberti, P.A. Magnetic Polymer Particles. U.S. Patent 4,795,698, 3 January 1989. [Google Scholar]
- Feng, Y.; White, A.; Hein, J.; Appel, E.; Fordyce, P. MRBLES 2.0: High-throughput generation of chemically functionalized spectrally and magnetically-encoded hydrogel beads using a simple single-layer microfluidic device. bioRxiv 2020. [Google Scholar] [CrossRef]
- Park, J.; An, K.; Hwang, Y.; Park, J.-G.; Noh, H.-J.; Kim, J.-Y.; Park, J.-H.; Hwang, N.-M.; Hyeon, T. Ultra-large-scale syntheses of monodisperse nanocrystals. Nat. Mater. 2004, 3, 891–895. [Google Scholar] [CrossRef]
- Yu, W.W.; Falkner, J.C.; Yavuz, C.T.; Colvin, V.L. Synthesis of monodisperse iron oxide nanocrystals by thermal decomposition of iron carboxylate salts. Chem. Commun. 2004, 20, 2306–2307. [Google Scholar] [CrossRef] [PubMed]
- Ge, Q.; Su, J.; Chung, T.-S.; Amy, G. Hydrophilic Superparamagnetic Nanoparticles: Synthesis, Characterization, and Performance in Forward Osmosis Processes. Ind. Eng. Chem. Res. 2011, 50, 382–388. [Google Scholar] [CrossRef]
- Nielsen, M.A.; Resende, M.; de Jongh, W.A.; Ditlev, S.B.; Mordmüller, B.; Houard, S.; Ndam, N.T.; Agerbæk, M.Ø.; Hamborg, M.; Massougbodji, A.; et al. The Influence of Sub-Unit Composition and Expression System on the Functional Antibody Response in the Development of a VAR2CSA Based Plasmodium falciparum Placental Malaria Vaccine. PLoS ONE 2015, 10, e0135406. [Google Scholar] [CrossRef] [Green Version]
- Hassan, E.M.; Willmore, W.G.; DeRosa, M.C. Aptamers: Promising Tools for the Detection of Circulating Tumor Cells. Nucleic Acid Ther. 2016, 26, 335–347. [Google Scholar] [CrossRef]
- Rao, C.G.; Chianese, D.; Doyle, G.V.; Miller, M.C.; Russell, T.; Sanders, R.A.; Terstappen, L.W. Expression of epithelial cell adhesion molecule in carcinoma cells present in blood and primary and metastatic tumors. Int. J. Oncol. 2005, 27, 49–57. [Google Scholar] [CrossRef]
- Went, P.T.; Lugli A Fau-Meier, S.; Meier S Fau-Bundi, M.; Bundi M Fau-Mirlacher, M.; Mirlacher M Fau-Sauter, G.; Sauter G Fau-Dirnhofer, S.; Dirnhofer, S. Frequent EpCam protein expression in human carcinomas. Hum. Pathol. 2004, 35, 122–128. [Google Scholar] [CrossRef]
- Cristofanilli, M.; Hayes, D.F.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Reuben, J.M.; Doyle, G.V.; Matera, J.; Allard, W.J.; Miller, M.C.; et al. Circulating tumor cells: A novel prognostic factor for newly diagnosed metastatic breast cancer. J. Clin. Oncol. 2005, 23, 1420–1430. [Google Scholar] [CrossRef]
- de Bono, J.S.; Scher, H.I.; Montgomery, R.B.; Parker, C.; Miller, M.C.; Tissing, H.; Doyle, G.V.; Terstappen, L.W.; Pienta, K.J.; Raghavan, D. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin. Cancer Res. 2008, 14, 6302–6309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayes, D.F.; Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Miller, M.C.; Matera, J.; Allard, W.J.; Doyle, G.V.; Terstappen, L.W. Circulating tumor cells at each follow-up time point during therapy of metastatic breast cancer patients predict progression-free and overall survival. Clin. Cancer Res. 2006, 12, 4218–4224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alix-Panabières, C.; Pantel, K. Challenges in circulating tumour cell research. Nat. Rev. Cancer 2014, 14, 623. [Google Scholar] [CrossRef] [PubMed]
- Galletti, G.; Sung, M.S.; Vahdat, L.T.; Shah, M.A.; Santana, S.M.; Altavilla, G.; Kirby, B.J.; Giannakakou, P. Isolation of breast cancer and gastric cancer circulating tumor cells by use of an anti HER2-based microfluidic device. Lab Chip 2014, 14, 147–156. [Google Scholar] [CrossRef]
- Lee, H.J.; Cho, H.Y.; Oh, J.H.; Namkoong, K.; Lee, J.G.; Park, J.M.; Lee, S.S.; Huh, N.; Choi, J.W. Simultaneous capture and in situ analysis of circulating tumor cells using multiple hybrid nanoparticles. Biosens. Bioelectron. 2013, 47, 508–514. [Google Scholar] [CrossRef]
- Que, Z.; Luo, B.; Zhou, Z.; Dong, C.; Jiang, Y.; Wang, L.; Shi, Q.; Tian, J. Establishment and characterization of a patient-derived circulating lung tumor cell line in vitro and in vivo. Cancer Cell Int. 2019, 19, 21. [Google Scholar] [CrossRef] [Green Version]
- Thege, F.I.; Gruber, C.N.; Cardle, I.I.; Cong, S.H.; Lannin, T.B.; Kirby, B.J. anti-EGFR capture mitigates EMT- and chemoresistance-associated heterogeneity in a resistance-profiling CTC platform. Anal. Biochem. 2019, 577, 26–33. [Google Scholar] [CrossRef]
- Ohnaga, T.; Takei, Y.; Nagata, T.; Shimada, Y. Highly efficient capture of cancer cells expressing EGFR by microfluidic methods based on antigen-antibody association. Sci. Rep. 2018, 8, 12005. [Google Scholar] [CrossRef]
- Onstenk, W.; Kraan, J.; Mostert, B.; Timmermans, M.M.; Charehbili, A.; Smit, V.T.; Kroep, J.R.; Nortier, J.W.; van de Ven, S.; Heijns, J.B.; et al. Improved Circulating Tumor Cell Detection by a Combined EpCAM and MCAM CellSearch Enrichment Approach in Patients with Breast Cancer Undergoing Neoadjuvant Chemotherapy. Mol. Cancer 2015, 14, 821–827. [Google Scholar] [CrossRef] [Green Version]
- Bande, M.F.; Santiago, M.; Muinelo-Romay, L.; Blanco, M.J.; Mera, P.; Capeans, C.; Pardo, M.; Piñeiro, A. Detection of circulating melanoma cells in choroidal melanocytic lesions. BMC Res. Notes 2015, 8, 452. [Google Scholar] [CrossRef] [Green Version]
- Li, N.; Zong, S.; Zhang, Y.; Wang, Z.; Wang, Y.; Zhu, K.; Yang, K.; Wang, Z.; Chen, B.; Cui, Y. A SERS-colorimetric dual-mode aptasensor for the detection of cancer biomarker MUC1. Anal. Bioanal. Chem. 2020, 412, 5707–5718. [Google Scholar] [CrossRef] [PubMed]
- Thege, F.I.; Lannin, T.B.; Saha, T.N.; Tsai, S.; Kochman, M.L.; Hollingsworth, M.A.; Rhim, A.D.; Kirby, B.J. Microfluidic immunocapture of circulating pancreatic cells using parallel EpCAM and MUC1 capture: Characterization, optimization and downstream analysis. Lab Chip 2014, 14, 1775–1784. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Liu, S.; Li, C.; Wang, Y.; Yan, C. Dual-target recognition sandwich assay based on core-shell magnetic mesoporous silica nanoparticles for sensitive detection of breast cancer cells. Talanta 2018, 182, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Chen, B.; He, M.; Hu, B. A Multifunctional Platform for the Capture, Release, And Enumeration of Circulating Tumor Cells Based on Aptamer Binding, Nicking Endonuclease-Assisted Amplification, And Inductively Coupled Plasma Mass Spectrometry Detection. Anal. Chem. 2020, 92, 10308–10315. [Google Scholar] [CrossRef]
- Salanti, A.; Clausen, T.M.; Agerbæk, M.Ø.; AlNakouzi, N.; Dahlbäck, M.; Oo, H.Z.; Lee, S.; Gustavsson, T.; Rich, J.R.; Hedberg, B.J.; et al. Targeting Human Cancer by a Glycosaminoglycan Binding Malaria Protein. Cancer Cell 2015, 28, 500–514. [Google Scholar] [CrossRef] [Green Version]
- Agerbæk, M.Ø.; Bang-Christensen, S.; Salanti, A. Fighting Cancer Using an Oncofetal Glycosaminoglycan-Binding Protein from Malaria Parasites. Trends Parasitol. 2019, 35, 178–181. [Google Scholar] [CrossRef]
- Agerbæk, M.Ø.; Bang-Christensen, S.R.; Yang, M.-H.; Clausen, T.M.; Pereira, M.A.; Sharma, S.; Ditlev, S.B.; Nielsen, M.A.; Choudhary, S.; Gustavsson, T.; et al. The VAR2CSA malaria protein efficiently retrieves circulating tumor cells in an EpCAM-independent manner. Nat. Commun. 2018, 9, 3279. [Google Scholar] [CrossRef] [Green Version]
- Bang-Christensen, S.R.; Pedersen, R.S.; Pereira, M.A.; Clausen, T.M.; Løppke, C.; Sand, N.T.; Ahrens, T.D.; Jørgensen, A.M.; Lim, Y.C.; Goksøyr, L. Capture and Detection of Circulating Glioma Cells Using the Recombinant VAR2CSA Malaria Protein. Cells 2019, 8, 998. [Google Scholar] [CrossRef] [Green Version]
- Sand, N.T.; Petersen, T.B.; Bang-Christensen, S.R.; Ahrens, T.D.; Loppke, C.; Jorgensen, A.M.; Gustavsson, T.; Choudhary, S.; Theander, T.G.; Salanti, A.; et al. Optimization of rVAR2-Based Isolation of Cancer Cells in Blood for Building a Robust Assay for Clinical Detection of Circulating Tumor Cells. Int. J. Mol. Sci. 2020, 21, 2401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andree, K.C.; Mentink, A.; Nguyen, A.T.; Goldsteen, P.; van Dalum, G.; Broekmaat, J.J.; van Rijn, C.J.M.; Terstappen, L.W.M.M. Tumor cell capture from blood by flowing across antibody-coated surfaces. Lab A Chip 2019, 19, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Martin, V.M.; Siewert C Fau-Scharl, A.; Scharl A Fau-Harms, T.; Harms T Fau-Heinze, R.; Heinze R Fau-Ohl, S.; Ohl S Fau-Radbruch, A.; Radbruch A Fau-Miltenyi, S.; Miltenyi S Fau-Schmitz, J.; Schmitz, J. Immunomagnetic enrichment of disseminated epithelial tumor cells from peripheral blood by MACS. Exp. Hematol. 1998, 26, 252–264. [Google Scholar] [PubMed]
- Schneck, H.; Gierke, B.; Uppenkamp, F.; Behrens, B.; Niederacher, D.; Stoecklein, N.H.; Templin, M.F.; Pawlak, M.; Fehm, T.; Neubauer, H.; et al. EpCAM-Independent Enrichment of Circulating Tumor Cells in Metastatic Breast Cancer. PLoS ONE 2015, 10, e0144535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neves, M.; Azevedo, R.; Lima, L.; Oliveira, M.I.; Peixoto, A.; Ferreira, D.; Soares, J.; Fernandes, E.; Gaiteiro, C.; Palmeira, C.; et al. Exploring sialyl-Tn expression in microfluidic-isolated circulating tumour cells: A novel biomarker and an analytical tool for precision oncology applications. New Biotechnol. 2019, 49, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, Z.; Chen, C.; Ding, P.; Sun, N.; Pei, R. Dual-antibody Modified PLGA Nanofibers for Specific Capture of Epithelial and Mesenchymal CTCs. Colloids Surf. 2019, 181, 143–148. [Google Scholar] [CrossRef]
- Wang, Z.; Sun, N.; Liu, H.; Chen, C.; Ding, P.; Yue, X.; Zou, H.; Xing, C.; Pei, R. High-Efficiency Isolation and Rapid Identification of Heterogeneous Circulating Tumor Cells (CTCs) Using Dual-Antibody-Modified Fluorescent-Magnetic Nanoparticles. ACS Appl. Mater. Interfaces 2019, 11, 39586–39593. [Google Scholar] [CrossRef]
- Po, J.W.; Roohullah, A.; Lynch, D.; DeFazio, A.; Harrison, M.; Harnett, P.R.; Kennedy, C.; de Souza, P.; Becker, T.M. Improved ovarian cancer EMT-CTC isolation by immunomagnetic targeting of epithelial EpCAM and mesenchymal N-cadherin. J. Circ. Biomark 2018, 7, 1849454418782617. [Google Scholar] [CrossRef] [Green Version]
- Esmaeilsabzali, H.; Payer, R.T.M.; Guo, Y.; Cox, M.E.; Parameswaran, A.M.; Beischlag, T.V.; Park, E.J. Development of a microfluidic platform for size-based hydrodynamic enrichment and PSMA-targeted immunomagnetic isolation of circulating tumour cells in prostate cancer. Biomicrofluidics 2019, 13, 014110. [Google Scholar] [CrossRef]
- Yin, C.; Wang, Y.; Ji, J.; Cai, B.; Chen, H.; Yang, Z.; Wang, K.; Luo, C.; Zhang, W.; Yuan, C.; et al. Molecular Profiling of Pooled Circulating Tumor Cells from Prostate Cancer Patients Using a Dual-Antibody-Functionalized Microfluidic Device. Anal. Chem. 2018, 90, 3744–3751. [Google Scholar] [CrossRef]
- KOLOSTOVA, K.; PINTEROVA, D.; HOFFMAN, R.M.; BOBEK, V. Circulating Human Prostate Cancer Cells from an Orthotopic Mouse Model Rapidly Captured by Immunomagnetic Beads and Imaged by GFP Expression. Anticancer Res. 2011, 31, 1535–1539. [Google Scholar]
- Hughes, A.D.; Mattison, J.; Powderly, J.D.; Greene, B.T.; King, M.R. Rapid isolation of viable circulating tumor cells from patient blood samples. J. Vis. Exp. 2012, 64, e4248. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, M.J.; Castellanos, C.A.; King, M.R. Immobilized surfactant-nanotube complexes support selectin-mediated capture of viable circulating tumor cells in the absence of capture antibodies. J. Biomed. Mater. Res. A 2015, 103, 3407–3418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Launiere, C.; Gaskill, M.; Czaplewski, G.; Myung, J.H.; Hong, S.; Eddington, D.T. Channel surface patterning of alternating biomimetic protein combinations for enhanced microfluidic tumor cell isolation. Anal. Chem. 2012, 84, 4022–4028. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Myung, J.H.; Launiere, C.A.; Eddington, D.T.; Hong, S. Enhanced tumor cell isolation by a biomimetic combination of E-selectin and anti-EpCAM: Implications for the effective separation of circulating tumor cells (CTCs). Langmuir 2010, 26, 8589–8596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beck, T.N.; Boumber, Y.A.; Aggarwal, C.; Pei, J.; Thrash-Bingham, C.; Fittipaldi, P.; Vlasenkova, R.; Rao, C.; Borghaei, H.; Cristofanilli, M.; et al. Circulating tumor cell and cell-free RNA capture and expression analysis identify platelet-associated genes in metastatic lung cancer. BMC Cancer 2019, 19, 603. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Raba, K.; Guglielmi, R.; Behrens, B.; Van Dalum, G.; Flügen, G.; Koch, A.; Patel, S.; Knoefel, W.T.; Stoecklein, N.H.; et al. Magnetic-Based Enrichment of Rare Cells from High Concentrated Blood Samples. Cancers 2020, 12, 933. [Google Scholar] [CrossRef] [Green Version]
- Jain, A.; Cheng, K. The principles and applications of avidin-based nanoparticles in drug delivery and diagnosis. J. Control. Release 2017, 245, 27–40. [Google Scholar] [CrossRef] [Green Version]
- Diamandis, E.P.; Christopoulos, T.K. The biotin-(strept)avidin system: Principles and applications in biotechnology. Clin. Chem. 1991, 37, 625–636. [Google Scholar] [CrossRef]
- Weber, P.C.; Ohlendorf, D.H.; Wendoloski, J.J.; Salemme, F.R. Structural origins of high-affinity biotin binding to streptavidin. Science 1989, 243, 85. [Google Scholar] [CrossRef]
- Kuzuya, A.; Numajiri, K.; Kimura, M.; Komiyama, M. Single-Molecule Accommodation of Streptavidin in Nanometer-Scale Wells Formed in DNA Nanostructures. Nucleic Acids Symp. Ser. 2008, 52, 681–682. [Google Scholar] [CrossRef] [Green Version]
- Avvakumova, S.; Colombo, M.; Galbiati, E.; Mazzucchelli, S.; Rotem, R.; Prosperi, D. Chapter 6-Bioengineered Approaches for Site Orientation of Peptide-Based Ligands of Nanomaterials. In Biomedical Applications of Functionalized Nanomaterials; Sarmento, B., das Neves, J., Eds.; Elsevier: Amsterdan, The Netherlands, 2018; pp. 139–169. [Google Scholar] [CrossRef]
- Gao, X.; Cui, Y.; Levenson, R.M.; Chung, L.W.K.; Nie, S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotechnol. 2004, 22, 969–976. [Google Scholar] [CrossRef]
- Li, S.; Liu, H.; He, N. Covalent Binding of Streptavidin on Gold Magnetic Nanoparticles for Bead Array Fabrication. J. Nanosci. Nanotechnol. 2010, 10, 4875–4882. [Google Scholar] [CrossRef] [PubMed]
- Andree, K.C.; Mentink, A.; Zeune, L.L.; Terstappen, L.W.M.M.; Stoecklein, N.H.; Neves, R.P.; Driemel, C.; Lampignano, R.; Yang, L.; Neubauer, H.; et al. Toward a real liquid biopsy in metastatic breast and prostate cancer: Diagnostic LeukApheresis increases CTC yields in a European prospective multicenter study (CTCTrap). Int. J. Cancer 2018, 143, 2584–2591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andree, K.C.; van Dalum, G.; Terstappen, L.W.M.M. Challenges in circulating tumor cell detection by the CellSearch system. Mol. Oncol. 2016, 10, 395–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stoecklein, N.H.; Fischer, J.C.; Niederacher, D.; Terstappen, L.W. Challenges for CTC-based liquid biopsies: Low CTC frequency and diagnostic leukapheresis as a potential solution. Expert Rev. Mol. Diagn. 2016, 16, 147–164. [Google Scholar] [CrossRef]
- Correa, I.; Plunkett, T.; Vlad, A.; Mungul, A.; Candelora-Kettel, J.; Burchell, J.M.; Taylor–papadimitriou, J.; Finn, O.J. Form and pattern of MUC1 expression on T cells activated in vivo or in vitro suggests a function in T-cell migration. Immunology 2003, 108, 32–41. [Google Scholar] [CrossRef]
- Yu, D.; Tang, L.; Dong, Z.; Loftis, K.A.; Ding, Z.; Cheng, J.; Qin, B.; Yan, J.; Li, W. Effective reduction of non-specific binding of blood cells in a microfluidic chip for isolation of rare cancer cells. Biomater. Sci. 2018, 6, 2871–2880. [Google Scholar] [CrossRef]
- Sanchez, L.M.; Martin, D.A.; Alvarez, V.A.; Gonzalez, J.S. Polyacrylic acid-coated iron oxide magnetic nanoparticles: The polymer molecular weight influence. Colloids Surf. A 2018, 543, 28–37. [Google Scholar] [CrossRef] [Green Version]
- Rose, P.A.; Praseetha, P.K.; Bhagat, M.; Alexander, P.; Abdeen, S.; Chavali, M. Drug embedded PVP coated magnetic nanoparticles for targeted killing of breast cancer cells. Technol. Cancer Res. Treat. 2013, 12, 463–472. [Google Scholar] [CrossRef]
- Ma, Y.R.; Zhang, X.L.; Zeng, T.; Cao, D.; Zhou, Z.; Li, W.H.; Niu, H.; Cai, Y.Q. Polydopamine-coated magnetic nanoparticles for enrichment and direct detection of small molecule pollutants coupled with MALDI-TOF-MS. ACS Appl. Mater. Interfaces 2013, 5, 1024–1030. [Google Scholar] [CrossRef]
- Hong, R.Y.; Feng, B.; Chen, L.L.; Liu, G.H.; Li, H.Z.; Zheng, Y.; Wei, D.G. Synthesis, characterization and MRI application of dextran-coated Fe3O4 magnetic nanoparticles. Biochem. Eng. J. 2008, 42, 290–300. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, X.; Liu, F.; Cheng, Y.; Shi, D. Effective Reduction of Nonspecific Binding by Surface Engineering of Quantum Dots with Bovine Serum Albumin for Cell-Targeted Imaging. Langmuir 2012, 28, 16605–16613. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang, M.; Wang, L.; Gu, Y. Effective reduction of non-specific binding by bovine serum albumin modified quantum dot surface for cell targeted imaging. In Twelfth International Conference on Photonics and Imaging in Biology and Medicine (PIBM 2014); SPIE Press: Bellingham, WA, USA, 2014. [Google Scholar]
- Zhu, N.; Ji, H.; Yu, P.; Niu, J.; Farooq, M.U.; Akram, M.W.; Udego, I.O.; Li, H.; Niu, X. Surface Modification of Magnetic Iron Oxide Nanoparticles. Nanomaterials 2018, 8, 810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, J.H.; Krause, S.; Tobin, H.; Mammoto, A.; Kanapathipillai, M.; Ingber, D.E. A combined micromagnetic-microfluidic device for rapid capture and culture of rare circulating tumor cells. Lab A Chip 2012, 12, 2175–2181. [Google Scholar] [CrossRef] [PubMed]
- Cann, G.M.; Gulzar, Z.G.; Cooper, S.; Li, R.; Luo, S.; Tat, M.; Stuart, S.; Schroth, G.; Srinivas, S.; Ronaghi, M.; et al. mRNA-Seq of single prostate cancer circulating tumor cells reveals recapitulation of gene expression and pathways found in prostate cancer. PLoS ONE 2012, 7, e49144. [Google Scholar] [CrossRef]
- Hoshino, K.; Huang, Y.-Y.; Lane, N.; Huebschman, M.; Uhr, J.W.; Frenkel, E.P.; Zhang, X. Microchip-based immunomagnetic detection of circulating tumor cells. Lab A Chip 2011, 11, 3449–3457. [Google Scholar] [CrossRef]
- Chen, P.; Huang, Y.-Y.; Hoshino, K.; Zhang, J.X.J. Microscale Magnetic Field Modulation for Enhanced Capture and Distribution of Rare Circulating Tumor Cells. Sci. Rep. 2015, 5, 8745. [Google Scholar] [CrossRef] [Green Version]
- Yu, X.; Feng, X.; Hu, J.; Zhang, Z.-L.; Pang, D.-W. Controlling the Magnetic Field Distribution on the Micrometer Scale and Generation of Magnetic Bead Patterns for Microfluidic Applications. Langmuir 2011, 27, 5147–5156. [Google Scholar] [CrossRef]
- Stroock, A.D.; Dertinger, S.K.W.; Ajdari, A.; Mezić, I.; Stone, H.A.; Whitesides, G.M. Chaotic Mixer for Microchannels. Science 2002, 295, 647. [Google Scholar] [CrossRef] [Green Version]
- Stott, S.L.; Hsu, C.-H.; Tsukrov, D.I.; Yu, M.; Miyamoto, D.T.; Waltman, B.A.; Rothenberg, S.M.; Shah, A.M.; Smas, M.E.; Korir, G.K.; et al. Isolation of circulating tumor cells using a microvortex-generating herringbone-chip. Proc. Natl. Acad. Sci. USA 2010, 107, 18392. [Google Scholar] [CrossRef] [Green Version]
- Shi, W.; Wang, S.; Maarouf, A.; Uhl, C.G.; He, R.; Yunus, D.; Liu, Y. Magnetic particles assisted capture and release of rare circulating tumor cells using wavy-herringbone structured microfluidic devices. Lab A Chip 2017, 17, 3291–3299. [Google Scholar] [CrossRef]
- Zhang, L.; Xu, Z.; Kang, Y.; Xue, P. Three-dimensional microfluidic chip with twin-layer herringbone structure for high efficient tumor cell capture and release via antibody-conjugated magnetic microbeads. Electrophoresis 2018, 39, 1452–1459. [Google Scholar] [CrossRef] [PubMed]
- Kwak, B.; Lee, J.; Lee, D.; Lee, K.; Kwon, O.; Kang, S.; Kim, Y. Selective isolation of magnetic nanoparticle-mediated heterogeneity subpopulation of circulating tumor cells using magnetic gradient based microfluidic system. Biosens. Bioelectron. 2017, 88, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-L.; Huang, W.; Jalal, S.I.; Chan, B.-D.; Mahmood, A.; Shahda, S.; O’Neil, B.H.; Matei, D.E.; Savran, C.A. Circulating tumor cell detection using a parallel flow micro-aperture chip system. Lab A Chip 2015, 15, 1677–1688. [Google Scholar] [CrossRef] [PubMed]
- Earhart, C.M.; Hughes, C.E.; Gaster, R.S.; Ooi, C.C.; Wilson, R.J.; Zhou, L.Y.; Humke, E.W.; Xu, L.; Wong, D.J.; Willingham, S.B.; et al. Isolation and mutational analysis of circulating tumor cells from lung cancer patients with magnetic sifters and biochips. Lab A Chip 2014, 14, 78–88. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Han, S.-I.; Park, M.-J.; Jeon, C.-W.; Joo, Y.-D.; Choi, I.-H.; Han, K.-H. Circulating Tumor Cell Microseparator Based on Lateral Magnetophoresis and Immunomagnetic Nanobeads. Anal. Chem. 2013, 85, 2779–2786. [Google Scholar] [CrossRef]
- Cho, H.; Kim, J.; Han, S.-I.; Han, K.-H. Analytical evaluation for somatic mutation detection in circulating tumor cells isolated using a lateral magnetophoretic microseparator. Biomed. Microdevices 2016, 18, 91. [Google Scholar] [CrossRef]
- Park, J.W.; Lee, N.-R.; Cho, S.M.; Jung, M.Y.; Ihm, C.; Lee, D.-S. Microdevice for Separation of Circulating Tumor Cells Using Embedded Magnetophoresis with V-shaped Ni-Co Nanowires and Immuno-nanomagnetic Beads. ETRI J. 2015, 37, 233–240. [Google Scholar] [CrossRef]
- Ozkumur, E.; Shah, A.M.; Ciciliano, J.C.; Emmink, B.L.; Miyamoto, D.T.; Brachtel, E.; Yu, M.; Chen, P.I.; Morgan, B.; Trautwein, J.; et al. Inertial focusing for tumor antigen-dependent and -independent sorting of rare circulating tumor cells. Sci. Transl. Med. 2013, 5, 179ra147. [Google Scholar] [CrossRef] [Green Version]
- Mishra, A.; Dubash, T.D.; Edd, J.F.; Jewett, M.K.; Garre, S.G.; Karabacak, N.A.-O.; Rabe, D.A.-O.; Mutlu, B.A.-O.; Walsh, J.A.-O.; Kapur, R.; et al. Ultrahigh-throughput magnetic sorting of large blood volumes for epitope-agnostic isolation of circulating tumor cells. Proc. Natl. Acad. Sci. USA 2020, 117, 16839–16847. [Google Scholar] [CrossRef]
- Powell, A.A.; Talasaz, A.H.; Zhang, H.; Coram, M.A.; Reddy, A.; Deng, G.; Telli, M.L.; Advani, R.H.; Carlson, R.W.; Mollick, J.A.; et al. Single Cell Profiling of Circulating Tumor Cells: Transcriptional Heterogeneity and Diversity from Breast Cancer Cell Lines. PLoS ONE 2012, 7, e33788. [Google Scholar] [CrossRef] [Green Version]
- Wu, S.; Gu, L.; Qin, J.; Zhang, L.; Sun, F.; Liu, Z.; Wang, Y.; Shi, D. Rapid Label-Free Isolation of Circulating Tumor Cells from Patients’ Peripheral Blood Using Electrically Charged Fe3O4 Nanoparticles. ACS Appl. Mater. Interfaces 2020, 12, 4193–4203. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Xia, H.-F.; Xu, C.-M.; Feng, J.; Ren, J.-G.; Miao, F.; Wu, M.; Wu, L.-L.; Pang, D.-W.; Chen, G.; et al. Magnetic chip based extracorporeal circulation: A new tool for circulating tumor cell in vivo detection. Anal. Chem. 2019, 91, 15260–15266. [Google Scholar] [CrossRef] [PubMed]
- Vermesh, O.; Aalipour, A.; Ge, T.J.; Saenz, Y.; Guo, Y.; Alam, I.S.; Park, S.-m.; Adelson, C.N.; Mitsutake, Y.; Vilches-Moure, J.; et al. An intravascular magnetic wire for the high-throughput retrieval of circulating tumour cells in vivo. Nat. Biomed. Eng. 2018, 2, 696–705. [Google Scholar] [CrossRef] [PubMed]
- Valihrach, L.; Androvic, P.; Kubista, M. Platforms for Single-Cell Collection and Analysis. Int. J. Mol. Sci. 2018, 19, 807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, Y.; Xu, X.; Wang, W.; Tian, T.; Zhu, Z.; Yang, C. Single cell transcriptomics: Moving towards multi-omics. Analyst 2019, 144, 3172–3189. [Google Scholar] [CrossRef]
- Baslan, T.; Hicks, J. Unravelling biology and shifting paradigms in cancer with single-cell sequencing. Nat. Rev. Cancer 2017, 17, 557–569. [Google Scholar] [CrossRef]
- Paolillo, C.; Londin, E.; Fortina, P. Single-Cell Genomics. Clin. Chem. 2019, 65, 972–985. [Google Scholar] [CrossRef]





| Method | Size | Morphology | Magnetization (Emu/g) | Ligand | Surface Groups Used | Supplier | Photos | Reference |
|---|---|---|---|---|---|---|---|---|
| Co-precipitation | 30–1000 nm | Roughly spherical | - | Dextran | −OH | Miltenyi biotec (MACS) |  (A) (A) | [34,35] |
| 100 nm | Irregular shape | - | BSA | −NH2−COOH | Veridex Ferrofluid™ |  (B) (B) | [29] | |
| 1.0/2.8/4.5 μm | Spherical | 10.8–23.5 | Polymer | −NH2 | InvitrogenTM Dynal |  (C) (C) | [36] | |
| Thermal decomposition | 20.3 ± 1.5 nm | Spherical | 85.8 | PEG | −COOH | N/A |  (D) (D) | [37] |
| 30 nm | Spherical | - | ABC triblock copolymer | −COOH | N/A |  (E) (E) | [38] | |
| Solvothermal | 80 nm | Spherical nanocluster | 80 | leukocyte membrane | Hydro-phobic inter-action | N/A |  (F) (F) | [39] |
| 100–300 nm | Spherical nanocluster | 58.48 | Silica | −OH | AMO LIFESCIENCE Inc., Korea |  (G) (G) | [40] | |
| Microemulsion | 10 nm | Spherical | 33.2 | Oleyla-mine | −NH2 | N/A |  (H) (H) | [41] |
| CD #/Abbr. | Name | Expression | Location | Used for CTC Capture |
|---|---|---|---|---|
| Epithelial cells and no or few hematopoietic cells | ||||
| CD44 | ep | cs, ic | Yes [76] | |
| CD49f | Integrin Alpha 6 | ep | cs | Yes [76] |
| CD66e | CEA (CAM5) | ep (colon) | cs | No |
| CD66f | PSG1 | ep, pl, li | cs | No |
| CD104 | Integrin beta4 | ep, ed, ne, ke | cs | No |
| CD118 | LIFR | ep | cs | No |
| CD133 | Prominin-1 | ep, hs, ed, ne | cs | No |
| CD164 (1) | MUC-24 | ep (prostate cancer, bone mets) | cs | No |
| CD164 (2) | ep (bowl, lung, thyroid) | cs | No | |
| CD164 (3) | hs, sc (bone marrow) | cs | No | |
| CD175 | Tn | ep, sc | cs | No |
| CD175s | Sialyl Tn | ep, ed, erythroblasts, | cs | Yes [77] |
| CD176 | Thomsen–Friedenreich antigen | ep, sc | cs | No |
| CD227 | MUC-1 | ep, sc | cs | Yes [65,66,67,68] |
| CD283 | TLR3. | ep, fi, ne, dc | cs, ic | No |
| CDw293 | BMPR-1B | ep, ms, bc | cs | No |
| CD318 | CDCP1 | ep, hs | cs | No |
| CD325 | N-Cadherin | ep, ne, mu (cardiac), fi, pa, li | cs, ic | Yes [78,79,80] |
| CD326 | EpCAM | ep, sc (embryonic) | cs | Yes [29,54,55] |
| CD331 | FGR1 | ep, ed, fi, ms, mu (cardio), ne | cs | No |
| CD331-4 | FGFR1-4 | ne, ki, te | No | |
| CD332 | FGFR2 | ep, li, fi, ne, | cs, ic | No |
| CD340 | erbB-2 kinase/ Her-2 | ep (breast), ed, ke, hs, sc | cs, ic | Yes [58,59] |
| CD344 | Frizzled-4 | ep, ms, ne, ki, te | cs | No |
| CD338 | ABCG2 | ep, li, ki, lu, pl | cs | No |
| CD350 | Frizzled-10 | ep (colon), ki, lu, br, pl | cs | No |
| CK | Cytokeratins 1-23 | ep | ic | Yes [75] |
| AR | Androgen receptor | ep | ic, in | No |
| EGFR | Epithelial Growth Factor/Her-1 | ep, pl, pa, ki, li | cs | Yes [59,60,61,62] |
| ER | Estrogen receptor | ep | ic, in | No |
| PSMA | Prostate-Specific Membrane Antigen | ep (prostate) | cs, ic | Yes [81,82,83] |
| Trop2 | Tumor-associated Ca signal transducer 2 | ep | cs | Yes [76] |
| Endothelial cells and no or few hematopoietic cells | ||||
| CD62E | E-selectin | ed (activated) | cs | Yes [84,85,86,87] |
| CD144 | VE Cadherin | ed, sc | cs | No |
| CD146 | MCAM | ed, sm, me, dc, T-cells (activated) | cs | Yes [13,63] |
| CD201 | EPCR | ed | cs | No |
| CD202b | TIE2 | ed, angioblasts | cs | No |
| CD228 | Melanotransferrin | ed, sc, me | cs | No |
| CD248 | Endosialin | ed (tumor), fi | cs | No |
| CD266 | TWEAK R | ed | cs | No |
| CD299 | C-type lectin domain family 4 member M | ed, li, lymph node | cs | No |
| CD300g | CLM-9 | ed | cs, ic | No |
| CD309 | VEGFR-2 | ed, sc | cs | No |
| CD322 | JAM-B | ed (high) | cs | No |
| CD362 | Syndecan-2, | ed, sm (vascular), fi, me | cs | No |
| Neural cells and no or few hematopoietic cells | ||||
| CD246 | ALK | ne (embryonic CNS & PNS | cs, ic, in | No |
| CD271 | NGF receptor | ne, ms (bone marrow) | cs | No |
| Mesenchymal cells & no or few hematopoietic cells | ||||
| CD90 | Thy-1 | fi | cs | No |
| CD167b | DDR2 | bc, he, lu | cs, ic | No |
| CD292 | BMPR-1A | mu (skeletal), bc, ms | cs | No |
| CD331-4 | FGFR1-4 | ne, ki, te | cs | No |
| CD339 | Jagged1, | st (bone marrow), ep (thymic), ed, ne, ke, ov, pr, pa, pl, he | cs | No |
| CD333 | FGFR | ne, ki, te | cs | No |
| c-Met/HGF | Hepatocyte growth factor | pl | cs | No |
| Vimentin | Vimentin | ms | ic | No |
| FAP | Fibroblast activation protein, Seprase | ep, ms, fi | cs | No |
| No or few hematopoietic cells | ||||
| VAR2 | Malaria-encoded VAR2CSA | pl | cs | [69,70,71,72,73] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, P.; Jonkheijm, P.; Terstappen, L.W.M.M.; Stevens, M. Magnetic Particles for CTC Enrichment. Cancers 2020, 12, 3525. https://doi.org/10.3390/cancers12123525
Liu P, Jonkheijm P, Terstappen LWMM, Stevens M. Magnetic Particles for CTC Enrichment. Cancers. 2020; 12(12):3525. https://doi.org/10.3390/cancers12123525
Chicago/Turabian StyleLiu, Peng, Pascal Jonkheijm, Leon W. M. M. Terstappen, and Michiel Stevens. 2020. "Magnetic Particles for CTC Enrichment" Cancers 12, no. 12: 3525. https://doi.org/10.3390/cancers12123525
APA StyleLiu, P., Jonkheijm, P., Terstappen, L. W. M. M., & Stevens, M. (2020). Magnetic Particles for CTC Enrichment. Cancers, 12(12), 3525. https://doi.org/10.3390/cancers12123525






