Breast Cancer Mortality among Women with a BRCA1 or BRCA2 Mutation in a Magnetic Resonance Imaging Plus Mammography Screening Program
Abstract
Simple Summary
Abstract
1. Introduction
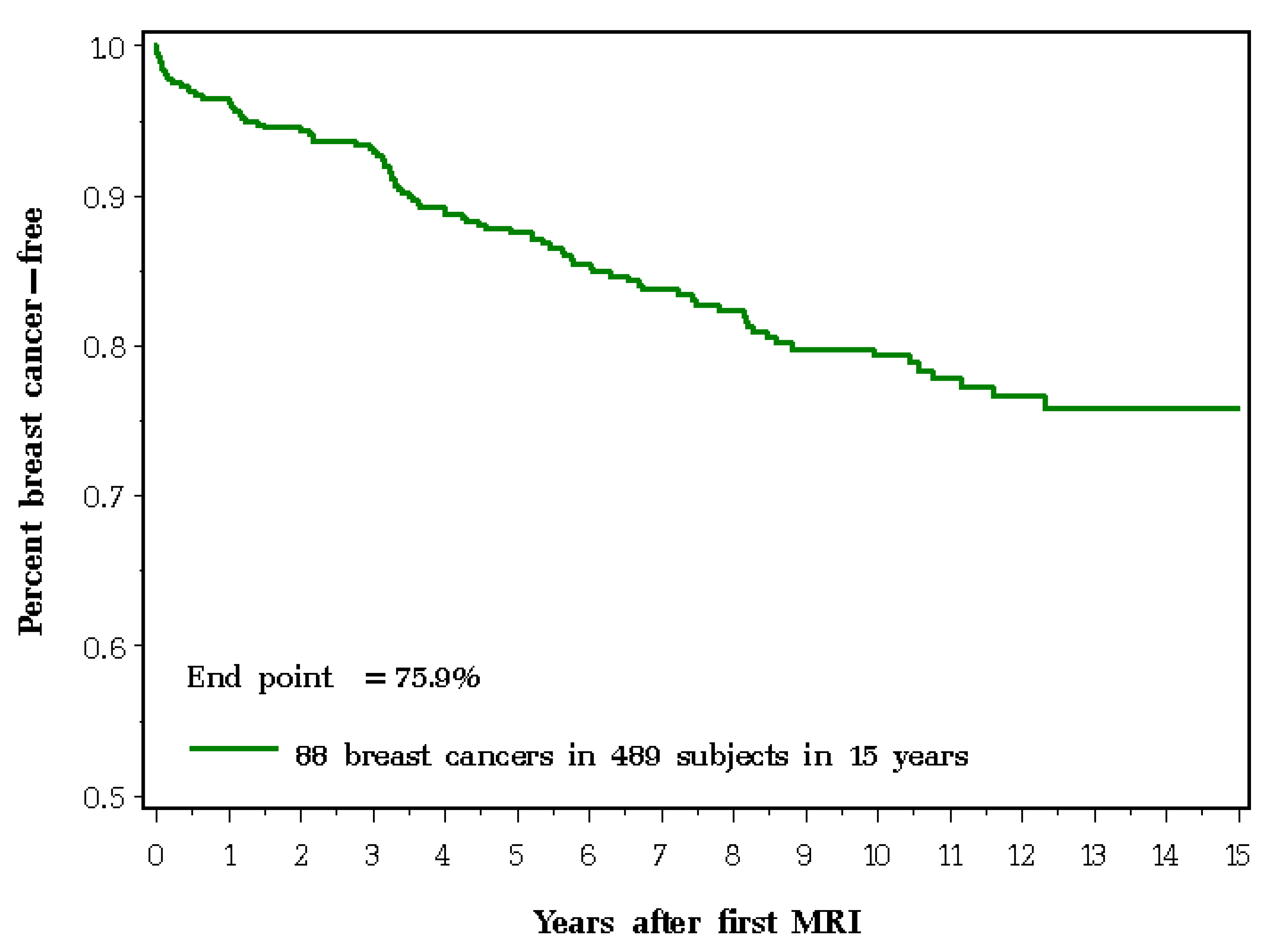
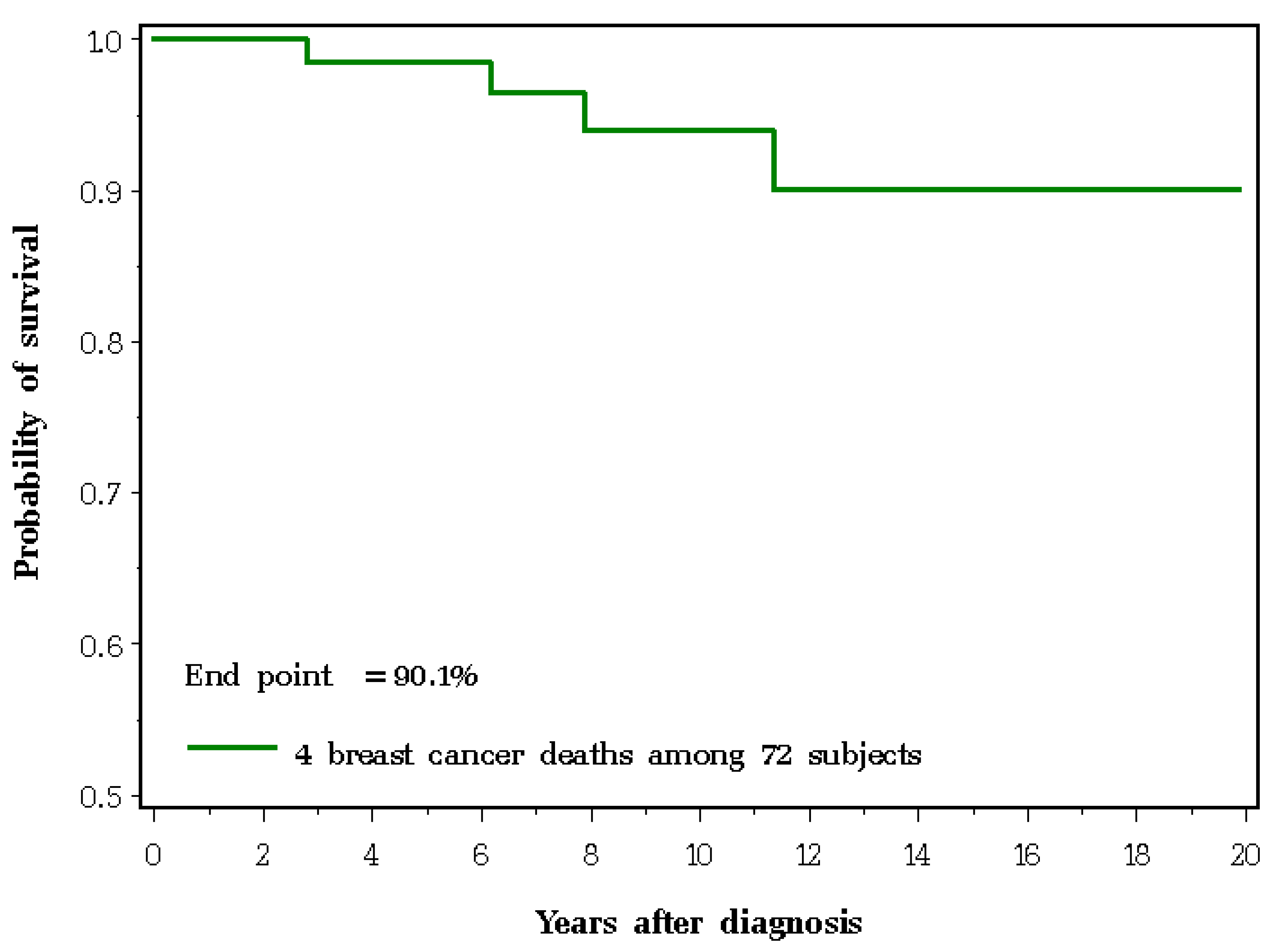
2. Results
3. Discussion
4. Materials and Methods
4.1. Study Subjects
4.2. Screening Protocol
4.3. Record Linkage
4.4. Statistical Analysis
4.5. Standardized Mortality Ratio
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Antoniou, A.; Pharoah, P.; Narod, S.; Risch, H.; Eyfjord, J.; Hopper, J.; Loman, N.; Olsson, H.; Johannsson, O.; Borg, Å.; et al. Average Risks of Breast and Ovarian Cancer Associated with BRCA1 or BRCA2 Mutations Detected in Case Series Unselected for Family History: A Combined Analysis of 22 Studies. Am. J. Hum. Genet. 2003, 72, 1117–1130. [Google Scholar] [PubMed]
- Chen, J.; Bae, E.; Zhang, L.; Hughes, K.; Parmigiani, G.; Braun, D.; Rebbeck, T.R. Penetrance of Breast and Ovarian Cancer in Women Who Carry a BRCA1/2 Mutation and Do Not Use Risk-Reducing Salpingo-Oophorectomy: An Updated Meta-Analysis. JNCI Cancer Spectr. 2020, 4. [Google Scholar] [CrossRef]
- Kuchenbaecker, K.B.; Hopper, J.L.; Barnes, D.R.; Phillips, K.-A.; Mooij, T.M.; Roos-Blom, M.-J.; Jervis, S.; Leeuwen, F.E.V.; Milne, R.L.; Andrieu, N.; et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 2017, 317, 2402. [Google Scholar] [PubMed]
- Carbine, N.E.; Lostumbo, L.; Wallace, J.; Ko, H. Risk-reducing mastectomy for the prevention of primary breast cancer. Cochrane Database Syst. Rev. 2018, 4, CD002748. [Google Scholar] [CrossRef]
- Kurian, A.W.; Sigal, B.M.; Plevritis, S.K. Survival analysis of cancer risk reduction strategies for BRCA1/2mutation carriers. J. Clin. Oncol. 2010, 28, 222–231. [Google Scholar]
- Domchek, S. Risk-Reducing Mastectomy in BRCA1 and BRCA2 Mutation Carriers: A Complex Discussion. JAMA 2019, 321, 27. [Google Scholar]
- Warner, E. Screening BRCA1 and BRCA2 Mutation Carriers for Breast Cancer. Cancers 2018, 10, 477. [Google Scholar]
- NICE Clinical Guideline. Familial Breast Cancer: Classification, Care and Managing Breast Cancer and Related Risks in People with a Family History of Breast Cancer. Available online: www.nice.org.uk/guidance/cg164 (accessed on 7 October 2020).
- NCCN Clinical Practice Guidelines in Oncology. (NCCN Guidelines): Genetic/Familial High Risk Assessment: Breast, Ovarian and Pancreatic. Available online: https://www.nccn.org/professionals/physician_gls/pdf/genetics_bop.pdf (accessed on 7 October 2020).
- Paluch-Shimon, S.; Cardoso, F.; Sessa, C.; Balmana, J.; Cardoso, M.J.; Gilbert, F.; Senkus, E. ESMO Guidelines Committee. Prevention and screening in BRCA mutation carriers and other breast/ovarian hereditary cancer syndromes: ESMO Clinical Practice Guidelines for cancer prevention and screening. Ann. Oncol. 2016, 27, v103–v110. [Google Scholar]
- Cancer Care Ontario Clinical Practice Guideline. Magnetic Resonance Imaging Screening of Women at High Risk for Breast Cancer v.3. Available online: www.cancercareontario.ca/en/guidelines-advice/types-of-cancer/2051 (accessed on 7 October 2020).
- Chiarelli, A.M.; Blackmore, K.M.; Muradali, D.; Done, S.J.; Majpruz, V.; Weerasinghe, A.; Mirea, L.; Eisen, A.; Rabeneck, L.; Warner, E. Performance Measures of Magnetic Resonance Imaging Plus Mammography in the High Risk Ontario Breast Screening Program. J. Natl. Cancer Inst. 2019, 112, 136–144. [Google Scholar]
- Tedeschi, E.; Caranci, F.; Giordano, F.; Angelini, V.; Cocozza, S.; Brunetti, A. Gadolinium retention in the body: What we know and what we can do. Radiol. Med. 2017, 122, 589–600. [Google Scholar]
- Heemskerk-Gerritsen, B.A.M.; Jager, A.; Koppert, L.B.; Obdeijn, A.I.-M.; Collée, M.; Meijers-Heijboer, H.E.J.; Jenner, D.J.; Oldenburg, H.S.A.; Engelen, K.V.; Vries, J.D.; et al. Survival after bilateral risk-reducing mastectomy in healthy BRCA1 and BRCA2 mutation carriers. Breast Cancer Res. Treat. 2019, 177, 723–733. [Google Scholar] [PubMed]
- Tilanus-Linthorst, M.M.; Obdeijn, I.M.; Hop, W.; Causer, P.A.; Leach, M.O.; Warner, E.; Pointon, L.; Hill, K.; Klijn, J.G.; Warren, R.M.; et al. A BRCA1 mutation and young age predict fast breast cancer growth in the Dutch, UK, and Canadian MRI screening trials. Clin. Cancer Res. 2007, 13, 7357–7362. [Google Scholar] [PubMed]
- Saadatmand, S.; Obdeijn, I.-M.; Rutgers, E.J.; Oosterwijk, J.C.; Tollenaar, R.A.; Woldringh, G.H.; Bergers, E.; Verhoef, C.; Heijnsdijk, E.A.; Hooning, M.J.; et al. Survival benefit in women with BRCA1mutation or familial risk in the MRI screening study (MRISC). Int. J. Cancer 2015, 137, 1729–1738. [Google Scholar] [PubMed]
- Gareth, E.D.; Nisha, K.; Yit, L.; Soujanye, G.; Emma, H.; Massat, N.J.; Maxwell, A.J.; Sarah, I.; Rosalind, E.; Leach, M.O.; et al. MRI breast screening in high-risk women: Cancer detection and survival analysis. Breast Cancer Res. Treat. 2014, 145, 663–672. [Google Scholar]
- Hadar, T.; Mor, P.; Amit, G.; Lieberman, S.; Gekhtman, D.; Rabinovitch, R.; Levy-Lahad, E. Presymptomatic awareness of germline pathogenic BRCA Variants and associated outcomes in women with Breast Cancer. JAMA Oncol. 2020, 6, 1460–1463. [Google Scholar]
- Saphner, T.; Tormey, D.C.; Gray, R. Annual hazard rates of recurrence for breast cancer after primary therapy. J. Clin. Oncol. 1996, 14, 2738–2746. [Google Scholar]
- Evans, D.G.; Howell, S.J.; Howell, A. Should unaffected female BRCA2 pathogenic variant mutation carriers be told there is little or no advantage from risk reducing mastectomy? Fam. Cancer 2019, 18, 377–379. [Google Scholar]
- Lobbezoo, D.J.A.; Van Kampen, R.J.W.; Voogd, A.C.; Dercksen, M.W.; Van Den Berkmortel, F.; Smilde, T.J.; Van De Wouw, A.J.; Peters, F.P.J.; Van Riel, J.M.G.H.; Peters, N.A.J.B.; et al. Prognosis of metastatic breast cancer: Are there differences between patients with de novo and recurrent metastatic breast cancer? Br. J. Cancer 2015, 112, 1445–1451. [Google Scholar]
- Carmichael, H.; Matsen, C.; Freer, P.; Kohlmann, W.; Stein, M.; Buys, S.S.; Colonna, S. Breast cancer screening of pregnant and breastfeeding women with BRCA mutations. Breast Cancer Res. Treat. 2017, 162, 225–230. [Google Scholar]
- Guindalini, R.S.C.; Zheng, Y.; Abe, H.; Whitaker, K.; Yoshimatsu, T.F.; Walsh, T.; Schacht, D.; Kulkarni, K.; Sheth, D.; Verp, M.S.; et al. Intensive Surveillance with Biannual Dynamic Contrast-Enhanced Magnetic Resonance Imaging Downstages Breast Cancer in BRCA1 Mutation Carriers. Clin. Cancer Res. 2018, 25, 1786–1794. [Google Scholar]
- Tutt, A.N.J.; Kaufman, B.; Gelber, R.D.; Mcfadden, E.; Goessl, C.D.; Viale, G.; Arahmani, A.; Fumagalli, D.; Azim, H.A.; Wu, W.; et al. OlympiA: A randomized phase III trial of olaparib as adjuvant therapy in patients with high-risk HER2-negative breast cancer and a germline BRCA1/2 mutation. J. Clin. Oncol. 2015, 33, 15. [Google Scholar]
- Lee, Y.-J.; Lee, S.-W.; Kim, K.-R.; Jung, K.-H.; Lee, J.-W.; Kim, Y.-M. Pathologic findings at risk-reducing salpingo-oophorectomy (RRSO) in germline BRCA mutation carriers with breast cancer: Significance of bilateral RRSO at the optimal age in germline BRCA mutation carriers. J. Gynecol. Oncol. 2017, 28. [Google Scholar] [CrossRef]
- Kauff, N.D.; Domchek, S.M.; Friebel, T.M.; Robson, M.E.; Lee, J.; Garber, J.E.; Isaacs, C.; Evans, D.G.; Lynch, H.; Eeles, R.A.; et al. Risk-Reducing Salpingo-Oophorectomy for the Prevention of BRCA1- and BRCA2-Associated Breast and Gynecologic Cancer: A Multicenter, Prospective Study. J. Clin. Oncol. 2008, 26, 1331–1337. [Google Scholar] [PubMed]
- Domchek, S.M.; Friebel, T.M.; Singer, C.F. Association of Risk-Reducing Surgery in BRCA1 or BRCA2 Mutation Carriers With Cancer Risk and Mortality. JAMA 2010, 304, 967–975. [Google Scholar]
- Mavaddat, N.; Antoniou, A.C. Risk-reducing salpingo-oophorectomy, natural menopause, and breast cancer risk: An international prospective cohort of BRCA1 and BRCA2 mutation carriers. Breast Cancer Res. 2020, 22, 8. [Google Scholar]
- Rodowa, M.; Salehi, A.H.; McGee, J. Primary Peritoneal Cancer Two Decades after a Bilateral Salpingo-Oophorectomy. Case Rep. Obstet. Gynecol. 2019, 2019, 1870834. [Google Scholar]
- Longo, D.L. Personalized medicine for primary treatment of serous ovarian cancer. N. Engl. J. Med. 2019, 381, 2471–2474. [Google Scholar]
- Warner, E.; Plewes, D.B.; Hill, K.A.; Causer, P.A.; Zubovits, J.T.; Jong, R.A.; Cutrara, M.R.; DeBoer, G.; Yaffe, M.J.; Messner, S.J.; et al. Surveillance of BRCA1 and BRCA2 mutation carriers with magnetic resonance imaging, ultrasound, mammography, and clinical breast examination. JAMA 2004, 292, 1317–1325. [Google Scholar]
- Passaperuma, K.; Warner, E.; Causer, P.A.; Hill, K.A.; Messner, S.; Wong, J.W.; Jong, R.A.; Wright, F.C.; Yaffe, M.J.; Ramsay, E.A.; et al. Long-term results of screening with magnetic resonance imaging in women with BRCA mutations. Br. J. Cancer 2012, 107, 24–30. [Google Scholar]
| Variables | Frequency or Mean Value |
|---|---|
| Year of Birth | 1962 (1936–1986) |
| Date of 1st MRI | 2005 (1997–2012) |
| Age at 1st MRI | 42.8 (25–66) |
| Mutation | |
| BRCA1 | 248 (50.7%) |
| BRCA2 | 241 (49.3%) |
| Preventive Mastectomy in Follow-up | |
| No | 394 (80.6%) |
| Yes | 95 (19.4%) |
| Oophorectomy | |
| No/missing | 237 (48.5%) |
| Yes | 252 (51.5%) |
| Breast Cancer | |
| No | 398 (81/4%) |
| Yes | 91 (18.6%) |
| Year of Diagnosis | 2009 (1998–2016) |
| Ovarian Cancer | |
| No | 476 (97.3%) |
| Yes | 13 (2.7%) |
| Year of diagnosis | 2008 (2000–2017) |
| Other Cancer | |
| No | 468 (95.7%) |
| Yes | 21 (4.3%) |
| Pancreas | 4 |
| Skin | 4 |
| Cervix | 3 |
| Colorectal | 2 |
| Thyroid | 2 |
| Endometrial | 1 |
| Kidney | 1 |
| Lung | 1 |
| Lymphoma | 1 |
| Unknown | 2 |
| Alive | 471 (96.3%) |
| Dead | 18 (3.7%) |
| Date of death | 2012 (2004–2018) |
| Cause of death | |
| Breast Cancer | 4 |
| Ovarian cancer | 5 |
| Pancreatic cancer | 4 |
| Lung cancer | 1 |
| Other/missing | 4 |
| Variables | Invasive (n = 72) | DCIS (n = 19) | |
|---|---|---|---|
| Year of birth: mean (range) | 1960 (1937–1981) | 1959 (1946–1974) | |
| Age at diagnosis: mean (range) | 48.6 (31–69) | 48.4(35–67) | |
| Tumour size (cm): mean (range) | 1.03 (0–3.0) (n = 69) | 0.64 (0–2.2) (n = 14) | |
| Node status | |||
| Negative | 58 (86.6%) | 6 (100%) | |
| Positive | 9 (13.4%) | 0 | |
| Missing | 5 | 13 | |
| Stage | |||
| 1 | 49 (74.2%) | n/a | |
| 2 or higher | 17 (25.8%) | ||
| Estrogen Receptor status | |||
| Positive | 41 (59.4%) | 6 (66.7%) | |
| Negative | 28 (40.6%) | 3 (33.3%) | |
| Missing | 3 | 10 | |
| Chemotherapy | |||
| No | 32 (47.8%) | 14 (100%) | |
| Yes | 35 (52.2%) | 0 | |
| Missing | 5 | 5 |
| Study ID | Gene | Yr of Birth | Age at 1st MRI | Age at Diagnosis | Means of Detection | Size (cm) | Node Status | ER | Chemo | HT | Age at Recurrence | Age of Death |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 296 | BRCA2 | 1942 | 59 | 63 | MRI | 0.7 | - | + | No | No | 70 | 74 |
| 424 | BRCA1 | 1974 | 30 | 33 | Self | 1.5 | + | - | Yes | n/a | 35 | 37 |
| 455 | BRCA1 | 1955 | 48 | 48 | MRI | 3.0 | + | + | Yes | Yes | 54 | 55 |
| 566 | BRCA1 | 1965 | 41 | 45 | MRI | 0.11 | - | - | No | n/a | 50 | 53 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warner, E.; Zhu, S.; Plewes, D.B.; Hill, K.; Ramsay, E.A.; Causer, P.A.; Seely, J.; Jong, R.A.; Lenkov, P.; Elser, C.; et al. Breast Cancer Mortality among Women with a BRCA1 or BRCA2 Mutation in a Magnetic Resonance Imaging Plus Mammography Screening Program. Cancers 2020, 12, 3479. https://doi.org/10.3390/cancers12113479
Warner E, Zhu S, Plewes DB, Hill K, Ramsay EA, Causer PA, Seely J, Jong RA, Lenkov P, Elser C, et al. Breast Cancer Mortality among Women with a BRCA1 or BRCA2 Mutation in a Magnetic Resonance Imaging Plus Mammography Screening Program. Cancers. 2020; 12(11):3479. https://doi.org/10.3390/cancers12113479
Chicago/Turabian StyleWarner, Ellen, Siqi Zhu, Donald B. Plewes, Kimberley Hill, Elizabeth A. Ramsay, Petrina A. Causer, Jean Seely, Roberta A. Jong, Pamela Lenkov, Christine Elser, and et al. 2020. "Breast Cancer Mortality among Women with a BRCA1 or BRCA2 Mutation in a Magnetic Resonance Imaging Plus Mammography Screening Program" Cancers 12, no. 11: 3479. https://doi.org/10.3390/cancers12113479
APA StyleWarner, E., Zhu, S., Plewes, D. B., Hill, K., Ramsay, E. A., Causer, P. A., Seely, J., Jong, R. A., Lenkov, P., Elser, C., Crystal, P., Yaffe, M. J., Giannakeas, V., Sun, P., & Narod, S. A. (2020). Breast Cancer Mortality among Women with a BRCA1 or BRCA2 Mutation in a Magnetic Resonance Imaging Plus Mammography Screening Program. Cancers, 12(11), 3479. https://doi.org/10.3390/cancers12113479







