Nimotuzumab Site-Specifically Labeled with 89Zr and 225Ac Using SpyTag/SpyCatcher for PET Imaging and Alpha Particle Radioimmunotherapy of Epidermal Growth Factor Receptor Positive Cancers
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
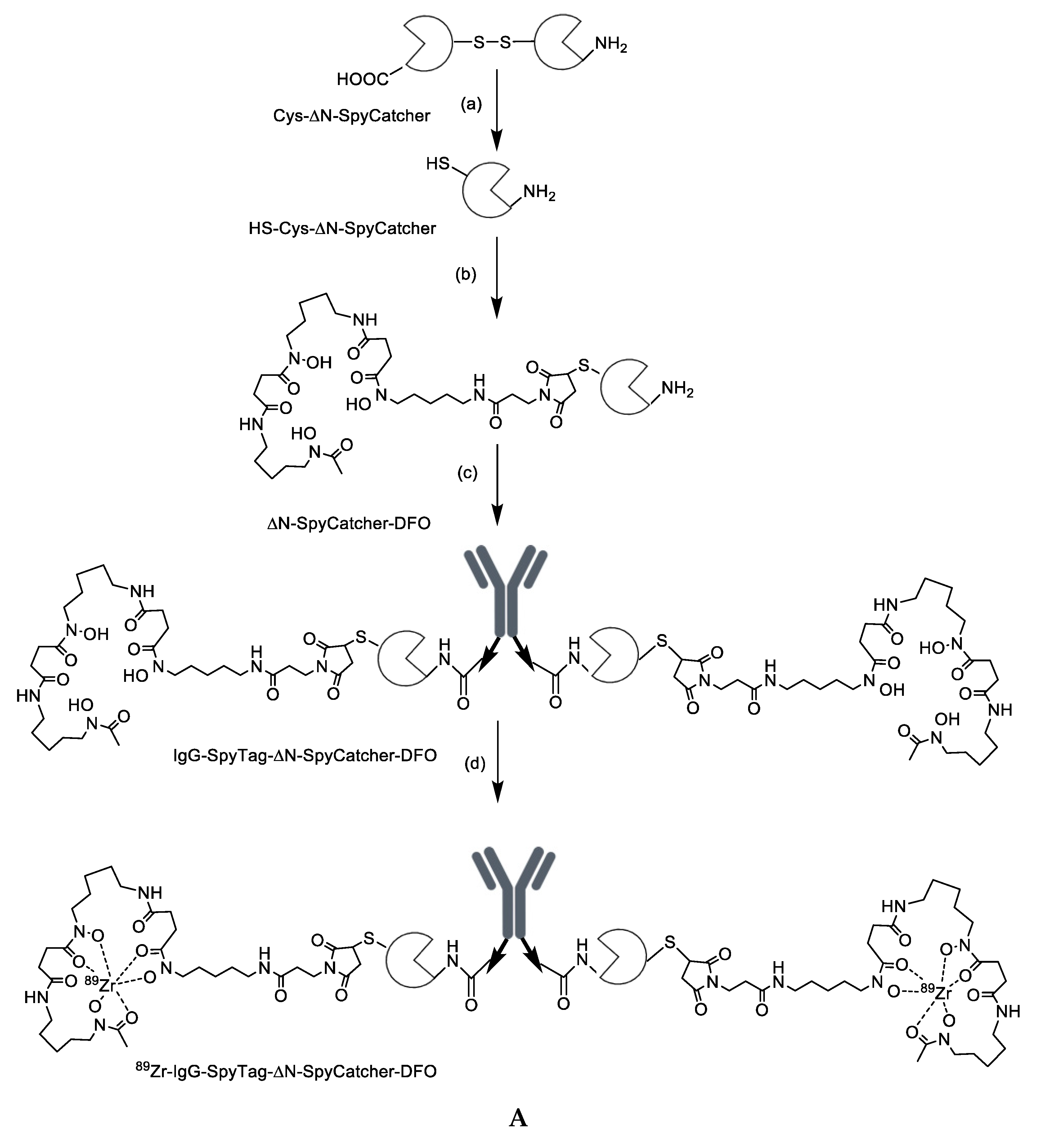
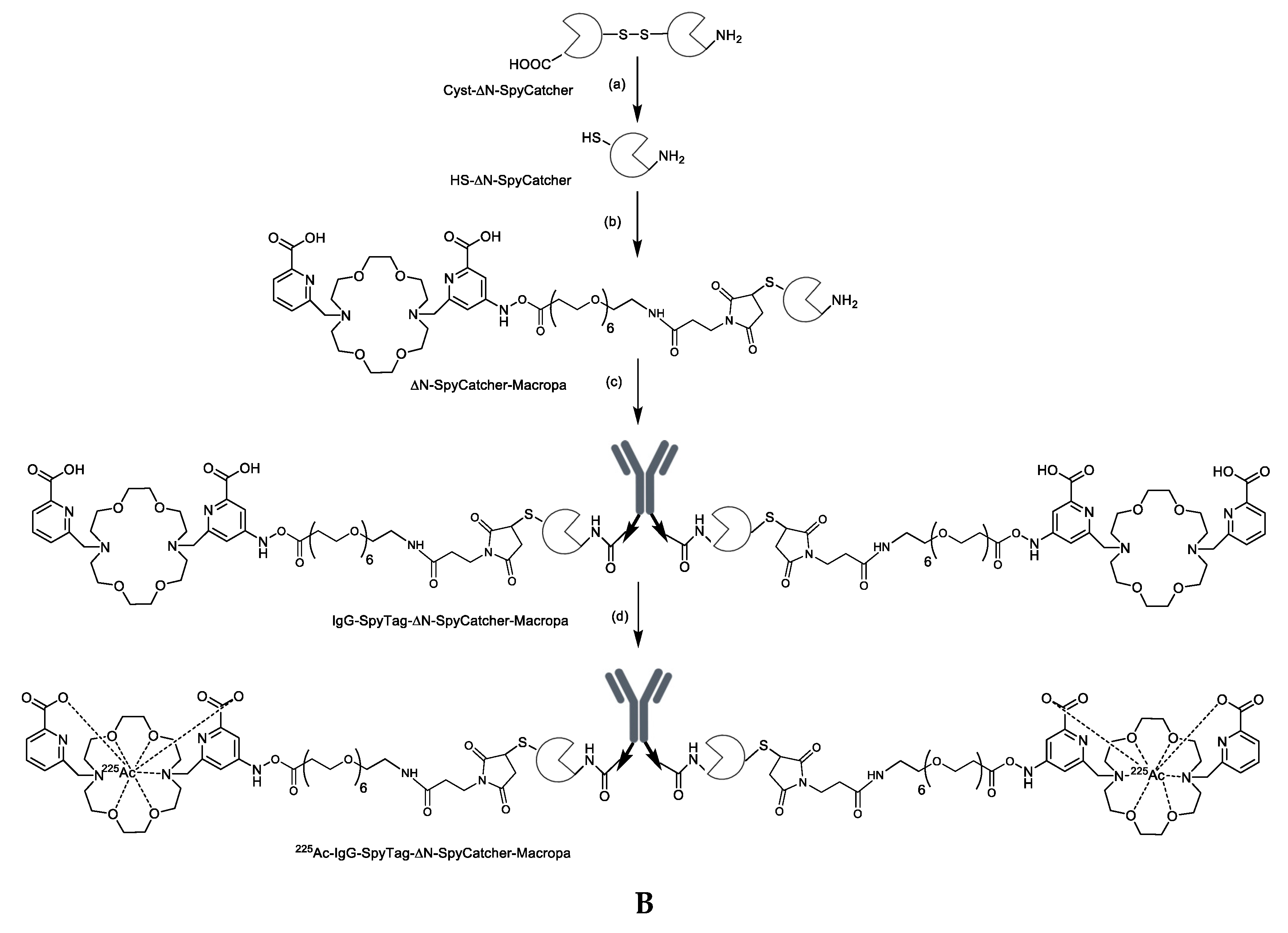
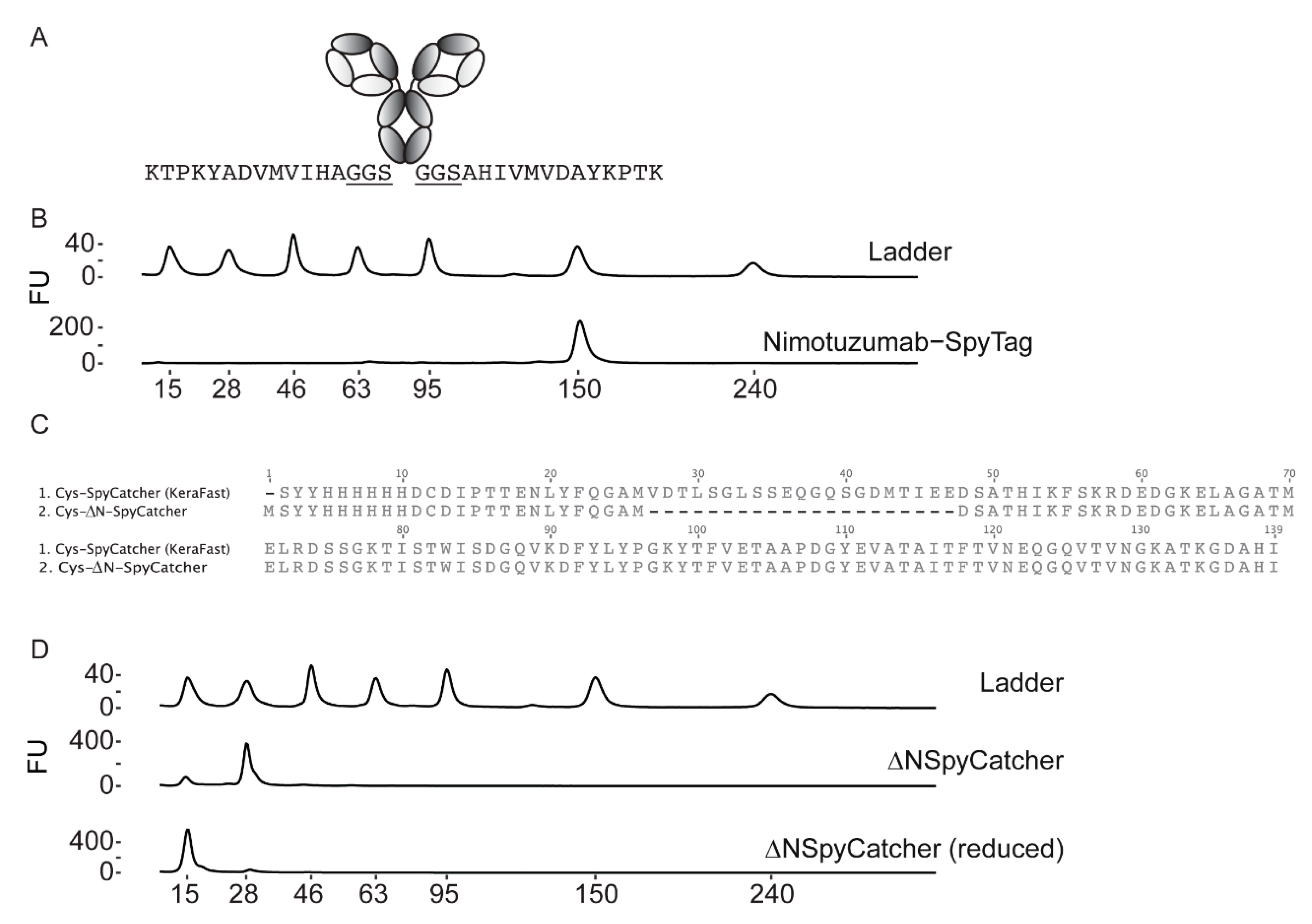
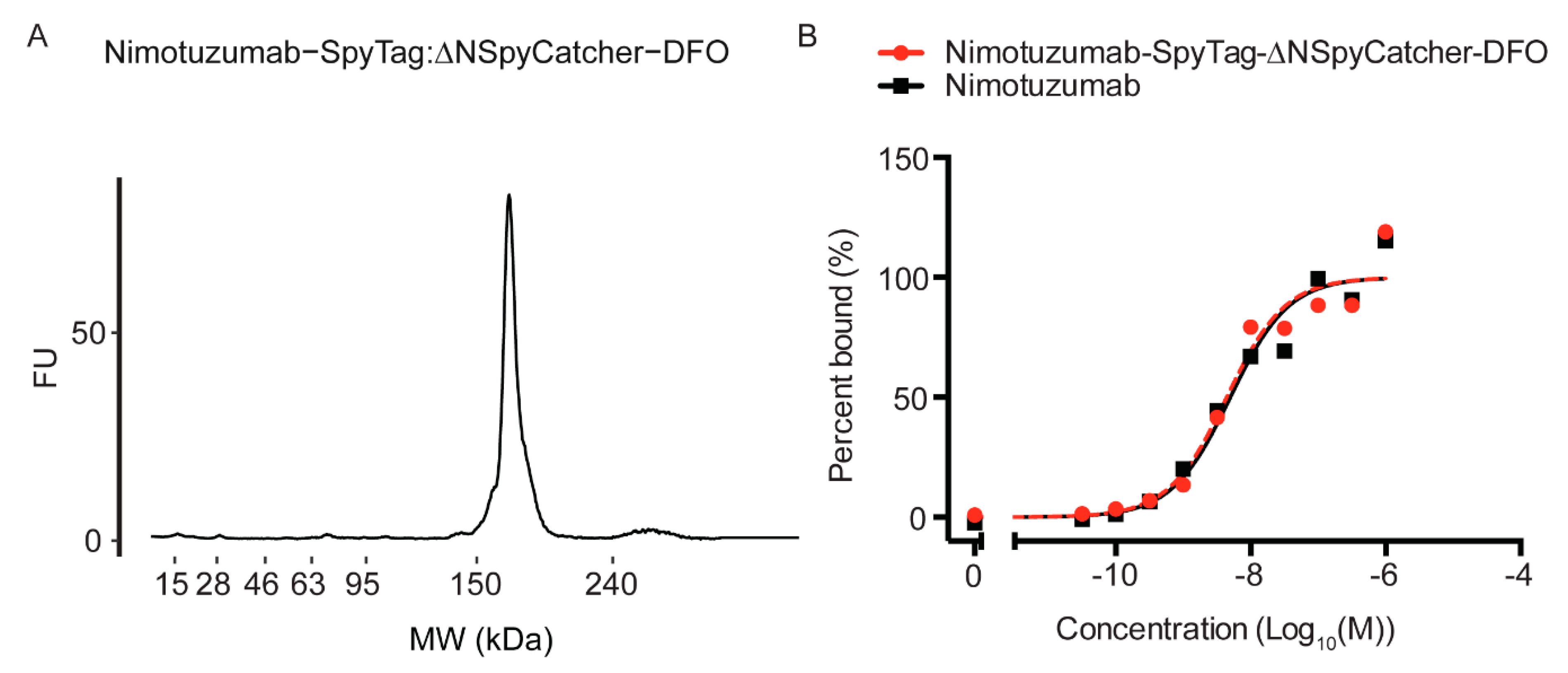
2.1. Synthesis and Characterization of Antibody-SpyTag and DFO-Cysteine-∆N-SpyCatcher
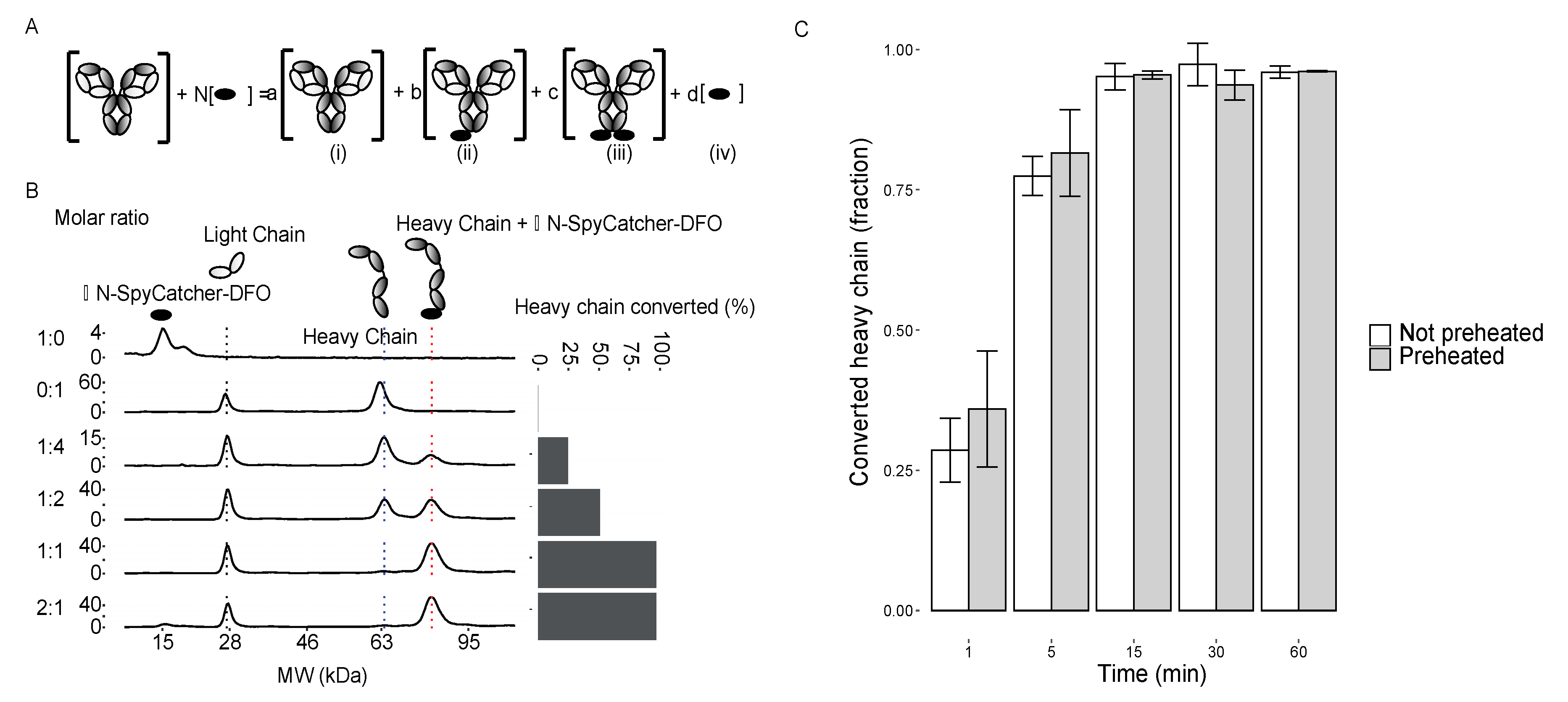
2.2. Characterization of the DFO-∆N-SpyCatcher IgG-SpyTag Immunoconjugate
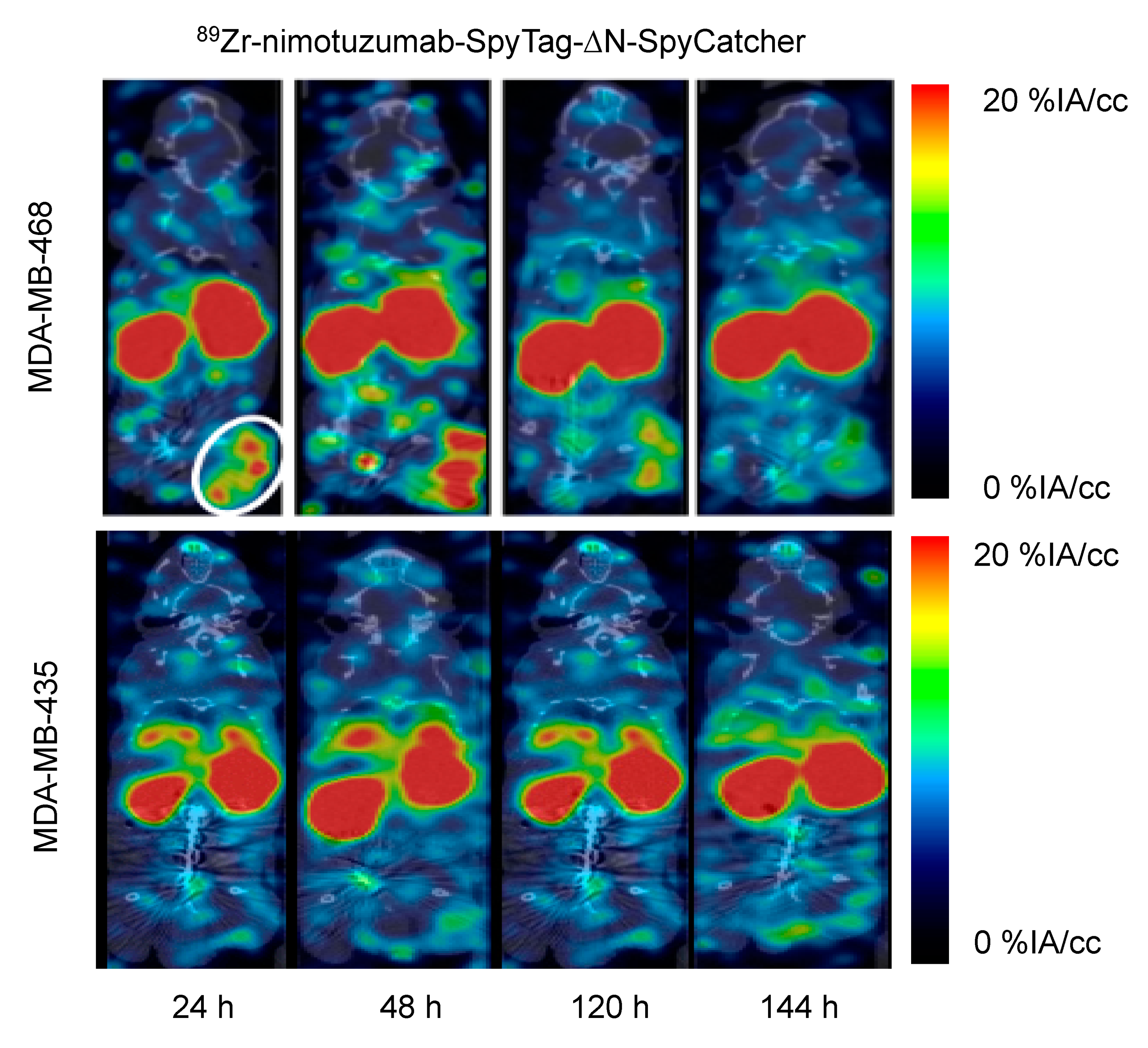
2.3. 89Zr radiolabeling, Characterization, and MicroPET/CT Imaging
2.4. 225Ac radiolabeling, Characterization, and In Vitro Cytotoxicity
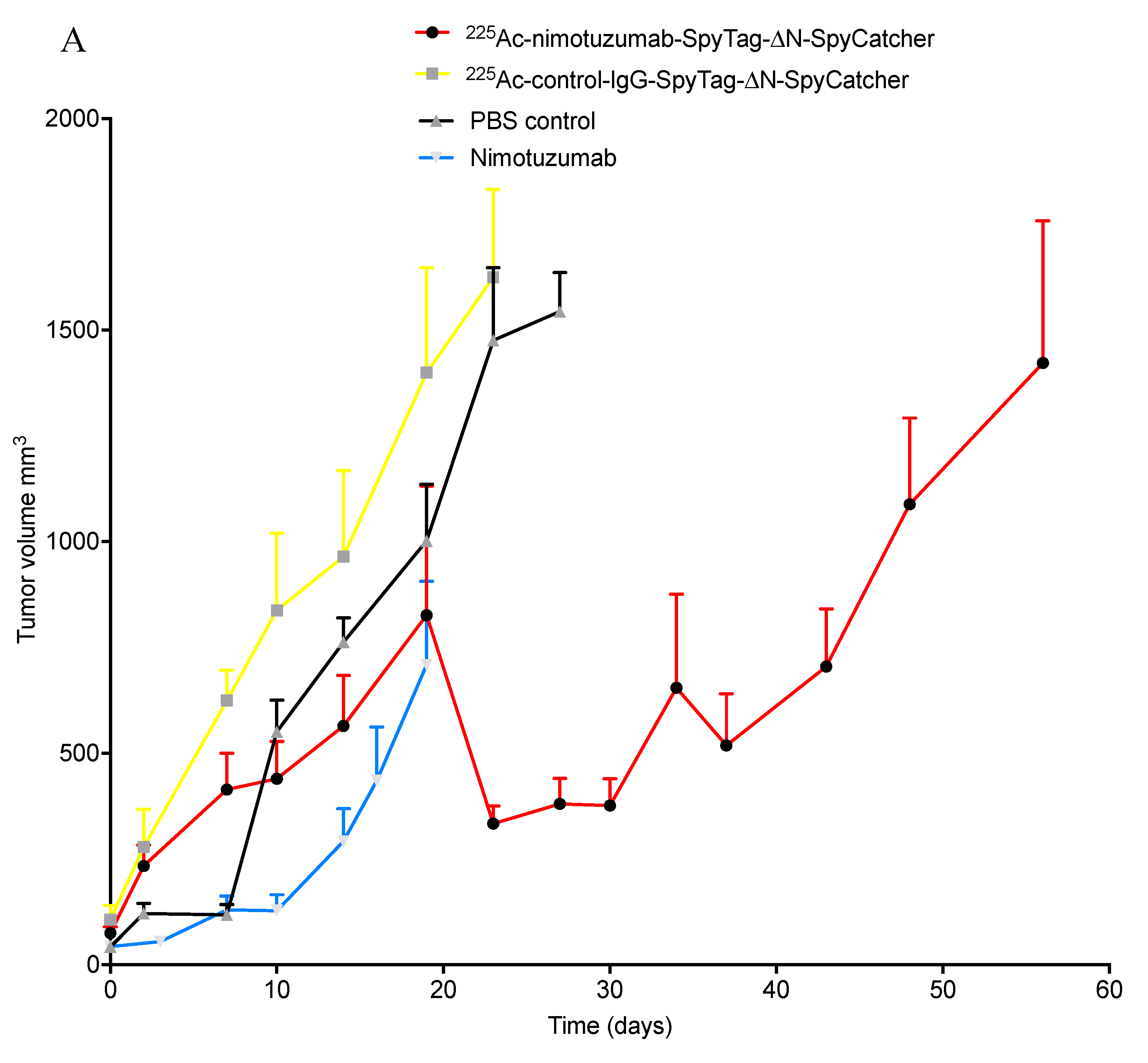
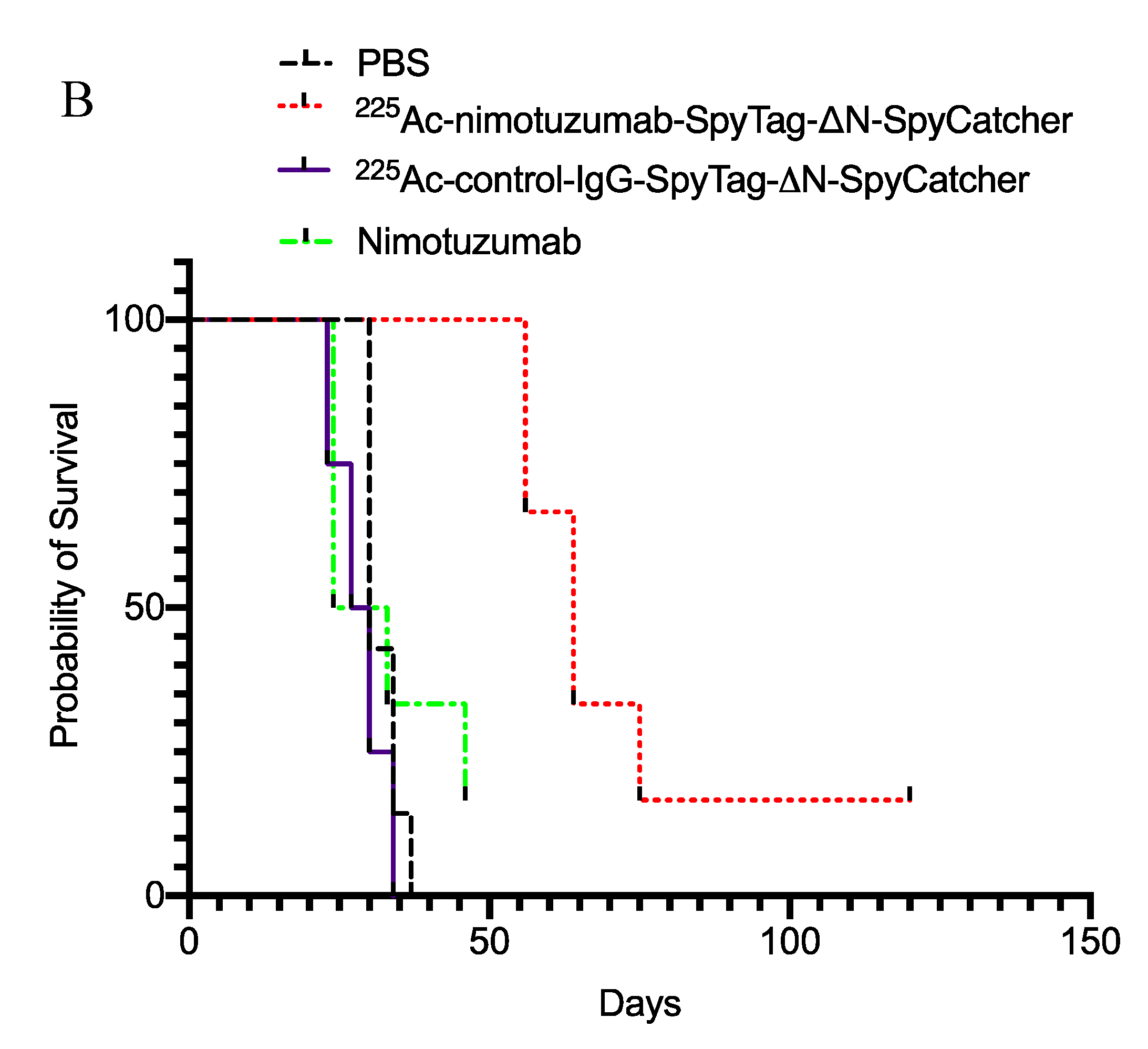
2.5. In Vivo Efficacy of 225Ac-Radioimmunoconjugates
3. Discussion
4. Experimental Section
4.1. General
4.2. Plasmids
4.3. Expression and Purification of His6-Cysteine-ΔN-SpyCatcher and Antibodies
4.4. Conjugation of Chelator to Cys-∆N-SpyCatcher
4.5. Bioconjugation of IgG-SpyTag with Chelator-∆N-SpyCatcher
4.6. Quality Control of Immunoconjugates
4.7. Cell Culture
4.8. Flow Cytometry
4.9. Radiolabeling with 89Zr and 225Ac
4.10. Stability of Radioimmunoconjugates
4.11. Biodistribution, Tumor Xenografts and MicroPET/CT Imaging
4.12. In Vitro Cytotoxicity
4.13. 225Ac Radioimmunotherapy
4.14. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| HEPES | 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid |
| CT | Computed tomography |
| DFO | Desferrioxamine |
| EC50 | Effective concentration |
| EGFR | Epidermal growth factor receptor I |
| EDTA | Ethylenediaminetetraacetic acid |
| LET | linear energy transfer |
| PET | Positron emission tomography |
References
- Addeo, R.; Caraglia, M.; Cerbone, D.; Frega, N.; Cimmino, G.; Abbruzzese, A.; Del Prete, S. Panitumumab: A new frontier of target therapy for the treatment of metastatic colorectal cancer. Expert Rev. Anticancer. Ther. 2010, 10, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Alshenawy, H.A. Immunohistochemical expression of epidermal growth factor receptor, E-cadherin, and matrix metalloproteinase-9 in ovarian epithelial cancer and relation to patient deaths. Ann. Diagn. Pathol. 2010, 14, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Bellone, S.; Frera, G.; Landolfi, G.; Romani, C.; Bandiera, E.; Tognon, G.; Roman, J.J.; Burnett, A.F.; Pecorelli, S.; Santin, A.D. Overexpression of epidermal growth factor type-1 receptor (EGF-R1) in cervical cancer: Implications for Cetuximab-mediated therapy in recurrent/metastatic disease. Gynecol. Oncol. 2007, 106, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Bonner, J.A.; Harari, P.M.; Giralt, J.; Azarnia, N.; Shin, D.M.; Cohen, R.B.; Jones, C.U.; Sur, R.; Raben, D.; Jassem, J.; et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N. Engl. J. Med. 2006, 354, 567–578. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.H.; Ely, K.; McGavran, L.; Varella-Garcia, M.; Parker, J.; Parker, N.; Jarrett, C.; Carter, J.; Murphy, B.A.; Netterville, J.; et al. Increased epidermal growth factor receptor gene copy number is associated with poor prognosis in head and neck squamous cell carcinomas. J. Clin. Oncol. 2006, 24, 4170–4176. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, D.; Humblet, Y.; Siena, S.; Khayat, D.; Bleiberg, H.; Santoro, A.; Bets, D.; Mueser, M.; Harstrick, A.; Verslype, C.; et al. Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N. Engl. J. Med. 2004, 351, 337–345. [Google Scholar] [CrossRef]
- Garnock-Jones, K.P. Necitumumab: First Global Approval. Drugs 2016, 76, 283–289. [Google Scholar] [CrossRef]
- Giltnane, J.M.; Ryden, L.; Cregger, M.; Bendahl, P.O.; Jirstrom, K.; Rimm, D.L. Quantitative measurement of epidermal growth factor receptor is a negative predictive factor for tamoxifen response in hormone receptor positive premenopausal breast cancer. J. Clin. Oncol. 2007, 25, 3007–3014. [Google Scholar] [CrossRef]
- Crombet, T.; Osorio, M.; Cruz, T.; Roca, C.; del Castillo, R.; Mon, R.; Iznaga-Escobar, N.; Figueredo, R.; Koropatnick, J.; Renginfo, E.; et al. Use of the humanized anti-epidermal growth factor receptor monoclonal antibody h-R3 in combination with radiotherapy in the treatment of locally advanced head and neck cancer patients. J. Clin. Oncol. 2004, 22, 1646–1654. [Google Scholar] [CrossRef]
- Keating, G.M. Panitumumab: A review of its use in metastatic colorectal cancer. Drugs 2010, 70, 1059–1078. [Google Scholar] [CrossRef]
- Molinari, E.; De Quatrebarbes, J.; Andre, T.; Aractingi, S. Cetuximab-induced acne. Dermatology 2005, 211, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Ramos, T.C.; Figueredo, J.; Catala, M.; Gonzalez, S.; Selva, J.C.; Cruz, T.M.; Toledo, C.; Silva, S.; Pestano, Y.; Ramos, M.; et al. Treatment of high-grade glioma patients with the humanized anti-epidermal growth factor receptor (EGFR) antibody h-R3: Report from a phase I/II trial. Cancer Biol. Ther. 2006, 5, 375–379. [Google Scholar] [CrossRef]
- Rojo, F.; Gracias, E.; Villena, N.; Cruz, T.; Corominas, J.M.; Corradino, I.; Cedeno, M.; Campas, C.; Osorio, M.; Iznaga, N.; et al. Pharmacodynamic trial of nimotuzumab in unresectable squamous cell carcinoma of the head and neck: A SENDO Foundation study. Clin. Cancer Res. 2010, 16, 2474–2482. [Google Scholar] [CrossRef] [PubMed]
- Garrido, G.; Tikhomirov, I.A.; Rabasa, A.; Yang, E.; Gracia, E.; Iznaga, N.; Fernandez, L.E.; Crombet, T.; Kerbel, R.S.; Perez, R. Bivalent binding by intermediate affinity of nimotuzumab: A contribution to explain antibody clinical profile. Cancer Biol. Ther. 2011, 11, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Hartimath, S.V.; Alizadeh, E.; Solomon, V.R.; Chekol, R.; Bernhard, W.; Hill, W.; Parada, A.C.; Barreto, K.; Geyer, C.R.; Fonge, H. Preclinical Evaluation of 111In-Labeled PEGylated Maytansine Nimotuzumab Drug Conjugates in EGFR-Positive Cancer Models. J. Nucl. Med. 2019, 60, 1103–1110. [Google Scholar] [CrossRef]
- Sutherland, A.R.; Alam, M.K.; Geyer, C.R. Post-translational Assembly of Protein Parts into Complex Devices by Using SpyTag/SpyCatcher Protein Ligase. Chembiochem Eur. J. Chem. Biol. 2019, 20, 319–328. [Google Scholar] [CrossRef]
- Aghevlian, S.; Lu, Y.; Winnik, M.A.; Hedley, D.W.; Reilly, R.M. Panitumumab Modified with Metal-Chelating Polymers (MCP) Complexed to 111In and 177Lu-An EGFR-Targeted Theranostic for Pancreatic Cancer. Mol. Pharm. 2018, 15, 1150–1159. [Google Scholar] [CrossRef]
- Bensch, F.; van der Veen, E.L.; Lub-de Hooge, M.N.; Jorritsma-Smit, A.; Boellaard, R.; Kok, I.C.; Oosting, S.F.; Schroder, C.P.; Hiltermann, T.J.N.; van der Wekken, A.J.; et al. 89Zr-atezolizumab imaging as a non-invasive approach to assess clinical response to PD-L1 blockade in cancer. Nat. Med. 2018, 24, 1852–1858. [Google Scholar] [CrossRef]
- Jauw, Y.W.; Menke-van der Houven van Oordt, C.W.; Hoekstra, O.S.; Hendrikse, N.H.; Vugts, D.J.; Zijlstra, J.M.; Huisman, M.C.; van Dongen, G.A. Immuno-Positron Emission Tomography with Zirconium-89-Labeled Monoclonal Antibodies in Oncology: What Can We Learn from Initial Clinical Trials? Front. Pharmacol. 2016, 7, 131. [Google Scholar] [CrossRef]
- McKnight, B.N.; Viola-Villegas, N.T. 89 Zr-ImmunoPET companion diagnostics and their impact in clinical drug development. J. Label. Compd. Radiopharm. 2018, 61, 727–738. [Google Scholar] [CrossRef]
- Deri, M.A.; Zeglis, B.M.; Francesconi, L.C.; Lewis, J.S. PET imaging with 89Zr: From radiochemistry to the clinic. Nucl. Med. Biol. 2013, 40, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Morgenstern, A.; Apostolidis, C.; Kratochwil, C.; Sathekge, M.; Krolicki, L.; Bruchertseifer, F. An Overview of Targeted Alpha Therapy with 225Actinium and 213Bismuth. Curr. Radiopharm. 2018, 11, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Zakeri, B.; Fierer, J.O.; Celik, E.; Chittock, E.C.; Schwarz-Linek, U.; Moy, V.T.; Howarth, M. Peptide tag forming a rapid covalent bond to a protein, through engineering a bacterial adhesin. Proc. Natl. Acad. Sci. USA 2012, 109, E690–E697. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, N.B.; Pandya, D.N.; Wadas, T.J. Recent Advances in Zirconium-89 Chelator Development. Molecules 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhou, H.; Wang, W.; Tan, W.; Fu, Y.X.; Zhu, M. A novel method for synthetic vaccine construction based on protein assembly. Sci. Rep. 2014, 4, 7266. [Google Scholar] [CrossRef] [PubMed]
- Keeble, A.H.; Banerjee, A.; Ferla, M.P.; Reddington, S.C.; Anuar, I.; Howarth, M. Evolving Accelerated Amidation by SpyTag/SpyCatcher to Analyze Membrane Dynamics. Angew. Chem. 2017, 56, 16521–16525. [Google Scholar] [CrossRef] [PubMed]
- Chekol, R.; Solomon, V.R.; Alizadeh, E.; Bernhard, W.; Fisher, D.; Hill, W.; Barreto, K.; DeCoteau, J.F.; Parada, A.C.; Geyer, C.R.; et al. 89Zr-nimotuzumab for immunoPET imaging of epidermal growth factor receptor I. Oncotarget 2018, 9, 17117–17132. [Google Scholar] [CrossRef]
- Alam, M.K.; El-Sayed, A.; Barreto, K.; Bernhard, W.; Fonge, H.; Geyer, C.R. Site-Specific Fluorescent Labeling of Antibodies and Diabodies Using SpyTag/SpyCatcher System for In Vivo Optical Imaging. Mol. Imaging Biol. 2019, 21, 54–66. [Google Scholar] [CrossRef]
- Heskamp, S.; Raave, R.; Boerman, O.; Rijpkema, M.; Goncalves, V.; Denat, F. 89Zr-Immuno-Positron Emission Tomography in Oncology: State-of-the-Art 89Zr Radiochemistry. Bioconjugate Chem. 2017, 28, 2211–2223. [Google Scholar] [CrossRef]
- McDevitt, M.R.; Ma, D.; Lai, L.T.; Simon, J.; Borchardt, P.; Frank, R.K.; Wu, K.; Pellegrini, V.; Curcio, M.J.; Miederer, M.; et al. Tumor therapy with targeted atomic nanogenerators. Science 2001, 294, 1537–1540. [Google Scholar] [CrossRef]
- McDevitt, M.R.; Ma, D.; Simon, J.; Frank, R.K.; Scheinberg, D.A. Design and synthesis of 225Ac radioimmunopharmaceuticals. Appl. Radiat. Isot. 2002, 57, 841–847. [Google Scholar] [CrossRef]
- Pandya, D.N.; Hantgan, R.; Budzevich, M.M.; Kock, N.D.; Morse, D.L.; Batista, I.; Mintz, A.; Li, K.C.; Wadas, T.J. Preliminary Therapy Evaluation of 225Ac-DOTA-c(RGDyK) Demonstrates that Cerenkov Radiation Derived from 225Ac Daughter Decay Can Be Detected by Optical Imaging for In Vivo Tumor Visualization. Theranostics 2016, 6, 698–709. [Google Scholar] [CrossRef] [PubMed]
- Pruszynski, M.; D’Huyvetter, M.; Bruchertseifer, F.; Morgenstern, A.; Lahoutte, T. Evaluation of an Anti-HER2 Nanobody Labeled with 225Ac for Targeted alpha-Particle Therapy of Cancer. Mol. Pharm. 2018, 15, 1457–1466. [Google Scholar] [CrossRef] [PubMed]
- Thiele, N.A.; Brown, V.; Kelly, J.M.; Amor-Coarasa, A.; Jermilova, U.; MacMillan, S.N.; Nikolopoulou, A.; Ponnala, S.; Ramogida, C.F.; Robertson, A.K.H.; et al. An Eighteen-Membered Macrocyclic Ligand for Actinium-225 Targeted Alpha Therapy. Angew. Chem. 2017, 56, 14712–14717. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, S.D.; Reid, R.; Robinson, L.; Ashley, G.W.; Santi, D.V. Long-term stabilization of maleimide-thiol conjugates. Bioconjugate Chem. 2015, 26, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Ross, B.; Mehta, S.; Zhang, J. Molecular tools for acute spatiotemporal manipulation of signal transduction. Curr. Opin. Chem. Biol. 2016, 34, 135–142. [Google Scholar] [CrossRef]
- Solomon, V.R.; Alizadeh, E.; Bernhard, W.; Hartimath, S.V.; Hill, W.; Chekol, R.; Barreto, K.M.; Geyer, C.R.; Fonge, H. 111In- and 225Ac-Labeled Cixutumumab for Imaging and alpha-Particle Radiotherapy of IGF-1R Positive Triple-Negative Breast Cancer. Mol. Pharm. 2019. [Google Scholar] [CrossRef]
- Solomon, V.R.; Gonzalez, C.; Alizadeh, E.; Bernhard, W.; Hartimath, S.V.; Barreto, K.; Geyer, C.R.; Fonge, H. (99m)Tc(CO)3(+) labeled domain I/II-specific anti-EGFR (scFv)2 antibody fragment for imaging EGFR expression. Eur. J. Med. Chem. 2018, 157, 437–446. [Google Scholar] [CrossRef]
| Organs | 89Zr-Nimotuzumab-SpyTag-∆N-SpyCatcher | 89Zr-Control-IgG-SpyTag-∆N-SpyCatcher | ||
|---|---|---|---|---|
| 24 h | 144 h | 24 h | 144 h | |
| Kidney | 19.2 ± 5.4 | 24.4 ± 1.8 | 14.4 ± 2.9 | 12.6 ± 1.4 |
| Liver | 3.8 ± 1.3 | 2.4 ± 0.3 | 4.6 ± 1.8 | 2.0 ± 0.4 |
| Pancreas | 0.4 ± 0.1 | 0.2 ± 0.0 | 0.2 ± 0.1 | 0.2 ± 0.0 |
| Spleen | 2.2 ± 1.0 | 2.1 ± 0.1 | 1.2 ± 0.7 | 3.5 ± 0.6 |
| Lungs | 1.7 ± 0.3 | 0.7 ± 0.3 | 0.5 ± 0.3 | 0.7 ± 0.3 |
| Heart | 1.8 ± 1.9 | 0.4 ± 0.1 | 0.5 ± 0.2 | 0.4 ± 0.2 |
| Stomach | 0.3 ± 0.2 | 0.1 ± 0.1 | 0.2 ± 0.0 | 0.1 ± 0.0 |
| Intestine | 0.4 ± 0.1 | 0.2 ± 0.1 | 0.3 ± 0.1 | 0.2 ± 0.0 |
| Blood | 3.6 ± 0.8 | 1.0 ± 0.3 | 1.1 ± 0.3 | 1.0 ± 0.8 |
| Bone | 0.7 ± 0.2 | 0.3 ± 0.0 | 0.4 ± 0.2 | 0.4 ± 0.0 |
| Muscle | 0.3 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.1 | 0.2 ± 0.2 |
| Skin | 1.2 ± 0.2 | 0.7 ± 0.1 | 0.8 ± 0.2 | 0.8 ± 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solomon, V.R.; Barreto, K.; Bernhard, W.; Alizadeh, E.; Causey, P.; Perron, R.; Gendron, D.; Alam, M.K.; Carr, A.; Geyer, C.R.; et al. Nimotuzumab Site-Specifically Labeled with 89Zr and 225Ac Using SpyTag/SpyCatcher for PET Imaging and Alpha Particle Radioimmunotherapy of Epidermal Growth Factor Receptor Positive Cancers. Cancers 2020, 12, 3449. https://doi.org/10.3390/cancers12113449
Solomon VR, Barreto K, Bernhard W, Alizadeh E, Causey P, Perron R, Gendron D, Alam MK, Carr A, Geyer CR, et al. Nimotuzumab Site-Specifically Labeled with 89Zr and 225Ac Using SpyTag/SpyCatcher for PET Imaging and Alpha Particle Radioimmunotherapy of Epidermal Growth Factor Receptor Positive Cancers. Cancers. 2020; 12(11):3449. https://doi.org/10.3390/cancers12113449
Chicago/Turabian StyleSolomon, Viswas Raja, Kris Barreto, Wendy Bernhard, Elahe Alizadeh, Patrick Causey, Randy Perron, Denise Gendron, Md. Kausar Alam, Adriana Carr, C. Ronald Geyer, and et al. 2020. "Nimotuzumab Site-Specifically Labeled with 89Zr and 225Ac Using SpyTag/SpyCatcher for PET Imaging and Alpha Particle Radioimmunotherapy of Epidermal Growth Factor Receptor Positive Cancers" Cancers 12, no. 11: 3449. https://doi.org/10.3390/cancers12113449
APA StyleSolomon, V. R., Barreto, K., Bernhard, W., Alizadeh, E., Causey, P., Perron, R., Gendron, D., Alam, M. K., Carr, A., Geyer, C. R., & Fonge, H. (2020). Nimotuzumab Site-Specifically Labeled with 89Zr and 225Ac Using SpyTag/SpyCatcher for PET Imaging and Alpha Particle Radioimmunotherapy of Epidermal Growth Factor Receptor Positive Cancers. Cancers, 12(11), 3449. https://doi.org/10.3390/cancers12113449






