Impact of the First Wave of COVID-19 on Pediatric Oncology and Hematology: A Report from the French Society of Pediatric Oncology
Abstract
Simple Summary
Abstract
1. Introduction
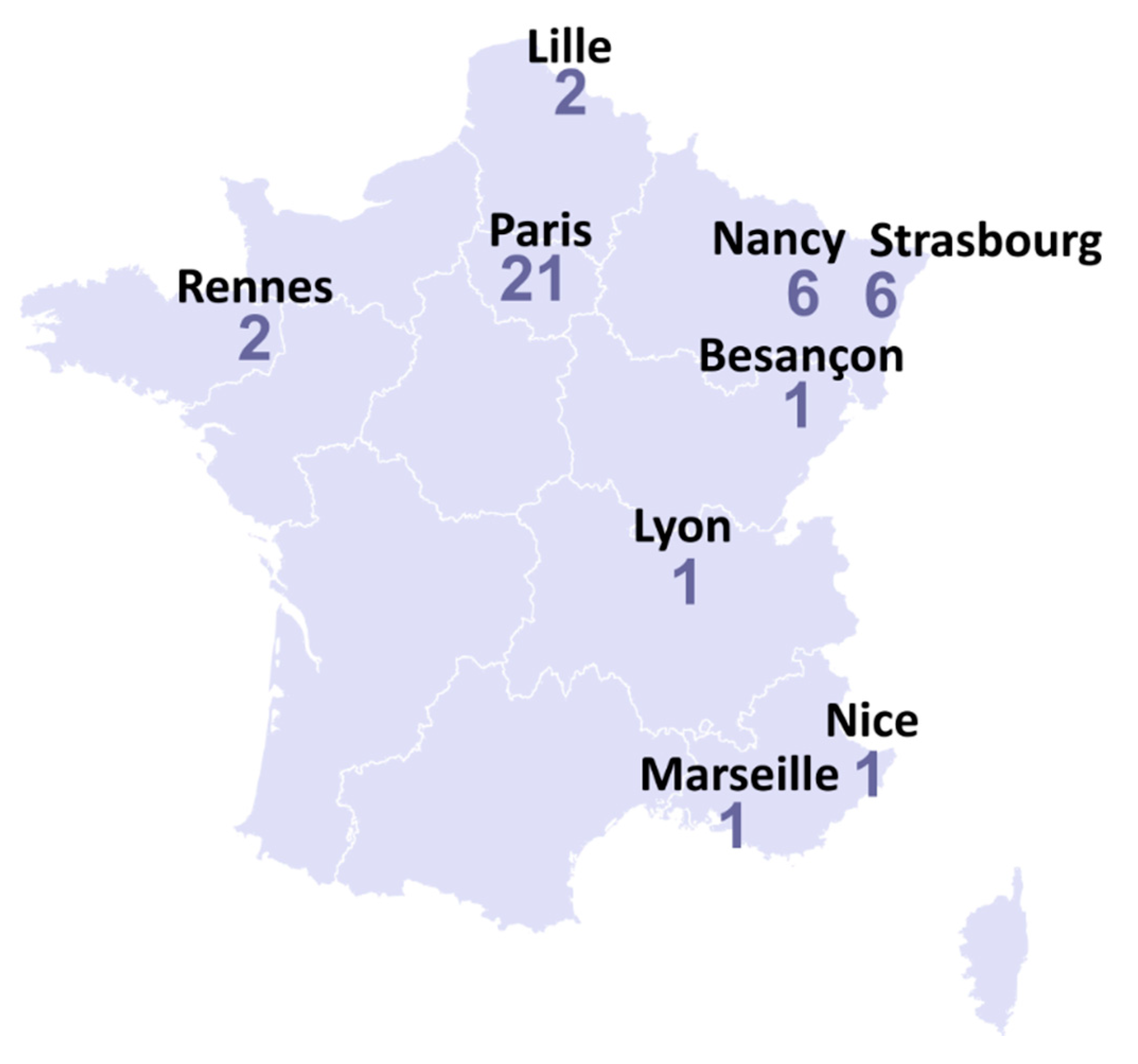
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jiatong, S.; Lanqin, L.; Wenjun, L. COVID-19 epidemic: Disease characteristics in children. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Assaad, S.; Avrillon, V.; Fournier, M.-L.; Mastroianni, B.; Russias, B.; Swalduz, A.; Cassier, P.; Eberst, L.; Steineur, M.-P.; Kazes, M.; et al. High mortality rate in cancer patients with symptoms of COVID-19 with or without detectable SARS-COV-2 on RT-PCR. Eur. J. Cancer 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhu, F.; Xie, L.; Wang, C.; Wang, J.; Chen, R.; Jia, P.; Guan, H.Q.; Peng, L.; Chen, Y.; et al. Clinical characteristics of COVID-19-infected cancer patients: A retrospective case study in three hospitals within Wuhan, China. Ann. Oncol. 2020, 31, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Boulad, F.; Kamboj, M.; Bouvier, N.; Mauguen, A.; Kung, A.L. COVID-19 in Children with Cancer in New York City. JAMA Oncol. 2020, 6, 1459–1460. [Google Scholar] [CrossRef] [PubMed]
- De Rojas, T.; Pérez-Martínez, A.; Cela, E.; Baragaño, M.; Galán, V.; Mata, C.; Peretó, A.; Madero, L. COVID-19 infection in children and adolescents with cancer in Madrid. Pediatr. Blood Cancer 2020, 67, e28397. [Google Scholar] [CrossRef] [PubMed]
- Terenziani, M.; Massimino, M.; Biassoni, V.; Casanova, M.; Chiaravalli, S.; Ferrari, A.; Luksch, R.; Meazza, C.; Podda, M.; Schiavello, E.; et al. SARS-CoV-2 disease and children under treatment for cancer. Pediatr. Blood Cancer 2020, 67, e28346. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.-Y.; Ramakrishna, S.; Long, A.H.; Phillips, C.A.; Montiel-Esparza, R.; Diorio, C.J.; Bailey, L.C.; Maude, S.L.; Aplenc, R.; Batra, V.; et al. Delayed cancer diagnoses and high mortality in children during the COVID-19 pandemic. Pediatr. Blood Cancer 2020, 67, e28427. [Google Scholar] [CrossRef] [PubMed]
- André, N. Covid-19: Breaking bad news with social distancing in pediatric oncology. Pediatr. Blood Cancer 2020, 67, e28524. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, M.; Bouffet, E.; Rodriguez-Galindo, C.; Luna-Fineman, S.; Khan, M.S.; Kearns, P.; Hawkins, D.S.; Challinor, J.; Morrissey, L.; Fuchs, J.; et al. The COVID-19 pandemic: A rapid global response for children with cancer from SIOP, COG, SIOP-E, SIOP-PODC, IPSO, PROS, CCI, and St Jude Global. Pediatr. Blood Cancer 2020, 67, e28409. [Google Scholar] [CrossRef] [PubMed]
- Johns Hopkins Coronavirus Resource Center. COVID-19 Map. Available online: https://coronavirus.jhu.edu/map.html (accessed on 14 April 2020).
- André, N.; Rouger-Gaudichon, J.; Brethon, B.; Phulpin, A.; Thébault, É.; Pertuisel, S.; Gandemer, V. COVID-19 in pediatric oncology from French pediatric oncology and hematology centers: High risk of severe forms? Pediatr. Blood Cancer 2020, 67, e28392. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.; Zecca, M.; Rizzari, C.; Porta, F.; Provenzi, M.; Marinoni, M.; Schumacher, R.F.; Luksch, R.; Terenziani, M.; Casanova, M.; et al. Children with cancer in the time of COVID-19: An 8-week report from the six pediatric onco-hematology centers in Lombardia, Italy. Pediatr. Blood Cancer 2020, 67, e28410. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.V.; Hu, Y. Poor clinical outcomes for patients with cancer during the COVID-19 pandemic. Lancet Oncol. 2020, 21, 862–864. [Google Scholar] [CrossRef]
- Liang, W.; Guan, W.; Chen, R.; Wang, W.; Li, J.; Xu, K.; Li, C.; Ai, Q.; Lu, W.; Liang, H.; et al. Cancer patients in SARS-CoV-2 infection: A nationwide analysis in China. Lancet Oncol. 2020, 21, 335–337. [Google Scholar] [CrossRef]
- Rouger-Gaudichon, J.; Gariazzo, L.; Thébault, E.; Brethon, B.; Fenwarth, L.; Gambart, M.; Alimi, A.; Réguerre, Y.; Piguet, C.; Jubert, C.; et al. Impact of COVID-19 on cancer care: A survey from the French Society of Pediatric Oncology (SFCE). Pediatr. Blood Cancer 2020, e28554. [Google Scholar] [CrossRef] [PubMed]
- Gampel, B.; Lucas, A.G.T.; Broglie, L.; Gartrell-Corrado, R.D.; Lee, M.T.; Levine, J.; Orjuela-Grimm, M.; Satwani, P.; Glade-Bender, J.; Roberts, S.S. COVID-19 disease in New York City pediatric hematology and oncology patients. Pediatr. Blood Cancer 2020, 67, e28420. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Mo, X.; Hu, Y.; Qi, X.; Jiang, F.; Jiang, Z.; Tong, S. Epidemiological Characteristics of 2143 Pediatric Patients with 2019 Coronavirus Disease in China. Pediatrics 2020, 58, 712–713. [Google Scholar] [CrossRef] [PubMed]
- Bisogno, G.; Provenzi, M.; Zama, D.; Tondo, A.; Meazza, C.; Colombini, A.; Galaverna, F.; Compagno, F.; Carraro, F.; De Santis, R.; et al. Clinical Characteristics and Outcome of Severe Acute Respiratory Syndrome Coronavirus 2 Infection in Italian Pediatric Oncology Patients: A Study from the Infectious Diseases Working Group of the Associazione Italiana di Oncologia e Ematologia Pediatrica. J. Pediatr. Infect. Dis. Soc. 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Suleyman, G.; Fadel, R.A.; Malette, K.M.; Hammond, C.; Abdulla, H.; Entz, A.; Demertzis, Z.; Hanna, Z.; Failla, A.; Dagher, C.; et al. Clinical Characteristics and Morbidity Associated with Coronavirus Disease 2019 in a Series of Patients in Metropolitan Detroit. JAMA Netw. Open 2020, 3, e2012270. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Number of Patients | Percentage |
|---|---|---|
| Sex | ||
| - Male | 18 | 49 |
| - Female | 19 | 51 |
| Age | ||
| - 0–2 years | 3 | 8 |
| - 2–6 years | 8 | 22 |
| - 6–12 years | 9 | 24 |
| - 12–18 years | 11 | 30 |
| - Above 18 years | 6 | 16 |
| Pathological condition | ||
| Hematological malignancy | 16 | 43 |
| - ALL | 10 | 27 |
| ○ t (9;22) negative | 9 | 24 |
| ○ t (9;22) positive | 1 | 3 |
| - AML | 1 | 3 |
| - CML | 1 | 3 |
| - NHL | 4 | 11 |
| Solid tumor | 17 | 46 |
| - Localized | 11 | 30 |
| - Metastatic | 6 | 16 |
| - CNS tumor | 7 | 19 |
| - Medulloblastoma | 2 | 5 |
| - CNS NB-FOXR2 | 1 | 3 |
| - Pinealoblastoma | 1 | 3 |
| - High-grade glioma | 1 | 3 |
| - Low-grade glioma | 1 | 3 |
| - Cerebral ATRT | 1 | 3 |
| - Bone tumor | 4 | 11 |
| - Osteosarcoma | 2 | 5 |
| - Ewing sarcoma | 2 | 5 |
| - MPNST | 1 | 3 |
| - Wilms’ tumor | 1 | 3 |
| - Neuroblastoma | 1 | 3 |
| - ATRT of the kidney | 1 | 3 |
| Non-oncologic condition | 4 | 11 |
| - Aplastic anemia | 1 | 3 |
| - Sickle cell disease | 1 | 3 |
| - EBV-induced macrophage activation syndrome | 1 | 3 |
| - Familial septic granulomatosis | 1 | 3 |
| COVID-19 diagnosis method | ||
| - Positive SARS-CoV-2 PCR | 34 | 92 |
| - Positive serology | 2 | 5 |
| - Clinical and radiological diagnosis | 1 | 3 |
| Symptoms | ||
| - Yes | 28 | 76 |
| - No | 9 | 24 |
| Treatments one month prior to COVID-19 diagnosis | ||
| - Chemotherapy | 24 | 65 |
| - Corticosteroids | 9 | 24 |
| - G-CSF | 7 | 19 |
| - Immunosupressive agents | 6 | 16 |
| - Targeted therapy or moncolonal antibody | 5 | 14 |
| - Radiotherapy | 2 | 5 |
| - Surgery | 2 | 5 |
| - None | 1 | 3 |
| Symptoms | Number of Patients | Percentage of Symptomatic Cases |
|---|---|---|
| Fever | 20 | 71% |
| >38.5 °C | 14 | 50% |
| Between 38 °C and 38.5 °C | 6 | 21% |
| Cough | 14 | 50% |
| Rhinorrhea | 12 | 43% |
| Asthenia | 12 | 43% |
| Loss of smell/taste | 8 | 29% |
| Diarrhea | 7 | 25% |
| Chest pain | 6 | 21% |
| Myalgia | 5 | 18% |
| Respiratory distress signs | 5 | 18% |
| Tachycardia | 4 | 14% |
| Headaches | 3 | 11% |
| Skin rash | 2 | 7% |
| Neurological signs | 2 | 7% |
| Age (Years) | Sex | Pathology | Time to ICU Admission (days) | Type of Respiratory Support | Specific Treatment Against SARS-CoV-2 | Evolution | ICU Stay (Days) | Biology | Comments |
|---|---|---|---|---|---|---|---|---|---|
| 12 | M | Relapsed B-ALL, HSCT 2 months prior SARS-CoV-2 infection, aGVHD | 1 | Non-invasive ventilation | No | Favorable | 5 | Neutro:3.2 G/L Ly: 0.07 CRP 39 mg/L | Developed cerebral toxoplasmosis after ICU stay |
| 5 | F | SS sickle cell disease with cerebral vasculopathy, HSCT 1 month prior to SARS-CoV-2 infection | 2 | Mechanic ventilation for 5 days | Remdesivir Tocilizumab (2 injections) | Favorable | 29 | Neutro:5.12 G/L Ly: 1.28 CRP <10 mg/L Ferritin 4400 µg/L | Neurological complications (bilateral facial palsy, progressive acute polyneuropathy) SARS-CoV-2 still detectable in stool at day 28 of infection |
| 8 | F | Relapsed high-grade astrocytoma | 10 | Non-invasive ventilation | No | Favorable | Unknown | WBC: 2.7 G/L Neutro: 0.5 G/L CRP max: 251 mg/L | Repeat PCR negative at day 8 |
| 18 | F | Relapsed B-ALL (2nd relapse), treated with vincristine only | 4 | Non-invasive ventilation | Hydroxychloroquine | Favorable | 10 | Neutro: 0.7 G/L Ly: 0.48 G/L CRP max: 267 mg/L | CAR-T cell treatment delayed because of SARS-CoV-2 infection |
| 4 | M | Relapsed B-ALL | 5 | Mechanic ventilation for 2 days | Hydroxychloroquine Tocilizumab (2 injections) | Deceased (19 days after COVID-19 diagnosis) | 14 | WBC: 0.12 G/L CRP <10 mg/L Ferritin > 300,000 µg/L | Severe macrophage activation syndrome |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rouger-Gaudichon, J.; Thébault, E.; Félix, A.; Phulpin, A.; Paillard, C.; Alimi, A.; Brethon, B.; Gouache, E.; Raimbault, S.; de Berranger, E.; et al. Impact of the First Wave of COVID-19 on Pediatric Oncology and Hematology: A Report from the French Society of Pediatric Oncology. Cancers 2020, 12, 3398. https://doi.org/10.3390/cancers12113398
Rouger-Gaudichon J, Thébault E, Félix A, Phulpin A, Paillard C, Alimi A, Brethon B, Gouache E, Raimbault S, de Berranger E, et al. Impact of the First Wave of COVID-19 on Pediatric Oncology and Hematology: A Report from the French Society of Pediatric Oncology. Cancers. 2020; 12(11):3398. https://doi.org/10.3390/cancers12113398
Chicago/Turabian StyleRouger-Gaudichon, Jérémie, Eric Thébault, Arthur Félix, Aurélie Phulpin, Catherine Paillard, Aurélia Alimi, Benoît Brethon, Elodie Gouache, Sandra Raimbault, Eva de Berranger, and et al. 2020. "Impact of the First Wave of COVID-19 on Pediatric Oncology and Hematology: A Report from the French Society of Pediatric Oncology" Cancers 12, no. 11: 3398. https://doi.org/10.3390/cancers12113398
APA StyleRouger-Gaudichon, J., Thébault, E., Félix, A., Phulpin, A., Paillard, C., Alimi, A., Brethon, B., Gouache, E., Raimbault, S., de Berranger, E., Poirée, M., Bouttefroy, S., André, N., Gandemer, V., & on behalf of Société Française de lutte contre les Cancers et leucémies de l’Enfant et de l’adolescent (SFCE). (2020). Impact of the First Wave of COVID-19 on Pediatric Oncology and Hematology: A Report from the French Society of Pediatric Oncology. Cancers, 12(11), 3398. https://doi.org/10.3390/cancers12113398





