SFCE-RAPIRI Phase I Study of Rapamycin Plus Irinotecan: A New Way to Target Intra-Tumor Hypoxia in Pediatric Refractory Cancers
Simple Summary
Abstract
1. Introduction
2. Results
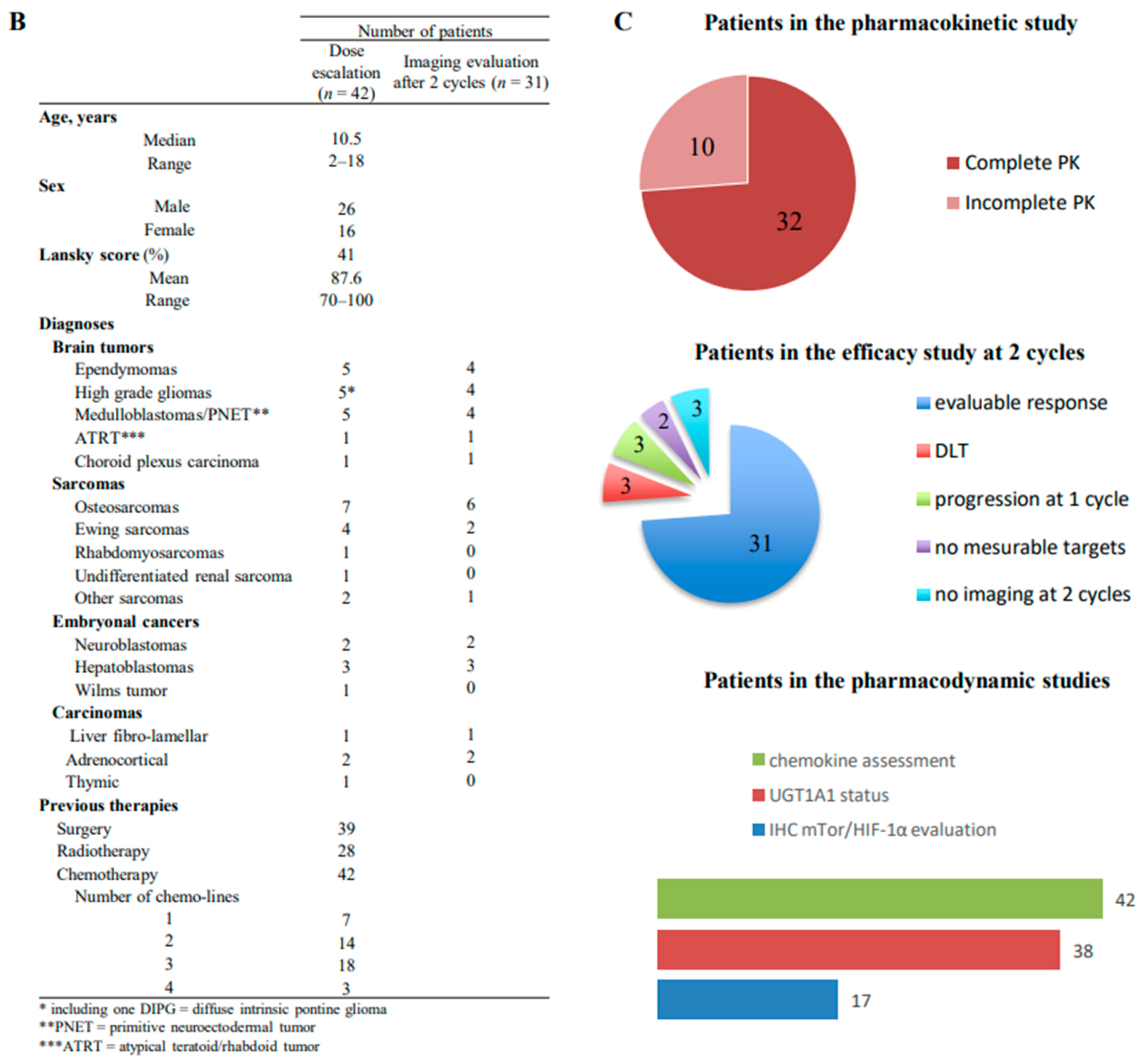
2.1. Patients’ Characteristics
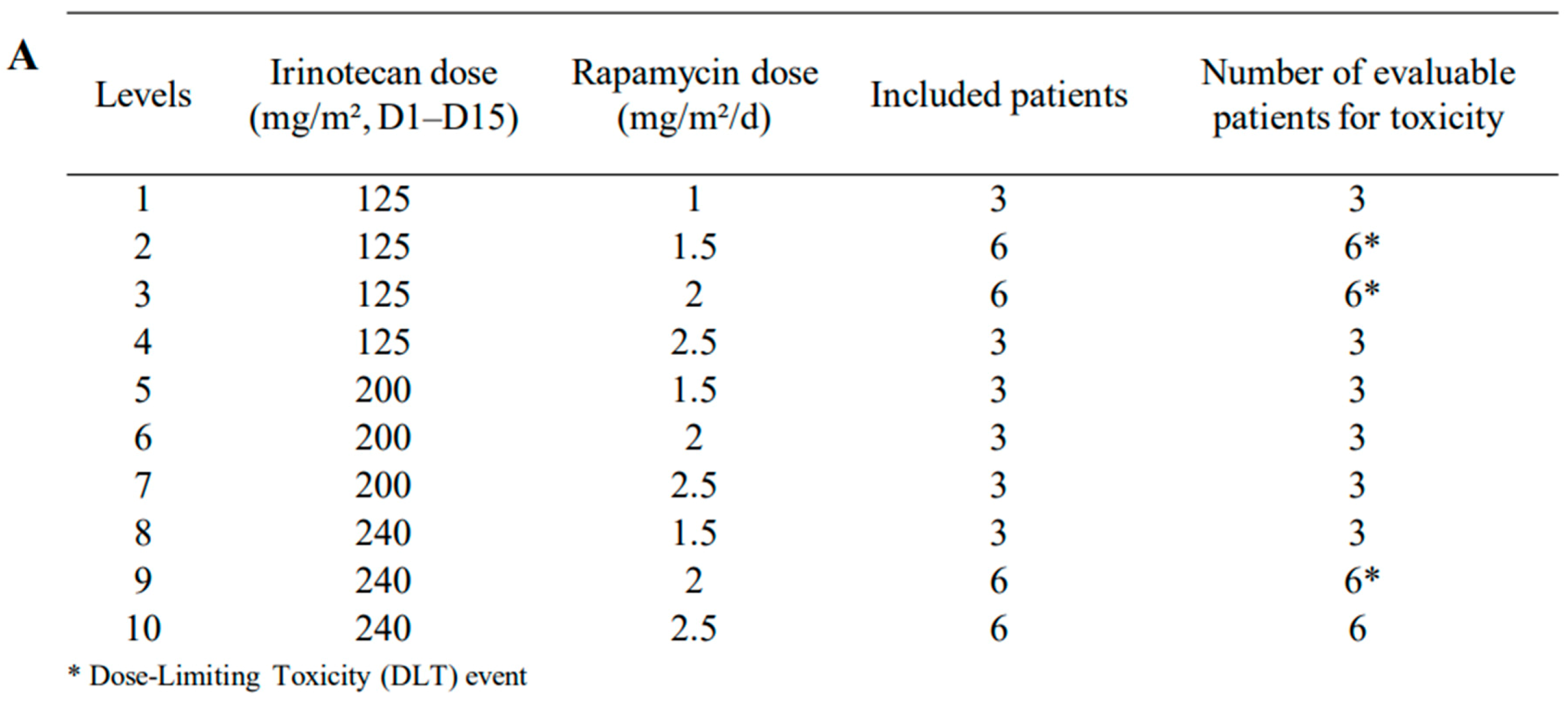
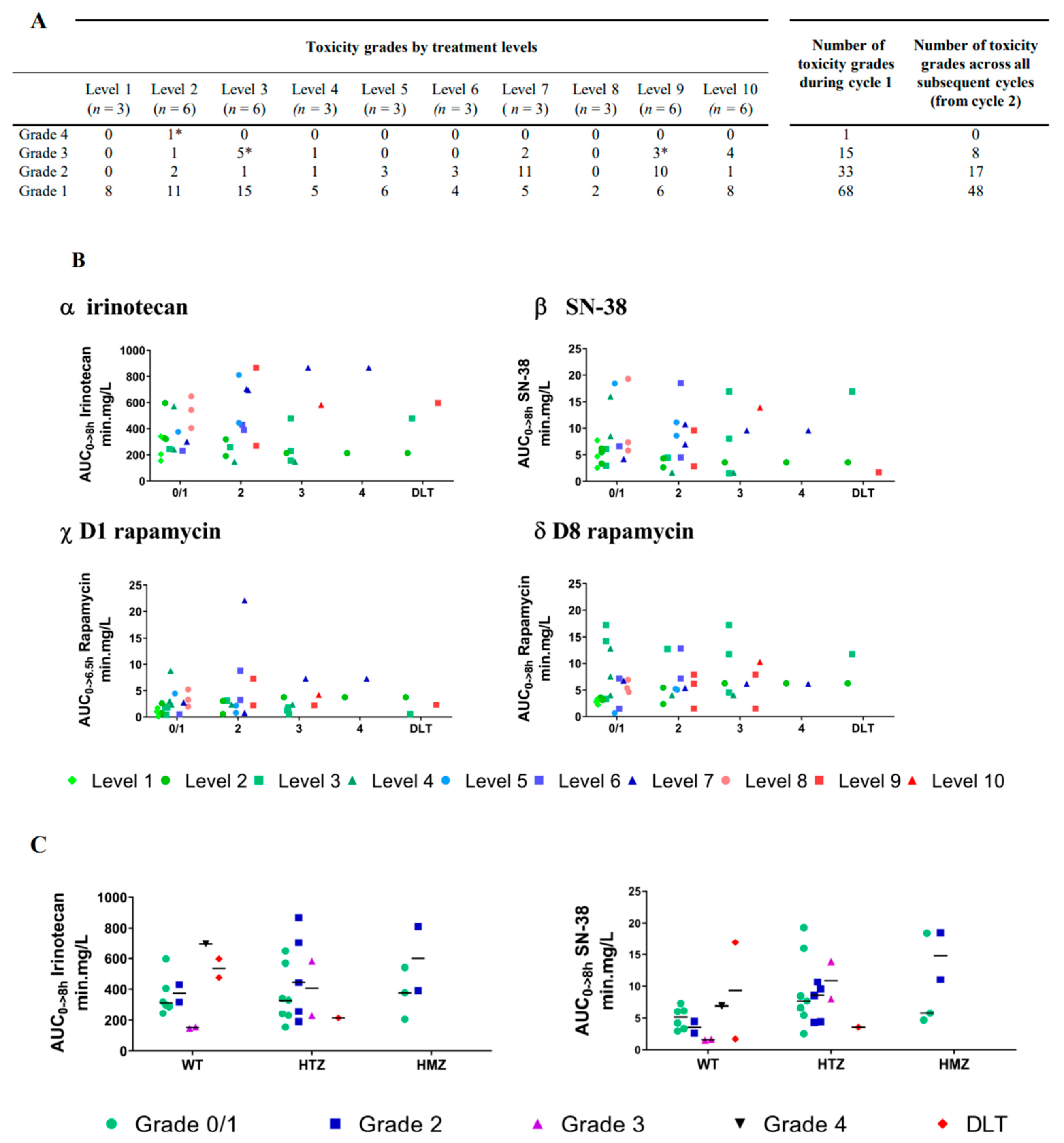
2.2. Dose Escalation and Toxicity
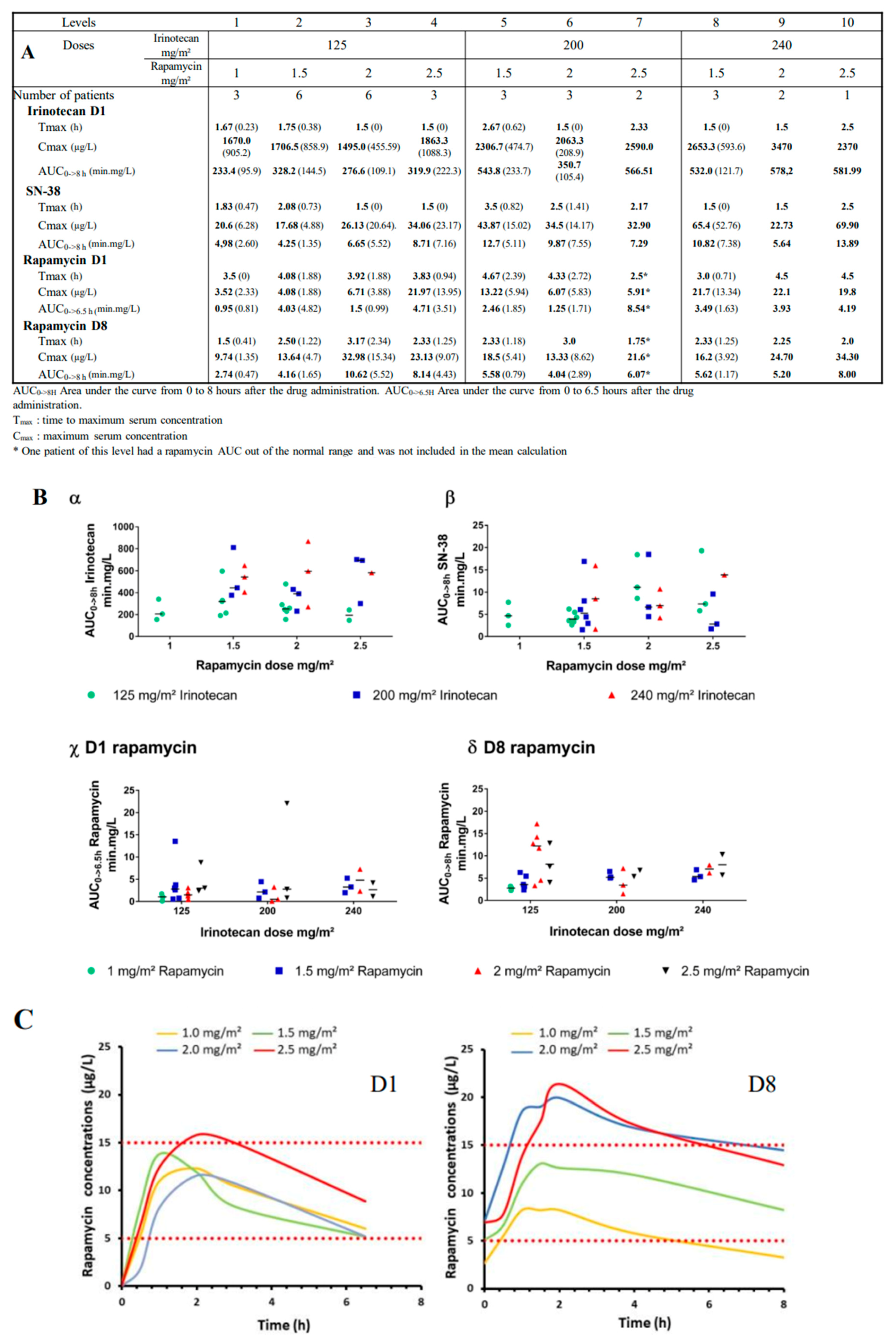
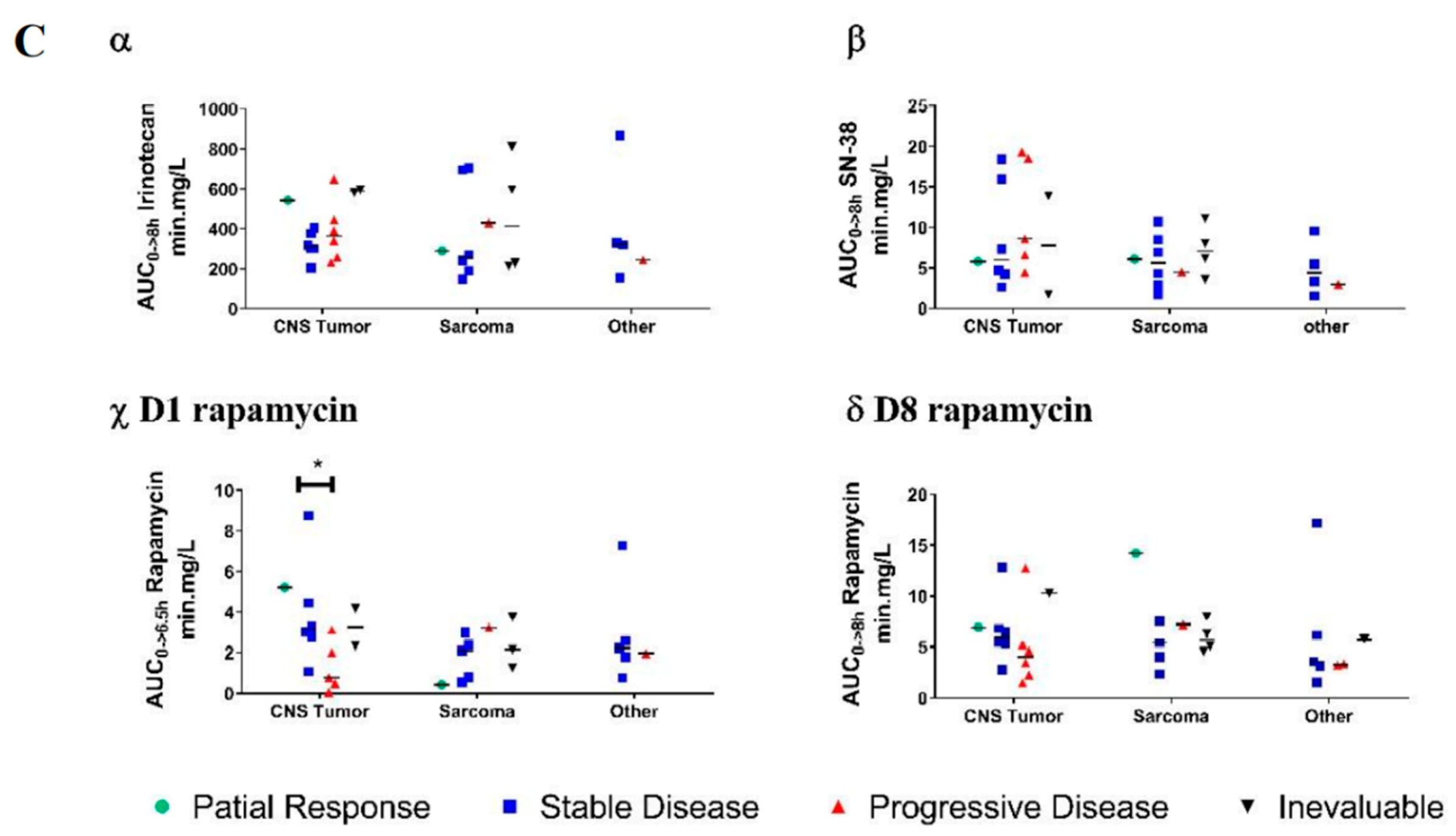
2.3. Pharmacokinetics (PK)
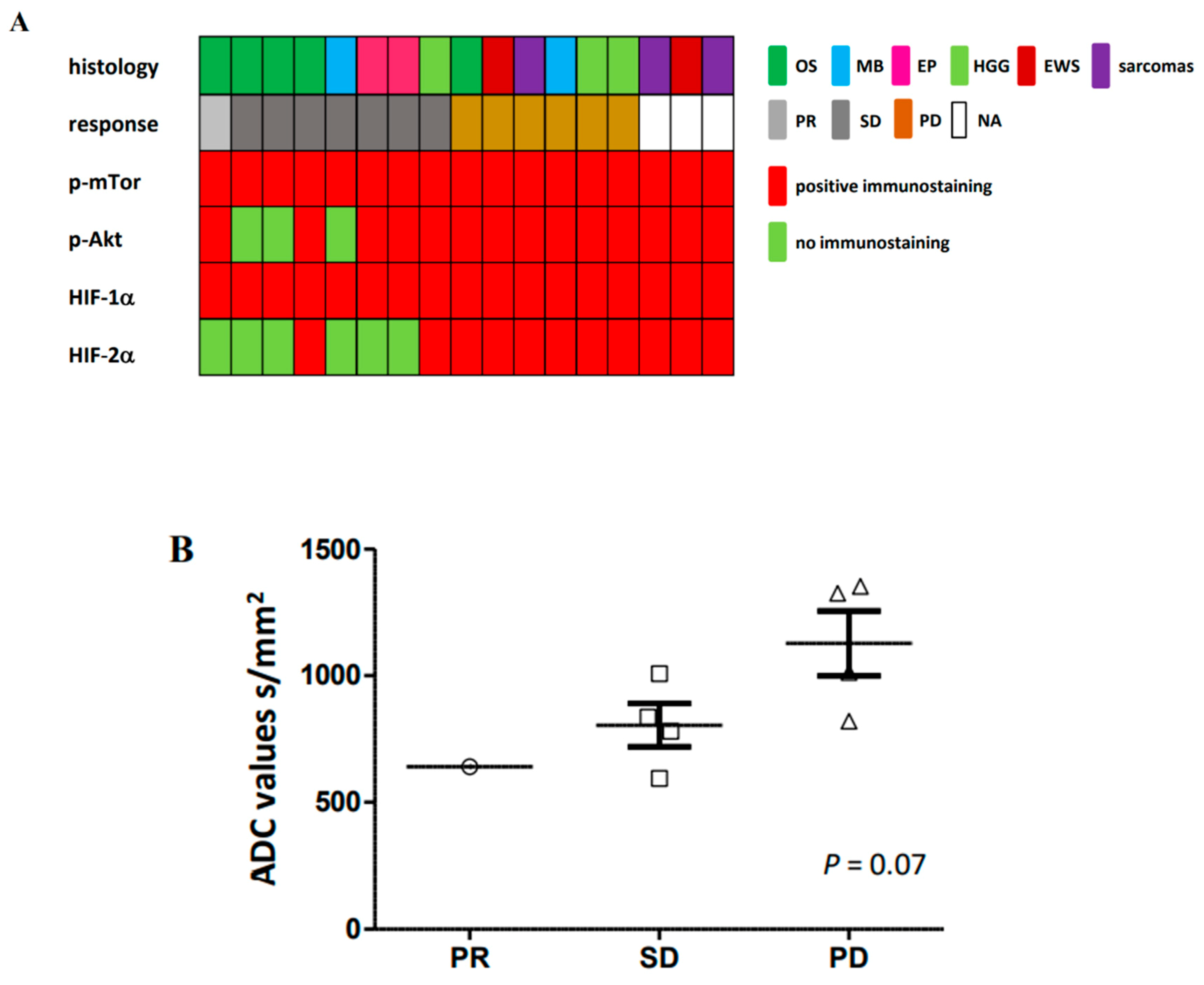
2.4. Anti-Tumor Effect, Links with PK, and Groups of Tumors
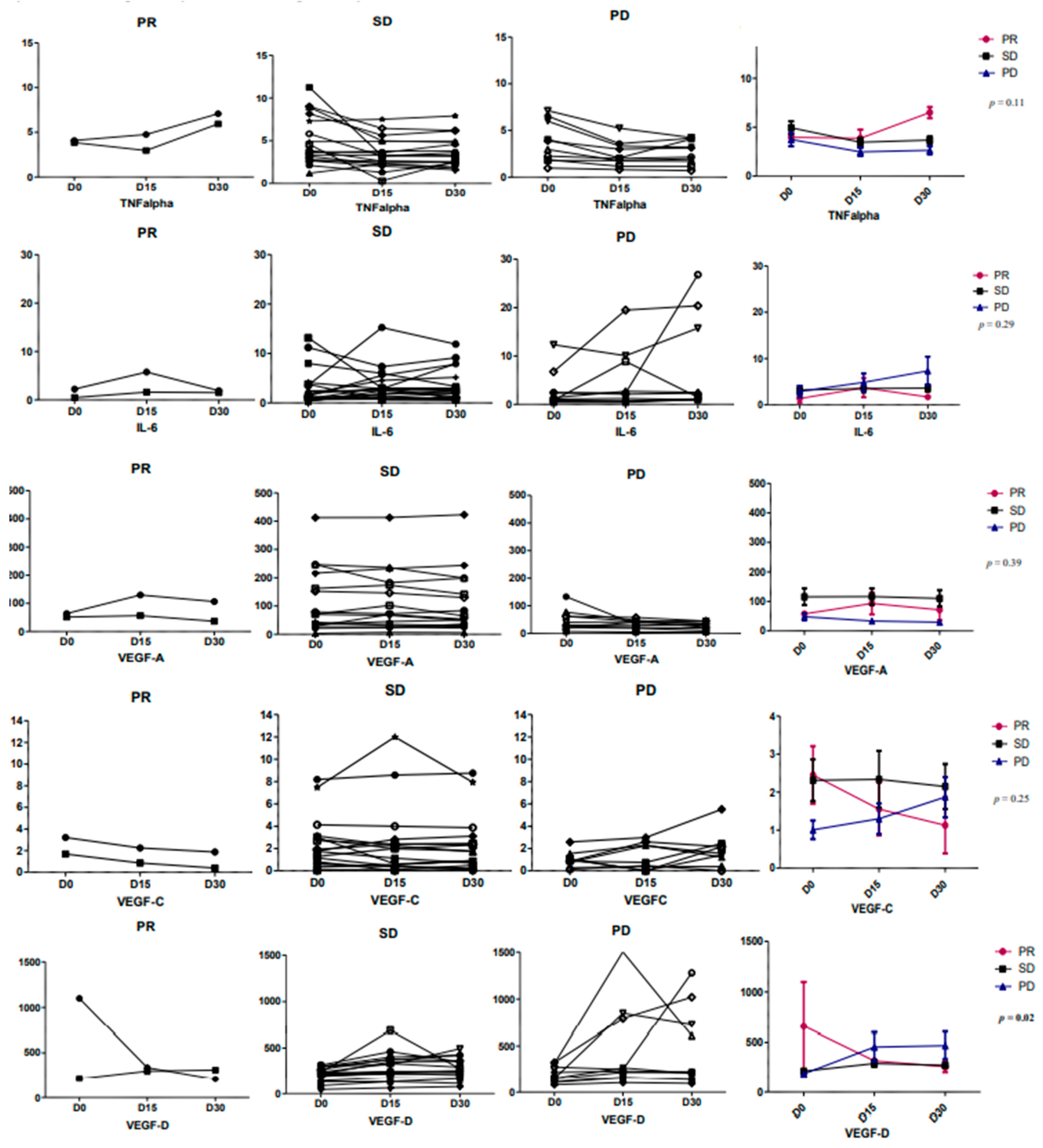
2.5. PDics Studies Were Able to Underline Biomarkers of Tumor Resistance to Hypoxia Targeting
3. Discussion
4. Materials and Methods
4.1. Patient Eligibility
4.2. Protocol Design and Drug Administration
4.3. Toxicity Assessment
4.4. Pharmacokinetics (PK)
4.5. Tumor Response
4.6. Pharmacodynamics (PDics)
4.6.1. UGT1A1 Genotyping
4.6.2. Immunohistochemistry (IHC) on FFPE Diagnostic Tumor Samples
4.6.3. Plasmatic Cytokine Measures
4.7. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Das, B.; Yeger, H.; Tsuchida, R.; Torkin, R.; Gee, M.F.; Thorner, P.S.; Shibuya, M.; Malkin, D.; Baruchel, S. A hypoxia-drivenvascular endothelial growth factor/Flt1 autocrine loop interacts with hypoxia-inducible factor-1alpha through mitogen-activated protein kinase/extracellular signal-regulated kinase 1/2 pathway in neuroblastoma. Cancer Res. 2005, 65, 7267–7275. [Google Scholar] [CrossRef] [PubMed]
- Mizobuchi, H.; García-Castellano, J.M.; Philip, S.; Healey, J.H.; Gorlick, R. Hypoxia markers in human osteosarcoma: An exploratory study. Clin. Orthop. 2008, 466, 2052–2059. [Google Scholar] [CrossRef] [PubMed]
- Hamidian, A.; von Stedingk, K.; Thorén, M.M.; Mohlin, S.; Påhlman, S. Differential regulation of HIF-1α and HIF-2α in neuroblastoma: Estrogen-related receptor alpha (ERRα) regulates HIF2A transcription and correlates to poor outcome. Biochem. Biophys. Res. Commun. 2015, 461, 560–567. [Google Scholar] [CrossRef]
- Nakamura, K.; Martin, K.C.; Jackson, J.K.; Beppu, K.; Woo, C.W.; Thiele, C.J. Brain-derived neurotrophic factor activation of TrkB induces vascular endothelial growth factor expression via hypoxia-inducible factor-1alpha in neuroblastoma cells. Cancer Res. 2006, 66, 4249–4255. [Google Scholar] [CrossRef]
- Preusser, M.; Wolfsberger, S.; Haberler, C.; Breitschopf, H.; Czech, T.; Slavc, I.; Harris, A.L.; Acker, T.; Budka, H.; Hainfellner, J.A. Vascularization and expression of hypoxia-related tissue factors in intracranial ependymoma and their impact on patient survival. Acta Neuropathol. (Berl.) 2005, 109, 211–216. [Google Scholar] [CrossRef]
- Gillespie, D.L.; Whang, K.; Ragel, B.T.; Flynn, J.R.; Kelly, D.A.; Jensen, R.L. Silencing of hypoxia inducible factor-1alpha by RNA interference attenuates human glioma cell growth in vivo. Clin. Cancer Res. 2007, 13, 2441–2448. [Google Scholar] [CrossRef]
- Påhlman, S.; Mohlin, S. Hypoxia and hypoxia-inducible factors in neuroblastoma. Cell Tissue Res. 2018, 372, 269–275. [Google Scholar] [CrossRef]
- Vander Heiden, M.G.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef]
- Cairns, R.A.; Harris, I.S.; Mak, T.W. Regulation of cancer cell metabolism. Nat. Rev. Cancer 2011, 11, 85–95. [Google Scholar] [CrossRef]
- Schito, L.; Semenza, G.L. Hypoxia-Inducible Factors: Master Regulators of Cancer Progression. Trends Cancer 2016, 2, 758–770. [Google Scholar] [CrossRef]
- Masson, N.; Ratcliffe, P.J. Hypoxia signaling pathways in cancer metabolism: The importance of co-selecting interconnected physiological pathways. Cancer Metab. 2014, 2, 3. [Google Scholar] [CrossRef]
- Land, S.C.; Tee, A.R. Hypoxia-inducible factor 1alpha is regulated by the mammalian targetof rapamycin (mTOR) via an mTOR signaling motif. J. Biol. Chem. 2007, 282, 20534–20543. [Google Scholar] [CrossRef] [PubMed]
- Bjornsti, M.-A.; Houghton, P.J. The TOR pathway: A target for cancer therapy. Nat. Rev. Cancer 2004, 4, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Bagatell, R.; Norris, R.; Ingle, A.M.; Ahern, C.; Voss, S.; Fox, E.; Little, A.R.; Weigel, B.J.; Adamson, P.C.; Blaney, S. Phase 1 trial of temsirolimus in combination with irinotecan and temozolomide in children, adolescents and young adults with relapsed or refractory solid tumors: A Children’s Oncology Group Study. Pediatr. Blood Cancer 2014, 61, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Houghton, P.J.; Morton, C.L.; Kolb, E.A.; Gorlick, R.; Lock, R.; Carol, H.; Reynolds, C.P.; Maris, J.M.; Keir, S.T.; Billups, C.A.; et al. Initial testing (stage 1) of the mTOR inhibitor rapamycin by the pediatric preclinical testing program. Pediatr. Blood Cancer 2008, 50, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Fouladi, M.; Laningham, F.; Wu, J.; O’Shaughnessy, M.A.; Molina, K.; Broniscer, A.; Spunt, S.L.; Luckett, I.; Stewart, C.F.; Houghton, P.J.; et al. Phase I study of everolimus in pediatric patients with refractory solid tumors. J. Clin Oncol 2007, 25, 4806–4812. [Google Scholar] [CrossRef]
- Geoerger, B.; Kieran, M.W.; Grupp, S.; Perek, D.; Clancy, J.; Krygowski, M.; Ananthakrishnan, R.; Boni, J.P.; Berkenblit, A.; Spunt, S.L. Phase II trial of temsirolimus in children with high-grade glioma, neuroblastoma and rhabdomyosarcoma. Eur. J. Cancer 2012, 48, 253–262. [Google Scholar] [CrossRef]
- Gore, L.; Trippett, T.M.; Katzenstein, H.M.; Boklan, J.; Narendran, A.; Smith, A.; Macy, M.E.; Rolla, K.; Narashimhan, N.; Squillace, R.M.; et al. A multicenter, first-in-pediatrics, phase 1, pharmacokinetic and pharmacodynamic study of ridaforolimus in patients with refractory solid tumors. Clin. Cancer Res. 2013, 19, 3649–3658. [Google Scholar] [CrossRef]
- Spunt, S.L.; Grupp, S.A.; Vik, T.A.; Santana, V.M.; Greenblatt, D.J.; Clancy, J.; Berkenblit, A.; Krygowski, M.; Ananthakrishnan, R.; Boni, J.P.; et al. Phase I study of temsirolimus in pediatric patients with recurrent/refractory solid tumors. J. Clin. Oncol. 2011, 29, 2933–2940. [Google Scholar] [CrossRef]
- Wagner, L.M.; Fouladi, M.; Ahmed, A.; Krailo, M.D.; Weigel, B.; DuBois, S.G.; Doyle, L.A.; Chen, H.; Blaney, S.M. Phase II study of cixutumumab in combination with temsirolimus in pediatric patients and young adults with recurrent or refractory sarcoma: A report from the Children’s Oncology Group. Pediatr. Blood Cancer 2015, 62, 440–444. [Google Scholar] [CrossRef]
- Deyell, R.J.; Wu, B.; Rassekh, S.R.; Tu, D.; Samson, Y.; Fleming, A.; Bouffet, E.; Sun, X.; Powers, J.; Seymour, L.; et al. Phase I study of vinblastine and temsirolimus in pediatric patients with recurrent or refractory solid tumors: Canadian Cancer Trials Group Study IND.218. Pediatr. Blood Cancer 2019, 66, e27540. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.Q.; Yang, Q.K.; Lu, B.W.; Yi, W.; Cantin, G.; Chen, Y.L.; Fearns, C.; Yates, J.R.; Lee, J.D. CDC25B mediates rapamycin-induced oncogenic responses in cancer cells. Cancer Res. 2009, 69, 2663–2668. [Google Scholar] [CrossRef] [PubMed]
- Zitzmann, K.; von Rüden, J.; Brand, S.; Göke, B.; Lichtl, J.; Spöttl, G.; Auernhammer, C.J. Compensatory activation of Akt in response to mTOR and Raf inhibitors—A rationale for dual-targeted therapy approaches in neuroendocrine tumor disease. Cancer Lett. 2010, 295, 100–199. [Google Scholar] [CrossRef]
- Butler, D.E.; Marlein, C.; Walker, H.F.; Frame, F.M.; Mann, V.M.; Simms, M.S.; Davies, B.R.; Collins, A.T.; Maitland, N.J. Inhibition of the PI3K/AKT/mTOR pathway activates autophagy and compensatory Ras/Raf/MEK/ERK signalling in prostate cancer. Oncotarget 2017, 8, 56698–56713. [Google Scholar] [CrossRef]
- Nguyen, A.; Moussallieh, F.M.; Mackay, A.; Cicek, A.E.; Coca, A.; Chenard, M.P.; Weingertner, N.; Lhermitte, B.; Letouzé, E.; Guérin, E.; et al. Characterization of the transcriptional and metabolic responses of pediatric high-grade gliomas to mTOR-HIF-1α axis inhibition. Oncotarget 2017, 8, 71597–71617. [Google Scholar] [CrossRef]
- Guérin, E.; Raffelsberger, W.; Pencreach, E.; Maier, A.; Neuville, A.; Schneider, A.; Bachellier, P.; Rohr, S.; Petitprez, A.; Poch, O.; et al. In vivo topoisomerase I inhibition attenuates the expression of hypoxia-inducible factor 1α target genes and decreases tumor angiogenesis. Mol. Med. 2012, 18, 83–94. [Google Scholar] [CrossRef]
- Pencreach, E.; Guérin, E.; Nicolet, C.; Lelong-Rebel, I.; Voegeli, A.C.; Oudet, P.; Larsen, A.K.; Gaub, M.P.; Guenot, D. Marked activity of irinotecan and rapamycin combination toward colon cancer cells in vivo and in vitro is mediated through cooperative modulation of the mammalian target of rapamycin/hypoxia-inducible factor-1alpha axis. Clin. Cancer Res. 2009, 15, 1297–1307. [Google Scholar] [CrossRef]
- Vredenburgh, J.J.; Desjardins, A.; Herndon, J.E.; Marcello, J.; Reardon, D.A.; Quinn, J.A.; Rich, J.N.; Sathornsumetee, S.; Gururangan, S.; Sampson, J.; et al. Bevacizumab plus irinotecan in recurrent glioblastoma multiforme. J. Clin. Oncol. 2007, 25, 4722–4729. [Google Scholar] [CrossRef]
- Iyer, L.; Hall, D.; Das, S.; Mortell, M.A.; Ramírez, J.; Kim, S.; Di Rienzo, A.; Ratain, M.J. Phenotype-genotype correlation of in vitro SN-38 (active metabolite of irinotecan) and bilirubin glucuronidation in human liver tissue with UGT1A1 promoter polymorphism. Clin. Pharmacol. Ther. 1999, 65, 576–582. [Google Scholar] [CrossRef]
- Femke, M.; Goey, A.K.; van Schaik, R.H.; Mathijssen, R.H.; Bins, S. Individualization of Irinotecan Treatment: A Review of Pharmacokinetics, Pharmacodynamics, and Pharmacogenetics. Clin. Pharmacokinet. 2018, 57, 1229–1254. [Google Scholar]
- Thompson, P.A.; Gupta, M.; Rosner, G.L.; Yu, A.; Barrett, J.; Bomgaars, L.; Bernstein, M.L.; Blaney, S.M.; Mondick, J. Pharmacokinetics of irinotecan and its metabolites in pediatric cancer patients: A report from the children’s oncology group. Cancer Chemother. Pharmacol. 2008, 62, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Barajas, R.F., Jr.; Phillips, J.J.; Vandenberg, S.R.; McDermott, M.W.; Berger, M.S.; Dillon, W.P.; Cha, S. Pro-angiogenic cellular and genomic expression patterns within glioblastoma influences dynamic susceptibility weighted perfusion MRI. Radiol. 2015, 70, 1087–1095. [Google Scholar] [CrossRef] [PubMed]
- Gadda, D.; Mazzoni, L.N.; Pasquini, L.; Busoni, S.; Simonelli, P.; Giordano, G.P. Relationship between Apparent Diffusion Coefficients and MR Spectroscopy Findings in High-Grade Gliomas. J. Neuroimag. 2017, 27, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Entz-Werle, N.; Chammas, A.; Ruhland, E.; Sebastia, C.; Gabor, F.; Durand, A.; Guerin, E.; Kremer, S.; Namer, I.J.; Dontenwill, M. Integrative analyses of MRI parameters, metabolomics and hypoxic biomarkers are showing predictive correlation with patient characteristics and outcome. Neuro-Oncology 2018, 20, i95. [Google Scholar] [CrossRef]
- Swartz, J.E.; Driessen, J.P.; van Kempen, P.M.; de Bree, R.; Janssen, L.M.; Pameijer, F.A.; Terhaard, C.H.; Philippens, M.E.; Willems, S. Influence of tumor and microenvironment characteristics on diffusion-weighted imaging in oropharyngeal carcinoma: A pilot study. Oral Oncol. 2018, 77, 9–15. [Google Scholar] [CrossRef]
- Sathyakumar, K.; Mani, S.; Pathak, G.H.; Prabhu, K.; Chacko, A.G.; Chacko, G. Neuroimaging of pediatric infratentorial tumors and the value of diffusion-weighted imaging (DWI) in determining tumor grade. Acta Radiol. 2020. [Google Scholar] [CrossRef]
- Ueda, S.; Saeki, T.; Osaki, A.; Yamane, T.; Kuji, I. Bevacizumab Induces Acute Hypoxia and Cancer Progression in Patients with Refractory Breast Cancer: Multimodal Functional Imaging and Multiplex Cytokine Analysis. Clin. Cancer Res. 2017, 23, 5769–5778. [Google Scholar] [CrossRef]
- Boige, V.; Malka, D.; Bourredjem, A.; Dromain, C.; Baey, C.; Jacques, N.; Pignon, J.P.; Vimond, N.; Bouvet-Forteau, N.; De Baere, T.; et al. Efficacy, safety, and biomarkers of single-agent bevacizumab therapy in patients with advanced hepatocellular carcinoma. Oncologist 2012, 17, 1063–1072. [Google Scholar] [CrossRef]
- Byers, L.A.; Holsinger, F.C.; Kies, M.S.; William, W.N.; El-Naggar, A.K.; Lee, J.J.; Hu, J.; Lopez, A.; Tran, H.T.; Yan, S.; et al. Serum signature of hypoxia-regulated factors is associated with progression after induction therapy in head and neck squamous cell cancer. Mol. Cancer Ther. 2010, 9, 1755–1763. [Google Scholar] [CrossRef]
- Bomgaars, L.; Kerr, J.; Berg, S.; Kuttesch, J.; Klenke, R.; Blaney, S.M. A phase I study of irinotecan administered on a weekly schedule in pediatric patients. Pediatr. Blood Cancer 2006, 46, 50–55. [Google Scholar] [CrossRef]
- Takano, S.; Kamiyama, H.; Mashiko, R.; Osuka, S.; Ishikawa, E.; Matsumura, A. Metronomic treatment of malignant glioma xenografts with irinotecan (CPT-11) inhibits angiogenesis and tumor growth. J. Neurooncol. 2010, 99, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Ghiringhelli, F.; Guiu, B.; Chauffert, B.; Ladoire, S. bevacizumab, 5-Fluorouracil and irinotecan for advanced colorectal cancer: A pilot study. World J. Gastroenterol. 2009, 15, 4278–4283. [Google Scholar] [CrossRef] [PubMed]
- Mody, R.; Naranjo, A.; Van Ryn, C.; Alice, L.Y.; London, W.B.; Shulkin, B.L.; Parisi, M.T.; Diccianni, M.B.; Sondel, P.M.; Bender, J.G.; et al. Phase I Irinotecan-temozolomide with temsirolimus or dinutuximab in children with refractory or relapsed neuroblastoma (COG ANBL1221): An open-label, randomised, phase 2 trial. Lancet Oncol. 2017, 18, 946–957. [Google Scholar] [CrossRef]
- Ando, Y.; Saka, H.; Ando, M.; Sawa, T.; Muro, K.; Ueoka, H.; Yokoyama, A.; Saitoh, S.; Shimokata, K.; Hasegawa, Y. Polymorphisms of UDP-glucuronosyltransferase gene and irinotecan toxicity: A pharmacogenetic analysis. Cancer Res. 2000, 60, 6921–6926. [Google Scholar] [PubMed]
- Innocenti, F.; Undevia, S.D.; Iyer, L.; Xian Chen, P.; Das, S.; Kocherginsky, M.; Karrison, T.; Janisch, L.; Ramírez, J.; Rudin, C.M.; et al. Genetic variants in the UDP-glucuronosyltransferase 1A1 gene predict the risk of severe neutropenia of irinotecan. J. Clin. Oncol. 2004, 22, 1382–1388. [Google Scholar] [CrossRef]
- Tang, S.C.; Sparidans, R.W.; Cheung, K.L.; Fukami, T.; Durmus, S.; Wagenaar, E.; Yokoi, T.; van Vlijmen, B.J.; Beijnen, J.H.; Schinkel, A.H. P-glycoprotein, CYP3A, and plasma carboxylesterase determine brain and blood disposition of the mTOR Inhibitor everolimus (Afinitor) in mice. Clin. Cancer Res. 2014, 20, 3133–3145. [Google Scholar] [CrossRef]
- Filipski, E.; Berland, E.; Ozturk, N.; Guettier, C.; van der Horst, G.T.; Lévi, F.; Okyar, A. Optimization of irinotecan chronotherapy with P-glycoprotein inhibition. Toxicol. Appl. Pharmacol. 2014, 274, 471–479. [Google Scholar] [CrossRef]
- Berg, A.K.; Buckner, J.C.; Galanis, E.; Jaeckle, K.A.; Ames, M.M.; Reid, J.M. Quantification of the impact of enzyme-inducing antiepileptic drugs on irinotecan pharmacokinetics and SN-38 exposure. J. Clin Pharmacol. 2015, 55, 1303–1312. [Google Scholar] [CrossRef]
- Ki, D.H.; Oppel, F.; Durbin, A.D.; Look, A.T. Mechanisms underlying synergy between DNA topoisomerase I-targeted drugs and mTOR kinase inhibitors in NF1-associated malignant peripheral nerve sheath tumors. Oncogene 2019, 38, 6585–6598. [Google Scholar] [CrossRef]
- McGregor, L.M.; Stewart, C.F.; Crews, K.R.; Tagen, M.; Wozniak, A.; Wu, J.; McCarville, M.B.; Navid, F.; Santana, V.M.; Houghton, P.J.; et al. Dose escalation of intravenous irinotecan using oral cefpodoxime: A phase I study in pediatric patients with refractory solid tumors. Pediatr. Blood Cancer 2012, 58, 372–379. [Google Scholar] [CrossRef]
- McMillan, K.M.; Rogers, B.P.; Field, A.S.; Laird, A.R.; Fine, J.P.; Meyerand, M.E. Physiologic characterisation of glioblastoma multiforme using MRI-based hypoxia mapping, chemical shift imaging, perfusion and diffusion maps. J. Clin. Neurosci. 2006, 13, 811–817. [Google Scholar] [CrossRef]
- Lin, N.U.; Lee, E.Q.; Aoyama, H.; Barani, I.J.; Barboriak, D.P.; Baumert, B.G.; Bendszus, M.; Brown, P.D.; Camidge, D.R.; Chang, S.M.; et al. Response Assessment in Neuro-Oncology (RANO) group. Response assessment criteria for brain metastases: Proposal from the RANO group. Lancet Oncol. 2015, 16, e270–e278. [Google Scholar] [CrossRef]
- Entz-Werle, N.; Schneider, A.; Kalifa, C.; Voegeli, A.C.; Tabone, M.D.; Marec-Berard, P.; Marcellin, L.; Pacquement, H.; Terrier, P.; Boutard, P.; et al. Genetic alterations in primary osteosarcoma from 54 children and adolescents by targeted allelotyping. Br. J. Cancer 2003, 88, 1925–1931. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jannier, S.; Kemmel, V.; Sebastia Sancho, C.; Chammas, A.; Sabo, A.-N.; Pencreach, E.; Farace, F.; Chenard, M.P.; Lhermitte, B.; Geoerger, B.; et al. SFCE-RAPIRI Phase I Study of Rapamycin Plus Irinotecan: A New Way to Target Intra-Tumor Hypoxia in Pediatric Refractory Cancers. Cancers 2020, 12, 3051. https://doi.org/10.3390/cancers12103051
Jannier S, Kemmel V, Sebastia Sancho C, Chammas A, Sabo A-N, Pencreach E, Farace F, Chenard MP, Lhermitte B, Geoerger B, et al. SFCE-RAPIRI Phase I Study of Rapamycin Plus Irinotecan: A New Way to Target Intra-Tumor Hypoxia in Pediatric Refractory Cancers. Cancers. 2020; 12(10):3051. https://doi.org/10.3390/cancers12103051
Chicago/Turabian StyleJannier, Sarah, Véronique Kemmel, Consuelo Sebastia Sancho, Agathe Chammas, Amelia-Naomie Sabo, Erwan Pencreach, Françoise Farace, Marie Pierre Chenard, Benoit Lhermitte, Birgit Geoerger, and et al. 2020. "SFCE-RAPIRI Phase I Study of Rapamycin Plus Irinotecan: A New Way to Target Intra-Tumor Hypoxia in Pediatric Refractory Cancers" Cancers 12, no. 10: 3051. https://doi.org/10.3390/cancers12103051
APA StyleJannier, S., Kemmel, V., Sebastia Sancho, C., Chammas, A., Sabo, A.-N., Pencreach, E., Farace, F., Chenard, M. P., Lhermitte, B., Geoerger, B., Aerts, I., Frappaz, D., Leblond, P., André, N., Ducassou, S., Corradini, N., Bertozzi, A. I., Guérin, E., Vincent, F., ... Entz-Werle, N. (2020). SFCE-RAPIRI Phase I Study of Rapamycin Plus Irinotecan: A New Way to Target Intra-Tumor Hypoxia in Pediatric Refractory Cancers. Cancers, 12(10), 3051. https://doi.org/10.3390/cancers12103051






