Progress in Clinical Trials of Photodynamic Therapy for Solid Tumors and the Role of Nanomedicine
Simple Summary
Abstract
1. Introduction
2. Photodynamic Therapy Mechanism and Advantages
3. Clinical Application of PDT for Solid Tumors
3.1. Brain Tumor
3.2. Lung Cancer
3.3. Urological Tumors: Bladder Cancer
3.4. Gastroenterological Cancer
4. Photoimmunotherapy for Solid Cancer
5. Nanotechnology in PDT
5.1. Advantages of Nanocarriers for PDT
5.2. Innovative Strategies of Using Nanocarriers in PDT
6. Photothermal Therapy (PTT)
7. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Yakavets, I.; Millard, M.; Zorin, V.; Lassalle, H.-P.; Bezdetnaya, L. Current state of the nanoscale delivery systems for temoporfin-based photodynamic therapy: Advanced delivery strategies. J. Control. Release 2019, 304, 268–287. [Google Scholar]
- Lucky, S.S.; Soo, K.C.; Zhang, Y. Nanoparticles in photodynamic therapy. Chem. Rev. 2015, 115, 1990–2042. [Google Scholar]
- Sztandera, K.; Gorzkiewicz, M.; Klajnert-Maculewicz, B. Nanocarriers in photodynamic therapy—in vitro and in vivo studies. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1509. [Google Scholar]
- Phua, S.Z.F.; Xue, C.; Lim, W.Q.; Yang, G.; Chen, H.; Zhang, Y.; Wijaya, C.F.; Luo, Z.; Zhao, Y. Light-responsive prodrug-based supramolecular nanosystems for site-specific combination therapy of cancer. Chem. Mater. 2019, 31, 3349–3358. [Google Scholar]
- Wu, H.; Minamide, T.; Yano, T. Role of photodynamic therapy in the treatment of esophageal cancer. Dig. Endosc. 2019, 31, 508–516. [Google Scholar]
- Nackiewicz, J.; Kliber-Jasik, M.; Skonieczna, M. A novel pro-apoptotic role of zinc octacarboxyphthalocyanine in melanoma me45 cancer cell’s photodynamic therapy (PDT). J. Photochem. Photobiol. B Biol. 2019, 190, 146–153. [Google Scholar]
- Aniogo, E.C.; George, B.P.A.; Abrahamse, H. The role of photodynamic therapy on multidrug resistant breast cancer. Cancer cell Int. 2019, 19, 1–14. [Google Scholar]
- Oda, D.; Duarte, M.; Andrade, F.; Moriyama, L.; Bagnato, V.; de Moraes, I. Antimicrobial action of photodynamic therapy in root canals using LED curing light, curcumin and carbopol gel. Int End. J. 2019, 52, 1010–1019. [Google Scholar]
- Spring, B.Q.; Rizvi, I.; Xu, N.; Hasan, T. The role of photodynamic therapy in overcoming cancer drug resistance. Photochem. Photobiol. Sci. 2015, 14, 1476–1491. [Google Scholar]
- Hu, S.; Yang, Y.; Jiang, B.; Su, D.; Zhang, L.; Huang, Z.; Zhang, F. Treatment of condyloma acuminatum using the combination of laser ablation and ALA-PDT. Photodiagnosis Photodyn. Ther. 2019, 25, 193–196. [Google Scholar]
- Van Straten, D.; Mashayekhi, V.; De Bruijn, H.S.; Oliveira, S.; Robinson, D.J. Oncologic photodynamic therapy: Basic principles, current clinical status and future directions. Cancers 2017, 9, 19. [Google Scholar]
- Zhang, Y.; Wang, B.; Zhao, R.; Zhang, Q.; Kong, X. Multifunctional nanoparticles as photosensitizer delivery carriers for enhanced photodynamic cancer therapy. Mater. Sci. Eng. C 2020, 115, 111099. [Google Scholar]
- Li, W.T. Nanoparticles for photodynamic therapy. Handb. Biophotonics 2013, 321–336. [Google Scholar] [CrossRef]
- Obaid, G.; Broekgaarden, M.; Bulin, A.-L.; Huang, H.-C.; Kuriakose, J.; Liu, J.; Hasan, T. Photonanomedicine: A convergence of photodynamic therapy and nanotechnology. Nanoscale 2016, 8, 12471–12503. [Google Scholar]
- Dalgaty, F.J. Principles underpinning the treatment of cancer with drugs. Med. J. 2013, 2, 47–52. [Google Scholar]
- Huang, Y.-Y.; Sharma, S.K.; Dai, T.; Chung, H.; Yaroslavsky, A.; Garcia-Diaz, M.; Chang, J.; Chiang, L.Y.; Hamblin, M.R. Can nanotechnology potentiate photodynamic therapy? Nanotechnol. Rev. 2012, 1, 111–146. [Google Scholar] [CrossRef]
- Konan, Y.N.; Cerny, R.; Favet, J.; Berton, M.; Gurny, R.; Allémann, E. Preparation and characterization of sterile sub-200 nm meso-tetra (4-hydroxylphenyl) porphyrin-loaded nanoparticles for photodynamic therapy. Eur. J. Pharm. Biopharm. 2003, 55, 115–124. [Google Scholar] [CrossRef]
- Abrahamse, H.; Hamblin, M.R. New photosensitizers for photodynamic therapy. Biochem. J. 2016, 473, 347–364. [Google Scholar] [CrossRef]
- Robertson, C.A.; Evans, D.H.; Abrahamse, H. Photodynamic therapy (PDT): A short review on cellular mechanisms and cancer research applications for PDT. J. Photochem. Photobiol. B Biol. 2009, 96, 1–8. [Google Scholar]
- Mroz, P.; Yaroslavsky, A.; Kharkwal, G.B.; Hamblin, M.R. Cell death pathways in photodynamic therapy of cancer. Cancers 2011, 3, 2516–2539. [Google Scholar]
- Konan, Y.N.; Gurny, R.; Allémann, E. State of the art in the delivery of photosensitizers for photodynamic therapy. J. Photochem. Photobiol. B Biol. 2002, 66, 89–106. [Google Scholar] [CrossRef]
- Lim, C.-K.; Heo, J.; Shin, S.; Jeong, K.; Seo, Y.H.; Jang, W.-D.; Park, C.R.; Park, S.Y.; Kim, S.; Kwon, I.C. Nanophotosensitizers toward advanced photodynamic therapy of cancer. Cancer Lett. 2013, 334, 176–187. [Google Scholar] [CrossRef]
- Chen, B.; Pogue, B.W.; Hoopes, P.J.; Hasan, T. Vascular and cellular targeting for photodynamic therapy. Crit. Rev. Eukaryot. Gene Expr. 2006, 16, 279–306. [Google Scholar] [CrossRef]
- Dolmans, D.E.; Fukumura, D.; Jain, R.K. Photodynamic therapy for cancer. Nat. Rev. Cancer 2003, 3, 380–387. [Google Scholar] [CrossRef]
- Kleinovink, J.W.; van Driel, P.B.; Snoeks, T.J.; Prokopi, N.; Fransen, M.F.; Cruz, L.J.; Mezzanotte, L.; Chan, A.; Löwik, C.W.; Ossendorp, F. Combination of photodynamic therapy and specific immunotherapy efficiently eradicates established tumors. Clin. Cancer Res. 2016, 22, 1459–1468. [Google Scholar] [CrossRef]
- Triesscheijn, M.; Baas, P.; Schellens, J.H.; Stewart, F.A. Photodynamic therapy in oncology. Oncologist 2006, 11, 1034–1044. [Google Scholar] [CrossRef]
- Abrahamse, H.; Kruger, C.A.; Kadanyo, S.; Mishra, A. Nanoparticles for advanced photodynamic therapy of cancer. Photomed. Laser Surg. 2017, 35, 581–588. [Google Scholar] [CrossRef]
- Chatterjee, D.K.; Fong, L.S.; Zhang, Y. Nanoparticles in photodynamic therapy: An emerging paradigm. Adv. Drug Deliv. Rev. 2008, 60, 1627–1637. [Google Scholar] [CrossRef]
- Kim, K.H.; Park, J.-J. The effects of photodynamic therapy in upper-gastrointestinal malignant diseases. Gut Liver 2010, 4, S39. [Google Scholar] [CrossRef]
- Kelly, J.; Snell, M. Hematoporphyrin derivative: A possible aid in the diagnosis and therapy of carcinoma of the bladder. J. Urol. 1976, 115, 150–151. [Google Scholar] [CrossRef]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D. Photodynamic therapy of cancer: An update. CA A Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef] [PubMed]
- Hayata, Y.; Kato, H.; Konaka, C.; Hayashi, N.; Tahara, M.; Saito, T.; Ono, J. Fiberoptic bronchoscopic photoradiation in experimentally induced canine lung cancer. Cancer 1983, 51, 50–56. [Google Scholar] [CrossRef]
- Kato, H.; Konaka, C.; Ono, J.; Matsushima, Y.; Saito, M.; Tahara, M.; Kawate, N.; Yoneyama, K.; Nishimiya, K.; Iimura, I. Cancer localization by detection of fluorescence by means of HpD administration and krypton ion laser photoradiation in canine lung cancer. Lung Cancer 1981, 21, 439–445. [Google Scholar]
- Fayter, D.; Corbett, M.; Heirs, M.; Fox, D.; Eastwood, A. A systematic review of photodynamic therapy in the treatment of pre-cancerous skin conditions, barrett’s oesophagus and cancers of the biliary tract, brain, head and neck, Lung, oesophagus and skin. In NIHR Health Technology Assessment Programme: Executive Summaries; NIHR Journals Library, Centre for Reviews and Dissemination (CRD), University of York: York, UK, 2010. [Google Scholar]
- Gao, F.; Bai, Y.; Ma, S.-R.; Liu, F.; Li, Z.-S. Systematic review: Photodynamic therapy for unresectable cholangiocarcinoma. J. Hepato-Biliary-Pancreat. Sci. 2010, 17, 125–131. [Google Scholar] [CrossRef]
- Hahn, S.M.; Smith, R.P.; Friedberg, J. Photodynamic therapy for mesothelioma. Curr. Treat. Options Oncol. 2001, 2, 375–383. [Google Scholar] [CrossRef]
- Hahn, S.M.; Fraker, D.L.; Mick, R.; Metz, J.; Busch, T.M.; Smith, D.; Zhu, T.; Rodriguez, C.; Dimofte, A.; Spitz, F. A phase II trial of intraperitoneal photodynamic therapy for patients with peritoneal carcinomatosis and sarcomatosis. Clin. Cancer Res. 2006, 12, 2517–2525. [Google Scholar] [CrossRef]
- Hendren, S.K.; Hahn, S.M.; Spitz, F.R.; Bauer, T.W.; Rubin, S.C.; Zhu, T.; Glatstein, E.; Fraker, D.L. Phase II trial of debulking surgery and photodynamic therapy for disseminated intraperitoneal tumors. Ann. Surg. Oncol. 2001, 8, 65–71. [Google Scholar] [CrossRef]
- Yano, T.; Wang, K.K. Photodynamic therapy for gastrointestinal cancer. Photochem. Photobiol. 2020, 96, 517–523. [Google Scholar] [CrossRef]
- Eljamel, M.S.; Goodman, C.; Moseley, H. ALA and Photofrin® Fluorescence-guided resection and repetitive PDT in glioblastoma multiforme: A single centre Phase III randomised controlled trial. Lasers Med. Sci. 2008, 23, 361–367. [Google Scholar] [CrossRef]
- Gaspar, L.E.; Fisher, B.J.; Macdonald, D.R.; Leber, D.V.; Halperin, E.C.; Schold, S.C., Jr.; Cairncross, J.G. Supratentorial malignant glioma: Patterns of recurrence and implications for external beam local treatment. Int. J. Radiat. Oncol. Biol. Phys. 1992, 24, 55–57. [Google Scholar] [CrossRef]
- Wilson, C. Glioblastoma: The past, the present, and the future. Clin. Neurosurg. 1992, 38, 32–48. [Google Scholar] [PubMed]
- Quirk, B.J.; Brandal, G.; Donlon, S.; Vera, J.C.; Mang, T.S.; Foy, A.B.; Lew, S.M.; Girotti, A.W.; Jogal, S.; LaViolette, P.S. Photodynamic therapy (PDT) for malignant brain tumors–where do we stand? Photodiagnosis Photodyn. Ther. 2015, 12, 530–544. [Google Scholar] [CrossRef] [PubMed]
- Stylli, S.S.; Kaye, A.H.; MacGregor, L.; Howes, M.; Rajendra, P. Photodynamic therapy of high grade glioma–long term survival. J. Clin. Neurosci. 2005, 12, 389–398. [Google Scholar] [PubMed]
- Mahmoudi, K.; Garvey, K.; Bouras, A.; Cramer, G.; Stepp, H.; Raj, J.J.; Bozec, D.; Busch, T.; Hadjipanayis, C. 5-aminolevulinic acid photodynamic therapy for the treatment of high-grade gliomas. J. Neuro-Oncol. 2019, 141, 595–607. [Google Scholar]
- Beck, T.J.; Kreth, F.W.; Beyer, W.; Mehrkens, J.H.; Obermeier, A.; Stepp, H.; Stummer, W.; Baumgartner, R. Interstitial photodynamic therapy of nonresectable malignant glioma recurrences using 5-aminolevulinic acid induced protoporphyrin IX. Lasers Surg. Med. Off. J. Am. Soc. Laser Med. Surg. 2007, 39, 386–393. [Google Scholar] [CrossRef]
- Dupont, C.; Vermandel, M.; Leroy, H.-A.; Quidet, M.; Lecomte, F.; Delhem, N.; Mordon, S.; Reyns, N. INtraoperative photoDYnamic Therapy for GliOblastomas (INDYGO): Study protocol for a phase I clinical trial. Neurosurgery 2019, 84, E414–E419. [Google Scholar]
- Johansson, A.; Faber, F.; Kniebühler, G.; Stepp, H.; Sroka, R.; Egensperger, R.; Beyer, W.; Kreth, F.W. Protoporphyrin IX fluorescence and photobleaching during interstitial photodynamic therapy of malignant gliomas for early treatment prognosis. Lasers Surg. Med. 2013, 45, 225–234. [Google Scholar] [CrossRef]
- Schwartz, C.; Rühm, A.; Tonn, J.-C.; Kreth, S.; Kreth, F.-W. Surg-25interstitial photodynamic therapy of de-novo glioblastoma multiforme who IV. Neuro Oncol. 2015, 17 (Suppl. 5), v219. [Google Scholar] [CrossRef]
- Vermandel, M.; Dupont, C.; Quidet, M.; Lecomte, F.; Lerhun, E.; Mordon, S.; Betrouni, N.; Reyns, N. Set-up of the first pilot study on intraopertive 5-ALA PDT: INDYGO trial. Photodiagnosis Photodyn. Ther. 2017, 100, A21. [Google Scholar]
- Shafirstein, G.; Battoo, A.; Harris, K.; Baumann, H.; Gollnick, S.O.; Lindenmann, J.; Nwogu, C.E. Photodynamic therapy of non–small cell lung cancer. Narrative review and future directions. Ann. Am. Thorac. Soc. 2016, 13, 265–275. [Google Scholar] [CrossRef]
- Usuda, J.; Kato, H.; Okunaka, T.; Furukawa, K.; Tsutsui, H.; Yamada, K.; Suga, Y.; Honda, H.; Nagatsuka, Y.; Ohira, T. Photodynamic therapy (PDT) for lung cancers. J. Thorac. Oncol. 2006, 1, 489–493. [Google Scholar] [PubMed]
- Hayata, Y.; Kato, H.; Konaka, C.; Ono, J.; Takizawa, N. Hematoporphyrin derivative and laser photoradiation in the treatment of lung cancer. Chest 1982, 81, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Ono, J.; Konaka, C.; Kawate, N.; Yoneyama, K.; Kinoshita, K.; Nishimiya, K.; Sakai, H.; Noguchi, M.; Tomono, T. Clinical measurement of tumor fluorescence using a new diagnostic system with hematoporphyrin derivative, laser photoradiation, and a spectroscope. Lasers Surg. Med. 1984, 4, 49–58. [Google Scholar] [CrossRef]
- Kato, H.; Konaka, C.; Kawate, N.; Shinohara, H.; Kinoshita, K.; Noguchi, M.; Ootomo, S.; Hayata, Y. Five-year disease-free survival of a lung cancer patient treated only by photodynamic therapy. Chest 1986, 90, 768–770. [Google Scholar] [CrossRef]
- Manyak, M.J.; Ogan, K. Photodynamic therapy for refractory superficial bladder cancer: Long-term clinical outcomes of single treatment using intravesical diffusion medium. J. Endourol. 2003, 17, 633–639. [Google Scholar] [CrossRef]
- Nseyo, U.O.; DeHaven, J.; Dougherty, T.J.; Potter, W.R.; Merrill, D.L.; Lundahl, S.L.; Lamm, D.L. Photodynamic therapy (PDT) in the treatment of patients with resistant superficial bladder cancer: A long term experience. J. Clin. Laser Med. Surg. 1998, 16, 61–68. [Google Scholar] [CrossRef]
- Railkar, R.; Agarwal, P.K. Photodynamic therapy in the treatment of bladder cancer: Past challenges and current innovations. Eur. Urol. Focus 2018, 4, 509–511. [Google Scholar] [CrossRef]
- Filonenko, E.; Kaprin, A.; Alekseev, B.; Apolikhin, O.; Slovokhodov, E.; Ivanova-Radkevich, V.; Urlova, A. 5-Aminolevulinic acid in intraoperative photodynamic therapy of bladder cancer (results of multicenter trial). Photodiagnosis Photodyn. Ther. 2016, 16, 106–109. [Google Scholar] [CrossRef]
- Kubba, A. Role of photodynamic therapy in the management of gastrointestinal cancer. Digestion 1999, 60, 1–10. [Google Scholar] [CrossRef]
- Ono, H.; Kondo, H.; Gotoda, T.; Shirao, K.; Yamaguchi, H.; Saito, D.; Hosokawa, K.; Shimoda, T.; Yoshida, S. Endoscopic mucosal resection for treatment of early gastric cancer. Gut 2001, 48, 225–229. [Google Scholar] [CrossRef]
- Yoshida, K.; Suzuki, S.; Mimura, S.; Ichii, M.; Sakai, H.; Shimao, H.; Kato, H.; Ito, Y.; Hiki, Y.; Hayashi, K. Photodynamic therapy for superficial esophageal cancer: A phase III study using PHE and excimer dye laser. Gan to kagaku ryoho. Cancer Chemother. 1993, 20, 2063. [Google Scholar]
- Kuwano, H.; Nishimura, Y.; Oyama, T.; Kato, H.; Kitagawa, Y.; Kusano, M.; Shimada, H.; Takiuchi, H.; Toh, Y.; Doki, Y. Guidelines for diagnosis and treatment of carcinoma of the esophagus April 2012 edited by the Japan Esophageal Society. Esophagus 2015, 12, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Overholt, B.F.; Lightdale, C.J.; Wang, K.K.; Canto, M.I.; Burdick, S.; Haggitt, R.C.; Bronner, M.P.; Taylor, S.L.; Grace, M.G.; Depot, M. Photodynamic therapy with porfimer sodium for ablation of high-grade dysplasia in Barrett’s esophagus: International, partially blinded, randomized phase III trial. Gastrointest. Endosc. 2005, 62, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Matono, S.; Nagano, T.; Murata, K.; Sueyoshi, S.; Yamana, H.; Shirouzu, K.; Fujita, H. Photodynamic therapy for large superficial squamous cell carcinoma of the esophagus. Gastrointest. Endosc. 2011, 73, 1–6. [Google Scholar] [CrossRef]
- Yano, T.; Muto, M.; Minashi, K.; Iwasaki, J.; Kojima, T.; Fuse, N.; Doi, T.; Kaneko, K.; Ohtsu, A. Photodynamic therapy as salvage treatment for local failure after chemoradiotherapy in patients with esophageal squamous cell carcinoma: A phase II study. Int. J. Cancer 2012, 131, 1228–1234. [Google Scholar] [CrossRef]
- Hasuike, N.; Ono, H.; Boku, N.; Mizusawa, J.; Takizawa, K.; Fukuda, H.; Oda, I.; Doyama, H.; Kaneko, K.; Hori, S. A non-randomized confirmatory trial of an expanded indication for endoscopic submucosal dissection for intestinal-type gastric cancer (cT1a): The Japan Clinical Oncology Group study (JCOG0607). Gastric Cancer 2018, 21, 114–123. [Google Scholar] [CrossRef]
- Mimura, S.; Ito, Y.; Nagayo, T.; Ichii, M.; Kato, H.; Sakai, H.; Goto, K.; Noguchi, Y.; Tanimura, H.; Nagai, Y. Cooperative clinical trial of photodynamic therapy with photofrin II and excimer dye laser for early gastric cancer. Lasers Surg. Med. Off. J. Am. Soc. Laser Med. Surg. 1996, 19, 168–172. [Google Scholar] [CrossRef]
- Park, D.H.; Lee, S.S.; Park, S.E.; Lee, J.L.; Choi, J.H.; Choi, H.J.; Jang, J.W.; Kim, H.J.; Eum, J.B.; Seo, D.-W. Randomised phase II trial of photodynamic therapy plus oral fluoropyrimidine, S-1, versus photodynamic therapy alone for unresectable hilar cholangiocarcinoma. Eur. J. Cancer 2014, 50, 1259–1268. [Google Scholar] [CrossRef]
- Kobayashi, H.; Choyke, P.L. Near-infrared photoimmunotherapy of cancer. Acc. Chem. Res. 2019, 52, 2332–2339. [Google Scholar] [CrossRef]
- Alsaab, H.O.; Sau, S.; Alzhrani, R.; Tatiparti, K.; Bhise, K.; Kashaw, S.K.; Iyer, A.K. PD-1 and PD-L1 checkpoint signaling inhibition for cancer immunotherapy: Mechanism, combinations, and clinical outcome. Front. Pharmacol. 2017, 8, 561. [Google Scholar] [CrossRef]
- Decker, W.K.; da Silva, R.F.; Sanabria, M.H.; Angelo, L.S.; Guimarães, F.; Burt, B.M.; Kheradmand, F.; Paust, S. Cancer immunotherapy: Historical perspective of a clinical revolution and emerging preclinical animal models. Front. Immunol. 2017, 8, 829. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.R.; Huang, Z.; Korbelik, M.; Nordquist, R.E.; Liu, H. Photoimmunotherapy for cancer treatment. J. Environ. Pathol. Toxicol. Oncol. 2006, 25, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Mitsunaga, M.; Ogawa, M.; Kosaka, N.; Rosenblum, L.T.; Choyke, P.L.; Kobayashi, H. Cancer cell–selective in vivo near infrared photoimmunotherapy targeting specific membrane molecules. Nat. Med. 2011, 17, 1685–1691. [Google Scholar] [CrossRef] [PubMed]
- Nagaya, T.; Nakamura, Y.; Sato, K.; Harada, T.; Choyke, P.L.; Hodge, J.W.; Schlom, J.; Kobayashi, H. Near infrared photoimmunotherapy with avelumab, an anti-programmed death-ligand 1 (PD-L1) antibody. Oncotarget 2017, 8, 8807. [Google Scholar] [CrossRef] [PubMed]
- Bhise, K.; Sau, S.; Alsaab, H.; Kashaw, S.K.; Tekade, R.K.; Iyer, A.K. Nanomedicine for cancer diagnosis and therapy: Advancement, success and structure–activity relationship. Ther. Deliv. 2017, 8, 1003–1018. [Google Scholar] [CrossRef] [PubMed]
- Overchuk, M.; Zheng, G. Overcoming obstacles in the tumor microenvironment: Recent advancements in nanoparticle delivery for cancer theranostics. Biomaterials 2018, 156, 217–237. [Google Scholar] [CrossRef]
- Sau, S.; Tatiparti, K.; Alsaab, H.O.; Kashaw, S.K.; Iyer, A.K. A tumor multicomponent targeting chemoimmune drug delivery system for reprograming the tumor microenvironment and personalized cancer therapy. Drug Discov. Today 2018, 23, 1344. [Google Scholar] [CrossRef]
- Sztandera, K.; Działak, P.; Marcinkowska, M.; Stańczyk, M.; Gorzkiewicz, M.; Janaszewska, A.; Klajnert-Maculewicz, B. Sugar Modification Enhances Cytotoxic Activity of PAMAM-Doxorubicin Conjugate in Glucose-Deprived MCF-7 Cells–Possible Role of GLUT1 Transporter. Pharm. Res. 2019, 36, 140. [Google Scholar] [CrossRef]
- Wang, Z.; Sau, S.; Alsaab, H.O.; Iyer, A.K. CD44 directed nanomicellar payload delivery platform for selective anticancer effect and tumor specific imaging of triple negative breast cancer. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 1441–1454. [Google Scholar] [CrossRef]
- Master, A.; Livingston, M.; Gupta, A.S. Photodynamic nanomedicine in the treatment of solid tumors: Perspectives and challenges. J. Control. Release 2013, 168, 88–102. [Google Scholar] [CrossRef]
- Alsaab, H.O.; Sau, S.; Alzhrani, R.M.; Cheriyan, V.T.; Polin, L.A.; Vaishampayan, U.; Rishi, A.K.; Iyer, A.K. Tumor hypoxia directed multimodal nanotherapy for overcoming drug resistance in renal cell carcinoma and reprogramming macrophages. Biomaterials 2018, 183, 280–294. [Google Scholar] [CrossRef] [PubMed]
- Saczko, J.; Chwilkowska, A.; Kulbacka, J.; Berdowska, I.; Zielinski, B.; Drag-Zalesinska, M.; Wysocka, T.; Lugowski, M.; Banas, T. Photooxidative action in cancer and normal cells induced by the use of Photofrin® in photodynamic therapy. Folia Biol. Praha 2008, 54, 24. [Google Scholar] [PubMed]
- Yano, S.; Hirohara, S.; Obata, M.; Hagiya, Y.; Ogura, S.-i.; Ikeda, A.; Kataoka, H.; Tanaka, M.; Joh, T. Current states and future views in photodynamic therapy. Photochem. Photobiol. C Photochem. Rev. 2011, 12, 46–67. [Google Scholar] [CrossRef]
- Debele, T.A.; Peng, S.; Tsai, H.-C. Drug carrier for photodynamic cancer therapy. Int. J. Mol. Sci. 2015, 16, 22094–22136. [Google Scholar] [CrossRef]
- Tian, G.; Zhang, X.; Gu, Z.; Zhao, Y. Recent advances in upconversion nanoparticles-based multifunctional nanocomposites for combined cancer therapy. Adv. Mater. 2015, 27, 7692–7712. [Google Scholar] [CrossRef]
- He, C.; Duan, X.; Guo, N.; Chan, C.; Poon, C.; Weichselbaum, R.R.; Lin, W. Core-shell nanoscale coordination polymers combine chemotherapy and photodynamic therapy to potentiate checkpoint blockade cancer immunotherapy. Nat. Commun. 2016, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.-O.; Ha, K.-S. New insights into the mechanisms for photodynamic therapy-induced cancer cell death. In International Review of Cell and Molecular Biology; Elsevier: Amsterdam, The Netherlands, 2012; Volume 295, pp. 139–174. [Google Scholar]
- Sadasivam, M.; Avci, P.; Gupta, G.K.; Lakshmanan, S.; Chandran, R.; Huang, Y.-Y.; Kumar, R.; Hamblin, M.R. Self-assembled liposomal nanoparticles in photodynamic therapy. Eur. J. Nanomed. 2013, 5, 115–129. [Google Scholar] [CrossRef]
- Avci, P.; Erdem, S.S.; Hamblin, M.R. Photodynamic therapy: One step ahead with self-assembled nanoparticles. J. Biomed. Nanotechnol. 2014, 10, 1937–1952. [Google Scholar] [CrossRef]
- Shah, Z.; Nazir, S.; Mazhar, K.; Abbasi, R.; Samokhvalov, I.M. PEGylated doped-and undoped-TiO2 nanoparticles for photodynamic Therapy of cancers. Photodiagnosis Photodyn. Ther. 2019, 27, 173–183. [Google Scholar] [CrossRef]
- He, Z.; Dai, Y.; Li, X.; Guo, D.; Liu, Y.; Huang, X.; Jiang, J.; Wang, S.; Zhu, G.; Zhang, F. Hybrid Nanomedicine Fabricated from Photosensitizer-Terminated Metal–Organic Framework Nanoparticles for Photodynamic Therapy and Hypoxia-Activated Cascade Chemotherapy. Small 2019, 15, 1804131. [Google Scholar] [CrossRef]
- R Mokoena, D.; P George, B.; Abrahamse, H. Enhancing breast cancer treatment using a combination of cannabidiol and gold nanoparticles for photodynamic therapy. Int. J. Mol. Sci. 2019, 20, 4771. [Google Scholar] [CrossRef] [PubMed]
- Chi, Y.-f.; Qin, J.-j.; Li, Z.; Ge, Q.; Zeng, W.-h. Enhanced anti-tumor efficacy of 5-aminolevulinic acid-gold nanoparticles-mediated photodynamic therapy in cutaneous squamous cell carcinoma cells. Braz. J. Med. Biol. Res. 2020, 53. [Google Scholar] [CrossRef] [PubMed]
- AlSalhi, M.S.; Aziz, M.H.; Atif, M.; Fatima, M.; Shaheen, F.; Devanesan, S.; Farooq, W.A. Synthesis of NiO nanoparticles and their evaluation for photodynamic therapy against HeLa cancer cells. J. King Saud Univ. Sci. 2020, 32, 1395–1402. [Google Scholar] [CrossRef]
- Zhang, T.; Jiang, Z.; Chen, L.; Pan, C.; Sun, S.; Liu, C.; Li, Z.; Ren, W.; Wu, A.; Huang, P. PCN-Fe (III)-PTX nanoparticles for MRI guided high efficiency chemo-photodynamic therapy in pancreatic cancer through alleviating tumor hypoxia. Nano Res. 2020, 13, 273–281. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Lee, H.-I.; Kim, J.-K.; Kim, C.-H.; Kim, Y.-J. Peptide 18-4/chlorin e6-conjugated polyhedral oligomeric silsesquioxane nanoparticles for targeted photodynamic therapy of breast cancer. Colloids Surf. B 2020, 189, 110829. [Google Scholar] [CrossRef]
- Feng, J.; Wang, S.; Wang, Y.; Wang, L. Stem cell membrane–camouflaged bioinspired nanoparticles for targeted photodynamic therapy of lung cancer. J. Nanopart. Res. 2020, 22, 1–11. [Google Scholar] [CrossRef]
- Zhang, T.; Bao, J.; Zhang, M.; Ge, Y.; Wei, J.; Li, Y.; Wang, W.; Li, M.; Jin, Y. Chemo-photodynamic therapy by pulmonary delivery of gefitinib nanoparticles and 5-aminolevulinic acid for treatment of primary lung cancer of rats. Photodiagnosis Photodyn. Ther. 2020, 31, 101807. [Google Scholar] [CrossRef]
- Huang, X.; Chen, J.; Wu, W.; Yang, W.; Zhong, B.; Qing, X.; Shao, Z. Delivery of MutT homolog 1 inhibitor by functionalized graphene oxide nanoparticles for enhanced chemo-photodynamic therapy triggers cell death in osteosarcoma. Acta Biomater. 2020, 109, 229–243. [Google Scholar] [CrossRef]
- Wan, G.; Cheng, Y.; Song, J.; Chen, Q.; Chen, B.; Liu, Y.; Ji, S.; Chen, H.; Wang, Y. Nucleus-targeting near-infrared nanoparticles based on TAT peptide-conjugated IR780 for photo-chemotherapy of breast cancer. Chem. Eng. Sci. 2020, 380, 122458. [Google Scholar] [CrossRef]
- Khaniabadi, P.M.; Shahbazi-Gahrouei, D.; Aziz, A.A.; Dheyab, M.A.; Khaniabadi, B.M.; Mehrdel, B.; Jameel, M.S. Trastuzumab conjugated porphyrin-superparamagnetic iron oxide nanoparticle: A potential PTT-MRI bimodal agent for herceptin positive breast cancer. Photodiagnosis Photodyn. Ther. 2020, 31, 101896. [Google Scholar] [CrossRef]
- Damke, G.M.Z.F.; Damke, E.; de Souza Bonfim-Mendonça, P.; Ratti, B.A.; de Freitas Meirelles, L.E.; da Silva, V.R.S.; Gonçalves, R.S.; César, G.B.; de Oliveira Silva, S.; Caetano, W. Selective photodynamic effects on cervical cancer cells provided by P123 Pluronic®-based nanoparticles modulating hypericin delivery. Life Sci. 2020, 255, 117858. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Kuang, G.; He, S.; Lu, H.; Cheng, Y.; Zhou, D.; Huang, Y. Photoactivatable Prodrug-Backboned Polymeric Nanoparticles for Efficient Light-Controlled Gene Delivery and Synergistic Treatment of Platinum-Resistant Ovarian Cancer. Nano Lett. 2020, 20, 3039–3049. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Tian, J.; Zhu, H.; Hong, L.; Mao, Z.; Oliveira, J.M.; Reis, R.L.; Li, X. Tumor-Targeting Polycaprolactone Nanoparticles with Codelivery of Paclitaxel and IR780 for Combinational Therapy of Drug-Resistant Ovarian Cancer. ACS Biomater. Sci. Eng. 2020, 6, 2175–2185. [Google Scholar] [CrossRef]
- Gaio, E.; Conte, C.; Esposito, D.; Reddi, E.; Quaglia, F.; Moret, F. CD44 Targeting Mediated by Polymeric Nanoparticles and Combination of Chlorine TPCS2a-PDT and Docetaxel-Chemotherapy for Efficient Killing of Breast Differentiated and Stem Cancer Cells in Vitro. Cancers 2020, 12, 278. [Google Scholar] [CrossRef]
- Nann, T. Nanoparticles in Photodynamic Therapy. Nano Biomed. Eng. 2011, 3, 137–143. [Google Scholar] [CrossRef]
- Calixto, G.M.F.; Bernegossi, J.; De Freitas, L.M.; Fontana, C.R.; Chorilli, M. Nanotechnology-based drug delivery systems for photodynamic therapy of cancer: A review. Molecules 2016, 21, 342. [Google Scholar] [CrossRef]
- Sanchez-Moreno, P.; Ortega-Vinuesa, J.L.; Peula-Garcia, J.M.; Marchal, J.A.; Boulaiz, H. Smart drug-delivery systems for cancer nanotherapy. Curr. Drug Targets 2018, 19, 339–359. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, J. Using nanoparticles to enable simultaneous radiation and photodynamic therapies for cancer treatment. Nanosci. Nanotechnol. 2006, 6, 1159–1166. [Google Scholar] [CrossRef]
- Vatansever, F.; Chandran, R.; Sadasivam, M.; Chiang, L.Y.; Hamblin, M.R. Multi-functionality in theranostic nanoparticles: Is more always better? J. Nanomed. Nanotechnol. 2012, 3, 120. [Google Scholar] [CrossRef]
- Bechet, D.; Couleaud, P.; Frochot, C.; Viriot, M.-L.; Guillemin, F.; Barberi-Heyob, M. Nanoparticles as vehicles for delivery of photodynamic therapy agents. Trends Biotechnol. 2008, 26, 612–621. [Google Scholar] [CrossRef]
- Friedman, D.; Claypool, A.S.E.; Liu, R. The smart targeting of nanoparticles. Curr. Pharm. Des. 2013, 19, 6315–6329. [Google Scholar] [CrossRef] [PubMed]
- Sibani, S.A.; McCarron, P.A.; Woolfson, A.D.; Donnelly, R.F. Photosensitiser delivery for photodynamic therapy. Part 2: Systemic carrier platforms. Expert Opin. Drug Deliv. 2008, 5, 1241–1254. [Google Scholar] [CrossRef] [PubMed]
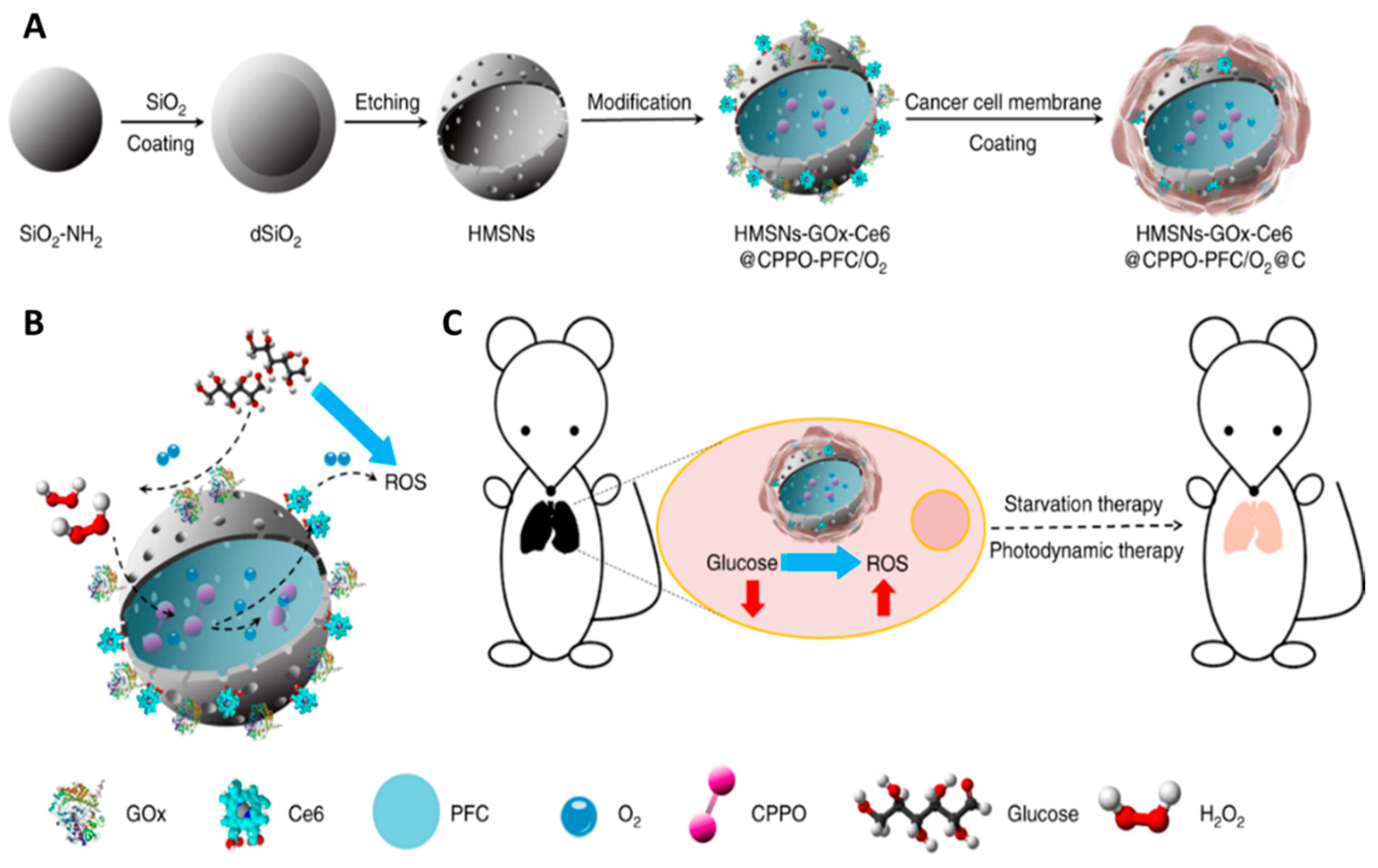
- Yu, Z.; Zhou, P.; Pan, W.; Li, N.; Tang, B. A biomimetic nanoreactor for synergistic chemiexcited photodynamic therapy and starvation therapy against tumor metastasis. Nat. Commun. 2018, 9, 1–9. [Google Scholar] [CrossRef]
- Shakiba, M.; Chen, J.; Zheng, G. Porphyrin nanoparticles in photomedicine. In Applications of Nanoscience in Photomedicine; Elsevier: Amsterdam, The Netherlands, 2015; pp. 511–526. [Google Scholar]
- Estelrich, J.; Busquets, M.A. Iron oxide nanoparticles in photothermal therapy. Molecules 2018, 23, 1567. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.; Kim, A.H. Laser Interstitial Thermal Therapy. MO Med. 2020, 117, 50. [Google Scholar]
- Mohammadi, A.M.; Sharma, M.; Beaumont, T.L.; Juarez, K.O.; Kemeny, H.; Dechant, C.; Seas, A.; Sarmey, N.; Lee, B.S.; Jia, X. Upfront magnetic resonance imaging-guided stereotactic laser-ablation in newly diagnosed glioblastoma: A multicenter review of survival outcomes compared to a matched cohort of biopsy-only patients. Neurosurgery 2019, 85, 762–772. [Google Scholar] [CrossRef]
- Willie, J.T.; Laxpati, N.G.; Drane, D.L.; Gowda, A.; Appin, C.; Hao, C.; Brat, D.J.; Helmers, S.L.; Saindane, A.; Nour, S.G. Real-time magnetic resonance-guided stereotactic laser amygdalohippocampotomy for mesial temporal lobe epilepsy. Neurosurgery 2014, 74, 569–585. [Google Scholar] [CrossRef]
- Wiebe, S.; Blume, W.T.; Girvin, J.P.; Eliasziw, M. A randomized, controlled trial of surgery for temporal-lobe epilepsy. MO Med. 2001, 345, 311–318. [Google Scholar] [CrossRef]
- Babu, A.; Templeton, A.K.; Munshi, A.; Ramesh, R. Nanoparticle-based drug delivery for therapy of lung cancer: Progress and challenges. J. Nanomater. 2013, 2013. [Google Scholar] [CrossRef]
- St Denis, T.G.; Hamblin, M.R. Synthesis, bioanalysis and biodistribution of photosensitizer conjugates for photodynamic therapy. Bioanalysis 2013, 5, 1099–1114. [Google Scholar] [CrossRef]
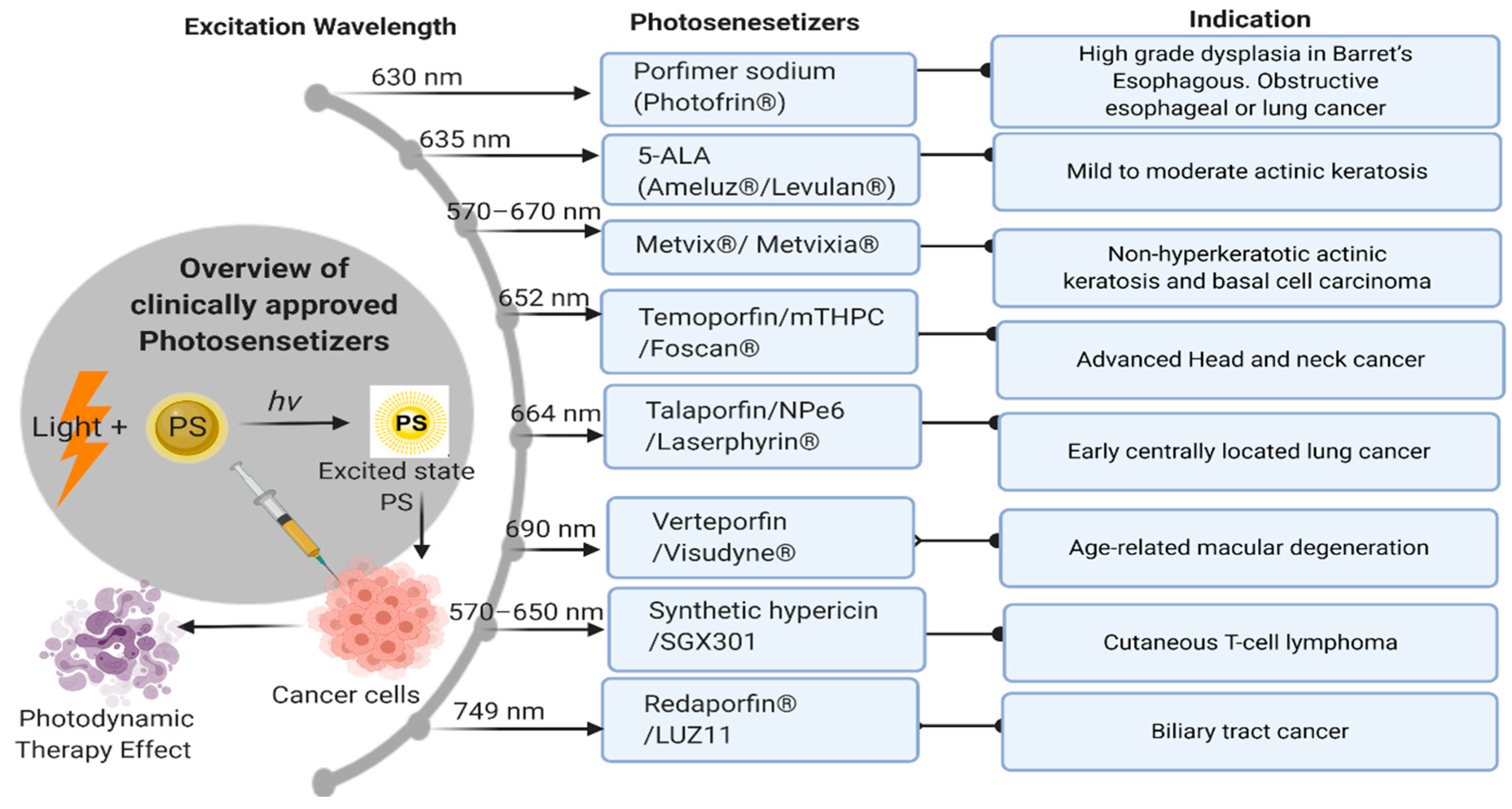
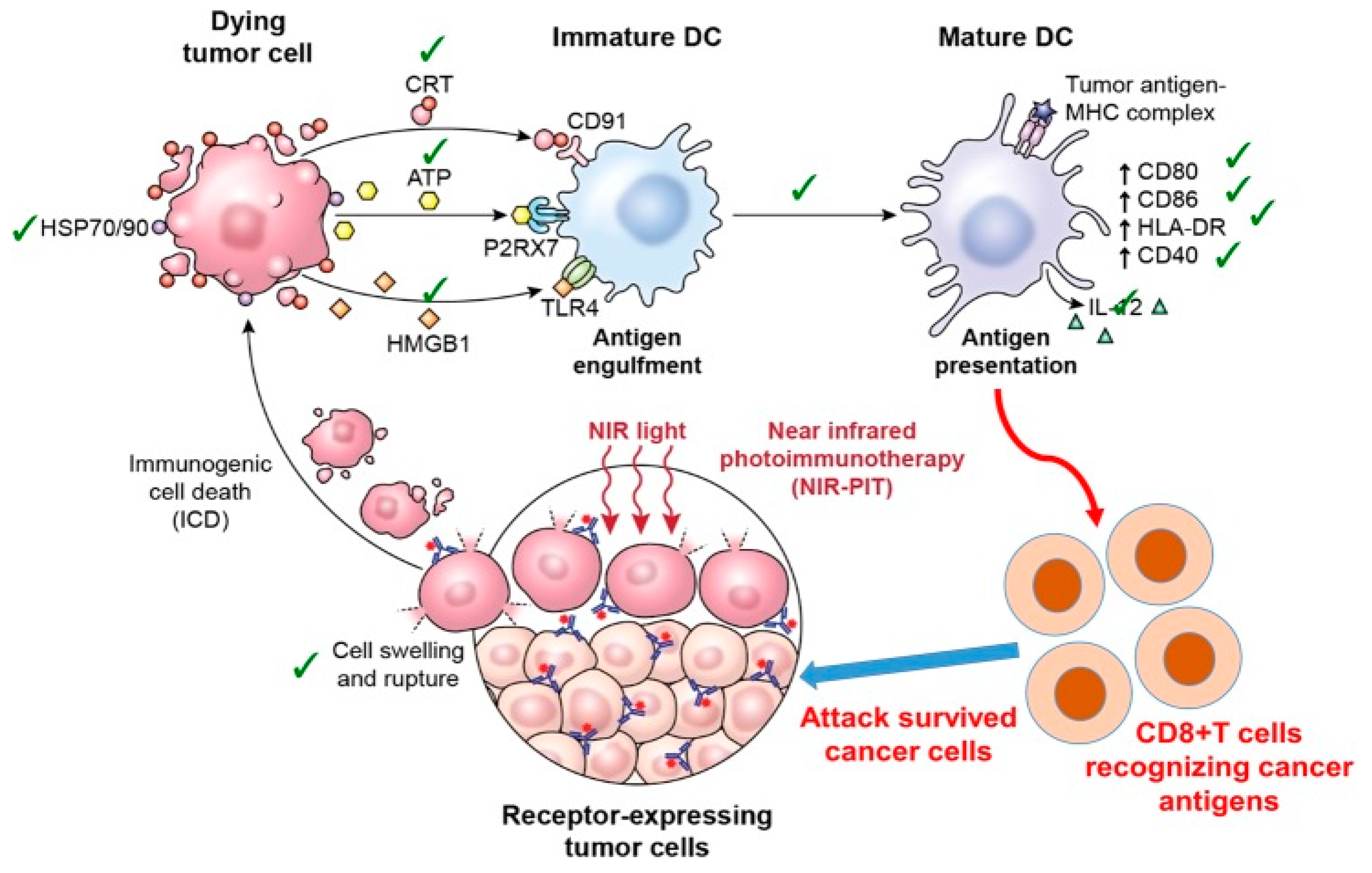
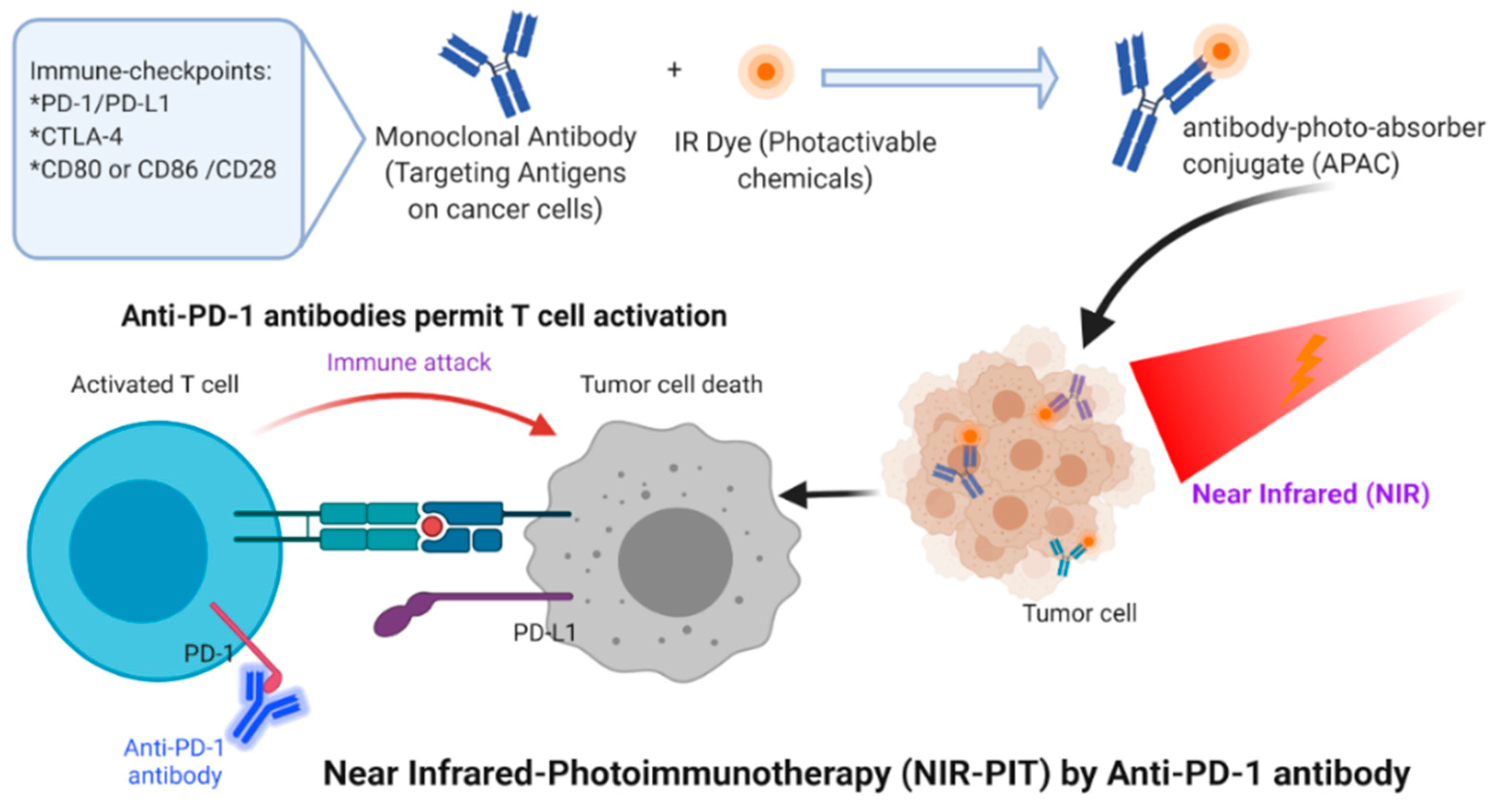

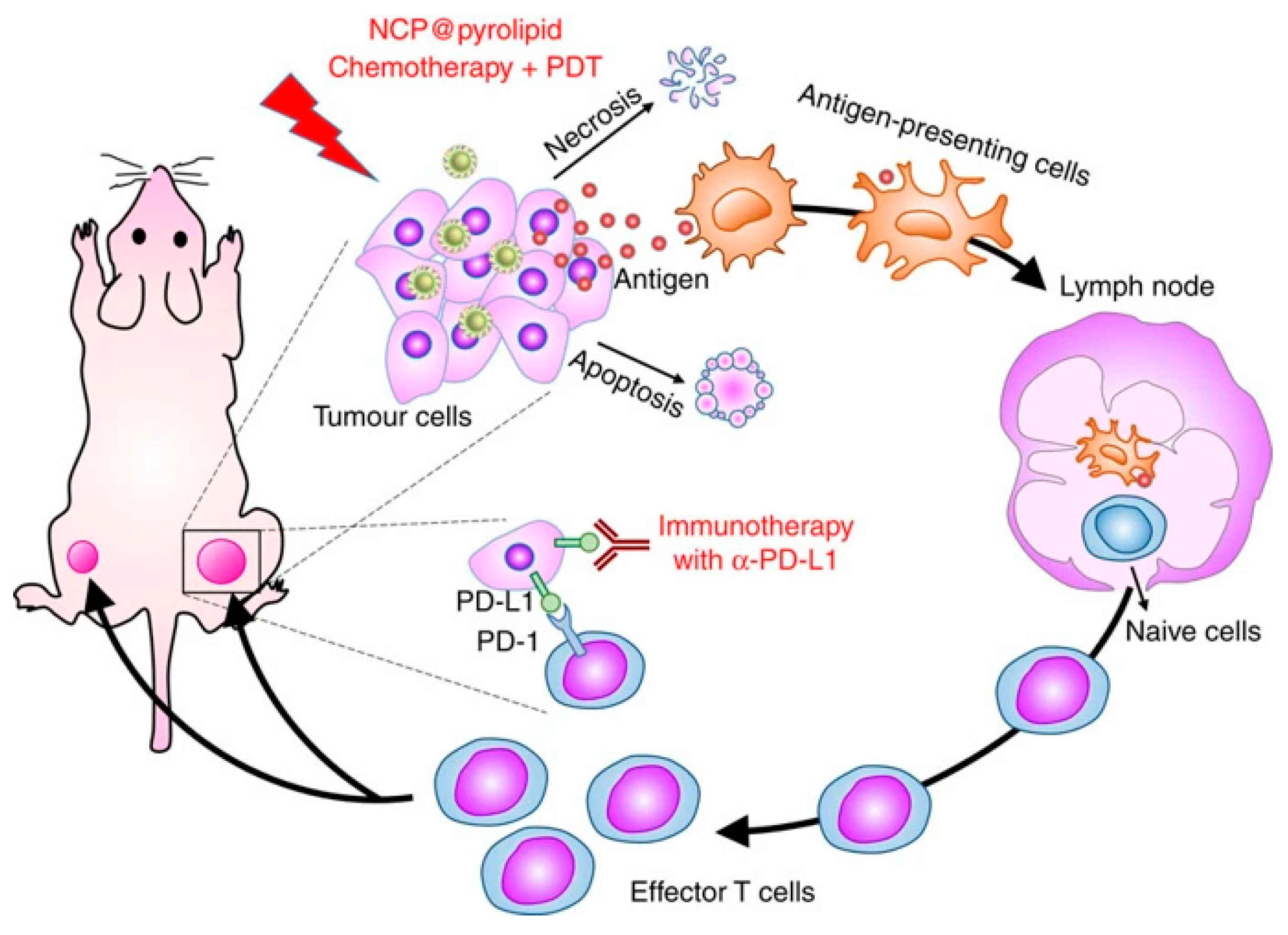
| # | Clinical Trial Phase (NCT) | Status | Type of Cancer or Indication | Drug Used/Light Applicator | Sponsor |
|---|---|---|---|---|---|
| 1 | Phase I (NCT01682746) | completed | Brain tumor (Recurrent) | Photofrin (porfimer sodium) & PDT | Harry T Whelan, MD Pinnacle Biologics, Inc. |
| 2 | Not provided (NCT02632084) | completed | Pituitary Neoplasms | Not Provided | The Leeds Teaching Hospitals NHS Trust |
| 3 | Phase III (NCT00118222) | completed | Brain and CNS Tumors | Drug: porfimer sodium + adjuvant therapy + conventional surgery | Case Comprehensive Cancer Center National Cancer Institute (NCI) |
| 4 | Phase I (NCT00002647) | ** Unknown | Brain and CNS Tumors Metastatic Cancer | Drug: verteporfin Procedure: conventional surgery | Medical College of Wisconsin |
| 5 | Phase III (NCT00003788) | ** Unknown | Brain and CNS Tumors | Drug: carmustine, lomustine porfimer sodium, procarbazine hydrochloride Procedure: neoadjuvant therapy, surgical procedure. Radiation: radiatiotherapy | Colorado Health Foundation |
| 6 | Not provided (NCT00984243) | Completed | Lung cancer | Photofrin II of intravenously. An argon-dye or an excimer-dye laser (620–630 nm) | Mayo Clinic |
| 7 | Phase II (NCT00601848) | Active, not recruiting | Lung Cancer and Metastatic Cancer | Chemotherapy porfimer sodium | Abramson Cancer Center of the University of Pennsylvania |
| 8 | Not provided (NCT00754910) | Unknown | Lung cancer | porfimer sodium | Ohio State University Comprehensive Cancer Center |
| 9 | Phase I (NCT00025571) | Completed | Lung cancer | HPPH | Roswell Park Cancer Institute |
| 10 | Phase I (NCT01854684) | Recruiting | Recurrent NSCLC Stage IIA, IIB, IIIA, and IIIB NSCLC | Temoporfin | Roswell Park Cancer Institute |
| 11 | Phase I (NCT00526461) | Completed | NSCLC in Situ or NSCLC Microinvasive Bronchogenic Carcinoma | HPPH | Roswell Park Cancer Institute |
| 12 | Phase I (NCT01668823) | Completed | Adenocarcinoma of the Lung; Large Cell Lung Cancer; Recurrent NSCLC; Squamous Cell Lung Cancer; Stage 0 NSCLC | HPPH + PDT | Roswell Park Cancer Institute |
| 13 | Phase I (NCT00014066) | Completed | Lung cancer | Drug: hematoporphyrin derivative Radiation: brachytherapy | Roswell Park Cancer Institute |
| 14 | Phase I (NCT02916745) | Not yet recruiting | NSCLC | Porfimer sodium | Concordia Laboratories Inc. |
| 15 | Not provided (NCT01842555) | Recruiting | Lung Cancer; Esophageal | PDT | Main Line Health |
| 16 | Phase II (NCT00054002) | Completed | Malignant Mesothelioma | Procedure: adjuvant therapy Procedure: conventional surgery Drug: porfimer sodium | Roswell Park Cancer Institute |
| 17 | Phase I (NCT02464761) | Recruiting | Vertebral Metastases | Visudyne | Sunnybrook Health Sciences Centre |
| 18 | Not provided (NCT02514226) | Not yet recruiting | Bronchiectasis; Periodontal Disease | PDT | University of Nove de Julho |
| 19 | Phase II (NCT02497053) | Recruiting | Malignant Pleural Mesothelioma | Pemetrexed/platinum Chemotherapy | Ain Shams University |
| 20 | Phase I (NCT03053635) | completed | Non-Muscle Invasive Bladder Cancer (NMIBC) Refractory to BCG | Drug: TLD1433 infusion and photodynamic therapy (PDT) treatment | Theralase Inc. University Health Network, Toronto Medelis Inc. WCCT Global |
| 21 | phase II | Recruiting | Non-Muscle Invasive Bladder Cancer (NMIBC) Refractory to BCG | TLD-1433 Bladder infusion + PDT | Theralase Inc. |
| 22 | Phase I (NCT01303991) | Active, not recruiting | Intermediate or High-risk Bladder Cancer | Hexvix PDT with Karl Storz T-Light | Photocure Karl Storz |
| 23 | phase I phase II (NCT00322699) | completed | Superficial Bladder Cancer | Procedure: Whole bladder laser light treatment as an alternative to radical cystectomy. Drug: Photofrin | North Florida/South Georgia Veterans Health System North Florida Foundation for Research and Education Axcan Pharma |
| # | Clinical Trial Phase (NCT#) | Status | Type of Cancer or Indication | Drug Used | Sponsor |
|---|---|---|---|---|---|
| 1 | Phase I, II (NCT02070432) | Recruiting | Head and neck cancer | LUZ11 | Luzitin SA |
| 2 | Phase I (NCT00978081) | Active, not recruiting | Head and Neck Cancer Precancerous Condition | aminolevulinic acid hydrochloride | Abramson Cancer Center of the University of Pennsylvania |
| 3 | Phase II (NCT00003856) | ** Unknown | Head and Neck Cancer | temoporfin | Quintiles, Inc. |
| 4 | Phase I (NCT01019954) | Completed | Head and Neck Tumors | Levulan | Abramson Cancer Center of the University of Pennsylvania |
| 5 | Phase I (NCT00670397) | Completed | Head and Neck Cancer Precancerous/Nonmalignant Condition | porfimer sodium + PDT | Roswell Park Cancer Institute |
| 6 | Phase I (NCT00028405) | Completed | Liver Metastasis Pelvic Cancer Head and Neck Breast, Colorectal, rectal and Mouth cancer, sarcoma. | Drug: LS 11(Taporfin Sodium) Device: Lumaflex Light Delivery Catheter | Light Sciences LLC |
| 7 | Phase I (NCT01043016) | ** Unknown | Skin Cancer Esophageal Cancer | Photocyanine Injection | Fujian Longhua Pharmaceutical Co. Ltd |
| 8 | Phase II (NCT01086488) | ** Unknown | Nasopharyngeal Carcinoma | FOSCAN | Ministry of Health, Malaysia |
| 9 | Phase I, II (NCT00060268) | Completed | Esophageal Cancer | HPPH | Roswell Park Cancer Institute |
| 10 | Phase II (NCT00002935) | Completed | Esophageal Cancer | porfimer sodium | Roswell Park Cancer Institute |
| 11 | Phase III (NCT02628665) | Recruiting | Stage I, II and III of Esophageal Adenocarcinoma and Esophageal Squamous Cell Carcinoma | photosensitizer(photofrin) Device: 630 nm laser irradiation (DIOMED) | The First Affiliated Hospital of Henan University of Science and Technology |
| 12 | Phase II (NCT00217087) | Completed | Early Stage Esophageal Adenocarcinoma. Barrett Esophagus | Porfimer sodium 2 mg/kg | Mayo Clinic |
| 13 | Phase I (NCT01366833) | ** Unknown | Malignant Dysphagia; Esophageal Cancer | Radiation: Brachytherapy; Procedure: Stent insertion | McGill University Health Center |
| 14 | (NCT00587314) | Enrolling by invitation | Barrett’s Esophagus; Early Esophageal Adenocarcinoma | Biopsy | Mayo Clinic |
| 15 | Phase IV (NCT00155337) | Completed | Oral Leukoplakia | Not provided | National Taiwan University Hospital |
| Type of Nanoparticles | Drug Used | Targeted Cancer | Results | Reference |
|---|---|---|---|---|
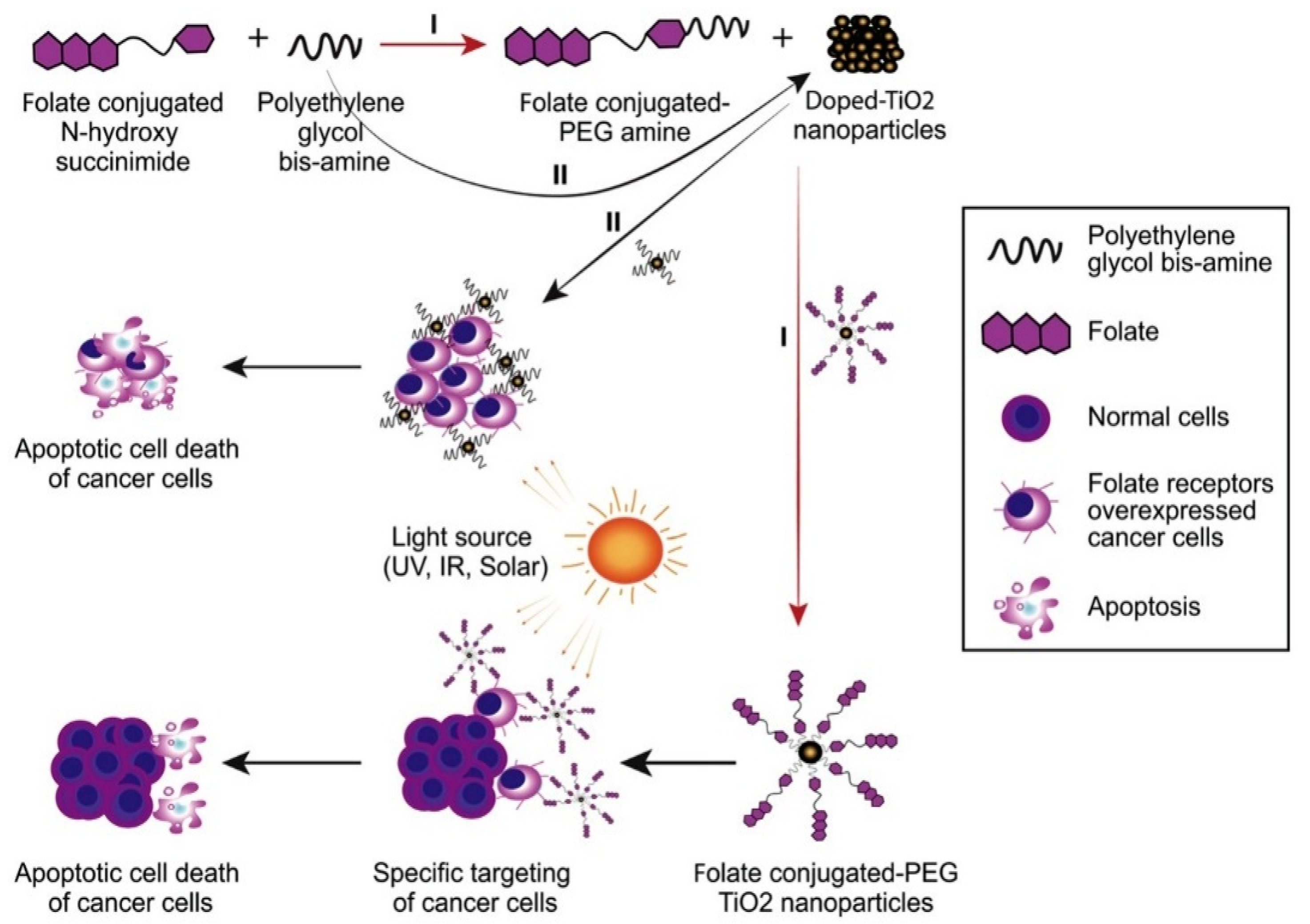
| doped- and undoped-TiO2 NPs stabilized by PEG | Titanium dioxide | cervical cancer cells (HeLa) | NPs significantly reduced the survival of human cervical cancer cells (HeLa). | [91] |
| nanoscale metal-organic frameworks (UiO-66-H/N3 NMOFs) | azido-/photosensitizer-terminated UiO-66 + the bioreductive prodrug banoxantrone (AQ4N) | not specified | Enhanced therapeutic efficacy Reduced systemic toxicity | [92] |
| gold nanoparticle (AuNP) conjugated photodynamic therapy (PDT) in combination with cannabidiol (CBD) | Cannabidiol (CBD) | breast cancer | Reduced side effects and toxicity to normal cells PDT and CBD are promising for hindering breast cancer progression and development | [93] |
| conjugation of gold nanoparticles (GNPs) | 5-aminolevulinic acid (5-ALA) | cutaneous squamous cell carcinoma (cSCC) non-melanoma skin cancer | PDT with 5-ALA and GNPs-conjugated 5-ALA (5-ALA-GNPs) significantly suppressed cell viability and increased cell apoptosis and singlet oxygen generation in both HaCat and A431 cells | [94] |
| metal-based nanoparticles (NPs) | NiO NPs | cervical cancer cells (HeLa) | A light dose of 100 J/cm2 and a NiO NP concentration of 180 μg/mL exhibited an effective PDT outcome on cervical cancer cells. The photokilling effect of NiO NPs as a potential treatment for cervical malignancy was supported | [95] |
| (PCN-Fe(III)-PTX) nanoparticles (NPs) | Fe(III)-complexed | pancreatic cancer (PaC) | NPs represented an ideal agent for mediating effective MRI-guided chemotherapy-PDT; great promise for the clinical treatment of pancreatic cancer | [96] |
| peptide p 18-4/chlorin e6 (Ce6)-conjugated polyhedral oligomeric silsesquioxane (PPC) nanoparticles | Chlorin e6 (Ce6) | breast cancer cells | PPC NPs are highly effective PDT agents for breast cancer therapy. | [97] |
| Stem cell membrane –camouflaged bioinspired nanoparticles | Chlorin e6 (Ce6)- | lung cancer | Enhanced antitumor effect of Ng/Ce6@SCV after NIR irradiation by significantly suppressing primary tumor growth with fewer side effects. | [98] |
| Gefitinib PLGA nanoparticles (GNPs) | 5-aminolevulinic acid (5-ALA) | lung cancer | The synergistic effect of CPDT was confirmed | [99] |
| graphene oxide nanoparticles | Polyethylene glycol (PEG), folic acid (FA), PS indocyanine green (ICG), and doxorubicin. | osteosarcoma | NPs with combined Chemo–PDT inhibited the proliferation and migration of osteosarcoma cells. | [100] |
| TID nanoparticles | Doxorubicin (DOX) | breast cancer | TID NPs rapidly destroyed the genetic substances and potently induced the apoptosis of breast cancer cells. | [101] |
| Fe3O4 nanoparticles (IONs) | Trastuzumab | breast cancer | No cytotoxicity was observed after incubating MCF 7 cells under various Fe concentrations of nanoparticles and Theranostic agents. | [102] |
| P123 Pluronic®-based nanoparticles | Hypericin | cervical cancer | HYP/P123 micelles had effective and selective time- and dose-dependent phototoxic effects on cervical cancer cells | [103] |
| photoactivatable Pt(IV) prodrug-backboned polymeric nanoparticle system (CNPPtCP/si(c-fos)) | Platinum | ovarian Cancer | CNPPtCP/si(c-fos) displayed excellent synergistic therapeutic efficacy on PROC with low toxicity. | [104] |
| poly-ε-caprolactone nanoparticles (PCL NPs) | IR780 and paclitaxel (PTX) | ovarian cancer | LHRH peptide modified PCL (PCL-LHRH) NPs demonstrated increased internalization in ovarian tumor cells in vitro and selective targeting in tumor xenografts in vivo. | [105] |
| Hyaluronic acid (HA) coated polymeric nanoparticles (HA-NPs) | docetaxel (DTX) and PS meso-tetraphenyl chlorine disulfonate (TPCS2a) | breast cancer | Combination therapy using co-loaded NPs (HA@DTX/TPCS2a-NPs) had superior efficacy over monotherapies (HA@DTX-NPs or HA@TPCS2a-NPs) in reducing the self-renewal capacity and eradicating the CSC population evaluated with an aldehyde dehydrogenase activity assay and CD44/CD24 immunostaining. | [106] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alsaab, H.O.; Alghamdi, M.S.; Alotaibi, A.S.; Alzhrani, R.; Alwuthaynani, F.; Althobaiti, Y.S.; Almalki, A.H.; Sau, S.; Iyer, A.K. Progress in Clinical Trials of Photodynamic Therapy for Solid Tumors and the Role of Nanomedicine. Cancers 2020, 12, 2793. https://doi.org/10.3390/cancers12102793
Alsaab HO, Alghamdi MS, Alotaibi AS, Alzhrani R, Alwuthaynani F, Althobaiti YS, Almalki AH, Sau S, Iyer AK. Progress in Clinical Trials of Photodynamic Therapy for Solid Tumors and the Role of Nanomedicine. Cancers. 2020; 12(10):2793. https://doi.org/10.3390/cancers12102793
Chicago/Turabian StyleAlsaab, Hashem O., Maha S. Alghamdi, Albatool S. Alotaibi, Rami Alzhrani, Fatimah Alwuthaynani, Yusuf S. Althobaiti, Atiah H. Almalki, Samaresh Sau, and Arun K. Iyer. 2020. "Progress in Clinical Trials of Photodynamic Therapy for Solid Tumors and the Role of Nanomedicine" Cancers 12, no. 10: 2793. https://doi.org/10.3390/cancers12102793
APA StyleAlsaab, H. O., Alghamdi, M. S., Alotaibi, A. S., Alzhrani, R., Alwuthaynani, F., Althobaiti, Y. S., Almalki, A. H., Sau, S., & Iyer, A. K. (2020). Progress in Clinical Trials of Photodynamic Therapy for Solid Tumors and the Role of Nanomedicine. Cancers, 12(10), 2793. https://doi.org/10.3390/cancers12102793







