18F-Facbc in Prostate Cancer: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Study Selection
2.2. Data Extraction
2.3. Statistical Method
3. Results
3.1. Search Results
3.2. Study Characteristics
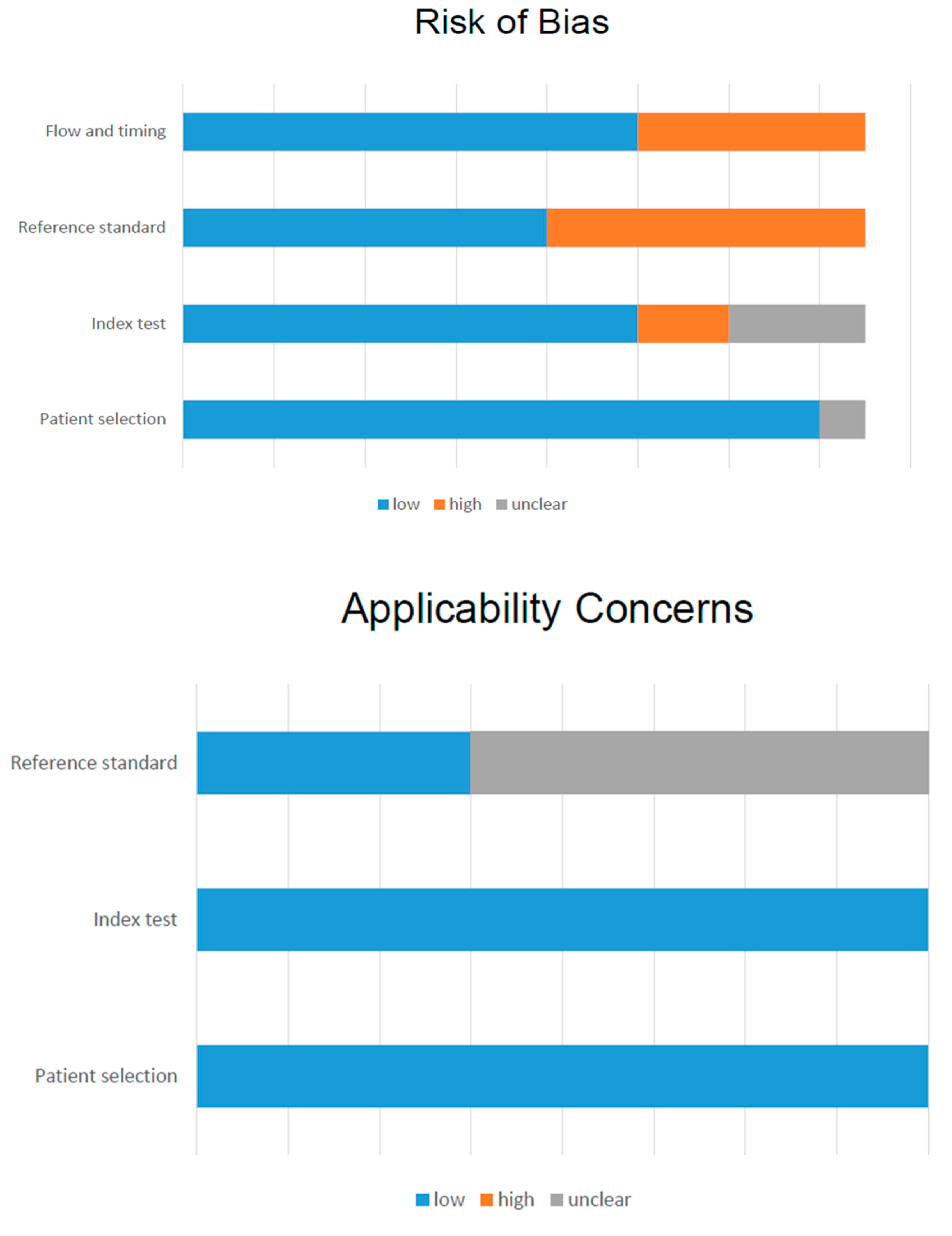
3.3. Methodological Quality
3.4. Qualitative Results
3.5. Quantitative Results
4. Discussion
- 18F-FACBC is more performant than 111In-capromab SPECT/CT and 11C-Choline for the detection of PCa recurrence. Therefore, if available it should be preferred in patients with a PSA increase, after primary treatments. However, data about the comparison with 18F-Choline PET/CT are missing and should be explored, also considering the radioisotope properties.
- The combination of 18F-FACBC PET/CT with mpMRI (or with a PET/MRI) seems useful for the detection of primary PCa, and therefore, it would be suggested in case of undetectable tumors in patients with a negative biopsy but a persistent PSA level increase. However, the interpretation of this sophisticated imaging required a great experience and a significant learning curve.
- The sensitivity for the evaluation of lymph node metastasis in the initial staging of disease is moderate (45%–66%; [31,36]), like for the other radiopharmaceuticals (radiolabeled PSMA and Choline; [41,42]). Probably the recent introduction of new imaging modalities, such as digital PET/CT or PET/MRI that has a higher spatial resolution, would improve the pathological lymph node detection.
- The pooled sensitivity for the identification of recurrence in prostate bed is high, being >90% with a limited pooled specificity (about 45%), probably due to the FP findings in case of inflamed cells, as reported by Oka et al. [43]. However, the absent uptake of radiopharmaceutical in the bladder represents a great advantage for the identification of peri-anastomotic PCa recurrence. Further data about the complementary role of 18F-FACBC and MRI are required for the assessment of prostatic bed recurrence, at different PSA levels.
- The recurrence in the extra-prostatic site may be assessed by 18F-FACBC PET/CT with a moderate sensitivity and specificity, independently from the PSA levels. However, the correlation with PSA kinetics is warranted in a selected large cohort of patients, thus testing the final impact on the patient management.
- Despite some articles have defined a potential impact of 18F-FACBC PET/CT on therapeutic management, there is still a lack information with regard to its role in radiotherapy planning and other adapted therapy.
5. Future Researches
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2016. CA Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.M.; Hoffman, K.E.; Levy, L.B.; Frank, S.J.; Pugh, T.J.; Choi, S.; Nguyen, Q.N.; McGuire, S.E.; Lee, A.K.; Kuban, D.A. Improvement in prostate cancer survival over time: A 20-year analysis. Cancer J. 2012, 18, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bruce, J.Y.; Lang, J.M.; McNeel, D.G.; Liu, G. Current controversies in the management of biochemical failure in prostate cancer. Clin. Adv. Hematol. Oncol. 2012, 10, 716–722. [Google Scholar] [PubMed]
- Roehl, K.A.; Han, M.; Ramos, C.G.; Antenor, J.A.V.; Catalona, W.J. Cancer progression and survival rates following anatomical radical retropubic prostatectomy in 3478 consecutive patients: Long-term results. J. Urol. 2004, 172, 910–914. [Google Scholar] [CrossRef] [PubMed]
- Simmons, M.N.; Stephenson, A.J.; Klein, E.A. Natural history of biochemical recurrence after radical prostatectomy: Risk assessment for secondary therapy. Eur. Urol. 2007, 51, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, H.U.; El-ShaterBosaily, A.; Brown, L.C.; Gabe, R.; Kaplan, R.; Parmar, M.K.; Collaco-Moraes, Y.; Ward, K.; Hindley, R.G.; Freeman, A.; et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): A paired validating confirmatory study. Lancet (Lond. Engl.) 2017, 389, 815–822. [Google Scholar] [CrossRef]
- Panebianco, V.; Barchetti, F.; Grompone, M.D.; Colarieti, A.; Salvo, V.; Cardone, G.; Catalano, C. Magnetic resonance imaging for localization of prostate cancer in the setting of biochemical recurrence. Urol. Oncol. Semin. Orig. Investig. 2016, 34, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Abd-Alazeez, M.; Ramachandran, N.; Dikaios, N.; Ahmed, H.U.; Emberton, M.; Kirkham, A.; Arya, M.; Taylor, S.; Halligan, S.; Punwani, S. Multiparametric MRI for detection of radiorecurrent prostate cancer: Added value of apparent diffusion coefficient maps and dynamic contrast-enhanced images. Prostate Cancer Prostatic Dis. 2015, 18, 128–136. [Google Scholar] [CrossRef]
- Johnson, D.C.; Reiter, R.E. Multi-parametric magnetic resonance imaging as a management decision tool. Transl. Androl. Urol. 2017, 6, 472–482. [Google Scholar] [CrossRef]
- Wallitt, K.L.; Khan, S.R.; Dubash, S.; Tam, H.H.; Khan, S.; Barwick, T.D. Clinical PET Imaging in Prostate Cancer. Radiographics 2017, 37, 1512–1536. [Google Scholar] [CrossRef] [PubMed]
- Farolfi, A.; Ceci, F.; Castellucci, P.; Graziani, T.; Siepe, G.; Lambertini, A.; Schiavina, R.; Lodi, F.; Morganti, A.G.; Fanti, S. 68Ga-PSMA-11 PET/CT in prostate cancer patients with biochemical recurrence after radical prostatectomy and PSA. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Nye, J.A.; Schuster, D.M.; Yu, W.; Camp, V.M.; Goodman, M.M.; Votaw, J.R. Biodistribution and radiation dosimetry of the synthetic nonmetabolized amino acid analogue anti-18F-FACBC in humans. J. Nucl. Med. 2007, 48, 1017–1020. [Google Scholar] [CrossRef] [PubMed]
- Sörensen, J.; Owenius, R.; Lax, M.; Johansson, S. Regional distribution and kinetics of [18F] fluciclovine (anti-[18F]FACBC), a tracer of amino acid transport, in subjects with primary prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Bach-Gansmo, T.; Nanni, C.; Nieh, P.T.; Zanoni, L.; Bogsrud, T.V.; Sletten, H.; Korsan, K.A.; Kieboom, J.; Tade, F.I.; Odewole, O.; et al. Multisite Experience of the Safety, Detection Rate and Diagnostic Performance of Fluciclovine (18F) Positron Emission Tomography/Computerized Tomography Imaging in the Staging of Biochemically Recurrent Prostate Cancer. J. Urol. 2017, 197, 676–683. [Google Scholar] [CrossRef]
- Ren, J.; Yuan, L.; Wen, G.; Yang, J. The value of anti-1-amino-3-18F-fluorocyclobutane-1-carboxylic acid PET/CT in the diagnosis of recurrent prostate carcinoma: A meta-analysis. Actaradiologica 2016, 57, 487–493. [Google Scholar] [CrossRef]
- Yu, C.-Y.; Desai, B.; Ji, L.; Groshen, S.; Jadvar, H. Comparative performance of PET tracers in biochemical recurrence of prostate cancer: A critical analysis of literature. Am. J. Nucl. Med. Mol. Imaging 2014, 4, 580. [Google Scholar]
- DA Approves 18F-Fluciclovine and 68Ga-DOTATATE Products. Available online: http://jnm.snmjournals.org/content/57/8/9N.full.pdf (accessed on 1 September 2019).
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Whiting, P.F.; Rutjes, A.W.S.; Westwood, M.E.; Mallett, S.; Deeks, J.J.; Reitsma, J.B.; Leeflang, M.M.G.; Sterne, J.A.C.; Bossuyt, P.M.M.; QUADAS-2 Group. QUADAS-2: A revised tool for the quality assessment of diagnostic accuracy studies. Ann. Intern. Med. 2011, 155, 529–536. [Google Scholar] [CrossRef]
- Wallace, B.C.; Schmid, C.H.; Lau, J.; Trikalinos, T.A. Meta-Analyst: Software for meta-analysis of binary, continuous and diagnostic data. BMC Med. Res. Methodol. 2009, 9, 80. [Google Scholar] [CrossRef] [PubMed]
- Schuster, D.M.; Votaw, J.R.; Nieh, P.T.; Yu, W.; Nye, J.A.; Master, V.; Bowman, F.D.; Issa, M.M.; Goodman, M.M. Initial experience with the radiotracer anti-1-amino-3-18F-fluorocyclobutane-1-carboxylic acid with PET/CT in prostate carcinoma. J. Nucl. Med. 2007, 48, 56–63. [Google Scholar] [PubMed]
- Schuster, D.M.; Savir-Baruch, B.; Nieh, P.T.; Master, V.A.; Halkar, R.K.; Rossi, P.J.; Lewis, M.M.; Nye, J.A.; Yu, W.; Bowman, F.D.; et al. Detection of recurrent prostate carcinoma with anti-1-amino-3-18F-fluorocyclobutane-1-carboxylic acid PET/CT and 111In-capromab pendetide SPECT/CT. Radiology 2011, 259, 852–861. [Google Scholar] [CrossRef] [PubMed]
- Turkbey, B.; Mena, E.; Shih, J.; Pinto, P.A.; Merino, M.J.; Lindenberg, M.L.; Bernardo, M.; McKinney, Y.L.; Adler, S.; Owenius, R.; et al. Localized prostate cancer detection with 18F FACBC PET/CT: Comparison with MR imaging and histopathologic analysis. Radiology 2014, 270, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Kairemo, K.; Rasulova, N.; Partanen, K.; Joensuu, T. Preliminary clinical experience of trans-1-Amino-3-(18)F-fluorocyclobutanecarboxylic Acid (anti-(18)F-FACBC) PET/CT imaging in prostate cancer patients. BioMed Res. Int. 2014, 2014, 305182. [Google Scholar] [CrossRef] [PubMed]
- Nanni, C.; Schiavina, R.; Brunocilla, E.; Borghesi, M.; Ambrosini, V.; Zanoni, L.; Gentile, G.; Vagnoni, V.; Romagnoli, D.; Martorana, G.; et al. 18F-FACBC compared with 11C-choline PET/CT in patients with biochemical relapse after radical prostatectomy: A prospective study in 28 patients. Clin. Genitourin. Cancer 2014, 12, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Nanni, C.; Schiavina, R.; Brunocilla, E.; Boschi, S.; Borghesi, M.; Zanoni, L.; Pettinato, C.; Martorana, G.; Fanti, S. 18F-Fluciclovine PET/CT for the Detection of Prostate Cancer Relapse: A Comparison to 11C-Choline PET/CT. Clin. Nucl. Med. 2015, 40, e386–e391. [Google Scholar] [CrossRef] [PubMed]
- Odewole, O.A.; Tade, F.I.; Nieh, P.T.; Savir-Baruch, B.; Jani, A.B.; Master, V.A.; Rossi, P.J.; Halkar, R.K.; Osunkoya, A.O.; Akin-Akintayo, O.; et al. Recurrent prostate cancer detection with anti-3-[(18)F]FACBC PET/CT: Comparison with CT. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1773–1783. [Google Scholar] [CrossRef] [PubMed]
- Akin-Akintayo, O.O.; Jani, A.B.; Odewole, O.; Tade, F.I.; Nieh, P.T.; Master, V.A.; Bellamy, L.M.; Halkar, R.K.; Zhang, C.; Chen, Z.; et al. Change in Salvage Radiotherapy Management Based on Guidance With FACBC (Fluciclovine) PET/CT in Postprostatectomy Recurrent Prostate Cancer. Clin. Nucl. Med. 2017, 42, e22–e28. [Google Scholar] [CrossRef]
- Selnæs, K.M.; Krüger-Stokke, B.; Elschot, M.; Willoch, F.; Størkersen, Ø.; Sandsmark, E.; Moestue, S.A.; Tessem, M.-B.; Halvorsen, D.; Kjøbli, E.; et al. 18F-Fluciclovine PET/MRI for preoperative lymph node staging in high-risk prostate cancer patients. Eur. Radiol. 2018, 28, 3151–3159. [Google Scholar] [CrossRef]
- Jambor, I.; Kuisma, A.; Kähkönen, E.; Kemppainen, J.; Merisaari, H.; Eskola, O.; Teuho, J.; Perez, I.M.; Pesola, M.; Aronen, H.J.; et al. Prospective evaluation of 18F-FACBC PET/CT and PET/MRI versus multiparametric MRI in intermediate- to high-risk prostate cancer patients (FLUCIPRO trial). Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Akin-Akintayo, O.; Tade, F.; Mittal, P.; Moreno, C.; Nieh, P.T.; Rossi, P.; Patil, D.; Halkar, R.; Fei, B.; Master, V.; et al. Prospective evaluation of fluciclovine (18F) PET-CT and MRI in detection of recurrent prostate cancer in non-prostatectomy patients. Eur. J. Radiol. 2018, 102, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Andriole, G.L.; Kostakoglu, L.; Chau, A.; Duan, F.; Mahmood, U.; Mankoff, D.A.; Schuster, D.M.; Siegel, B.A.; LOCATE Study Group. The Impact of Positron Emission Tomography with 18F-Fluciclovine on the Treatment of Biochemical Recurrence of Prostate Cancer: Results from the LOCATE Trial. J. Urol. 2019, 201, 322–331. [Google Scholar] [CrossRef] [PubMed]
- England, J.R.; Paluch, J.; Ballas, L.K.; Jadvar, H. 18F-Fluciclovine PET/CT Detection of Recurrent Prostate Carcinoma in Patients With Serum PSA ≤ 1 ng/mL After Definitive Primary Treatment. Clin. Nucl. Med. 2019, 44, e128–e132. [Google Scholar] [CrossRef]
- Suzuki, H.; Jinnouchi, S.; Kaji, Y.; Kishida, T.; Kinoshita, H.; Yamaguchi, S.; Tobe, T.; Okamura, T.; Kawakita, M.; Furukawa, J.; et al. Diagnostic performance of 18F-fluciclovine PET/CT for regional lymph node metastases in patients with primary prostate cancer: A multicenter phase II clinical trial. Jpn. J. Clin. Oncol. 2019. Epub ahead of print. Available online: https://academic.oup.com/jjco/advance-article/doi/10.1093/jjco/hyz072/5490163 (accessed on 1 September 2019).
- Mottet, N.; Bellmunt, J.; Bolla, M.; Briers, E.; Cumberbatch, M.G.; De Santis, M.; Fossati, N.; Gross, T.; Henry, A.M.; Joniau, S.; et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol. 2017, 71, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Pernthaler, B.; Kulnik, R.; Gstettner, C.; Salamon, S.; Aigner, R.M.; Kvaternik, H. A Prospective Head-to-Head Comparison of 18F-Fluciclovine with 68Ga-PSMA-11 in Biochemical Recurrence of Prostate Cancer in PET/CT. Clin. Nucl. Med. 2019. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Calais, J.; Ceci, F.; Eiber, M.; Hope, T.A.; Hofman, M.S.; Rischpler, C.; Bach-Gansmo, T.; Nanni, C.; Savir-Baruch, B.; Elashoff, D.; et al. 18F-fluciclovine PET-CT and 68Ga-PSMA-11 PET-CT in patients with early biochemical recurrence after prostatectomy: A prospective, single-centre, single-arm, comparative imaging trial. Lancet Oncol. 2019. Epub ahead of print. [Google Scholar] [CrossRef]
- Lawhn-Heath, C.; Flavell, R.R.; Behr, S.C.; Yohannan, T.; Greene, K.L.; Feng, F.; Carroll, P.R.; Hope, T.A. SingleCenter Prospective Evaluation of 68Ga-PSMA-11 PET in Biochemical Recurrence of Prostate Cancer. AJR Am. J. Roentgenol. 2019, 1–8, Epub ahead of print. [Google Scholar] [CrossRef]
- Evangelista, L.; Guttilla, A.; Zattoni, F.; Muzzio, P.C.; Zattoni, F. Utility of choline positron emission tomography/computed tomography for lymph node involvement identification in intermediate- to high-risk prostate cancer: A systematic literature review and meta-analysis. Eur. Urol. 2013, 63, 1040–1048. [Google Scholar] [CrossRef]
- Yaxley, J.W.; Raveenthiran, S.; Nouhaud, F.X.; Samartunga, H.; Yaxley, A.J.; Coughlin, G.; Delahunt, B.; Egevad, L.; McEwan, L.; Wong, D. Outcomes of Primary Lymph Node Staging of Intermediate and High Risk Prostate Cancer with 68 Ga-PSMA Positron Emission Tomography/Computerized Tomography Compared to Histological Correlation of Pelvic Lymph Node Pathology. J. Urol. 2019, 201, 815–820. [Google Scholar] [CrossRef]
- Oka, S.; Hattori, R.; Kurosaki, F.; Toyama, M.; Williams, L.A.; Yu, W.; Votaw, J.R.; Yoshida, Y.; Goodman, M.M.; Ito, O. A preliminary study of Anti-1-amino-3-18F-fluorocyclobutyl-1-carboxylic acid for the detection of prostate cancer. J. Nucl. Med. 2007, 48, 46–55. [Google Scholar] [PubMed]

| Study Characteristics | Patient Characteristics | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Authors | Year | Journal | Country | Study Design | Setting | N. pts | Mean Age (Range) | Gleason Score (n) | Type of Treatment (n) | Mean PSA (Range) | Mean PSA Doubling Time (Range) |
| Schuster et al. [23] | 2007 | JNM | USA | Prospective | Staging (n = 9) Restaging (n = 6) | 15 | 62y (45–76) | 6 (2) 7 (2) 8 (2) 9 (2) 10 (1) NA (6) | SP (1), BCT + RT + CTR (1), BCT (2), BCT + RT (1), RP + RT (1), naive (9) | 15 ng/mL (1.9–71) | NA |
| Schuster et al. [24] | 2011 | Radiology | USA | Prospective | Restaging | 50 | 68.3y (50–90) | NA | RP (13), CTR, HFUS, EBRT, and/or BCT (37) | 6.62 ng/mL (0.11–44.74) | NA |
| Turkbey et al. [25] | 2014 | Radiology | USA | Prospective | Staging | 21 | 62y (44–73) | 6 (3) 7 (12) 8 (5) 9 (1) | RARP + LND (21) | 13.5 ng/mL (3.55–37.3) | NA |
| Kairemo et al. [26] | 2014 | BioMed Research Intern | Finland | Retrospective | Restaging | 26 * | 68.1y (56–77) | 5 (3) 6 (7) 7 (7) 8 (3) 9 (5) | RP + RT (12), RT (13), ADT (20), BT (11), CHT(5), 153Sm-EDTMP (7), Denosumab (1) | 7.9 ng/mL (0.11–69) | positive FACBC 3.2mo (0.3–6) negative FACBC 31.2mo (8–84) |
| Nanni et al. [27] | 2014 | ClinGenitourin Cancer | Italy | Prospective | Restaging | 28 | 67y (55–78) | 6 (1) 7 (16) 8 (6) 9 (4) 10 (1) | RP (28), RT (11), ADT (14) | 2.9 ng/mL (0.2–14.6) | NA |
| Nanni et al. [28] | 2015 | ClinNucl Med | Italy | Prospective | Restaging | 50 | 67y (55–78) | ≤6 (4) 7 (31) 8–10 (15) | RP (50), RT (23), ADT (21) | 3.2 ng/mL (0.24–15.6) | NA |
| Odewole et al. [29] | 2016 | EJNMMI | USA | Retrospective | Staging | 53 | 67.57y (49–90) | 7 (49) NA (4) | RP (7), EBRT (5), BCT (6), CTR (4), HT (1), 2 or more treatment (30) | 7.2 ng/mL (0.11–44.8) | 18.6mo ## (−31.6–357.8) |
| Bach-Gansmo et al. [15] | 2017 | J Urol | Norway Italy USA | Retrospective | Restaging | 596 | 67y (42–90) | 6.7 (110) § 7.4 (355) §§ | RP (130), RP + other but no RT (62), RT (76), RT + other (266), other but no RT/RP (41) | 5.43 ng/mL (0.05–82.0) | NA |
| Akin-Akintayo et al. [30] | 2017 | ClinNucl Med | USA | Prospective | Restaging | 42 | 62y (42–75) | 7 (42) # | RP (42) | 2.1 ng/mL (0.07–11.15) | NA |
| Selnaes et al. [31] | 2018 | EurRadiol | Norway | Prospective | Staging | 26 | 66.2y (55–71.9) | 7 (11) 8 (8) 9 (7) | RARP + LND (26) | 14.6 ng/mL (3.7–56.9) | NA |
| Jambor et al. [32] | 2018 | EJNMMI | Finland | Prospective | Staging | 26 | 65y ** (49–76) | 6 (1) 7 (17) 8 (2) 9 (6) | RARP + LND (26) | 12 ng/mL (4.1–35) | NA |
| Akin-Akintayo et al. [33] | 2018 | Eur J Radiol | USA | Prospective | Staging | 24 | 70.8y (60–83) | 7 (24) # | BCT (3), RT (3), PT (1), CTR (1), CTR + HT (1), BCT + other treatment but no RP (13), other treatment but no BCT (2) | 8.5 ng/mL (2.2–29.3) | NA |
| Andriole et al. [34] | 2019 | J Urol | USA | Prospective | Restaging | 213 | 66.4y (46–90) | ≤6 (27) 7 (134) ≥8 (50) NA (2) | RP (121), RP + RT (43), EBRT (21), BCT (1), EBRT + BCT (2), EBRT + ADT (17), EBRT + CTR (2), CTR (1), BCT + ADT (1), EBRT + BCT + ADT (2), HIFU (1), High-dose BCT (1) | 4.24 ng/mL (0.2–93.5) | NA |
| England et al. [35] | 2019 | Clin Nucl Med | USA | Retrospective | Restaging | 28 | 67.1y (53–77) | 7 (19) 8 (3) 9 (6) | Primary treatment RP (22), RP+ EBRT (3), RP + EBRT + ADT (1), EBRT + ADT (2) Salvage therapy RT (6), ADT (1), RT + ADT (1), LND (1) | 0.44 ng/mL (0.1–1.0) | 6.38mo (1.6–16.8) |
| Suzuki et al. [36] | 2019 | Japanese J Clin Oncol | Japan | Prospective | Staging | 28 | 67.9 (57–77) | <6 (1) 7 (12) 8 (8) 9 (8) | RARP + LND (28) | 17.94 ng/mL (1.20–82.38) | NA |
| Author, (Ref) | Year | Journal | Country | N pts | Outcome | DR | TP | TN | FP | FN |
|---|---|---|---|---|---|---|---|---|---|---|
| Schuster et al. [23] | 2007 | JNM | USA | 9 | Accuracy LN (patient-based) | NA | 2 | 5 | 0 | 2 |
| Schuster et al. [24] | 2011 | Radiology | USA | 50 | Accuracy (PB) FACBC (region-based) | NA | 32 | 8 | 4 | 4 |
| Acc (extra-p) FACBC (region-based) | 10 | 7 | 0 | 0 | ||||||
| Acc (PB) Capromab (region-based) | 25 | 7 | 5 | 11 | ||||||
| Ac (extra-p) Capromab (region-based) | 1 | 7 | 0 | 9 | ||||||
| Turkbey et al. [25] | 2014 | Radiology | USA | 21 | DR for primary | 19/21 (90%) | ||||
| Lesion-based | 33 | 0 | 38 | 15 | ||||||
| Accuracy MRI (les-based) | 34 | 0 | 21 | 14 | ||||||
| Kairemo et al. [26] | 2014 | BioMed Research Intern | Finland | 26 ** | DR | 17/26 (65%) | ||||
| Patient-based | 11 | 12 | 3 | 0 | ||||||
| Nanni et al. [27] | 2014 | ClinGenitourin Cancer | Italy | 28 | DR (comparison with Choline) | 10/28 (36%) | NA | NA | NA | NA |
| Nanni et al. [28] | 2015 | ClinNucl Med | Italy | 50 | DR (comparison with Choline) | 17/50 (34%) | NA | NA | NA | NA |
| Odewole et al. [29] | 2016 | EJNMMI | USA | 53 | DR (all PSA levels and clinical data) | 41/53 (77.4%) | ||||
| Accuracy (PB) FACBC | 31 | 9 | 7 | 4 | ||||||
| Accuracy (PB) CT | 4 | 14 | 2 | 31 | ||||||
| Accuracy (extra-pr) FACBC | 12 | 15 | 0 | 15 | ||||||
| Accuracy (extra-pr) CT | 3 | 15 | 0 | 23 | ||||||
| Bach-Gasmo et al. [15] | 2017 | J Urol | Norway Italy USA | 596 | DR | 403/595 (67.7%) | ||||
| Lesion-based | 153 | 216 | 93 | 91 | ||||||
| Region-based (PB) | 74 | 14 | 20 | 10 | ||||||
| Region-based (Extra-prost) | 36 | 1 | 3 | 4 | ||||||
| Patient-based | 98 | 14 | 21 | 10 | ||||||
| Akin-Akintayo et al. [30] | 2017 | ClinNucl Med | USA | 42 | DR (change in radiotherapy strategy) | 34/42 (81%) | NA | NA | NA | NA |
| Selnaes et al. [31] | 2018 | EurRadiol | Norway | 26 | Accuracy for LN | NA | ||||
| Patient-based | NA | 4 | 16 | 0 | 6 | |||||
| Region-based | NA | 6 | 185 | 0 | 14 | |||||
| Jambor et al. [32] | 2018 | EJNMMI | Finland | 26 | Accuracy LN | NA | ||||
| Patient-based | 7 | 19 | 0 | 0 | ||||||
| Region-based | NA | NA | NA | NA | ||||||
| Akin-Akintayo et al. [33] | 2018 | Eur J Radiol | USA | 24 | Accuracy (PB) FACBC * | NA | 13 | 1 | 8 | 0 |
| Accuracy (PB) MRI * | 5 | 5 | 4 | 8 | ||||||
| Accuracy (extra-p) FACBC | 7 | 9 | 1 | 1 | ||||||
| Accuracy (extra-p) MRI * | 4 | 7 | 3 | 4 | ||||||
| Andriole et al. [34] | 2019 | J Urol | USA | 213 | DR (also for PSA level) | 122/213 (57%) | NA | NA | NA | NA |
| England et al. [35] | 2019 | ClinNucl Med | USA | 28 | DR (for site and clinical data) | 13/28 (46%) | NA | NA | NA | NA |
| Suzuki et al. [36] | 2019 | Japanese J ClinOncol | Japan | 28 | Accuracy LN | NA | ||||
| Patient-based | 4 | 19 | 3 | 2 | ||||||
| Lesion-based | 4 | 28 | 5 | 3 |
| Type of Analysis | Study Name (Year), Ref | Setting (Site) | TP | FN | TN | FP | Sensitivity | Specificity |
|---|---|---|---|---|---|---|---|---|
| Patient-based analysis | Suzuki et al. (2019), [36] | Staging (LN) | 4 | 2 | 19 | 3 | 66.6% | 86.3% |
| Selnaes et al. (2018), [31] | Staging (LN) | 4 | 6 | 16 | 0 | 45% | 80.8% | |
| Jambor et al. (2018), [32] | Staging (primary) | 7 | 0 | 19 | 0 | 70.6% | 82.8% | |
| Bach-Gasmo et al. (2017), [15] | Restaging (all) | 98 | 10 | 14 | 21 | 90.7% | 40% | |
| Kairemo et al. (2014), [26] | Restaging (all) | 11 | 0 | 12 | 3 | 76.2% | 68% | |
| Schuster et al. (2007), [23] | Staging/restaging (all) | 2 | 2 | 5 | 0 | 50% | 66.7% | |
| Region-based analysis (PB) | Schuster et al. (2011), [24] | Restaging | 32 | 4 | 8 | 4 | 88.9% | 66.7% |
| Bach-Gasmo et al. (2017), [15] | Restaging | 74 | 10 | 14 | 20 | 88.1% | 41.2% | |
| Akin-Akintayo et al. (2018), [33] | Staging | 13 | 0 | 1 | 8 | 78.3% | 31.6% | |
| Odewole et al. (2016), [29] | Staging | 31 | 4 | 9 | 7 | 88.6% | 56.3% | |
| Region-based analysis (extra-PB) | Schuster et al. (2011), [24] | Restaging | 10 | 0 | 7 | 0 | 75% | 70.6% |
| Bach-Gasmo et al. (2017), [15] | Restaging | 36 | 4 | 1 | 3 | 90% | 25% | |
| Akin-Akintayo et al. (2018), [33] | Staging | 7 | 1 | 9 | 1 | 87.5% | 90% | |
| Odewole et al. (2016), [29] | Staging | 12 | 15 | 15 | 0 | 45.9% | 80% |
| Meta-Analysis Results | Patient-Based Analysis (95% CI) | Region-Based Analysis (PB) (95% CI) | Region-Based Analysis (ex-PB) (95% CI) | |||
|---|---|---|---|---|---|---|
| Value | I2 | Value | I2 | Value | I2 | |
| Pooled sensitivity, % | 86.3% (79.6–91.4%) | 78.6% | 90.4% (84.8–94.4%) | 22.1% | 76.5% (66–85%) | 87.3% |
| Pooled specificity, % | 75.9% (66.9–83.5%) | 88.7% | 45.1% (33.2–57.3%) | 63.3% | 88.9% (73.9–96.9%) | 78.7% |
| DOR | 16.453 (5.241–51.646) | 29.9% | 8.026 (3.841–16.769) | 3.5% | 24.820 (3.777–163.12) | 36% |
| LR+ | 4.557 (1.685–12.324) | 72.9% | 1.598 (1.088–2.349) | 70% | 6.024 (0.568–63.943) | 85.6% |
| LR− | 0.337 (0.166–0.681) | 63.6% | 0.221 (0.130–0.375) | 0% | 0.251 (0.058–1.090) | 71.6% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laudicella, R.; Albano, D.; Alongi, P.; Argiroffi, G.; Bauckneht, M.; Baldari, S.; Bertagna, F.; Boero, M.; Vincentis, G.D.; Sole, A.D.; et al. 18F-Facbc in Prostate Cancer: A Systematic Review and Meta-Analysis. Cancers 2019, 11, 1348. https://doi.org/10.3390/cancers11091348
Laudicella R, Albano D, Alongi P, Argiroffi G, Bauckneht M, Baldari S, Bertagna F, Boero M, Vincentis GD, Sole AD, et al. 18F-Facbc in Prostate Cancer: A Systematic Review and Meta-Analysis. Cancers. 2019; 11(9):1348. https://doi.org/10.3390/cancers11091348
Chicago/Turabian StyleLaudicella, Riccardo, Domenico Albano, Pierpaolo Alongi, Giovanni Argiroffi, Matteo Bauckneht, Sergio Baldari, Francesco Bertagna, Michele Boero, Giuseppe De Vincentis, Angelo Del Sole, and et al. 2019. "18F-Facbc in Prostate Cancer: A Systematic Review and Meta-Analysis" Cancers 11, no. 9: 1348. https://doi.org/10.3390/cancers11091348
APA StyleLaudicella, R., Albano, D., Alongi, P., Argiroffi, G., Bauckneht, M., Baldari, S., Bertagna, F., Boero, M., Vincentis, G. D., Sole, A. D., Rubini, G., Fantechi, L., Frantellizzi, V., Ganduscio, G., Guglielmo, P., Nappi, A. G., Evangelista, L., & on the behalf of Young AIMN Working Group. (2019). 18F-Facbc in Prostate Cancer: A Systematic Review and Meta-Analysis. Cancers, 11(9), 1348. https://doi.org/10.3390/cancers11091348










