Radiation Tolerance in Tardigrades: Current Knowledge and Potential Applications in Medicine
Abstract
1. Introduction
2. The Patterns of Tolerance to Radiation in Tardigrades
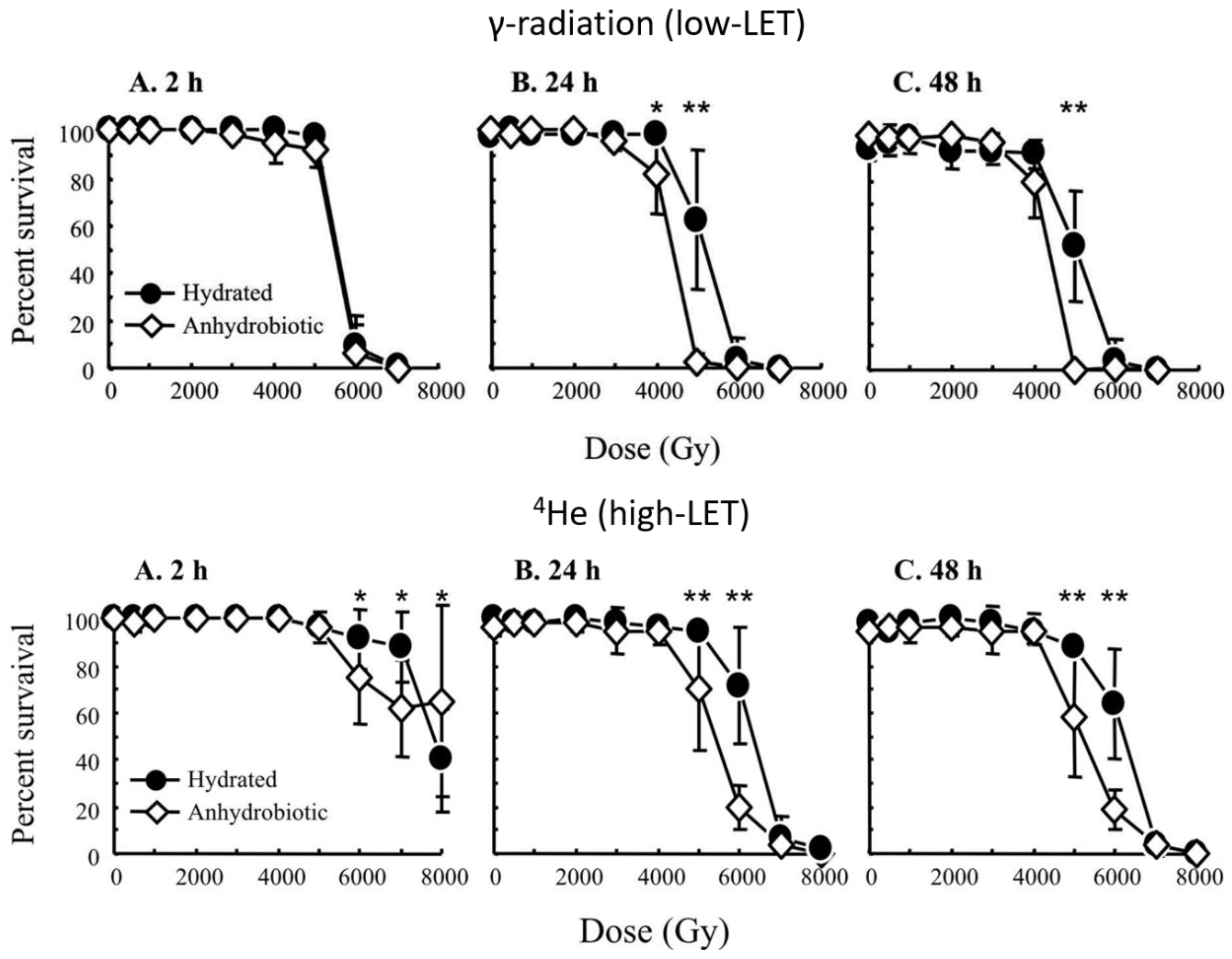
2.1. Low Linear Energy Transfer (Low-LET) Irradiation
2.2. High Linear Energy Transfer (High-LET) Irradiation
2.3. Irradiation with UV
2.4. Effects on Fertility and Hatchability of Laid Eggs
2.5. Direct Irradiation of Early Developmental Stages
3. Molecular Mechanisms of Radiation and Desiccation Tolerance in Tardigrades
3.1. Damage Induced by Radiation
3.2. Molecular Responses to Radiation
3.3. Molecular Mechanisms of Desiccation Tolerance
3.4. Cross-Tolerance between Desiccation and Radiation
4. Relevance of Tardigrade Stress Mechanisms for Research on Cancer and Other Medical Fields
4.1. Preservation of Cells, Tissues and Bioreagents in the Dry State
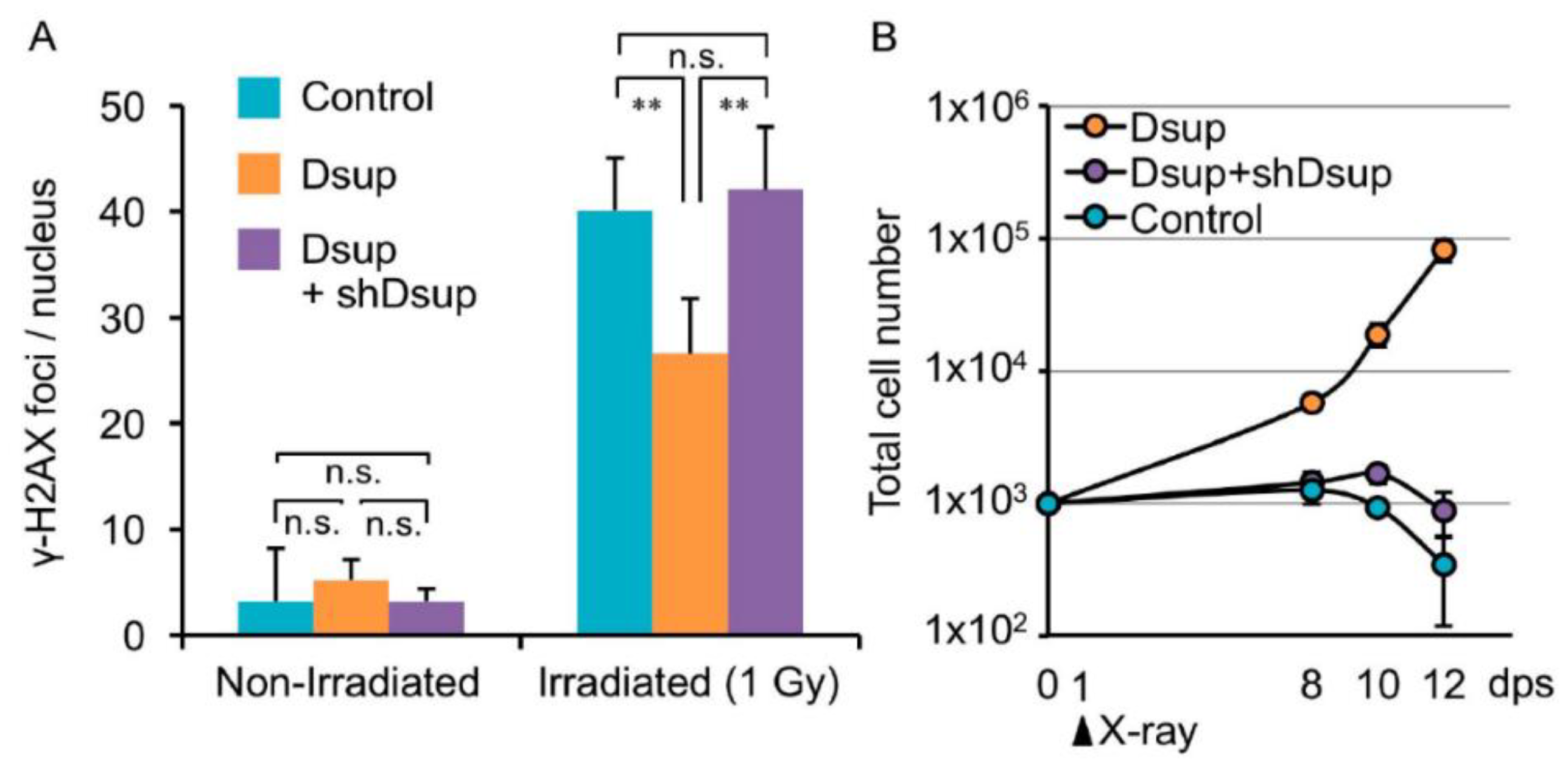
4.2. Cell and Genomic Integrity—A Field of Common Interest in Research on Tardigrades and Cancer
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Degma, P.; Bertolani, R.; Guidetti, R. Actual Checklist of Tardigrada Species (2009–2018, 34th Edition: 30-06-2018). Available online: http://www.tardigrada.modena.unimo.it/miscellanea/Actual%20checklist%20of%20Tardigrada.pdf (accessed on 5 July 2019).
- Guidetti, R.; Bertolani, R. Tardigrade taxonomy: An updated check list of the taxa and a list of characters for their identification. Zootaxa 2005, 845, 1–46. [Google Scholar] [CrossRef]
- Degma, P.; Guidetti, R. Notes to the current checklist of Tardigrada. Zootaxa 2007, 1579, 41–53. [Google Scholar] [CrossRef]
- Jørgensen, A.; Kristensen, R.M.; Møbjerg, N. Phylogeny and Integrative Taxonomy of Tardigrada. In Water Bears: The Biology of Tardigrades; Schill, R.O., Ed.; Springer: Cham, Switzerland, 2018; pp. 95–114. [Google Scholar]
- Yoshida, Y.; Koutsovoulos, G.; Laetsch, D.R.; Stevens, L.; Kumar, S.; Horikawa, D.D.; Ishino, K.; Komine, S.; Kunieda, T.; Tomita, M.; et al. Comparative genomics of the tardigrades Hypsibius dujardini and Ramazzottius varieornatus. PLoS Biol. 2017, 15, e2002266. [Google Scholar] [CrossRef] [PubMed]
- Gąsiorek, P.; Stec, D.; Morek, W.; Michalczyk, Ł. An integrative redescription of Hypsibius dujardini (Doyere, 1840), the nominal taxon for Hypsibioidea (Tardigrada: Eutardigrada). Zootaxa 2018, 4415, 45–75. [Google Scholar] [CrossRef] [PubMed]
- Koutsovoulos, G.; Kumar, S.; Laetsch, D.R.; Stevens, L.; Daub, J.; Conlon, C.; Maroon, H.; Thomas, F.; Aboobaker, A.A.; Blaxter, M. No evidence for extensive horizontal gene transfer in the genome of the tardigrade Hypsibius dujardini. Proc. Natl. Acad. Sci. USA 2016, 113, 5053–5058. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Horikawa, D.D.; Saito, Y.; Kuwahara, H.; Kozuka-Hata, H.; Shin-I, T.; Minakuchi, Y.; Ohishi, K.; Motoyama, A.; Aizu, T.; et al. Extremotolerant tardigrade genome and improved radiotolerance of human cultured cells by tardigrade-unique protein. Nat. Commun. 2016, 7, 12808. [Google Scholar] [CrossRef]
- Schill, R.O.; Hengherr, S. Environmental Adaptations: Desiccation Tolerance. In Water Bears: The Biology of Tardigrades; Schill, R.O., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 273–293. [Google Scholar]
- Hengherr, S.; Schill, R.O. Environmental Adaptations: Cryobiosis. In Water Bears: The Biology of Tardigrades; Schill, R.O., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 295–310. [Google Scholar]
- Keilin, D. The Leeuwenhoek Lecture. The problem of anabiosis or latent life: History and current concept. Proc. R. Soc. Lond. Ser. B 1959, 150, 149–191. [Google Scholar]
- Kikawada, T.; Minakawa, N.; Watanabe, M.; Okuda, T. Factors Inducing Successful Anhydrobiosis in the African Chironomid Polypedilum vanderplanki: Significance of the Larval Tubular Nest. Integr. Comp. Biol. 2005, 45, 710–714. [Google Scholar] [CrossRef]
- Lavens, P.; Sorgeloos, P. The cryptobiotic state of Artemia cysts, its diapause deactivation and hatching: A review. In Artemia Research and its Applications, Vol. 3, Ecology, Culturing, Use in Agriculture; Sorgeloos, P., Bengtson, D.A., Decleir, W., Jaspers, E., Eds.; Universa Press: Wetteren, Belgium, 1987; pp. 27–63. [Google Scholar]
- Schill, R.O.; Mali, B.; Dandekar, T.; Schnolzer, M.; Reuter, D.; Frohme, M. Molecular mechanisms of tolerance in tardigrades: New perspectives for preservation and stabilization of biological material. Biotech. Adv. 2009, 27, 348–352. [Google Scholar] [CrossRef]
- Crowe, J.H. Anhydrobiosis: An unsolved problem with applications in human welfare. In Membrane Hydration: The Role of Water in the Structure and Function of Biological Membranes; Disalvo, E.A., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 263–280. [Google Scholar]
- Jönsson, K.I.; Levine, E.B.; Wojcik, A.; Haghdoost, S.; Harms-Ringdahl, M. Environmental Adaptations: Radiation Tolerance. In Water Bears: The Biology of Tardigrades; Schill, R.O., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 311–330. [Google Scholar]
- Gladyshev, E.; Meselson, M. Extreme resistance of bdelloid rotifers to ionizing radiation. Proc. Natl. Acad. Sci. USA 2008, 105, 5139–5144. [Google Scholar] [CrossRef]
- Iwasaki, T. Sensitivity of Artemia Eggs to the γ-irradiation: I. Hatchability of Encysted Dry Eggs. J. Radiat. Res. 1964, 5, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Nakahara, Y.; Sakashita, T.; Kikawada, T.; Fujita, A.; Hamada, N.; Horikawa, D.D.; Wada, S.; Kobayashi, Y.; Okuda, T. Physiological changes leading to anhydrobiosis improve radiation tolerance in Polypedilum vanderplanki larvae. J. Insect Physiol. 2007, 53, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Mattimore, V.; Battista, J.R. Radioresistance of Deinococcus radiodurans: Functions necessary to survive ionizing radiation are also necessary to survive prolonged desiccation. J. Bacteriol. 1996, 178, 633–637. [Google Scholar] [CrossRef] [PubMed]
- Beblo-Vranesevic, K.; Bohmeier, M.; Perras, A.K.; Schwendner, P.; Rabbow, E.; Moissl-Eichinger, C.; Cockell, C.S.; Vannier, P.; Marteinsson, V.T.; Monaghan, E.P.; et al. Lack of correlation of desiccation and radiation tolerance in microorganisms from diverse extreme environments tested under anoxic conditions. FEMS Microbiol. Lett. 2018, 365. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, K.I. Causes and Consequences of Excess Resistance in Cryptobiotic Metazoans. Physiol. Biochem. Zool. 2003, 76, 429–435. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jönsson, K.I. Tardigrades as a potential model organism in space research. Astrobiology 2007, 7, 757–766. [Google Scholar] [CrossRef]
- Greven, H. From Johann August Ephraim Goeze to Ernst Marcus: A Ramble Through the History of Early Tardigrade Research (1773 Until 1929). In Water Bears: The Biology of Tardigrades; Schill, R.O., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 1–55. [Google Scholar]
- May, R.M.; Maria, M.; Guimard, J. Actions différentielles des rayons x et Ultraviolets sur le tardigrade Macrobiotus areolatus, à l´état actif et desséché. Bull. Biol. Fr. Belg. 1964, 98, 349–367. [Google Scholar]
- Jönsson, K.I.; Harms-Ringdahl, M.; Torudd, J. Radiation tolerance in the eutardigrade Richtersius coronifer. Int. J. Radiat. Biol. 2005, 81, 649–656. [Google Scholar] [CrossRef]
- Horikawa, D.D.; Sakashita, T.; Katagiri, C.; Watanabe, M.; Kikawada, T.; Nakahara, Y.; Hamada, N.; Wada, S.; Funayama, T.; Higashi, S.; et al. Radiation tolerance in the tardigrade Milnesium tardigradum. Int. J. Radiat. Biol. 2006, 82, 843–848. [Google Scholar] [CrossRef]
- Beltrán-Pardo, E.; Jönsson, K.I.; Harms-Ringdahl, M.; Haghdoost, S.; Wojcik, A. Tolerance to Gamma Radiation in the Tardigrade Hypsibius dujardini from Embryo to Adult Correlate Inversely with Cellular Proliferation. PLoS ONE 2015, 10, e0133658. [Google Scholar] [CrossRef]
- Hashimoto, T.; Kunieda, T. DNA Protection Protein, a Novel Mechanism of Radiation Tolerance: Lessons from Tardigrades. Life 2017, 7, 26. [Google Scholar] [CrossRef]
- Jönsson, K.I.; Hygum, T.L.; Andersen, K.N.; Clausen, L.K.B.; Møbjerg, N. Tolerance to Gamma Radiation in the Marine Heterotardigrade, Echiniscoides sigismundi. PLoS ONE 2016, 11, e0168884. [Google Scholar] [CrossRef]
- Hall, E.J.; Giaccia, A.J. Radiobiology for the Radiologist, 8th ed.; Wolters Kluwer: Philadelphia, PA, USA, 2019; p. 597. [Google Scholar]
- Fernandez, C.; Vasanthan, T.; Kissoon, N.; Karam, G.; Duquette, N.; Seymour, C.; Stone, J.R. Radiation tolerance and bystander effects in the eutardigrade species Hypsibius dujardini (Parachaela: Hypsibiidae). Zool. J. Linn. Soc. 2016, 178, 919–923. [Google Scholar] [CrossRef][Green Version]
- Nilsson, E.J.; Jönsson, K.I.; Pallon, J. Tolerance to proton irradiation in the eutardigrade Richtersius coronifer—A nuclear microprobe study. Int. J. Radiat. Biol. 2010, 86, 420–427. [Google Scholar] [CrossRef]
- Jönsson, K.I.; Wojcik, A. Tolerance to X-rays and Heavy Ions (Fe, He) in the Tardigrade Richtersius coronifer and the Bdelloid Rotifer Mniobia russeola. Astrobiology 2017, 17, 163–167. [Google Scholar] [CrossRef]
- Altiero, T.; Guidetti, R.; Caselli, V.; Cesari, M.; Rebecchi, L. Ultraviolet radiation tolerance in hydrated and desiccated eutardigrades. J. Zool. Syst. Evol. Res. 2011, 49, 104–110. [Google Scholar] [CrossRef]
- Horikawa, D.D.; Cumbers, J.; Sakakibara, I.; Rogoff, D.; Leuko, S.; Harnoto, R.; Arakawa, K.; Katayama, T.; Kunieda, T.; Toyoda, A.; et al. Analysis of DNA repair and protection in the Tardigrade Ramazzottius varieornatus and Hypsibius dujardini after exposure to UVC radiation. PLoS ONE 2013, 8, e64793. [Google Scholar] [CrossRef]
- Jönsson, K.I.; Rabbow, E.; Schill, R.O.; Harms-Ringdahl, M.; Rettberg, P. Tardigrades survive exposure to space in low Earth orbit. Curr. Biol. 2008, 18, R729–R731. [Google Scholar] [CrossRef]
- Jönsson, K.I.; Schill, R.O.; Rabbow, E.; Rettberg, P.; Harms-Ringdahl, M. The fate of the TARDIS offspring: No intergenerational effects of space exposure. Zool. J. Linn. Soc. 2016, 178, 924–930. [Google Scholar] [CrossRef]
- Bergonié, J.; Tribondeau, L. De Quelques Résultats de la Radiothérapie et Essai de Fixation d’une Technique Rationnelle. C. R. Hebd. Séances L’Acad. Sci. 1906, 143, 983–985. [Google Scholar]
- Beltran-Pardo, E.; Jönsson, K.I.; Wojcik, A.; Haghdoost, S.; Harms-Ringdahl, M.; Bermudez-Cruz, R.M.; Bernal Villegas, J.E. Effects of ionizing radiation on embryos of the tardigrade Milnesium cf. tardigradum at different stages of development. PLoS ONE 2013, 8, e72098. [Google Scholar] [CrossRef]
- Jönsson, I.; Beltran-Pardo, E.; Haghdoost, S.; Wojcik, A.; Bermúdez-Cruz, R.M.; Bernal Villegas, J.E.; Harms-Ringdahl, M. Tolerance to gamma-irradiation in eggs of the tardigrade Richtersius coronifer depends on stage of development. J. Limnol. 2013, 72, 73–79. [Google Scholar] [CrossRef]
- Schill, R.O.; Fritz, G.B. Desiccation tolerance in embryonic stages of the tardigrade. J. Zool. 2008, 276, 103–107. [Google Scholar] [CrossRef]
- Horikawa, D.D.; Yamaguchi, A.; Sakashita, T.; Tanaka, D.; Hamada, N.; Yukuhiro, F.; Kuwahara, H.; Kunieda, T.; Watanabe, M.; Nakahara, Y.; et al. Tolerance of anhydrobiotic eggs of the Tardigrade Ramazzottius varieornatus to extreme environments. Astrobiology 2012, 12, 283–289. [Google Scholar] [CrossRef]
- Iwasaki, T. Sensitivity of Artemia eggs to the γ-irradiation. II. Effects of water content. J. Radiat. Res. 1954, 5, 76–81. [Google Scholar] [CrossRef]
- Watanabe, M.; Sakashita, T.; Fujita, A.; Kikawada, T.; Horikawa, D.D.; Nakahara, Y.; Wada, S.; Funayama, T.; Hamada, N.; Kobayashi, Y.; et al. Biological effects of anhydrobiosis in an African chironomid, Polypedilum vanderplanki on radiation tolerance. Int. J. Radiat. Biol. 2006, 82, 587–592. [Google Scholar] [CrossRef]
- Watanabe, M.; Sakashita, T.; Fujita, A.; Kikawada, T.; Nakahara, Y.; Hamada, N.; Horikawa, D.D.; Wada, S.; Funayama, T.; Kobayashi, Y.; et al. Estimation of radiation tolerance to high LET heavy ions in an anhydrobiotic insect, Polypedilum vanderplanki. Int. J. Radiat. Biol. 2006, 82, 835–842. [Google Scholar] [CrossRef]
- Neumann, S.; Reuner, A.; Brummer, F.; Schill, R.O. DNA damage in storage cells of anhydrobiotic tardigrades. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2009, 153, 425–429. [Google Scholar] [CrossRef]
- Kuzmic, M.; Richaud, M.; Cuq, P.; Frelon, S.; Galas, S. Carbonylation accumulation of the Hypsibius exemplaris anhydrobiote reveals age-associated marks. PLoS ONE 2018, 13, e0208617. [Google Scholar] [CrossRef]
- Fedorova, M.; Bollineni, R.C.; Hoffmann, R. Protein carbonylation as a major hallmark of oxidative damage: Update of analytical strategies. Mass Spectrom. Rev. 2014, 33, 79–97. [Google Scholar] [CrossRef]
- Schokraie, E.; Hotz-Wagenblatt, A.; Warnken, U.; Frohme, M.; Dandekar, T.; Schill, R.O.; Schnölzer, M. Investigating heat shock proteins of tardigrades in active versus anhydrobiotic state using shotgun proteomics. J. Zool. Syst. Evol. Res. 2011, 49, 111–119. [Google Scholar] [CrossRef]
- Schokraie, E.; Warnken, U.; Hotz-Wagenblatt, A.; Grohme, M.A.; Hengherr, S.; Forster, F.; Schill, R.O.; Frohme, M.; Dandekar, T.; Schnolzer, M. Comparative proteome analysis of Milnesium tardigradum in early embryonic state versus adults in active and anhydrobiotic state. PLoS ONE 2012, 7, e45682. [Google Scholar] [CrossRef]
- Wang, C.; Grohme, M.A.; Mali, B.; Schill, R.O.; Frohme, M. Towards decrypting cryptobiosis—Analyzing anhydrobiosis in the tardigrade Milnesium tardigradum using transcriptome sequencing. PLoS ONE 2014, 9, e92663. [Google Scholar] [CrossRef]
- Boothby, T.C.; Tapia, H.; Brozena, A.H.; Piszkiewicz, S.; Smith, A.E.; Giovannini, I.; Rebecchi, L.; Pielak, G.J.; Koshland, D.; Goldstein, B. Tardigrades Use Intrinsically Disordered Proteins to Survive Desiccation. Mol. Cell 2017, 65, 975–984. [Google Scholar] [CrossRef]
- Beltràn-Pardo, E.A.; Jönsson, I.; Wojcik, A.; Haghdoost, S.; Bermúdez Cruz, R.M.; Bernal Villegas, J.E. Sequence analysis of the DNA-repair gene rad51 in the tardigrades Milnesium cf. tardigradum, Hypsibius dujardini and Macrobiotus cf. harmsworthi. J. Limnol. 2013, 72, 80–91. [Google Scholar] [CrossRef]
- Jönsson, K.I.; Schill, R.O. Induction of Hsp70 by desiccation, ionising radiation and heat-shock in the eutardigrade Richtersius coronifer. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2007, 146, 456–460. [Google Scholar] [CrossRef]
- Mayer, M.P.; Bukau, B. Hsp70 chaperones: Cellular functions and molecular mechanism. Cell. Mol. Life Sci. 2005, 62, 670. [Google Scholar] [CrossRef]
- Murphy, M.E. The HSP70 family and cancer. Carcinogenesis 2013, 34, 1181–1188. [Google Scholar] [CrossRef]
- Broca, P. Rapport sur la question soumise à la Société de Biologie au suject de la reviviscence des animaux desséchés. Mémoires Soc. Biol. Paris 1860, 2, 1–140. [Google Scholar]
- Welnicz, W.; Grohme, M.A.; Kaczmarek, L.; Schill, R.O.; Frohme, M. Anhydrobiosis in tardigrades—The last decade. J. Insect Physiol. 2011, 57, 577–583. [Google Scholar] [CrossRef]
- Crowe, J.H. Trehalose as a “chemical chaperone”: Fact and fantasy. Adv. Exp. Med. Biol. 2008, 594, 143–158. [Google Scholar] [CrossRef]
- Crowe, J.H.; Carpenter, J.F.; Crowe, L.M. The role of vitrification in anhydrobiosis. Ann. Rev. Physiol. 1998, 60, 73–103. [Google Scholar] [CrossRef]
- Westh, P.; Ramløv, H. Trehalose accumulation in the tardigrade Adorybiotus coronifer during anhydrobiosis. J. Exp. Zool. 1991, 258, 303–311. [Google Scholar] [CrossRef]
- Hengherr, S.; Heyer, A.G.; Kohler, H.R.; Schill, R.O. Trehalose and anhydrobiosis in tardigrades—Evidence for divergence in responses to dehydration. FEBS J. 2008, 275, 281–288. [Google Scholar] [CrossRef]
- Jönsson, K.I.; Persson, O. Trehalose in Three Species of Desiccation Tolerant Tardigrades. Open Zool. J. 2010, 3, 1–5. [Google Scholar] [CrossRef]
- Hengherr, S.; Worland, M.R.; Reuner, A.; Brummer, F.; Schill, R.O. High-temperature tolerance in anhydrobiotic tardigrades is limited by glass transition. Physiol. Biochem. Zool. 2009, 82, 749–755. [Google Scholar] [CrossRef]
- Leprince, O.; Buitink, J. Desiccation tolerance: From genomics to the field. Plant Sci. 2010, 179, 554–564. [Google Scholar] [CrossRef]
- Yamaguchi, A.; Tanaka, S.; Yamaguchi, S.; Kuwahara, H.; Takamura, C.; Imajoh-Ohmi, S.; Horikawa, D.D.; Toyoda, A.; Katayama, T.; Arakawa, K.; et al. Two novel heat-soluble protein families abundantly expressed in an anhydrobiotic tardigrade. PLoS ONE 2012, 7, e44209. [Google Scholar] [CrossRef]
- Tanaka, S.; Tanaka, J.; Miwa, Y.; Horikawa, D.D.; Katayama, T.; Arakawa, K.; Toyoda, A.; Kubo, T.; Kunieda, T. Novel mitochondria-targeted heat-soluble proteins identified in the anhydrobiotic tardigrade improve osmotic tolerance of human cells. PLoS ONE 2015, 10, e0118272. [Google Scholar] [CrossRef]
- Kamilari, M.; Jørgensen, A.; Schiøtt, M.; Møbjerg, N. Comparative transcriptomics suggest unique molecular adaptations within tardigrade lineages. BMC Genom. 2019, 20, 607. [Google Scholar] [CrossRef]
- França, M.B.; Panek, A.D.; Eleutherio, E.C.A. Oxidative stress and its effects during dehydration. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2007, 146, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, A.M.; Negroni, M.; Altiero, T.; Montorfano, G.; Corsetto, P.A.; Valeria, P.; Berselli, R.; Berra, B.; Guidetti, R.; Rebecchi, L. Antioxidant defences in hydrated and desiccated states of the tardigrade Paramacrobiotus richtersi. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2010, 156, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Förster, F.; Beisser, D.; Grohme, M.A.; Liang, C.; Mali, B.; Siegl, A.M.; Engelmann, J.C.; Shkumatov, A.V.; Schokraie, E.; Müller, T.; et al. Transcriptome analysis in tardigrade species reveals specific molecular pathways for stress adaptations. Bioinform. Biol. Insights 2012, 6, 69–96. [Google Scholar] [CrossRef] [PubMed]
- Schokraie, E.; Hotz-Wagenblatt, A.; Warnken, U.; Mali, B.; Frohme, M.; Forster, F.; Dandekar, T.; Hengherr, S.; Schill, R.O.; Schnolzer, M. Proteomic analysis of tardigrades: Towards a better understanding of molecular mechanisms by anhydrobiotic organisms. PLoS ONE 2010, 5, e9502. [Google Scholar] [CrossRef] [PubMed]
- Hygum, T.L.; Clausen, L.K.B.; Halberg, K.A.; Jørgensen, A.; Møbjerg, N. Tun formation is not a prerequisite for desiccation tolerance in the marine tidal tardigrade Echiniscoides sigismundi. Zool. J. Linn. Soc. 2016, 178, 907–911. [Google Scholar] [CrossRef]
- Sørensen-Hygum, T.L.; Stuart, R.M.; Jørgensen, A.; Møbjerg, N. Modelling extreme desiccation tolerance in a marine tardigrade. Sci. Rep. 2018, 8, 11495. [Google Scholar] [CrossRef]
- Jönsson, K.I.; Rebecchi, L. Experimentally induced anhydrobiosis in the tardigrade Richtersius coronifer: Phenotypic factors affecting survival. J. Exp. Zool. 2002, 293, 578–584. [Google Scholar] [CrossRef]
- Wright, J.C. Desiccation Tolerance and Water-Retentive Mechanisms in Tardigrades. J. Exp. Biol. 1989, 142, 267–292. [Google Scholar]
- Horikawa, D.D.; Kunieda, T.; Abe, W.; Watanabe, M.; Nakahara, Y.; Yukuhiro, F.; Sakashita, T.; Hamada, N.; Wada, S.; Funayama, T.; et al. Establishment of a rearing system of the extremotolerant tardigrade Ramazzottius varieornatus: A new model animal for astrobiology. Astrobiology 2008, 8, 549–556. [Google Scholar] [CrossRef]
- Gusev, O.; Nakahara, Y.; Vanyagina, V.; Malutina, L.; Cornette, R.; Sakashita, T.; Hamada, N.; Kikawada, T.; Kobayashi, Y.; Okuda, T. Anhydrobiosis-associated nuclear DNA damage and repair in the sleeping chironomid: Linkage with radioresistance. PLoS ONE 2010, 5, e14008. [Google Scholar] [CrossRef]
- Ryabova, A.; Mukae, K.; Cherkasov, A.; Cornette, R.; Shagimardanova, E.; Sakashita, T.; Okuda, T.; Kikawada, T.; Gusev, O. Genetic background of enhanced radioresistance in an anhydrobiotic insect: Transcriptional response to ionizing radiations and desiccation. Extremophiles 2017, 21, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.Q.; Leopold, A.C.; Crowe, L.M.; Crowe, J.H. Stability of dry liposomes in sugar glasses. Biophys. J. 1996, 70, 1769–1776. [Google Scholar] [CrossRef]
- Wolkers, W.F.; Walker, N.J.; Tablin, F.; Crowe, J.H. Human Platelets Loaded with Trehalose Survive Freeze-Drying. Cryobiology 2001, 42, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Gläfke, C.; Akhoondi, M.; Oldenhof, H.; Sieme, H.; Wolkers, W.F. Cryopreservation of platelets using trehalose: The role of membrane phase behavior during freezing. Biotechnol. Prog. 2012, 28, 1347–1354. [Google Scholar] [CrossRef] [PubMed]
- Piszkiewicz, S.; Gunn, K.H.; Warmuth, O.; Propst, A.; Mehta, A.; Nguyen, K.H.; Kuhlman, E.; Guseman, A.J.; Stadmiller, S.S.; Boothby, T.C.; et al. Protecting activity of desiccated enzymes. Protein Sci. 2019, 28, 941–951. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z. Genomic instability and cancer: An introduction. J. Mol. Cell Biol. 2011, 3, 1–3. [Google Scholar] [CrossRef] [PubMed]
- West, C.M.; Barnett, G.C. Genetics and genomics of radiotherapy toxicity: Towards prediction. Genome Med. 2011, 3, 52. [Google Scholar] [CrossRef] [PubMed]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef] [PubMed]
- Rebecchi, L. Dry up and survive: The role of antioxidant defences in anhydrobiotic organisms. J. Limnol. 2013, 72, 62–72. [Google Scholar] [CrossRef]
- Kyriakakis, E.; Markaki, M.; Tavernarakis, N. Caenorhabditis elegans as a model for cancer research. Mol. Cell Oncol. 2015, 2, e975027. [Google Scholar] [CrossRef] [PubMed]
- Villegas, S.N. One hundred years of Drosophila cancer research: No longer in solitude. Dis. Models Mech. 2019, 12, dmm039032. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jönsson, K.I. Radiation Tolerance in Tardigrades: Current Knowledge and Potential Applications in Medicine. Cancers 2019, 11, 1333. https://doi.org/10.3390/cancers11091333
Jönsson KI. Radiation Tolerance in Tardigrades: Current Knowledge and Potential Applications in Medicine. Cancers. 2019; 11(9):1333. https://doi.org/10.3390/cancers11091333
Chicago/Turabian StyleJönsson, K. Ingemar. 2019. "Radiation Tolerance in Tardigrades: Current Knowledge and Potential Applications in Medicine" Cancers 11, no. 9: 1333. https://doi.org/10.3390/cancers11091333
APA StyleJönsson, K. I. (2019). Radiation Tolerance in Tardigrades: Current Knowledge and Potential Applications in Medicine. Cancers, 11(9), 1333. https://doi.org/10.3390/cancers11091333




