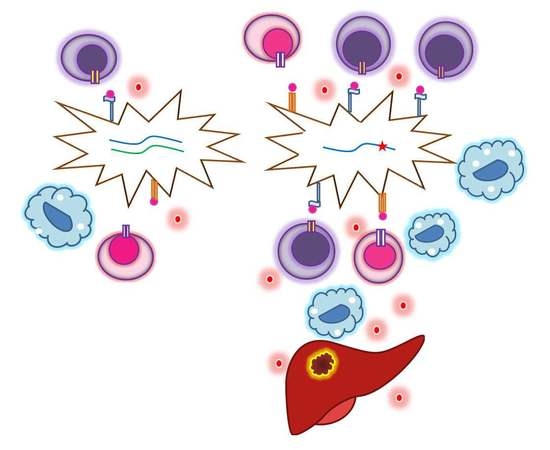
HLA Expression in Uveal Melanoma: An Indicator of Malignancy and a Modifiable Immunological Target
Abstract
1. Introduction
2. HLA Expression in Uveal Melanoma
2.1. Variability of HLA Expression in UM
2.2. HLA Expression and Metastatic Potential
2.3. Coordination between HLA Expression and Tumor Infiltrates
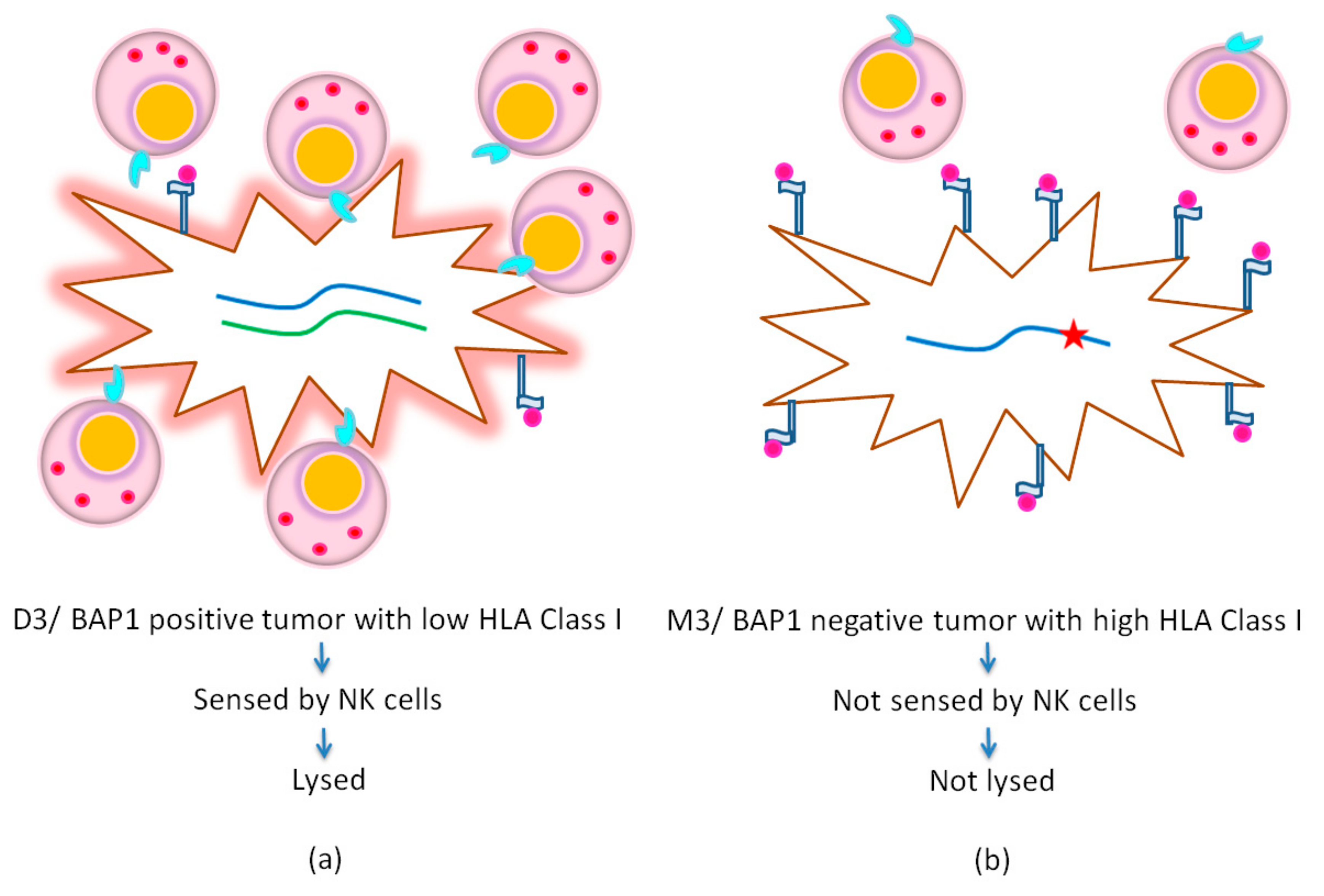
2.4. Locus and Allele-Specific Loss as an Escape Strategy
3. HLA Class I Regulation in UM
3.1. HLA Expression is Related to Genetic Factors
3.2. Locus-Specific Differences between HLA-A and HLA-B
4. Other Types of HLA Class I in UM
5. Genetics Play a Role in the Development of UM
5.1. HLA Allele Frequencies in UM
5.2. Somatic Genetic Abnormalities and HLA Expression in Uveal Melanoma
6. Metastases and HLA Expression
7. The Effect of Different Treatments on HLA Expression in UM
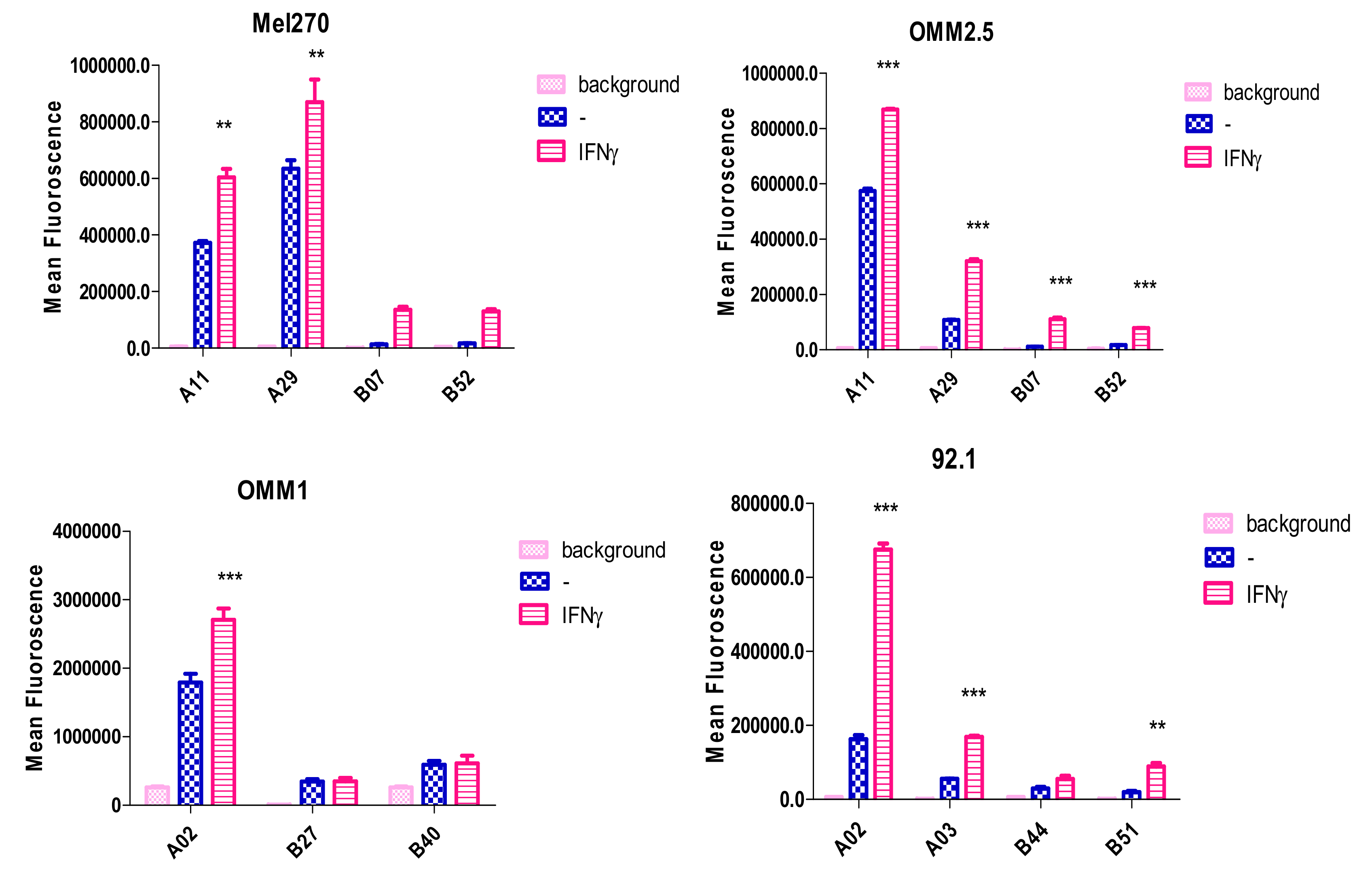
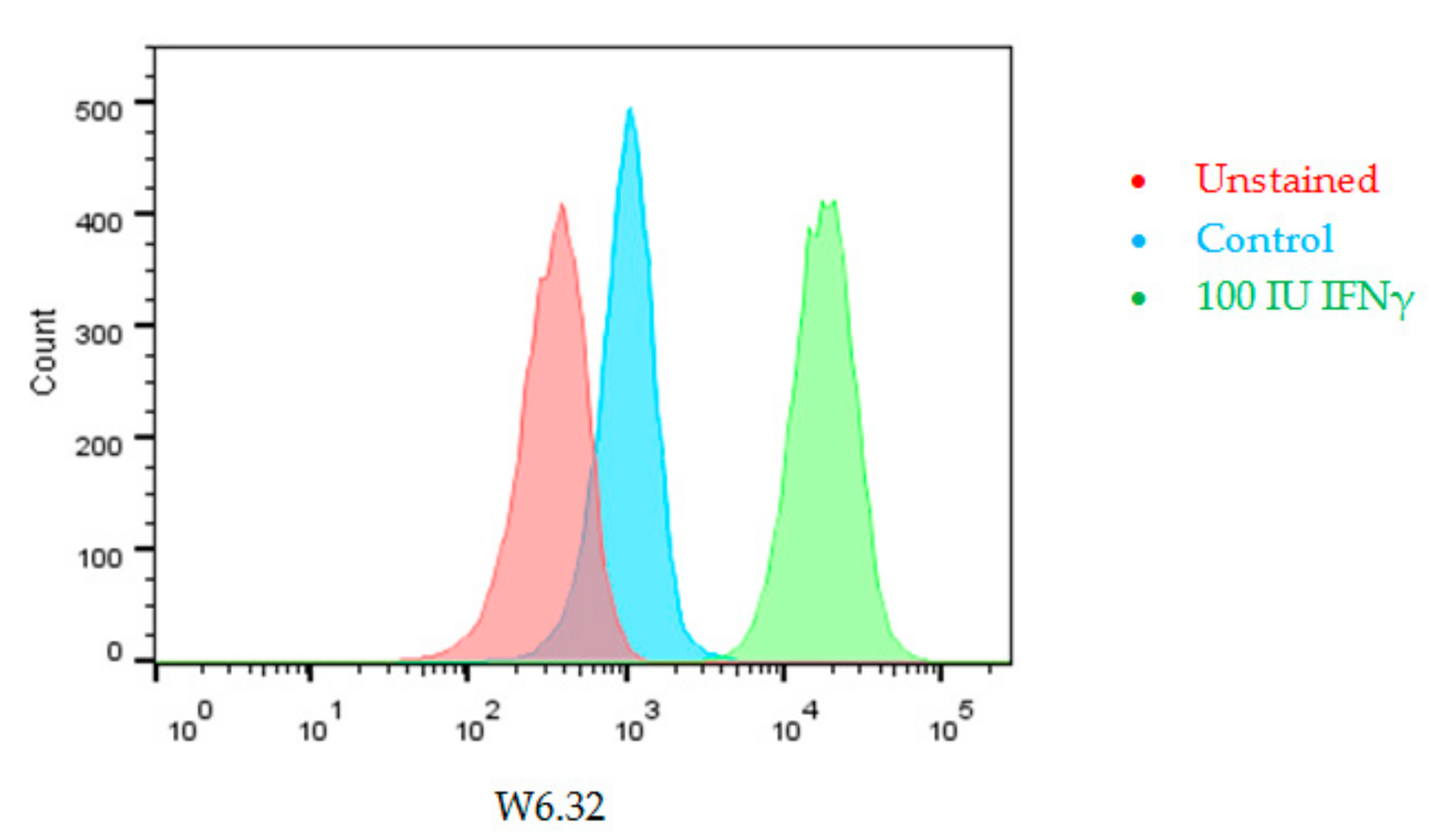
Interferon Induces HLA Expression
8. HLA Class II in UM
8.1. Irradiation and HLA Class II Expression
8.2. Regulation of HLA Class II
9. Environmental Influences
10. Potential for Immunotherapy: Function of HLA Antigens in Antigen Presentation
11. New Technologies Might Serve to Improve Knowledge about HLA Expression in UM
12. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Krantz, B.A.; Dave, N.; Komatsubara, K.M.; Marr, B.P.; Carvajal, R.D. Uveal melanoma: Epidemiology, etiology, and treatment of primary disease. Clin. Ophthalmol. 2017, 11, 279–289. [Google Scholar] [CrossRef]
- Singh, A.D.; Bergman, L.; Seregard, S. Uveal Melanoma: Epidemiologic Aspects. Ophthalmol. Clin. North Am. 2005, 18, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Metzelaar-Blok, J.A.W.; Ter Huurne, J.A.C.; Hurks, H.M.H.; Keunen, J.E.E.; Jager, M.J.; Gruis, N.A. Characterization of Melanocortin-1 Receptor gene variants in uveal melanoma patients. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1951–1954. [Google Scholar]
- Weis, E.; Shah, C.P.; Lajous, M.; Shields, J.A.; Shields, C.L. The association between host susceptibility factors and uveal melanoma: A meta-analysis. Arch. Ophthalmol. 2006, 124, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Dogrusöz, M.; Jager, M.J.; Damato, B. Uveal melanoma treatment and prognostication. Asia Pac. J. Ophthalmol. 2017, 6, 186–196. [Google Scholar] [CrossRef]
- Kaliki, S.; Shields, C.S. Uveal melanoma: Relatively rare but deadly cancer. Eye 2017, 31, 241–257. [Google Scholar] [CrossRef] [PubMed]
- Dogrusöz, M.; Jager, M.J. Genetic prognostication in uveal melanoma. Acta Ophthalmol. 2018, 96, 331–347. [Google Scholar] [CrossRef]
- Damato, B.; Coupland, S.E. Translating uveal melanoma cytogenetics into clinical care. Arch. Ophthalmol. 2009, 127, 423–429. [Google Scholar] [CrossRef]
- Kujala, E.; Mäkitie, T.; Kivelä, T. Very long-term prognosis of patients with malignant uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2003, 44, 4651–4659. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Fouad, Y.A.; Aanei, C. Revisiting the hallmarks of cancer. Am. J. Cancer Res. 2017, 7, 1016–1036. [Google Scholar] [PubMed]
- Colotta, F.; Allavena, P.; Sica, A.; Garlanda, C.; Mantovani, A. Cancer-related inflammation, the seventh hallmark of cancer: Links to genetic instability. Carcinogenesis 2009, 30, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Streilein, J.W. Unraveling immune privilege. Science 1995, 270, 1158–1159. [Google Scholar] [CrossRef] [PubMed]
- De Waard-Siebinga, I.; Hilders, C.G.; Hansen, B.E.; van Delft, J.L.; Jager, M.J. HLA expression and tumor-infiltrating immune cells in uveal melanoma. Graefe’s Arch. Clin. Exp. Ophthalmol. 1996, 234, 34–42. [Google Scholar] [CrossRef]
- Gezgin, G.; Dogrusöz, M.; van Essen, T.H.; Kroes, W.G.; Luyten, G.P.; van der Velden, P.A.; Walter, V.; Verdijk, R.M.; van Hall, T.; van der Burg, S.H.; et al. Genetic evolution of uveal melanoma guides the development of an inflammatory microenvironment. Cancer Immunol. Immunother. 2017, 66, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Jager, M.J.; Hurks, H.M.; Levitskaya, J.; Kiessling, R. HLA expression in uveal melanoma: There is no rule without some exception. Hum. Immunol. 2002, 63, 444–451. [Google Scholar] [CrossRef]
- Maat, W.; Ly, L.V.; Jordanova, E.S.; de Wolff-Rouendaal, D.; Schalij-Delfos, N.E.; Jager, M.J. Monosomy of chromosome 3 and an inflammatory phenotype occur together in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2008, 49, 505–510. [Google Scholar] [CrossRef]
- Robertson, A.G.; Shih, J.; Yau, C.; Gibb, E.A.; Oba, J.; Mungall, K.L.; Hess, J.M.; Uzunangelov, V.; Walter, V.; Danilova, L.; et al. Integrative analysis identifies four molecular and clinical subsets in uveal melanoma. Cancer Cell 2017, 32, 204–220. [Google Scholar] [CrossRef]
- Bronkhorst, I.H.; Jager, M.J. Uveal melanoma: The inflammatory microenvironment. J. Innate Immunol. 2012, 4, 454–462. [Google Scholar] [CrossRef]
- Harbour, J.W.; Onken, M.D.; Roberson, E.D.; Duan, S.; Cao, L.; Worley, L.A.; Council, M.L.; Matatall, K.A.; Helms, C.; Bowcock, A.M. Frequent mutation of BAP1 in metastasizing uveal melanomas. Science 2010, 330, 1410–1413. [Google Scholar] [CrossRef]
- Van Essen, T.H.; van Pelt, S.I.; Versluis, M.; Bronkhorst, I.H.G.; van Duinen, S.G.; Marinkovic, M.; Kroes, W.G.M.; Ruivenkamp, C.A.L.; de Klein, A.; Kilic, E.; et al. Prognostic parameters in Uveal Melanoma and their association with BAP1 expression. Br. J. Ophthalmol. 2014, 98, 1738–1743. [Google Scholar] [CrossRef]
- Szalai, E.; Wells, J.R.; Ward, L.; Grossniklaus, H.E. Uveal melanoma nuclear BRCA-1-Associated Protein-1 Immunoreactivity is an indicator of metastasis. Ophthalmology 2018, 125, 203–209. [Google Scholar] [CrossRef]
- Mosaad, Y.M. Clinical role of human leukocyte antigen in health and disease. Scand J. Immunol. 2015, 82, 283–306. [Google Scholar] [CrossRef]
- Ruiter, D.J.; Bergman, W.; Welvaart, K.; Scheffer, E.; van Vloten, W.A.; Russo, C.; Ferrone, S. Immunohistochemical analysis of malignant melanomas and nevocellular nevi with monoclonal antibodies to distinct monomorphic determinants of HLA antigens. Cancer Res. 1984, 44, 3930–3935. [Google Scholar] [PubMed]
- Jager, M.J.; de Wolff-Rouendaal, D.; Breebaart, A.C.; Ruiter, D.J. Expression of HLA antigens in paraffin sections of uveal melanomas. Doc. Ophthalmol. 1986, 64, 69–76. [Google Scholar] [CrossRef]
- De Waard-Siebinga, I.; Houbiers, J.G.; Hilders, C.G.; de Wolff-Rouendaal, D.; Jager, M.J. Differential expression of HLA-A and B-alleles on uveal melanoma as determined by immuno-histology. Ocul. Immunol. Inflamm. 1996, 4, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Blom, D.J.; Luyten, G.P.; Mooy, C.; Kerkvliet, S.; Zwinderman, A.H.; Jager, M.J. Human leukocyte antigen class I expression. Marker of poor prognosis in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 1997, 38, 1865–1872. [Google Scholar]
- Ma, D.; Niederkorn, J.Y. Transforming growth factor-beta down-regulates major histocompatibility complex class I antigen expression and increases the susceptibility of uveal melanoma cells to natural killer cell-mediated cytolysis. Immunology 1995, 86, 263–269. [Google Scholar] [PubMed]
- Ericsson, C.; Seregard, S.; Bartolazzi, A.; Levitskaya, E.; Ferrone, S.; Kiessling, R.; Larsson, O. Association of HLA class I and class II antigen expression and mortality in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2153–2156. [Google Scholar]
- Dithmar, S.; Crowder, J.; Jager, M.J.; Vigniswaran, N.; Grossniklaus, H.E. HLA class I antigen expression correlates with histological cell type in uveal melanoma. Der Ophthalmol. 2002, 99, 625–628. [Google Scholar] [CrossRef]
- Stam, N.J.; Vroom, T.M.; Peters, P.J.; Pastoors, E.B.; Ploegh, H.L. HLA-A-and HLA-B-specific monoclonal antibodies reactive with free heavy chains in western blots, in formalin-fixed, paraffin-embedded tissue sections and in cryo-immuno-electron microscopy. Int. Immunol. 1990, 2, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Meecham, W.J.; Char, D.H.; Kaleta-Michaels, S. Infiltrating lymphocytes and antigen expression in uveal melanoma. Ophthalmol. Res. 1992, 24, 20–26. [Google Scholar] [CrossRef]
- Van Essen, T.H.; van Pelt, S.I.; Bronkhorst, I.H.; Versluis, M.; Némati, F.; Laurent, C.; Luyten, G.P.; van Hall, T.; van den Elsen, P.J.; van der Velden, P.A.; et al. Upregulation of HLA expression in primary uveal melanoma by infiltrating leukocytes. PLoS ONE 2016, 11. [Google Scholar] [CrossRef]
- Anastassiou, G.; Rebmann, V.; Wagner, S.; Bornfeld, N.; Grosse-Wilde, H. Expression of classic and nonclassic HLA class I antigens in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2003, 44, 2016–2019. [Google Scholar] [CrossRef]
- Hurks, H.M.; Metzelaar-Blok, J.A.; Mulder, A.; Claas, F.H.; Jager, M.J. High frequency of allele-specific down-regulation of HLA class I expression in uveal melanoma cell lines. Int. J. Cancer 2000, 85, 697–702. [Google Scholar] [CrossRef]
- De Waard-Siebinga, I.; Creyghton, W.M.; Kool, J.; Jager, M.J. Effects of interferon alfa and gamma on human uveal melanoma cells in vitro. Br. J. Ophthalmol. 1995, 79, 847–855. [Google Scholar] [CrossRef]
- Jager, M.J.; Magner, J.A.; Ksander, B.R.; Dubovy, S.R. Uveal melanoma cell lines: Where do they come from? (An American Ophthalmological Society Thesis). Trans. Am. Ophthalmol. Soc. 2016, 114, T5. [Google Scholar]
- Van Essen, T.H.; Bronkhorst, I.H.; Maat, W.; Verduyn, W.; Roelen, D.L.; Luyten, G.P.; Jager, M.J. A comparison of HLA genotype with inflammation in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2640–2646. [Google Scholar] [CrossRef]
- Versteeg, R.; Krüse-Wolters, K.M.; Plomp, A.C.; Van Leeuwen, A.; Stam, N.J.; Ploegh, H.L.; Ruiter, D.J.; Schrier, P.I. Suppression of class I human histocompatibility leukocyte antigen by c-myc is locus specific. J. Exp. Med. 1989, 170, 621–635. [Google Scholar] [CrossRef]
- Blom, D.J.; Mooy, C.M.; Luyten, G.P.; Kerkvliet, S.; Ouwerkerk, I.; Zwinderman, A.H.; Schrier, P.I.; Jager, M.J. Inverse correlation between expression of HLA-B and c-myc in uveal melanoma. J. Pathol. 1997, 181, 75–79. [Google Scholar] [CrossRef]
- Kaklamanis, L.; Townsend, A.; Doussis-Anagnostopoulou, I.A.; Mortensen, N.; Harris, A.L.; Gatter, K.C. Loss of major histocompatibility complex-encoded transporter associated with antigen presentation (TAP) in colorectal cancer. Am. J. Pathol. 1994, 145, 505–509. [Google Scholar] [PubMed]
- Ling, A.; Löfgren-Burström, A.; Larsson, P.; Li, X.; Wikberg, M.L.; Öberg, Å.; Stenling, R.; Edin, S.; Palmqvist, R. TAP1 down-regulation elicits immune escape and poor prognosis in colorectal cancer. Oncoimmunology 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Dissemond, J.; Götte, P.; Mörs, J.; Lindeke, A.; Goos, M.; Ferrone, S.; Wagner, S.N. Association of TAP1 downregulation in human primary melanoma lesions with lack of spontaneous regression. Melanoma Res. 2003, 13, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Kamarashev, J.; Ferrone, S.; Seifert, B.; Böni, R.; Nestle, F.; Burg, G.; Dummer, R. TAP1 down-regulation in primary melanoma lesions: An independent marker of poor prognosis. Int J. Cancer 2001, 95, 23–28. [Google Scholar] [CrossRef]
- Krishnakumar, S.; Abhyankar, D.; Sundaram, A.L.; Pushparaj, V.; Shanmugam, M.P.; Biswas, J. Major histocompatibility antigens and antigen-processing molecules in uveal melanoma. Clin. Cancer Res. 2003, 9, 4159–4164. [Google Scholar] [PubMed]
- Girdlestone, J.; Isamat, M.; Gewert, D.; Milstein, C. Transcriptional regulation of HLA-A and-B: Differential binding of members of the Rel and IRF families of transcription factors. Proc. Natl. Acad. Sci. USA 1993, 90, 11568–11572. [Google Scholar] [CrossRef]
- Johnson, D.R.; Pober, J.S. HLA class I heavy-chain gene promoter elements mediating synergy between tumor necrosis factor and interferons. Mol. Cell. Biol. 1994, 14, 1322–1332. [Google Scholar] [CrossRef]
- Blais, M.E.; Dong, T.; Rowland-Jones, S. HLA-C as a mediator of natural killer and T-cell activation: Spectator or key player? Immunology 2011, 133, 1–7. [Google Scholar] [CrossRef]
- Ugurel, S.; Rebmann, V.; Ferrone, S.; Tilgen, W.; Grosse-Wilde, H.; Reinhold, U. Soluble human leukocyte antigen-G serum level is elevated in melanoma patients and is further increased by interferon-α immunotherapy. Cancer 2001, 92, 369–376. [Google Scholar] [CrossRef]
- Derré, L.; Corvaisier, M.; Charreau, B.; Moreau, A.; Godefroy, E.; Moreau-Aubry, A.; Jotereau, F.; Gervois, N. Expression and release of HLA-E by melanoma cells and melanocytes: Potential impact on the response of cytotoxic effector cells. J. Immunol. 2006, 177, 3100–3107. [Google Scholar] [CrossRef]
- Rouas-Freiss, N.; Moreau, P.; Ferrone, S.; Carosella, E.D. HLA-G proteins in cancer: Do they provide tumor cells with an escape mechanism? Cancer Res. 2005, 65, 10139–10144. [Google Scholar] [CrossRef]
- Hurks, H.M.; Valter, M.M.; Wilson, L.; Hilgert, I.; van den Elsen, P.J.; Jager, M.J. Uveal melanoma: No expression of HLA-G. Investig. Ophthalmol. Vis. Sci. 2001, 42, 3081–3084. [Google Scholar]
- Metzelaar-Blok, J.A.; Hurks, H.M.; Naipal, A.; de Lange, P.; Keunen, J.E.; Claas, F.H.; Doxiadis, I.; Jager, M.J. Normal HLA class I, II, and MICA gene distribution in uveal melanoma patients. Mol. Vis. 2005, 11, 1166–1172. [Google Scholar]
- Maat, W.; Haasnoot, G.W.; Claas, F.H.; Schalij-Delfos, N.E.; Schreuder, G.M.; Jager, M.J. HLA Class I and II genotype in uveal melanoma: Relation to occurrence and prognosis. Investig. Ophthalmol. Vis. Sci. 2006, 47, 3–6. [Google Scholar] [CrossRef]
- Jager, M.J.; Völker-Dieben, H.J.; de Wolff-Rouendaal, D.; Kakebeeke-Kemme, H.; D’Amaro, J. Possible relation between HLA and ABO type and prognosis of uveal melanoma. Doc. Ophthalmol. 1992, 82, 43–47. [Google Scholar] [CrossRef]
- Helgadottir, H.; Höiom, V. The genetics of uveal melanoma: Current insights. Appl. Clin. Genet. 2016, 9, 147–155. [Google Scholar] [CrossRef]
- Kilic, E.; van Gils, W.; Lodder, E.; Beverloo, H.B.; van Til, M.E.; Mooy, C.M.; Paridaens, D.; de Klein, A.; Luyten, G.P.M. Clinical and cytogenetic analyses in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2006, 47, 3703–3707. [Google Scholar] [CrossRef]
- Shields, C.L.; Say, E.A.; Hasanreisoglu, M.; Saktanasate, J.; Lawson, B.M.; Landy, J.E.; Badami, A.U.; Sivalingam, M.D.; Mashayekhi, A.; Shields, J.A.; et al. Cytogenetic abnormalities in uveal melanoma based on tumor features and size in 1059 patients: The 2016 W. Richard Green Lecture. Ophthalmology 2017, 124, 609–618. [Google Scholar] [CrossRef]
- Cassoux, N.; Rodrigues, M.J.; Plancher, C.; Asselain, B.; Levy-Gabriel, C.; Lumbroso-Le Rouic, L.; Piperno-Neumann, S.; Dendale, R.; Sastre, X.; Desjardins, L.; et al. Genome-wide profiling is a clinically relevant and affordable prognostic test in posterior uveal melanoma. Br. J. Ophthalmol. 2014, 98, 769–774. [Google Scholar] [CrossRef]
- Versluis, M.; de Lange, M.J.; van Pelt, S.I.; Ruivenkamp, C.A.; Kroes, W.G.; Cao, J.; Jager, M.J.; Luyten, G.P.M.; van der Velden, P.A. Digital PCR validates 8q dosage as prognostic tool in uveal melanoma. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Singh, N.; Singh, A.D.; Hide, W. Inferring an evolutionary tree of uveal melanoma from genomic copy number aberrations. Investig. Ophthalmol. Vis. Sci. 2015, 56, 6801–6809. [Google Scholar] [CrossRef] [PubMed]
- Metzelaar-Blok, J.A.W.; Jager, M.J.; Hanifi Moghaddam, P.; van der Slik, A.; Giphart, M.J. Frequent loss of heterozygosity on chromosome 6p in uveal melanoma. Hum. Immunol. 1999, 60, 962–969. [Google Scholar] [CrossRef]
- Blom, D.J.; Schurmans, L.R.; de Waard-Siebinga, I.; de Wolff-Rouendaal, D.; Keunen, J.E.; Jager, M.J. HLA expression in a primary uveal melanoma, its cell line, and four of its metastases. Br. J. Ophthalmol. 1997, 81, 989–993. [Google Scholar] [CrossRef] [PubMed]
- Verbik, D.J.; Murray, T.G.; Tran, J.M.; Ksander, B.R. Melanomas that develop within the eye inhibit lymphocyte proliferation. Int J. Cancer 1997, 73, 470–478. [Google Scholar] [CrossRef]
- Gezgin, G.; Luk, S.J.; Cao, J.; Dogrusöz, M.; van der Steen, D.M.; Hagedoorn, R.S.; Krijgsman, D.; van der Velden, P.A.; Field, M.G.; Luyten, G.P.; et al. PRAME as a potential target for immunotherapy in metastatic uveal melanoma. JAMA Ophthalmol. 2017, 135, 541–549. [Google Scholar] [CrossRef]
- Field, M.G.; Decatur, C.L.; Kurtenbach, S.; Gezgin, G.; van der Velden, P.A.; Jager, M.J.; Kozak, K.N.; Harbour, J.W. PRAME as an independent biomarker for metastasis in uveal melanoma. Clin. Cancer Res. 2016, 22, 1234–1242. [Google Scholar] [CrossRef] [PubMed]
- Meir, T.; Dror, R.; Yu, X.; Qian, J.; Simon, I.; Pe’er, J.; Chowers, I. Molecular characteristics of liver metastases from uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2007, 48, 4890–4896. [Google Scholar] [CrossRef]
- De Waard-Siebinga, I.; Kool, J.; Jager, M.J. HLA antigen expression on uveal melanoma cells in vivo and in vitro. Hum. Immunol. 1995, 44, 111–117. [Google Scholar] [CrossRef]
- De Waard-Siebinga, I.; Blom, D.J.; Griffioen, M.; Schrier, P.I.; Hoogendoorn, E.; Beverstock, G.; Danen, E.H.; Jager, M.J. Establishment and characterization of an uveal-melanoma cell line. Int. J. Cancer 1995, 62, 155–161. [Google Scholar] [CrossRef]
- Seliger, B.; Kloor, M.; Ferrone, S. HLA class II antigen-processing pathway in tumors: Molecular defects and clinical relevance. Oncoimmunology 2017, 6. [Google Scholar] [CrossRef]
- Krishnakumar, S.; Abhyankar, D.; Lakshmi, S.A.; Shanmugam, M.P.; Pushparaj, V.; Biswas, J. HLA class II antigen expression in uveal melanoma: Correlation with clinicopathological features. Exp. Eye Res. 2003, 77, 175–180. [Google Scholar] [CrossRef]
- Jager, M.J.; van der Pol, J.P.; de Wolff-Rouendaal, D.; de Jong, P.T.V.M.; Ruiter, D.J. Decreased expression of HLA Class II antigens on human uveal melanoma cells after in vivo X-ray irradiation. Am. J. Ophthalmol. 1988, 105, 78–86. [Google Scholar] [CrossRef]
- Radosevich, M.; Jager, M.; Ono, S.J. Inhibition of MHC class II gene expression in uveal melanoma cells is due to methylation of the CIITA gene or an upstream activator. Exp. Mol. Pathol. 2007, 82, 68–76. [Google Scholar] [CrossRef]
- Holling, T.M.; Schooten, E.; Langerak, A.W.; van den Elsen, P.J. Regulation of MHC class II expression in human T-cell malignancies. Blood 2004, 103, 1438–1444. [Google Scholar] [CrossRef]
- Holling, T.M.; Bergevoet, M.W.; Wilson, L.; van Eggermond, M.C.; Schooten, E.; Steenbergen, R.D.; Snijders, P.J.; Jager, M.J.; van den Elsen, P.J. A role for EZH2 in silencing of IFN-γ inducible MHC2TA transcription in uveal melanoma. J. Immunol. 2007, 179, 5317–5325. [Google Scholar] [CrossRef]
- Boyd, N.H.; Morgan, J.E.; Greer, S.F. Polycomb recruitment at the Class II transactivator gene. Mol. Immunol. 2015, 67, 482–491. [Google Scholar] [CrossRef]
- Hehlgans, T.; Strominger, J.L. Activation of transcription by binding of NF-E1 (YY1) to a newly identified element in the first exon of the human DR alpha gene. J. Immunol. 1995, 154, 5181–5187. [Google Scholar]
- Griffioen, M.; Ouwerkerk, I.J.; Harten, V.; Schrier, P.I. HLA-B locus-specific downregulation in human melanoma requires enhancer A as well as a sequence element located downstream of the transcription initiation site. Immunogenetics 2000, 52, 121–128. [Google Scholar] [CrossRef]
- Blom, D.J.; Schuitmaker, H.J.; de Waard-Siebinga, I.; Dubbelman, T.M.; Jager, M.J. Decreased expression of HLA class I on ocular melanoma cells following in vitro photodynamic therapy. Cancer Lett. 1997, 112, 239–243. [Google Scholar] [CrossRef]
- Blom, D.J.; de Waard-Siebinga, I.; Apte, R.S.; Luyten, G.P.; Niederkorn, J.Y.; Jager, M.J. Effect of hyperthermia on expression of histocompatibility antigens and heat-shock protein molecules on three human ocular melanoma cell lines. Melanoma Res. 1997, 7, 103–109. [Google Scholar] [CrossRef]
- Sutmuller, R.P.; Schurmans, L.R.; van Duivenvoorde, L.M.; Tine, J.A.; van der Voort, E.I.; Toes, R.E.; Melief, C.J.; Jager, M.J.; Offringa, R. Adoptive T cell immunotherapy of human uveal melanoma targeting gp100. J. Immunol. 2000, 165, 7308–7315. [Google Scholar] [CrossRef]
- Luyten, G.P.M.; van der Spek, C.W.; Brand, I.; Sintnicolaas, K.; de Waard-Siebinga, I.; Jager, M.J.; de Jong, P.T.; Schrier, P.I.; Luider, T.M. Expression of MAGE, gp100 and tyrosinase genes in uveal melanoma cell lines. Melanoma Res. 1998, 8, 11–16. [Google Scholar] [CrossRef]
- Bosch, J.J.; Thompson, J.A.; Srivastava, M.K.; Iheagwara, U.K.; Murray, T.G.; Lotem, M.; Ksander, B.R.; Ostrand-Rosenberg, S. MHC Class II-transduced tumor cells originating in the immune-privileged eye prime and boost CD4+ T lymphocytes that cross-react with primary and metastatic uveal melanoma cells. Cancer Res. 2007, 67, 4499–4506. [Google Scholar] [CrossRef]
- Mondanelli, G.; Bianchi, R.; Pallotta, M.T.; Orabona, C.; Albini, E.; Iacono, A.; Belladonna, M.L.; Vacca, C.; Fallarino, F.; Macchiarulo, A.; et al. A relay pathway between Arginine and Tryptophan metabolism confers immunosuppressive properties on dendritic cells. Immunity 2017, 46, 233–244. [Google Scholar] [CrossRef]
- Li, Y.; Tredget, E.E.; Ghahary, A. Cell surface expression of MHC Class I antigen is suppressed in Indoleamine 2,3-Dioxygenase genetically modified keratinocytes: Implications in allogeneic skin substitute engraftment. Hum. Immunol. 2004, 65, 114–123. [Google Scholar] [CrossRef]
- Chen, P.W.; Mellon, J.K.; Mayhew, E.; Wang, S.; He, Y.G.; Hogan, N.; Niederkorn, J.Y. Uveal melanoma expression of indoleamine 2,3-deoxygenase: Establishment of an immune privileged environment by tryptophan depletion. Exp. Eye Res. 2007, 85, 617–625. [Google Scholar] [CrossRef]
- Stalhammer, G.; Seregard, S.; Grossniklaus, H.E. Expression of immune checkpoint receptors Indolamine 2,3-dioxygenase and T cell ig and ITIM domain in metastatic versus nonmetastatic choroidal melanoma. Cancer Med. 2019, 8, 2784–2792. [Google Scholar] [CrossRef]
- Zoutman, W.H.; Nell, R.J.; van der Velden, P.A. Usage of Droplet Digital PCR (ddPCR) assays for T cell quantification in cancer. Methods Mol. Biol. 2019, 1884, 1–14. [Google Scholar] [CrossRef]
- De Lange, M.J.; Nell, R.J.; Lalai, R.N.; Versluis, M.; Jordanova, E.S.; Luyten, G.P.M.; Jager, M.J.; van der Burg, S.H.; Zoutman, W.H.; van Hall, T.; et al. Digital PCR-based T cell quantification-assisted deconvolution of the microenvironment reveals that activated macrophages drive tumor inflammation in uveal melanoma. Mol. Cancer Res. 2018, 16, 1902–1911. [Google Scholar] [CrossRef]
- Subrahmanyam, P.B.; Maecker, H.T. CyTOF measurement of immunocompetence across Major Immune Cell Types. Curr. Protoc. Cytom. 2018, 82, 9–54. [Google Scholar] [CrossRef]
- Lingblom, C.M.D.; Kowli, S.; Swaminathan, N.; Maecker, H.T. Baseline immune profile by CyTOF can predict response to an investigational adjuvanted vaccine in elderly adults. J. Transl. Med. 2018, 16, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Buart, S.; Terry, S.; Noman, M.Z.; Lanoy, E.; Boutros, C.; Fogel, P.; Dessen, P.; Meurice, G.; Gaston-Mathé, Y.; Vielh, P.; et al. Transcriptional response to hypoxic stress in melanoma and prognostic potential of GBE1 and BNIP3. Oncotarget 2017, 8, 108786–108801. [Google Scholar] [CrossRef]
- Brouwer, N.J.; Wierenga, A.P.A.; Gezgin, G.; Marinkovic, M.; Luyten, G.P.M.; Kroes, W.G.M.; Versluis, M.; van der Velden, P.A.; Verdijk, R.M.; Jager, M.J. Ischemia Is Related to Tumour Genetics in Uveal Melanoma. Cancers 2019, 11, 1004. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; You, S.; Zhang, Q.; Osuka, S.; Devi, N.S.; Kaluz, S.; Ferguson, J.H.; Yang, H.; Chen, G.; Wang, B.; et al. Arylsulfonamide 64B inhibits hypoxia/HIF-induced expression of c-Met and CXCR4 and reduces primary tumor growth and metastasis of uveal melanoma. Clin Cancer Res. 2019, 25, 2206–2218. [Google Scholar] [CrossRef] [PubMed]
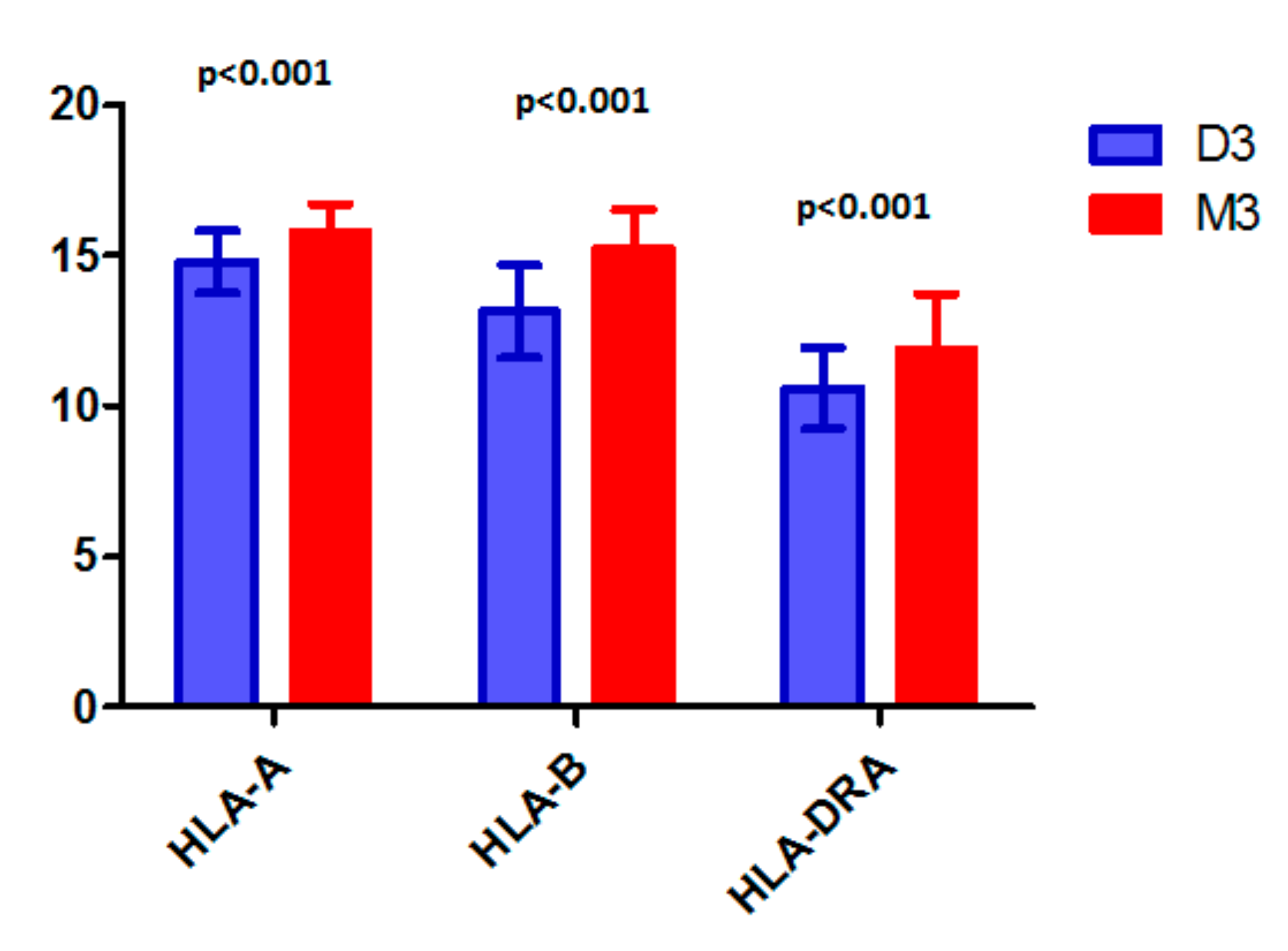
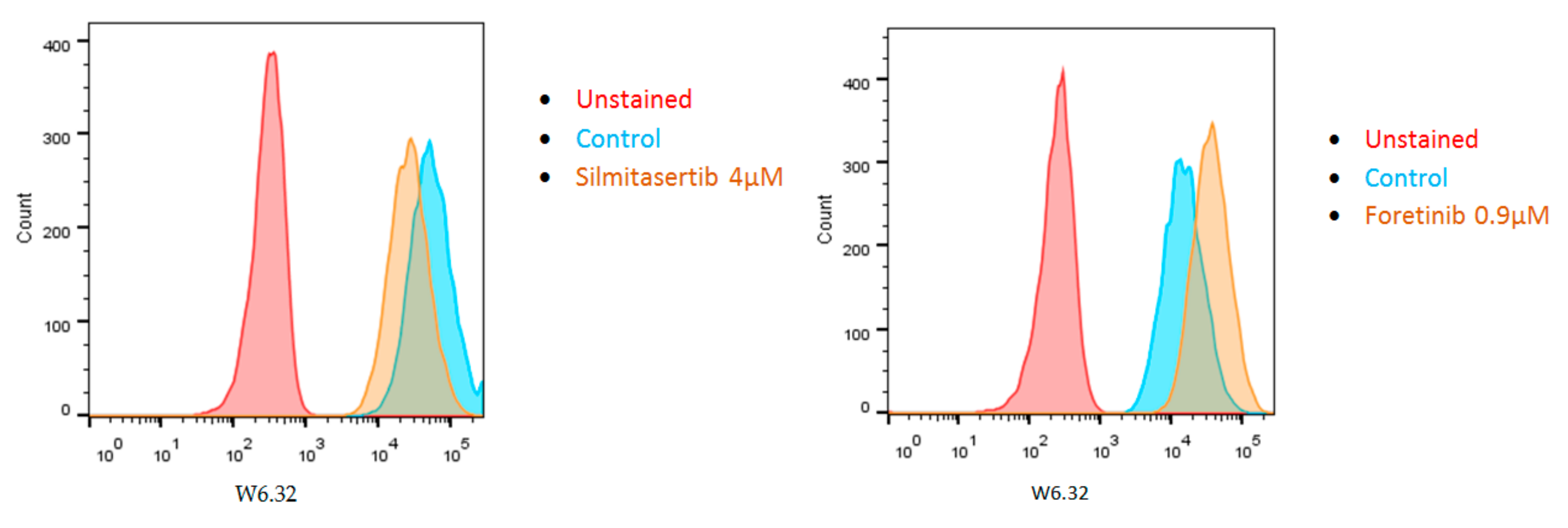
- Souri, Z.; Wierenga, A.P.A.; van Weeghel, C.; van der Velden, P.A.; Kroes, W.G.M.; Luyten, G.P.M.; van der Burg, S.H.; Jochemsen, A.G.; Jager, M.J. Loss of BAP1 Is Associated with Upregulation of the NFkB Pathway and Increased HLA Class I Expression in Uveal Melanoma. Cancers 2019, 11, 1102. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souri, Z.; Wierenga, A.P.A.; Mulder, A.; Jochemsen, A.G.; Jager, M.J. HLA Expression in Uveal Melanoma: An Indicator of Malignancy and a Modifiable Immunological Target. Cancers 2019, 11, 1132. https://doi.org/10.3390/cancers11081132
Souri Z, Wierenga APA, Mulder A, Jochemsen AG, Jager MJ. HLA Expression in Uveal Melanoma: An Indicator of Malignancy and a Modifiable Immunological Target. Cancers. 2019; 11(8):1132. https://doi.org/10.3390/cancers11081132
Chicago/Turabian StyleSouri, Zahra, Annemijn P.A. Wierenga, Arend Mulder, Aart G. Jochemsen, and Martine J. Jager. 2019. "HLA Expression in Uveal Melanoma: An Indicator of Malignancy and a Modifiable Immunological Target" Cancers 11, no. 8: 1132. https://doi.org/10.3390/cancers11081132
APA StyleSouri, Z., Wierenga, A. P. A., Mulder, A., Jochemsen, A. G., & Jager, M. J. (2019). HLA Expression in Uveal Melanoma: An Indicator of Malignancy and a Modifiable Immunological Target. Cancers, 11(8), 1132. https://doi.org/10.3390/cancers11081132