Surgery or Locoregional Approaches for Hepatic Oligometastatic Pancreatic Cancer: Myth, Hope, or Reality?
Abstract
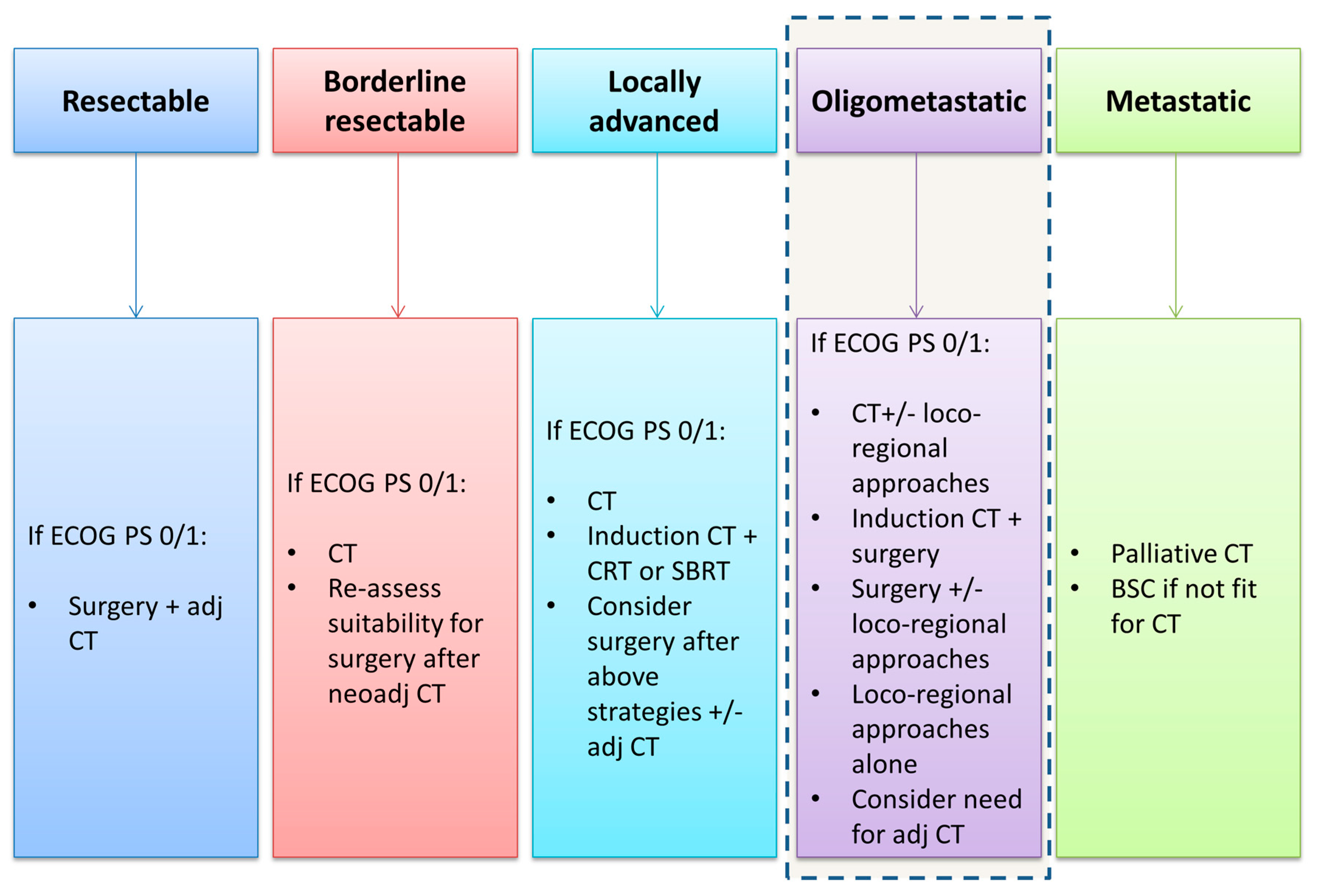
1. Introduction
2. Materials and Methods
3. Results
3.1. The Role of Liver Metastasectomy
3.2. The Role of Radiofrequency Ablation (RFA)
3.3. The Role of Irreversible Electroporation (IRE)
3.4. The Role of Stereotactic Body Radiation Therapy (SBRT)
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the united states. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Burris, H.A.; Moore, M.J.; Andersen, J.; Green, M.R.; Rothenberg, M.L.; Modiano, M.R.; Cripps, M.C.; Portenoy, R.K.; Storniolo, A.M.; Tarassoff, P.; et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997, 15, 2403–2413. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.-L.; Gourgou-Bourgade, S.; de la Fouchardière, C.; et al. FOLFIRINOX versus Gemcitabine for Metastatic Pancreatic Cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, D.; El-Maraghi, R.H.; Hammel, P.; Heinemann, V.; Kunzmann, V.; Sastre, J.; Scheithauer, W.; Siena, S.; Tabernero, J.; Teixeira, L.; et al. Nab-paclitaxel plus gemcitabine for metastatic pancreatic cancer: Long-term survival from a phase III trial. J. Natl. Cancer Inst. 2015, 107, dju413. [Google Scholar] [CrossRef] [PubMed]
- Oettle, H.; Post, S.; Neuhaus, P.; Gellert, K.; Langrehr, J.; Ridwelski, K.; Schramm, H.; Fahlke, J.; Zuelke, C.; Burkart, C.; et al. Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: A randomized controlled trial. J. Am. Med. Assoc. 2007, 297, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Neoptolemos, J.P.; Stocken, D.D.; Bassi, C.; Ghaneh, P.; Cunningham, D.; Goldstein, D.; Padbury, R.; Moore, M.J.; Gallinger, S.; Mariette, C.; et al. Adjuvant chemotherapy with fluorouracil plus folinic acid vs gemcitabine following pancreatic cancer resection: A randomized controlled trial. JAMA J. Am. Med. Assoc. 2010, 304, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Sinn, M.; Bahra, M.; Liersch, T.; Gellert, K.; Messmann, H.; Bechstein, W.; Waldschmidt, D.; Jacobasch, L.; Wilhelm, M.; Rau, B.M.; et al. CONKO-005: Adjuvant chemotherapy with gemcitabine plus erlotinib versus gemcitabine alone in patients after r0 resection of pancreatic cancer: A multicenter randomized phase III trial. J. Clin. Oncol. 2017, 35, 3330–3337. [Google Scholar] [CrossRef] [PubMed]
- De Ruysscher, D.; Peeters, S.; Dooms, C. Oligometastases. New Ther. Strateg. Lung Cancers 2015, 13, 219–223. [Google Scholar]
- Gillams, A.; Goldberg, N.; Ahmed, M.; Bale, R.; Breen, D.; Callstrom, M.; Chen, M.H.; Choi, B.I.; de Baere, T.; Dupuy, D.; et al. Thermal ablation of colorectal liver metastases: A position paper by an international panel of ablation experts, the interventional oncology sans frontières meeting 2013. Eur. Radiol. 2015, 25, 3438–3454. [Google Scholar] [CrossRef] [PubMed]
- Petrelli, F.; Coinu, A.; Cabiddu, M.; Ghilardi, M.; Borgonovo, K.; Lonati, V.; Barni, S. Hepatic resection for gastric cancer liver metastases: A systematic review and meta-analysis. J. Surg. Oncol. 2015, 111, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Tempero, M.A.; Malafa, M.P.; Al-Hawary, M.; Asbun, H.; Bain, A.; Behrman, S.W.; Benson, A.B.; Binder, E.; Cardin, D.B.; Cha, C.; et al. Pancreatic Adenocarcinoma, Version 2.2017, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2017, 15, 1028–1061. [Google Scholar] [CrossRef] [PubMed]
- Hackert, T.; Niesen, W.; Hinz, U.; Tjaden, C.; Strobel, O.; Ulrich, A.; Michalski, C.W.; Büchler, M.W. Radical surgery of oligometastatic pancreatic cancer. Eur. J. Surg. Oncol. 2017, 43, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Crippa, S.; Bittoni, A.; Sebastiani, E.; Partelli, S.; Zanon, S.; Lanese, A.; Andrikou, K.; Muffatti, F.; Balzano, G.; Reni, M.; et al. Is there a role for surgical resection in patients with pancreatic cancer with liver metastases responding to chemotherapy? Eur. J. Surg. Oncol. 2016, 42, 1533–1539. [Google Scholar] [CrossRef] [PubMed]
- Klein, F.; Puhl, G.; Guckelberger, O.; Pelzer, U.; Pullankavumkal, J.R.; Guel, S.; Neuhaus, P.; Bahra, M. The impact of simultaneous liver resection for occult liver metastases of pancreatic adenocarcinoma. Gastroenterol. Res. Pract. 2012, 2012, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Dünschede, F.; Will, L.; Von Langsdorf, C.; Möhler, M.; Galle, P.R.; Otto, G.; Vahl, C.F.; Junginger, T. Treatment of metachronous and simultaneous liver metastases of pancreatic cancer. Eur. Surg. Res. 2010, 44, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Frigerio, I.; Regi, P.; Giardino, A.; Scopelliti, F.; Girelli, R.; Bassi, C.; Gobbo, S.; Martini, P.T.; Capelli, P.; D’Onofrio, M.; et al. Downstaging in Stage IV Pancreatic Cancer: A New Population Eligible for Surgery? Ann. Surg. Oncol. 2017, 24, 2397–2403. [Google Scholar] [CrossRef] [PubMed]
- De Jong, M.C.; Farnell, M.B.; Sclabas, G.; Cunningham, S.C.; Cameron, J.L.; Geschwind, J.F.; Wolfgang, C.L.; Herman, J.M.; Edil, B.H.; Choti, M.A.; et al. Liver-directed therapy for hepatic metastases in patients undergoing pancreaticoduodenectomy: A dual-center analysis. Ann. Surg. 2010, 252, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Gleisner, A.L.; Assumpcao, L.; Cameron, J.L.; Wolfgang, C.L.; Choti, M.A.; Herman, J.M.; Schulick, R.D.; Pawlik, T.M. Is resection of periampullary or pancreatic adenocarcinoma with synchronous hepatic metastasis justified? Cancer 2007, 110, 2484–2492. [Google Scholar] [CrossRef] [PubMed]
- Adam, R.; Chiche, L.; Aloia, T.; Elias, D.; Salmon, R.; Rivoire, M.; Jaeck, D.; Saric, J.; Le Treut, Y.P.; Belghiti, J.; et al. Hepatic resection for noncolorectal nonendocrine liver metastases: Analysis of 1452 patients and development of a prognostic model. Ann. Surg. 2006, 244, 524–533. [Google Scholar] [CrossRef]
- Andreou, A.; Knitter, S.; Klein, F.; Malinka, T.; Schmelzle, M.; Struecker, B.; Schmuck, R.B.; Noltsch, A.R.; Lee, D.; Pelzer, U.; et al. The role of hepatectomy for synchronous liver metastases from pancreatic adenocarcinoma. Surg. Oncol. 2018, 27, 688–694. [Google Scholar] [CrossRef]
- Wright, G.P.; Poruk, K.E.; Zenati, M.S.; Steve, J.; Bahary, N.; Hogg, M.E.; Zuriekat, A.H.; Wolfgang, C.L.; Zeh, H.J.; Weiss, M.J. Primary Tumor Resection Following Favorable Response to Systemic Chemotherapy in Stage IV Pancreatic Adenocarcinoma with Synchronous Metastases: a Bi-institutional Analysis. J. Gastrointest. Surg. 2016, 20, 1830–1835. [Google Scholar] [CrossRef] [PubMed]
- Kandel, P.; Wallace, M.B.; Stauffer, J.; Bolan, C.; Raimondo, M.; Woodward, T.A.; Gomez, V.; Ritter, A.W.; Asbun, H.; Mody, K. Survival of Patients with Oligometastatic Pancreatic Ductal Adenocarcinoma Treated with Combined Modality Treatment Including Surgical Resection: A Pilot Study. J. Pancreat. Cancer 2018, 4, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Bahra, M.; Pratschke, J.; Klein, F.; Neuhaus, P.; Boas-Knoop, S.; Puhl, G.; Denecke, T.; Pullankavumkal, J.R.; Sinn, M.; Riess, H.; et al. Cytoreductive surgery for pancreatic cancer improves overall outcome of gemcitabine-based chemotherapy. Pancreas 2015, 44, 930–936. [Google Scholar] [CrossRef] [PubMed]
- Zanini, N.; Lombardi, R.; Masetti, M.; Giordano, M.; Landolfo, G.; Jovine, E. Surgery for isolated liver metastases from pancreatic cancer. Updates Surg. 2015, 67, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Tachezy, M.; Gebauer, F.; Janot, M.; Uhl, W.; Zerbi, A.; Montorsi, M.; Perinel, J.; Adham, M.; Dervenis, C.; Agalianos, C.; et al. Synchronous resections of hepatic oligometastatic pancreatic cancer: Disputing a principle in a time of safe pancreatic operations in a retrospective multicenter analysis. Surgery 2016, 160, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Slotta, J.E.; Schuld, J.; Distler, S.; Richter, S.; Schilling, M.K.; Kollmar, O. Hepatic resection of non-colorectal and non-neuroendocrine liver metastases - Survival benefit for patients with non-gastrointestinal primary cancers - A case-controlled study. Int. J. Surg. 2014, 12, 163–168. [Google Scholar] [CrossRef][Green Version]
- Schiergens, T.S.; Lüning, J.; Renz, B.W.; Thomas, M.; Pratschke, S.; Feng, H.; Lee, S.M.L.; Engel, J.; Rentsch, M.; Guba, M.; et al. Liver Resection for Non-colorectal Non-neuroendocrine Metastases: Where Do We Stand Today Compared to Colorectal Cancer? J. Gastrointest. Surg. 2016, 20, 1163–1172. [Google Scholar] [CrossRef]
- Shrikhande, S.V.; Kleeff, J.; Reiser, C.; Weitz, J.; Hinz, U.; Esposito, I.; Schmidt, J.; Friess, H.; Büchler, M.W. Pancreatic resection for M1 pancreatic ductal adenocarcinoma. Ann. Surg. Oncol. 2007, 14, 118–127. [Google Scholar] [CrossRef]
- Park, J.B.; Kim, Y.H.; Kim, J.; Chang, H.M.; Kim, T.W.; Kim, S.C.; Kim, P.N.; Han, D.J. Radiofrequency ablation of liver metastasis in patients with locally controlled pancreatic ductal adenocarcinoma. J. Vasc. Interv. Radiol. 2012, 23, 635–641. [Google Scholar] [CrossRef]
- Hua, Y.Q.; Wang, P.; Zhu, X.Y.; Shen, Y.H.; Wang, K.; Shi, W.D.; Lin, J.H.; Meng, Z.Q.; Chen, Z.; Chen, H.; et al. Radiofrequency ablation for hepatic oligometastatic pancreatic cancer: An analysis of safety and efficacy. Pancreatology 2017, 17, 967–973. [Google Scholar] [CrossRef]
- Takaki, H.; Yamakado, K.; Nakatsuka, A.; Yamada, T.; Shiraki, K.; Takei, Y.; Takeda, K. Frequency of and risk factors for complications after liver radiofrequency ablation under ct fluoroscopic guidance in 1500 sessions: Single-center experience. Am. J. Roentgenol. 2013, 200, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Jing, X.; Liu, J.; Wang, Y.; Wang, F.; Wang, Y.; Du, Z. Complications of thermal ablation of hepatic tumours: Comparison of radiofrequency and microwave ablative techniques. Clin. Radiol. 2013, 68, 608–615. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.C.G.; Kwon, D.; Chalikonda, S.; Sellers, M.; Kotz, E.; Scoggins, C.; McMasters, K.M.; Watkins, K. Treatment of 200 Locally Advanced (Stage III) Pancreatic Adenocarcinoma Patients With Irreversible Electroporation. Ann. Surg. 2015, 262, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Kluger, M.D.; Epelboym, I.; Schrope, B.A.; Mahendraraj, K.; Hecht, E.M.; Susman, J.; Weintraub, J.L.; Chabot, J.A. Single-Institution Experience with Irreversible Electroporation for T4 Pancreatic Cancer: First 50 Patients. Ann. Surg. Oncol. 2016, 23, 1736–1743. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Rice, J.; Sharma, D.; Martin, R.C.G. The use of IRE in multi-modality treatment for oligometastatic pancreatic cancer. Am. J. Surg. 2018, 216, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Syed, Y.A.; Patel-Yadav, A.K.; Rivers, C.; Singh, A.K. Stereotactic radiotherapy for prostate cancer: A review and future directions. World J. Clin. Oncol. 2017, 8, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Rosen, J.E.; Salazar, M.C.; Wang, Z.; Yu, J.B.; Decker, R.H.; Kim, A.W.; Detterbeck, F.C.; Boffa, D.J. Lobectomy versus stereotactic body radiotherapy in healthy patients with stage i lung cancer. J. Thorac. Cardiovasc. Surg. 2016, 152, 44–54.e9. [Google Scholar] [CrossRef]
- Rule, W.; Timmerman, R.; Tong, L.; Abdulrahman, R.; Meyer, J.; Boike, T.; Schwarz, R.E.; Weatherall, P.; Cho, L.C. Phase i dose-escalation study of stereotactic body radiotherapy in patients with hepatic metastases. Ann. Surg. Oncol. 2011, 18, 1081–1087. [Google Scholar] [CrossRef]
- Meyer, J.J.; Foster, R.D.; Lev-Cohain, N.; Yokoo, T.; Dong, Y.; Schwarz, R.E.; Rule, W.; Tian, J.; Xie, Y.; Hannan, R.; et al. A Phase I Dose-Escalation Trial of Single-Fraction Stereotactic Radiation Therapy for Liver Metastases. Ann. Surg. Oncol. 2016, 23, 218–224. [Google Scholar] [CrossRef]
- Yuan, Z.Y.; Meng, M.B.; Liu, C.L.; Wang, H.H.; Jiang, C.; Song, Y.C.; Zhuang, H.Q.; Yang, D.; Wang, J.S.; Wei, W.; et al. Stereotactic body radiation therapy using the CyberKnife® system for patients with liver metastases. Onco. Targets. Ther. 2014, 7, 915–923. [Google Scholar] [CrossRef]
- Andratschke, N.; Alheid, H.; Allgäuer, M.; Becker, G.; Blanck, O.; Boda-Heggemann, J.; Brunner, T.; Duma, M.; Gerum, S.; Guckenberger, M.; et al. The SBRT database initiative of the German Society for Radiation Oncology (DEGRO): patterns of care and outcome analysis of stereotactic body radiotherapy (SBRT) for liver oligometastases in 474 patients with 623 metastases. BMC Cancer 2018, 18, 283. [Google Scholar] [CrossRef] [PubMed]
- Mahadevan, A.; Blanck, O.; Lanciano, R.; Peddada, A.; Sundararaman, S.; D’Ambrosio, D.; Sharma, S.; Perry, D.; Kolker, J.; Davis, J. Stereotactic Body Radiotherapy (SBRT) for liver metastasis – clinical outcomes from the international multi-institutional RSSearch® Patient Registry. Radiat. Oncol. 2018, 13, 26. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.T.; Kim, J.J.; Dinniwell, R.; Brierley, J.; Lockwood, G.; Wong, R.; Cummings, B.; Ringash, J.; Tse, R.V.; Knox, J.J.; et al. Phase i study of individualized stereotactic body radiotherapy of liver metastases. J. Clin. Oncol. 2009, 27, 1585–1591. [Google Scholar] [CrossRef] [PubMed]
- Rusthoven, K.E.; Kavanagh, B.D.; Burri, S.H.; Chen, C.; Cardenes, H.; Chidel, M.A.; Pugh, T.J.; Kane, M.; Gaspar, L.E.; Schefter, T.E. Multi-Institutional Phase I/II Trial of Stereotactic Body Radiation Therapy for Lung Metastases. J. Clin. Oncol. 2009, 27, 1579–1584. [Google Scholar] [CrossRef] [PubMed]
- Michalski, C.W.; Erkan, M.; Hüser, N.; Müller, M.W.; Hartel, M.; Friess, H.; Kleeff, J. Resection of Primary Pancreatic Cancer and Liver Metastasis: A Systematic Review. Dig. Surg. 2008, 25, 473–480. [Google Scholar] [CrossRef]
- Petrelli, F.; Coinu, A.; Borgonovo, K.; Cabiddu, M.; Ghilardi, M.; Lonati, V.; Aitini, E.; Barni, S. FOLFIRINOX-based neoadjuvant therapy in borderline resectable or unresectable pancreatic cancer: A meta-analytical review of published studies. Pancreas 2015, 44, 515–521. [Google Scholar] [CrossRef]
- Ogawa, K.; Karasawa, K.; Ito, Y.; Ogawa, Y.; Jingu, K.; Onishi, H.; Aoki, S.; Wada, H.; Kokubo, M.; Etoh, H.; et al. Intraoperative Radiotherapy for Resected Pancreatic Cancer: A Multi-Institutional Retrospective Analysis of 210 Patients. Int. J. Radiat. Oncol. 2010, 77, 734–742. [Google Scholar] [CrossRef]
- Cai, S.; Hong, T.S.; Goldberg, S.I.; Fernandez-del Castillo, C.; Thayer, S.P.; Ferrone, C.R.; Ryan, D.P.; Blaszkowsky, L.S.; Kwak, E.L.; Willett, C.G.; et al. Updated long-term outcomes and prognostic factors for patients with unresectable locally advanced pancreatic cancer treated with intraoperative radiotherapy at the Massachusetts General Hospital, 1978 to 2010. Cancer 2013, 119, 4196–4204. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Kobayashi, K.; Ogura, Y.; Nakamura, K.; Nakano, K.; Mizumoto, K.; Tanaka, M. Radiation therapy, bypass operation and celiac plexus block in patients with unresectable locally advanced pancreatic cancer. Hepatogastroenterology. 2005, 52, 1605–1612. [Google Scholar]
- Petrelli, F.; Inno, A.; Barni, S.; Ghidini, A.; Labianca, R.; Falconi, M.; Reni, M.; Cascinu, S. Borderline resectable pancreatic cancer: More than an anatomical concept. Dig. Liver Dis. 2017, 49, 223–226. [Google Scholar] [CrossRef]
- Collisson, E.A.; Bailey, P.; Chang, D.K.; Biankin, A.V. Molecular subtypes of pancreatic cancer. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Aung, K.L.; Fischer, S.E.; Denroche, R.E.; Jang, G.H.; Dodd, A.; Creighton, S.; Southwood, B.; Liang, S.B.; Chadwick, D.; Zhang, A.; et al. Genomics-driven precision medicine for advanced pancreatic cancer: Early results from the COMPASS trial. Clin. Cancer Res. 2018, 24, 1344–1354. [Google Scholar] [CrossRef]
- Rochefort, P.; Lardy-Cleaud, A.; Sarabi, M.; Desseigne, F.; Cattey-Javouhey, A.; de la Fouchardière, C. Long-Term Survivors in Metastatic Pancreatic Ductal Adenocarcinoma: A Retrospective and Matched Pair Analysis. Oncologist 2019. [Google Scholar] [CrossRef] [PubMed]
- Al-Batran, S.-E.; Goetze, T.O.; Mueller, D.W.; Vogel, A.; Winkler, M.; Lorenzen, S.; Novotny, A.; Pauligk, C.; Homann, N.; Jungbluth, T.; et al. The RENAISSANCE (AIO-FLOT5) trial: Effect of chemotherapy alone vs. chemotherapy followed by surgical resection on survival and quality of life in patients with limited-metastatic adenocarcinoma of the stomach or esophagogastric junction—A phase III trial. BMC Cancer 2017, 17, 893. [Google Scholar] [CrossRef] [PubMed]
| Name (Year) Country | N | Patient Characteristics | Treatment in Addition to Surgery | Surgery Details | Morbidity Rate % | Complication Type (%) | Mortality Rate % | Median OS Months |
|---|---|---|---|---|---|---|---|---|
| Hackert [12] (2016) Germany | 128 | - mean age 61 - 48% male - res of liver (n = 85) or ILN mets (n = 43) | - neo-adj treatment given n = 20, (16%) - adj CT completed n = 73 (57%), incomplete in n = 22 (17%) and unk for n = 33 (26%) | - ILN + sync pancreatic res (n = 43) - 72.9% primary + sync liver res - 22.4% meta liver res (mean of 18.4 months after primary) | - 45 for sync res - 22 for meta res | wound infection n = 14 (11), percutaneous drainage n = 11 (9), delayed gastric emptying n = 11 (9) | - 3 for sync res - 4 for meta res | - 12.3 from liver res sync or meta - 12.3 for ILN res |
| Crippa [13] (2016) Italy | 4 | - median age 65- 60% male - all treated with CT - res of primary only (n = 7) - sync liver res (n = 3) - meta liver res (n = 1) - 1 lesion (n = 3) - 2 lesions (n = 1) | pre-op CT for the 11 res pts - FOLFIRINOX n = 3 - GEMOX n = 2 - PEXG n = 4 - PDXG n = 1 - PEFG n = 1 | - res: median 12 months from diagnosis (range 6–20 months) - sync liver res (n = 3) - meta liver res (n = 1) | - 27 | - fistula, n = 2 (18) - post-op pneumonia, n = 1 (9) | - 0 | - 39 for res pts (n = 11) vs. 11 without res(n = 116), p < 0.0001 - 39 for pts proceeding to res (n = 11) vs. 12 for pts with partial response to CT managed without res (n = 45), p < 0.0001 - 11 for whole population (n = 127) |
| Klein [14] (2012) Germany | 22 | - mean age 58 - male 64% - incidental liver mets at surgery | -adj CT with gemcitabine in all pts | - Sync liver res in all pts | - 18 | - fistula, n = 2 (9) - hemorrhage, n = 2 (9) | - 0 | - 7.5 in pts having sync liver res (n = 22) vs. 14.4 in control group having res of primary alone (n = 22), p = 0.15 |
| Dunschede [15] (2010) Germany | 13 | sync liver res pts - mean age 55 - 56% male - median number of liver mets = 3 (range 1–5) meta liver res pts - mean age 42–50% male - median number of liver mets = 1.75 (1–2) | - 2 pts had adj CT after primary res | - sync liver res pts (n = 9) - meta liver res pts (n = 4) | - 33 for sync res - 0 for meta res | NA | - 0 for sync res - 0 for meta res | - 8 in sync res group (n = 9) vs. 11 in the CT alone group (n = 10) - 31 from detection of mets in meta res group (n = 4) vs. 11 in the CT alone group (n = 10) |
| Frigerio [16] (2017) Italy | 24 | - mean age 58 - male 46% - number of liver mets at baseline: 1, n = 5 2, n = 5 multiple n = 14 | pre-op CT in all pts: - gemcitabine n = 5 - FOLFIRINOX n = 16 - gemcitabine + nab-paclitaxel n = 3 - adj CT in 15/24 (63%) | - surgery only for pts with downstaging after CT with disappearance of liver lesions - If liver lesion evident at surgery, fine needle aspiration and surgery aborted if positive - R0 res in 21/24 (88%) | - 63 | - grade B/C -fistula n = 5 (21), - bleeding n = 1 (4) - sepsis n = 3 (13) | - 0 | - 56 |
| De Jong [17] (2010) USA | 126 | - PDAC n = 42 - others n = 84 - median age 56- male 59% - median number of treated liver mets = 2 (range 1–15) | - res only n = 57 (45%) - res + ablation n = 14 (11%) - ablation only n = 10 (8%) - TACE only n = 6 (5%) - whole liver RT n = 28 (22%) - unk n = 11 (9%) | - sync liver-directed therapy with primary res n = 57 - staged primary res and liver -directed therapy n = 69 | 34 overall - sync n = 15 (26) - staged n = 28 (41) | - liver abscess n = 14 (11) - surgical site infection n = 11 (9) - sepsis n = 8 (6) | 2 overall - sync n = 1 (2) - staged n = 2 (3) | - 17.7 for PDAC + liver-directed therapy - 20.1 for whole population |
| Gleisner [18] (2007) USA | 22 | - mean age 65 - 46% male - PDAC n = 17 - others n = 5 | 6 pts had adj CT - 5-fluorourcil (n = 3) - gemcitabine (n = 3) | - all sync liver res (n = 22) | 46 | - urinary retention n = 2 (9) - delayed gastric emptying n = 2 (9) - fistula n = 2 (9) | 9 | - 5.9 for PDAC vs. 9.9 for non-PDAC, p = 0.43 |
| Adam [19] (2006) France | 40 | - PDAC n = 40 - mean age for all pts = 53 (multiple primary tumors) | NA | NA | NA | NA | NA | for PDAC: - 20 - 25% 5 year survival rate |
| Andreou [20] (2018) Germany | 76 | - median age 64 - 60% male - 36% multiple liver mets - median number of mets = 1 (range 1–5) | - pre-op CT n = 4, (5%) - adj CT n = 55, (72%) | - all sync liver res (n = 76) | 50 | - fistula n = 13 (17) - hemorrhage n = 5 (7) | 5 | survival rates: - 41% 1 year - 13% 3 year - 7% 5 year |
| Wright [21] (2016) USA | 11 | - 23 pts had primary res - mean age 58 - 61% male - 11/23 had metastasectomy - liver res n = 9 - lung res n = 2 | pre-op CT in all pts: - FOLFIRINOX n = 14 (61%) - gemcitabine based n = 9 (39%) | - median time from diagnosis to surgery 9.7 months for all 23 pts | - 13 | NA | - 0 | - 18.2 from time of surgery survival rates: - 72.7% 1 year - 21.5% 3 year |
| Kandel [22] (2018) USA | 6 | - median age 64 - 100% male | - pre-op CT n = 5 - pre-op CT + RT n = 1 - adj CT n = 5 - adj CT + RT n = 1 | - sync liver res n = 1 - sync liver res + RFA to liver n = 2 - radio-embolization to liver only n = 1 - RFA only for lung mets n = 2 | NA | NA | NA | - 33 for surgery (n = 6) vs. 11.8 in patients with M1 disease and no surgery (n = 18), p = 0.01 |
| Bahra [23] (2015) Germany | 21 | - median age 60 - 58% male - 21 had liver res | - all pts had gemcitabine based adj CT | - sync liver res (n = 21) | 20 | - fistula grade C n = 3 (7) - hemorrhage N = 2 (4) | 2 | - 10.4 for cytoreductive surgery + gemcitabine based CT vs. 7.2 for CT alone p = 0.009 |
| Zanini [24] (2015) Italy | 15 | - median age 55 - 53% male - single lesions n = 9 (60%) - multiple lesions n = 6 (30%) | - all pts had gemcitabine based adj CT | - 11 had sync liver res - 4 had meta liver res. - median disease-free interval between primary surgery and diagnosis of metastases 8 months | 60 | - fistula grade B/C n = 2 (13) | 0 | - 9.1 -11.4 for meta vs. 8.3 for sync, p = 0.038 |
| Tachezy [25] (2016) 6 European pancreas centres | 69 | - median age 65 - 57% male - median number of mets = 2 (range 1–11) | - no pre -op CT n = 59 pre-op CT: - gemcitabine n = 3 - FOLFIRINOX n = 4 - unk n = 2 - RFA n = 1 - Adj CT: - gemcitabine n = 35 - FOLFIRINOX n = 3 - other CT n = 5 - none n = 11 | - 69 sync liver res | 68 | - fistula grade B/C n = 9 (13) - wound infection n = 12 (17) - hemorrhage n = 6 (9) | 1% | - 14.5 for sync res vs. 7.5 for a control group having no res p < 0.001 |
| Slotta [26] (2014) Germany | 13 | - multiple different primary tumor types, PDAC n = 13 - mean age for whole population = 59 (multiple primary tumors) and 44% male | NA | NA | NA | NA | NA | - 8.2 for PDAC |
| Schiergens [27] (2016) Germany | 19 | - multiple different primary tumor types, PDAC n = 13 - mean age for the 43 pts with gastrointestinal tumors = 64, 53% male | NA | NA | NA | NA | NA | - 7 for PDAC |
| Shrikhande [28] (2007) Germany | 29 | - median age 65 - 38% male - incidental liver mets discovered at surgery n = 14 - liver mets discovered after surgery n = 15 | - neoadjuvant CT + RT n = 1 - adj CT n = 23 gemcitabine n = 13 fluorouracil n = 6 experimental tumor vaccine n = 2 other CT n = 2 | - liver res n = 10 - ILN res n = 10 - peritoneal n = 8 - ILN+liver res n = 1 | 24% | - fistula n = 2 (7) - delayed gastric emptying n = 1 (3) - intra-abdominal abscess n = 2 (7) - hemorrhage + delayed gastric emptying n = 1 (3) | 0 | - 13.8 - 1 year survival rate of 58.9% - 27 for ILN mets - 11.4 for liver mets - 12.9 for peritoneal mets |
| Name(Year) | N | Design | Histology % Metastatic Site % | Preop CT/ RT Yes/No (%) | Type of Procedure % | Morbidity Rate % | Complication Type % | mPFS Mo | mOS Mo |
|---|---|---|---|---|---|---|---|---|---|
| Park [29] (2012) Korea | 34 | retro | - PDAC 100 - liver mets 100 | no (100) | - intraop orpostop RFA 100 - surgery 100 | NA | - pleural effusion 8.8 - liver abscess 3 | 2 (range: 0–32) | 18 14 (OS after liver mets) |
| Hua [30] (2017) China | 102 | retro | - PDAC 100 - liver mets 100 | no (100) | RFA 100 | 9.8 | - vomiting 2 - biliary-cardiac reflex 2 - abdominal pain 4 - fever 2 | NA | 11.4 |
| Martin [33] (2015) USA | 200 | pros | - PDAC 100 - locally advanced 100 | CT (100) CT + RT (52) | - IRE alone (75) - pancreatic res plus IRE (25) | 18 mortality 1.5 | - gastrointestinal 23 - liver 10 - infection 9 - vascular 5.5 | 12 | 24.9 |
| Hong [35] (2018) USA | 7 | retro | - PDAC 100 - liver 57.1 - omentum 42.8 - peritoneum 42.8 | CT (100) | - IRE plus resection 86 - IRE on mets 14.3 | NA | NA | NA | 16 |
| Kluger [34] (2016) USA | 50 | pros | - PDAC 94 - neuroen 6 | CT (94) RT (83) | IRE 100 | 38 mortality 11 | NA | NA | 12 |
| Yuan [40] (2014) China | 57 | retro | PDAC 14 | NA | SBRT 100 | fatigue, nausea, vomiting, and changes in liver function tests | NA | NA | 2-year OS: 38 |
| Andratschke [41] (2018) Switzerland | 474 | retro | - PDAC 5.1 - liver 100 | NA for PDAC | SBRT 100 | fatigue, nausea, and diarrhea | Hepatitis 0.4 Liver fibrosis 1.4 Necrotic reaction 0.4 | NA | NA for PDAC |
| Mahadevan [42] (2018) USA | 427 | retro | - PDAC 4.9 - liver 100 | NA for PDAC | SBRT 100 | NA | NA | NA | 6 1-year OS 18% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghidini, M.; Petrillo, A.; Salati, M.; Khakoo, S.; Varricchio, A.; Tomasello, G.; Grossi, F.; Petrelli, F. Surgery or Locoregional Approaches for Hepatic Oligometastatic Pancreatic Cancer: Myth, Hope, or Reality? Cancers 2019, 11, 1095. https://doi.org/10.3390/cancers11081095
Ghidini M, Petrillo A, Salati M, Khakoo S, Varricchio A, Tomasello G, Grossi F, Petrelli F. Surgery or Locoregional Approaches for Hepatic Oligometastatic Pancreatic Cancer: Myth, Hope, or Reality? Cancers. 2019; 11(8):1095. https://doi.org/10.3390/cancers11081095
Chicago/Turabian StyleGhidini, Michele, Angelica Petrillo, Massimiliano Salati, Shelize Khakoo, Antonio Varricchio, Gianluca Tomasello, Francesco Grossi, and Fausto Petrelli. 2019. "Surgery or Locoregional Approaches for Hepatic Oligometastatic Pancreatic Cancer: Myth, Hope, or Reality?" Cancers 11, no. 8: 1095. https://doi.org/10.3390/cancers11081095
APA StyleGhidini, M., Petrillo, A., Salati, M., Khakoo, S., Varricchio, A., Tomasello, G., Grossi, F., & Petrelli, F. (2019). Surgery or Locoregional Approaches for Hepatic Oligometastatic Pancreatic Cancer: Myth, Hope, or Reality? Cancers, 11(8), 1095. https://doi.org/10.3390/cancers11081095






