Impact of Baseline ALBI Grade on the Outcomes of Hepatocellular Carcinoma Patients Treated with Lenvatinib: A Multicenter Study
Abstract
1. Introduction
2. Methods
2.1. Patients
2.2. Treatment Protocol
2.3. Evaluation of Treatment Response
2.4. Efficacy Outcome and ALBI Score
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. ORR and DCR According to ALBI Grade and Child–Pugh Score
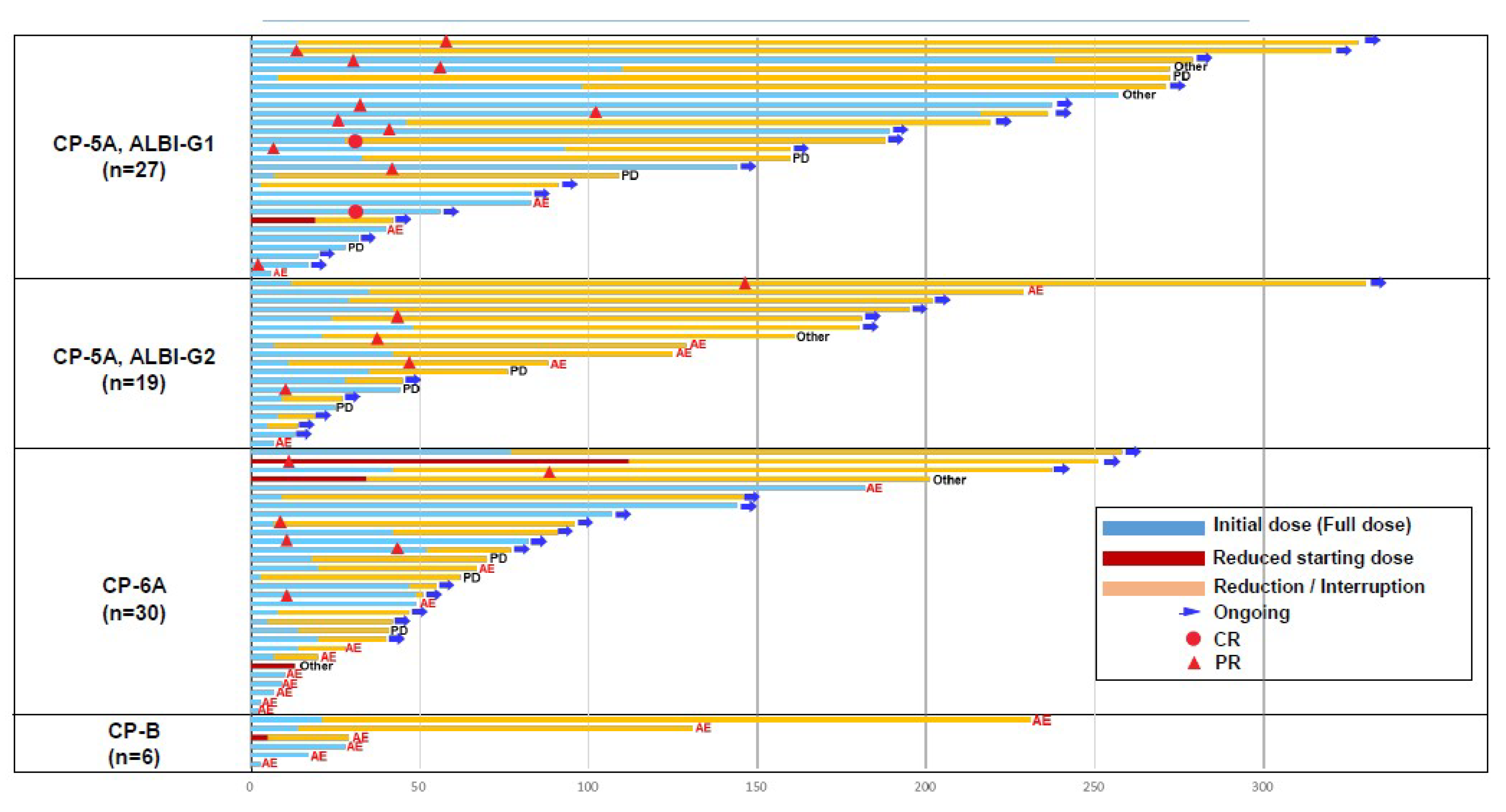
3.3. Dose Reduction, Interruption, and Discontinuation due to AEs according to Liver Function Status
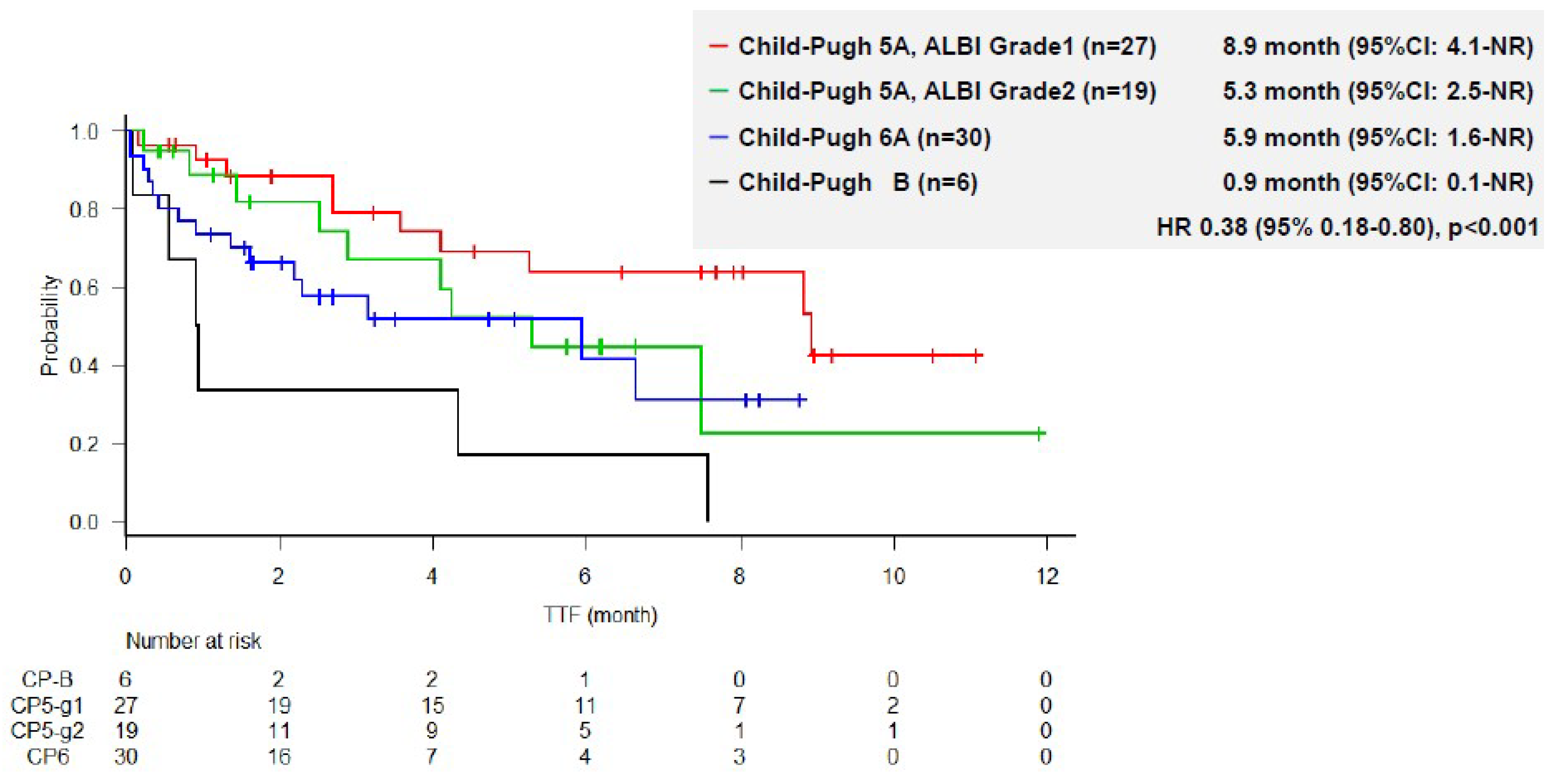
3.4. TTF and Duration of Treatment According to ALBI Grade and Child–Pugh Score
3.5. Univariate and Multivariate Regression Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- El-Serag, H.B.; Rudolph, K.L. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Balogh, J.; Victor, D., 3rd; Asham, E.H.; Burroughs, S.G.; Boktour, M.; Saharia, A.; Li, X.; Ghobrial, R.M.; Monsour, H.P., Jr. Hepatocellular carcinoma: A review. J. Hepatocell. Carcinoma 2016, 3, 41–53. [Google Scholar] [CrossRef] [PubMed]
- EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [CrossRef] [PubMed]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.L.; Kang, Y.K.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Kudo, M.; Izumi, N.; Sakamoto, M.; Matsuyama, Y.; Ichida, T.; Nakashima, O.; Matsui, O.; Ku, Y.; Kokudo, N.; Makuuchi, M. Survival Analysis over 28 Years of 173,378 Patients with Hepatocellular Carcinoma in Japan. Liver Cancer 2016, 5, 190–197. [Google Scholar] [CrossRef]
- Kudo, M. Locoregional Therapy for Hepatocellular Carcinoma. Liver Cancer 2015, 4, 163–164. [Google Scholar] [CrossRef]
- Matsui, J.; Funahashi, Y.; Uenaka, T.; Watanabe, T.; Tsuruoka, A.; Asada, M. Multi-kinase inhibitor E7080 suppresses lymph node and lung metastases of human mammary breast tumor MDA-MB-231 via inhibition of vascular endothelial growth factor-receptor (VEGF-R) 2 and VEGF-R3 kinase. Clin. Cancer Res. 2008, 14, 5459–5465. [Google Scholar] [CrossRef]
- Matsui, J.; Yamamoto, Y.; Funahashi, Y.; Tsuruoka, A.; Watanabe, T.; Wakabayashi, T.; Uenaka, T.; Asada, M. E7080, a novel inhibitor that targets multiple kinases, has potent antitumor activities against stem cell factor producing human small cell lung cancer H146, based on angiogenesis inhibition. Int. J. Cancer 2008, 122, 664–671. [Google Scholar] [CrossRef]
- Yamada, K.; Yamamoto, N.; Yamada, Y.; Nokihara, H.; Fujiwara, Y.; Hirata, T.; Koizumi, F.; Nishio, K.; Koyama, N.; Tamura, T. Phase I dose-escalation study and biomarker analysis of E7080 in patients with advanced solid tumors. Clin. Cancer Res. 2011, 17, 2528–2537. [Google Scholar] [CrossRef]
- Boss, D.S.; Glen, H.; Beijnen, J.H.; Keesen, M.; Morrison, R.; Tait, B.; Copalu, W.; Mazur, A.; Wanders, J.; O’Brien, J.P.; et al. A phase I study of E7080, a multitargeted tyrosine kinase inhibitor, in patients with advanced solid tumours. Br. J. Cancer 2012, 106, 1598–1604. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Okusaka, T.; Mitsunaga, S.; Ueno, H.; Tamai, T.; Suzuki, T.; Hayato, S.; Kadowaki, T.; Okita, K.; Kumada, H. Safety and Pharmacokinetics of Lenvatinib in Patients with Advanced Hepatocellular Carcinoma. Clin. Cancer Res. 2016, 22, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M. Albumin-Bilirubin Grade and Hepatocellular Carcinoma Treatment Algorithm. Liver Cancer 2017, 6, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.J.; Berhane, S.; Kagebayashi, C.; Satomura, S.; Teng, M.; Reeves, H.L.; O’Beirne, J.; Fox, R.; Skowronska, A.; Palmer, D.; et al. Assessment of liver function in patients with hepatocellular carcinoma: A new evidence-based approach-the ALBI grade. J. Clin. Oncol. 2015, 33, 550–558. [Google Scholar] [CrossRef]
- Hiraoka, A.; Kumada, T.; Kudo, M.; Hirooka, M.; Tsuji, K.; Itobayashi, E.; Kariyama, K.; Ishikawa, T.; Tajiri, K.; Ochi, H.; et al. Albumin-Bilirubin (ALBI) Grade as Part of the Evidence-Based Clinical Practice Guideline for HCC of the Japan Society of Hepatology: A Comparison with the Liver Damage and Child–Pugh Classifications. Liver Cancer 2017, 6, 204–215. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Michitaka, K.; Kumada, T.; Kudo, M. ALBI Score as a Novel Tool in Staging and Treatment Planning for Hepatocellular Carcinoma: Advantage of ALBI Grade for Universal Assessment of Hepatic Function. Liver Cancer 2017, 6, 377–379. [Google Scholar] [CrossRef] [PubMed]
- Fang, K.C.; Kao, W.Y.; Su, C.W.; Chen, P.C.; Lee, P.C.; Huang, Y.H.; Huo, T.I.; Chang, C.C.; Hou, M.C.; Lin, H.C.; et al. The Prognosis of Single Large Hepatocellular Carcinoma Was Distinct from Barcelona Clinic Liver Cancer Stage A or B: The Role of Albumin-Bilirubin Grade. Liver Cancer 2018, 7, 335–358. [Google Scholar] [CrossRef]
- National Cancer Institute. Protocol development. Cancer therapy evaluation program. Available online: https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm (accessed on 21 March 2017).
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef]
- Hiraoka, A.; Kumada, T.; Michitaka, K.; Toyoda, H.; Tada, T.; Ueki, H.; Kaneto, M.; Aibiki, T.; Okudaira, T.; Kawakami, T.; et al. Usefulness of albumin-bilirubin grade for evaluation of prognosis of 2584 Japanese patients with hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2016, 31, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Michitaka, K.; Kumada, T.; Izumi, N.; Kadoya, M.; Kokudo, N.; Kubo, S.; Matsuyama, Y.; Nakashima, O.; Sakamoto, M.; et al. Validation and Potential of Albumin-Bilirubin Grade and Prognostication in a Nationwide Survey of 46,681 Hepatocellular Carcinoma Patients in Japan: The Need for a More Detailed Evaluation of Hepatic Function. Liver Cancer 2017, 6, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Yang, X.; Li, Q.L.; Zhou, Q.X.; Xiong, L.; Wen, Y. A Comparative Study of Albumin-Bilirubin Score with Child–Pugh Score, Model for End-Stage Liver Disease Score and Indocyanine Green R15 in Predicting Posthepatectomy Liver Failure for Hepatocellular Carcinoma Patients. Dig. Dis. 2018, 36, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Su, T.S.; Yang, H.M.; Zhou, Y.; Huang, Y.; Liang, P.; Cheng, T.; Chen, L.; Li, L.Q.; Liang, S.X. Albumin - bilirubin (ALBI) versus Child-Turcotte-Pugh (CTP) in prognosis of HCC after stereotactic body radiation therapy. Radiat. Oncol. 2019, 14, 50. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Kumada, T.; Kudo, M.; Hirooka, M.; Koizumi, Y.; Hiasa, Y.; Tajiri, K.; Toyoda, H.; Tada, T.; Ochi, H.; et al. Hepatic Function during Repeated TACE Procedures and Prognosis after Introducing Sorafenib in Patients with Unresectable Hepatocellular Carcinoma: Multicenter Analysis. Dig. Dis. 2017, 35, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, S.; Chiba, T.; Ooka, Y.; Kanogawa, N.; Motoyama, T.; Suzuki, E.; Tawada, A.; Kanai, F.; Yoshikawa, M.; Yokosuka, O. Efficacy of Sorafenib in Intermediate-Stage Hepatocellular Carcinoma Patients Refractory to Transarterial Chemoembolization. Oncology 2014, 87, 330–341. [Google Scholar] [CrossRef] [PubMed]
- Arizumi, T.; Ueshima, K.; Minami, T.; Kono, M.; Chishina, H.; Takita, M.; Kitai, S.; Inoue, T.; Yada, N.; Hagiwara, S.; et al. Effectiveness of Sorafenib in patients with transcatheter arterial chemoembolization (TACE) refractory and intermediate-stage hepatocellular carcinoma. Liver Cancer 2015, 4, 253–262. [Google Scholar] [CrossRef] [PubMed]
| Parameter | All Patients (n = 82) | |
|---|---|---|
| Median age, years (range) | 71.5 (21–92) | |
| Sex, n | Male/Female | 59/23 |
| Body weight, n | <60 kg/≥60 kg | 36/46 |
| Etiology, n | HBV/HCV/nonBnonC | 12 */37 */34 |
| Starting dose, n | 12 mg/8 mg/4 mg | 37/40/5 |
| Median ALB (g/dL) | 3.6 (2.5–4.4) | |
| Median BIL (mg/dL) | 0.7 (0.2–2.9) | |
| Median PT (%) | 88.0 (14.1–130.0) | |
| Child–Pugh score | 5/6/≥7 | 46/30/6 |
| ALBI Grade | 1/2/3 | 29/52/1 |
| BCLC stage | A/B/C | 7/27/48 |
| Median AFP (ng/mL) | 106.8 (1–4,503,000) | |
| Median DCP (mAU/mL) | 878 (8–538,430) | |
| Median AFP-L3 (%) | 20.9 (0–99.5) | |
| Sorafenib | Naïve/Experience | 50/32 |
| Regorafenib | Naïve/Experience | 70/12 |
| Median number of previous TACE | Child–Pugh A/Child–Pugh B | 2/7 |
| Median number of previous TACE | Child–Pugh 5A, ALBI Grade1/Child–Pugh5A, ALBI Grade2/Child–Pugh 6A | 1/2/4 |
| Liver Function | n | CR | PR | SD | PD | ORR | DCR |
|---|---|---|---|---|---|---|---|
| Child–Pugh A | 55 | 2 | 21 | 24 | 8 | 41.8% | 85.5% |
| 5A, ALBI Grade1 | 21 | 2 | 10 | 8 | 1 | 57.1% | 95.2% |
| 5A, ALBI Grade2 | 13 | 0 | 5 | 5 | 3 | 38.5% | 76.9% |
| 6A | 21 | 0 | 6 | 11 | 4 | 28.6% | 81.0% |
| Child–Pugh B | 4 | 0 | 0 | 2 | 2 | 0.0% | 50.0% |
| Total | 59 | 2 | 21 | 26 | 10 | 39.0% | 83.1% |
| Liver Function | n | Reduction or Interruption Due to AE | Discontinuation Due to AE | ||
|---|---|---|---|---|---|
| Child–Pugh A | 76 | 47 | 61.8% | 16 | 21.1% |
| 5A, ALBI Grade1 | 27 | 13 | 48.1% | 3 | 11.1% |
| 5A, ALBI Grade2 | 19 | 15 | 78.9% | 5 | 26.3% |
| 6A | 30 | 19 | 63.3% | 10 | 33.3% |
| Child–Pugh B | 6 | 2 | 33.3% | 6 | 100.% |
| Total | 82 | 49 | 59.8% | 22 | 26.8% |
| Parameters | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| Odds Ratio (95% CI) | p | Odds Ratio (95% CI) | p | |
| ALBI grade | ||||
| Grade 1 | 2.64 (0.81–8.97) | p < 0.1 | 3.32 (1.04–10.50) | p < 0.05 |
| Grade ≥2 | 1 | 1 | ||
| Baseline AFP level | ||||
| AFP < 200 ng/mL | 2.89 (0.90–9.90) | p < 0.1 | 3.58 (1.14–11.30) | p < 0.01 |
| AFP ≥ 200 ng/mL | 1 | 1 | ||
| Parameters | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| Odds Ratio (95% CI) | p | Odds Ratio (95% CI) | p | |
| ALBI grade | ||||
| Grade 1 | 0.19 (0.03–0.76) | p < 0.01 | 0.22 (0.06–0.69) | p < 0.01 |
| Grade ≥2 | 1 | 1 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ueshima, K.; Nishida, N.; Hagiwara, S.; Aoki, T.; Minami, T.; Chishina, H.; Takita, M.; Minami, Y.; Ida, H.; Takenaka, M.; et al. Impact of Baseline ALBI Grade on the Outcomes of Hepatocellular Carcinoma Patients Treated with Lenvatinib: A Multicenter Study. Cancers 2019, 11, 952. https://doi.org/10.3390/cancers11070952
Ueshima K, Nishida N, Hagiwara S, Aoki T, Minami T, Chishina H, Takita M, Minami Y, Ida H, Takenaka M, et al. Impact of Baseline ALBI Grade on the Outcomes of Hepatocellular Carcinoma Patients Treated with Lenvatinib: A Multicenter Study. Cancers. 2019; 11(7):952. https://doi.org/10.3390/cancers11070952
Chicago/Turabian StyleUeshima, Kazuomi, Naoshi Nishida, Satoru Hagiwara, Tomoko Aoki, Tomohiro Minami, Hirokazu Chishina, Masahiro Takita, Yasunori Minami, Hiroshi Ida, Mamoru Takenaka, and et al. 2019. "Impact of Baseline ALBI Grade on the Outcomes of Hepatocellular Carcinoma Patients Treated with Lenvatinib: A Multicenter Study" Cancers 11, no. 7: 952. https://doi.org/10.3390/cancers11070952
APA StyleUeshima, K., Nishida, N., Hagiwara, S., Aoki, T., Minami, T., Chishina, H., Takita, M., Minami, Y., Ida, H., Takenaka, M., Sakurai, T., Watanabe, T., Morita, M., Ogawa, C., Hiraoka, A., Johnson, P., & Kudo, M. (2019). Impact of Baseline ALBI Grade on the Outcomes of Hepatocellular Carcinoma Patients Treated with Lenvatinib: A Multicenter Study. Cancers, 11(7), 952. https://doi.org/10.3390/cancers11070952










