Use of Machine-Learning Algorithms in Intensified Preoperative Therapy of Pancreatic Cancer to Predict Individual Risk of Relapse
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Elegibility
2.2. Neoadjuvant Therapy
2.3. Histological Data
2.4. Postoperative Therapy and Follow Up
2.5. Toxicity
2.6. Statistical Analysis
2.7. Machine-Learning Algorithms
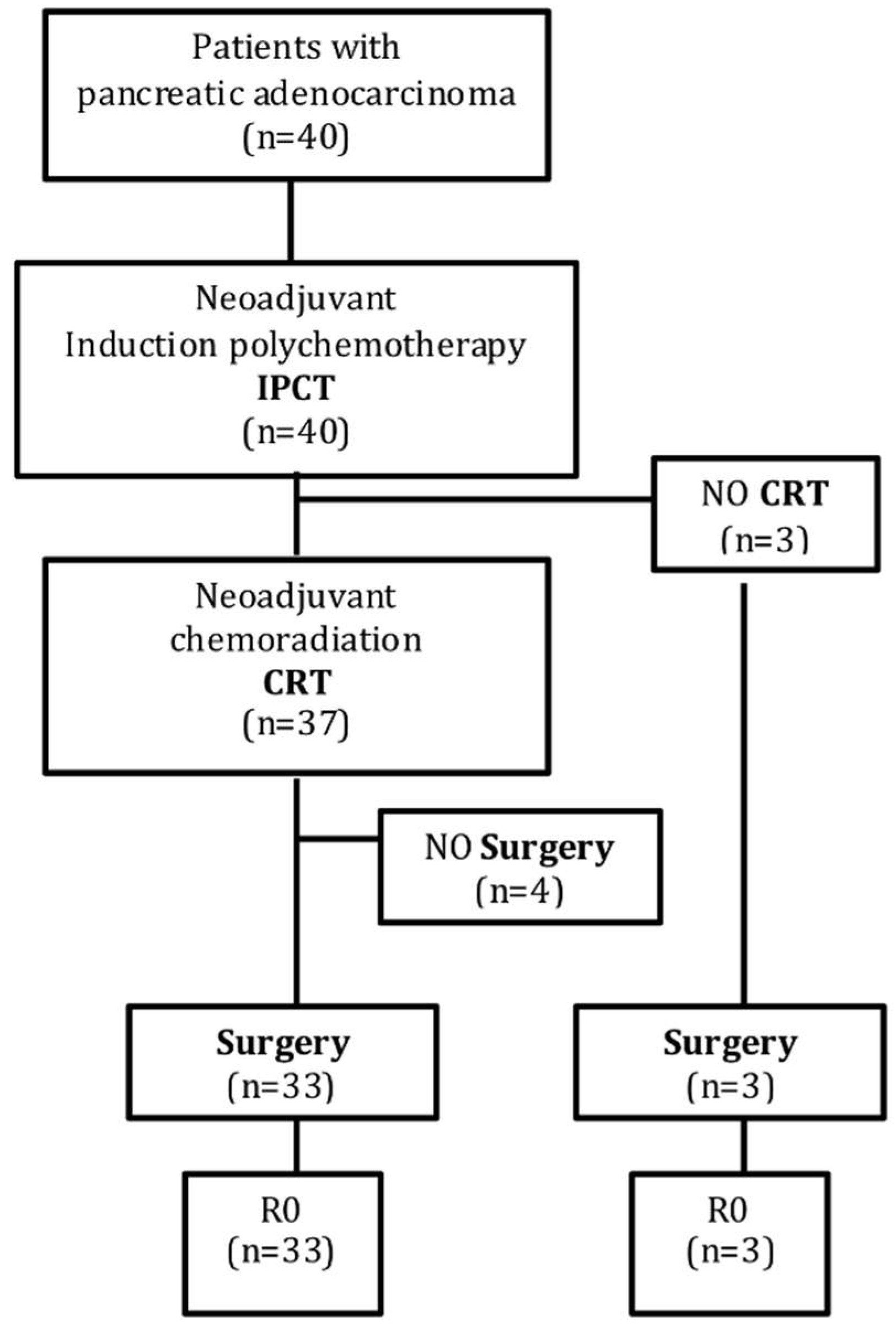
3. Results
3.1. Clinical Data
3.1.1. Patients Characteristics
3.1.2. Multimodality Therapy Completion
3.1.3. Surgical Outcome and Pathological Results
3.1.4. Toxicity Profile
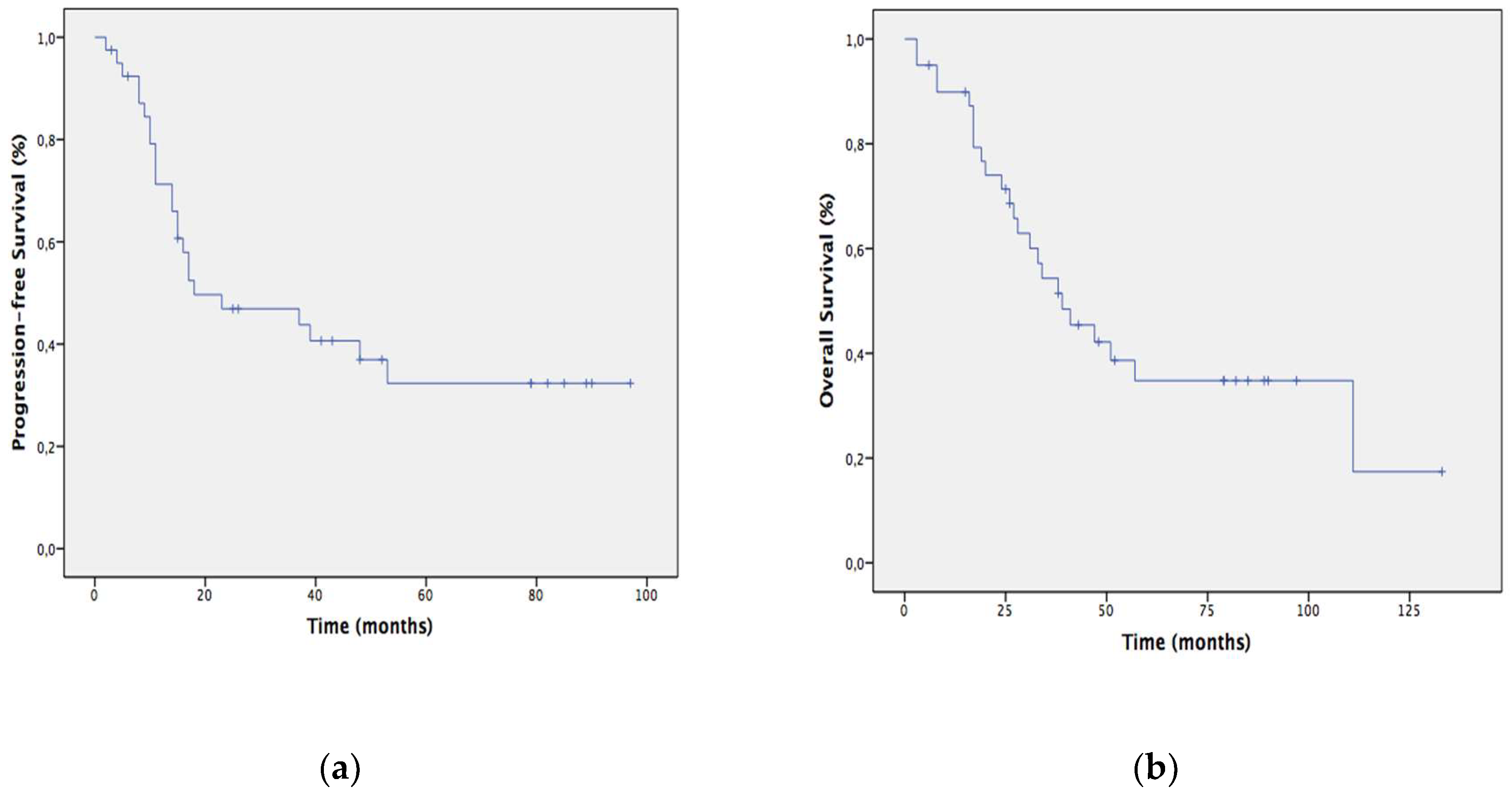
3.1.5. Patients Long-Term Outcome
3.2. Predictive Population Model
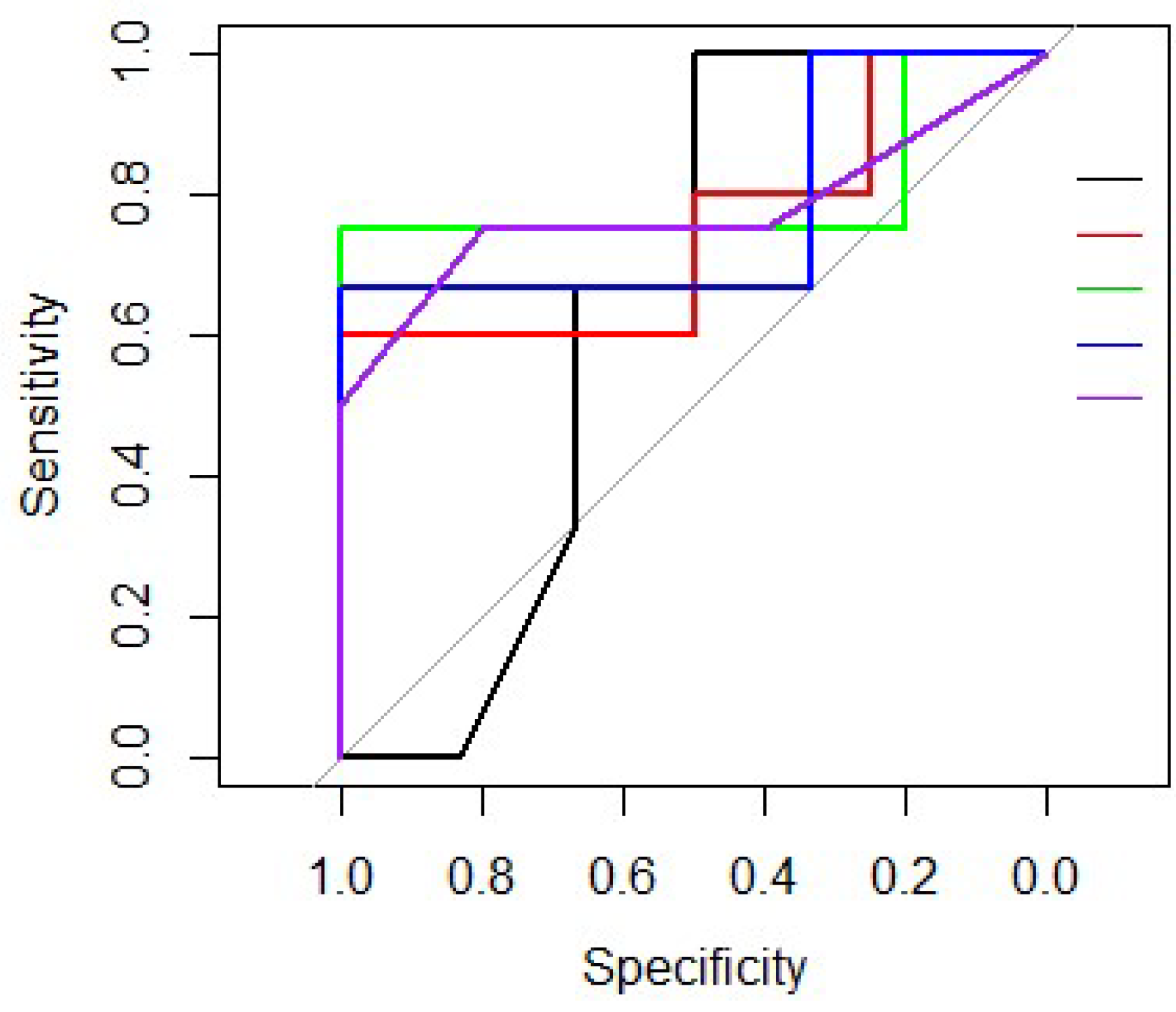
3.2.1. Model Development
3.2.2. External Validation of the Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Different Machine-Learning Techniques Used to Perform a Predictive Population Model
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting Cancer Incidence and Deaths to 2030: The Unexpected Burden of Thyroid, Liver, and Pancreas Cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Oettle, H.; Neuhaus, P.; Hochhaus, A.; Hartmann, J.T.; Gellert, K.; Ridwelski, K.; Niedergethmann, M.; Zülke, C.; Fahlke, J.; Arning, M.B.; et al. Adjuvant Chemotherapy With Gemcitabine and Long-term Outcomes Among Patients With Resected Pancreatic Cancer. JAMA 2013, 310, 1473. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Stocken, D.D.; Bassi, C.; Ghaneh, P.; Cunningham, D.; Goldstein, D.; Padbury, R.; Moore, M.J.; Gallinger, S.; Mariette, C.; et al. Adjuvant Chemotherapy With Fluorouracil Plus Folinic Acid vs. Gemcitabine Following Pancreatic Cancer Resection. JAMA 2010, 304, 1073. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Stocken, D.D.; Friess, H.; Bassi, C.; Dunn, J.A.; Hickey, H.; Beger, H.; Fernandez-Cruz, L.; Dervenis, C.; Lacaine, F.; et al. A Randomized Trial of Chemoradiotherapy and Chemotherapy after Resection of Pancreatic Cancer. N. Engl. J. Med. 2004, 350, 1200–1210. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Palmer, D.H.; Ghaneh, P.; Psarelli, E.E.; Valle, J.W.; Halloran, C.M.; Faluyi, O.; O’Reilly, D.A.; Cunningham, D.; Wadsley, J.; et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): A multicentre, open-label, randomised, phase 3 trial. Lancet 2017, 389, 1011–1024. [Google Scholar] [CrossRef]
- Tempero, M.A.; Malafa, M.P.; Al-Hawary, M.; Asbun, H.; Bain, A.; Behrman, S.W.; Benson, A.B., 3rd; Binder, E.; Cardin, D.B.; Cha, C.; et al. Pancreatic Adenocarcinoma, Version 2.2017, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2017, 15, 1028–1061. [Google Scholar] [CrossRef]
- Lutz, M.P.; Zalcberg, J.R.; Ducreux, M.; Aust, D.; Bruno, M.J.; Büchler, M.W.; Delpero, J.R.; Gloor, B.; Glynne-Jones, R.; Hartwig, W.; et al. 3rd St. Gallen EORTC Gastrointestinal Cancer Conference: Consensus recommendations on controversial issues in the primary treatment of pancreatic cancer ScienceDirect. Eur. J. Cancer. 2017, 79, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Khorana, A.A.; Mangu, P.B.; Berlin, J.; Engebretson, A.; Hong, T.S.; Maitra, A.; Mohile, S.G.; Mumber, M.; Schulick, R.; Shapiro, M.; et al. Potentially Curable Pancreatic Cancer: American Society of Clinical Oncology Clinical Practice Guideline Update. J. Clin. Oncol. 2017, 35, 2324–2328. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.H.; Ellenberg, S.S. Pancreatic Cancer: Adjuvant Combined Radiation and Chemotherapy Following Curative Resection. Arch. Surg. 1985, 120, 899–903. [Google Scholar] [CrossRef]
- Klinkenbijl, J.H.; Jeekel, J.; Sahmoud, T.; van Pel, R.; Couvreur, M.L.; Veenhof, C.H.; Arnaud, J.P.; Gonzalez, D.G.; de Wit, L.T.; Hennipman, A.; et al. Adjuvant radiotherapy and 5-fluorouracil after curative resection of cancer of the pancreas and periampullary region: Phase III trial of the EORTC gastrointestinal tract cancer cooperative group. Ann. Surg. 1999, 230, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Mayo, S.C.; Gilson, M.M.; Herman, J.M.; Cameron, J.L.; Nathan, H.; Edil, B.H.; Choti, M.A.; Schulick, R.D.; Wolfgang, C.L.; Pawlik, T.M. Management of Patients with Pancreatic Adenocarcinoma: National Trends in Patient Selection, Operative Management, and Use of Adjuvant Therapy. J. Am. Coll. Surg. 2012, 214, 33–45. [Google Scholar] [CrossRef]
- Van Tienhoven, G.; Versteijne, E.; Suker, M.; Groothuis, K.B.C.; Busch, O.R.; Bonsing, B.A.; de Hingh, I.H.J.T.; Festen, S.; Patijn, G.A.; Vos-Geelen, J.; et al. Preoperative chemoradiotherapy versus immediate surgery for resectable and borderline resectable pancreatic cancer (PREOPANC-1): A randomized, controlled, multicenter phase III trial. J. Clin. Oncol. 2018, 36. [Google Scholar] [CrossRef]
- De W Marsh, R.; Talamonti, M.S.; Baker, M.S.; Posner, M.; Roggin, K.; Matthews, J.; Catenacci, D.; Kozloff, M.; Polite, B.; Britto, M.; et al. Primary systemic therapy in resectable pancreatic ductal adenocarcinoma using mFOLFIRINOX: A pilot study. J. Surg. Oncol. 2018, 117, 354–362. [Google Scholar] [CrossRef]
- Weiser, M.R.; Landmann, R.G.; Kattan, M.W.; Gonen, M.; Shia, J.; Chou, J.; Paty, P.B.; Guillem, J.G.; Temple, L.K.; Schrag, D.; et al. Individualized prediction of colon cancer recurrence using a nomogram. J. Clin. Oncol. 2008, 26, 380–385. [Google Scholar] [CrossRef]
- Song, W.; Miao, D.-L.; Chen, L. Nomogram for predicting survival in patients with pancreatic cancer. Oncol. Targets Ther. 2018, 11, 539–545. [Google Scholar] [CrossRef]
- Pu, N.; Li, J.; Xu, Y.; Lee, W.; Fang, Y.; Han, X.; Zhao, G.; Zhang, L.; Nuerxiati, A.; Yin, H.; et al. Comparison of prognostic prediction between nomogram based on lymph node ratio and AJCC 8th staging system for patients with resected pancreatic head carcinoma: A SEER analysis. Cancer Manag. Res. 2018, 10, 227–238. [Google Scholar] [CrossRef]
- Bailey, P.; Chang, D.K.; Nones, K.; Johns, A.L.; Patch, A.M.; Gingras, M.C.; Miller, D.K.; Christ, A.N.; Bruxner, T.J.; Quinn, M.C.; et al. Genomic analyses identify molecular subtypes of pancreatic cancer. Nature 2016, 531, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Buil-Bruna, N.; Lopez-Picazo, J.-M.; Martin-Algarra, S.; Troconiz, I.F. Bringing Model-Based Prediction to Oncology Clinical Practice: A Review of Pharmacometrics Principles and Applications. Oncologist 2016, 21, 220–232. [Google Scholar] [CrossRef]
- Bender, B.C.; Schindler, E.; Friberg, L.E. Population pharmacokinetic-pharmacodynamic modelling in oncology: A tool for predicting clinical response. Br. J. Clin. Pharmacol. 2015, 79, 56–71. [Google Scholar] [CrossRef]
- Buil-Bruna, N.; López-Picazo, J.-M.; Moreno-Jiménez, M.; Martín-Algarra, S.; Ribba, B.; Trocóniz, I.F. A Population Pharmacodynamic Model for Lactate Dehydrogenase and Neuron Specific Enolase to Predict Tumor Progression in Small Cell Lung Cancer Patients. AAPS J. 2014, 16, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Buil-Bruna, N.; Sahota, T.; Lopez-Picazo, J.-M.; Moreno-Jiménez, M.; Martín-Algarra, S.; Ribba, B.; Trocóniz, I.F. Early Prediction of Disease Progression in Small Cell Lung Cancer: Toward Model-Based Personalized Medicine in Oncology. Cancer Res. 2015, 75, 2416–2425. [Google Scholar] [CrossRef]
- Kukar, M.; Bešič, N.; Kononenko, I.; Auersperg, M.; Robnik-Šikonja, M. Prognosing the Survival Time of Patients with Anaplastic Thyroid Carcinoma using Machine Learning. In Intelligent Data Analysis in Medicine and Pharmacology; Springer: New Yrok, NY, USA, 1997; pp. 115–129. [Google Scholar]
- Hansson, E.K.; Amantea, M.A.; Westwood, P.; Milligan, P.A.; Houk, B.E.; French, J.; Karlsson, M.O.; Friberg, L.E. PKPD Modeling of VEGF, sVEGFR-2, sVEGFR-3, and sKIT as Predictors of Tumor Dynamics and Overall Survival Following Sunitinib Treatment in GIST. CPT Pharmacomet. Syst. Pharmacol. 2013, 2, e84. [Google Scholar] [CrossRef]
- Desmée, S.; Mentré, F.; Veyrat-Follet, C.; Guedj, J. Nonlinear Mixed-Effect Models for Prostate-Specific Antigen Kinetics and Link with Survival in the Context of Metastatic Prostate Cancer: A Comparison by Simulation of Two-Stage and Joint Approaches. AAPS J. 2015, 17, 691–699. [Google Scholar] [CrossRef]
- Hayward, J.; Alvarez, S.A.; Ruiz, C.; Sullivan, M.; Tseng, J.; Whalen, G. Machine learning of clinical performance in a pancreatic cancer database. Artif. Intell. Med. 2010, 49, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Sadot, E.; Doussot, A.; O’Reilly, E.M.; Lowery, M.A.; Goodman, K.A.; Do, R.K.; Tang, L.H.; Gönen, M.; D’Angelica, M.I.; DeMatteo, R.P.; et al. FOLFIRINOX Induction Therapy for Stage 3 Pancreatic Adenocarcinoma. Ann. Surg. Oncol. 2015, 22, 3512–3521. [Google Scholar] [CrossRef] [PubMed]
- Hess, V.; Pratsch, S.; Potthast, S.; Lee, L.; Winterhalder, R.; Widmer, L.; Cescato, C.; Lohri, A.; Jost, L.; Stillhart, P.; et al. Combining gemcitabine, oxaliplatin and capecitabine (GEMOXEL) for patients with advanced pancreatic carcinoma (APC): A phase I/II trial. Ann. Oncol. 2010, 21, 2390–2395. [Google Scholar] [CrossRef]
- Lassen, U.; Jensen, L.H.; Sorensen, M.; Rohrberg, K.S.; Ujmajuridze, Z.; Jakobsen, A. A Phase I–II dose escalation study of fixed-dose rate gemcitabine, oxaliplatin and capecitabine every two weeks in advanced cholangiocarcinomas. Acta Oncol. 2011, 50, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Combs, S.E.; Habermehl, D.; Kessel, K.; Bergmann, F.; Werner, J.; Brecht, I.; Schirmacher, P.; Jäger, D.; Büchler, M.W.; Debus, J. Intensity modulated radiotherapy as neoadjuvant chemoradiation for the treatment of patients with locally advanced pancreatic cancer. Strahlenther. Onkol. 2013, 189, 738–744. [Google Scholar] [CrossRef]
- Washington, K.; Berlin, J.; Branton, P.; Burgart, L.J.; Carter, D.; Compton, C.; Fitzgibbons, P.; Frankel, W.; Jessup, J.; Tang, L.; et al. Protocol for the Examination of Specimens from Patients with Carcinoma of the Pancreas. Available online: http://www.cap.org (accessed on 4 April 2018).
- Smith, I.C.; Heys, S.D.; Hutcheon, A.W.; Miller, I.D.; Payne, S.; Gilbert, F.J.; Ah-See, A.K.; Eremin, O.; Walker, L.G.; Sarkar, T.K.; et al. Neoadjuvant Chemotherapy in Breast Cancer: Significantly Enhanced Response with Docetaxel. J. Clin. Oncol. 2002, 20, 1456–1466. [Google Scholar] [CrossRef]
- Ishikawa, O.; Ohigashi, H.; Imaoka, S.; Sasaki, Y.; Iwanaga, T.; Matayoshi, Y.; Inoue, T. Is the Long-term Survival Rate Improved by Preoperative Irradiation Prior to Whipple’s Procedure for Adenocarcinoma of the Pancreatic Head? Arch. Surg. 1994, 129, 1075–1080. [Google Scholar] [CrossRef]
- O’Reilly, E.M.; Perelshteyn, A.; Jarnagin, W.R.; Schattner, M.; Gerdes, H.; Capanu, M.; Tang, L.H.; LaValle, J.; Winston, C.; DeMatteo, R.P.; et al. A single-arm, nonrandomized phase II trial of neoadjuvant gemcitabine and oxaliplatin in patients with resectable pancreas adenocarcinoma. Ann. Surg. 2014, 260, 142–148. [Google Scholar] [CrossRef]
- Heinrich, S.; Pestalozzi, B.C.; Schäfer, M.; Weber, A.; Bauerfeind, P.; Knuth, A.; Clavien, P.A. Prospective Phase II Trial of Neoadjuvant Chemotherapy with Gemcitabine and Cisplatin for Resectable Adenocarcinoma of the Pancreatic Head. J. Clin. Oncol. 2008, 26, 2526–2531. [Google Scholar] [CrossRef]
- Palmer, D.H.; Stocken, D.D.; Hewitt, H.; Markham, C.E.; Hassan, A.B.; Johnson, P.J.; Buckels, J.A.; Bramhall, S.R. A Randomized Phase 2 Trial of Neoadjuvant Chemotherapy in Resectable Pancreatic Cancer: Gemcitabine Alone Versus Gemcitabine Combined with Cisplatin. Ann. Surg. Oncol. 2007, 14, 2088–2096. [Google Scholar] [CrossRef]
- Evans, D.B.; Rich, T.A.; Byrd, D.R.; Cleary, K.R.; Connelly, J.H.; Levin, B.; Charnsangavej, C.; Fenoglio, C.J.; Ames, F.C. Preoperative chemoradiation and pancreaticoduodenectomy for adenocarcinoma of the pancreas. Arch. Surg. 1992, 127, 1335–1339. [Google Scholar] [CrossRef]
- Evans, D.B.; Varadhachary, G.R.; Crane, C.H.; Sun, C.C.; Lee, J.E.; Pisters, P.W.; Vauthey, J.N.; Wang, H.; Cleary, K.R.; Staerkel, G.A.; et al. Preoperative gemcitabine-based chemoradiation for patients with resectable adenocarcinoma of the pancreatic head. J. Clin. Oncol. 2008, 26, 3496–3502. [Google Scholar] [CrossRef]
- Turrini, O.; Viret, F.; Moureau-Zabotto, L.; Guiramand, J.; Moutardier, V.; Lelong, B.; de Chaisemartin, C.; Giovannini, M.; Delpero, J.R. Neoadjuvant 5 fluorouracil-cisplatin chemoradiation effect on survival in patients with resectable pancreatic head adenocarcinoma: A ten-year single institution experience. Oncology 2009, 76, 413–419. [Google Scholar] [CrossRef]
- Le Scodan, R.; Mornex, F.; Girard, N.; Mercier, C.; Valette, P.J.; Ychou, M.; Bibeau, F.; Roy, P.; Scoazec, J.Y.; Partensky, C. Preoperative chemoradiation in potentially resectable pancreatic adenocarcinoma: Feasibility, treatment effect evaluation and prognostic factors, analysis of the SFRO-FFCD 9704 trial and literature review. Ann. Oncol. 2009, 20, 1387–1396. [Google Scholar] [CrossRef]
- Kim, E.J.; Ben-Josef, E.; Herman, J.M.; Bekaii-Saab, T.; Dawson, L.A.; Griffith, K.A.; Francis, I.R.; Greenson, J.K.; Simeone, D.M.; Lawrence, T.S.; et al. A Multi-Institutional Phase 2 Study of Neoadjuvant Gemcitabine and Oxaliplatin With Radiation Therapy in Patients with Pancreatic Cancer. Cancer 2013, 119, 2692–2700. [Google Scholar] [CrossRef]
- Golcher, H.; Brunner, T.B.; Witzigmann, H.; Marti, L.; Bechstein, W.O.; Bruns, C.; Jungnickel, H.; Schreiber, S.; Grabenbauer, G.G.; Meyer, T.; et al. Neoadjuvant chemoradiation therapy with gemcitabine/cisplatin and surgery versus immediate surgery in resectable pancreatic cancer. Strahlenther. Onkol. 2015, 191, 7–16. [Google Scholar] [CrossRef]
- Casadei, R.; Di Marco, M.; Ricci, C.; Santini, D.; Serra, C.; Calculli, L.; D’Ambra, M.; Guido, A.; Morselli-Labate, A.M.; Minni, F. Neoadjuvant Chemoradiotherapy and Surgery Versus Surgery Alone in Resectable Pancreatic Cancer: A Single-Center Prospective, Randomized, Controlled Trial Which Failed to Achieve Accrual Targets. J. Gastrointest. Surg. 2015, 19, 1802–1812. [Google Scholar] [CrossRef]
- Varadhachary, G.R.; Wolff, R.A.; Crane, C.H.; Sun, C.C.; Lee, J.E.; Pisters, P.W.; Vauthey, J.N.; Abdalla, E.; Wang, H.; Staerkel, G.A.; et al. Preoperative gemcitabine and cisplatin followed by gemcitabine-based chemoradiation for resectable adenocarcinoma of the pancreatic head. J. Clin. Oncol. 2008, 26, 3487–3495. [Google Scholar] [CrossRef]
- Cooper, A.B.; Parmar, A.D.; Riall, T.S.; Hall, B.L.; Katz, M.H.; Aloia, T.A.; Pitt, H.A. Does the Use of Neoadjuvant Therapy for Pancreatic Adenocarcinoma Increase Postoperative Morbidity and Mortality Rates? J. Gastrointest. Surg. 2015, 19, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Mokdad, A.A.; Minter, R.M.; Zhu, H.; Augustine, M.M.; Porembka, M.R.; Wang, S.C.; Yopp, A.C.; Mansour, J.C.; Choti, M.A.; Polanco, P.M. Neoadjuvant therapy followed by resection versus upfront resection for resectable pancreatic cancer: A propensity score matched analysis. J. Clin. Oncol. 2017, 35, 515–522. [Google Scholar] [CrossRef]
- Reni, M.; Cordio, S.; Milandri, C.; Passoni, P.; Bonetto, E.; Oliani, C.; Luppi, G.; Nicoletti, R.; Galli, L.; Bordonaro, R.; et al. Gemcitabine versus cisplatin, epirubicin, fluorouracil, and gemcitabine in advanced pancreatic cancer: A randomised controlled multicentre phase III trial. Lancet Oncol. 2005, 6, 369–376. [Google Scholar] [CrossRef]
- Reni, M.; Cereda, S.; Rognone, A.; Belli, C.; Ghidini, M.; Longoni, S.; Fugazza, C.; Rezzonico, S.; Passoni, P.; Slim, N.; et al. A randomized phase II trial of two different 4-drug combinations in advanced pancreatic adenocarcinoma: Cisplatin, capecitabine, gemcitabine plus either epirubicin or docetaxel (PEXG or PDXG regimen). Cancer Chemother. Pharmacol. 2012, 69, 115–123. [Google Scholar] [CrossRef]
- Petrioli, R.; Roviello, G.; Fiaschi, A.I.; Laera, L.; Marrelli, D.; Roviello, F.; Francini, E. Gemcitabine, oxaliplatin, and capecitabine (GEMOXEL) compared with gemcitabine alone in metastatic pancreatic cancer: A randomized phase II study. Cancer Chemother. Pharmacol. 2015, 75, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.L.; Gourgou-Bourgade, S.; de la Fouchardière, C.; et al. FOLFIRINOX versus Gemcitabine for Metastatic Pancreatic Cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef] [PubMed]
- Pingpank, J.F.; Hoffman, J.P.; Ross, E.A.; Cooper, H.S.; Meropol, N.J.; Freedman, G.; Pinover, W.H.; LeVoyer, T.E.; Sasson, A.R.; Eisenberg, B.L. Effect of preoperative chemoradiotherapy on surgical margin status of resected adenocarcinoma of the head of the pancreas. J. Gastrointest. Surg. 2001, 5, 121–130. [Google Scholar] [CrossRef]
- Takahashi, H.; Ogawa, H.; Ohigashi, H.; Gotoh, K.; Yamada, T.; Ohue, M.; Miyashiro, I.; Noura, S.; Kishi, K.; Motoori, M.; et al. Preoperative chemoradiation reduces the risk of pancreatic fistula after distal pancreatectomy for pancreatic adenocarcinoma. Surgery 2011, 150, 547–556. [Google Scholar] [CrossRef]
- Lowy, A.M.; Lee, J.E.; Pisters, P.W.T.; Davidson, B.S.; Fenoglio, C.J.; Stanford, P.; Jinnah, R.; Evans, D.B. Prospective, Randomized Trial of Octreotide to Prevent Pancreatic Fistula After Pancreaticoduodenectomy for Malignant Disease. Ann. Surg. 1997, 226, 632–641. [Google Scholar] [CrossRef] [PubMed]
- Lamarca, A.; Rigby, C.; McNamara, M.G.; Hubner, R.A.; Valle, J.W. Impact of biliary stent-related events in patients diagnosed with advanced pancreatobiliary tumors receiving palliative chemotherapy. World J. Gastroenterol. 2016, 22, 6065–6075. [Google Scholar] [CrossRef]
- Adams, M.A.; Anderson, M.A.; Myles, J.D.; Khalatbari, S.; Scheiman, J.M. Self-expanding metal stents (SEMS) provide superior outcomes compared to plastic stents for pancreatic cancer patients undergoing neoadjuvant therapy. J. Gastrointest. Oncol. 2012, 3, 309–313. [Google Scholar]
- Stocken, D.D.; Hassan, A.B.; Altman, D.G.; Billingham, L.J.; Bramhall, S.R.; Johnson, P.J.; Freemantle, N. Modelling prognostic factors in advanced pancreatic cancer. Br. J. Cancer. 2008, 99, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Bilici, A. Prognostic factors related with survival in patients with pancreatic adenocarcinoma. World J. Gastroenterol. 2014, 20, 10802–10812. [Google Scholar] [CrossRef]
- Ethun, C.G.; Kooby, D.A. The importance of surgical margins in pancreatic cancer. J. Surg. Oncol. 2016, 113, 283–288. [Google Scholar] [CrossRef]
- Slidell, M.B.; Chang, D.C.; Cameron, J.L.; Wolfgang, C.; Herman, J.M.; Schulick, R.D.; Choti, M.A.; Pawlik, T.M. Impact of total lymph node count and lymph node ratio on staging and survival after pancreatectomy for pancreatic adenocarcinoma: A large, population-based analysis. Ann. Surg. Oncol. 2008, 15, 165–174. [Google Scholar] [CrossRef]
- Showalter, T.N.; Winter, K.A.; Berger, A.C.; Regine, W.F.; Abrams, R.A.; Safran, H.; Hoffman, J.P.; Benson, A.B.; MacDonald, J.S.; Willett, C.G. The influence of total nodes examined, number of positive nodes, and lymph node ratio on survival after surgical resection and adjuvant chemoradiation for pancreatic cancer: A secondary analysis of RTOG 9704. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 1328–1335. [Google Scholar] [CrossRef]
- Chatterjee, D.; Katz, M.H.; Rashid, A.; Wang, H.; Iuga, A.C.; Varadhachary, G.R.; Wolff, R.A.; Lee, J.E.; Pisters, P.W.; Crane, C.H.; et al. Perineural and intraneural invasion in posttherapy pancreaticoduodenectomy specimens predicts poor prognosis in patients with pancreatic ductal adenocarcinoma. Am. J. Surg. Pathol. 2012, 36, 409–417. [Google Scholar] [CrossRef]
- Lyman, G.H.; Yau, L.; Nakov, R.; Krendyukov, A. Overall Survival and Risk of Second Malignancies with Cancer Chemotherapy and G-CSF Support. Ann. Oncol. 2018, 29, 1903–1910. [Google Scholar] [CrossRef]
- Lee, J.; Kim, J.W.; Ahn, S.; Kim, H.W.; Lee, J.; Kim, Y.H.; Paik, K.H.; Kim, J.; Hwang, J.H. Optimal dose reduction of FOLFIRINOX for preserving tumour response in advanced pancreatic cancer: Using cumulative relative dose intensity. Eur. J. Cancer 2017, 76, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Yabusaki, N.; Fujii, T.; Yamada, S.; Murotani, K.; Sugimoto, H.; Kanda, M.; Nakayama, G.; Koike, M.; Fujiwara, M.; Kodera, Y. The significance of relative dose intensity in adjuvant chemotherapy of pancreatic ductal adenocarcinoma-including the analysis of clinicopathological factors influencing relative dose intensity. Medicine 2016, 95, e4282. [Google Scholar] [CrossRef] [PubMed]
| Variables | n (%) |
|---|---|
| Age-years | |
| Median | 63 |
| Range | 35–82 |
| Gender | |
| Male | 23 (57.5) |
| Female | 17 (42.5) |
| ECOG | |
| 0 | 5 (12.5) |
| 1 | 33 (82.5) |
| 2 | 2 (5) |
| Location | |
| Head-Isthmus | 32 (80) |
| Body-Tail | 8 (20) |
| Baseline EUS-T stage | |
| T1-T2 | 3 (7.5) |
| T3 | 37 (92.5) |
| Baseline EUS-N stage | |
| N0 | 21 (52.5) |
| N+ | 11 (27.5) |
| Nx | 8 (20) |
| Biliary stent | |
| No | 19 (47.5) |
| Yes | 21 (52.5) |
| Neoadjuvant chemotherapy regimen | |
| Gemcitabine-based | 26 (65) |
| mFOLFOXIRI | 14 (35) |
| Neoadjuvant radiotherapy technique | |
| 3D-RT | 20 (54.1) |
| IMRT | 17 (45.9) |
| Adverse Event | IPCT 1 (n = 40) | CRT 2 (n = 37) | ||||
|---|---|---|---|---|---|---|
| Type | Grade 1/2 n (%) | Grade 3 n (%) | Grade 4 n (%) | Grade 1/2 n (%) | Grade 3 n (%) | Grade 4 n (%) |
| Hematological | ||||||
| Anemia | 37 (92.5) | - | - | 33 (89.2) | - | - |
| Leukopenia | 19 (47.5) | 2 (5) | 3 (7.5) | 29 (78.4) | - | - |
| Neutropenia | 7 (17.5) | 9 (22.5) | 1 (2.5) | 8 (21.6) | - | - |
| Febrile neutropenia | 1 (2.5) | 4 (10) | 1 (2.5) | 1 (2.7) | - | - |
| Thrombocytopenia | 26 (65) | - | - | 27 (73) | 4 (10.8) | - |
| Non-hematological | ||||||
| Nausea/Vomiting | 12 (30) | - | - | 15 (40.5) | - | - |
| Anorexia | 18 (45) | - | - | 20 (54) | - | - |
| Diarrhea | 15 (37.5) | 3 (7.5) | - | 6 (16.2) | - | - |
| Gastritis | 7 (17.5) | 1 (2.5) | - | 13 (35.1) | - | - |
| Mucositis | 10 (25) | - | - | 2 (5.4) | - | - |
| Asthenia | 32 (80) | 2 (5) | - | 21 (56.7) | 2 (5.4) | - |
| Peripheral neuropathy | 22 (55) | - | - | 6 (16.2) | - | - |
| Hand-foot syndrome | 1 (2.5) | - | - | 1 (2.7) | - | - |
| Cholangitis | - | 5 (12.5) | - | - | 5 (13.5) | - |
| Variables | n (%) |
|---|---|
| Age-years | |
| Median | 64 |
| Range | 44-80 |
| Gender | |
| Male | 23 (51.1) |
| Female | 22 (48.9) |
| ECOG | |
| 0 | 8 (17.8) |
| 1 | 35 (77.8) |
| 2 | 2 (4.4) |
| Location | |
| Head-Isthmus | 34 (75.6) |
| Body-Tail | 10 (22.2) |
| Multifocal | 1 (2.2) |
| Baseline EUS-T stage | |
| T1-T2 | 4 (8.9) |
| T3 | 40 (88.9) |
| T4 | 1 (2.2) |
| Baseline EUS-N stage | |
| N0 | 27 (60) |
| N+ | 11 (24.4) |
| Nx | 7 (15.6) |
| Resectability | |
| Resectable | 37 (82.2) |
| Borderline-resectable | 8 (17.8) |
| Neoadjuvant approach | |
| IPCT + CRT | 42 (93.3) |
| IPCT | 3 (6.7) |
| Duration of IPCT-days | |
| Median | 53 |
| Range | 40-125 |
| Number of CRT session | |
| Median | 25 |
| Range | 18-30 |
| Type of surgery | |
| Cephalic duodenopancreatectomy | 37 (82.2) |
| Distal pancreatectomy | 8 (17.8) |
| Adjuvant treatment | |
| Yes | 13 (28.9) |
| No | 32 (71.1) |
| Relapse | |
| Yes | 30 (66.7) |
| No | 15 (33.3) |
| Relapse at 2 years | |
| Yes | 22 (48.9) |
| No | 23 (51.1) |
| Type of relapse | |
| Local | 2 (6.7) |
| Distant | 22 (73.3) |
| Local and distant | 6 (20) |
| Variables | n (%) |
|---|---|
| Gender | |
| Male | 23 (51.1) |
| Female | 22 (48.9) |
| Age-years | |
| Min. | 44 |
| Median | 64 |
| Mean | 63 |
| Max. | 80 |
| Resectability | |
| Resectable | 37 (82.2) |
| Borderline-resectable | 8 (17.8) |
| ECOG | |
| 0 | 8 (17.8) |
| 1 | 35 (77.8) |
| 2 | 2 (4.4) |
| Neoadjuvant chemotherapy regimen | |
| mFOLFOXIRI | 18 (40) |
| Gemcitabine-based | 27 (60) |
| Granulocyte colony-stimulating factors | |
| No | 37 (82.2) |
| Yes | 8 (17.8) |
| Neoadjuvant radiotherapy technique | |
| 3D-RT | 18 (40) |
| IMRT | 21 (46.7) |
| Not reported | 6 (13.3) |
| Type of surgery | |
| Cephalic duodenopancreatectomy | 37 (82.2) |
| Distal pancreatectomy | 8 (17.8) |
| ypT | |
| ypT0 | 6 (13.3) |
| ypT1 | 19 (42.2) |
| ypT2 | 6 (13.3) |
| ypT3 | 12 (26.7) |
| ypTx | 2 (4.4) |
| ypN | |
| ypN0 | 41 (91.1) |
| ypN1 | 4 (8.9) |
| CAP | |
| 0 | 6 (13.3) |
| 1 | 23 (51.1) |
| 2 | 10 (22.2) |
| 3 | 6 (13.3) |
| CAP Group | |
| CAP 0–1 | 29 (64.4) |
| CAP 2–3 | 16 (35.6) |
| Pathological complete response | |
| No | 37 (82.2) |
| Yes | 8 (17.8) |
| Resected lymph nodes | |
| Min. | 2 |
| Median | 9 |
| Mean | 10.93 |
| Max. | 27 |
| Pathological lymph nodes | |
| Min. | 0.00 |
| Median | 0.00 |
| Mean | 0.16 |
| Max. | 3.00 |
| Modified LNR: 1 + n° pathological/1 + n° resected | |
| Min. | 0.036 |
| Median | 0.100 |
| Mean | 0.125 |
| Max. | 0.333 |
| Miller & Payne nodal response | |
| A | 41 (91.1) |
| B | 1 (2.2) |
| C | 3 (6.7) |
| D | 0 (0) |
| Vascular invasion | |
| No | 40 (88.9) |
| Yes | 5 (11.1%) |
| Perineural invasion | |
| No | 31 (68.9) |
| Yes | 14 (31.1) |
| Surgical margins | |
| R0 (Not involved, >1 mm) | 43 (95.6) |
| R1 (Involved, <1 mm) | 2 (4.4) |
| SCORE 1 | |
| 1 | 6 (13.3) |
| 2 | 25 (55.6) |
| 3 | 14 (31.1) |
| Dose intensity > 80% 2 | |
| No | 12 (27.3) |
| Yes | 32 (72.7) |
| Progression disease at 2 years | |
| No | 23 (51.1) |
| Yes | 22 (48.9) |
| Variable | Logistic Regression | Decision Tree | Random Forest | Support Vector Machine | K-Nearest Neighbours |
|---|---|---|---|---|---|
| Accuracy | 0.71 | 0.60 | 0.67 | 0.60 | 0.58 |
| Sensitivity | 0.70 | 0.83 | 0.74 | 0.65 | 0.65 |
| Specificity | 0.73 | 0.36 | 0.59 | 0.55 | 0.50 |
| PPV 1 | 0.73 | 0.58 | 0.65 | 0.60 | 0.58 |
| NPV 2 | 0.70 | 0.67 | 0.68 | 0.60 | 0.58 |
| AUC | 0.75 | 0.61 | 0.67 | 0.61 | 0.58 |
| Variables | n (%) |
|---|---|
| Granulocyte colony-stimulating factors | |
| No | 4 (36.4) |
| Yes | 7 (63.6) |
| Perineural invasion | |
| No | 2 (18.2) |
| Yes | 9 (81.8) |
| Surgical margins | |
| R0 (Not involved) | 8 (72.7) |
| R1 (Involved) | 3 (27.3) |
| Resected lymph nodes | |
| Min. | 9 |
| Median | 17 |
| Mean | 15.55 |
| Max. | 19 |
| Progression disease at 2 years | |
| No | 4 (36.4) |
| Yes | 7 (63.6) |
| Study | NA | ChT | RT | CRT | n | Stage | Resection | R0 Rate G/Res | Median OS (Months) | Survival Rate (%) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| G | Res | NRes | G | Res | NRes | |||||||||
| Ishikawa (1994) [33] | RT | - | 50 Gy | - | 54 | R | 74% | - | 15 | - | 9 | 30 (2y), 22 (5y) | 28 (3y), 22 (5y) | 17 (1y), 0 (2y) |
| Evans (1992) [37] | CRT | - | - | 5-FU, 50.4Gy | 28 | R | 61% | 50%/82% | - | - | - | - | - | - |
| Evans (2008) [38] | CRT | - | - | Gemcitabine 30 Gy | 86 | R | 74% | 66%/95% | 22.7 | 34 | 7 | 27 (5y) | 36 (5y) | 0 (5y) |
| BR | ||||||||||||||
| Turrini (2009) [39] | CRT | - | - | 5-FU Cisplatin 45 Gy | 102 | R | 61% | 56%/92% | 17 | 23 | 11 | 10 (5y) | 18 (5y) | 0 (5y) |
| Le Scodan (2009) [40] | CRT | - | - | 5-FU Cisplatin 50 Gy | 41 | R | 63% | 51%/80.7% | 9.4 | 11.7 | 5.7 | 41 (1y), 20 (2y) | 48 (1y), 32 (2y) | 40 (1y), 0 (2y) |
| Kim (2013) [41] | CRT | - | - | Gemcitabine | 68 | R | 63% | 53%/84% | 18.2 | 27.1 | 10.9 | 62 (1y), 44 (2y) | 82 (1y), 62 (2y) | 33 (1y), 17 (2y) |
| Oxaliplatin | BR | |||||||||||||
| 30Gy | I | |||||||||||||
| Golcher (2015) [42] | CRT | - | - | Gemcitabine Cisplatin 55.8 to 50.4Gy | 66 | R | 58% | 51%/89% | 17.4 | 25 | - | 39 (2y), 12 (3y) | - | - |
| BR | ||||||||||||||
| Casadei (2015) [43] | ChT-CRT | Gemcitabine | - | Gemcitabine 54 Gy | 38 | R | 61.1% | 38.9%/64% | 22.4 | - | - | - | - | - |
| Varadhachary (2008) [44] | ChT-CRT | Gemcitabine Cisplatin | - | Gemcitabine 30 Gy | 90 | R | 58% | 55%/96% | 17.4 | 31 | 10.5 | 37 (2y), 19 (4y) | 60 (2y), 36 (4y) | - |
| BR | ||||||||||||||
| O’Reilly (2014) [34] | ChT | Gemcitabine Oxaliplatin | - | - | 38 | BR | 71% | 53%/74% | 27.2 | NR | 15 | 63 (18m) | 78 (18m) | 25 (18 m) |
| Heinrich (2008) [35] | ChT | Gemcitabine Cisplatin | - | - | 28 | R | 89% | 71%/80% | 26.5 | 19.1 | - | - | - | - |
| Palmer (2007) [36] | ChT | Gemcitabine Cisplatin Vs. Gemcitabine alone | - | - | 50 | R | 70% Vs. 38% | 46%/75% Vs. 25%/75% | 15.6 Vs. 9.9 | 28.4 (global) | - | 62 (1y) Vs. 42 (1y) | 77.8 (global) | - |
| De W Marsh (2017) [14] | ChT | mFOLFIRINOX | - | - | 21 | R | 81% | 76%/94% | 34 | 35.5 | 10.1 | 80 (1y), 60(2y) | 81 (1y), 71 (2y) | 33 (1y), 0 (2y) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sala Elarre, P.; Oyaga-Iriarte, E.; Yu, K.H.; Baudin, V.; Arbea Moreno, L.; Carranza, O.; Chopitea Ortega, A.; Ponz-Sarvise, M.; Mejías Sosa, L.D.; Rotellar Sastre, F.; et al. Use of Machine-Learning Algorithms in Intensified Preoperative Therapy of Pancreatic Cancer to Predict Individual Risk of Relapse. Cancers 2019, 11, 606. https://doi.org/10.3390/cancers11050606
Sala Elarre P, Oyaga-Iriarte E, Yu KH, Baudin V, Arbea Moreno L, Carranza O, Chopitea Ortega A, Ponz-Sarvise M, Mejías Sosa LD, Rotellar Sastre F, et al. Use of Machine-Learning Algorithms in Intensified Preoperative Therapy of Pancreatic Cancer to Predict Individual Risk of Relapse. Cancers. 2019; 11(5):606. https://doi.org/10.3390/cancers11050606
Chicago/Turabian StyleSala Elarre, Pablo, Esther Oyaga-Iriarte, Kenneth H. Yu, Vicky Baudin, Leire Arbea Moreno, Omar Carranza, Ana Chopitea Ortega, Mariano Ponz-Sarvise, Luis D. Mejías Sosa, Fernando Rotellar Sastre, and et al. 2019. "Use of Machine-Learning Algorithms in Intensified Preoperative Therapy of Pancreatic Cancer to Predict Individual Risk of Relapse" Cancers 11, no. 5: 606. https://doi.org/10.3390/cancers11050606
APA StyleSala Elarre, P., Oyaga-Iriarte, E., Yu, K. H., Baudin, V., Arbea Moreno, L., Carranza, O., Chopitea Ortega, A., Ponz-Sarvise, M., Mejías Sosa, L. D., Rotellar Sastre, F., Larrea Leoz, B., Iragorri Barberena, Y., Subtil Iñigo, J. C., Benito Boíllos, A., Pardo, F., & Rodríguez Rodríguez, J. (2019). Use of Machine-Learning Algorithms in Intensified Preoperative Therapy of Pancreatic Cancer to Predict Individual Risk of Relapse. Cancers, 11(5), 606. https://doi.org/10.3390/cancers11050606






