Influence of Tea Consumption on the Development of Second Esophageal Neoplasm in Patients with Head and Neck Cancer
Abstract
1. Introduction
2. Results
2.1. Factors Associated with the Development of Synchronous Esophageal Neoplasm in HNSCC Patients
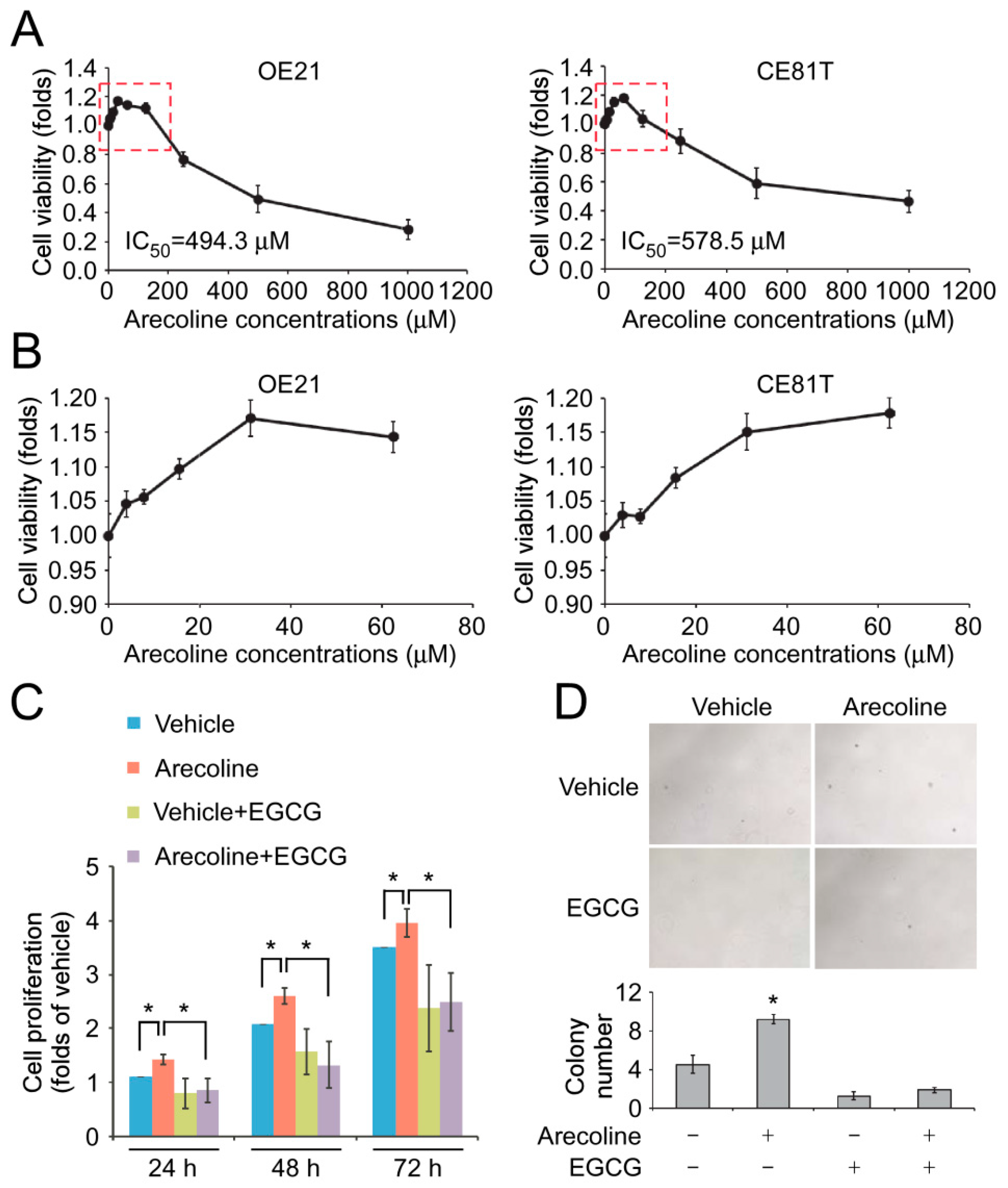
2.2. EGCG Suppressed Low-Concentration Arecoline-Induced Proliferation and Colony Formation of ESCC Cells
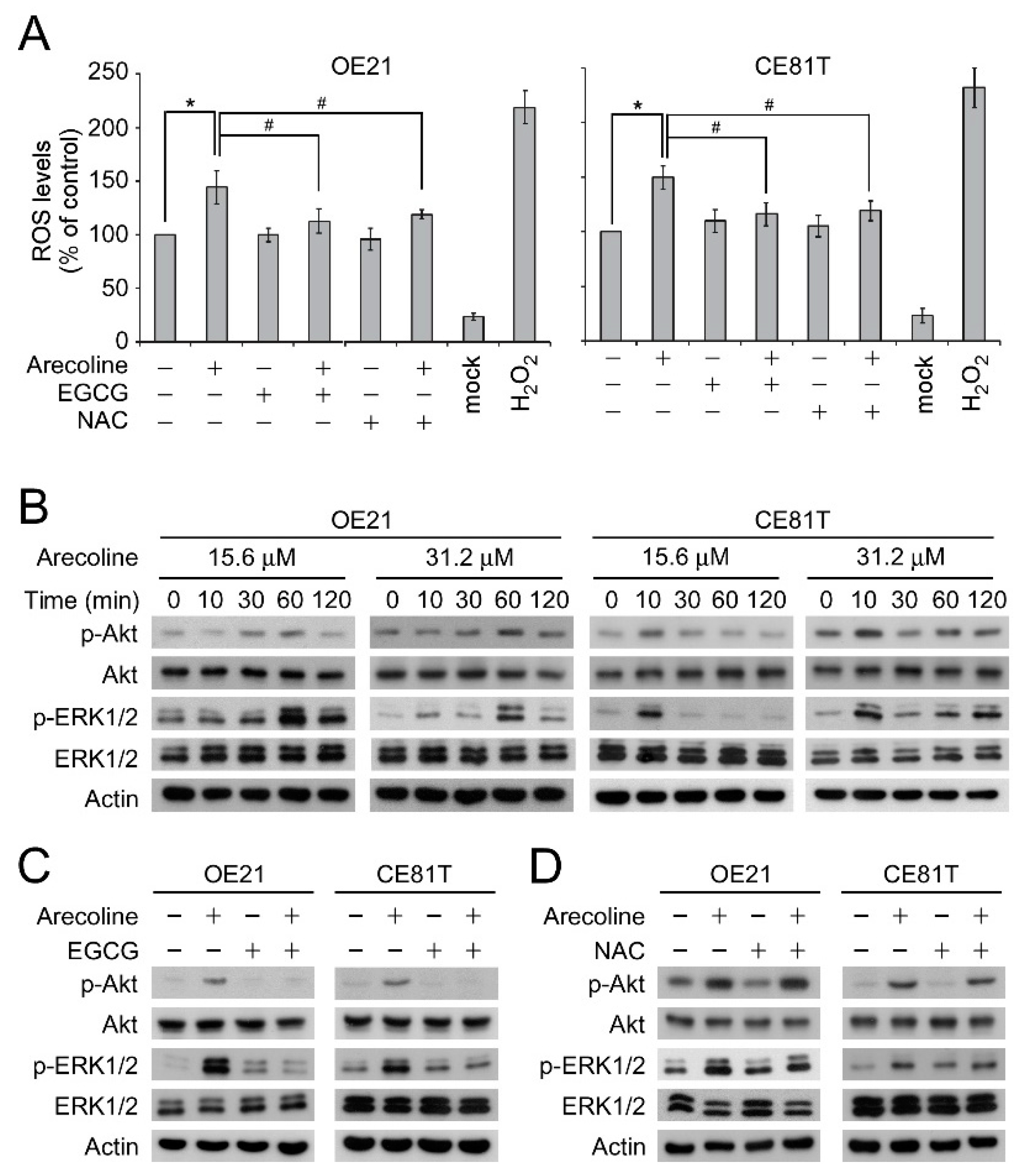
2.3. EGCG Suppressed Arecoline-Induced Akt and ERK1/2 Phosphorylation Through An ROS-Independent Pathway
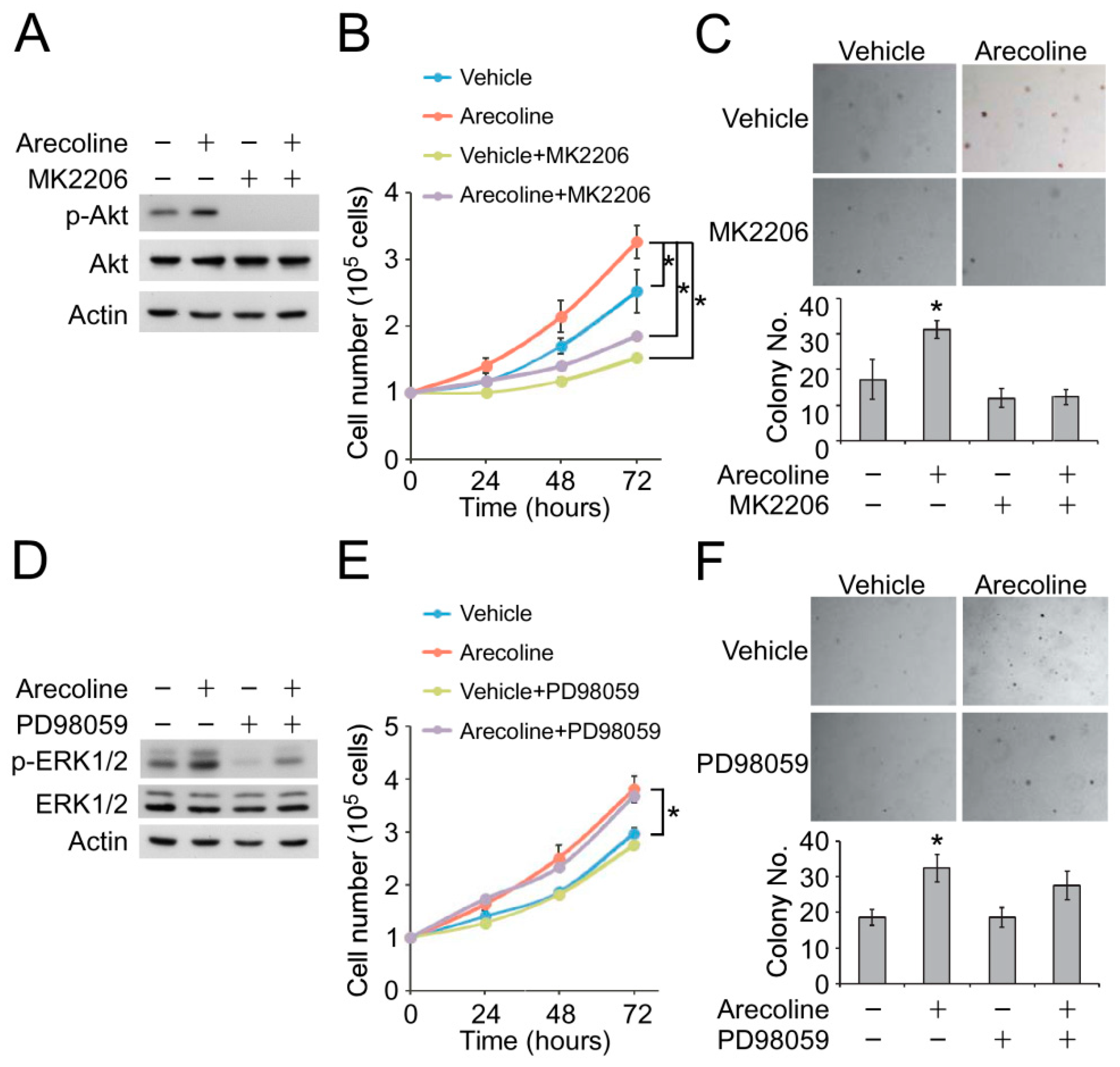
2.4. Inhibition of Akt Phosphorylation Suppressed Arecoline-Induced ESCC Cell Proliferation and Colony Formation
3. Discussion
4. Materials and Methods
4.1. Study Population and Diagnosis of Esophageal Squamous Cell Neoplasm
4.2. Substance Use and Demographic Data Collection
4.3. Esophageal Cancer Cell Lines and Chemicals
4.4. Cell Viability Assay and Cell Proliferation
DCFH-DA Cellular ROS Assay
4.5. Soft Agar Colony Formation Assay
4.6. Western Blot
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chuang, S.C.; Scelo, G.; Tonita, J.M.; Tamaro, S.; Jonasson, J.G.; Kliewer, E.V.; Hemminki, K.; Weiderpass, E.; Pukkala, E.; Tracey, E.; et al. Risk of second primary cancer among patients with head and neck cancers: A pooled analysis of 13 cancer registries. Int. J. Cancer 2008, 123, 2390–2396. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.J.; Chou, H.W.; Wang, C.T.; Chung, C.S.; Lai, M.S. The impact of second primary malignancies on head and neck cancer survivors: A nationwide cohort study. PLoS ONE 2013, 8, e62116. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.K.; Chuang, Y.S.; Wu, T.S.; Lee, K.W.; Wu, C.W.; Wang, H.C.; Kuo, C.T.; Lee, C.H.; Kuo, W.R.; Chen, C.H.; et al. Endoscopic screening for synchronous esophageal neoplasia among patients with incident head and neck cancer: Prevalence, risk factors, and outcomes. Int. J. Cancer 2017, 141, 1987–1996. [Google Scholar] [CrossRef] [PubMed]
- Secretan, B.; Straif, K.; Baan, R.; Grosse, Y.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Freeman, C.; Galichet, L.; et al. A review of human carcinogens—Part E: Tobacco, areca nut, alcohol, coal smoke, and salted fish. Lancet Oncol. 2009, 10, 1033–1034. [Google Scholar] [CrossRef]
- Chung, C.S.; Liao, L.J.; Lo, W.C.; Chou, Y.H.; Chang, Y.C.; Lin, Y.C.; Hsu, W.F.; Shueng, P.W.; Lee, T.H. Risk factors for second primary neoplasia of esophagus in newly diagnosed head and neck cancer patients: A case-control study. BMC Gastroenterol. 2013, 13, 154. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Gong, E.J.; Jung, H.Y.; Lim, H.; Ahn, J.Y.; Choi, K.S.; Lee, J.H.; Choi, K.D.; Song, H.J.; Lee, G.H.; et al. Clinical significance of intensive endoscopic screening for synchronous esophageal neoplasm in patients with head and neck squamous cell carcinoma. Scand. J. Gastroenterol. 2014, 49, 1486–1492. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.C.; Lee, Y.C.; Tseng, P.H.; Chen, T.C.; Yang, T.L.; Lou, P.J.; Ko, J.Y.; Liao, L.J.; Hsu, W.L.; Chang, Y.L.; et al. Regular screening of esophageal cancer for 248 newly diagnosed hypopharyngeal squamous cell carcinoma by unsedated transnasal esophagogastroduodenoscopy. Oral Oncol. 2016, 55, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Gong, E.J.; Kim, D.H.; Ahn, J.Y.; Choi, K.S.; Jung, K.W.; Lee, J.H.; Choi, K.D.; Song, H.J.; Lee, G.H.; Jung, H.Y.; et al. Routine endoscopic screening for synchronous esophageal neoplasm in patients with head and neck squamous cell carcinoma: A prospective study. Dis. Esophagus 2016, 29, 752–759. [Google Scholar] [CrossRef]
- Chung, C.S.; Lo, W.C.; Lee, Y.C.; Wu, M.S.; Wang, H.P.; Liao, L.J. Image-enhanced endoscopy for detection of second primary neoplasm in patients with esophageal and head and neck cancer: A systematic review and meta-analysis. Head Neck 2016, 38 (Suppl. 1), E2343–E2349. [Google Scholar] [CrossRef]
- Zheng, P.; Zheng, H.M.; Deng, X.M.; Zhang, Y.D. Green tea consumption and risk of esophageal cancer: A meta-analysis of epidemiologic studies. BMC Gastroenterol. 2012, 12, 165. [Google Scholar] [CrossRef]
- Oze, I.; Matsuo, K.; Kawakita, D.; Hosono, S.; Ito, H.; Watanabe, M.; Hatooka, S.; Hasegawa, Y.; Shinoda, M.; Tajima, K.; et al. Coffee and green tea consumption is associated with upper aerodigestive tract cancer in Japan. Int. J. Cancer 2014, 135, 391–400. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Lujan-Barroso, L.; Bueno-de-Mesquita, H.B.; Dik, V.K.; Boeing, H.; Steffen, A.; Tjonneland, A.; Olsen, A.; Bech, B.H.; Overvad, K.; et al. Tea and coffee consumption and risk of esophageal cancer: The European prospective investigation into cancer and nutrition study. Int. J. Cancer 2014, 135, 1470–1479. [Google Scholar] [CrossRef]
- Chen, Y.K.; Lee, C.H.; Wu, I.C.; Liu, J.S.; Wu, D.C.; Lee, J.M.; Goan, Y.G.; Chou, S.H.; Huang, C.T.; Lee, C.Y.; et al. Food intake and the occurrence of squamous cell carcinoma in different sections of the esophagus in Taiwanese men. Nutrition 2009, 25, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Lambert, J.D.; Ju, J.; Lu, G.; Sang, S. Tea and cancer prevention: Molecular mechanisms and human relevance. Toxicol. Appl. Pharmacol. 2007, 224, 265–273. [Google Scholar] [CrossRef]
- Yang, C.S.; Wang, X.; Lu, G.; Picinich, S.C. Cancer prevention by tea: Animal studies, molecular mechanisms and human relevance. Nat. Rev. Cancer 2009, 9, 429–439. [Google Scholar] [CrossRef]
- Chikara, S.; Nagaprashantha, L.D.; Singhal, J.; Horne, D.; Awasthi, S.; Singhal, S.S. Oxidative stress and dietary phytochemicals: Role in cancer chemoprevention and treatment. Cancer Lett. 2018, 413, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Wang, H. Cancer Preventive Activities of Tea Catechins. Molecules 2016, 21, 1679. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.F.; Yang, C.C.; Kao, S.Y.; Liu, C.J.; Lin, S.C.; Chang, K.W. MicroRNA-211 enhances the oncogenicity of carcinogen-induced oral carcinoma by repressing TCF12 and increasing antioxidant activity. Cancer Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Hsu, N.Y.; Yeh, K.T.; Chiang, I.P.; Pai, L.Y.; Chen, C.Y.; Ho, H.C. Cortactin overexpression in the esophageal squamous cell carcinoma and its involvement in the carcinogenesis. Dis. Esophagus 2008, 21, 402–408. [Google Scholar] [CrossRef]
- Yen, C.Y.; Lin, M.H.; Liu, S.Y.; Chiang, W.F.; Hsieh, W.F.; Cheng, Y.C.; Hsu, K.C.; Liu, Y.C. Arecoline-mediated inhibition of AMP-activated protein kinase through reactive oxygen species is required for apoptosis induction. Oral Oncol. 2011, 47, 345–351. [Google Scholar] [CrossRef]
- Hung, T.C.; Huang, L.W.; Su, S.J.; Hsieh, B.S.; Cheng, H.L.; Hu, Y.C.; Chen, Y.H.; Hwang, C.C.; Chang, K.L. Hemeoxygenase-1 expression in response to arecoline-induced oxidative stress in human umbilical vein endothelial cells. Int. J. Cardiol. 2011, 151, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.T.; Yang, S.R.; Chen, J.Y.; Cheng, Y.P.; Lee, Y.R.; Chiang, M.K.; Chen, H.R. Arecoline downregulates levels of p21 and p27 through the reactive oxygen species/mTOR complex 1 pathway and may contribute to oral squamous cell carcinoma. Cancer Sci. 2012, 103, 1221–1229. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.S.; Lin, C.P.; Chen, Y.P.; Chao, M.R.; Li, C.C.; Liu, K.L. CYP450-mediated mitochondrial ROS production involved in arecoline N-oxide-induced oxidative damage in liver cell lines. Environ. Toxicol. 2018, 33, 1029–1038. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Lee, W.T.; Tsai, S.T.; Ou, C.Y.; Lo, H.I.; Wong, T.Y.; Fang, S.Y.; Chen, K.C.; Huang, J.S.; Wu, J.L.; et al. Tea consumption and risk of head and neck cancer. PLoS ONE 2014, 9, e96507. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yang, Y.; Zhang, W.; Wu, W. Association of tea consumption and the risk of oral cancer: A meta-analysis. Oral Oncol. 2014, 50, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Abnet, C.C.; Corley, D.A.; Freedman, N.D.; Kamangar, F. Diet and upper gastrointestinal malignancies. Gastroenterology 2015, 148, 1234–1243. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Wang, H.; Li, G.X.; Yang, Z.; Guan, F.; Jin, H. Cancer prevention by tea: Evidence from laboratory studies. Pharmacol. Res. 2011, 64, 113–122. [Google Scholar] [CrossRef]
- Oliva, J.; Bardag-Gorce, F.; Tillman, B.; French, S.W. Protective effect of quercetin, EGCG, catechin and betaine against oxidative stress induced by ethanol in vitro. Exp. Mol. Pathol. 2011, 90, 295–299. [Google Scholar] [CrossRef]
- Shi, J.; Liu, F.; Zhang, W.; Liu, X.; Lin, B.; Tang, X. Epigallocatechin-3-gallate inhibits nicotine-induced migration and invasion by the suppression of angiogenesis and epithelial-mesenchymal transition in non-small cell lung cancer cells. Oncol. Rep. 2015, 33, 2972–2980. [Google Scholar] [CrossRef]
- Tu, S.H.; Ku, C.Y.; Ho, C.T.; Chen, C.S.; Huang, C.S.; Lee, C.H.; Chen, L.C.; Pan, M.H.; Chang, H.W.; Chang, C.H.; et al. Tea polyphenol (-)-epigallocatechin-3-gallate inhibits nicotine- and estrogen-induced alpha9-nicotinic acetylcholine receptor upregulation in human breast cancer cells. Mol. Nutr. Food Res. 2011, 55, 455–466. [Google Scholar] [CrossRef]
- Lee, S.S.; Tsai, C.H.; Ho, Y.C.; Yu, C.C.; Chang, Y.C. Heat shock protein 27 expression in areca quid chewing-associated oral squamous cell carcinomas. Oral Dis. 2012, 18, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.S.; Tsai, C.H.; Yu, C.C.; Chang, Y.C. Elevated snail expression mediates tumor progression in areca quid chewing-associated oral squamous cell carcinoma via reactive oxygen species. PLoS ONE 2013, 8, e67985. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.P.; Chen, H.M.; Chang, J.Z.; Chiang, C.P.; Deng, Y.T.; Kuo, M.Y. Arecoline stimulated early growth response-1 production in human buccal fibroblasts: Suppression by epigallocatechin-3-gallate. Head Neck 2015, 37, 493–497. [Google Scholar] [CrossRef]
- Lee, S.S.; Chen, Y.J.; Tsai, C.H.; Huang, F.M.; Chang, Y.C. Elevated transglutaminase-2 expression mediates fibrosis in areca quid chewing-associated oral submucocal fibrosis via reactive oxygen species generation. Clin. Oral Investig. 2016, 20, 1029–1034. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.P.; Wu, K.J.; Chen, H.M.; Deng, Y.T. Arecoline activates latent transforming growth factor beta1 via mitochondrial reactive oxygen species in buccal fibroblasts: Suppression by epigallocatechin-3-gallate. J. Formos Med. Assoc. 2018, 117, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Ghelardini, C.; Galeotti, N.; Lelli, C.; Bartolini, A. M1 receptor activation is a requirement for arecoline analgesia. Farmaco 2001, 56, 383–385. [Google Scholar] [CrossRef]
- Yang, Y.R.; Chang, K.C.; Chen, C.L.; Chiu, T.H. Arecoline excites rat locus coeruleus neurons by activating the M2-muscarinic receptor. Chin. J. Physiol. 2000, 43, 23–28. [Google Scholar]
- Xie, D.P.; Chen, L.B.; Liu, C.Y.; Zhang, C.L.; Liu, K.J.; Wang, P.S. Arecoline excites the colonic smooth muscle motility via M3 receptor in rabbits. Chin. J. Physiol. 2004, 47, 89–94. [Google Scholar]
- Castillo-Gonzalez, A.C.; Nieto-Ceron, S.; Pelegrin-Hernandez, J.P.; Montenegro, M.F.; Noguera, J.A.; Lopez-Moreno, M.F.; Rodriguez-Lopez, J.N.; Vidal, C.J.; Hellin-Meseguer, D.; Cabezas-Herrera, J. Dysregulated cholinergic network as a novel biomarker of poor prognostic in patients with head and neck squamous cell carcinoma. BMC Cancer 2015, 15, 385. [Google Scholar] [CrossRef]
- Chiu, C.C.; Chen, B.H.; Hour, T.C.; Chiang, W.F.; Wu, Y.J.; Chen, C.Y.; Chen, H.R.; Chan, P.T.; Liu, S.Y.; Chen, J.Y. Betel quid extract promotes oral cancer cell migration by activating a muscarinic M4 receptor-mediated signaling cascade involving SFKs and ERK1/2. Biochem. Biophys. Res. Commun. 2010, 399, 60–65. [Google Scholar] [CrossRef]
- Cui, Q.L.; Fogle, E.; Almazan, G. Muscarinic acetylcholine receptors mediate oligodendrocyte progenitor survival through Src-like tyrosine kinases and PI3K/Akt pathways. Neurochem. Int. 2006, 48, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Smedlund, K.; Tano, J.Y.; Margiotta, J.; Vazquez, G. Evidence for operation of nicotinic and muscarinic acetylcholine receptor-dependent survival pathways in human coronary artery endothelial cells. J. Cell. Biochem. 2011, 112, 1978–1984. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Li, H.; Chen, L.; Yang, L.; Li, L.; Tao, Y.; Li, W.; Li, Z.; Liu, H.; Tang, M.; et al. (-)-Epigallocatechin-3-gallate inhibition of Epstein-Barr virus spontaneous lytic infection involves ERK1/2 and PI3-K/Akt signaling in EBV-positive cells. Carcinogenesis 2013, 34, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Sah, J.F.; Balasubramanian, S.; Eckert, R.L.; Rorke, E.A. Epigallocatechin-3-gallate inhibits epidermal growth factor receptor signaling pathway. Evidence for direct inhibition of ERK1/2 and AKT kinases. J. Biol. Chem. 2004, 279, 12755–12762. [Google Scholar] [CrossRef] [PubMed]
- Van Aller, G.S.; Carson, J.D.; Tang, W.; Peng, H.; Zhao, L.; Copeland, R.A.; Tummino, P.J.; Luo, L. Epigallocatechin gallate (EGCG), a major component of green tea, is a dual phosphoinositide-3-kinase/mTOR inhibitor. Biochem. Biophys. Res. Commun. 2011, 406, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Satonaka, H.; Ishida, K.; Takai, M.; Koide, R.; Shigemasa, R.; Ueyama, J.; Ishikawa, T.; Hayashi, K.; Goto, H.; Wakusawa, S. (-)-Epigallocatechin-3-gallate Down-regulates Doxorubicin-induced Overexpression of P-glycoprotein Through the Coordinate Inhibition of PI3K/Akt and MEK/ERK Signaling Pathways. Anticancer Res. 2017, 37, 6071–6077. [Google Scholar] [CrossRef] [PubMed]
- Lagiou, P.; Talamini, R.; Samoli, E.; Lagiou, A.; Ahrens, W.; Pohlabeln, H.; Benhamou, S.; Bouchardy, C.; Slamova, A.; Schejbalova, M.; et al. Diet and upper-aerodigestive tract cancer in Europe: The ARCAGE study. Int. J. Cancer 2009, 124, 2671–2676. [Google Scholar] [CrossRef]
| Characteristics | No Esophageal Neoplasm (n = 836) | Esophageal Low Grade Dysplasia (n = 88) | Esophageal High Grade Dysplasia/SCC (n = 63) | p Value |
|---|---|---|---|---|
| n (%) | n (%) | n (%) | ||
| Age (years) | 0.019 | |||
| <50 | 279 (33.4) | 20 (22.7) | 28 (44.4) | |
| ≥50 | 557 (66.6) | 68 (77.3) | 35 (55.6) | |
| Gender (Male) | 836 (100) | 88 (100) | 63 (100) | |
| Smoking | 0.013 | |||
| Non-smoker | 109 (13.0) | 7 (8.0) | 1 (1.6) | |
| Smoker | 727 (87.0) | 81 (92.0) | 62 (98.4) | |
| Alcohol drinking | <0.001 | |||
| Non-drinker | 233 (27.9) | 10 (11.6) | 1 (1.6) | |
| Drinker | 603 (72.1) | 78 (88.6) | 62 (98.4) | |
| Betel nut | 0.953 | |||
| Non-chewer | 184 (22.0) | 22 (25.0) | 13 (20.6) | |
| Chewer | 651 (77.9) | 66 (75.0) | 50 (79.1) | |
| Tea drinking | 0.025 | |||
| Non-consumer | 562 (67.2) | 53 (60.2) | 51 (81.0) | |
| Consumer | 274 (32.8) | 35 (39.8) | 12 (19.0) | |
| Cancer Stage | 0.029 | |||
| 0–I | 136 (16.3) | 14 (15.9) | 3 (4.8) | |
| II | 117 (14.0) | 15 (17.0) | 6 (9.5) | |
| III | 154 (18.4) | 8 (9.1) | 11 (17.5) | |
| IV | 429 (51.3) | 51 (58.0) | 43 (68.3) | |
| Location of index cancer | <0.001 | |||
| Oral cavity | 623 (74.5) | 55 (62.5) | 17 (27.0) | |
| Oropharynx | 117 (14.0) | 19 (21.6) | 15 (23.8) | |
| Hypopharynx | 68 (8.1) | 13 (14.8) | 26 (41.3) | |
| Larynx | 28 (3.3) | 1 (1.1) | 5 (7.9) |
| Characteristics | Low Grade Dysplasia vs. No Neoplasm | High Grade Dysplasia/SCC vs. No Neoplasm | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| cOR | 95% CI | aOR | 95% CI | p | cOR | 95% CI | aOR | 95% CI | p | |
| Age | ||||||||||
| <50 | 1 | 1 | 1 | 1 | ||||||
| ≥50 | 1.7 | 1.0–2.9 | 1.8 | 1.1–3.1 | 0.03 | 0.6 | 0.4–1.1 | 0.7 | 0.4–1.3 | 0.25 |
| Smoking | ||||||||||
| Non-smoker | 1 | 1 | 1 | 1 | ||||||
| Smoker | 1.7 | 0.8–3.9 | 1.7 | 0.7–4.0 | 0.22 | 9.3 | 1.3–67.7 | 5.6 | 0.8–42.2 | 0.09 |
| Alcohol | ||||||||||
| Non-drinker | 1 | 1 | 1 | 1 | ||||||
| Drinker | 3.0 | 1.5–5.9 | 3.4 | 1.7–7.0 | <0.01 | 23.9 | 3.3–173.4 | 21.3 | 2.9–156.6 | <0.01 |
| Betel nut | ||||||||||
| Non-chewer | 1 | 1 | 1 | 1 | ||||||
| Chewer | 0.8 | 0.5–1.4 | 0.6 | 0.4–1.1 | 0.09 | 1.1 | 0.6–2.0 | 0.6 | 0.3–1.1 | 0.08 |
| Tea drinking | ||||||||||
| Non-consumer | 1 | 1 | 1 | 1 | ||||||
| Consumer | 1.4 | 0.9–2.1 | 1.3 | 0.8–2.0 | 0.33 | 0.5 | 0.3–0.9 | 0.5 | 0.3–0.9 | 0.03 |
| Stage | ||||||||||
| 0–I | 1 | 1 | 1 | 1 | ||||||
| II | 1.2 | 0.6–2.7 | 1.4 | 0.6–3.0 | 0.45 | 2.3 | 0.6–9.5 | 2.8 | 0.7–11.7 | 0.16 |
| III | 0.5 | 0.2–1.2 | 0.5 | 0.2–1.2 | 0.11 | 3.2 | 0.9–11.8 | 2.9 | 0.8–10.7 | 0.11 |
| IV | 1.2 | 0.6–2.2 | 1.1 | 0.6–2.1 | 0.69 | 4.5 | 1.4–14.9 | 4.3 | 1.3–14.3 | 0.02 |
| Characteristics | No Neoplasm | High Grade Dysplasia/SCC | High Grade Dysplasia/SCC vs. No Neoplasm | ||
|---|---|---|---|---|---|
| n | n | aOR | 95% CI | p value | |
| Betel nut | |||||
| Betel nut non-chewer | |||||
| Tea non-consumer | 123 | 10 | 1 | ||
| Tea consumer | 61 | 3 | 0.8 | 0.2–3.3 | 0.705 |
| Betel nut chewer | |||||
| Tea non-consumer | 438 | 41 | 1 | ||
| Tea consumer | 213 | 9 | 0.5 | 0.2–0.9 | 0.045 |
| Alcohol | |||||
| Alcohol non-drinker | |||||
| Tea non-consumer | 151 | 1 | 1 | ||
| Tea consumer | 82 | 0 | - | - | 0.924 |
| Alcohol drinker | |||||
| Tea non-consumer | 411 | 50 | 1 | ||
| Tea consumer | 192 | 12 | 0.5 | 0.26–0.99 | 0.045 |
| Smoking | |||||
| Cigarette non-smoker | |||||
| Tea non-consumer | 78 | 1 | 1 | ||
| Tea consumer | 31 | 0 | - | - | 0.938 |
| Cigarette smoker | |||||
| Tea non-consumer | 484 | 50 | 1 | ||
| Tea consumer | 243 | 12 | 0.5 | 0.266–0.996 | 0.049 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.-K.; Chen, W.-C.; Lai, Y.-H.; Chen, Y.-H.; Wu, M.-T.; Kuo, C.-T.; Wang, Y.-Y.; Yuan, S.-S.F.; Liu, Y.-P.; Wu, I.-C. Influence of Tea Consumption on the Development of Second Esophageal Neoplasm in Patients with Head and Neck Cancer. Cancers 2019, 11, 387. https://doi.org/10.3390/cancers11030387
Wang Y-K, Chen W-C, Lai Y-H, Chen Y-H, Wu M-T, Kuo C-T, Wang Y-Y, Yuan S-SF, Liu Y-P, Wu I-C. Influence of Tea Consumption on the Development of Second Esophageal Neoplasm in Patients with Head and Neck Cancer. Cancers. 2019; 11(3):387. https://doi.org/10.3390/cancers11030387
Chicago/Turabian StyleWang, Yao-Kuang, Wei-Chung Chen, Ying-Ho Lai, Yi-Hsun Chen, Ming-Tsang Wu, Chie-Tong Kuo, Yen-Yun Wang, Shyng-Shiou F. Yuan, Yu-Peng Liu, and I-Chen Wu. 2019. "Influence of Tea Consumption on the Development of Second Esophageal Neoplasm in Patients with Head and Neck Cancer" Cancers 11, no. 3: 387. https://doi.org/10.3390/cancers11030387
APA StyleWang, Y.-K., Chen, W.-C., Lai, Y.-H., Chen, Y.-H., Wu, M.-T., Kuo, C.-T., Wang, Y.-Y., Yuan, S.-S. F., Liu, Y.-P., & Wu, I.-C. (2019). Influence of Tea Consumption on the Development of Second Esophageal Neoplasm in Patients with Head and Neck Cancer. Cancers, 11(3), 387. https://doi.org/10.3390/cancers11030387








