The Role of Single-Nucleotide Polymorphisms in Pituitary Adenomas Tumorigenesis
Abstract
1. Introduction
2. Single-Nucleotide Polymorphisms (SNPs) in Pituitary Adenomas by Tumor Type
2.1. Definitions–Mutation vs. Polymorphism
2.2. Brief Background on Mutations in Pituitary Adenomas
2.3. SNPs Found in PAs in General (Functional and Nonfunctional)
2.4. SNPs in Nonfunctional Pituitary Adenomas
2.5. SNPs in Functional Pituitary Adenomas
2.5.1. SNPs in Prolactin-Secreting PAs (Prolactinomas)
2.5.2. SNPs in Growth Hormone-Secreting PAs
2.5.3. SNPs in Adrenocorticotropin-Secreting PAs
2.5.4. SNPs in Other Hormone-Secreting PAs
3. Future Directions
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lake, M.G.; Krook, L.S.; Cruz, S.V. Pituitary adenomas: An overview. Am. Fam. Phys. 2013, 88, 319–327. [Google Scholar]
- Ostrom, Q.T.; Gittleman, H.; Truitt, G.; Boscia, A.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2011–2015. Neuro. Oncol. 2018, 20, iv1–iv86. [Google Scholar] [CrossRef] [PubMed]
- Daly, A.F.; Rixhon, M.; Adam, C.; Dempegioti, A.; Tichomirowa, M.A.; Beckers, A. High prevalence of pituitary adenomas: A cross-sectional study in the province of Liege, Belgium. J. Clin. Endocrinol. Metab. 2006, 91, 4769–4775. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, A.; Karavitaki, N.; Wass, J.A. Prevalence of pituitary adenomas: A community-based, cross-sectional study in Banbury (Oxfordshire, UK). Clin. Endocrinol. 2010, 73, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Ezzat, S.; Asa, S.L.; Couldwell, W.T.; Barr, C.E.; Dodge, W.E.; Vance, M.L.; McCutcheon, I.E. The prevalence of pituitary adenomas: A systematic review. Cancer 2004, 101, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Lopez, M.B.S. The 2017 World Health Organization classification of tumors of the pituitary gland: A summary. Acta Neuropathol. 2017, 134, 521–535. [Google Scholar] [CrossRef] [PubMed]
- Al-Brahim, N.Y.; Asa, S.L. My approach to pathology of the pituitary gland. J. Clin. Pathol. 2006, 59, 1245–1253. [Google Scholar] [CrossRef]
- Rick, J.; Jahangiri, A.; Flanigan, P.M.; Chandra, A.; Kunwar, S.; Blevins, L.; Aghi, M.K. Growth hormone and prolactin-staining tumors causing acromegaly: A retrospective review of clinical presentations and surgical outcomes. J. Neurosurg. 2018, 1, 1–7. [Google Scholar] [CrossRef]
- Molitch, M.E. Diagnosis and treatment of pituitary adenomas: A review. JAMA 2017, 317, 516–524. [Google Scholar] [CrossRef]
- Joshi, S.M.; Cudlip, S. Transsphenoidal surgery. Pituitary 2008, 11, 353–360. [Google Scholar] [CrossRef]
- Rolston, J.D.; Han, S.J.; Aghi, M.K. Nationwide shift from microscopic to endoscopic transsphenoidal pituitary surgery. Pituitary 2016, 19, 248–250. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhong, C.; Wang, Y.; Xu, S.; Guo, Y.; Dai, C.; Zheng, Y.; Wang, Y.; Luo, Q.; Jiang, J. Endoscopic versus microscopic transsphenoidal pituitary adenoma surgery: A meta-analysis. World J. Surg. Oncol. 2014, 12, 94. [Google Scholar] [CrossRef] [PubMed]
- Meji, B.P.; Lopex, M.B.; Ellegala, D.B.; Alden, T.D.; Laws, E.R. The long-term significance of microscopic dural invasion in 354 patients with pituitary adenomas treated with transsphenoidal surgery. J. Neurosurg. 2002, 96, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Javanmard, P.; Duan, D.; Geer, E.B. Mortality in patients with endogenous Cushing’s syndrome. Endocrinol. Metab. Clin. N. Am. 2018, 47, 313–333. [Google Scholar] [CrossRef]
- Bernabeu, I.; Aller, J.; Álvarez-Escolá, C.; Fajardo-Montañana, C.; Gálvez-Moreno, Á.; Guillín-Amarelle, C.; Sesmilo, G. Criteria for diagnosis and postoperative control of acromegaly, and screening and management of its comorbidities: Expert consensus. Endocrinol. Diabetes Nutr. 2018, 65, 297–305. [Google Scholar] [CrossRef]
- Rutkowski, M.; Zada, G. Managament of pituitary adenomas invading the cavernous sinus. Neurosurg. Clin. N. Am. 2019, 30, 445–455. [Google Scholar] [CrossRef]
- Faltermeier, C.M.; Magill, S.T.; Blevins, L.S.; Aghi, M.K. Molecular biology of pituitary adenomas. Neurosurg. Clin. N. Am. 2019, 30, 391–400. [Google Scholar] [CrossRef]
- Amundadottir, L.T.; Sulem, P.; Gudmundsson, J.; Helgason, A.; Baker, A.; Agnarsson, B.A.; Sigurdsson, A.; Benediktsdottir, K.R.; Cazier, J.B.; Sainz, J.; et al. A common variant associated with prostate cancer in European and African populations. Nat. Genet. 2006, 38, 652–658. [Google Scholar] [CrossRef]
- Rae, J.M.; Skaar, T.C.; Hilsenbeck, S.G.; Oesterreich, S. The role of single nucleotide polymorphisms in breast cancer metastasis. Breast Cancer Res. 2008, 10, 301. [Google Scholar] [CrossRef]
- Zlatkutė, E.; Liutkevičienė, R.; Vilkevičiūtė, A.; Glebauskienė, B.; Kriaučiūnienė, L.; Jakštienė, S.; Žaliūnienė, D. The role of MMP-1 and FGFR4-R388 gene polymorphisms in pituitary adenoma. Acta Med. Litu. 2017, 24, 177–190. [Google Scholar]
- Altaş, M.; Bayrak, O.F.; Ayan, E.; Bolukbasi, F.; Silav, G.; Coskun, K.K.; Culha, M.; Sahin, F.; Sevli, S.; Elmaci, I. The effect of polymorphisms in the promotor region of the MMP-1 gene on the occurrence and invasiveness of hypophyseal adenoma. Acta Neurochir. 2010, 152, 1611–1617. [Google Scholar] [CrossRef] [PubMed]
- Glebauskiene, B.; Liutkeviciene, R.; Vilkeviciute, A.; Kriauciuniene, L.; Jakstiene, S.; Zlatkute, E.; Tamosiunas, A.; Zemaitiene, R.; Vaitkiene, P.; Zaliuniene, D. Does MMP-9 gene polymorphism play a role in pituitary adenoma development? Dis. Markers 2017, 2017, 5839528. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, H.; Kawamoto, K.; Mizoue, T.; Uozumi, T.; Arita, K.; Kurisu, K. Matrix metalloproteinase-9 secretion by human pituitary adenomas detected by cell immunoblot analysis. Acta Neurochir. 1996, 138, 1442–1448. [Google Scholar] [CrossRef] [PubMed]
- Tampourlou, M.; Fountas, A.; Ntali, G.; Karavitaki, N. Mortality in patients with non-functioning pituitary adenomas. Pituitary 2018, 21, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Pierantoni, G.M.; Finelli, P.; Valtorta, E.; Giardino, D.; Rodeschini, O.; Esposito, F.; Losa, M.; Fusco, A.; Larizza, L. High-mobility group A2 gene expression is frequently induced in non-functioning pituitary adenomas (NFPAs), even in the absence of chromosome 12 polysomy. Endocr. Relat. Cancer 2005, 12, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.H.; Lee, J.H.; Seol, H.J.; Lee, J.I.; Kim, J.H.; Kong, D.S.; Nam, D.H. Clinical concerns about recurrence of non-functioning pituitary adenomas. Brain Tumor. Res. Treat. 2016, 4, 1–7. [Google Scholar] [CrossRef]
- Levy, M.J.; Robertson, I.J.; Khalk, N.; Vitello, S.; Reddy, N.; Bhake, R.; Howlett, T.A. Long-term follow-up of a large prospective cohort of patients with nonfunctioning pituitary adenomas: The outcome of a conservative management policy. Clin. Endocrinol. 2018, 89, 354–359. [Google Scholar] [CrossRef]
- Yagnik, G.; Jahangiri, A.; Chen, R.; Wagner, J.R.; Aghi, M.K. Role of a p53 polymorphism in the development of nonfunctional pituitary adenomas. Mol. Cell. Endocrinol. 2017, 446, 81–90. [Google Scholar] [CrossRef]
- Fischer, M. Census and evaluation of p53 target genes. Oncogene 2017, 36, 3943–3956. [Google Scholar] [CrossRef]
- Zhu, B.; Wang, J.; Qin, L.; Wang, L.; Zheng, Y.; Zhang, L.; Wang, W. FGFR2 gene polymorphism rs2981582 is associated with non-functioning pituitary adenomas in Chinese Han population: A case-control study. Biosci. Rep. 2018, 38. [Google Scholar] [CrossRef]
- Kim, L.H.; Kim, J.H.; Namgoong, S.; Cheong, H.S.; Yoon, S.J.; Kim, E.H.; Kim, S.H.; Kim, S.H.; Chang, J.H.; Shin, H.D. A PHLDB1 variant associated with the nonfunctional pituitary adenoma. J. Neurooncol. 2019, 142, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Ruggeri, R.M.; Santarpia, L.; Curto, L.; Torre, M.L.; Galatioto, M.; Galatioto, S.; Trimarchi, F.; Cannavo, S. Non-functioning pituitary adenomas infrequently harbor G-protein gene mutations. J. Endocrinol. Investig. 2008, 31, 946–949. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Yang, J.; Chang, Y.; Ma, S.; Qi, J. SNPs in the aryl hydrocarbon receptor-interacting protein gene associated with sporadic non-functioning pituitary adenoma. Exp. Ther. Med. 2016, 11, 1142–1146. [Google Scholar] [CrossRef] [PubMed]
- Mete, O.; Lopes, M.B. Overview of the 2017 WHO Classification of Pituitary Tumors. Endocr. Pathol. 2017, 28, 228–243. [Google Scholar] [CrossRef] [PubMed]
- Trouillas, J.; Delgrange, E.; Wierinckx, A.; Vasiljevic, A.; Jouanneau, E.; Burman, P.; Raverot, G. Clinical, Pathological, and Molecular Factors of Aggressiveness in Lactotroph Tumours. Neuroendocrinology 2019, 109, 70–76. [Google Scholar] [CrossRef]
- Ikeda, H. Mutational analysis of transforming growth factor-beta receptor type II and Smad3 tumor suppressor genes in prolactinomas. Brain Tumor Pathol. 2006, 23, 7–12. [Google Scholar] [CrossRef]
- Ikeda, H.; Yoshimoto, T.; Shida, N.; Miyoshi, I.; Nakayama, K.; Nakayama, K.; Oshima, M.; Taketo, M.M. Morphologic and molecular analysis of estrogen-induced pituitary tumorigenesis in targeted disruption of transforming growth factor-beta receptor type II and/or p27 mice. Endocrine 2001, 16, 55–65. [Google Scholar] [CrossRef]
- Peculis, R.; Balcere, I.; Rovite, V.; Megnis, K.; Valtere, A.; Stukens, J.; Arnicane, L.; Nikitina-Zake, L.; Lejnieks, A.; Pirags, V.; et al. Polymorphisms in MEN1 and DRD2 genes are associated with the occurrence and characteristics of pituitary adenomas. Eur. J. Endocrinol. 2016, 175, 145–153. [Google Scholar] [CrossRef]
- Cander, S.; Ertürk, E.; Karkucak, M.; Oz Gül, O.; Görükmez, O.; Yakut, T.; Unal, O.K.; Ersoy, C.; Tuncel, E.; Imamoğlu, S. Effect of cyclin D1 (CCND1) gene polymorphism on tumor formation and behavior in patients with prolactinoma. Gene 2012, 509, 158–163. [Google Scholar] [CrossRef]
- Schuuring, E. The involvement of the chromosome 11q13 region in human malignancies: Cyclin D1 and EMS1 are two new candidate oncogenes-a review. Gene 1995, 159, 83–96. [Google Scholar] [CrossRef]
- Simpson, D.J.; Fryer, A.A.; Grossman, A.B.; Wass, J.A.; Pfeifer, M.; Kros, J.M.; Clayton, R.N.; Farrell, W.E. Cyclin D1 (CCND1) genotype is associated with tumour grade in sporadic pituitary adenomas. Carcinogenesis 2001, 22, 1801–1807. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Athanasoulia, A.P.; Sievers, C.; Ising, M.; Brockhaus, A.C.; Yassouridis, A.; Stalla, G.K.; Uhr, M. Polymorphisms of the drug transporter gene ABCB1 predict side effects of treatment with cabergoline in patients with PRL adenomas. Eur. J. Endocrinol. 2012, 167, 327–335. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kiseljak-Vassiliades, K.; Carlson, N.E.; Borges, M.T.; Kleinschmidt-DeMasters, B.K.; Lillehei, K.O.; Kerr, J.M.; Wierman, M.E. Growth hormone tumor histological subtypes predict response to surgical and medical therapy. Endocrine 2015, 49, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Melmed, S. Pituitary medicine from discovery to patient-focused outcomes. J. Clin. Endocrinol. Metab. 2016, 101, 769–777. [Google Scholar] [CrossRef] [PubMed]
- Välimäki, N.; Demir, H.; Pitkänen, E.; Kaasinen, E.; Karppinen, A.; Kivipelto, L.; Schalin-Jäntti, C.; Aaltonen, L.A.; Karhu, A. Whole-Genome Sequencing of Growth Hormone (GH)-Secreting Pituitary Adenomas. J. Clin. Endocrinol. Metab. 2015, 100, 3918–3927. [Google Scholar] [CrossRef] [PubMed]
- Meier, K.; Knepel, W.; Schofl, C. Potassium depolarization elevates cytosolic free calcium concentration in rat anterior pituitary cells through 1,4-dihydropyridine-sensitive, omega-conotoxin-insensitive calcium channels. Endocrinology 1988, 122, 2764–2770. [Google Scholar] [CrossRef]
- Canny, B.J.; Rawlings, S.R.; Leong, D.A. Pituitary adenylate cyclase-activating polypeptide specifically increases cytosolic calcium ion concentration in rat gonadotropes and somatotropes. Endocrinology 1992, 130, 211–215. [Google Scholar] [CrossRef]
- Chen, Z.P.; Kratzmeier, M.; Levy, A.; McArdle, C.A.; Poch, A.; Day, A.; Mukhopadhyay, A.K.; Lightman, S.L. Evidence for a role of pituitary ATP receptors in the regulation of pituitary function. Proc. Natl. Acad. Sci. USA 1995, 92, 5219–5223. [Google Scholar] [CrossRef]
- Tateno, T.; Asa, S.L.; Zheng, L.; Mayr, T.; Ullrich, A.; Ezzat, S. The FGFR4-G388R polymorphism promotes mitochondrial STAT3 serine phosphorylation to facilitate pituitary growth hormone cell tumorigenesis. PLoS Genet. 2011, 7, e1002400. [Google Scholar] [CrossRef]
- Ezzat, S.; Wang, R.; Pintilie, M.; Asa, S.L. FGFR4 polymorphic alleles modulate mitochondrial respiration: A novel target for somatostatin analog action in pituitary tumors. Oncotarget 2017, 8, 3481–3494. [Google Scholar] [CrossRef]
- Yarman, S.; Ogret, Y.D.; Oguz, F.S. Do the aryl hydrocarbon receptor interacting protein variants (Q228K and Q307R) play a role in patients with familial and sporadic hormone-secreting pituitary adenomas? Genet. Test. Mol. Biomark. 2015, 19, 394–398. [Google Scholar] [CrossRef] [PubMed]
- Cannavo, S.; Ferrau, F.; Ragonese, M.; Romeo, P.D.; Torre, M.L.; Puglisi, S.; De Menis, E.; Arnaldi, G.; Salpietro, C.; Cotta, O.R.; et al. Increased frequency of the rs2066853 variant of aryl hydrocarbon receptor gene in patients with acromegaly. Clin. Endocrinol. 2014, 81, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Amorim, P.V.G.H.; Grande, I.P.P.; Batista, R.L.; Silveira, L.F.G.; Freire, A.C.T.B.; Bronstein, M.D.; Jallad, R.S.; Trarbach, E.B. Association between KISS1 rs5780218 promoter polymorphism and onset of growth hormone secreting pituitary adenoma. Ann. Endocrinol. (Paris) 2019, 80, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Silveira, L.G.; Latronico, A.C.; Seminara, S.B. Kisspeptin and clinical disorders. Adv. Exp. Med. Biol. 2013, 784, 187–199. [Google Scholar] [PubMed]
- Gao, M.; Zhu, B.; Xu, Z.; Liu, S.; Liu, J.; Zhang, G.; Gao, Y.; Fan, Y.; Kang, X. Association between acromegaly and a single nucleotide polymorphism (rs2854744) in the IGFBP3 gene. BMC Med. Genet. 2018, 19, 182. [Google Scholar] [CrossRef]
- Gao, M.; Zhu, B.; Li, P.; Zhang, G.; Chen, K.; Lv, H.; Ma, R.; Zhang, L.; Fan, Y.; Kang, X. Influence of the IGFBP3-202A/C Gene Polymorphism on Clinical Features and Surgery Outcome in Acromegalic Patients. Front. Endocrinol. (Lausanne) 2018, 9, 751. [Google Scholar] [CrossRef]
- Herman, V.; Fagin, J.; Gonsky, R.; Kovacs, K.; Melmed, S. Clonal origin of pituitary adenomas. J. Clin. Endocrinol. Metab. 1990, 71, 1427–1433. [Google Scholar] [CrossRef]
- Gicquel, C.; Le Bouc, Y.; Luton, J.P.; Girard, F.; Bertagna, X. Monoclonality of corticotroph macroadenomas in Cushing’s disease. J. Clin. Endocrinol. Metab. 1992, 75, 472–475. [Google Scholar]
- Biller, B.M.; Alexander, J.M.; Zervas, N.T.; Hedley-Whyte, E.T.; Arnold, A.; Klibanski, A. Clonal origins of adrenocorticotropin-secreting pituitary tissue in Cushing’s disease. J. Clin. Endocrinol. Metab. 1992, 75, 1303–1309. [Google Scholar]
- Tritos, N.A.; Biller, B.M.K. Current management of Cushing’s disease. J. Intern. Med. 2019, 286, 526–541. [Google Scholar] [CrossRef]
- Cassarino, M.F.; Sesta, A.; Pagliardini, L.; Losa, M.; Lasio, G.; Cavagnini, F.; Pecori Giraldi, F. Proopiomelanocortin, glucocorticoid, and CRH receptor expression in human ACTH-secreting pituitary adenomas. Endocrine 2017, 55, 853–860. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Huizenga, N.A.; de Lange, P.; Koper, J.W.; Clayton, R.N.; Farrell, W.E.; van der Lely, A.J.; Brinkmann, A.O.; de Jong, F.H.; Lamberts, S.W. Human adrenocorticotropin-secreting pituitary adenomas show frequent loss of heterozygosity at the glucocorticoid receptor gene locus. J. Clin. Endocrinol. Metab. 1998, 83, 917–921. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Antonini, S.R.; Latronico, A.C.; Elias, L.L.; Cukiert, A.; Machado, H.R.; Liberman, B.; Mendonca, B.B.; Moreira, A.C.; Castro, M. Glucocorticoid receptor gene polymorphisms in ACTH-secreting pituitary tumours. Clin. Endocrinol. 2002, 57, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Nakano-Tateno, T.; Tateno, T.; Hlaing, M.M.; Zheng, L.; Yoshimoto, K.; Yamada, S.; Asa, S.L.; Ezzat, S. FGFR4 polymorphic variants modulate phenotypic features of Cushing disease. Mol. Endocrinol. 2014, 28, 525–533. [Google Scholar] [CrossRef]
- Brito, L.P.; Lerario, A.M.; Bronstein, M.D.; Soares, I.C.; Mendonca, B.B.; Fragoso, M.C. Influence of the fibroblast growth factor receptor 4 expression and the G388R functional polymorphism on Cushing’s disease outcome. J. Clin. Endocrinol. Metab. 2010, 95, E271–E279. [Google Scholar] [CrossRef] [PubMed]
- Kottler, M.L.; Seret-Bégué, D.; Lahlou, N.; Assayag, M.; Carré, M.C.; Lagarde, J.P.; Ajzenberg, C.; Christin-Maitre, S.; Bouchard, P.; Mikol, J.; et al. The GnRH receptor gene is preferentially expressed in functioning gonadotroph adenomas and displays a Mae III polymorphism site. Clin. Endocrinol. 1998, 49, 115–123. [Google Scholar]
- Cossu, G.; Daniel, R.T.; Pierzchala, K.; Berhouma, M.; Pitteloud, N.; Lamine, F.; Colao, A.; Messerer, M. Thyrotropin-secreting pituitary adenomas: A systematic review and meta-analysis of postoperative outcomes and management. Pituitary 2019, 22, 79–88. [Google Scholar] [CrossRef]
- Amlashi, F.G.; Tritos, N.A. Thyrotropin-secreting pituitary adenomas: Epidemiology, diagnosis, and management. Endocrine 2016, 52, 427–440. [Google Scholar] [CrossRef]
- Sapkota, S.; Horiguchi, K.; Tosaka, M.; Yamada, S.; Yamada, M. Whole-exome sequencing study of thyrotropin-secreting pituitary adenomas. J. Clin. Endocrinol. Metab. 2017, 102, 566–575. [Google Scholar] [CrossRef]
- Filopanti, M.; Ballarè, E.; Lania, A.G.; Bondioni, S.; Verga, U.; Locatelli, M.; Zavanone, L.M.; Losa, M.; Gelmini, S.; Peri, A.; et al. Loss of heterozygosity at the SS receptor type 5 locus in human GH- and TSH-secreting pituitary adenomas. J. Endocrinol. Investig. 2004, 27, 937–942. [Google Scholar] [CrossRef]
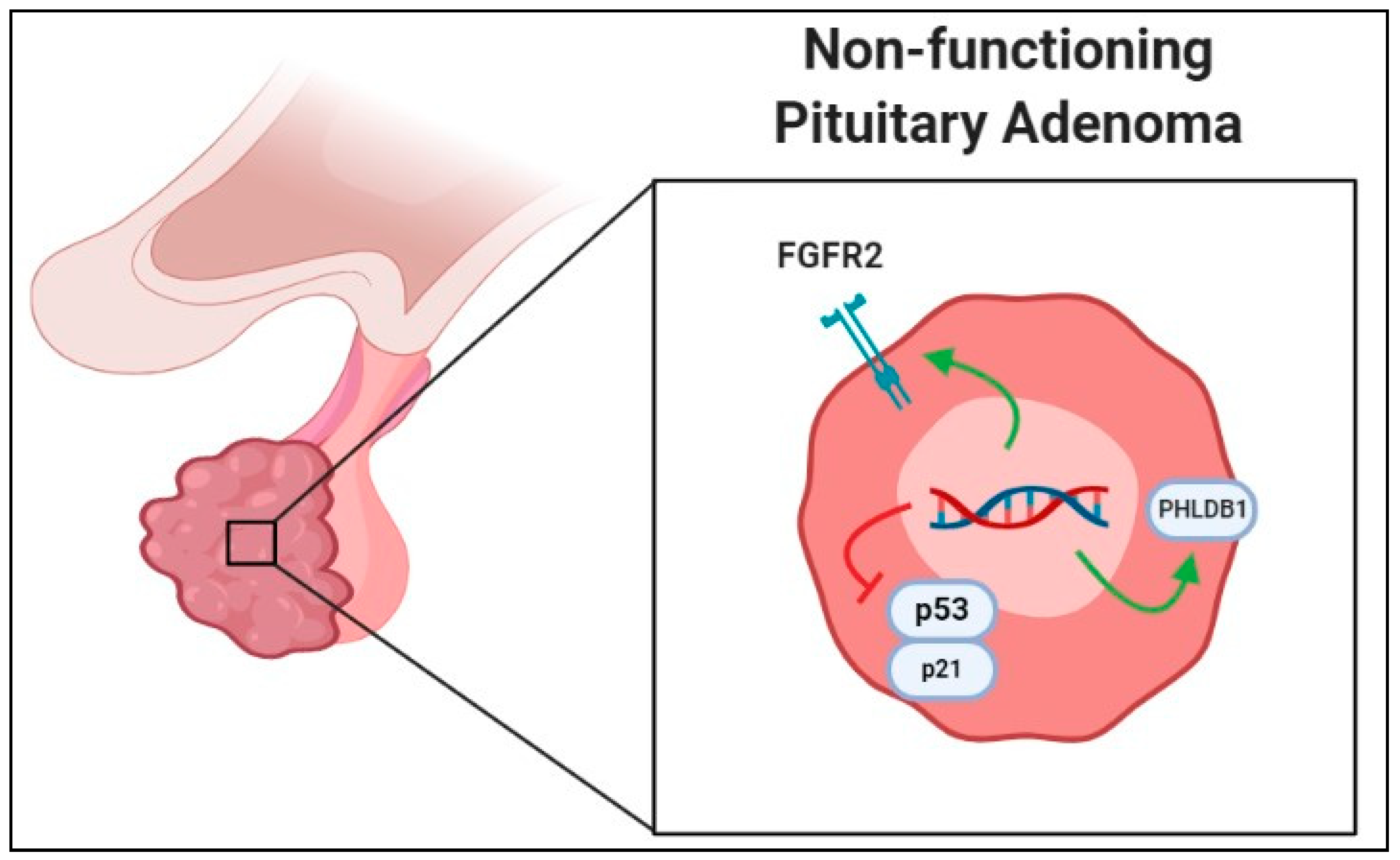
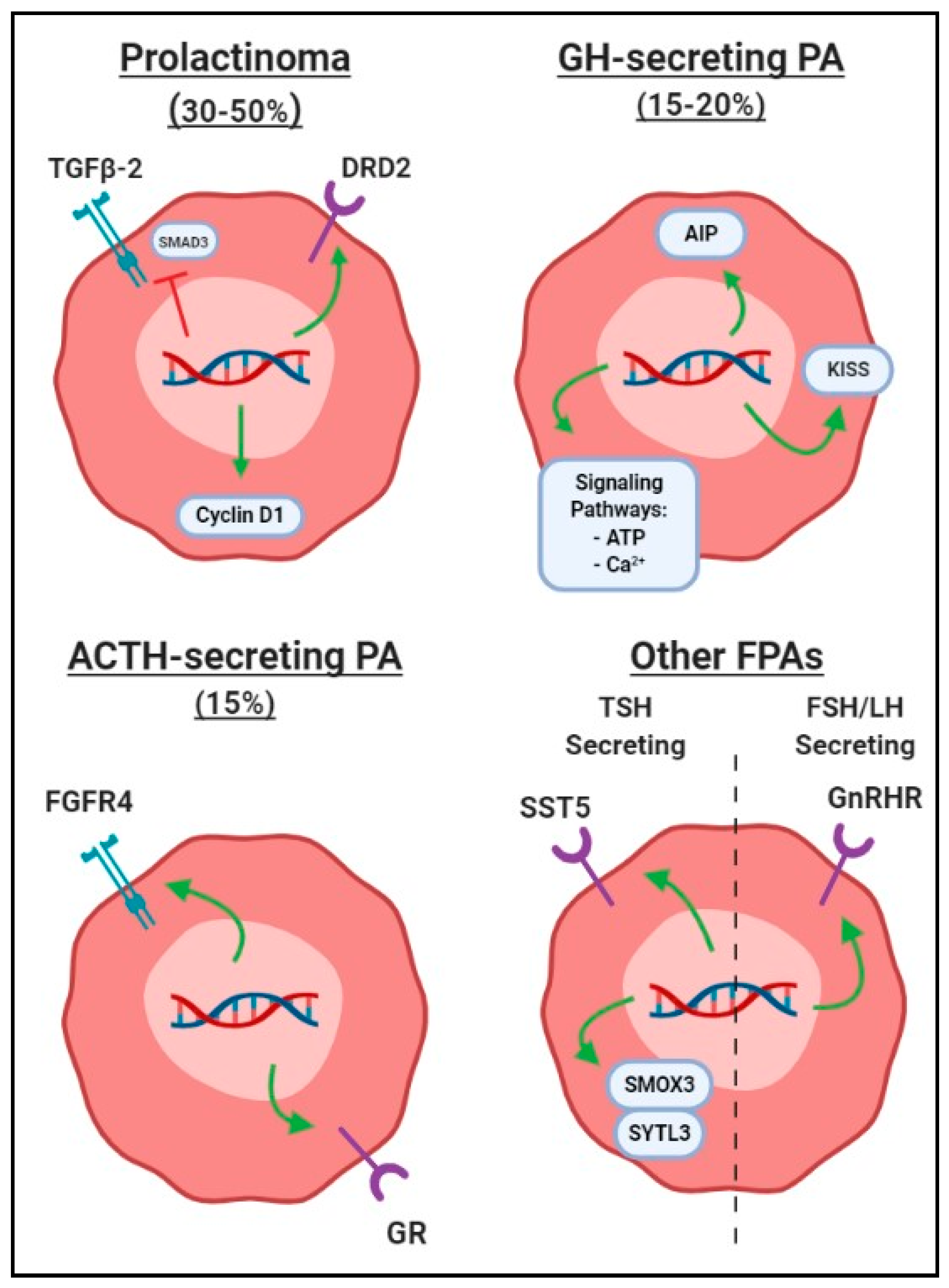
| Functional Category | Gene | Tumor Type | SNP | Relevance |
|---|---|---|---|---|
| Tumor Suppressor | p53 | NFPA | rs1042522 C > G | Increased cell proliferation and vascularity |
| TGF-B RII | PRL | rs2228048 G > A | Accelerated prolactinoma formation | |
| Growth Factor Receptor | FGFR2 | NFPA | rs2981582 | Increased activation of pro-mitogenic/survival downstream effectors |
| DRD2 | PRL | rs7131056 | Associated with extrasellar prolactinoma growth | |
| FGFR4 | GHPA | G388R | Increased cell proliferation, increase O2 consumption, and disruption of normal GH feedback response | |
| FGFR4 | ACTH-PA | G388R | Increased cell proliferation | |
| GR | ACTH-PA | N363S A > G | Unclear, higher in ACTH-PA patients | |
| N766N T > C | Unclear, higher in ACTH-PA patients | |||
| GnRHR | FSH/LSH | Nucl 453 C > T | Associated with functional gonadotropic adenomas > nonfunctional | |
| Cell Cycle Regulator | CCND1 | PRL-PA | G870A | Increased cell proliferation |
| Promoter | KISS1 | GHPA | rs57802180 | Increased KISS1 expression increases GH release |
| Drug-related | ABCB1 | PRL-PA | rs1045642 | Associated with central side effects in medically treated pts |
| rs2032582 | Associated with central side effects in medically treated pts | |||
| FGFR4 | GHPA | G388R | May modulate efficacy of somatostatin analog therapy | |
| Miscellaneous | PHCDB1 | NFPA | rs67307131 T > C | Unclear, possible genetic marker |
| AIP | GHPA | rs641081 Q228K | Associated with acromegalic patients > health controls | |
| rs4930195 Q307R | Associated with acromegalic patients > health controls | |||
| rs2066953 | Associated with acromegalic patients > health controls | |||
| IGFBP3 | GHPA | A > C (unspec) | Unclear, possible association with need for postoperative medical therapy | |
| MMP-1 | unspec | 1G/1G genotype | More frequent in invasive vs. non-invasive PAs | |
| MMP-9 | unspec | rs3918242 C > T | Higher MMP-9 observed in invasive vs. noninvasive PAs |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shah, S.S.; Aghi, M.K. The Role of Single-Nucleotide Polymorphisms in Pituitary Adenomas Tumorigenesis. Cancers 2019, 11, 1977. https://doi.org/10.3390/cancers11121977
Shah SS, Aghi MK. The Role of Single-Nucleotide Polymorphisms in Pituitary Adenomas Tumorigenesis. Cancers. 2019; 11(12):1977. https://doi.org/10.3390/cancers11121977
Chicago/Turabian StyleShah, Sumedh S., and Manish K. Aghi. 2019. "The Role of Single-Nucleotide Polymorphisms in Pituitary Adenomas Tumorigenesis" Cancers 11, no. 12: 1977. https://doi.org/10.3390/cancers11121977
APA StyleShah, S. S., & Aghi, M. K. (2019). The Role of Single-Nucleotide Polymorphisms in Pituitary Adenomas Tumorigenesis. Cancers, 11(12), 1977. https://doi.org/10.3390/cancers11121977




