Response to Lenvatinib Is Associated with Optimal RelativeDose Intensity in Hepatocellular Carcinoma: Experience in Clinical Settings
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Characteristics
2.2. Treatment Protocol and Relative Dose Intensity
2.3. Evaluation Criteria for Adverse Events and Response
2.4. Ethical Considerations
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Adverse Events
3.3. Response
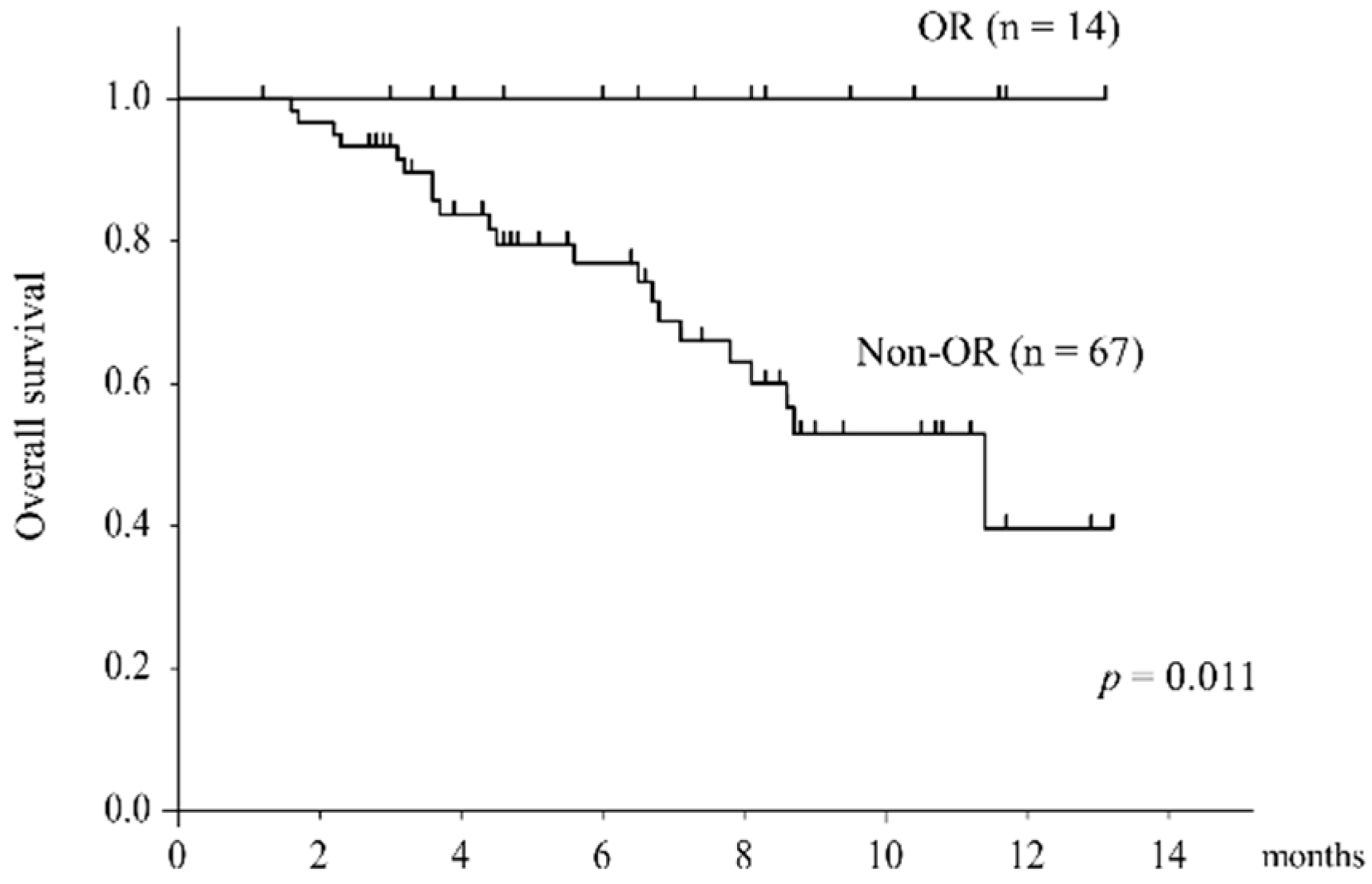
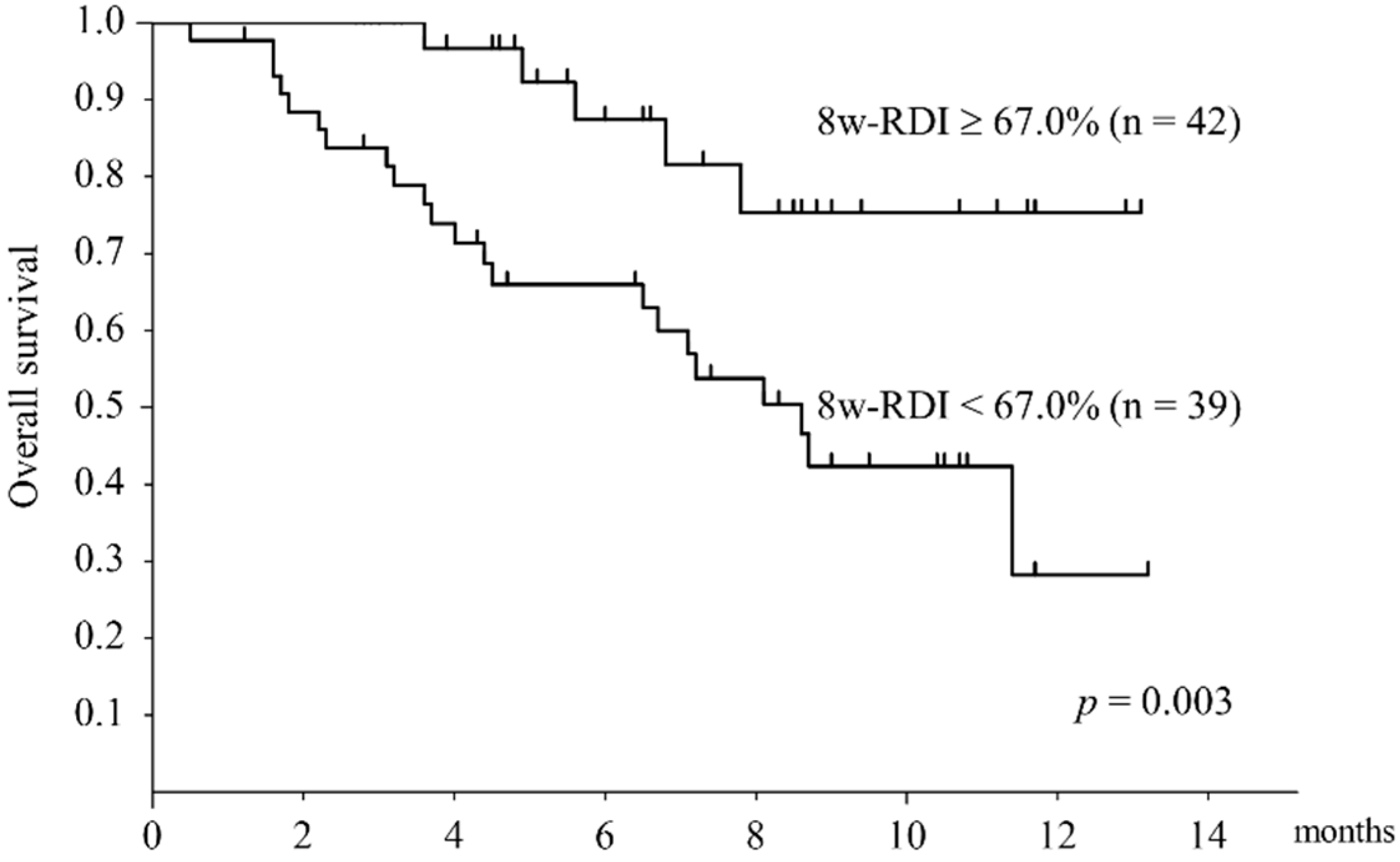
3.4. Overall Survival
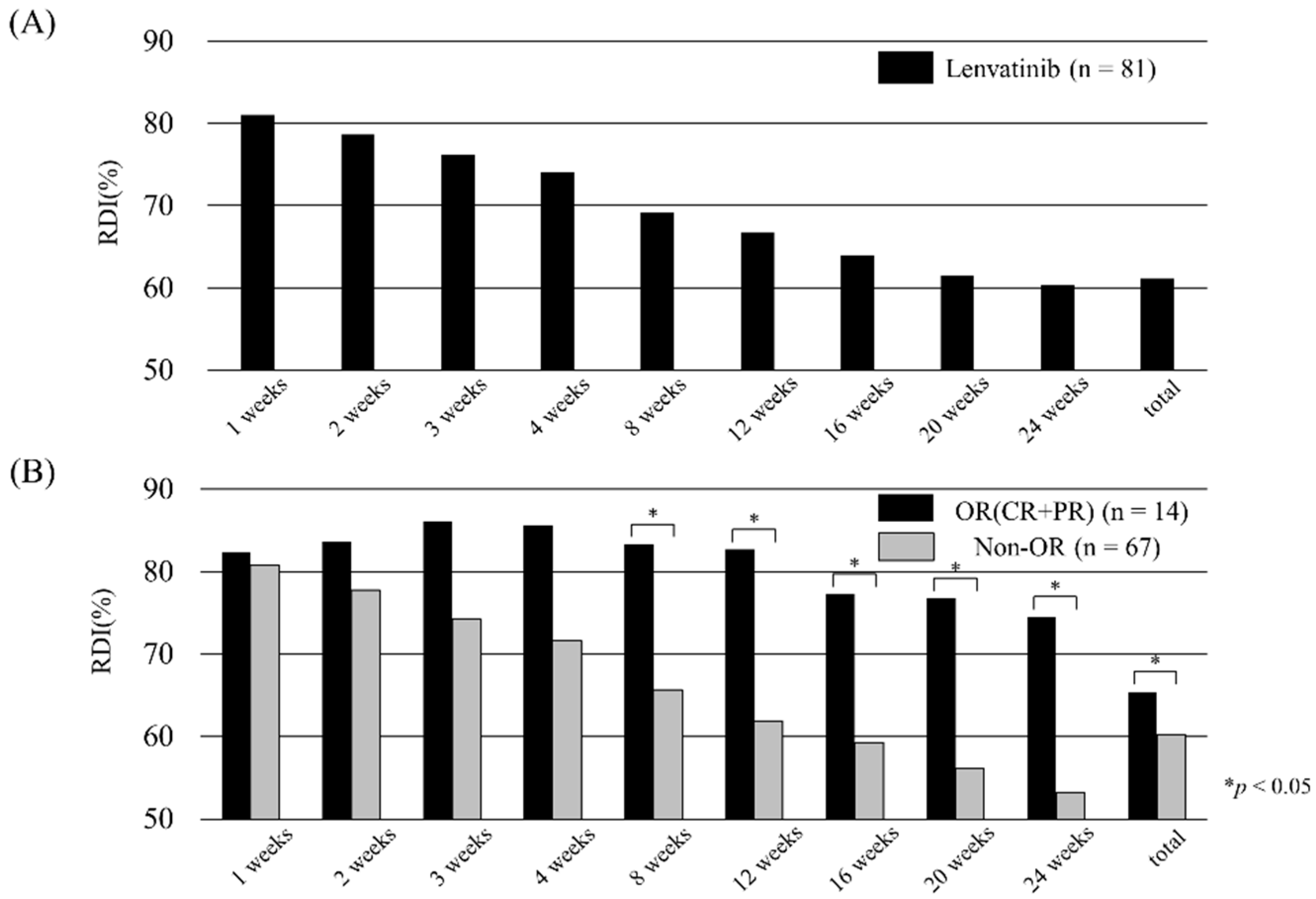
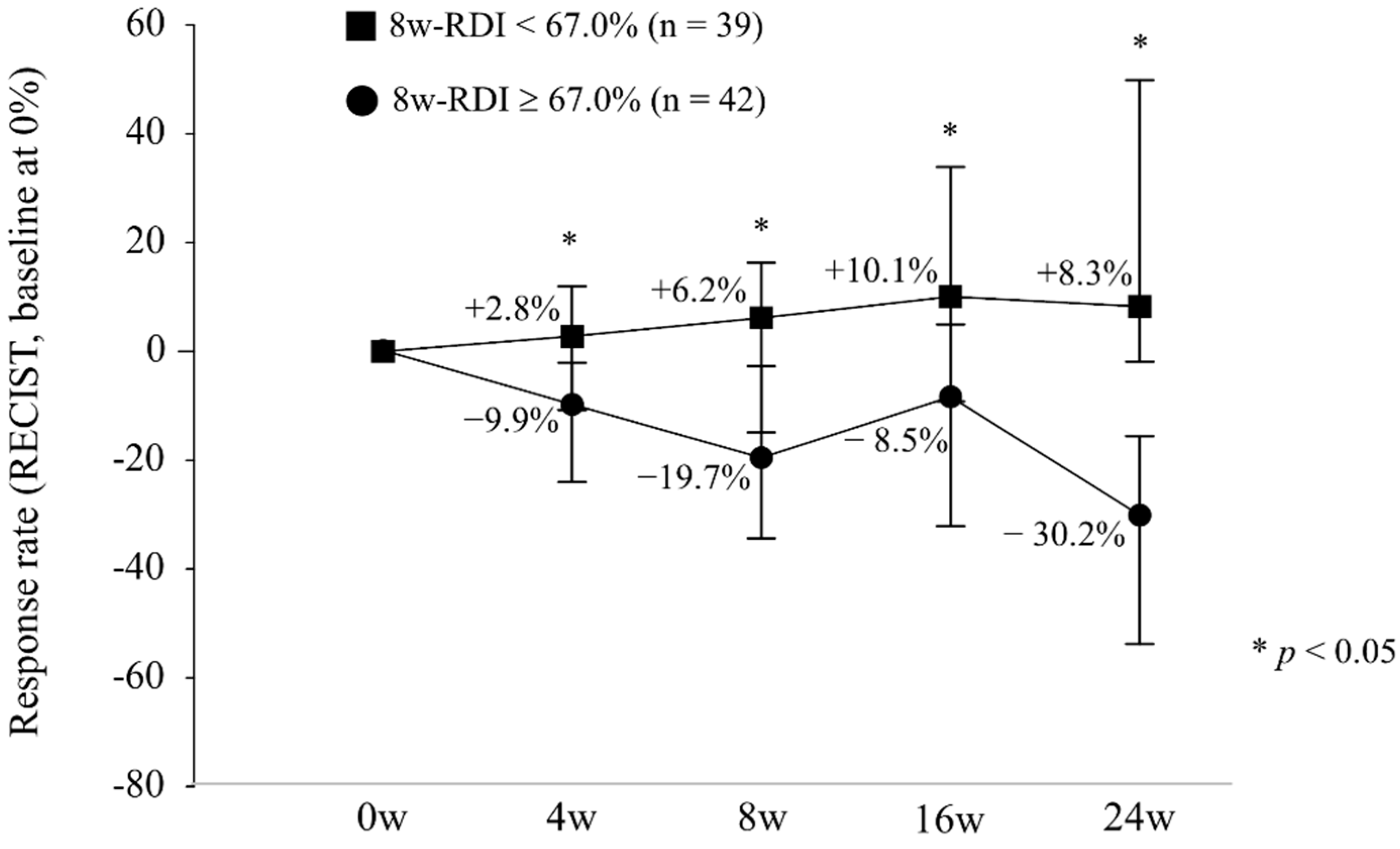
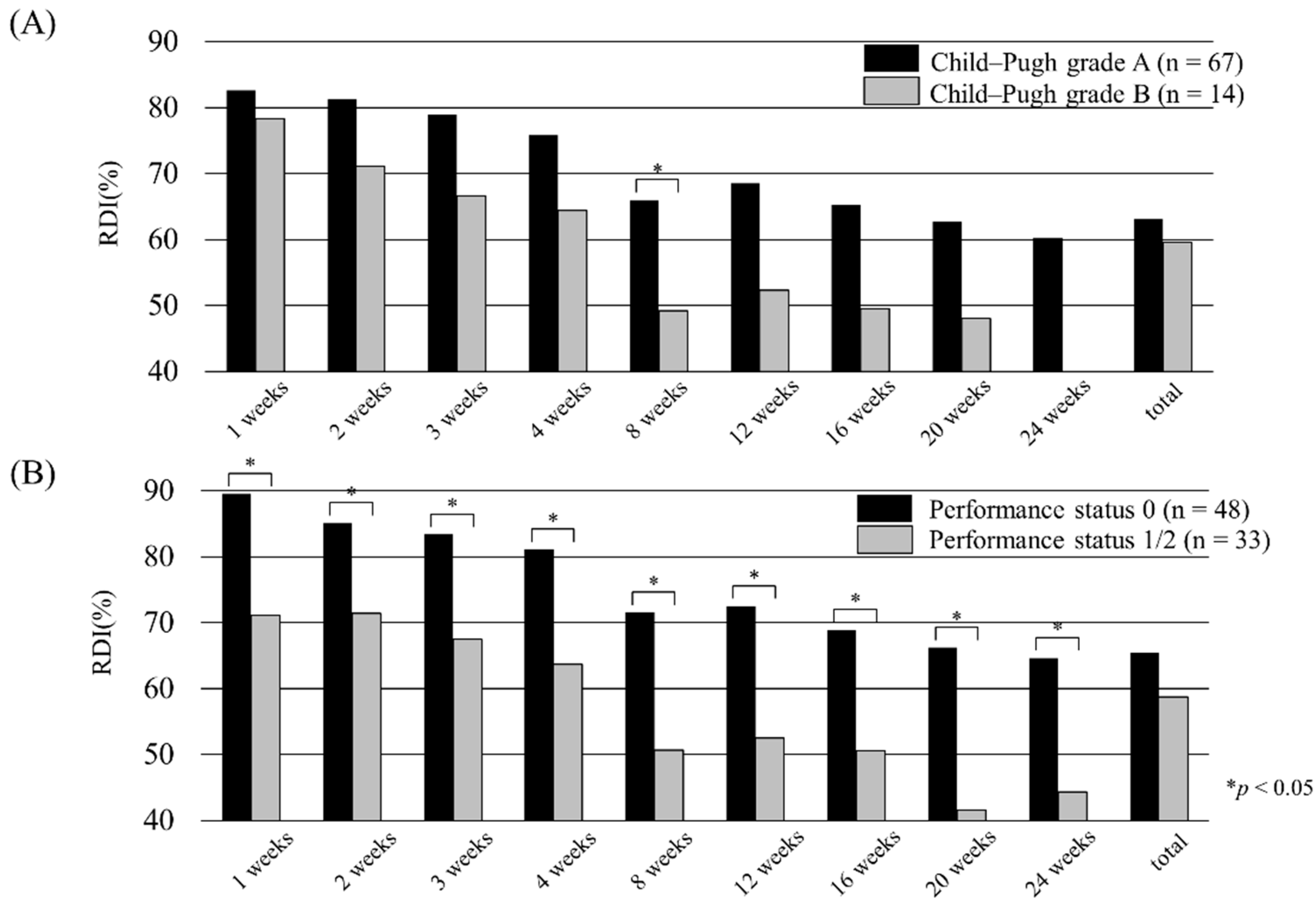
3.5. Relative Dose Intensity and Radiological Response
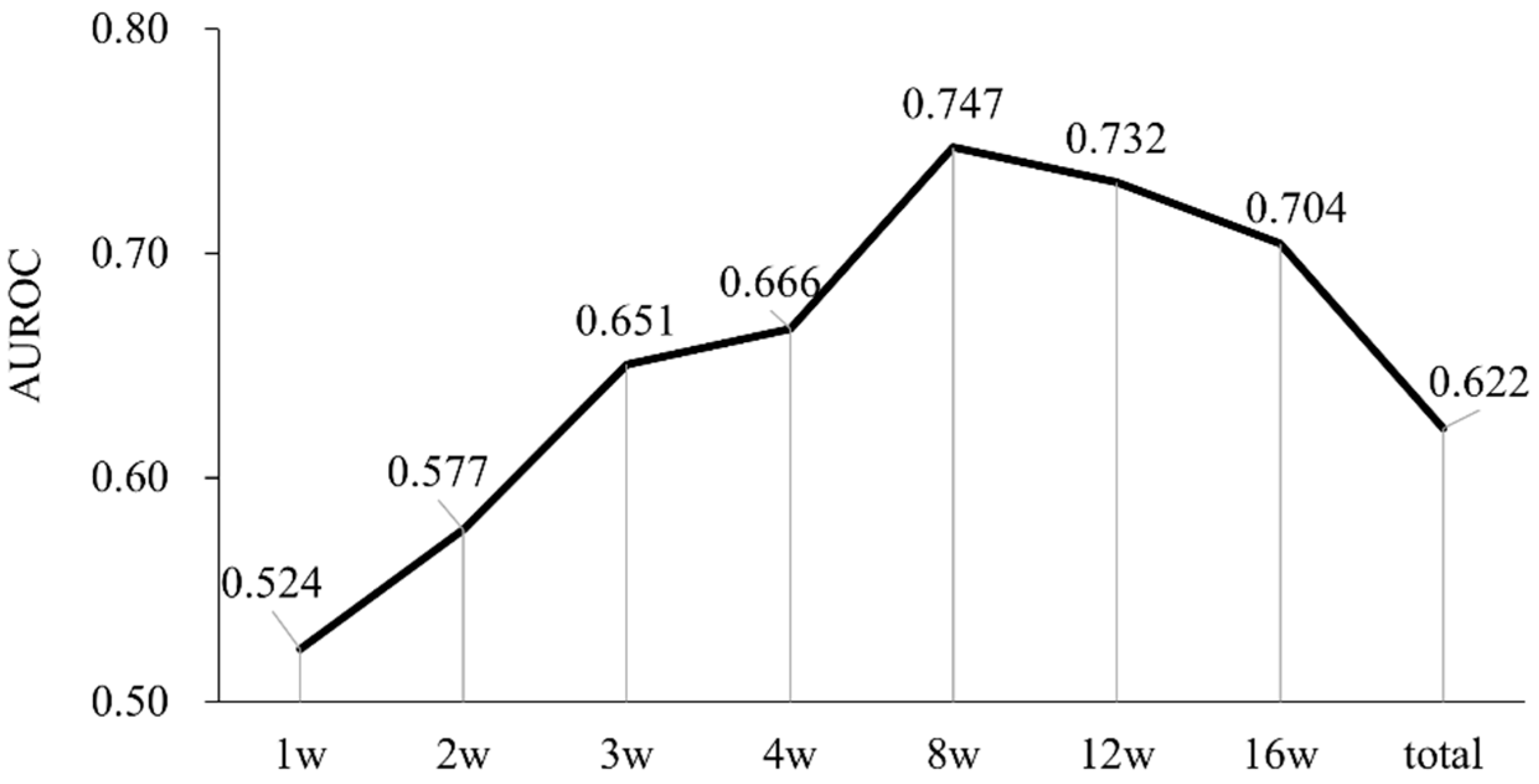
3.6. Predictive Ability of Relative Dose Intensity for Radiological Response
3.7. Overall Survival Stratified by Relative Dose Intensity
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bruix, J.; Llovet, J.M. Major achievements in hepatocellular carcinoma. Lancet 2009, 373, 614–616. [Google Scholar] [CrossRef]
- El-Serag, H.B. Hepatocellular carcinoma: An epidemiologic view. J. Clin. Gastroenterol. 2002, 35, S72–S78. [Google Scholar] [CrossRef] [PubMed]
- EASL Clinical Practice Guidelines. Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed]
- Omata, M.; Cheng, A.L.; Kokudo, N.; Kudo, M.; Lee, J.M.; Jia, J.; Tateishi, R.; Han, K.H.; Chawla, Y.K.; Shiina, S.; et al. Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: A 2017 update. Hepatol. Int. 2017, 11, 317–370. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Zhu, A.X.; Kang, Y.K.; Yen, C.J.; Finn, R.S.; Galle, P.R.; Llovet, J.M.; Assenat, E.; Brandi, G.; Pracht, M.; Lim, H.Y.; et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased alpha-fetoprotein concentrations (REACH-2): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019, 20, 282–296. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Meyer, T.; Cheng, A.L.; El-Khoueiry, A.B.; Rimassa, L.; Ryoo, B.Y.; Cicin, I.; Merle, P.; Chen, Y.; Park, J.W.; et al. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N. Engl. J. Med. 2018, 379, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.L.; Kang, Y.K.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: A phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Ueshima, K.; Nishida, N.; Hagiwara, S.; Aoki, T.; Minami, T.; Chishina, H.; Takita, M.; Minami, Y.; Ida, H.; Takenaka, M.; et al. Impact of Baseline ALBI Grade on the Outcomes of Hepatocellular Carcinoma Patients Treated with Lenvatinib: A Multicenter Study. Cancers (Basel) 2019, 11, 952. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Kumada, T.; Atsukawa, M.; Hirooka, M.; Tsuji, K.; Ishikawa, T.; Takaguchi, K.; Kariyama, K.; Itobayashi, E.; Tajiri, K.; et al. Prognostic factor of lenvatinib for unresectable hepatocellular carcinoma in real-world conditions-Multicenter analysis. Cancer Med. 2019, 8, 3719–3728. [Google Scholar] [CrossRef] [PubMed]
- Capdevila, J.; Newbold, K.; Licitra, L.; Popovtzer, A.; Moreso, F.; Zamorano, J.; Kreissl, M.; Aller, J.; Grande, E. Optimisation of treatment with lenvatinib in radioactive iodine-refractory differentiated thyroid cancer. Cancer Treat. Rev. 2018, 69, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin. Liver Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Matsui, J.; Yamamoto, Y.; Funahashi, Y.; Tsuruoka, A.; Watanabe, T.; Wakabayashi, T.; Uenaka, T.; Asada, M. E7080, a novel inhibitor that targets multiple kinases, has potent antitumor activities against stem cell factor producing human small cell lung cancer H146, based on angiogenesis inhibition. Int. J. Cancer 2008, 122, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Boss, D.S.; Glen, H.; Beijnen, J.H.; Keesen, M.; Morrison, R.; Tait, B.; Copalu, W.; Mazur, A.; Wanders, J.; O'Brien, J.P.; et al. A phase I study of E7080, a multitargeted tyrosine kinase inhibitor, in patients with advanced solid tumours. Br. J. Cancer 2012, 106, 1598–1604. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, K.; Kudo, M.; Kawazoe, S.; Osaki, Y.; Ikeda, M.; Okusaka, T.; Tamai, T.; Suzuki, T.; Hisai, T.; Hayato, S.; et al. Phase 2 study of lenvatinib in patients with advanced hepatocellular carcinoma. J. Gastroenterol. 2017, 52, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Okusaka, T.; Mitsunaga, S.; Ueno, H.; Tamai, T.; Suzuki, T.; Hayato, S.; Kadowaki, T.; Okita, K.; Kumada, H. Safety and Pharmacokinetics of Lenvatinib in Patients with Advanced Hepatocellular Carcinoma. Clin. Cancer Res. 2016, 22, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Hayato, S.; Shumaker, R.; Ferry, J.; Binder, T.; Dutcus, C.E.; Hussein, Z. Exposure-response analysis and simulation of lenvatinib safety and efficacy in patients with radioiodine-refractory differentiated thyroid cancer. Cancer Chemother. Pharmacol. 2018, 82, 971–978. [Google Scholar] [CrossRef] [PubMed]
- Houk, B.E.; Bello, C.L.; Poland, B.; Rosen, L.S.; Demetri, G.D.; Motzer, R.J. Relationship between exposure to sunitinib and efficacy and tolerability endpoints in patients with cancer: Results of a pharmacokinetic/pharmacodynamic meta-analysis. Cancer Chemother. Pharmacol. 2010, 66, 357–371. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Tsuchiya, K.; Kurosaki, M.; Yasui, Y.; Inada, K.; Kirino, S.; Yamashita, K.; Sekiguchi, S.; Hayakawa, Y.; Osawa, L.; et al. Sorafenib-Regorafenib Sequential Therapy in Japanese Patients with Unresectable Hepatocellular Carcinoma-Relative Dose Intensity and Post-Regorafenib Therapies in Real World Practice. Cancers (Basel) 2019, 11, 1517. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, A.; Moriguchi, M.; Seko, Y.; Ishikawa, H.; Yo, T.; Kimura, H.; Fujii, H.; Shima, T.; Mitsumoto, Y.; Ishiba, H.; et al. Impact of Relative Dose Intensity of Early-phase Lenvatinib Treatment on Therapeutic Response in Hepatocellular Carcinoma. Anticancer Res. 2019, 39, 5149–5156. [Google Scholar] [CrossRef] [PubMed]

| Variable | n = 81 | |
|---|---|---|
| Age | year | 72.0 (41–88) |
| Gender | male/female | 66/15 |
| BMI | kg/m2 | 24.10 (17.5–34.4) |
| Performance status | 0/1/2 | 48/28/5 |
| Child–Pugh grade | A/B | 67/14 |
| ALBI grade | 1/2/3 | 20/57/4 |
| Macroscopic PV invasion | Vp3 or Vp4 | 14 (17.2) |
| Extrahepatic spread | + | 34 (41.9) |
| BCLC stage | A/B/C | 1/19/61 |
| Etiology | HBV/HCV/NBNC | 22/26/33 |
| Platelet count | ×104/μL | 12.30 (4.0–38.1) |
| PT | % | 88.0 (47–128) |
| T.bil | mg/dL | 0.90 (0.4–2.6) |
| Albumin | g/dL | 3.50 (2.2–4.6) |
| ALT | IU/mL | 27.0 (11–198) |
| AFP | ng/ml | 68.8 (1.8–23,124) |
| DCP | mAU/ml | 484.0 (10–990,474) |
| Systemic therapy | naïve/experienced | 66/15 |
| Dose reduction | at administration | 32 (39.5) |
| Treatment-Emergent Adverse Events | Any Grade | Grade ≥ 3 |
|---|---|---|
| All adverse events | 79 (97.5%) | 35 (43.2%) |
| Hypertension | 50 (61.7%) | 11 (13.6%) |
| Fatigue | 47 (58.0%) | 1 (1.2%) |
| Decreased appetite | 46 (56.7%) | 3 (3.7%) |
| Hypothyroidism | 42 (51.8%) | 0 (0.0%) |
| Proteinuria | 37 (45.6%) | 7 (8.6%) |
| Palmar–plantar erythrodysesthesia syndrome | 31 (38.2%) | 0 (0.0%) |
| Thrombocytopenia | 30 (37.0%) | 3 (3.7%) |
| Elevated liver enzymes | 25 (30.8%) | 6 (7.4%) |
| Diarrhea | 20 (24.6%) | 0 (0.0%) |
| Weight loss | 18 (22.2%) | 1 (1.2%) |
| Dysphonia | 12 (14.8%) | 0 (0.0%) |
| edema | 11 (13.5%) | 1 (1.2%) |
| Rash | 5 (6.1%) | 0 (0.0%) |
| Loss of hair | 1 (1.2%) | 0 (0.0%) |
| Response Category | Lenvatinib (n = 81) | |
|---|---|---|
| mRECIST | RECIST | |
| Complete response (CR) | 7 (8.6%) | 3(3.7%) |
| Partial response (PR) | 21 (25.9%) | 11 (13.6%) |
| Stable disease (SD) | 23 (28.4%) | 38 (46.9%) |
| Progressive disease (PD) | 20 (24.7%) | 23 (28.4%) |
| Unknown or not evaluable | 10 (12.3%) | 6 (7.4%) |
| Objective response (OR) | 28 (34.6%) | 14 (17.3%) |
| Disease control (DC) | 51 (63.0%) | 52 (64.2%) |
| Factors | Child–Pugh Grade | Performance Status | ||
|---|---|---|---|---|
| grade A (n = 67) | grade B (n = 14) | PS 0 (n =48) | PS 1/2 (n = 33) | |
| Complete response (CR) | 3 (4.4%) | 0 (0.0%) | 2 (4.2%) | 1 (3.0%) |
| Partial response (PR) | 11 (16.4%) | 0 (0.0%) | 10 (20.8%) | 1 (3.0%) |
| Stable disease (SD) | 32 (47.7%) | 6 (42.9%) | 24 (50.0%) | 14 (42.4%) |
| Progressive disease (PD) | 18 (26.8%) | 5 (35.7%) | 10 (20.8%) | 13 (39.3%) |
| Unknown or not evaluable | 3 (4.4%) | 3 (21.4%) | 1 (2.1%) | 5 (15.1%) |
| Objective response (OR) | 14 (20.8%) | 0 (0.0%) | 12 (25.0%) | 2 (6.0%) |
| Disease control (DC) | 46 (68.6%) | 6 (42.9%) | 36 (75.0%) | 16 (48.4%) |
| Variable | 8w-RDI ≥ 67.0% | 8w-RDI < 67.0% | p-Value | |
|---|---|---|---|---|
| Age | year | 71.0 (46–84) | 76.0 (41–88) | 0.065 |
| Gender | male/female | 37/5 | 29/10 | 0.111 |
| BMI | kg/m2 | 24.65 (18.6–33.6) | 22.15 (17.5–34.4) | 0.019 |
| Performance status | 0/1/2 | 34/8/0 | 14/20/5 | <0.001 |
| Child–Pugh grade | A/B | 38/4 | 29/10 | 0.055 |
| ALBI grade | 1/2/3 | 11/31/0 | 9/26/4 | 0.103 |
| Macroscopic PV invasion | Vp3 or Vp4 | 7 (16.7) | 7 (17.9) | 0.878 |
| Extrahepatic spread | + | 16 (38.1) | 18 (46.2) | 0.462 |
| BCLC stage | A/B/C | 1/16/25 | 0/3/36 | 0.002 |
| Etiology | HBV/HCV/NBNC | 13/12/17 | 9/14/16 | 0.669 |
| Platelet count | ×104/μL | 14.85 (4.6–38.1) | 10.60 (4.0–28.0) | 0.015 |
| PT | % | 90.5 (64–128) | 84.0 (47–118) | 0.013 |
| T.bil | mg/dL | 0.90 (0.4–1.8) | 1.00 (0.4–2.6) | 0.307 |
| Albumin | g/dL | 3.70 (2.8–4.6) | 3.50 (2.2–4.5) | 0.028 |
| ALT | IU/mL | 27.5 (11–143) | 26.0 (11–198) | 0.909 |
| AFP | ng/mL | 43.0 (1.8–9926) | 140.0 (1.9–23,124) | 0.061 |
| DCP | mAU/mL | 572.5 (10–78884) | 458.0 (11–990,474) | 0.609 |
| Systemic therapy | naïve/experienced | 36/6 | 30/9 | 0.308 |
| Dose reduction | at administration | 9 (21.4) | 23 (59.0) | <0.001 |
| Results given as median (range) or n (%). | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sasaki, R.; Fukushima, M.; Haraguchi, M.; Miuma, S.; Miyaaki, H.; Hidaka, M.; Eguchi, S.; Matsuo, S.; Tajima, K.; Matsuzaki, T.; et al. Response to Lenvatinib Is Associated with Optimal RelativeDose Intensity in Hepatocellular Carcinoma: Experience in Clinical Settings. Cancers 2019, 11, 1769. https://doi.org/10.3390/cancers11111769
Sasaki R, Fukushima M, Haraguchi M, Miuma S, Miyaaki H, Hidaka M, Eguchi S, Matsuo S, Tajima K, Matsuzaki T, et al. Response to Lenvatinib Is Associated with Optimal RelativeDose Intensity in Hepatocellular Carcinoma: Experience in Clinical Settings. Cancers. 2019; 11(11):1769. https://doi.org/10.3390/cancers11111769
Chicago/Turabian StyleSasaki, Ryu, Masanori Fukushima, Masafumi Haraguchi, Satoshi Miuma, Hisamitsu Miyaaki, Masaaki Hidaka, Susumu Eguchi, Satoshi Matsuo, Kazuaki Tajima, Toshihisa Matsuzaki, and et al. 2019. "Response to Lenvatinib Is Associated with Optimal RelativeDose Intensity in Hepatocellular Carcinoma: Experience in Clinical Settings" Cancers 11, no. 11: 1769. https://doi.org/10.3390/cancers11111769
APA StyleSasaki, R., Fukushima, M., Haraguchi, M., Miuma, S., Miyaaki, H., Hidaka, M., Eguchi, S., Matsuo, S., Tajima, K., Matsuzaki, T., Hashimoto, S., Ooba, K., Kugiyama, Y., Yatsuhashi, H., Motoyoshi, Y., Shigeno, M., Kinoshita, N., & Nakao, K. (2019). Response to Lenvatinib Is Associated with Optimal RelativeDose Intensity in Hepatocellular Carcinoma: Experience in Clinical Settings. Cancers, 11(11), 1769. https://doi.org/10.3390/cancers11111769






