The Lung Immune Prognostic Index Discriminates Survival Outcomes in Patients with Solid Tumors Treated with Immune Checkpoint Inhibitors
Abstract
1. Introduction
2. Results
2.1. Baseline Characteristics
2.2. Overall Survival (OS)
2.3. Progression-Free Survival (PFS)
2.4. Objective-Response Rate (ORR)
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Borghaei, H.; Paz-Ares, L.; Horn, L.; Spigel, D.R.; Steins, M.; Ready, N.E.; Chow, L.Q.; Vokes, E.E.; Felip, E.; Holgado, E.; et al. Nivolumab versus Docetaxel in advanced Nonsquamous Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 1627–1639. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Pembrolizumab versus Chemotherapy for PD-L1-Positive Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2016, 375, 1823–1833. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Tannir, N.M.; McDermott, D.F.; Arén Frontera, O.; Melichar, B.; Choueiri, T.K.; Plimack, E.R.; Barthélémy, P.; Porta, C.; George, S.; et al. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2018, 378, 1277–1290. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Escudier, B.; McDermott, D.F.; George, S.; Hammers, H.J.; Srinivas, S.; Tykodi, S.S.; Sosman, J.A.; Procopio, G.; Plimack, E.R.; et al. Nivolumab versus Everolimus in Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2015, 373, 1803–1813. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Ribas, A.; Schachter, J.; Arance, A.; Grob, J.J.; Mortier, L.; Daud, A.; Carlino, M.S.; McNeil, C.M.; Lotem, M.; et al. Pembrolizumab versus Ipilimumab in advanced Melanoma (KEYNOTE-006): Post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 Study. Lancet Oncol. 2019, 20, 1239–1251. [Google Scholar] [CrossRef]
- Postow, M.A.; Hellmann, M.D. Adverse events associated with immune checkpoint blockade. N. Engl. J. Med. 2018, 378, 1163–1165. [Google Scholar] [CrossRef]
- Verma, V.; Sprave, T.; Haque, W.; Simone, C.B.; Chang, J.Y.; Welsh, J.W.; Thomas, C.R. A systematic review of the cost and cost-effectiveness studies of immune checkpoint inhibitors. J. Immuno Ther. Cancer 2018, 6, 128. [Google Scholar] [CrossRef]
- Postow, M.A.; Callahan, M.K.; Wolchok, J.D. Immune checkpoint blockade in cancer therapy. J. Clin. Oncol. 2015, 33, 1974–1982. [Google Scholar] [CrossRef]
- Patel, S.P.; Kurzrock, R. PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol. Cancer Ther. 2015, 14, 847–856. [Google Scholar] [CrossRef]
- Samstein, R.M.; Lee, C.H.; Shoushtari, A.N.; Hellmann, M.D.; Shen, R.; Janjigian, Y.Y.; Barron, D.A.; Zehir, A.; Jordan, E.J.; Omuro, A.; et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat. Genet. 2019, 51, 202–206. [Google Scholar] [CrossRef]
- Havel, J.J.; Chowell, D.; Chan, T.A. The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat. Rev. Cancer 2019, 19, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Mezquita, L.; Auclin, E.; Ferrara, R.; Charrier, M.; Remon, J.; Planchard, D.; Ponce, S.; Ares, L.P.; Leroy, L.; Audigier-Valette, C.; et al. Association of the Lung Immune Prognostic Index with immune checkpoint inhibitor outcomes in patients with advanced Non–Small Cell Lung Cancer. JAMA Oncol. 2018, 4, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Sorich, M.J.; Rowland, A.; Karapetis, C.S.; Hopkins, A.M. Evaluation of the Lung Immune Prognostic Index for prediction of survival and response in patients treated with Atezolizumab for Non-Small Cell Lung Cancer: Pooled analysis of clinical trials. J. Thorac. Oncol. 2019, 14, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Kazandjian, D.; Gong, Y.; Keegan, P.; Pazdur, R.; Blumenthal, G.M. Prognostic value of the Lung Immune Prognostic Index for patients treated for metastatic Non–Small Cell Lung Cancer. JAMA Oncol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Hamid, O.; Robert, C.; Daud, A.; Hodi, F.S.; Hwu, W.J.; Kefford, R.; Wolchok, J.D.; Hersey, P.; Joseph, R.; Weber, J.S.; et al. Five-year survival outcomes for patients with advanced melanoma treated with pembrolizumab in KEYNOTE-001. Ann. Oncol. 2019, 30, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Updated Analysis of KEYNOTE-024: Pembrolizumab versus platinum-based chemotherapy for advanced Non-Small-Cell Lung Cancer with PD-L1 tumor proportion ccore of 50% or greater. J. Clin. Oncol. 2019, 37, 537–546. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Hopkins, A.M.; Rowland, A.; Kichenadasse, G.; Wiese, M.D.; Gurney, H.; McKinnon, R.A.; Karapetis, C.S.; Sorich, M.J. Predicting response and toxicity to immune checkpoint inhibitors using routinely available blood and clinical markers. Br. J. Cancer 2017, 117, 913–920. [Google Scholar] [CrossRef]
- Buder-Bakhaya, K.; Hassel, J.C. Biomarkers for Clinical Benefit of Immune Checkpoint Inhibitor Treatment-A Review from the Melanoma Perspective and Beyond. Front. Immunol. 2018, 9, 1474. [Google Scholar] [CrossRef]
- Russo, A.; Franchina, T.; Ricciardi, G.R.R.; Battaglia, A.; Scimone, A.; Berenato, R.; Giordano, A.; Adamo, V. Baseline neutrophilia, derived neutrophil-to-lymphocyte ratio (dNLR), platelet-to-lymphocyte ratio (PLR), and outcome in non small cell lung cancer (NSCLC) treated with Nivolumab or Docetaxel. J. Cell. Physiol. 2018, 233, 6337–6343. [Google Scholar] [CrossRef]
- Bigot, F.; Castanon, E.; Baldini, C.; Hollebecque, A.; Carmona, A.; Postel-Vinay, S.; Angevin, E.; Armand, J.-P.; Ribrag, V.; Aspeslagh, S.; et al. Prospective validation of a prognostic score for patients in immunotherapy phase I trials: The Gustave Roussy Immune Score (GRIm-Score). Eur. J. Cancer 2017, 84, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, D.; Bajaj, S.; Yu, J.; Hsu, M.; Balar, A.; Pavlick, A.; Weber, J.; Osman, I.; Zhong, J. The complex relationship between body mass index and response to immune checkpoint inhibition in metastatic melanoma patients. J. Immuno Ther. Cancer 2019, 7, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Tomašev, N.; Glorot, X.; Rae, J.W.; Zielinski, M.; Askham, H.; Saraiva, A.; Mottram, A.; Meyer, C.; Ravuri, S.; Protsyuk, I.; et al. A clinically applicable approach to continuous prediction of future acute kidney injury. Nature 2019, 572, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Beam, A.L.; Kohane, I.S. Big data and machine learning in health care. JAMA 2018, 319, 1317–1318. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, G.M.; Karuri, S.W.; Zhang, H.; Zhang, L.; Khozin, S.; Kazandjian, D.; Tang, S.; Sridhara, R.; Keegan, P.; Pazdur, R. Overall response rate, progression-free survival, and overall survival with targeted and standard therapies in advanced non-small-cell lung cancer: US Food and Drug Administration trial-level and patient-level analyses. J. Clin. Oncol. 2015, 33, 1008–1014. [Google Scholar] [CrossRef]
- Alexander, B.M.; Schoenfeld, J.D.; Trippa, L. Hazards of Hazard Ratios-Deviations from Model Assumptions in Immunotherapy. N. Engl. J. Med. 2018, 378, 1158–1159. [Google Scholar] [CrossRef]
- Rahman, R.M.; Fell, G.; Ventz, S.; Arfe, A.; Vanderbeek, A.M.; Trippa, L.; Alexander, B.M. Deviation from the proportional hazards assumption in randomized phase 3 clinical trials in oncology: Prevalence, associated factors and implications. Clin. Cancer Res. 2019. [Google Scholar] [CrossRef]
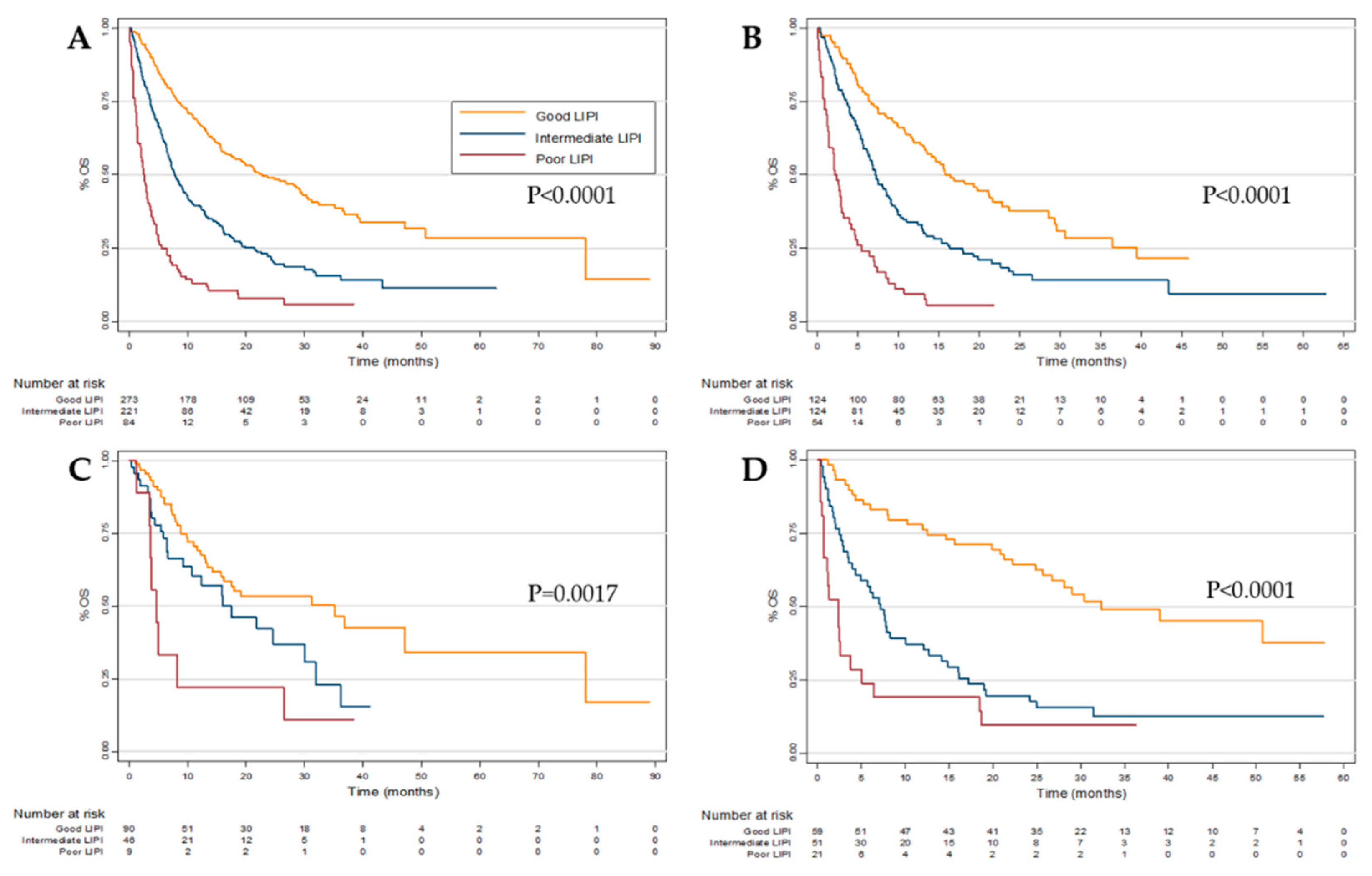
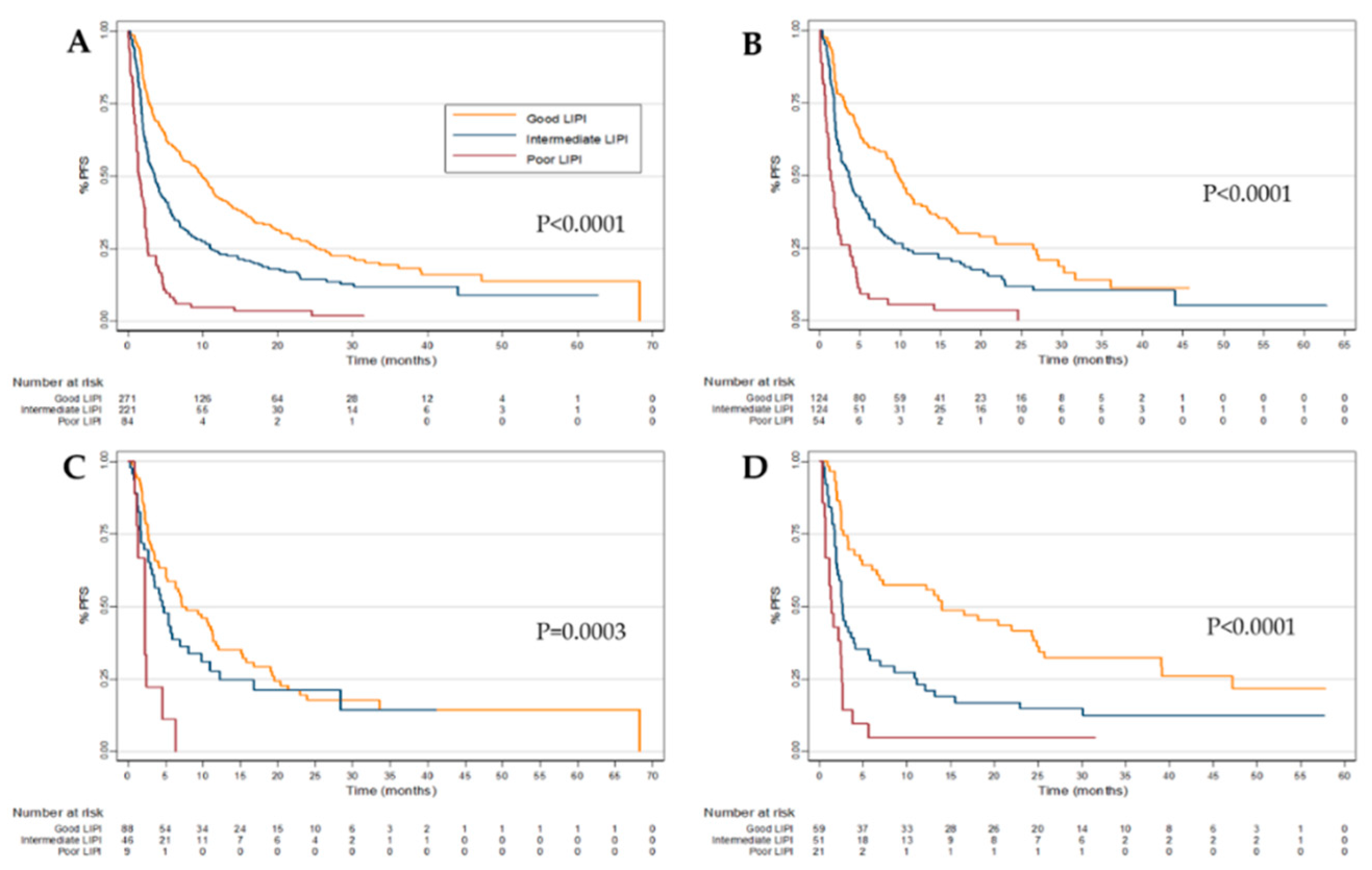
| Parameter | Pooled Cohort (N = 578) |
|---|---|
| LIPI Group—n (%) | |
| Good (0) | 273 (47.2) |
| Intermediate (1) | 221 (38.2) |
| Poor (2) | 84 (14.5) |
| Cohort—n (%) | |
| NSCLC | 302 (52.4) |
| RCC | 145 (25.1) |
| Melanoma | 131 (22.7) |
| Age | |
| Median (years) | 66.7 |
| Range (years) | 32.5–87.2 |
| <70—n (%) | 379 (65.6) |
| ≥70—n (%) | 199 (34.4) |
| Treatment Line—n (%) | |
| 1 | 147 (25.4) |
| ≥2 | 431 (74.6) |
| Range | 1–5 |
| ECOG PS—n (%) | |
| <2 | 425 (73.5) |
| ≥2 | 145 (25.1) |
| Unknown | 8 (1.4) |
| Alive at Analysis—n (%) | 180 (31.1) |
| Median follow-up (months) | 23.5 |
| Range (months) | 1.8–89.0 |
| Parameter | OS | PFS | ||
|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | |
| LIPI Group | ||||
| Good (0) | 1.0 (reference) | 1.0 (reference) | ||
| Intermediate (1) | 1.8 (1.4–2.3) | <0.001 | 1.3 (1.0–1.7) | 0.019 |
| Poor (2) | 3.6 (2.5–5.1) | <0.001 | 3.0 (2.0–4.5) | <0.001 |
| Cohort | ||||
| NSCLC | 1.0 (reference) | 1.0 (reference) | ||
| RCC | 0.6 (0.5–0.8) | 0.002 | 1.1 (0.8–1.4) | 0.65 |
| Melanoma | 0.8 (0.6–1.1) | 0.18 | 0.9 (0.7–1.3) | 0.68 |
| Age | ||||
| <70 | 1.0 (reference) | 1.0 (reference) | ||
| ≥70 | 0.9 (0.7–1.2) | 0.60 | 1.0 (0.8–1.2) | 0.83 |
| Treatment Line | ||||
| 1 | 1.0 (reference) | 1.0 (reference) | ||
| ≥2 | 1.7 (1.2–2.1) | <0.001 | 1.3 (1.0–1.8) | 0.040 |
| ECOG PS | ||||
| <2 | 1.0 (reference) | 1.0 (reference) | ||
| ≥2 | 2.3 (1.8–3.0) | <0.001 | 1.7 (1.2–2.2) | <0.001 |
| ORR | LIPI Group (n (%)) | p Value | |||
|---|---|---|---|---|---|
| Good (0) | Intermediate (1) | Poor (2) | |||
| Best Response | PR + CR | 90 (33.0%) | 48 (21.7%) | 4 (4.8%) | <0.001 |
| SD + PD | 183 (67.0%) | 173 (78.3%) | 80 (95.2%) | ||
| Parameter | ORR | |
|---|---|---|
| OR (95% CI) | p Value | |
| LIPI Group | ||
| Good (0) | 1.0 (reference) | |
| Intermediate (1) | 1.7 (1.1–2.6) | 0.018 |
| Poor (2) | 9.9 (3.4–28.5) | <0.001 |
| Cohort | ||
| NSCLC | 1.0 (reference) | |
| RCC | 1.3 (0.7–2.2) | 0.38 |
| Melanoma | 0.6 (0.4–1.0) | 0.044 |
| Age | ||
| <70 | 1.0 (reference) | |
| ≥70 | 0.8 (0.5–1.2) | 0.21 |
| Treatment Line | ||
| 1 | 1.0 (reference) | |
| ≥2 | 2.3 (1.5–3.6) | <0.001 |
| ECOG PS | ||
| <2 | 1.0 (reference) | |
| ≥2 | 2.0 (1.2–3.4) | 0.007 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meyers, D.E.; Stukalin, I.; Vallerand, I.A.; Lewinson, R.T.; Suo, A.; Dean, M.; North, S.; Pabani, A.; Cheng, T.; Heng, D.Y.C.; et al. The Lung Immune Prognostic Index Discriminates Survival Outcomes in Patients with Solid Tumors Treated with Immune Checkpoint Inhibitors. Cancers 2019, 11, 1713. https://doi.org/10.3390/cancers11111713
Meyers DE, Stukalin I, Vallerand IA, Lewinson RT, Suo A, Dean M, North S, Pabani A, Cheng T, Heng DYC, et al. The Lung Immune Prognostic Index Discriminates Survival Outcomes in Patients with Solid Tumors Treated with Immune Checkpoint Inhibitors. Cancers. 2019; 11(11):1713. https://doi.org/10.3390/cancers11111713
Chicago/Turabian StyleMeyers, Daniel E., Igor Stukalin, Isabelle A. Vallerand, Ryan T. Lewinson, Aleksi Suo, Michelle Dean, Scott North, Aliyah Pabani, Tina Cheng, Daniel Y.C. Heng, and et al. 2019. "The Lung Immune Prognostic Index Discriminates Survival Outcomes in Patients with Solid Tumors Treated with Immune Checkpoint Inhibitors" Cancers 11, no. 11: 1713. https://doi.org/10.3390/cancers11111713
APA StyleMeyers, D. E., Stukalin, I., Vallerand, I. A., Lewinson, R. T., Suo, A., Dean, M., North, S., Pabani, A., Cheng, T., Heng, D. Y. C., Bebb, D. G., & Morris, D. G. (2019). The Lung Immune Prognostic Index Discriminates Survival Outcomes in Patients with Solid Tumors Treated with Immune Checkpoint Inhibitors. Cancers, 11(11), 1713. https://doi.org/10.3390/cancers11111713





