Dual PDK1/Aurora Kinase A Inhibitors Reduce Pancreatic Cancer Cell Proliferation and Colony Formation
Abstract
:1. Introduction
2. Results
2.1. In Vitro Kinase Profiling of Dual PDK1/Aurora Kinase Inhibitors
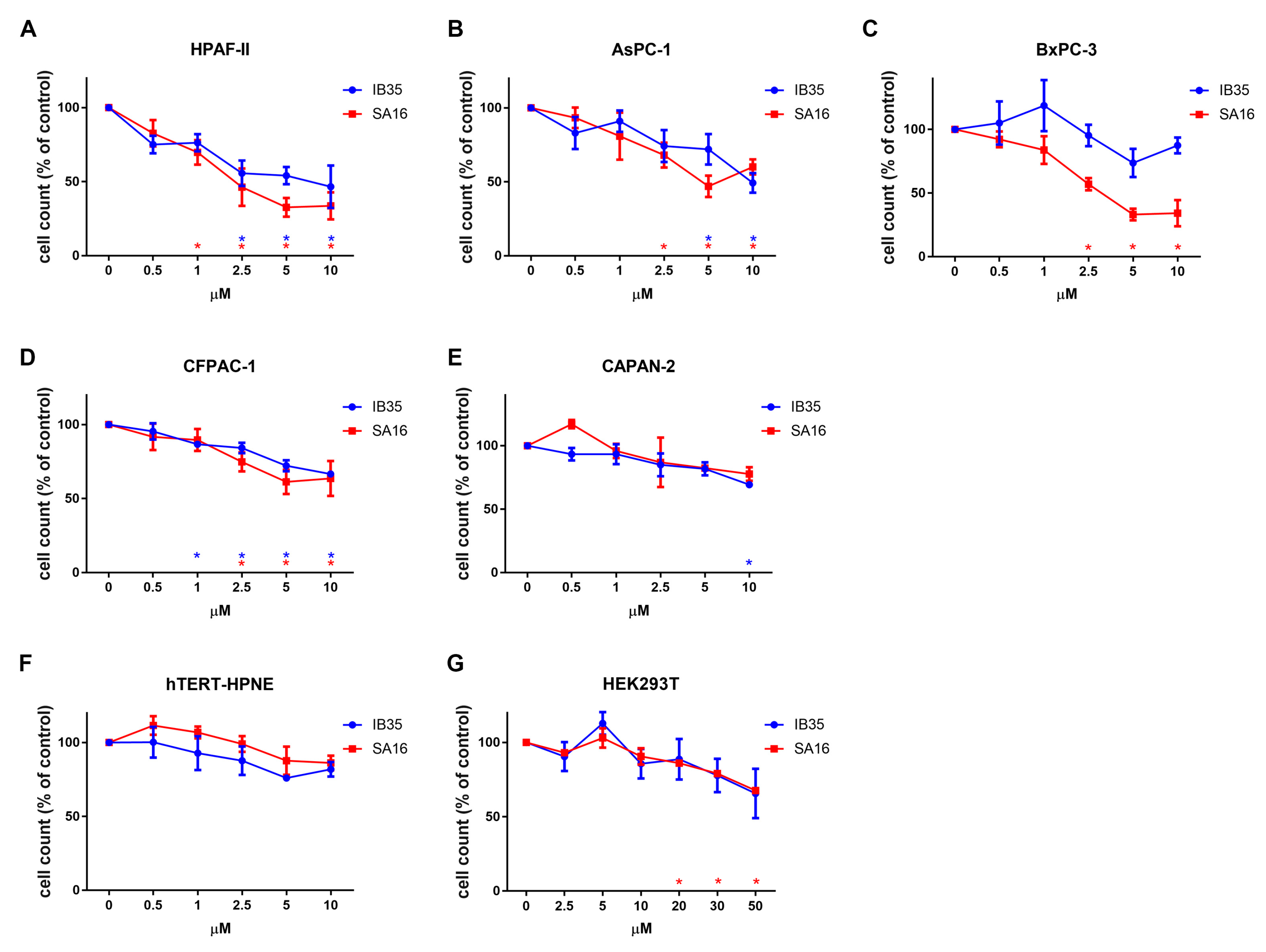
2.2. Dual Aurora Kinase/PDK1 Inhibitors Affect Pancreatic Cancer Cell Proliferation
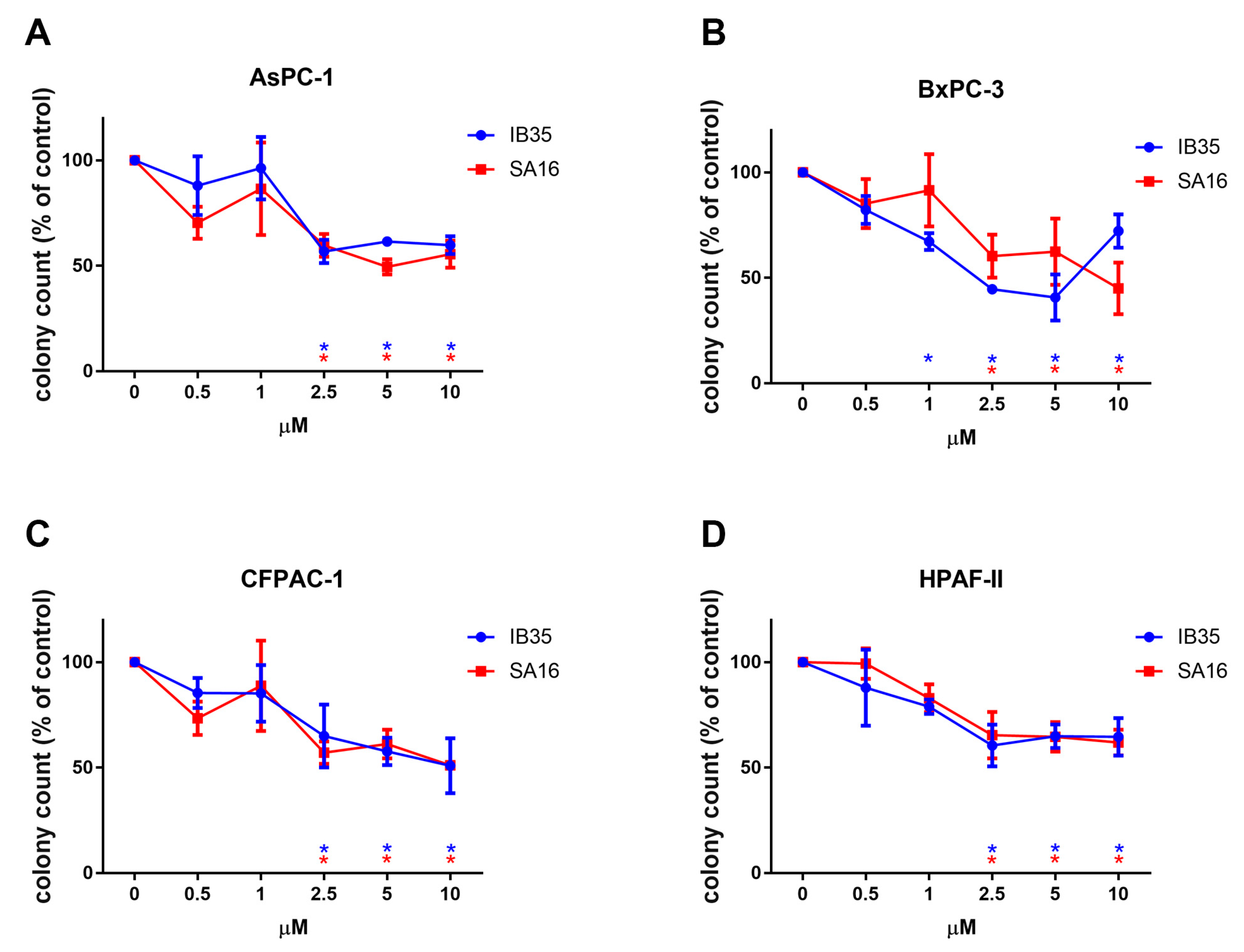
2.3. Dual Aurora Kinase/PDK1 Inhibitors Reduce Anchorage Independent Pancreatic Cancer Cell Growth
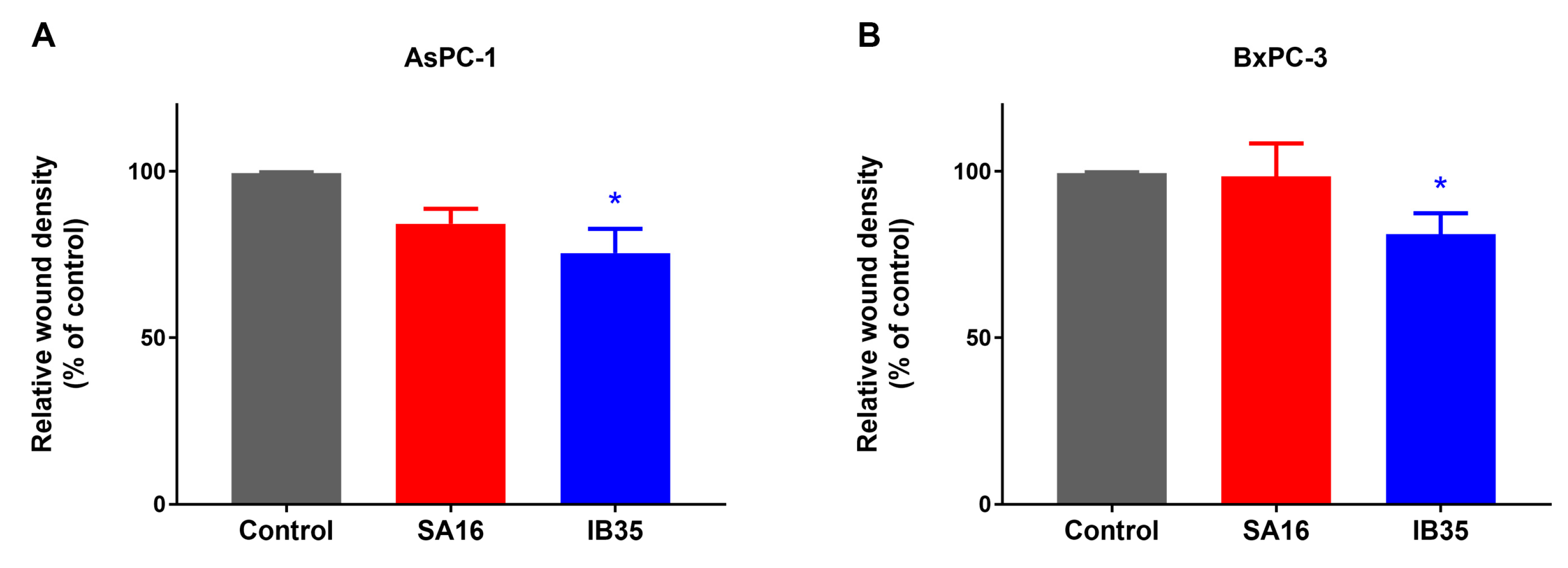
2.4. Dual Aurora Kinase/PDK1 Inhibitors Inhibit Pancreatic Cancer Cell Migration
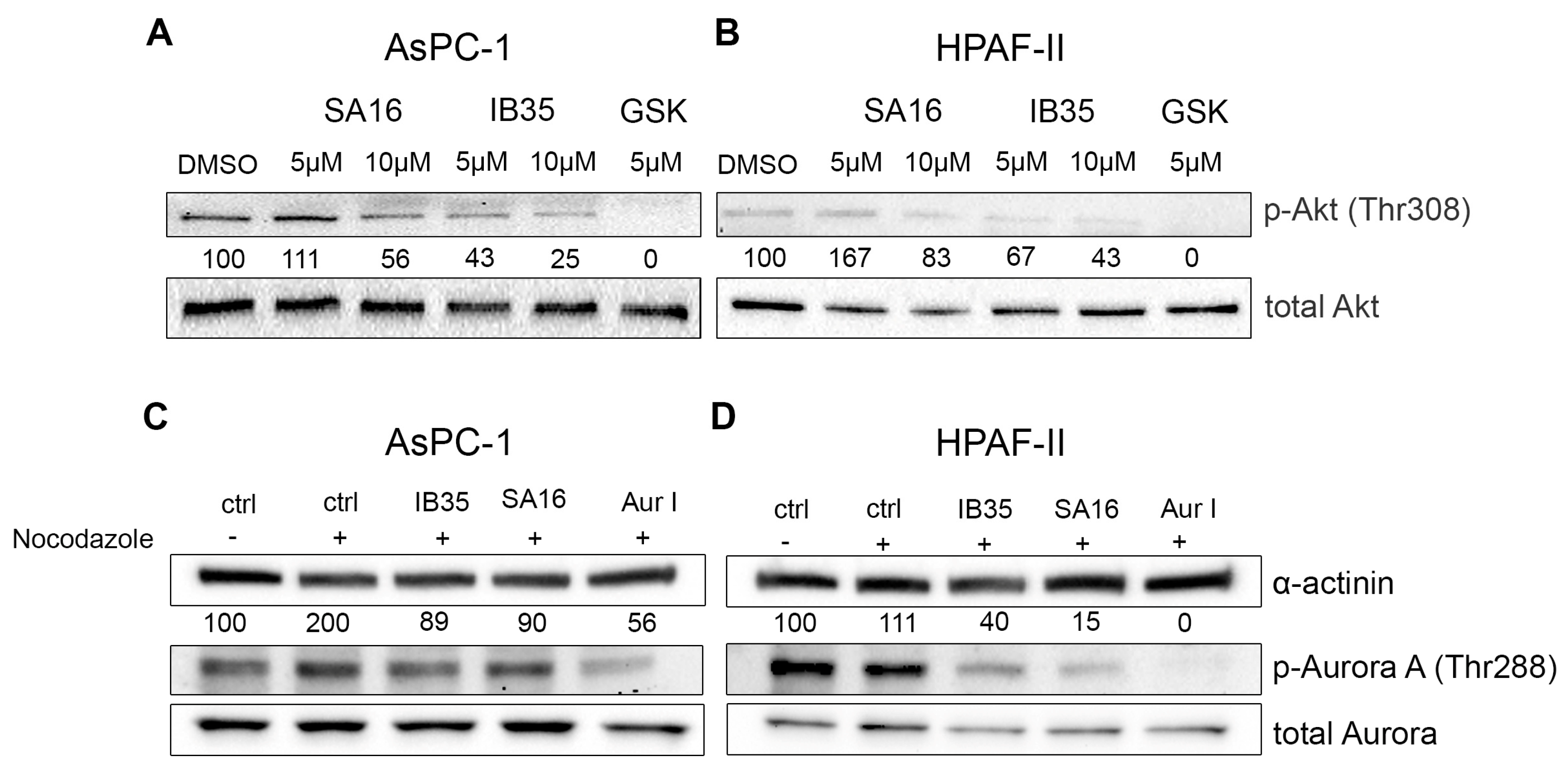
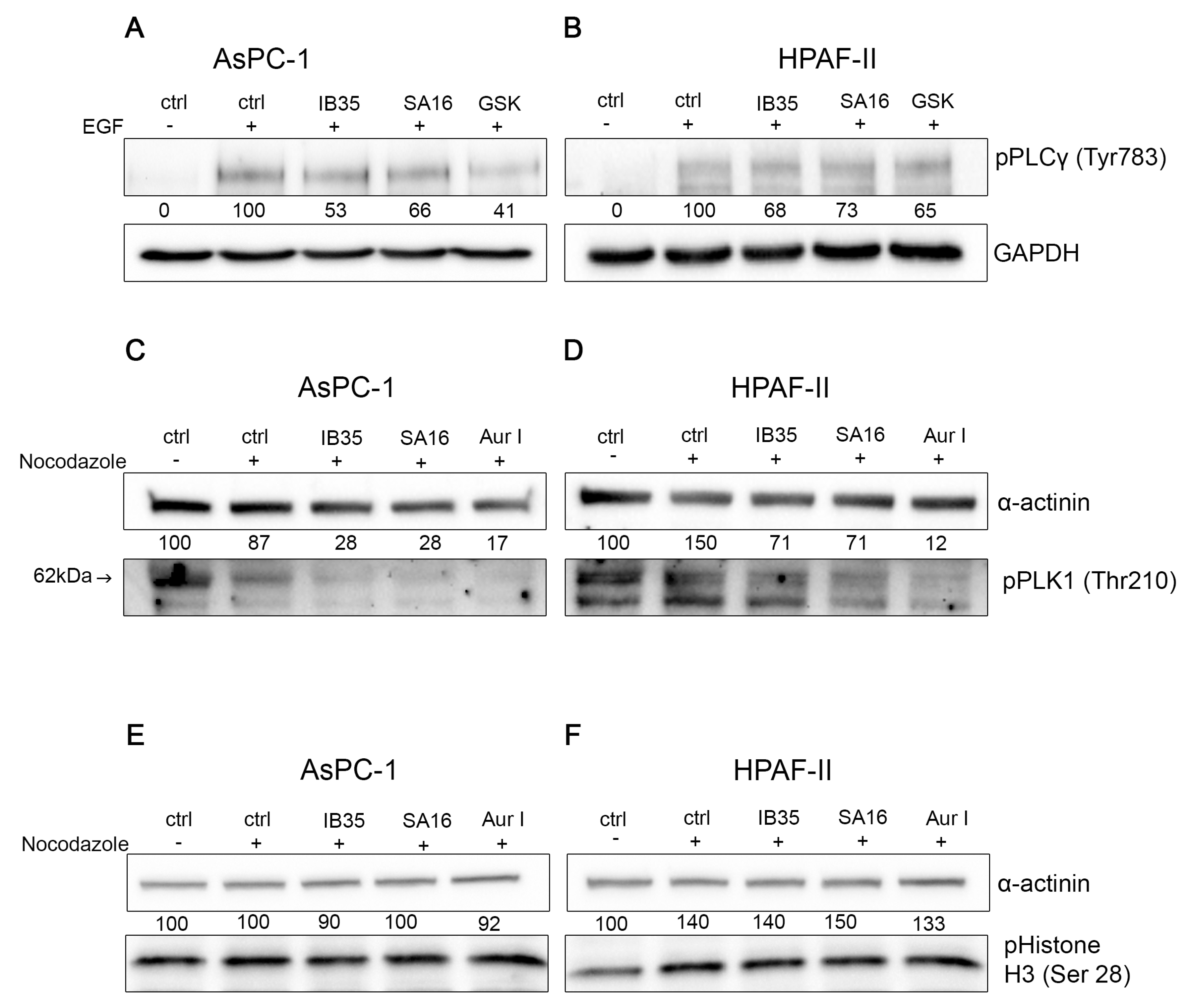
2.5. Functional Validation of Dual Aurora Kinase/PDK1 Inhibitors
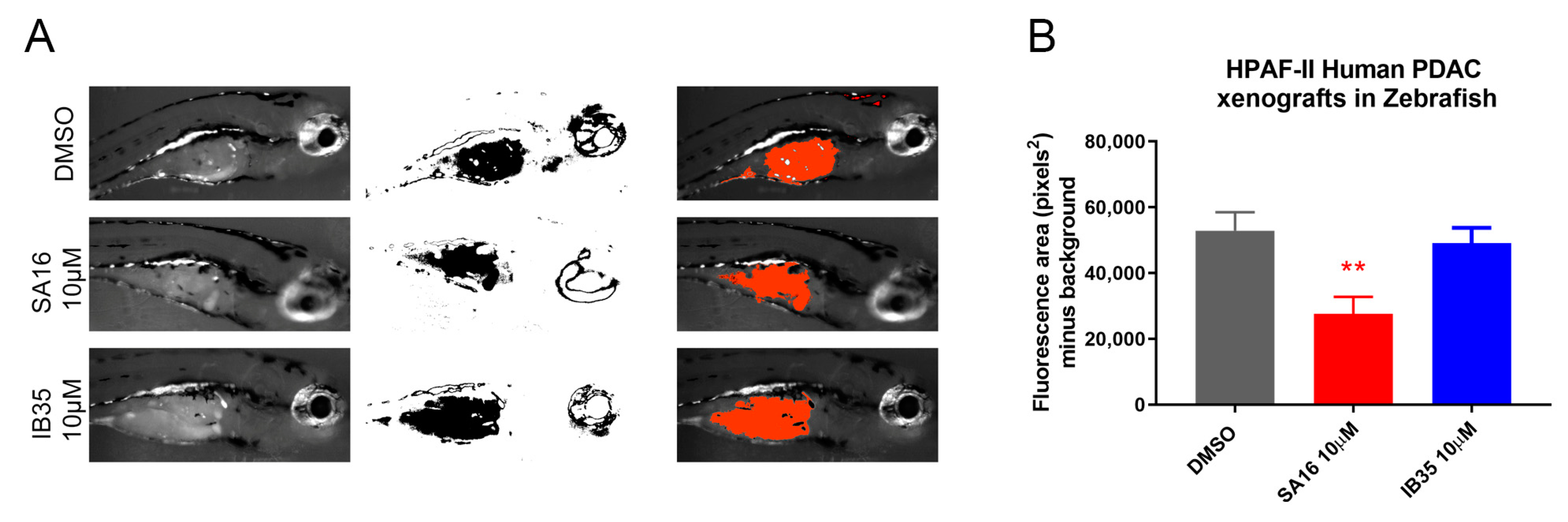
2.6. In Vivo Validation of Dual Aurora Kinase/PDK1 Inhibitors in Pancreatic Cancer Xenografts
3. Discussion
4. Materials and Methods
4.1. Inhibition of Aurora B
4.2. Cell Culture and Counting
4.3. 3D Colony Formation
4.4. Migration (Scratch Wound Assay)
4.5. Western Blotting Analysis
4.6. In Vivo Xenograft Experiment
4.7. Zebrafish Xenograft
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fruman, D.A.; Chiu, H.; Hopkins, B.D.; Bagrodia, S.; Cantley, L.C.; Abraham, R.T. The pi3k pathway in human disease. Cell 2017, 170, 605–635. [Google Scholar] [CrossRef]
- Falasca, M. Pi3k/akt signalling pathway specific inhibitors: A novel strategy to sensitize cancer cells to anti-cancer drugs. Curr. Pharm. Des. 2010, 16, 1410–1416. [Google Scholar] [CrossRef]
- Gagliardi, P.A.; Puliafito, A.; Primo, L. Pdk1: At the crossroad of cancer signaling pathways. Semin. Cancer Biol. 2018, 48, 27–35. [Google Scholar] [CrossRef]
- Mora, A.; Komander, D.; van Aalten, D.M.; Alessi, D.R. Pdk1, the master regulator of agc kinase signal transduction. Semin. Cell Dev. Biol. 2004, 15, 161–170. [Google Scholar] [CrossRef]
- Bhaskar, P.T.; Hay, N. The two torcs and akt. Dev. Cell 2007, 12, 487–502. [Google Scholar] [CrossRef]
- Toker, A.; Marmiroli, S. Signaling specificity in the akt pathway in biology and disease. Adv. Biol. Regul. 2014, 55, 28–38. [Google Scholar] [CrossRef]
- Di Blasio, L.; Gagliardi, P.A.; Puliafito, A.; Primo, L. Serine/threonine kinase 3-phosphoinositide-dependent protein kinase-1 (pdk1) as a key regulator of cell migration and cancer dissemination. Cancers 2017, 9, 25. [Google Scholar] [CrossRef]
- Manning, B.D.; Toker, A. Akt/pkb signaling: Navigating the network. Cell 2017, 169, 381–405. [Google Scholar] [CrossRef]
- Leroux, A.E.; Schulze, J.; Biondi, R.M. Agc kinases, mechanisms of regulation and innovative drug development. Semin. Cancer Biol. 2018, 48, 1–17. [Google Scholar] [CrossRef]
- Raimondi, C.; Falasca, M. Targeting pdk1 in cancer. Curr. Med. Chem. 2011, 18, 2763–2769. [Google Scholar] [CrossRef]
- Emmanouilidi, A.; Fyffe, C.A.; Ferro, R.; Edling, C.E.; Capone, E.; Sestito, S.; Rapposelli, S.; Lattanzio, R.; Iacobelli, S.; Sala, G.; et al. Preclinical Validation of 3-Phosphoinositide-Dependent Protein Kinase 1 Inhibition in Pancreatic Cancer. J. Exp. Clin. Cancer Res. 2019, 38, 191. [Google Scholar] [CrossRef] [PubMed]
- Damodaran, A.P.; Vaufrey, L.; Gavard, O.; Prigent, C. Aurora a kinase is a priority pharmaceutical target for the treatment of cancers. Trends Pharmacol. Sci. 2017, 38, 687–700. [Google Scholar] [CrossRef] [PubMed]
- D’Assoro, A.B.; Haddad, T.; Galanis, E. Aurora-a kinase as a promising therapeutic target in cancer. Front. Oncol. 2015, 5, 295. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J. The aurora kinase inhibitors in cancer research and therapy. J. Cancer Res. Clin. Oncol. 2016, 142, 1995–2012. [Google Scholar] [CrossRef]
- Bavetsias, V.; Linardopoulos, S. Aurora kinase inhibitors: Current status and outlook. Front. Oncol. 2015, 5, 278. [Google Scholar] [CrossRef]
- Bavetsias, V.; Crumpler, S.; Sun, C.; Avery, S.; Atrash, B.; Faisal, A.; Moore, A.S.; Kosmopoulou, M.; Brown, N.; Sheldrake, P.W.; et al. Optimization of imidazo [4, 5-b] pyridine-based kinase inhibitors: Identification of a dual flt3/aurora kinase inhibitor as an orally bioavailable preclinical development candidate for the treatment of acute myeloid leukemia. J. Med. Chem. 2012, 55, 8721–8734. [Google Scholar] [CrossRef]
- Ferro, R.; Falasca, M. Emerging role of the kras-pdk1 axis in pancreatic cancer. World J. Gastroenterol. 2014, 20, 10752–10757. [Google Scholar] [CrossRef]
- Bearss, D.J. Shining the light on aurora-a kinase as a drug target in pancreatic cancer. Mol. Cancer Ther. 2011, 10, 2012. [Google Scholar] [CrossRef]
- Daniele, S.; Sestito, S.; Pietrobono, D.; Giacomelli, C.; Chiellini, G.; Di Maio, D.; Marinelli, L.; Novellino, E.; Martini, C.; Rapposelli, S. Dual inhibition of pdk1 and aurora kinase a: An effective strategy to induce differentiation and apoptosis of human glioblastoma multiforme stem cells. ACS Chem. Neurosci. 2016, 8, 100–114. [Google Scholar] [CrossRef]
- Sestito, S.; Daniele, S.; Nesi, G.; Zappelli, E.; Di Maio, D.; Marinelli, L.; Digiacomo, M.; Lapucci, A.; Martini, C.; Novellino, E. Locking pdk1 in dfg-out conformation through 2-oxo-indole containing molecules: Another tools to fight glioblastoma. Eur. J. Med. Chem. 2016, 118, 47–63. [Google Scholar] [CrossRef]
- Raimondi, C.; Chikh, A.; Wheeler, A.P.; Maffucci, T.; Falasca, M. A novel regulatory mechanism links plcγ1 to pdk1. J. Cell Sci. 2012, 125, 3153–3163. [Google Scholar] [CrossRef] [PubMed]
- Raimondi, C.; Calleja, V.; Ferro, R.; Fantin, A.; Riley, A.M.; Potter, B.V.; Brennan, C.H.; Maffucci, T.; Larijani, B.; Falasca, M. A small molecule inhibitor of pdk1/plcγ1 interaction blocks breast and melanoma cancer cell invasion. Sci. Rep. 2016, 6, 26142. [Google Scholar] [CrossRef] [PubMed]
- Macůrek, L.; Lindqvist, A.; Lim, D.; Lampson, M.A.; Klompmaker, R.; Freire, R.; Clouin, C.; Taylor, S.S. Polo-like kinase-1 is activated by aurora A to promote checkpoint recovery. Nature 2008, 455, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Seki, A.; Coppinger, J.A.; Jang, C.-Y.; Yates, J.R.; Fang, G. Bora and the kinase Aurora a cooperatively activate the kinase Plk1 and control mitotic entry. Science 2008, 320, 1655–1658. [Google Scholar] [CrossRef]
- Goto, H.; Yasui, Y.; Nigg, E.A.; Inagaki, M. Aurora-B phosphorylates Histone H3 at serine28 with regard to the mitotic chromosome condensation. Genes Cells 2002, 7, 11–17. [Google Scholar] [CrossRef]
- De Groot, C.O.; Hsia, J.E.; Anzola, J.V.; Motamedi, A.; Yoon, M.; Wong, Y.L.; Jenkins, D.; Lee, H.J.; Martinez, M.B.; Davis, R.L.; et al. A Cell Biologist’s Field Guide to Aurora Kinase Inhibitors. Front. Oncol. 2015, 5, 285. [Google Scholar] [CrossRef]
- Polivka, J.; Janku, F. Molecular targets for cancer therapy in the pi3k/akt/mtor pathway. Pharmacol. Ther. 2014, 142, 164–175. [Google Scholar] [CrossRef]
- Konings, I.R.; Verweij, J.; Wiemer, E.A.; Sleijfer, S. The applicability of mtor inhibition in solid tumors. Curr. Cancer Drug Targets 2009, 9, 439–450. [Google Scholar] [CrossRef]
- LoRusso, P.M. Inhibition of the pi3k/akt/mtor pathway in solid tumors. J. Clin. Oncol. 2016, 34, 3803–3815. [Google Scholar] [CrossRef]
- Sabbah, D.A.; Brattain, M.G.; Zhong, H. Dual inhibitors of pi3k/mtor or mtor-selective inhibitors: Which way shall we go? Curr. Med. Chem. 2011, 18, 5528–5544. [Google Scholar] [CrossRef]
- Sestito, S.; Rapposelli, S. A patent update on PDK1 inhibitors (2015–present). Expert Opin. Ther. Pat. 2019, 29, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Lien, E.C.; Dibble, C.C.; Toker, A. Pi3k signaling in cancer: Beyond akt. Curr. Opin. Cell Biol. 2017, 45, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Castel, P.; Ellis, H.; Bago, R.; Toska, E.; Razavi, P.; Carmona, F.J.; Kannan, S.; Verma, C.S.; Dickler, M.; Chandarlapaty, S. Pdk1-sgk1 signaling sustains akt-independent mtorc1 activation and confers resistance to pi3kα inhibition. Cancer Cell 2016, 30, 229–242. [Google Scholar] [CrossRef] [PubMed]
- Alzahrani, A.S. PI3K/Akt/mTOR inhibitors in cancer: At the bench and bedside. Semin. Cancer Biol. 2019. [Google Scholar] [CrossRef]
- Friedberg, J.W.; Mahadevan, D.; Cebula, E.; Persky, D.; Lossos, I.; Agarwal, A.B.; Jung, J.; Burack, R.; Zhou, X.; Leonard, E.J.; et al. Phase II study of alisertib, a selective Aurora A kinase inhibitor, in relapsed and refractory aggressive B- and T-cell non-Hodgkin lymphomas. J. Clin. Oncol. 2014, 32, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Erlanson, D.A.; Arndt, J.W.; Cancilla, M.T.; Cao, K.; Elling, R.A.; English, N.; Friedman, J.; Hansen, S.K.; Hession, C.; Joseph, I.; et al. Discovery of a potent and highly selective pdk1 inhibitor via fragment-based drug discovery. Bioorganic Med. Chem. Lett. 2011, 21, 3078–3083. [Google Scholar] [CrossRef]
- Kusum, B.; Banji, D. Novel strategies for poorly water soluble drugs. Int. J. Pharm. Sci. Rev. Res. 2010, 4, 76–84. [Google Scholar]
- Westerfield, M. The Zebrafish Book: A Guide for the Laboratory Use of Zebrafish (Brachydanio Rerio); University of Oregon Press: Corvallis, OR, USA, 1995. [Google Scholar]
- Adamska, A.; Elaskalani, O.; Emmanouilidi, A.; Kim, M.; Razak, N.B.A.; Metharom, P.; Falasca, M. Molecular and cellular mechanisms of chemoresistance in pancreatic cancer. Adv. Biol. Regul. 2018, 68, 77–87. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casari, I.; Domenichini, A.; Sestito, S.; Capone, E.; Sala, G.; Rapposelli, S.; Falasca, M. Dual PDK1/Aurora Kinase A Inhibitors Reduce Pancreatic Cancer Cell Proliferation and Colony Formation. Cancers 2019, 11, 1695. https://doi.org/10.3390/cancers11111695
Casari I, Domenichini A, Sestito S, Capone E, Sala G, Rapposelli S, Falasca M. Dual PDK1/Aurora Kinase A Inhibitors Reduce Pancreatic Cancer Cell Proliferation and Colony Formation. Cancers. 2019; 11(11):1695. https://doi.org/10.3390/cancers11111695
Chicago/Turabian StyleCasari, Ilaria, Alice Domenichini, Simona Sestito, Emily Capone, Gianluca Sala, Simona Rapposelli, and Marco Falasca. 2019. "Dual PDK1/Aurora Kinase A Inhibitors Reduce Pancreatic Cancer Cell Proliferation and Colony Formation" Cancers 11, no. 11: 1695. https://doi.org/10.3390/cancers11111695
APA StyleCasari, I., Domenichini, A., Sestito, S., Capone, E., Sala, G., Rapposelli, S., & Falasca, M. (2019). Dual PDK1/Aurora Kinase A Inhibitors Reduce Pancreatic Cancer Cell Proliferation and Colony Formation. Cancers, 11(11), 1695. https://doi.org/10.3390/cancers11111695








