The Pivotal Roles of the Epithelial Membrane Protein Family in Cancer Invasiveness and Metastasis
Abstract
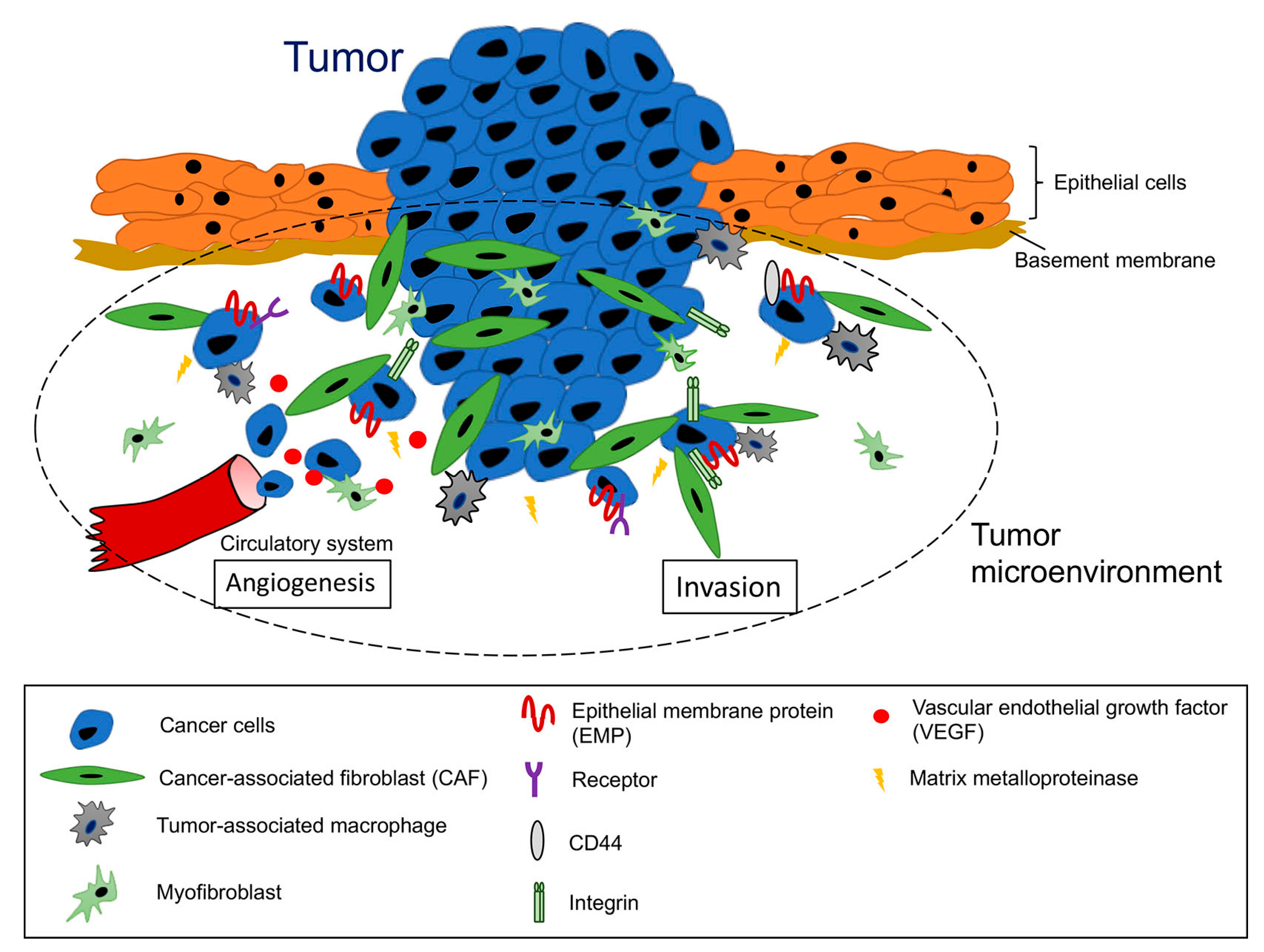
1. Introduction
2. The Structures, Expressions, and Functions of EMPs
2.1. EMP1
2.2. EMP2
2.3. EMP3
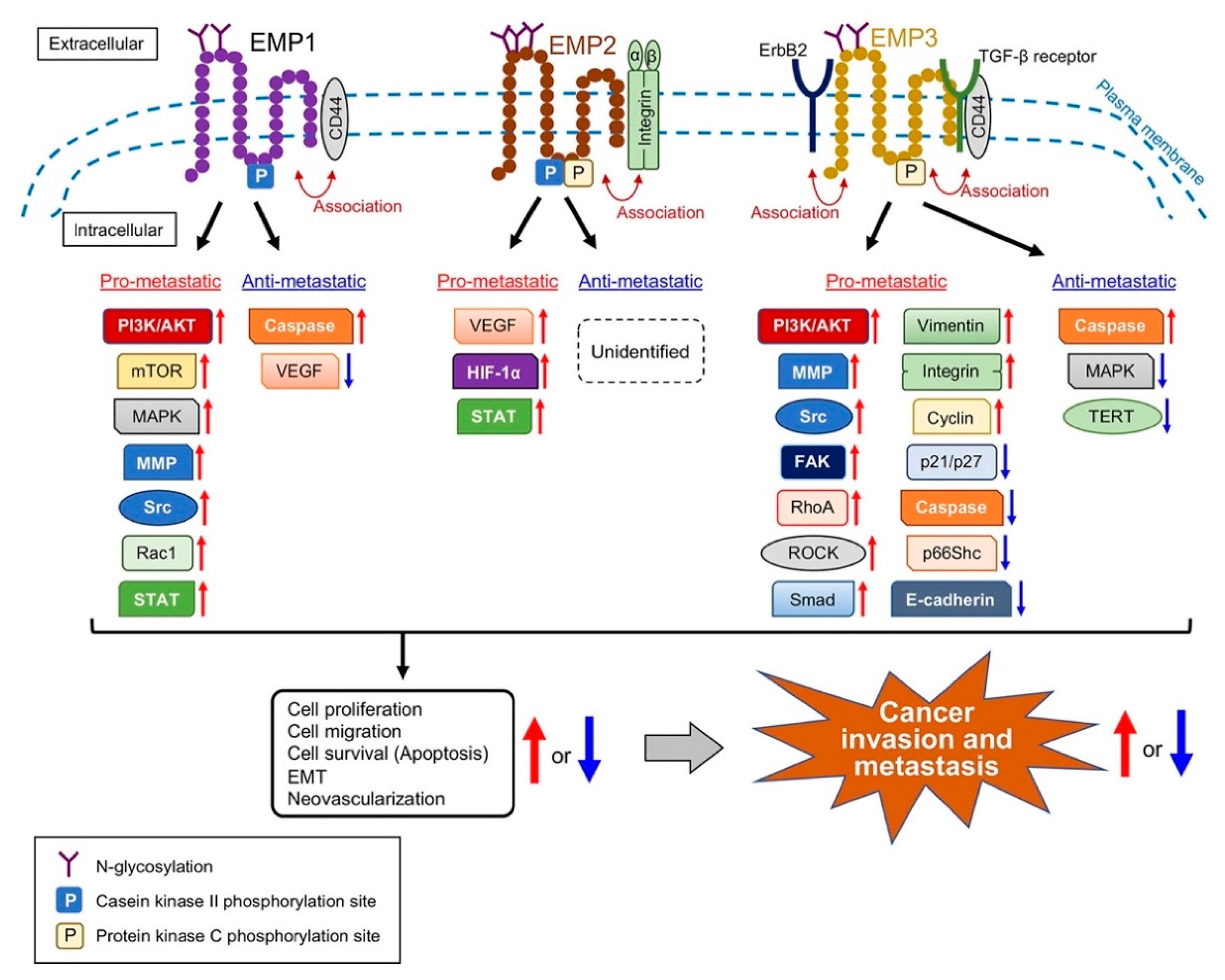
3. Involvement of EMPs in Cancer Metastasis
3.1. EMP1
3.1.1. Pro-Metastatic Roles of EMP1
3.1.2. Anti-Metastatic Roles of EMP1
3.1.3. Opposing Roles of EMP1 in Cancer Invasiveness and Metastasis in the Same Type of Cancer
3.2. EMP2
3.2.1. Pro-Metastatic Roles of EMP2
3.2.2. Anti-Metastatic Roles of EMP2
3.3. EMP3
3.3.1. Pro-Metastatic Roles of EMP3
3.3.2. Anti-Metastatic Roles of EMP3
3.4. PMP22
4. Therapeutic Implications of EMPs
4.1. Development of Monoclonal Antibodies
4.2. Perturbation of Protein−Protein Interactions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Steeg, P.S. Targeting metastasis. Nat. Rev. Cancer 2016, 16, 201–218. [Google Scholar] [CrossRef] [PubMed]
- Mehlen, P.; Puisieux, A. Metastasis: A question of life or death. Nat. Rev. Cancer 2006, 6, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Bacac, M.; Stamenkovic, I. Metastatic cancer cell. Annu. Rev. Pathol. 2008, 3, 221–247. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.J.; McGowan, P.M.; Gallagher, W.M. Cancer invasion and metastasis: Changing views. J. Pathol. 2008, 214, 283–293. [Google Scholar] [CrossRef]
- Paget, S. The distribution of secondary growths in cancer of the breast. Lancet 1889, 133, 571–573. [Google Scholar] [CrossRef]
- Andarawewa, K.L.; Motrescu, E.R.; Chenard, M.P.; Gansmuller, A.; Stoll, I.; Tomasetto, C.; Rio, M.C. Stromelysin-3 is a potent negative regulator of adipogenesis participating to cancer cell-adipocyte interaction/crosstalk at the tumor invasive front. Cancer Res. 2005, 65, 10862–10871. [Google Scholar] [CrossRef]
- Duda, D.G.; Cohen, K.S.; Kozin, S.V.; Perentes, J.Y.; Fukumura, D.; Scadden, D.T.; Jain, R.K. Evidence for incorporation of bone marrow-derived endothelial cells into perfused blood vessels in tumors. Blood 2006, 107, 2774–2776. [Google Scholar] [CrossRef]
- Levin, I. Changes in the tissue surrounding a growing tumor and the significance of the “precancerous state”. J. Exp. Med. 1912, 16, 149–154. [Google Scholar] [CrossRef]
- Lyden, D.; Hattori, K.; Dias, S.; Costa, C.; Blaikie, P.; Butros, L.; Chadburn, A.; Heissig, B.; Marks, W.; Witte, L.; et al. Impaired recruitment of bone-marrow-derived endothelial and hematopoietic precursor cells blocks tumor angiogenesis and growth. Nat. Med. 2001, 7, 1194–1201. [Google Scholar] [CrossRef]
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef]
- Begum, A.; McMillan, R.H.; Chang, Y.T.; Penchev, V.R.; Rajeshkumar, N.V.; Maitra, A.; Goggins, M.G.; Eshelman, J.R.; Wolfgang, C.L.; Rasheed, Z.A.; et al. Direct interactions with cancer-associated fibroblasts lead to enhanced pancreatic cancer stem cell function. Pancreas 2019, 48, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Coussens, L.M. Accessories to the crime: Functions of cells recruited to the tumor microenvironment. Cancer Cell 2012, 21, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Pantel, K.; Kang, Y. Tumor metastasis: Moving new biological insights into the clinic. Nat. Med. 2013, 19, 1450–1464. [Google Scholar] [CrossRef] [PubMed]
- Malanchi, I.; Santamaria-Martínez, A.; Susanto, E.; Peng, H.; Lehr, H.A.; Delaloye, J.F.; Huelsken, J. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 2011, 481, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Oskarsson, T.; Acharyya, S.; Zhang, X.H.; Vanharanta, S.; Tavazoie, S.F.; Morris, P.G.; Downey, R.J.; Manova-Todorova, K.; Brogi, E.; Massagué, J. Breast cancer cells produce tenascin C as a metastatic niche component to colonize the lungs. Nat. Med. 2011, 17, 867–874. [Google Scholar] [CrossRef]
- Lau, E.Y.; Lo, J.; Cheng, B.Y.; Ma, M.K.; Lee, J.M.; Ng, J.K.; Chai, S.; Lin, C.H.; Tsang, S.Y.; Ma, S.; et al. Cancer-associated fibroblasts regulate tumor-initiating cell plasticity in hepatocellular carcinoma through c-Met/FRA1/HEY1 signaling. Cell Rep. 2016, 15, 1175–1189. [Google Scholar] [CrossRef]
- Su, S.; Chen, J.; Yao, H.; Liu, J.; Yu, S.; Lao, L.; Wang, M.; Luo, M.; Xing, Y.; Chen, F.; et al. CD10+ GPR77+ cancer-associated fibroblasts promote cancer formation and chemoresistance by sustaining cancer stemness. Cell 2018, 172, 841–856. [Google Scholar] [CrossRef]
- Goulet, C.R.; Champagne, A.; Bernard, G.; Vandal, D.; Chabaud, S.; Pouliot, F.; Bolduc, S. Cancer-associated fibroblasts induce epithelial-mesenchymal transition of bladder cancer cells through paracrine IL-6 signalling. BMC Cancer 2019, 19, 137. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, W.; Sun, X.; Lin, Y.; Chen, W. Cancer-associated fibroblasts induce epithelial-mesenchymal transition through secreted cytokines in endometrial cancer cells. Oncol. Lett. 2018, 15, 5694–5702. [Google Scholar] [CrossRef]
- Yu, Y.; Xiao, C.H.; Tan, L.D.; Wang, Q.S.; Li, X.Q.; Feng, Y.M. Cancer-associated fibroblasts induce epithelial-mesenchymal transition of breast cancer cells through paracrine TGF-β signalling. Br. J. Cancer 2014, 110, 724–732. [Google Scholar] [CrossRef]
- Zhuang, J.; Lu, Q.; Shen, B.; Huang, X.; Shen, L.; Zheng, X.; Huang, R.; Yan, J.; Guo, H. TGFβ1 secreted by cancer-associated fibroblasts induces epithelial-mesenchymal transition of bladder cancer cells through lncRNA-ZEB2NAT. Sci. Rep. 2015, 5, 11924. [Google Scholar] [CrossRef] [PubMed]
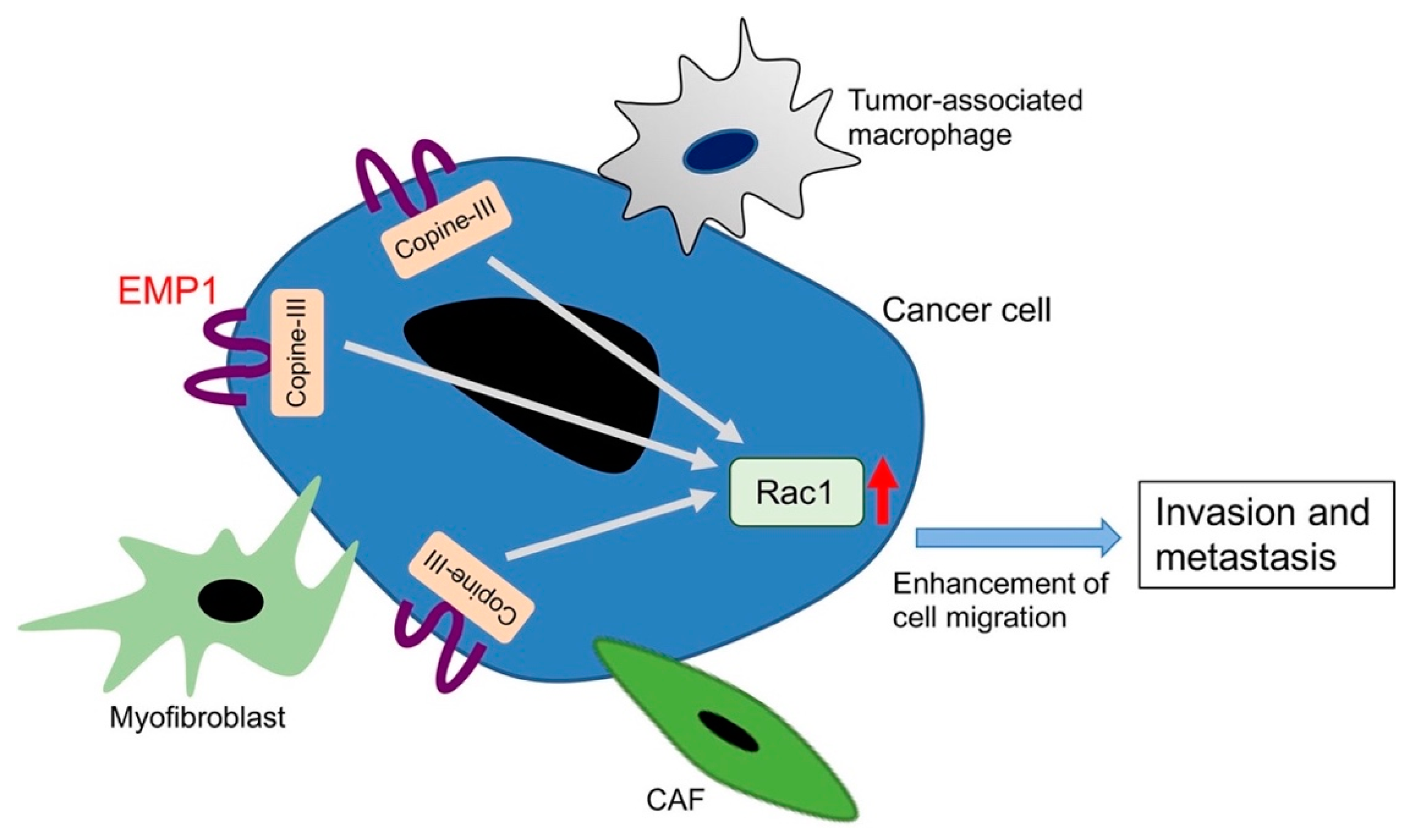
- Ahmat Amin, M.K.B.; Shimizu, A.; Zankov, D.P.; Sato, A.; Kurita, S.; Ito, M.; Maeda, T.; Yoshida, T.; Sakaue, T.; Higashiyama, S.; et al. Epithelial membrane protein 1 promotes tumor metastasis by enhancing cell migration via copine-III and Rac1. Oncogene 2018, 37, 5416–5434. [Google Scholar] [CrossRef] [PubMed]
- Wright, M.D.; Tomlinson, M.G. The ins and outs of the transmembrane 4 superfamily. Immunol. Today 1994, 15, 588–594. [Google Scholar] [CrossRef]
- Seigneuret, M.; Delaguillaumie, A.; Lagaudrière-Gesbert, C.; Conjeaud, H. Structure of the tetraspanin main extracellular domain. A partially conserved fold with a structurally variable domain insertion. J. Biol. Chem. 2001, 276, 40055–40064. [Google Scholar] [CrossRef] [PubMed]
- Stipp, C.S.; Kolesnikova, T.V.; Hemler, M.E. Functional domains in tetraspanin proteins. Trends Biochem. Sci. 2003, 28, 106–112. [Google Scholar] [CrossRef]
- Jetten, A.M.; Suter, U. The peripheral myelin protein 22 and epithelial membrane protein family. Prog. Nucleic Acid Res. Mol. Biol. 2000, 64, 97–129. [Google Scholar] [PubMed]
- Suter, U.; Snipes, G.J. Peripheral myelin protein 22: Facts and hypotheses. J. Neurosci. Res. 1995, 40, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Fabbretti, E.; Edomi, P.; Brancolini, C.; Schneider, C. Apoptotic phenotype induced by overexpression of wild-type gas3/PMP22: Its relation to the demyelinating peripheral neuropathy CMT1A. Genes Dev. 1995, 9, 1846–1856. [Google Scholar] [CrossRef]
- Zoidl, G.; Blass-Kampmann, S.; D’Urso, D.; Schmalenbach, C.; Müller, H.W. Retroviral-mediated gene transfer of the peripheral myelin protein PMP22 in Schwann cells: Modulation of cell growth. EMBO J. 1995, 14, 1122–1128. [Google Scholar] [CrossRef]
- Cai, W.; Chen, G.; Luo, Q.; Liu, J.; Guo, X.; Zhang, T.; Ma, F.; Yuan, L.; Li, B.; Cai, J. PMP22 regulates self-renewal and chemoresistance of gastric cancer cells. Mol. Cancer Ther. 2017, 16, 1187–1198. [Google Scholar] [CrossRef]
- Liu, S.; Chen, Z. The functional role of PMP22 gene in the proliferation and invasion of osteosarcoma. Med. Sci. Monit. 2015, 21, 1976–1982. [Google Scholar] [PubMed]
- Tong, D.; Heinze, G.; Pils, D.; Wolf, A.; Singer, C.F.; Concin, N.; Hofstetter, G.; Schiebel, I.; Rudas, M.; Zeillinger, R. Gene expression of PMP22 is an independent prognostic factor for disease-free and overall survival in breast cancer patients. BMC Cancer 2010, 10, 682. [Google Scholar] [CrossRef] [PubMed]
- EMP1 in The Human Protein Atlas. Available online: https://www.proteinatlas.org/ENSG00000134531-EMP1/tissue (accessed on 8 October 2019).
- Taylor, V.; Welcher, A.A.; Program, A.E.; Suter, U. Epithelial membrane protein-1, peripheral myelin protein 22, and lens membrane protein 20 define a novel gene family. J. Biol. Chem. 1995, 270, 28824–28833. [Google Scholar] [CrossRef] [PubMed]
- Wulf, P.; Suter, U. Embryonic expression of epithelial membrane protein 1 in early neurons. Brain Res. Dev. Brain Res. 1999, 116, 169–180. [Google Scholar] [CrossRef]
- Gnirke, A.U.; Weidle, U.H. Investigation of prevalence and regulation of expression of progression associated protein (PAP). Anticancer Res. 1998, 18, 4363–4369. [Google Scholar] [PubMed]
- Bangsow, T.; Baumann, E.; Bangsow, C.; Jaeger, M.H.; Pelzer, B.; Gruhn, P.; Wolf, S.; von Melchner, H.; Stanimirovic, D.B. The epithelial membrane protein 1 is a novel tight junction protein of the blood-brain barrier. J. Cereb. Blood Flow Metab. 2008, 28, 1249–1260. [Google Scholar] [CrossRef]
- EMP2 in The Human Protein Atlas. Available online: https://www.proteinatlas.org/ENSG00000213853-EMP2/tissue (accessed on 8 October 2019).
- Wadehra, M.; Sulur, G.G.; Braun, J.; Gordon, L.K.; Goodglick, L. Epithelial membrane protein-2 is expressed in discrete anatomical regions of the eye. Exp. Mol. Pathol. 2003, 74, 106–112. [Google Scholar] [CrossRef]
- Taylor, V.; Suter, U. Epithelial membrane protein-2 and epithelial membrane protein-3: Two novel members of the peripheral myelin protein 22 gene family. Gene 1996, 175, 115–120. [Google Scholar] [CrossRef]
- Wadehra, M.; Iyer, R.; Goodglick, L.; Braun, J. The tetraspan protein epithelial membrane protein-2 interacts with β1 integrins and regulates adhesion. J. Biol. Chem. 2002, 277, 41094–41100. [Google Scholar] [CrossRef]
- Wadehra, M.; Su, H.; Gordon, L.K.; Goodglick, L.; Braun, J. The tetraspan protein EMP2 increases surface expression of class I major histocompatibility complex proteins and susceptibility to CTL-mediated cell death. Clin. Immunol. 2003, 107, 129–136. [Google Scholar] [CrossRef]
- Wadehra, M.; Goodglick, L.; Braun, J. The tetraspan protein EMP2 modulates the surface expression of caveolins and glycosylphosphatidyl inositol-linked proteins. Mol. Biol. Cell 2004, 15, 2073–2083. [Google Scholar] [CrossRef] [PubMed]
- Wadehra, M.; Forbes, A.; Pushkarna, N.; Goodglick, L.; Gordon, L.K.; Williams, C.J.; Braun, J. Epithelial membrane protein-2 regulates surface expression of αvβ3 integrin in the endometrium. Dev. Biol. 2005, 287, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Wadehra, M.; Dayal, M.; Mainigi, M.; Ord, T.; Iyer, R.; Braun, J.; Williams, C.J. Knockdown of the tetraspan protein epithelial membrane protein-2 inhibits implantation in the mouse. Dev. Biol. 2006, 292, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Morales, S.A.; Mareninov, S.; Wadehra, M.; Zhang, L.; Goodglick, L.; Braun, J.; Gordon, L.K. FAK activation and the role of epithelial membrane protein 2 (EMP2) in collagen gel contraction. Investig. Ophthalmol. Vis. Sci. 2009, 50, 462–469. [Google Scholar] [CrossRef][Green Version]
- Parsons, J.T.; Martin, K.H.; Slack, J.K.; Taylor, J.M.; Weed, S.A. Focal adhesion kinase: A regulator of focal adhesion dynamics and cell movement. Oncogene 2000, 19, 5606–5613. [Google Scholar] [CrossRef]
- Schlaepfer, D.D.; Hauck, C.R.; Sieg, D.J. Signaling through focal adhesion kinase. Prog. Biophys. Mol. Biol. 1999, 71, 435–478. [Google Scholar] [CrossRef]
- EMP3 in The Human Protein Atlas. Available online: https://www.proteinatlas.org/ENSG00000142227-EMP3/tissue (accessed on 8 October 2019).
- Kusumoto, Y.; Okuyama, H.; Shibata, T.; Konno, K.; Takemoto, Y.; Maekawa, D.; Kononaga, T.; Ishii, T.; Akashi-Takamura, S.; Saitoh, S.I.; et al. Epithelial membrane protein 3 (Emp3) downregulates induction and function of cytotoxic T lymphocytes by macrophages via TNF-α production. Cell. Immunol. 2018, 324, 33–41. [Google Scholar] [CrossRef]
- Smith-Garvin, J.E.; Koretzky, G.A.; Jordan, M.S. T cell activation. Annu. Rev. Immunol. 2009, 27, 591–619. [Google Scholar] [CrossRef]
- Hotta, H.; Ross, A.H.; Huebner, K.; Isobe, M.; Wendeborn, S.; Chao, M.V.; Ricciardi, R.P.; Tsujimoto, Y.; Croce, C.M.; Koprowski, H. Molecular cloning and characterization of an antigen associated with early stages of melanoma tumor progression. Cancer Res. 1988, 48, 2955–2962. [Google Scholar]
- Oren, R.; Takahashi, S.; Doss, C.; Levy, R.; Levy, S. TAPA-1, the target of an antiproliferative antibody, defines a new family of transmembrane proteins. Mol. Cell. Biol. 1990, 10, 4007–4015. [Google Scholar] [CrossRef]
- Wright, M.D.; Henkle, K.J.; Mitchell, G.F. An immunogenic Mr 23,000 integral membrane protein of Schistosoma mansoni worms that closely resembles a human tumor-associated antigen. J. Immunol. 1990, 144, 3195–3200. [Google Scholar] [PubMed]
- Cabodi, S.; Di Stefano, P.; Leal, M.P.; Tinnirello, A.; Bisaro, B.; Morello, V.; Damiano, L.; Aramu, S.; Repetto, D.; Tornillo, G.; et al. Integrins and signal transduction. Adv. Exp. Med. Biol. 2010, 674, 43–54. [Google Scholar] [PubMed]
- Basak, S.; Dhar, R.; Das, C. Steroids modulate the expression of α4 integrin in mouse blastocysts and uterus during implantation. Biol. Reprod. 2002, 66, 1784–1789. [Google Scholar] [CrossRef] [PubMed]
- Lessey, B.A.; Castelbaum, A.J.; Wolf, L.; Greene, W.; Paulson, M.; Meyer, W.R.; Fritz, M.A. Use of integrins to date the endometrium. Fertil. Steril. 2000, 73, 779–787. [Google Scholar] [CrossRef]
- Lessey, B.A.; Gui, Y.; Apparao, K.B.; Young, S.L.; Mulholland, J. Regulated expression of heparin-binding EGF-like growth factor (HB-EGF) in the human endometrium: A potential paracrine role during implantation. Mol. Reprod. Dev. 2002, 62, 446–455. [Google Scholar] [CrossRef]
- Miao, L.; Jiang, Z.; Wang, J.; Yang, N.; Qi, Q.; Zhou, W.; Feng, Z.; Li, W.; Zhang, Q.; Huang, B.; et al. Epithelial membrane protein 1 promotes glioblastoma progression through the PI3K/AKT/mTOR signaling pathway. Oncol. Rep. 2019, 42, 605–614. [Google Scholar] [CrossRef]
- Wang, J.; Li, X.; Wu, H.; Wang, H.; Yao, L.; Deng, Z.; Zhou, Y. EMP1 regulates cell proliferation, migration, and stemness in gliomas through PI3K-AKT signaling and CD44. J. Cell. Biochem. 2019, 120, 17142–17150. [Google Scholar] [CrossRef]
- Wang, H.H.; Liao, C.C.; Chow, N.H.; Huang, L.L.; Chuang, J.I.; Wei, K.C.; Shin, J.W. Whether CD44 is an applicable marker for glioma stem cells. Am. J. Transl. Res. 2017, 9, 4785–4806. [Google Scholar]
- Ostrom, Q.T.; Gittleman, H.; Truitt, G.; Boscia, A.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS statistical report: Primary brain and other central nervous system tumors diagnosed in the United States in 2011-2015. Neuro Oncol. 2018, 20, iv1–iv86. [Google Scholar] [CrossRef]
- Onken, M.D.; Ehlers, J.P.; Worley, L.A.; Makita, J.; Yokota, Y.; Harbour, J.W. Functional gene expression analysis uncovers phenotypic switch in aggressive uveal melanomas. Cancer Res. 2006, 66, 4602–4609. [Google Scholar] [CrossRef]
- Emami, K.H.; Nguyen, C.; Ma, H.; Kim, D.H.; Jeong, K.W.; Eguchi, M.; Moon, R.T.; Teo, J.L.; Oh, S.W.; Kim, H.Y.; et al. A small molecule inhibitor of β-catenin/CREB-binding protein transcription [corrected]. Proc. Natl. Acad. Sci. USA 2004, 101, 12682–12687. [Google Scholar] [CrossRef] [PubMed]
- Kaochar, S.; Dong, J.; Torres, M.; Rajapakshe, K.; Nikolos, F.; Davis, C.M.; Ehli, E.A.; Coarfa, C.; Mitsiades, N.; Poulaki, V. ICG-001 exerts potent anticancer activity against uveal melanoma cells. Investig. Ophthalmol. Vis. Sci. 2018, 59, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Lai, S.; Wang, G.; Cao, X.; Li, Z.; Hu, J.; Wang, J. EMP-1 promotes tumorigenesis of NSCLC through PI3K/AKT pathway. J. Huazhong Univ. Sci. Technolog. Med. Sci. 2012, 32, 834–838. [Google Scholar] [CrossRef] [PubMed]
- De Marco, C.; Laudanna, C.; Rinaldo, N.; Oliveira, D.M.; Ravo, M.; Weisz, A.; Ceccarelli, M.; Caira, E.; Rizzuto, A.; Zoppoli, P.; et al. Specific gene expression signatures induced by the multiple oncogenic alterations that occur within the PTEN/PI3K/AKT pathway in lung cancer. PLoS ONE 2017, 12, e0178865. [Google Scholar] [CrossRef]
- Jain, A.; Tindell, C.A.; Laux, I.; Hunter, J.B.; Curran, J.; Galkin, A.; Afar, D.E.; Aronson, N.; Shak, S.; Natale, R.B.; et al. Epithelial membrane protein-1 is a biomarker of gefitinib resistance. Proc. Natl. Acad. Sci. USA 2005, 102, 11858–11863. [Google Scholar] [CrossRef]
- Ariës, I.M.; Jerchel, I.S.; van den Dungen, R.E.; van den Berk, L.C.; Boer, J.M.; Horstmann, M.A.; Escherich, G.; Pieters, R.; den Boer, M.L. EMP1, a novel poor prognostic factor in pediatric leukemia regulates prednisolone resistance, cell proliferation, migration and adhesion. Leukemia 2014, 28, 1828–1837. [Google Scholar] [CrossRef]
- Sun, G.G.; Lu, Y.F.; Fu, Z.Z.; Cheng, Y.J.; Hu, W.N. EMP1 inhibits nasopharyngeal cancer cell growth and metastasis through induction apoptosis and angiogenesis. Tumour Biol. 2014, 35, 3185–3193. [Google Scholar] [CrossRef]
- Wang, H.T.; Kong, J.P.; Ding, F.; Wang, X.Q.; Wang, M.R.; Liu, L.X.; Wu, M.; Liu, Z.H. Analysis of gene expression profile induced by EMP-1 in esophageal cancer cells using cDNA Microarray. World J. Gastroenterol. 2003, 9, 392–398. [Google Scholar] [CrossRef]
- Sun, G.; Zhao, G.; Lu, Y.; Wang, Y.; Yang, C. Association of EMP1 with gastric carcinoma invasion, survival and prognosis. Int. J. Oncol. 2014, 45, 1091–1098. [Google Scholar] [CrossRef]
- Sun, G.G.; Wang, Y.D.; Cui, D.W.; Cheng, Y.J.; Hu, W.N. Epithelial membrane protein 1 negatively regulates cell growth and metastasis in colorectal carcinoma. World J. Gastroenterol. 2014, 20, 4001–4010. [Google Scholar] [CrossRef]
- Demirag, G.G.; Kefeli, M.; Kemal, Y.; Yucel, I. Epithelial membrane protein 1 expression in ovarian serous tumors. Oncol. Lett. 2016, 11, 2140–2144. [Google Scholar] [CrossRef] [PubMed]
- Turashvili, G.; Bouchal, J.; Baumforth, K.; Wei, W.; Dziechciarkova, M.; Ehrmann, J.; Klein, J.; Fridman, E.; Skarda, J.; Srovnal, J.; et al. Novel markers for differentiation of lobular and ductal invasive breast carcinomas by laser microdissection and microarray analysis. BMC Cancer 2007, 7, 55. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.G.; Wang, Y.D.; Lu, Y.F.; Hu, W.N. EMP1, a member of a new family of antiproliferative genes in breast carcinoma. Tumour Biol. 2014, 35, 3347–3354. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.G.; Wang, Y.D.; Cui, D.W.; Cheng, Y.J.; Hu, W.N. EMP1 regulates caspase-9 and VEGFC expression and suppresses prostate cancer cell proliferation and invasion. Tumour Biol. 2014, 35, 3455–3462. [Google Scholar] [CrossRef]
- Newman, L.A.; Buzdar, A.U.; Singletary, S.E.; Kuerer, H.M.; Buchholz, T.; Ames, F.C.; Ross, M.I.; Hunt, K.K. A prospective trial of preoperative chemotherapy in resectable breast cancer: Predictors of breast-conservation therapy feasibility. Ann. Surg. Oncol. 2002, 9, 228–234. [Google Scholar] [CrossRef]
- Arpino, G.; Bardou, V.J.; Clark, G.M.; Elledge, R.M. Infiltrating lobular carcinoma of the breast: Tumor characteristics and clinical outcome. Breast Cancer Res. 2004, 6, R149–R156. [Google Scholar] [CrossRef]
- Mersin, H.; Yildirim, E.; Gülben, K.; Berberoğlu, U. Is invasive lobular carcinoma different from invasive ductal carcinoma. Eur. J. Surg. Oncol. 2003, 29, 390–395. [Google Scholar] [CrossRef]
- Chung, L.K.; Pelargos, P.E.; Chan, A.M.; Demos, J.V.; Lagman, C.; Sheppard, J.P.; Nguyen, T.; Chang, Y.L.; Hojat, S.A.; Prins, R.M.; et al. Tissue microarray analysis for epithelial membrane protein-2 as a novel biomarker for gliomas. Brain Tumor Pathol. 2018, 35, 1–9. [Google Scholar] [CrossRef]
- Freije, W.A.; Castro-Vargas, F.E.; Fang, Z.; Horvath, S.; Cloughesy, T.; Liau, L.M.; Mischel, P.S.; Nelson, S.F. Gene expression profiling of gliomas strongly predicts survival. Cancer Res. 2004, 64, 6503–6510. [Google Scholar] [CrossRef]
- Qin, Y.; Fu, M.; Takahashi, M.; Iwanami, A.; Kuga, D.; Rao, R.G.; Sudhakar, D.; Huang, T.; Kiyohara, M.; Torres, K.; et al. Epithelial membrane protein-2 (EMP2) activates Src protein and is a novel therapeutic target for glioblastoma. J. Biol. Chem. 2014, 289, 13974–13985. [Google Scholar] [CrossRef]
- Qin, Y.; Takahashi, M.; Sheets, K.; Soto, H.; Tsui, J.; Pelargos, P.; Antonios, J.P.; Kasahara, N.; Yang, I.; Prins, R.M.; et al. Epithelial membrane protein-2 (EMP2) promotes angiogenesis in glioblastoma multiforme. J. Neurooncol. 2017, 134, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, D.; Fan, M.; Yang, C.H.; Zbytek, B.; Finkelstein, D.; Roussel, M.F.; Pfeffer, L.M. The critical role that STAT3 plays in glioma-initiating cells: STAT3 addiction in glioma. Oncotarget 2018, 9, 22095–22112. [Google Scholar] [CrossRef] [PubMed]
- Kao, J.; Salari, K.; Bocanegra, M.; Choi, Y.L.; Girard, L.; Gandhi, J.; Kwei, K.A.; Hernandez-Boussard, T.; Wang, P.; Gazdar, A.F.; et al. Molecular profiling of breast cancer cell lines defines relevant tumor models and provides a resource for cancer gene discovery. PLoS ONE 2009, 4, e6146. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, Y. Bioinformatics analysis of gene expression data for the identification of critical genes in breast invasive carcinoma. Mol. Med. Rep. 2017, 16, 8657–8664. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.J.; Dahiya, S.; Richardson, E.; Erlander, M.; Sgroi, D.C. Gene expression profiling of the tumor microenvironment during breast cancer progression. Breast Cancer Res. 2009, 11, R7. [Google Scholar] [CrossRef]
- Watson, M.A.; Ylagan, L.R.; Trinkaus, K.M.; Gillanders, W.E.; Naughton, M.J.; Weilbaecher, K.N.; Fleming, T.P.; Aft, R.L. Isolation and molecular profiling of bone marrow micrometastases identifies TWIST1 as a marker of early tumor relapse in breast cancer patients. Clin. Cancer Res. 2007, 13, 5001–5009. [Google Scholar] [CrossRef]
- Fu, M.; Maresh, E.L.; Helguera, G.F.; Kiyohara, M.; Qin, Y.; Ashki, N.; Daniels-Wells, T.R.; Aziz, N.; Gordon, L.K.; Braun, J.; et al. Rationale and preclinical efficacy of a novel anti-EMP2 antibody for the treatment of invasive breast cancer. Mol. Cancer Ther. 2014, 13, 902–915. [Google Scholar] [CrossRef]
- Obermayr, E.; Sanchez-Cabo, F.; Tea, M.K.; Singer, C.F.; Krainer, M.; Fischer, M.B.; Sehouli, J.; Reinthaller, A.; Horvat, R.; Heinze, G.; et al. Assessment of a six gene panel for the molecular detection of circulating tumor cells in the blood of female cancer patients. BMC Cancer 2010, 10, 666. [Google Scholar] [CrossRef]
- Christgen, M.; Geffers, R.; Kreipe, H.; Lehmann, U. IPH-926 lobular breast cancer cells are triple-negative but their microarray profile uncovers a luminal subtype. Cancer Sci. 2013, 104, 1726–1730. [Google Scholar] [CrossRef]
- Chen, Q.; Yao, L.; Burner, D.; Minev, B.; Lu, L.; Wang, M.; Ma, W. Epithelial membrane protein 2: A novel biomarker for circulating tumor cell recovery in breast cancer. Clin. Transl. Oncol. 2019, 21, 433–442. [Google Scholar] [CrossRef]
- Murlidhar, V.; Reddy, R.M.; Fouladdel, S.; Zhao, L.; Ishikawa, M.K.; Grabauskiene, S.; Zhang, Z.; Lin, J.; Chang, A.C.; Carrott, P.; et al. Poor prognosis indicated by venous circulating tumor cell clusters in early-stage lung cancers. Cancer Res. 2017, 77, 5194–5206. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, A.; Lim, A.R.; Ghajar, C.M. Circulating and disseminated tumor cells: Harbingers or initiators of metastasis. Mol. Oncol. 2017, 11, 40–61. [Google Scholar] [CrossRef] [PubMed]
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W.; et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N. Engl. J. Med. 2004, 351, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Maresh, E.L.; Soslow, R.A.; Alavi, M.; Mah, V.; Zhou, Q.; Iasonos, A.; Goodglick, L.; Gordon, L.K.; Braun, J.; et al. Epithelial membrane protein-2 is a novel therapeutic target in ovarian cancer. Clin. Cancer Res. 2010, 16, 3954–3963. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Rao, R.; Sudhakar, D.; Hogue, C.P.; Rutta, Z.; Morales, S.; Gordon, L.K.; Braun, J.; Goodglick, L.; Wadehra, M. Epithelial membrane protein-2 promotes endometrial tumor formation through activation of FAK and Src. PLoS ONE 2011, 6, e19945. [Google Scholar] [CrossRef] [PubMed]
- Gordon, L.K.; Kiyohara, M.; Fu, M.; Braun, J.; Dhawan, P.; Chan, A.; Goodglick, L.; Wadehra, M. EMP2 regulates angiogenesis in endometrial cancer cells through induction of VEGF. Oncogene 2013, 32, 5369–5376. [Google Scholar] [CrossRef] [PubMed]
- Kiyohara, M.H.; Dillard, C.; Tsui, J.; Kim, S.R.; Lu, J.; Sachdev, D.; Goodglick, L.; Tong, M.; Torous, V.F.; Aryasomayajula, C.; et al. EMP2 is a novel therapeutic target for endometrial cancer stem cells. Oncogene 2017, 36, 5793–5807. [Google Scholar] [CrossRef]
- Habeeb, O.; Goodglick, L.; Soslow, R.A.; Rao, R.G.; Gordon, L.K.; Schirripa, O.; Horvath, S.; Braun, J.; Seligson, D.B.; Wadehra, M. Epithelial membrane protein-2 expression is an early predictor of endometrial cancer development. Cancer 2010, 116, 4718–4726. [Google Scholar] [CrossRef]
- Tang, M.; Liu, R.Y.; Zhou, C.; Yuan, M.Z.; Wu, D.M.; Yuan, Z.; Zhang, P.; Lang, J.Y. EMP2 re-expression inhibits growth and enhances radiosensitivity in nasopharyngeal carcinoma. Tumour Biol. 2017, 39, 1010428317695972. [Google Scholar] [CrossRef]
- Wang, M.; Li, S.; Zhang, P.; Wang, Y.; Wang, C.; Bai, D.; Jiang, X. EMP2 acts as a suppressor of melanoma and is negatively regulated by mTOR-mediated autophagy. J. Cancer 2019, 10, 3582–3592. [Google Scholar] [CrossRef]
- Wang, Y.W.; Li, W.M.; Wu, W.J.; Chai, C.Y.; Chang, T.Y.; Sun, Y.; Cheng, C.J.; Shiue, Y.L.; Su, S.J.; Cheng, H.L.; et al. Epithelial membrane protein 2 is a prognostic indictor for patients with urothelial carcinoma of the upper urinary tract. Am. J. Pathol. 2013, 183, 709–719. [Google Scholar] [CrossRef] [PubMed]
- Scrideli, C.A.; Carlotti, C.G.; Okamoto, O.K.; Andrade, V.S.; Cortez, M.A.; Motta, F.J.; Lucio-Eterovic, A.K.; Neder, L.; Rosemberg, S.; Oba-Shinjo, S.M.; et al. Gene expression profile analysis of primary glioblastomas and non-neoplastic brain tissue: Identification of potential target genes by oligonucleotide microarray and real-time quantitative PCR. J. Neurooncol. 2008, 88, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Shu, C.; Wang, Q.; Yan, X.; Wang, J. Whole-genome expression microarray combined with machine learning to identify prognostic biomarkers for high-grade glioma. J. Mol. Neurosci. 2018, 64, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Yue, H.; Xu, Q.; Xie, S. High EMP3 expression might independently predict poor overall survival in glioblastoma and its expression is related to DNA methylation. Medicine 2018, 97, e9538. [Google Scholar] [CrossRef]
- Gao, Y.F.; Zhu, T.; Mao, C.X.; Liu, Z.X.; Wang, Z.B.; Mao, X.Y.; Li, L.; Yin, J.Y.; Zhou, H.H.; Liu, Z.Q. PPIC, EMP3 and CHI3L1 are novel prognostic markers for high grade glioma. Int. J. Mol. Sci. 2016, 17, 1808. [Google Scholar] [CrossRef]
- Jun, F.; Hong, J.; Liu, Q.; Guo, Y.; Liao, Y.; Huang, J.; Wen, S.; Shen, L. Epithelial membrane protein 3 regulates TGF-β signaling activation in CD44-high glioblastoma. Oncotarget 2017, 8, 14343–14358. [Google Scholar] [CrossRef]
- Bhat, K.P.L.; Balasubramaniyan, V.; Vaillant, B.; Ezhilarasan, R.; Hummelink, K.; Hollingsworth, F.; Wani, K.; Heathcock, L.; James, J.D.; Goodman, L.D.; et al. Mesenchymal differentiation mediated by NF-κB promotes radiation resistance in glioblastoma. Cancer Cell 2013, 24, 331–346. [Google Scholar] [CrossRef]
- Guo, X.X.; Su, J.; He, X.F. A 4-gene panel predicting the survival of patients with glioblastoma. J. Cell. Biochem. 2019, 120, 16037–16043. [Google Scholar] [CrossRef]
- Ernst, A.; Hofmann, S.; Ahmadi, R.; Becker, N.; Korshunov, A.; Engel, F.; Hartmann, C.; Felsberg, J.; Sabel, M.; Peterziel, H.; et al. Genomic and expression profiling of glioblastoma stem cell-like spheroid cultures identifies novel tumor-relevant genes associated with survival. Clin. Cancer Res. 2009, 15, 6541–6550. [Google Scholar] [CrossRef]
- Zheng, Z.; Luan, X.; Zha, J.; Li, Z.; Wu, L.; Yan, Y.; Wang, H.; Hou, D.; Huang, L.; Huang, F.; et al. TNF-α inhibits the migration of oral squamous cancer cells mediated by miR-765-EMP3-p66Shc axis. Cell. Signal. 2017, 34, 102–109. [Google Scholar] [CrossRef]
- Han, M.; Xu, W. EMP3 is induced by TWIST1/2 and regulates epithelial-to-mesenchymal transition of gastric cancer cells. Tumour Biol. 2017, 39, 1010428317718404. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.H.; Hsieh, S.C.; Lee, C.H.; Yang, S.F.; Cheng, C.W.; Tang, M.J.; Lin, C.L.; Lin, C.L.; Chou, R.H. Targeting EMP3 suppresses proliferation and invasion of hepatocellular carcinoma cells through inactivation of PI3K/Akt pathway. Oncotarget 2015, 6, 34859–34874. [Google Scholar] [CrossRef] [PubMed]
- Hong, X.C.; Fen, Y.J.; Yan, G.C.; Hong, H.; Yan, C.H.; Bing, L.W.; Zhong, Y.H. Epithelial membrane protein 3 functions as an oncogene and is regulated by microRNA-765 in primary breast carcinoma. Mol. Med. Rep. 2015, 12, 6445–6450. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhou, W.; Jiang, Z.; Li, X.; Xu, F.; Liu, Y.; Wen, P.; Kong, L.; Hou, M.; Yu, J. EMP3 overexpression in primary breast carcinomas is not associated with epigenetic aberrations. J. Korean Med. Sci. 2009, 24, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Zhang, D.; Yang, X.; Song, Y. Estrogen receptor α activates MAPK signaling pathway to promote the development of endometrial cancer. J. Cell. Biochem. 2019, 120, 17593–17601. [Google Scholar] [CrossRef]
- Wang, Y.W.; Li, W.M.; Wu, W.J.; Chai, C.Y.; Liu, H.S.; Lai, M.D.; Chow, N.H. Potential significance of EMP3 in patients with upper urinary tract urothelial carcinoma: Crosstalk with ErbB2-PI3K-Akt pathway. J. Urol. 2014, 192, 242–251. [Google Scholar] [CrossRef]
- Fumoto, S.; Hiyama, K.; Tanimoto, K.; Noguchi, T.; Hihara, J.; Hiyama, E.; Noguchi, T.; Nishiyama, M. EMP3 as a tumor suppressor gene for esophageal squamous cell carcinoma. Cancer Lett. 2009, 274, 25–32. [Google Scholar] [CrossRef]
- Ma, Q.; Zhang, Y.; Liang, H.; Zhang, F.; Liu, F.; Chen, S.; Hu, Y.; Jiang, L.; Hao, Y.; Li, M.; et al. EMP3, which is regulated by miR-663a, suppresses gallbladder cancer progression via interference with the MAPK/ERK pathway. Cancer Lett. 2018, 430, 97–108. [Google Scholar] [CrossRef]
- Xue, Q.; Zhou, Y.; Wan, C.; Lv, L.; Chen, B.; Cao, X.; Ju, G.; Huang, Y.; Ni, R.; Mao, G. Epithelial membrane protein 3 is frequently shown as promoter methylation and functions as a tumor suppressor gene in non-small cell lung cancer. Exp. Mol. Pathol. 2013, 95, 313–318. [Google Scholar] [CrossRef]
- Pareek, S.; Notterpek, L.; Snipes, G.J.; Naef, R.; Sossin, W.; Laliberté, J.; Iacampo, S.; Suter, U.; Shooter, E.M.; Murphy, R.A. Neurons promote the translocation of peripheral myelin protein 22 into myelin. J. Neurosci 1997, 17, 7754–7762. [Google Scholar] [CrossRef]
- Li, J.; Kleeff, J.; Esposito, I.; Kayed, H.; Felix, K.; Giese, T.; Büchler, M.W.; Friess, H. Expression analysis of PMP22/Gas3 in premalignant and malignant pancreatic lesions. J. Histochem. Cytochem. 2005, 53, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Qu, H.; Zhu, M.; Tao, Y.; Zhao, Y. Suppression of peripheral myelin protein 22 (PMP22) expression by miR29 inhibits the progression of lung cancer. Neoplasma 2015, 62, 881–886. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Coulson, A.; Levy, A.; Gossell-Williams, M. Monoclonal antibodies in cancer therapy: Mechanisms, successes and limitations. West. Indian Med. J. 2014, 63, 650–654. [Google Scholar] [PubMed]
- Salles, G.; Barrett, M.; Foà, R.; Maurer, J.; O’Brien, S.; Valente, N.; Wenger, M.; Maloney, D.G. Rituximab in B-cell hematologic malignancies: A review of 20 years of clinical experience. Adv. Ther. 2017, 34, 2232–2273. [Google Scholar] [CrossRef]
- Strohl, W.R. Current progress in innovative engineered antibodies. Protein Cell 2018, 9, 86–120. [Google Scholar] [CrossRef]
- Shimazaki, K.; Lepin, E.J.; Wei, B.; Nagy, A.K.; Coulam, C.P.; Mareninov, S.; Fu, M.; Wu, A.M.; Marks, J.D.; Braun, J.; et al. Diabodies targeting epithelial membrane protein 2 reduce tumorigenicity of human endometrial cancer cell lines. Clin. Cancer Res. 2008, 14, 7367–7377. [Google Scholar] [CrossRef]
- Filippakopoulos, P.; Qi, J.; Picaud, S.; Shen, Y.; Smith, W.B.; Fedorov, O.; Morse, E.M.; Keates, T.; Hickman, T.T.; Felletar, I.; et al. Selective inhibition of BET bromodomains. Nature 2010, 468, 1067–1073. [Google Scholar] [CrossRef]
- Smith, B.J.; Lee, E.F.; Checco, J.W.; Evangelista, M.; Gellman, S.H.; Fairlie, W.D. Structure-guided rational design of α/β-peptide foldamers with high affinity for BCL-2 family prosurvival proteins. ChemBioChem 2013, 14, 1564–1572. [Google Scholar] [CrossRef]
- Zhao, Y.; Aguilar, A.; Bernard, D.; Wang, S. Small-molecule inhibitors of the MDM2−p53 protein−protein interaction (MDM2 inhibitors) in clinical trials for cancer treatment. J. Med. Chem. 2015, 58, 1038–1052. [Google Scholar] [CrossRef]
- Haase, H.S.; Peterson-Kaufman, K.J.; Lan Levengood, S.K.; Checco, J.W.; Murphy, W.L.; Gellman, S.H. Extending foldamer design beyond α-helix mimicry: α/β-peptide inhibitors of vascular endothelial growth factor signaling. J. Am. Chem. Soc. 2012, 134, 7652–7655. [Google Scholar] [CrossRef]
- Yoshida, A.; Shimizu, A.; Asano, H.; Kadonosono, T.; Kondoh, S.K.; Geretti, E.; Mammoto, A.; Klagsbrun, M.; Seo, M.K. VEGF-A/NRP1 stimulates GIPC1 and Syx complex formation to promote RhoA activation and proliferation in skin cancer cells. Biol. Open 2015, 4, 1063–1076. [Google Scholar] [CrossRef] [PubMed]
| Type of Cancer | In Vitro Model | In Vivo Model | Patient Samples | Remarks | |||
|---|---|---|---|---|---|---|---|
| mRNA | Protein | mRNA | Protein | mRNA | Protein | ||
| Pro-metastatic property | |||||||
| GBM | [59,60] | [59,60] | [59] | [59,60] | [59,60] | Promotion of cancer cell proliferation and invasion; Correlation with poor clinical outcome | |
| Uveal melanoma | [65] | [65] | [65] | [63] | Possible relationship with a high risk for metastatic death | ||
| NSCLC | [66] | [66,67] | [67] | [68] | [67] | Increase in cancer cell proliferation; Impairment of drug sensitivity | |
| ALL | [69] | [69] | [69] | Decrease in apoptosis; Increase in cancer cell migration, adhesion and proliferation; Impairment of drug sensitivity | |||
| Anti-metastatic property | |||||||
| Nasopharyngeal cancer | [70] | [70] | [70] | Inhibition of cancer cell migration and invasion; Increase in apoptosis; Improvement of the patients’ survival rates | |||
| Esophageal cancer | [71] | [71] | Decrease in cancer cell proliferation | ||||
| Gastric cancer | [72] | [72] | [72] | Correlation with reduced cancer invasion and metastasis and with elongation of the patients’ survival | |||
| Colorectal cancer | [73] | [73] | [73] | Decrease in cancer cell proliferation; Increase in apoptosis; Improvement of the patients’ survival rates | |||
| Ovarian cancer | [74] | Possible association with reduction of the severity of the cancer | |||||
| Paradoxical effect on metastasis | |||||||
| Breast cancer | [76] | [76] | [75,76] | Possible correlation with promotion of cancer invasion; Biomarker to distinguish the histological types of the cancer Inhibition of cancer cell migration, proliferation and invasion; Improvement of the patients’ survival rates | |||
| rostate cancer | [22,77] | [22,77] | [22] | [22] | [22,77] | Promotion of cancer cell migration, invasion and metastasis Inhibition of cancer cell migration and invasion | |
| Type of Cancer | In Vitro Model | In Vivo Model | Patient Samples | Remarks | |||
|---|---|---|---|---|---|---|---|
| mRNA | Protein | mRNA | Protein | mRNA | Protein | ||
| Pro-metastatic property | |||||||
| GBM | [84] | [83,84] | [83,84] | [82] | [81,83] | Promotion of cancer cell migration, invasion and angiogenesis | |
| Breast cancer | [92] | [90,93] | [92] | [86,87,88,89,91] | [90,93] | Promotion of cancer cell invasion and metastasis | |
| Ovarian cancer | [97] | [97] | [97] | Decrease in cancer cell death; Association with the malignant type of the cancer | |||
| Endometrial cancer | [99,100] | [98,99,100] | [98,99,100] | [101] | Promotion of angiogenesis; Correlation with cancer progression | ||
| Anti-metastatic property | |||||||
| Nasopharyngeal cancer | [102] | [102] | [102] | Decrease in cancer cell growth; Enhancement of the sensitivity of radiotherapy; Improvement of the clinical outcome | |||
| Cutaneous melanoma | [103] | [103] | [103] | Increase in apoptosis | |||
| Urothelial cancer | [104] | [104] | [104] | [104] | Decrease in cancer cell proliferation | ||
| Type of Cancer | In Vitro Model | In Vivo Model | Patient Samples | Remarks | |||
|---|---|---|---|---|---|---|---|
| mRNA | Protein | mRNA | Protein | mRNA | Protein | ||
| Pro-metastatic property | |||||||
| GBM | [109] | [109] | [109] | [105,106,107,108,109,111,112] | [112] | Increase in cancer cell proliferation; Decrease in apoptosis; Correlation with poor clinical outcome | |
| Oral squamous cancer | [113] | [113] | [113] | Increase in cancer cell migration | |||
| Gastric cancer | [114] | [114] | [114] | Induction of EMT; Correlation with poor clinical outcome | |||
| Hepatocellular cancer | [115] | [115] | [115] | Promotion of cancer cell proliferation, migration and invasion; Negative correlation with tumor differentiation | |||
| Breast cancer | [116] | [116] | [116,117] | [116] | Promotion of cancer cell proliferation, invasion and metastasis | ||
| Endometrial cancer | [118] | Possible correlation with development of the cancer | |||||
| Urothelial cancer | [119] | [119] | [119] | [119] | Increase in cancer cell proliferation and migration | ||
| Anti-metastatic property | |||||||
| Esophageal cancer | [120] | [120] | [120] | [120] | Increase in cancer cell death; Improvement of the survival rate | ||
| Gallbladder cancer | [121] | [121] | [121] | [121] | [121] | Inhibition of cancer cell proliferation, migration and invasion; Improvement of the patients’ survival rates | |
| NSCLC | [122] | [122] | Decrease in cancer cell proliferation | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmat Amin, M.K.B.; Shimizu, A.; Ogita, H. The Pivotal Roles of the Epithelial Membrane Protein Family in Cancer Invasiveness and Metastasis. Cancers 2019, 11, 1620. https://doi.org/10.3390/cancers11111620
Ahmat Amin MKB, Shimizu A, Ogita H. The Pivotal Roles of the Epithelial Membrane Protein Family in Cancer Invasiveness and Metastasis. Cancers. 2019; 11(11):1620. https://doi.org/10.3390/cancers11111620
Chicago/Turabian StyleAhmat Amin, Mohammad Khusni B., Akio Shimizu, and Hisakazu Ogita. 2019. "The Pivotal Roles of the Epithelial Membrane Protein Family in Cancer Invasiveness and Metastasis" Cancers 11, no. 11: 1620. https://doi.org/10.3390/cancers11111620
APA StyleAhmat Amin, M. K. B., Shimizu, A., & Ogita, H. (2019). The Pivotal Roles of the Epithelial Membrane Protein Family in Cancer Invasiveness and Metastasis. Cancers, 11(11), 1620. https://doi.org/10.3390/cancers11111620





