Variation in Clinical Application of Hyperthermic Intraperitoneal Chemotherapy: A Review
Abstract
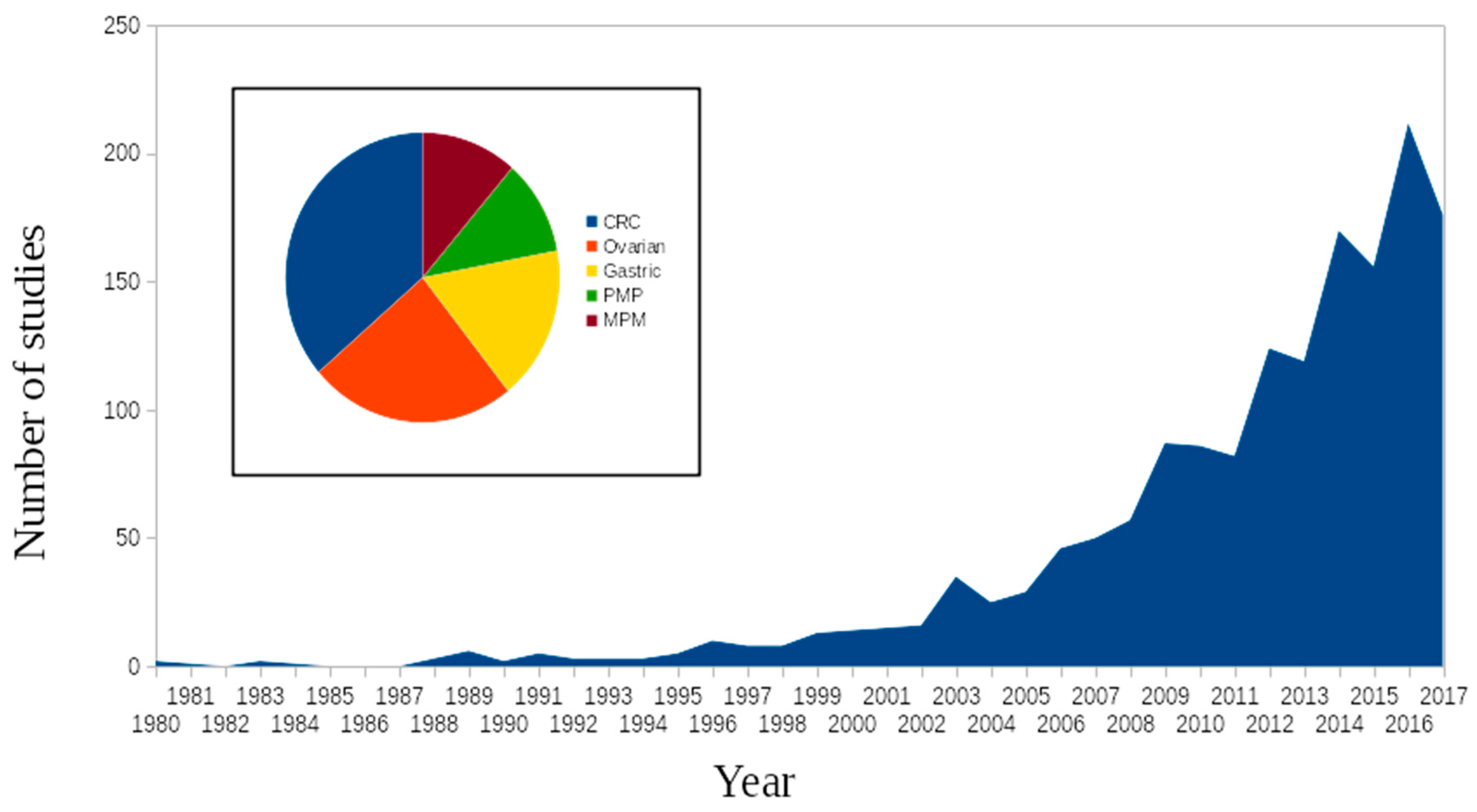
1. Introduction
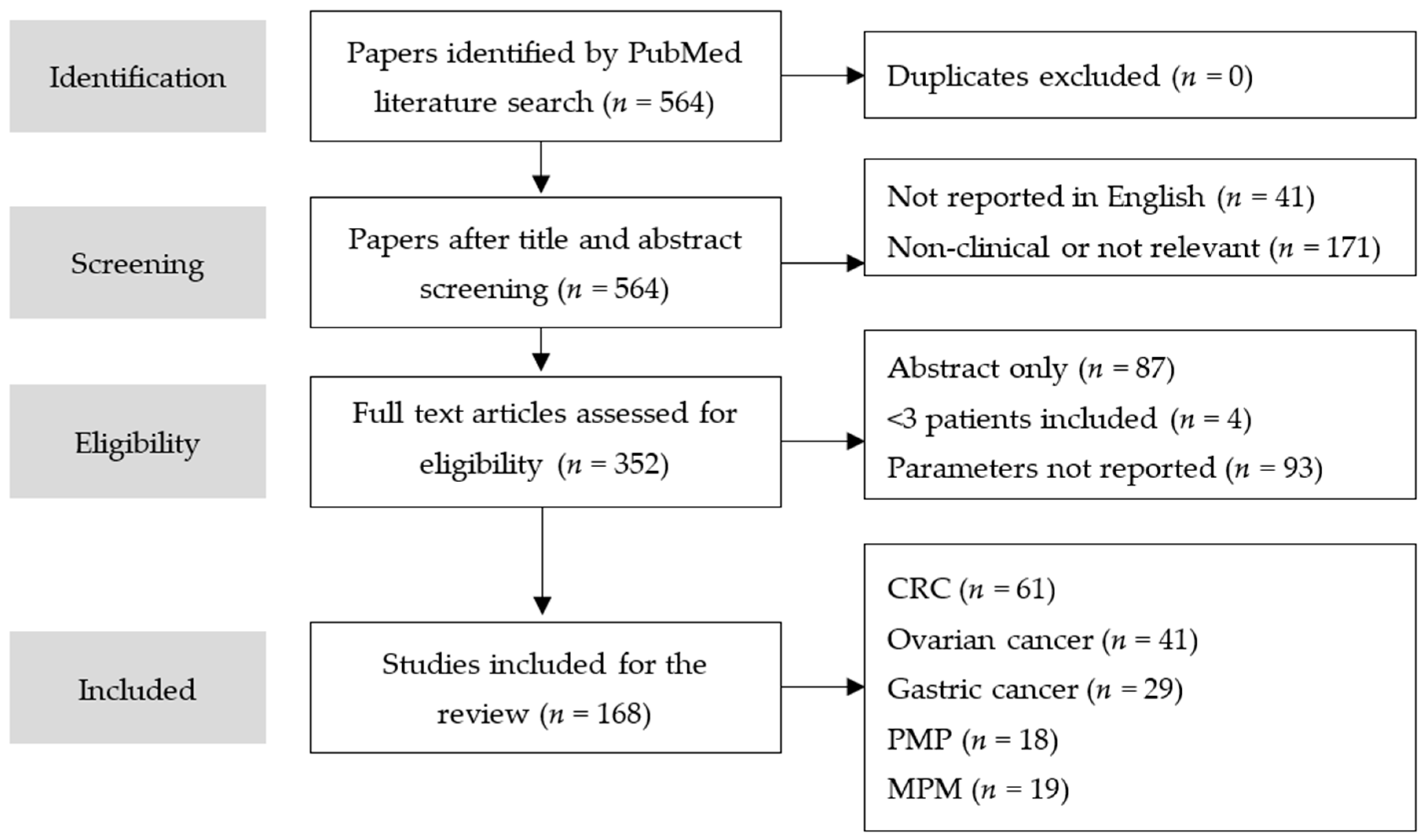
2. Methods
3. Parameters of HIPEC
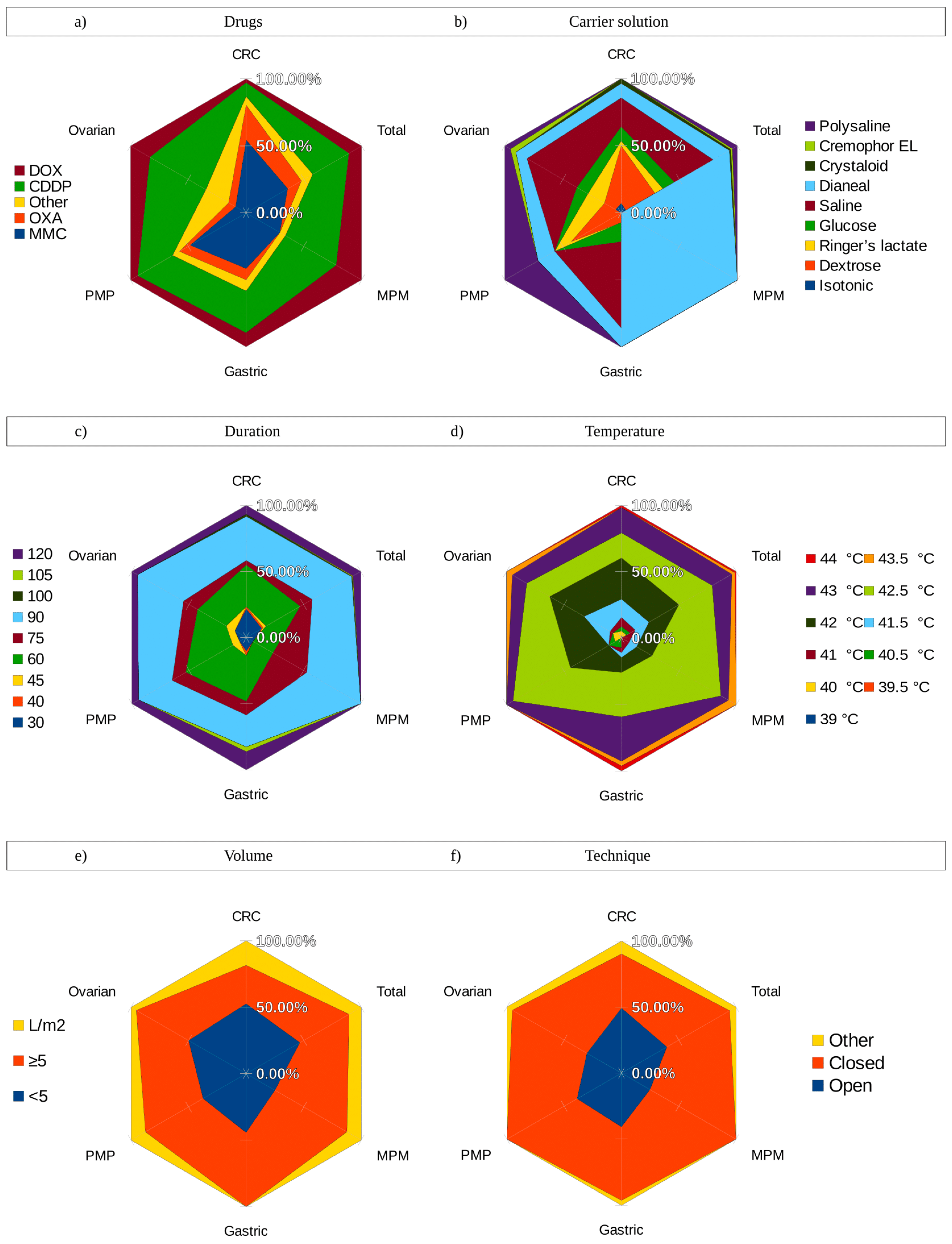
3.1. Chemotherapy
3.2. Carrier Solution
3.3. Dosage and Perfusate Volume
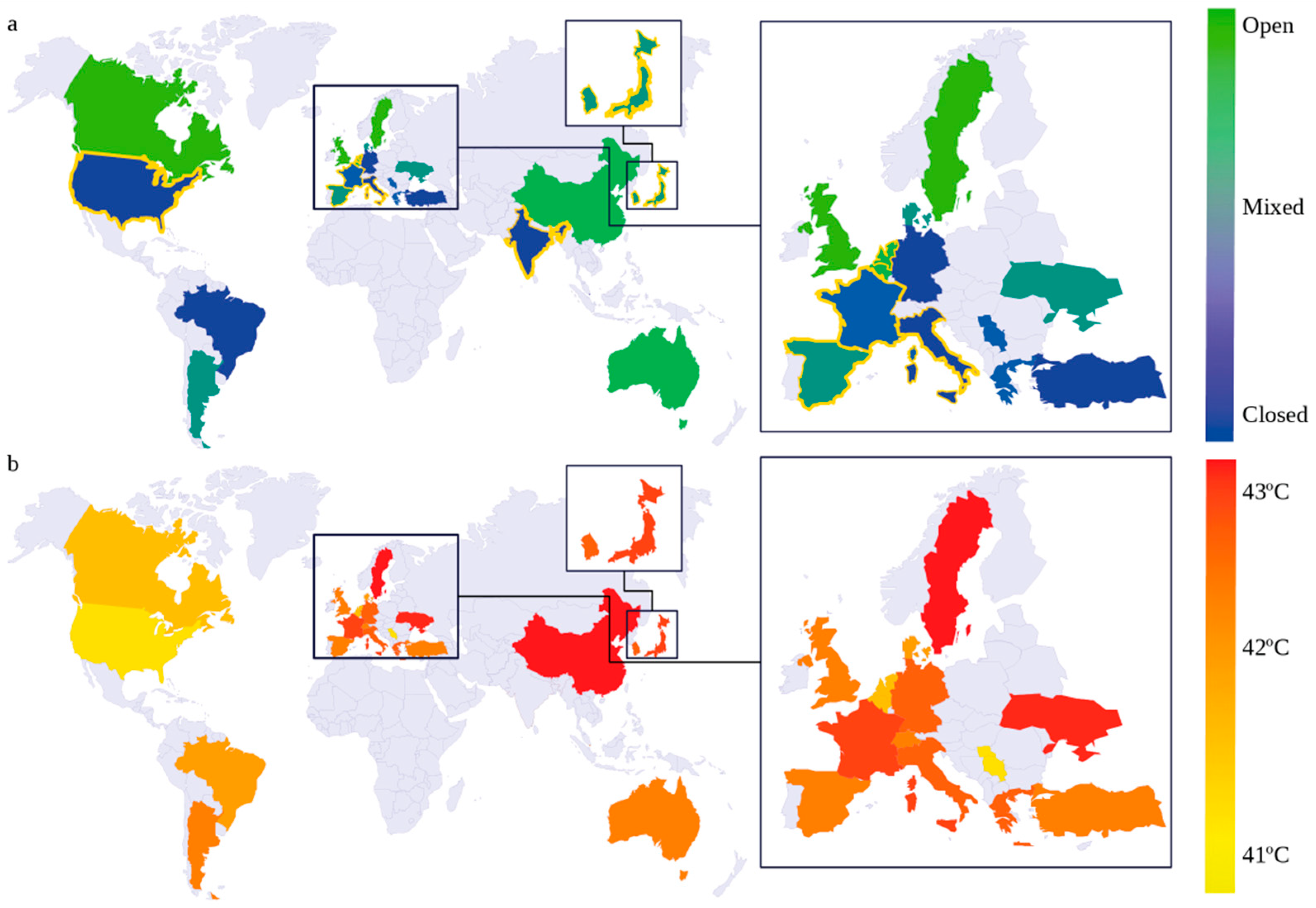
3.4. Temperature
3.5. Duration
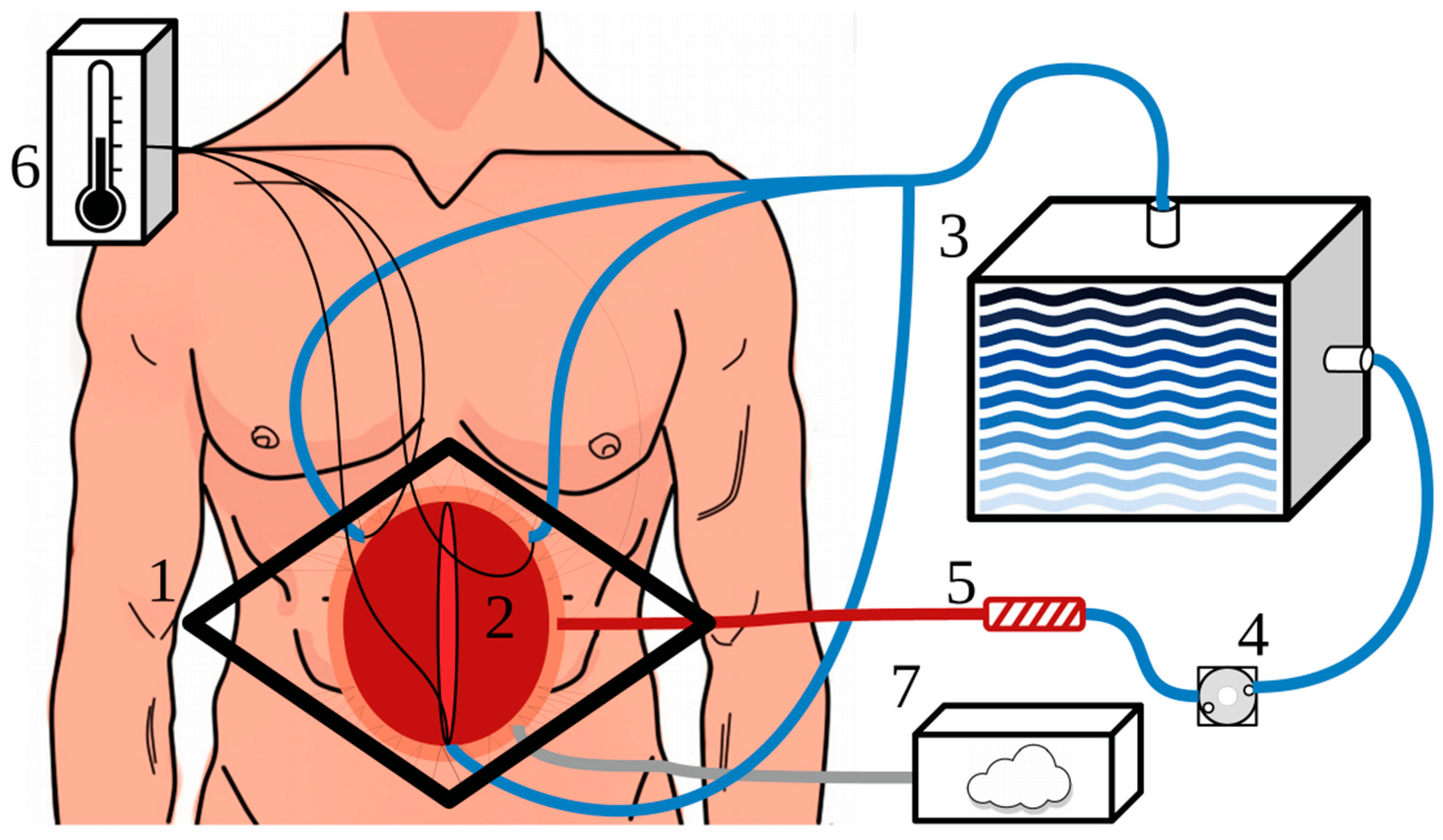
3.6. HIPEC Delivery Techniques
3.7. Patient Selection
3.8. Additional Parameters
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Koppe, M.J.; Boerman, O.C.; Oyen, W.J.; Bleichrodt, R.P. Peritoneal carcinomatosis of colorectal origin: Incidence and current treatment strategies. Ann. Surg. 2006, 243, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Mehlen, P.; Puisieux, A. Metastasis: A question of life or death. Nat. Rev. Cancer 2006, 6, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Jayne, D.G.; Fook, S.; Loi, C.; Seow-Choen, F. Peritoneal carcinomatosis from colorectal cancer. Br. J. Surg. 2002, 89, 1545–1550. [Google Scholar] [CrossRef] [PubMed]
- Sugarbaker, P.H. Colorectal cancer: Prevention and management of metastatic disease. Biomed. Res. Int. 2014, 2014, 782890. [Google Scholar] [CrossRef] [PubMed]
- Pelz, J.O.; Chua, T.C.; Esquivel, J.; Stojadinovic, A.; Doerfer, J.; Morris, D.L.; Maeder, U.; Germer, C.T.; Kerscher, A.G. Evaluation of best supportive care and systemic chemotherapy as treatment stratified according to the retrospective peritoneal surface disease severity score (PSDSS) for peritoneal carcinomatosis of colorectal origin. BMC Cancer 2010, 10, 689. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Alzahrani, N.A.; Chua, T.C.; Liauw, W.; Morris, D.L. Impacts of low peritoneal cancer index on the survival outcomes of patient with peritoneal carcinomatosis of colorectal origin. Int. J. Surg. 2015, 23, 181–185. [Google Scholar] [CrossRef]
- Tan, H.L.; Chia, C.S.; Tan, G.H.; Choo, S.P.; Tai, D.W.; Chua, C.W.; Ng, M.C.; Soo, K.C.; Teo, M.C. Gastric peritoneal carcinomatosis—A retrospective review. World J. Gastrointest. Oncol. 2017, 9, 121–128. [Google Scholar] [CrossRef]
- Thomassen, I.; van Gestel, Y.R.; van Ramshorst, B.; Luyer, M.D.; Bosscha, K.; Nienhuijs, S.W.; Lemmens, V.E.; de Hingh, I.H. Peritoneal carcinomatosis of gastric origin: A population-based study on incidence, survival and risk factors. Int. J. Cancer 2014, 134, 622–628. [Google Scholar] [CrossRef]
- Testa, U.; Petrucci, E.; Pasquini, L.; Castelli, G.; Pelosi, E. Ovarian Cancers: Genetic Abnormalities, Tumor Heterogeneity and Progression, Clonal Evolution and Cancer Stem Cells. Medicines 2018, 5, 16. [Google Scholar] [CrossRef]
- Amini, A.; Masoumi-Moghaddam, S.; Ehteda, A.; Morris, D.L. Secreted mucins in pseudomyxoma peritonei: Pathophysiological significance and potential therapeutic prospects. Orphanet J. Rare Dis. 2014, 9, 71. [Google Scholar] [CrossRef]
- Smeenk, R.M.; van Velthuysen, M.L.; Verwaal, V.J.; Zoetmulder, F.A. Appendiceal neoplasms and pseudomyxoma peritonei: A population based study. Eur. J. Surg. Oncol. 2008, 34, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Coccolini, F.; Gheza, F.; Lotti, M.; Virzi, S.; Iusco, D.; Ghermandi, C.; Melotti, R.; Baiocchi, G.; Giulini, S.M.; Ansaloni, L.; et al. Peritoneal carcinomatosis. World J. Gastroenterol. 2013, 19, 6979–6994. [Google Scholar] [CrossRef] [PubMed]
- Glehen, O.; Cotte, E.; Kusamura, S.; Deraco, M.; Baratti, D.; Passot, G.; Beaujard, A.C.; Noel, G.F. Hyperthermic intraperitoneal chemotherapy: Nomenclature and modalities of perfusion. J. Surg. Oncol. 2008, 98, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Jung, H. Interaction of Thermotolerance and Thermosensitization Induced in CHO Cells by Combined Hyperthermic Treatments at 40 and 43 °C. Radiat. Res. 1982, 91, 433–446. [Google Scholar] [CrossRef] [PubMed]
- Lutgens, L.; van der Zee, J.; Pijls-Johannesma, M.; De Haas-Kock, D.F.; Buijsen, J.; Mastrigt, G.A.; Lammering, G.; De Ruysscher, D.K.; Lambin, P. Combined use of hyperthermia and radiation therapy for treating locally advanced cervix carcinoma. Cochrane Database Syst. Rev. 2010. [Google Scholar] [CrossRef] [PubMed]
- Van der Zee, J.; Gonzalez Gonzalez, D.; van Rhoon, G.C.; van Dijk, J.D.; van Putten, W.L.; Hart, A.A. Comparison of radiotherapy alone with radiotherapy plus hyperthermia in locally advanced pelvic tumours: A prospective, randomised, multicentre trial. Dutch Deep Hyperthermia Group. Lancet 2000, 355, 1119–1125. [Google Scholar] [CrossRef]
- Roti, J.L. Cellular responses to hyperthermia (40-46 degrees C): Cell killing and molecular events. Int. J. Hyperth. 2008, 24, 3–15. [Google Scholar] [CrossRef]
- Hildebrandt, B.; Wust, P.; Ahlers, O.; Dieing, A.; Sreenivasa, G.; Kerner, T.; Felix, R.; Riess, H. The cellular and molecular basis of hyperthermia. Crit. Rev. Oncol. Hematol. 2002, 43, 33–56. [Google Scholar] [CrossRef]
- Oei, A.L.; Vriend, L.E.; Crezee, J.; Franken, N.A.; Krawczyk, P.M. Effects of hyperthermia on DNA repair pathways: One treatment to inhibit them all. Radiat. Oncol. 2015, 10, 165. [Google Scholar] [CrossRef]
- Roviello, F.; Caruso, S.; Marrelli, D.; Pedrazzani, C.; Neri, A.; De Stefano, A.; Pinto, E. Treatment of peritoneal carcinomatosis with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy: State of the art and future developments. Surg. Oncol. 2011, 20, E38–E54. [Google Scholar] [CrossRef]
- Spratt, J.S.; Adcock, R.A.; Muskovin, M.; Sherrill, W.; McKeown, J. Clinical delivery system for intraperitoneal hyperthermic chemotherapy. Cancer Res. 1980, 40, 256–260. [Google Scholar] [PubMed]
- Spratt, J.S.; Adcock, R.A.; Sherrill, W.; Travathen, S. Hyperthermic peritoneal perfusion system in canines. Cancer Res. 1980, 40, 253–255. [Google Scholar] [PubMed]
- Neuwirth, M.G.; Alexander, H.R.; Karakousis, G.C. Then and now: Cytoreductive surgery with hyperthermic intraperitoneal chemotherapy (HIPEC), a historical perspective. J. Gastrointest. Oncol. 2016, 7, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Quenet, F. Prodige 7 Trial; ASCO: Chicago, IL, USA, 2018. [Google Scholar]
- Van Driel, W.J.; Koole, S.N.; Sikorska, K.; Schagen van Leeuwen, J.H.; Schreuder, H.W.R.; Hermans, R.H.M.; de Hingh, I.; van der Velden, J.; Arts, H.J.; Massuger, L.; et al. Hyperthermic Intraperitoneal Chemotherapy in Ovarian Cancer. N. Engl. J. Med. 2018, 378, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Kusamura, S.; Dominique, E.; Baratti, D.; Younan, R.; Deraco, M. Drugs, carrier solutions and temperature in hyperthermic intraperitoneal chemotherapy. J. Surg. Oncol. 2008, 98, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Goodman, M.D.; McPartland, S.; Detelich, D.; Saif, M.W. Chemotherapy for intraperitoneal use: A review of hyperthermic intraperitoneal chemotherapy and early post-operative intraperitoneal chemotherapy. J. Gastrointest. Oncol. 2016, 7, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Jacquet, P.; Averbach, A.; Stephens, A.D.; Stuart, O.A.; Chang, D.; Sugarbaker, P.H. Heated intraoperative intraperitoneal mitomycin C and early postoperative intraperitoneal 5-fluorouracil: Pharmacokinetic studies. Oncology 1998, 55, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Urano, M.; Ling, C.C. Thermal enhancement of melphalan and oxaliplatin cytotoxicity in vitro. Int. J. Hyperth. 2002, 18, 307–315. [Google Scholar] [CrossRef]
- Pestieau, S.R.; Schnake, K.J.; Stuart, O.A.; Sugarbaker, P.H. Impact of carrier solutions on pharmacokinetics of intraperitoneal chemotherapy. Cancer Chemother. Pharmacol. 2001, 47, 269–276. [Google Scholar]
- Mehta, A.M.; Van den Hoven, J.M.; Rosing, H.; Hillebrand, M.J.X.; Nuijen, B.; Huitema, A.D.R.; Beijnen, J.H.; Verwaal, V.J. Stability of oxaliplatin in chloride-containing carrier solutions used in hyperthermic intraperitoneal chemotherapy. Int. J. Pharm. 2015, 479, 23–27. [Google Scholar] [CrossRef]
- Stewart, C.L.; Gleisner, A.; Halpern, A.; Ibrahim-Zada, I.; Luna, R.A.; Pearlman, N.; Gajdos, C.; Edil, B.; McCarter, M. Implications of Hyperthermic Intraperitoneal Chemotherapy Perfusion-Related Hyperglycemia. Ann. Surg. Oncol. 2018, 25, 655–659. [Google Scholar] [CrossRef] [PubMed]
- Valle, S.J.; Alzahrani, N.A.; Liauw, W.; Sugarbaker, P.H.; Bhatt, A.; Morris, D.L. Hyperthermic Intraperitoneal Chemotherapy (HIPEC) Methodology, Drugs and Bidirectional Chemotherapy. Indian J. Surg. Oncol. 2016, 7, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Passot, G.; Vaudoyer, D.; Villeneuve, L.; Kepenekian, V.; Beaujard, A.C.; Bakrin, N.; Cotte, E.; Gilly, F.N.; Glehen, O. What made hyperthermic intraperitoneal chemotherapy an effective curative treatment for peritoneal surface malignancy: A 25-year experience with 1,125 procedures. J. Surg. Oncol. 2016, 113, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Voron, T.; Eveno, C.; Jouvin, I.; Beaugerie, A.; Lo Dico, R.; Dagois, S.; Soyer, P.; Pocard, M. Cytoreductive surgery with a hyperthermic intraperitoneal chemotherapy program: Safe after 40 cases, but only controlled after 140 cases. Eur. J. Surg. Oncol. 2015, 41, 1671–1677. [Google Scholar] [CrossRef] [PubMed]
- Esquivel, J. Technology of hyperthermic intraperitoneal chemotherapy in the United States, Europe, China, Japan, and Korea. Cancer J. 2009, 15, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Van Eden, W.J.; Kok, N.F.M.; Woensdregt, K.; Huitema, A.D.R.; Boot, H.; Aalbers, A.G.J. Safety of intraperitoneal Mitomycin C versus intraperitoneal oxaliplatin in patients with peritoneal carcinomatosis of colorectal cancer undergoing cytoreductive surgery and HIPEC. Eur. J. Surg. Oncol. 2018, 44, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Verwaal, V.J.; Bruin, S.; Boot, H.; van Slooten, G.; van Tinteren, H. 8-year follow-up of randomized trial: Cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy in patients with peritoneal carcinomatosis of colorectal cancer. Ann. Surg. Oncol. 2008, 15, 2426–2432. [Google Scholar] [CrossRef] [PubMed]
- Verwaal, V.J.; van Ruth, S.; Witkamp, A.; Boot, H.; van Slooten, G.; Zoetmulder, F.A. Long-term survival of peritoneal carcinomatosis of colorectal origin. Ann. Surg. Oncol. 2005, 12, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Gabano, E.; Colangelo, D.; Ghezzi, A.R.; Osella, D. The influence of temperature on antiproliferative effects, cellular uptake and DNA platination of the clinically employed Pt(II)-drugs. J. Inorg. Biochem. 2008, 102, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Elias, D.; Lefevre, J.H.; Chevalier, J.; Brouquet, A.; Marchal, F.; Classe, J.M.; Ferron, G.; Guilloit, J.M.; Meeus, P.; Goere, D.; et al. Complete cytoreductive surgery plus intraperitoneal chemohyperthermia with oxaliplatin for peritoneal carcinomatosis of colorectal origin. J. Clin. Oncol. 2009, 27, 681–685. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Maeta, M.; Koga, S. Influence of local hyperthermia on the healing of small intestinal anastomoses in the rat. Br. J. Surg. 1991, 78, 57–59. [Google Scholar] [CrossRef] [PubMed]
- Murata, S.; Yamamoto, H.; Shimizu, T.; Naitoh, H.; Yamaguchi, T.; Kaida, S.; Takebayashi, K.; Miyake, T.; Tani, T.; Tani, M. 5-fluorouracil combined with cisplatin and mitomycin C as an optimized regimen for hyperthermic intraperitoneal chemotherapy in gastric cancer. J. Surg. Oncol. 2018, 117, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Deballon, P.; Facy, O.; Jambet, S.; Magnin, G.; Cotte, E.; Beltramo, J.L.; Chauffert, B.; Rat, P. Which method to deliver hyperthermic intraperitoneal chemotherapy with oxaliplatin? An experimental comparison of open and closed techniques. Ann. Surg. Oncol. 2010, 17, 1957–1963. [Google Scholar] [CrossRef] [PubMed]
- Elias, D.; Antoun, S.; Goharin, A.; Otmany, A.E.; Puizillout, J.M.; Lasser, P. Research on the best chemohyperthermia technique of treatment of peritoneal carcinomatosis after complete resection. Int. J. Surg. Investig. 2000, 1, 431–439. [Google Scholar] [PubMed]
- Sugarbaker, P.H. An instrument to provide containment of intraoperative intraperitoneal chemotherapy with optimized distribution. J. Surg. Oncol. 2005, 92, 142–146. [Google Scholar] [CrossRef]
- Sugarbaker, P.H.; Averbach, A.M.; Jacquet, P.; Stephens, A.D.; Stuart, O.A. A simplified approach to hyperthermic intraoperative intraperitoneal chemotherapy (HIIC) using a self retaining retractor. Cancer Treat. Res. 1996, 82, 415–421. [Google Scholar]
- Gonzalez-Bayon, L.; Gonzalez-Moreno, S.; Ortega-Perez, G. Safety considerations for operating room personnel during hyperthermic intraoperative intraperitoneal chemotherapy perfusion. Eur. J. Surg. Oncol. 2006, 32, 619–624. [Google Scholar] [CrossRef]
- Fujimura, T.; Yonemura, Y.; Fushida, S.; Urade, M.; Takegawa, S.; Kamata, T.; Sugiyama, K.; Hasegawa, H.; Katayama, K.; Miwa, K.; et al. Continuous hyperthermic peritoneal perfusion for the treatment of peritoneal dissemination in gastric cancers and subsequent second-look operation. Cancer 1990, 65, 65–71. [Google Scholar] [CrossRef]
- Rat, P.; Benoit, L.; Cheynel, N.; Osmak, L.; Favoulet, P.; Peschaud, F.; Chauffert, B.; Favre, J.P. Intraperitoneal chemo-hyperthermia with “overflow” open abdomen. Ann. Chir. 2001, 126, 669–671. [Google Scholar] [CrossRef]
- Esquivel, J.; Averbach, A. Laparoscopic Cytoreductive Surgery and HIPEC in Patients with Limited Pseudomyxoma Peritonei of Appendiceal Origin. Gastroenterol. Res. Pract. 2012, 2012, 981245. [Google Scholar] [CrossRef]
- de Mestier, L.; Volet, J.; Scaglia, E.; Msika, S.; Kianmanesh, R.; Bouche, O. Is palliative laparoscopic hyperthermic intraperitoneal chemotherapy effective in patients with malignant hemorrhagic ascites? Case Rep. Gastroenterol. 2012, 6, 166–170. [Google Scholar] [CrossRef]
- Klaver, C.E.; Musters, G.D.; Bemelman, W.A.; Punt, C.J.; Verwaal, V.J.; Dijkgraaf, M.G.; Aalbers, A.G.; van der Bilt, J.D.; Boerma, D.; Bremers, A.J.; et al. Adjuvant hyperthermic intraperitoneal chemotherapy (HIPEC) in patients with colon cancer at high risk of peritoneal carcinomatosis; the COLOPEC randomized multicentre trial. BMC Cancer 2015, 15, 428. [Google Scholar] [CrossRef] [PubMed]
- Facy, O.; Al Samman, S.; Magnin, G.; Ghiringhelli, F.; Ladoire, S.; Chauffert, B.; Rat, P.; Ortega-Deballon, P. High pressure enhances the effect of hyperthermia in intraperitoneal chemotherapy with oxaliplatin: An experimental study. Ann. Surg. 2012, 256, 1084–1088. [Google Scholar] [CrossRef] [PubMed]
- Halkia, E.; Tsochrinis, A.; Vassiliadou, D.T.; Pavlakou, A.; Vaxevanidou, A.; Datsis, A.; Efstathiou, E.; Spiliotis, J. Peritoneal carcinomatosis: Intraoperative parameters in open (coliseum) versus closed abdomen HIPEC. Int. J. Surg. Oncol. 2015, 2015, 610597. [Google Scholar] [CrossRef]
- Cavaliere, F.; De Simone, M.; Virzi, S.; Deraco, M.; Rossi, C.R.; Garofalo, A.; Di Filippo, F.; Giannarelli, D.; Vaira, M.; Valle, M.; et al. Prognostic factors and oncologic outcome in 146 patients with colorectal peritoneal carcinomatosis treated with cytoreductive surgery combined with hyperthermic intraperitoneal chemotherapy: Italian multicenter study S.I.T.I.L.O. Eur. J. Surg. Oncol. 2011, 37, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez Silva, C.; Moreno Ruiz, F.J.; Bellido Estevez, I.; Carrasco Campos, J.; Titos Garcia, A.; Ruiz Lopez, M.; Gonzalez Poveda, I.; Toval Mata, J.A.; Mera Velasco, S.; Santoyo Santoyo, J. Are there intra-operative hemodynamic differences between the Coliseum and closed HIPEC techniques in the treatment of peritoneal metastasis? A retrospective cohort study. World J. Surg. Oncol. 2017, 15, 51. [Google Scholar] [CrossRef] [PubMed]
- Simkens, G.A.; Rovers, K.P.; Nienhuijs, S.W.; de Hingh, I.H. Patient selection for cytoreductive surgery and HIPEC for the treatment of peritoneal metastases from colorectal cancer. Cancer Manag. Res. 2017, 9, 259–266. [Google Scholar] [CrossRef]
- Marz, L.; Piso, P. Treatment of peritoneal metastases from colorectal cancer. Gastroenterol. Rep. 2015, 3, 298–302. [Google Scholar] [CrossRef]
- Ashvin, R.; Nikhilesh, J. Preoperative Preparation and Patient Selection for Cytoreductive Surgery and HIPEC. Indian J. Surg. Oncol. 2016, 7, 208–214. [Google Scholar] [CrossRef]
- Yoon, W.; Alame, A.; Berri, R. Peritoneal Surface Disease Severity Score as a predictor of resectability in the treatment of peritoneal surface malignancies. Am. J. Surg. 2014, 207, 403–407. [Google Scholar] [CrossRef]
- Ng, J.L.; Ong, W.S.; Chia, C.S.; Tan, G.H.; Soo, K.C.; Teo, M.C. Prognostic Relevance of the Peritoneal Surface Disease Severity Score Compared to the Peritoneal Cancer Index for Colorectal Peritoneal Carcinomatosis. Int. J. Surg. Oncol. 2016, 2016, 2495131. [Google Scholar] [CrossRef] [PubMed]
- Lunt, S.J.; Fyles, A.; Hill, R.P.; Milosevic, M. Interstitial fluid pressure in tumors: Therapeutic barrier and biomarker of angiogenesis. Future Oncol. 2008, 4, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Esquis, P.; Consolo, D.; Magnin, G.; Pointaire, P.; Moretto, P.; Ynsa, M.D.; Beltramo, J.L.; Drogoul, C.; Simonet, M.; Benoit, L.; et al. High intra-abdominal pressure enhances the penetration and antitumor effect of intraperitoneal cisplatin on experimental peritoneal carcinomatosis. Ann. Surg. 2006, 244, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Jacquet, P.; Stuart, O.A.; Chang, D.; Sugarbaker, P.H. Effects of intra-abdominal pressure on pharmacokinetics and tissue distribution of doxorubicin after intraperitoneal administration. Anticancer Drugs 1996, 7, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Furman, M.J.; Picotte, R.J.; Wante, M.J.; Rajeshkumar, B.R.; Whalen, G.F.; Lambert, L.A. Higher flow rates improve heating during hyperthermic intraperitoneal chemoperfusion. J. Surg. Oncol. 2014, 110, 970–975. [Google Scholar] [CrossRef]
- Chua, T.C.; Robertson, G.; Liauw, W.; Farrell, R.; Yan, T.D.; Morris, D.L. Intraoperative hyperthermic intraperitoneal chemotherapy after cytoreductive surgery in ovarian cancer peritoneal carcinomatosis: Systematic review of current results. J. Cancer Res. Clin. Oncol. 2009, 135, 1637–1645. [Google Scholar] [CrossRef]
- Park, E.J.; Ahn, J.; Gwak, S.W.; Park, K.S.; Baik, S.H.; Hwang, S.-J. Pharmacologic Properties of the Carrier Solutions for Hyperthermic Intraperitoneal Chemotherapy: Comparative Analyses Between Water and Lipid Carrier Solutions in the Rat Model. Ann. Surg. Oncol. 2018, 25, 3185–3192. [Google Scholar] [CrossRef]

| Drug | Type | Dosage (mg/m2) | AUC Ratio | Synergistic with Heat | Penetration Depth (mm) | Cell-Cycle Specific |
|---|---|---|---|---|---|---|
| Mitomycin C | Antitumor antibiotic (methylazirinopyrroloindoledione antineoplastic) | 10–160 | 23.5 | Yes | 2 | No |
| Oxaliplatin | Alkylating agent | 160–460 | 16 | Yes | 1–2 | No |
| Cisplatin | Alkylating agent | 50–360 | 7.8 | Yes | 1–3 | No |
| Doxorubicin | Antitumor antibiotic (anthracycline topoisomerase inhibitor) | 15 | 230 | Yes | 4–6 cell layers | No |
| Irinotecan | Plant alkaloids (topoisomerase inhibitor) | 100–400 | N/A | No | N/A | Yes |
| Paclitaxel | Plant alkaloids (microtubule inhibitor) | 60–175 | 1000 | No | >80 cell layers | Yes |
| Docetaxel | Plant alkaloids (microtubule inhibitor) | 80 | 552 | No | N/A | Yes |
| 5-fluorouracil | Antimetabolite (nucleoside metabolic inhibitor) | 1000 | 250 | Minimal | 0.2 | Yes |
| Carboplatin | Alkylating agent | 350–800 | 10 | Yes | 0.5 | No |
| Type of Carrier Solution | Advantages | Disadvantages |
|---|---|---|
| Isotonic salt solutions and Dextrose solutions | Rapidly absorbed due to low molecular weight | Inability to maintain a prolonged high intraperitoneal fluid volume |
| Hypotonic solutions | Increases the cisplatin accumulation and enhances its cytotoxicity in vitro | Unexplained postoperative peritoneal bleeding |
| Hypertonic solutions | Allows prolonged high intraperitoneal volume Slows down the clearance of intraperitoneal fluid | Dilution of intraperitoneal drug due to fluid shift inward to the peritoneal cavity |
| Isotonic molecular weight solutions | Prolonged high intraperitoneal volume Reduced drug clearance from the peritoneal cavity | Drug exposure to the cancer cells is not significantly increased |
| Temperature | Type of Hyperthermia | Cytotoxic Effect | Thermosensitization | Vascular Effect | Immune Reaction |
|---|---|---|---|---|---|
| 39–41 °C | Mild | Minimal growth arrest | Synergism with cytotoxic drugs | Increased blood flow | Enhanced |
| 31–43 °C | Moderate | Reversible growth arrest | Significant increased effect combined with cytotoxic drugs | Increased blood flow | Enhanced |
| >43 °C | Severe | Exponential growth arrest, significant cytotoxicity in normal cells | Significant increased effect combined with cytotoxic drugs | Reduced blood flow | Suppressed |
| Type of Technique | Advantages | Disadvantages |
|---|---|---|
| “Colosseum” technique | Relatively uniform drug distribution Relatively uniform temperature distribution Surgical interceptions possible Manual creation of optimal conditions | Heat dissipation 1 Possible aerosolization of chemotherapy 1 |
| Closed abdomen | Limited heat dissipation Reduced risk for theater staff | Minimal surgical interception possibilities Drug inhomogeneities |
| Peritoneal Cavity Expander (PCE) | Drug/heat distribution homogeneous Increased perfusion volume increasing effective surface Minimized exposure to theater staff Enlarged accessibility | Complex technique Experienced staff required |
| Laparoscopic | Limited heat dissipation Minimally invasive Can be used in different settings | Can only be used in combination with low tumor burden Drug and heat inhomogeneities |
| Criteria | Factor | Inclusion Criteria | Details |
|---|---|---|---|
| Performance status | 0 (able to carry out all normal activity) 4 (completely disabled) | ≤2 | Measuring a patient’s level of functioning in terms of their ability to care for themselves, daily activity, and physical ability |
| Karnofsky index | 0 (dead/moribund) 100 (no evidence of Disease) | >70 | A standard way of measuring the ability of cancer patients to perform ordinary tasks |
| PCI | 0 (no tumor) 39 (tumors greater than 5 cm spread through the Peritoneum) | CRC + gastric cancer: PCI ≤ 10; MPM + PMP + ovarian cancer: no PCI limit | Extent of disease at the time of surgery |
| Metastatic extent | 0 (no distant metastasis), 1 (distant metastasis present) | M0 | Score to define distant metastasis |
| Lymph node involvement | 0 (no involvement, 1 (1–3 node), 2 (>4 node) | N0 or N1/2 | Score to define lymph node involvement metastasis |
| CCR Score | 0 (complete CRS), 1 (0–2.5 mm), 2 (>2.5 mm) | CCR-0 or CCR-1/2 | Completeness of cytoreduction score after cytoreduction to assess residual nodules |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Helderman, R.F.C.P.A.; Löke, D.R.; Kok, H.P.; Oei, A.L.; Tanis, P.J.; Franken, N.A.P.K.; Crezee, J. Variation in Clinical Application of Hyperthermic Intraperitoneal Chemotherapy: A Review. Cancers 2019, 11, 78. https://doi.org/10.3390/cancers11010078
Helderman RFCPA, Löke DR, Kok HP, Oei AL, Tanis PJ, Franken NAPK, Crezee J. Variation in Clinical Application of Hyperthermic Intraperitoneal Chemotherapy: A Review. Cancers. 2019; 11(1):78. https://doi.org/10.3390/cancers11010078
Chicago/Turabian StyleHelderman, Roxan F. C. P. A., Daan R. Löke, H. Petra Kok, Arlene L. Oei, Pieter J. Tanis, Nicolaas A. P. Klaas Franken, and Johannes Crezee. 2019. "Variation in Clinical Application of Hyperthermic Intraperitoneal Chemotherapy: A Review" Cancers 11, no. 1: 78. https://doi.org/10.3390/cancers11010078
APA StyleHelderman, R. F. C. P. A., Löke, D. R., Kok, H. P., Oei, A. L., Tanis, P. J., Franken, N. A. P. K., & Crezee, J. (2019). Variation in Clinical Application of Hyperthermic Intraperitoneal Chemotherapy: A Review. Cancers, 11(1), 78. https://doi.org/10.3390/cancers11010078







