Small Bowel Carcinomas Associated with Immune-Mediated Intestinal Disorders: The Current Knowledge
Abstract
1. Introduction
2. Epidemiology and Risk Factors
3. Histopathology and Molecular Biology
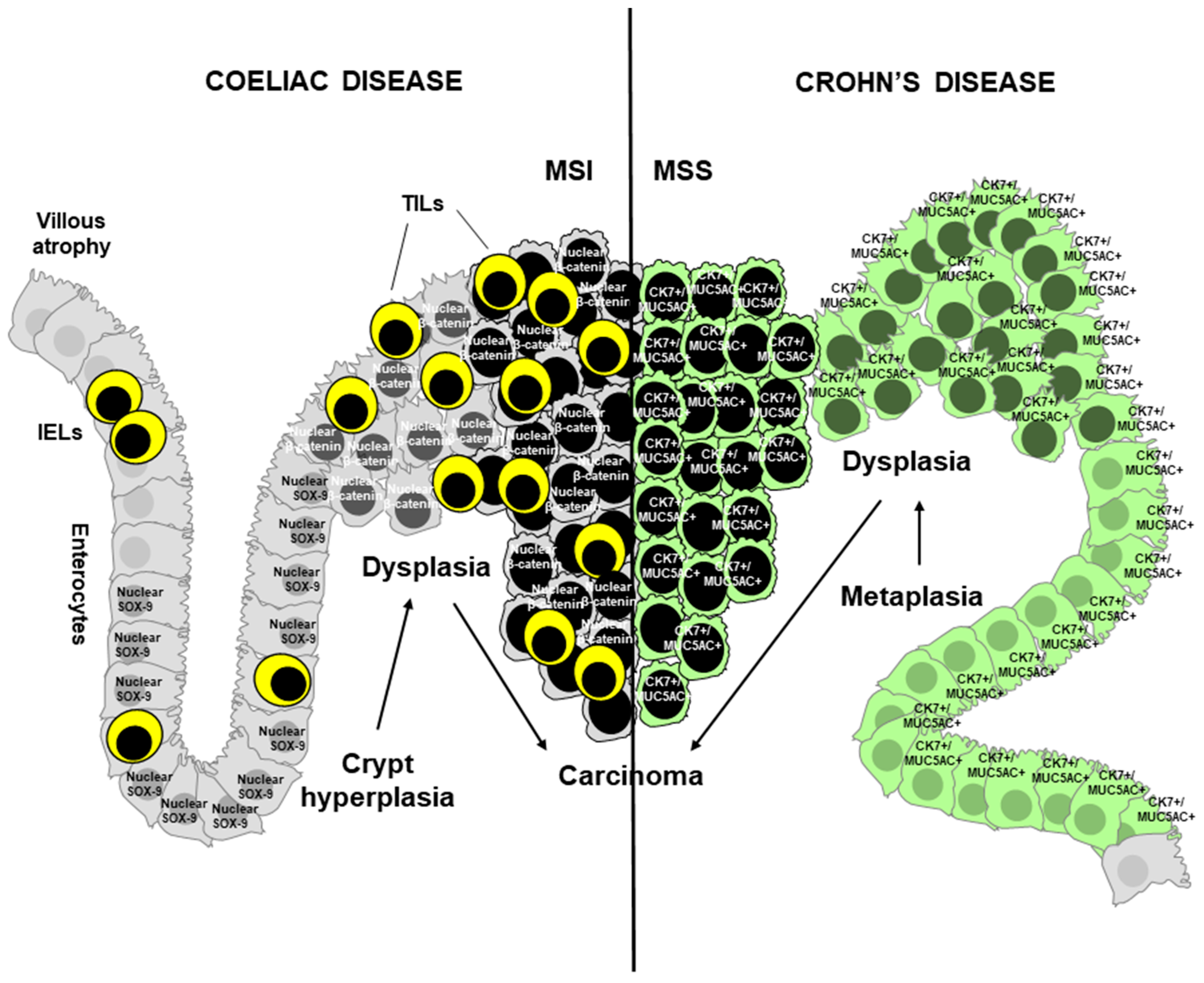
4. Pathogenesis and Preneoplastic Lesions
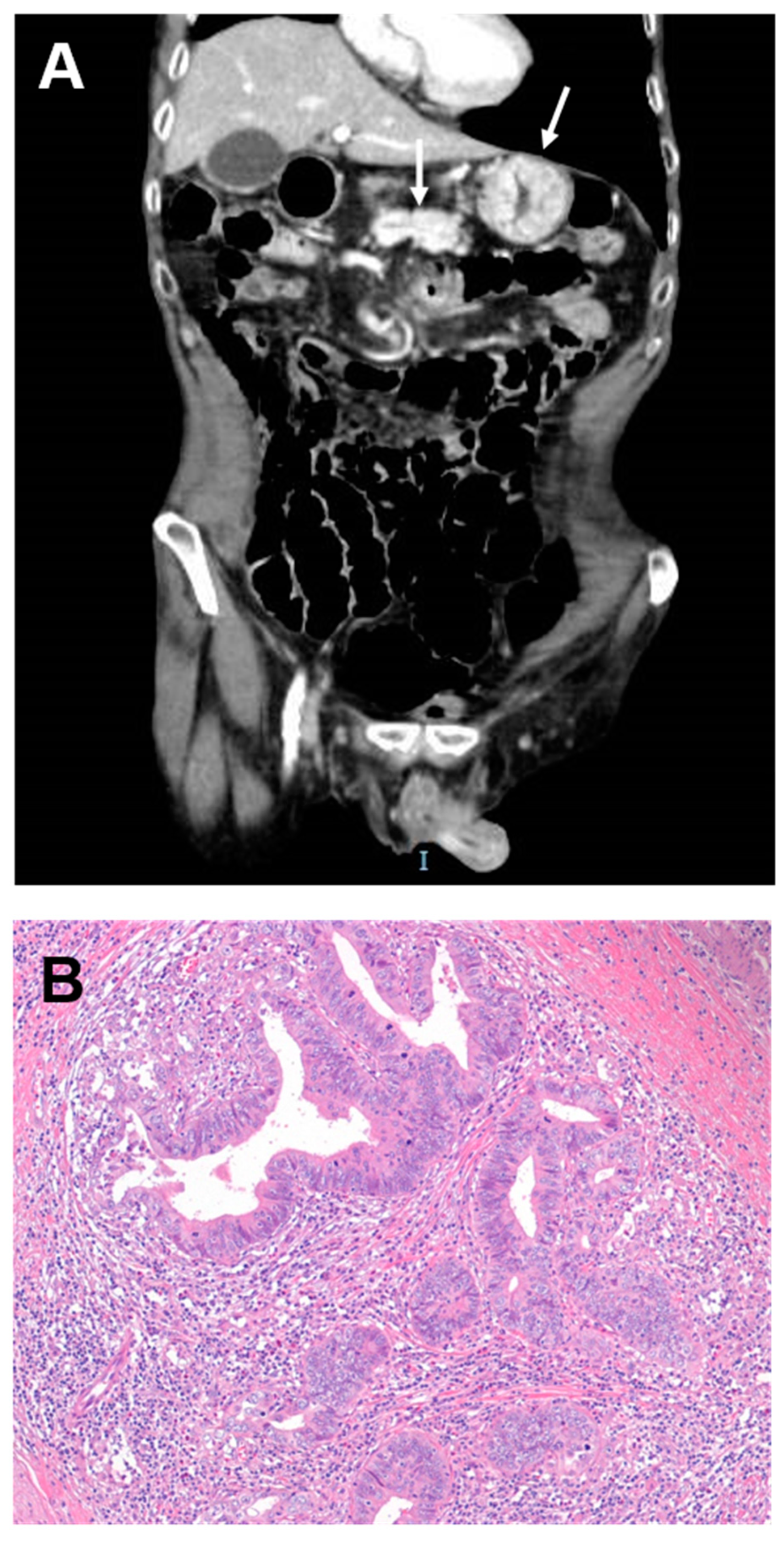
5. Clinical Presentation and Diagnosis
6. Prognosis and Treatment
7. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Raghav, K.; Overman, M.J. Small bowel adenocarcinomas—Existing evidence and evolving paradigms. Nat. Rev. Clin. Oncol. 2013, 10, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Vanoli, A.; Di Sabatino, A.; Furlan, D.; Klersy, C.; Grillo, F.; Fiocca, R.; Mescoli, C.; Rugge, M.; Nesi, G.; Fociani, P.; et al. Small Bowel Carcinomas in Coeliac or Crohn’s Disease: Clinico-pathological, Molecular, and Prognostic Features. A Study from the Small Bowel Cancer Italian Consortium. J. Crohns Colitis 2017, 11, 942–953. [Google Scholar] [CrossRef] [PubMed]
- Di Sabatino, A.; Lenti, M.V.; Giuffrida, P.; Vanoli, A.; Corazza, G.R. New insights into immune mechanisms underlying autoimmune diseases of the gastrointestinal tract. Autoimmun. Rev. 2015, 14, 1161–1169. [Google Scholar] [CrossRef] [PubMed]
- Di Sabatino, A.; Corazza, G.R. Coeliac disease. Lancet 2009, 373, 1480–1493. [Google Scholar] [CrossRef]
- Potter, D.D.; Murray, J.A.; Donohue, J.H.; Burgart, L.J.; Nagorney, D.M.; van Heerden, J.A.; Plevak, M.F.; Zinsmeister, A.R.; Thibodeau, S.N. The role of defective mismatch repair in small bowel adenocarcinoma in celiac disease. Cancer Res. 2004, 64, 7073–7077. [Google Scholar] [CrossRef] [PubMed]
- Diosdado, B.; Buffart, T.E.; Watkins, R.; Carvalho, B.; Ylstra, B.; Tijssen, M.; Bolijn, A.S.; Lewis, F.; Maude, K.; Verbeke, C.; et al. High-resolution array comparative genomic hybridization in sporadic and celiac disease-related small bowel adenocarcinomas. Clin. Cancer Res. 2010, 16, 1391–1401. [Google Scholar] [CrossRef]
- Vanoli, A.; Di Sabatino, A.; Martino, M.; Klersy, C.; Grillo, F.; Mescoli, C.; Nesi, G.; Volta, U.; Fornino, D.; Luinetti, O.; et al. Small bowel carcinomas in celiac or Crohn’s disease: Distinctive histophenotypic, molecular and histogenetic patterns. Mod. Pathol. 2017, 30, 1453–1466. [Google Scholar] [CrossRef]
- Svrcek, M.; Piton, G.; Cosnes, J.; Beaugerie, L.; Vermeire, S.; Geboes, K.; Lemoine, A.; Cervera, P.; El-Murr, N.; Dumont, S.; et al. Small bowel adenocarcinomas complicating Crohn’s disease are associated with dysplasia: A pathological and molecular study. Inflamm. Bowel Dis. 2014, 20, 1584–1592. [Google Scholar] [CrossRef]
- Giuffrida, P.; Corazza, G.R.; Di Sabatino, A. Old and New Lymphocyte Players in Inflammatory Bowel Disease. Dig. Dis. Sci. 2018, 63, 277–288. [Google Scholar] [CrossRef]
- Grolleau, C.; Pote, N.M.; Guedj, N.S.; Zappa, M.; Theou-Anton, N.; Bouhnik, Y.; Panis, Y.; Cazals-Hatem, D.L. Small bowel adenocarcinoma complicating Crohn’s disease: A single-centre experience emphasizing the importance of screening for dysplasia. Virchows Arch. 2017, 471, 611–617. [Google Scholar] [CrossRef]
- Rashid, A.; Hamilton, S.R. Genetic alterations in sporadic and Crohn’s-associated adenocarcinomas of the small intestine. Gastroenterology 1997, 113, 127–135. [Google Scholar] [CrossRef]
- Bojesen, R.D.; Riis, L.B.; Høgdall, E.; Nielsen, O.H.; Jess, T. Inflammatory Bowel Disease and Small Bowel Cancer Risk, Clinical Characteristics, and Histopathology: A Population-Based Study. Clin. Gastroenterol. Hepatol. 2017, 15, 1900–1907. [Google Scholar] [CrossRef] [PubMed]
- Howdle, P.D.; Jalal, P.K.; Holmes, G.K.; Houlston, R.S. Primary small-bowel malignancy in the UK and its association with coeliac disease. QJM 2003, 96, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Bruno, C.J.; Batts, K.P.; Ahlquist, D.A. Evidence against flat dysplasia as a regional field defect in small bowel adenocarcinoma associated with celiac sprue. Mayo Clin. Proc. 1997, 72, 320–322. [Google Scholar] [CrossRef] [PubMed]
- Michelassi, F.; Testa, G.; Pomidor, W.J.; Lashner, B.A.; Block, G.E. Adenocarcinoma complicating Crohn’s disease. Dis. Colon Rectum 1993, 36, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Sigel, J.E.; Petras, R.E.; Lashner, B.A.; Fazio, V.W.; Goldblum, J.R. Intestinal adenocarcinoma in Crohn’s disease: A report of 30 cases with a focus on coexisting dysplasia. Am. J. Surg. Pathol. 1999, 23, 651–655. [Google Scholar] [CrossRef]
- Palascak-Juif, V.; Bouvier, A.M.; Cosnes, J.; Flourié, B.; Bouché, O.; Cadiot, G.; Lémann, M.; Bonaz, B.; Denet, C.; Marteau, P.; et al. Small bowel adenocarcinoma in patients with Crohn’s disease compared with small bowel adenocarcinoma de novo. Inflamm. Bowel Dis. 2005, 11, 828–832. [Google Scholar] [CrossRef]
- Piton, G.; Cosnes, J.; Monnet, E.; Beaugerie, L.; Seksik, P.; Savoye, G.; Cadiot, G.; Flourie, B.; Capelle, P.; Marteau, P.; et al. Risk factors associated with small bowel adenocarcinoma in Crohn’s disease: A case-control study. Am. J. Gastroenterol. 2008, 103, 1730–1736. [Google Scholar] [CrossRef]
- Widmar, M.; Greenstein, A.J.; Sachar, D.B.; Harpaz, N.; Bauer, J.J.; Greenstein, A.J. Small bowel adenocarcinoma in Crohn’s disease. J. Gastrointest. Surg. 2011, 15, 797–802. [Google Scholar] [CrossRef]
- Whitcomb, E.; Liu, X.; Xiao, S.Y. Crohn enteritis-associated small bowel adenocarcinomas exhibit gastric differentiation. Hum. Pathol. 2014, 45, 359–367. [Google Scholar] [CrossRef]
- Weber, N.K.; Fletcher, J.G.; Fidler, J.L.; Barlow, J.M.; Pruthi, S.; Loftus, E.V., Jr.; Pardi, D.S.; Smyrk, T.C.; Becker, B.D.; Pasha, S.F.; et al. Clinical characteristics and imaging features of small bowel adenocarcinomas in Crohn’s disease. Abdom. Imaging 2015, 40, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Wieghard, N.; Mongoue-Tchokote, S.; Young, J.I.; Sheppard, B.C.; Tsikitis, V.L. Prognosis of small bowel adenocarcinoma in Crohn’s disease compares favourably with de novo small bowel adenocarcinoma. Colorectal. Dis. 2017, 19, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Warth, A.; Kloor, M.; Schirmacher, P.; Bläker, H. Genetics and epigenetics of small bowel adenocarcinoma: The interactions of CIN, MSI, and CIMP. Mod. Pathol. 2011, 24, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Overman, M.J.; Pozadzides, J.; Kopetz, S.; Wen, S.; Abbruzzese, J.L.; Wolff, R.A.; Wang, H. Immunophenotype and molecular characterisation of adenocarcinoma of the small intestine. Br. J. Cancer 2010, 102, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Vanoli, A.; Di Sabatino, A.; Biancone, L.; Martino, M.; Macciomei, M.C.; Zorzi, F.; Pallone, F.; Solcia, E.; Corazza, G.R. Small bowel Epstein-Barr virus-positive lympho-epithelioma-like carcinoma in Crohn’s disease. Histopathology 2017, 70, 837–839. [Google Scholar] [CrossRef] [PubMed]
- Vanoli, A.; Di Sabatino, A.; Martino, M.; Dallera, E.; Furlan, D.; Mescoli, C.; Macciomei, M.C.; Biancone, L.; Neri, B.; Grillo, F.; et al. Epstein-Barr virus-positive ileal carcinomas associated with Crohn’s disease. Virchows Arch. 2017, 471, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Von Rahden, B.H.; Langner, C.; Brücher, B.L.; Stein, H.J.; Sarbia, M. No association of primary adenocarcinomas of the small bowel with Epstein-Barr virus infection. Mol. Carcinog. 2006, 45, 349–352. [Google Scholar] [CrossRef] [PubMed]
- DeSesso, J.M.; Jacobson, C.F. Anatomical and physiological parameters affecting gastrointestinal absorption in humans and rats. Food Chem. Toxicol. 2001, 39, 209–228. [Google Scholar] [CrossRef]
- Aparicio, T.; Zaanan, A.; Svrcek, M.; Laurent-Puig, P.; Carrere, N.; Manfredi, S.; Locher, C.; Afchain, P. Small bowel adenocarcinoma: Epidemiology, risk factors, diagnosis and treatment. Dig. Liver Dis. 2014, 46, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Kummar, S.; Ciesielski, T.E.; Fogarasi, M.C. Management of small bowel adenocarcinoma. Oncology 2002, 16, 1364–1369. [Google Scholar]
- Bilimoria, K.Y.; Bentrem, D.J.; Wayne, J.D.; Ko, C.Y.; Bennett, C.L.; Talamonti, M.S. Small bowel cancer in the United States: Changes in epidemiology, treatment, and survival over the last 20 years. Ann. Surg. 2009, 249, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Faivre, J.; Trama, A.; De Angelis, R.; Elferink, M.; Siesling, S.; Audisio, R.; Bosset, J.F.; Cervantes, A.; Lepage, C.; RARECARE Working Group. Incidence, prevalence and survival of patients with rare epithelial digestive cancers diagnosed in Europe in 1995–2002. Eur. J. Cancer 2012, 48, 1417–1424. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Chen, W.; Li, P.; Ye, J. Association Between Coeliac Disease and Risk of Any Malignancy and Gastrointestinal Malignancy: A Meta-Analysis. Medicine 2015, 94, e1612. [Google Scholar] [CrossRef] [PubMed]
- Canavan, C.; Abrams, K.R.; Mayberry, J. Meta-analysis: Colorectal and small bowel cancer risk in patients with Crohn’s disease. Aliment. Pharmacol. Ther. 2006, 23, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Silano, M.; Volta., U.; Vincenzi, A.D.; Dessì, M.; Vincenzi, M.D.; Collaborating Centers of the Italian Registry of the Complications of Coeliac Disease. Effect of a gluten-free diet on the risk of enteropathy-associated T-cell lymphoma in celiac disease. Dig. Dis. Sci. 2008, 53, 972–976. [Google Scholar] [CrossRef] [PubMed]
- Elfström, P.; Granath, F.; Ye, W.; Ludvigsson, J.F. Low risk of gastrointestinal cancer among patients with celiac disease, inflammation, or latent celiac disease. Clin. Gastroenterol. Hepatol. 2012, 10, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, R.; Kelly, C.P.; Leffler, D.A. Gastrointestinal cancer in celiac disease: “The first days are the hardest days, don’t you worry anymore?”. Clin. Gastroenterol. Hepatol. 2012, 10, 4–6. [Google Scholar] [CrossRef]
- Silano, M.; Volta, U.; Mecchia, A.M.; Dessì, M.; Di Benedetto, R.; De Vincenzi, M.; Collaborating Centers of the Italian Registry of the Complications of Coeliac Disease. Delayed diagnosis of coeliac disease increases cancer risk. BMC Gastroenterol. 2007, 7, 8. [Google Scholar] [CrossRef]
- Di Sabatino, A.; Biagi, F.; Gobbi, P.G.; Corazza, G.R. How I treat enteropathy-associated T-cell lymphoma. Blood 2012, 119, 2458–2468. [Google Scholar] [CrossRef]
- Cantoro, L.; Di Sabatino, A.; Papi, C.; Margagnoni, G.; Ardizzone, S.; Giuffrida, P.; Giannarelli, D.; Massari, A.; Monterubbianesi, R.; Lenti, M.V.; et al. The Time Course of Diagnostic Delay in Inflammatory Bowel Disease Over the Last Sixty Years: An Italian Multicentre Study. J. Crohns Colitis 2017, 11, 975–980. [Google Scholar] [CrossRef]
- Cahill, C.; Gordon, P.H.; Petrucci, A.; Boutros, M. Small bowel adenocarcinoma and Crohn’s disease: Any further ahead than 50 years ago? World J. Gastroenterol. 2014, 20, 11486–11495. [Google Scholar] [CrossRef] [PubMed]
- Lashner, B.A. Risk factors for small bowel cancer in Crohn’s disease. Dig. Dis. Sci. 1992, 37, 1179–1184. [Google Scholar] [CrossRef]
- Solem, C.A.; Harmsen, W.S.; Zinsmeister, A.R.; Loftus, E.V., Jr. Small intestinal adenocarcinoma in Crohn’s disease: A case-control study. Inflamm. Bowel Dis. 2004, 10, 32–35. [Google Scholar] [CrossRef]
- Schottenfeld, D.; Beebe-Dimmer, J.L.; Vigneau, F.D. The epidemiology and pathogenesis of neoplasia in the small intestine. Ann. Epidemiol. 2009, 19, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Goodman, M.T.; Matsuno, R.K.; Shvetsov, Y.B. Racial and ethnic variation in the incidence of small-bowel cancer subtypes in the United States, 1995–2008. Dis. Colon Rectum 2013, 56, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Brcic, I.; Cathomas, G.; Vanoli, A.; Jilek, K.; Giuffrida, P.; Langner, C. Medullary carcinoma of the small bowel. Histopathology 2016, 69, 136–140. [Google Scholar] [CrossRef]
- Rosenbaum, M.W.; Bledsoe, J.R.; Morales-Oyarvide, V.; Huynh, T.G.; Mino-Kenudson, M. PD-L1 expression in colorectal cancer is associated with microsatellite instability, BRAF mutation, medullary morphology and cytotoxic tumor-infiltrating lymphocytes. Mod. Pathol. 2016, 29, 1104–1112. [Google Scholar] [CrossRef]
- De Rosa, S.; Sahnane, N.; Tibiletti, M.G.; Magnoli, F.; Vanoli, A.; Sessa, F.; Chiaravalli, A.M. EBV⁺ and MSI Gastric Cancers Harbor High PD-L1/PD-1 Expression and High CD8⁺ Intratumoral Lymphocytes. Cancers 2018, 10, 102. [Google Scholar] [CrossRef]
- Thota, R.; Gonzalez, R.S.; Berlin, J.; Cardin, D.B.; Shi, C. Could the PD-1 Pathway Be a Potential Target for Treating Small Intestinal Adenocarcinoma? Am. J. Clin. Pathol. 2017, 148, 208–214. [Google Scholar] [CrossRef]
- Schrock, A.B.; Devoe, C.E.; McWilliams, R.; Sun, J.; Aparicio, T.; Stephens, P.J.; Ross, J.S.; Wilson, R.; Miller, V.A.; Ali, S.M.; et al. Genomic Profiling of Small-Bowel Adenocarcinoma. JAMA Oncol. 2017, 3, 1546–1553. [Google Scholar] [CrossRef]
- Aparicio, T.; Svrcek, M.; Zaanan, A.; Beohou, E.; Laforest, A.; Afchain, P.; Mitry, E.; Taieb, J.; Di Fiore, F.; Gornet, J.M.; et al. Small bowel adenocarcinoma phenotyping, a clinicobiological prognostic study. Br. J. Cancer 2013, 109, 3057–3066. [Google Scholar] [CrossRef] [PubMed]
- Yaeger, R.; Shah, M.A.; Miller, V.A.; Kelsen, J.R.; Wang, K.; Heins, Z.J.; Ross, J.S.; He, Y.; Sanford, E.; Yantiss, R.K.; et al. Genomic Alterations Observed in Colitis-Associated Cancers Are Distinct from Those Found in Sporadic Colorectal Cancers and Vary by Type of Inflammatory Bowel Disease. Gastroenterology 2016, 151, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Younes, N.; Fulton, N.; Tanaka, R.; Wayne, J.; Straus, F.H., 2nd; Kaplan, E.L. The presence of K-12 ras mutations in duodenal adenocarcinomas and the absence of ras mutations in other small bowel adenocarcinomas and carcinoid tumors. Cancer 1997, 79, 1804–1808. [Google Scholar] [CrossRef]
- Bläker, H.; Helmchen, B.; Bönisch, A.; Aulmann, S.; Penzel, R.; Otto, H.F.; Rieker, R.J. Mutational activation of the RAS-RAF-MAPK and the Wnt pathway in small intestinal adenocarcinomas. Scand. J. Gastroenterol. 2004, 39, 748–753. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Lee, O.J.; Jang, K.T.; Bae, Y.K.; Chung, J.Y.; Eom, D.W.; Kim, J.M.; Yu, E.; Hong, S.M. Combined loss of E-cadherin and aberrant β-catenin protein expression correlates with a poor prognosis for small intestinal adenocarcinomas. Am. J. Clin. Pathol. 2013, 139, 167–176. [Google Scholar] [CrossRef]
- Fang, M.; Ou, J.; Hutchinson, L.; Green, M.R. The BRAF oncoprotein functions through the transcriptional repressor MAFG to mediate the CpG Island Methylator phenotype. Mol. Cell. 2014, 55, 904–915. [Google Scholar] [CrossRef]
- Serra, R.W.; Fang, M.; Park, S.M.; Hutchinson, L.; Green, M.R. A KRAS-directed transcriptional silencing pathway that mediates the CpG island methylator phenotype. Elife 2014, 3, e02313. [Google Scholar] [CrossRef]
- Laforest, A.; Aparicio, T.; Zaanan, A.; Silva, F.P.; Didelot, A.; Desbeaux, A.; Le Corre, D.; Benhaim, L.; Pallier, K.; Aust, D.; et al. ERBB2 gene as a potential therapeutic target in small bowel adenocarcinoma. Eur. J. Cancer 2014, 50, 1740–1746. [Google Scholar] [CrossRef]
- Rampertab, S.D.; Forde, K.A.; Green, P.H. Small bowel neoplasia in coeliac disease. Gut 2003, 52, 1211–1214. [Google Scholar] [CrossRef]
- Perzin, K.H.; Bridge, M.F. Adenomas of the small intestine: A clinicopathologic review of 51 cases and a study of their relationship to carcinoma. Cancer 1981, 48, 799–819. [Google Scholar] [CrossRef]
- Genta, R.M.; Feagins, L.A. Advanced precancerous lesions in the small bowel mucosa. Best. Pract. Res. Clin. Gastroenterol. 2013, 27, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Senger, S.; Sapone, A.; Fiorentino, M.R.; Mazzarella, G.; Lauwers, G.Y.; Fasano, A. Celiac Disease Histopathology Recapitulates Hedgehog Downregulation, Consistent with wound Healing Processes Activation. PLoS ONE 2015, 10, e0144634. [Google Scholar] [CrossRef] [PubMed]
- Simon, M.; Cosnes, J.; Gornet, J.M.; Seksik, P.; Stefanescu, C.; Blain, A.; Pariente, B.; Nancey, S.; Vuitton, L.; Nachury, M.; et al. Endoscopic Detection of Small Bowel Dysplasia and Adenocarcinoma in Crohn’s Disease: A Prospective Cohort-Study in High-Risk Patients. J. Crohns Colitis 2017, 11, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Ciccocioppo, R.; Racca, F.; Scudeller, L.; Piralla, A.; Formagnana, P.; Pozzi, L.; Betti, E.; Vanoli, A.; Riboni, R.; Kruzliak, P.; et al. Differential cellular localization of Epstein-Barr virus and human cytomegalovirus in the colonic mucosa of patients with active or quiescent inflammatory bowel disease. Immunol. Res. 2016, 64, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Genitsch, V.; Novotny, A.; Seiler, C.A.; Kröll, D.; Walch, A.; Langer, R. Epstein-barr virus in gastroesophageal adenocarcinomas—Single center experiences in the context of current literature. Front. Oncol. 2015, 5, 73. [Google Scholar] [CrossRef] [PubMed]
- Tomba, C.; Sidhu, R.; Sanders, D.S.; Mooney, P.D.; Branchi, F.; Locatelli, M.; Roncoroni, L.; Conte, D.; Bardella, M.T.; Elli, L. Celiac Disease and Double-Balloon Enteroscopy: What Can we Achieve? The Experience of 2 European Tertiary Referral Centers. J. Clin. Gastroenterol. 2016, 50, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Rieder, F.; Latella, G.; Magro, F.; Yuksel, E.S.; Higgins, P.D.; Di Sabatino, A.; de Bruyn, J.R.; Rimola, J.; Brito, J.; Bettenworth, D.; et al. European Crohn’s and Colitis Organisation Topical Review on Prediction, Diagnosis and Management of Fibrostenosing Crohn’s Disease. J. Crohns Colitis 2016, 10, 873–885. [Google Scholar] [CrossRef] [PubMed]
- Bergamaschi, G.; Di Sabatino, A.; Corazza, G.R. Pathogenesis, diagnosis and treatment of anaemia in immune-mediated gastrointestinal disorders. Br. J. Haematol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Baars, J.E.; Thijs, J.C.; Bac, D.J.; Ter Borg, P.C.; Kuipers, E.J.; van der Woude, C.J. Small bowel carcinoma mimicking a relapse of Crohn’s disease: A case series. J. Crohns Colitis 2011, 5, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Collier, P.E.; Turowski, P.; Diamond, D.L. Small intestinal adenocarcinoma complicating regional enteritis. Cancer 1985, 55, 516–521. [Google Scholar] [CrossRef]
- Overman, M.J.; Hu, C.Y.; Kopetz, S.; Abbruzzese, J.L.; Wolff, R.A.; Chang, G.J. A population-based comparison of adenocarcinoma of the large and small intestine: Insights into a rare disease. Ann. Surg. Oncol. 2012, 19, 1439–1445. [Google Scholar] [CrossRef] [PubMed]
- Halfdanarson, T.R.; McWilliams, R.R.; Donohue, J.H.; Quevedo, J.F. A single-institution experience with 491 cases of small bowel adenocarcinoma. Am. J. Surg. 2010, 199, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Overman, M.J.; Hu, C.Y.; Wolff, R.A.; Chang, G.J. Prognostic value of lymph node evaluation in small bowel adenocarcinoma: Analysis of the surveillance, epidemiology, and end results database. Cancer 2010, 116, 5374–5382. [Google Scholar] [CrossRef] [PubMed]
- Dabaja, B.S.; Suki, D.; Pro, B.; Bonnen, M.; Ajani, J. Adenocarcinoma of the small bowel: Presentation, prognostic factors, and outcome of 217 patients. Cancer 2004, 101, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Locher, C.; Batumona, B.; Afchain, P.; Carrère, N.; Samalin, E.; Cellier, C.; Aparicio, T.; Becouarn, Y.; Bedenne, L.; Michel, P.; et al. Small bowel adenocarcinoma: French intergroup clinical practice guidelines for diagnosis, treatments and follow-up (SNFGE, FFCD, GERCOR, UNICANCER, SFCD, SFED, SFRO). Dig. Liver Dis. 2018, 50, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Santini, D.; Fratto, M.E.; Spoto, C.; Russo, A.; Galluzzo, S.; Zoccoli, A.; Vincenzi, B.; Tonini, G. Cetuximab in small bowel adenocarcinoma: A new friend? Br. J. Cancer 2010, 103, 1305. [Google Scholar] [CrossRef] [PubMed]
- Falcone, R.; Roberto, M.; Filetti, M.; Anselmi, E.; Marchetti, P. Anti epidermal growth factor receptor therapy in small bowel adenocarcinoma: Case report and literature review. Medicine 2018, 97, e9672. [Google Scholar] [CrossRef]
- Gulhati, P.; Raghav, K.; Shroff, R.; Varadhachary, G.; Javle, M.; Qiao, W.; Wang, H.; Morris, J.; Wolff, R.; Overman, M.J. Phase II Study of Panitumumab in RAS Wild-Type Metastatic Adenocarcinoma of Small Bowel or Ampulla of Vater. Oncologist 2018, 23, 277-e26. [Google Scholar] [CrossRef]
- Venook, A.P. Right-sided vs left-sided colorectal cancer. Clin. Adv. Hematol. Oncol. 2017, 15, 22–24. [Google Scholar]
- Hamad, A.; Singhi, A.D.; Bahary, N.; McGrath, K.; Amarin, R.; Zeh, H.J.; Zureikat, A.H. Neoadjuvant Treatment with Trastuzumab and FOLFOX Induces a Complete Pathologic Response in a Metastatic ERBB2 (HER2)-Amplified Duodenal Cancer. J. Natl. Compr. Canc. Netw. 2017, 15, 983–988. [Google Scholar] [CrossRef]
- US National Library of Medicine. ClinicalTrials.gov. 2018. Available online: https://clinicaltrials.gov/ct2/show/NCT03000179?term=avelumab&cond=Small+Bowel+Carcinoma&rank=1 (accessed on 18 November 2018).
- US National Library of Medicine. ClinicalTrials.gov. 2018. Available online: https://clinicaltrials.gov/ct2/show/NCT03108131?term=atezolizumab&cond=Small+Bowel+Cancer&rank=1 (accessed on 18 November 2018).
- Le, D.T.; Uram, J.N.; Wang, H.; Bartlett, B.R.; Kemberling, H.; Eyring, A.D.; Skora, A.D.; Luber, B.S.; Azad, N.S.; Laheru, D.; et al. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N. Engl. J. Med. 2015, 372, 2509–2520. [Google Scholar] [CrossRef] [PubMed]
- US National Library of Medicine. ClinicalTrials.gov. 2018. Available online: https://clinicaltrials.gov/ct2/show/NCT02949219?term=pembrolizumab&cond=Small+Bowel+Cancer&rank=1 (accessed on 18 November 2018).
- US National Library of Medicine. ClinicalTrials.gov. 2018. Available online: https://clinicaltrials.gov/ct2/show/NCT03095781?term=pembrolizumab&cond=Small+Bowel+Cancer&rank=2 (accessed on 18 November 2018).
- US National Library of Medicine. ClinicalTrials.gov. 2018. Available online: https://clinicaltrials.gov/ct2/show/NCT02834013?term=ipilimumab&recrs=ab&cond=Small+Bowel+Adenocarcinoma&rank=1 (accessed on 18 November 2018).
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413. [Google Scholar] [CrossRef] [PubMed]
| Risk Factor |
|---|
| Inherited Tumour Syndromes |
| Familial adenomatous polyposis |
| Peutz-Jeghers syndrome |
| Hereditary nonpolyposis colon cancer syndrome (Lynch syndrome) |
| Juvenile polyposis syndrome |
| MUTYH-associated polyposis |
| Other Genetic Disorders |
| Cystic fibrosis |
| Immune-Mediated Intestinal Disorders |
| Coeliac disease |
| Crohn’s disease |
| Other Causes |
| Small bowel sporadic adenomatous polyps |
| Long-standing ileostomy |
| Feature | CD-SBC | CrD-SBC | Spo-SBC |
|---|---|---|---|
| Age at diagnosis | 53–62 yrs [2,5,6,13,14] | 42–73 yrs [2,8,10,11,12,15,16,17,18,19,20,21,22] | 56.5–72.1 yrs [2,5,6,10,17,20,22] |
| Site | Jejunum and duodenum [2,5,6,13,14] | Ileum [2,8,10,11,12,15,16,17,19,20,21,22] | Jejunum and duodenum [2,5,6,10,17,20] |
| MSI status | 65–73% [2,5,6] | 0–16% [2,8,10,11,12] | 9–35% [2,5,6,10,23,24] |
| Tumour cell phenotype | Intestinal [7] | Non-intestinal [7,20] | Intestinal [7] |
| Oncogenic viruses | Unknown | EBV latent infection [25,26] | No association with EBV infection [27] |
| Authors, Year | Pt | Age at SBC dgn (Median, Range, yrs) | Age at CD or CrD dgn (Median, Range, yrs) | CD or CrD Duration at SBC dgn (Median, Range, yrs) | Stage III/IV (%) | Overall Survival (%) | Main Findings |
|---|---|---|---|---|---|---|---|
| Small bowel carcinoma associated with coeliac disease (CD-SBC) | |||||||
| Bruno JC et al., 1997 [14] | 6 | 62, 45–75 | NA | 17, 0–40 | NA | NA | No evidence of flat dysplasia was present |
| Howdle PD et al., 2003 [13] | 23 | 62 *, 47–80 | NA | 8.2, 0.8–36 | NA | NA | CD-SBC account for 13% of all SBC |
| Potter DD et al., 2004 [5] | 17 | 59.5, 42–78 | 53, 25–77 | NA | 8/17 (47) | 64.2 at 5 yrs | CD-SBC have a high incidence of mismatch repair deficiency |
| Diosdado B et al., 2010 [6] | 15 | 61, 47–79 | 59, 18–79 | 2.5, 0–32.3 | NA | NA | CD-SBC have promoter hypermethylation of the APC gene |
| Vanoli A et al., 2017 [2,7] | 26 | 53, 28–80 | 49, 7–79 | 1.4, 0–25 | 8/26 (31) | 83 at 5 yrs | CD-SBC harbour MSI, high TILs and nuclear β-catenin expression frequently and show a better outcome in comparison with CrD-SBC |
| Small bowel carcinoma associated with Crohn’s disease (CrD-SBC) | |||||||
| Michelassi F et al., 1993 [15] | 7 | 47.7 *, 33–73 | 24, 11–57 | 20, 10–30 | NA | 6 mos (mean) | Survival is worse in CrD-SBC than in colorectal cancer complicating CrD |
| Rashid A et al., 1997 [11] | 8 | 45.5, 35–71 | 33.5 | NA, 0–30 | 0/7 (0) | 28.5 mos (median) | CrD-SBC have RAS and TP53 mutations |
| Sigel JE et al., 1999 [16] | 8 | 42, 35–71 | 35, 23–52 | 12, 0.6–19 | 2/8 (25) | NA | Most CrD-SBC have dysplasia adjacent to carcinoma |
| Palascak-Juif V et al., 2005 [17] | 20 | 47, 33–72 | 36, 15–54 | 16, 0–37 | 11/20 (55) | 35 at 5 yrs | Signet-ring cells were found in 7/20 CrD-SBC |
| Piton G et al., 2008 [18] | 29 | 45, 29–74 | 34, 13–63 | 7, 0–52 | NA | NA | Small bowel resection and salicylate intake ≥2 yrs protect against CrD-SBC |
| Widmar M et al., 2011 [19] | 29 | 55.4, 22–81 | 25, 13–63 | 25.2, 0.8–51.3 | 16/29 (55) | NA | Two clinical indicators of SBC were symptoms in longstanding quiescent CrD and obstruction refractory to medical therapy |
| Svrcek M et al., 2014 [8] | 41 | 47 | NA | 13.5 | 19/41 (46) | NA | 40/41 CrD-SBC were observed in inflamed mucosal areas. Flat or raised dysplasia was found in 20/41 patients with CrD-SBC |
| Whitcomb E et al., 2014 [20] | 11 | 47, 42–77 | 24, 6–33 | 25, 10–40 | NA | NA | 10/11 CrD-SBC expressed at least a gastric marker and 8/11 CrD-SBC expressed the pancreatobiliary marker CK7 |
| Weber NK et al., 2015 [21] | 34 | 52.9, 32–74 | 22.4, 13.0–69.3 | 22.3, 0–50.6 | NA | 52 at 2 yrs | Imaging features suggestive for CrD-SBC included annular mass, nodularity at the extraluminal margins of mass, and perforation |
| Grolleau C et al., 2017 [10] | 9 | 46, 37–67 | 36, 10–67 | 15, 0–32 | 5/9 (56) | 56 at 2 yrs | Adjacent dysplasia was present in 9/9 CrD-SBC |
| Bojesen RD et al., 2017 [12] | 23 | 53, 37–85 | NA | NA | NA | 26 at 5 yrs | 79% of CrD-SBC showed inflammation-dysplasia-carcinoma sequence |
| Wieghard N et al., 2017 [22] | 179 | 72.9 | NA | NA | 71/179 (40) | 3.9 yrs (median) | CrD-SBC have similar overal survival compared to sporadic SBC |
| Vanoli A et al., 2017 [2,7] | 25 | 59, 33–84 | 50, 22–84 | 13, 0–41 | 13/25 (52) | 38 at 5 yrs | CrD-SBC exhibit a low rate of MSI and TILs CrD-SBC are associated with dysplasia and metaplasia, both showing gastropancreatobiliary phenotype |
| Vanoli A et al., 2017 [26] | 31 | 59, 33–84 | NA | NA | 17/31 (55) | NA | EBV+ CrD-SBC may occur |
| Authors, Year | Pt | MSI Status N (%) | KRAS Mutation N (%) | NRAS Mutation N (%) | BRAF Mutation N (%) | PIK3CA Mutation N (%) | HER2 AmplificationN (%) | p53 Overexpression N (%) | Nuclear β-Catenin Expression N (%) |
|---|---|---|---|---|---|---|---|---|---|
| Small bowel carcinoma associated with coeliac disease (CD-SBC) | |||||||||
| Potter DD et al., 2004 [5] | 17 | 8/11 (73) | NA | NA | NA | NA | NA | NA | NA |
| Diosdado B et al., 2010 [6] | 15 | 6/9 (67) | NA | NA | NA | NA | NA | NA | NA |
| Vanoli A et al., 2017 [2,7] | 26 | 17/26 (65) | 8/26 (31) | 1/26 (4) | 0/26 (0) | 4/26 (15) | 2/26 (8) | 12/26 (46) | 24/26 (92) |
| Small bowel carcinoma associated with Crohn’s disease (CrD-SBC) | |||||||||
| Rashid A et al., 1997 [11] | 8 | 1/7 (14) | 3/7 (43) | NA | NA | NA | NA | 4/7 (57) | NA |
| Svrcek M et al., 2014 [8] | 41 | 1/36 (3) | 7/30 (23) | NA | 1/29 (4) | 0/23 (0) | NA | 21/35 (60) | 16/31 (52) |
| Grolleau C et al., 2017 [10] | 9 | 1/9 (11) | 1/8 (12.5) | NA | 0/8 (0) | NA | NA | NA | NA |
| Bojesen RD et al., 2017 [12] | 23 | 0/14 (0) | 2/14 (14) | NA | 1/14 (7) | NA | NA | NA | NA |
| Vanoli A et al., 2017 [2,7] | 25 | 4/25 (16) | 4/25 (12) | 1/25 (4) | 0/25 (0) | 2/25 (8) | 2/25 (8) | 12/25 (48) | 6/24 (25) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giuffrida, P.; Vanoli, A.; Arpa, G.; Bonometti, A.; Luinetti, O.; Solcia, E.; Corazza, G.R.; Paulli, M.; Di Sabatino, A. Small Bowel Carcinomas Associated with Immune-Mediated Intestinal Disorders: The Current Knowledge. Cancers 2019, 11, 31. https://doi.org/10.3390/cancers11010031
Giuffrida P, Vanoli A, Arpa G, Bonometti A, Luinetti O, Solcia E, Corazza GR, Paulli M, Di Sabatino A. Small Bowel Carcinomas Associated with Immune-Mediated Intestinal Disorders: The Current Knowledge. Cancers. 2019; 11(1):31. https://doi.org/10.3390/cancers11010031
Chicago/Turabian StyleGiuffrida, Paolo, Alessandro Vanoli, Giovanni Arpa, Arturo Bonometti, Ombretta Luinetti, Enrico Solcia, Gino Roberto Corazza, Marco Paulli, and Antonio Di Sabatino. 2019. "Small Bowel Carcinomas Associated with Immune-Mediated Intestinal Disorders: The Current Knowledge" Cancers 11, no. 1: 31. https://doi.org/10.3390/cancers11010031
APA StyleGiuffrida, P., Vanoli, A., Arpa, G., Bonometti, A., Luinetti, O., Solcia, E., Corazza, G. R., Paulli, M., & Di Sabatino, A. (2019). Small Bowel Carcinomas Associated with Immune-Mediated Intestinal Disorders: The Current Knowledge. Cancers, 11(1), 31. https://doi.org/10.3390/cancers11010031






