The Interaction between Laminin-332 and α3β1 Integrin Determines Differentiation and Maintenance of CAFs, and Supports Invasion of Pancreatic Duct Adenocarcinoma Cells
Abstract
1. Introduction
2. Results
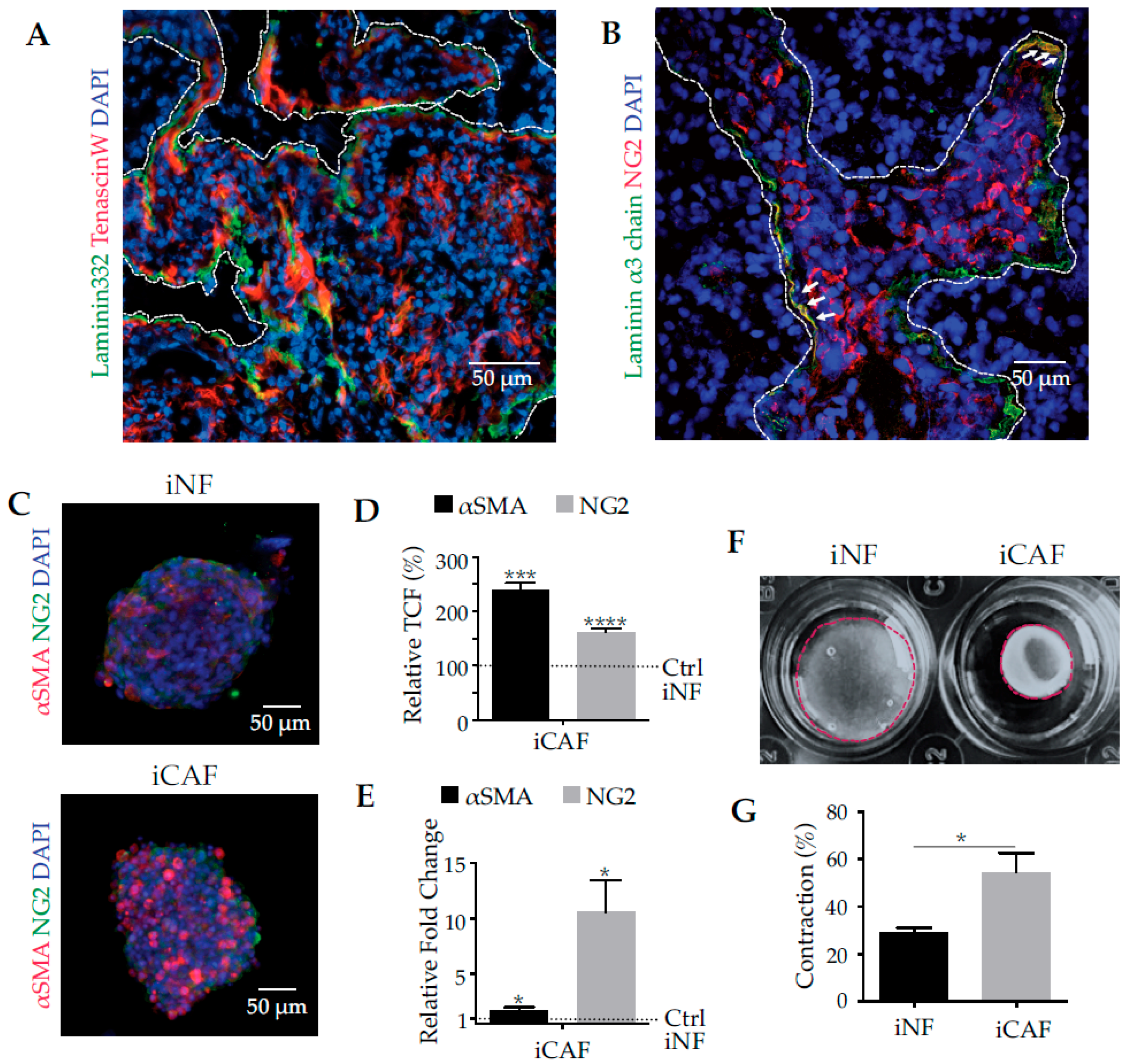
2.1. Pancreatic Tumour Stroma Is Characterized by Ectopic Expression of Laminin-332 and by the Presence of Cancer-Associated Fibroblasts (CAFs)
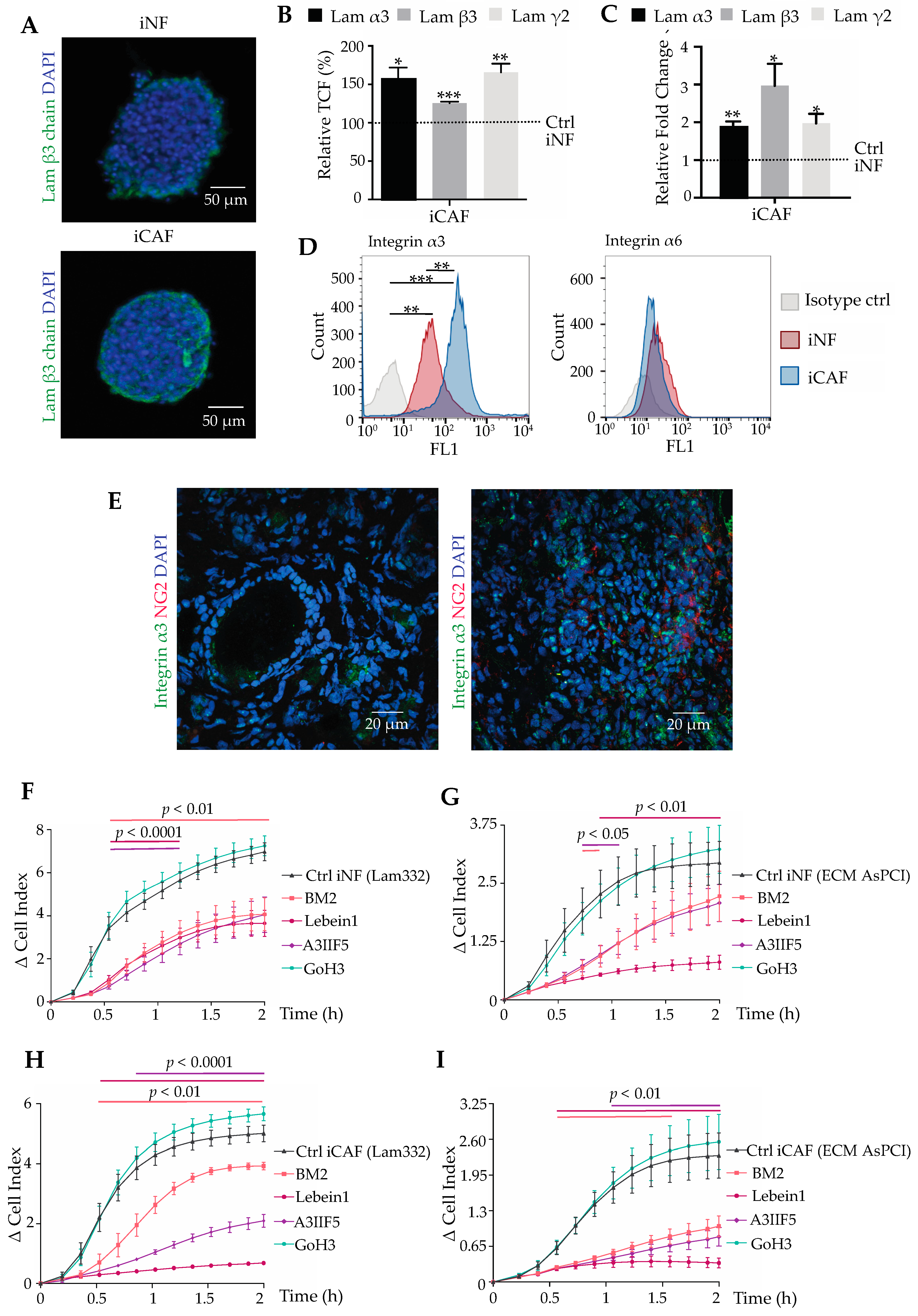
2.2. Comparison of Normal Fibroblasts and CAFs from Pancreatic Tumour Stroma Reveals That Integrin α3β1 and Laminin-332 Are Differentiation Markers
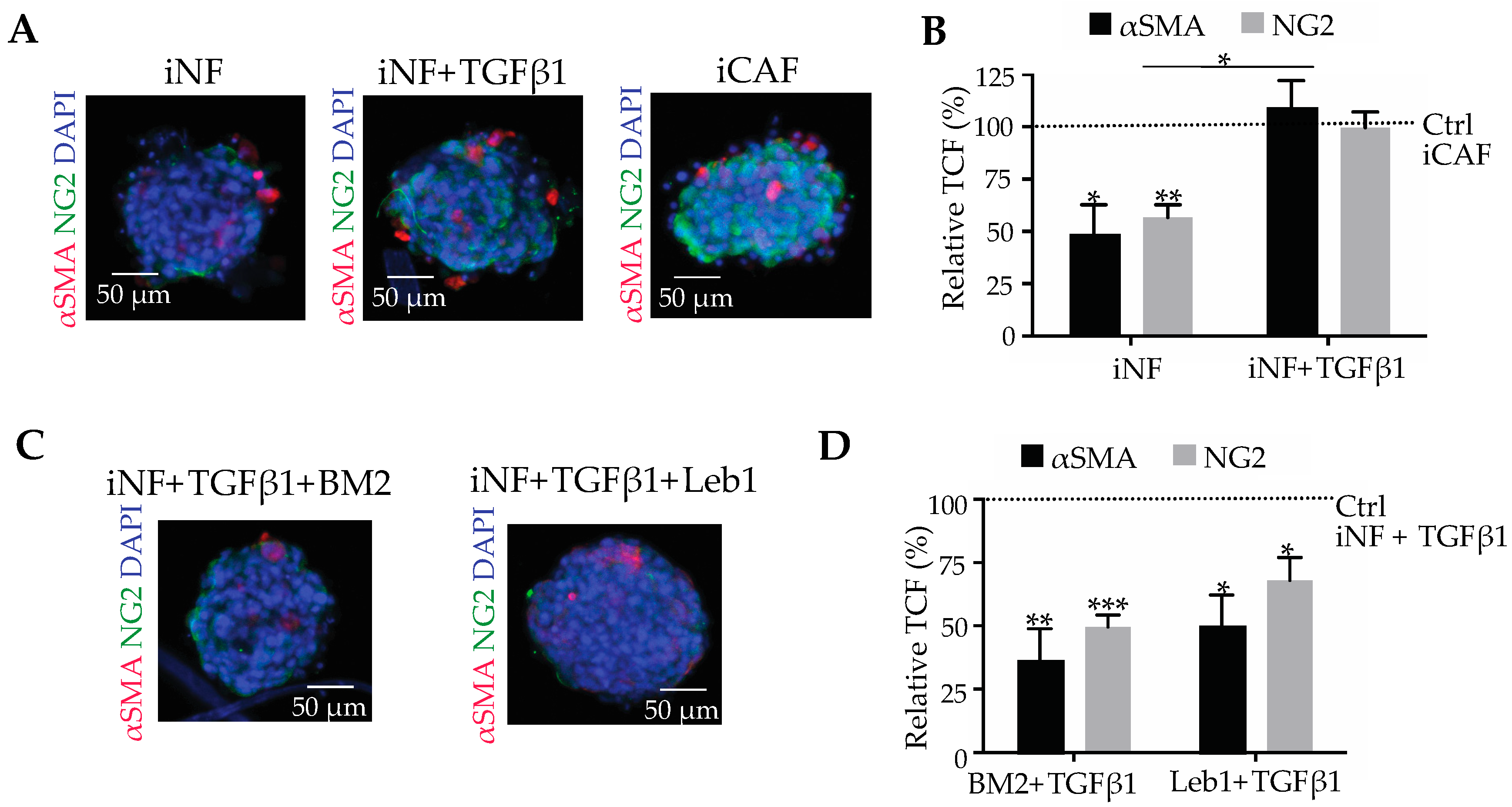
2.3. Stimulation of Pancreatic CAF Differentiation by TGF-β1 Depends on Laminin-332-Integrin Binding
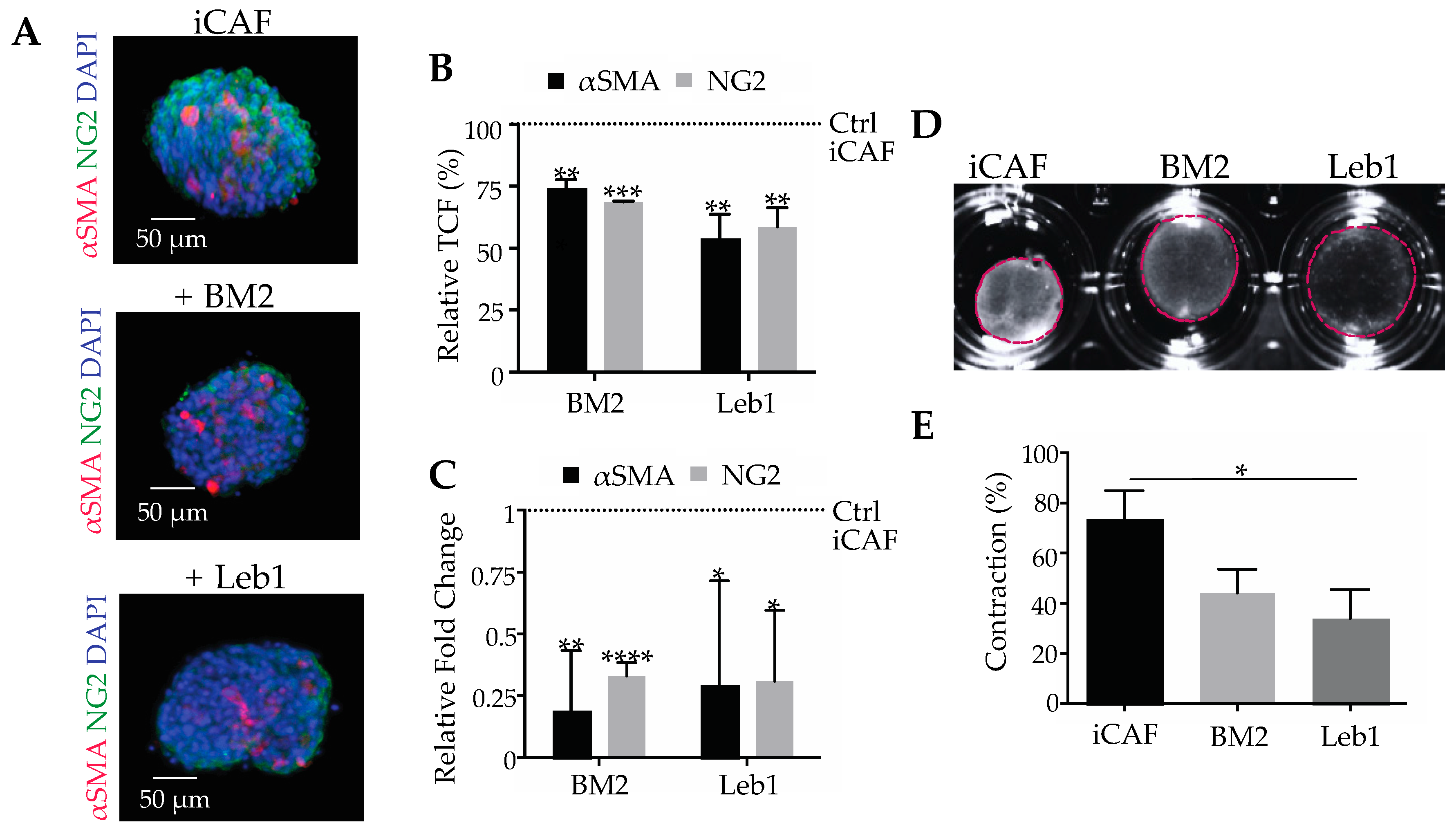
2.4. Laminin-332-Integrin Interaction Sustains the CAF Phenotype
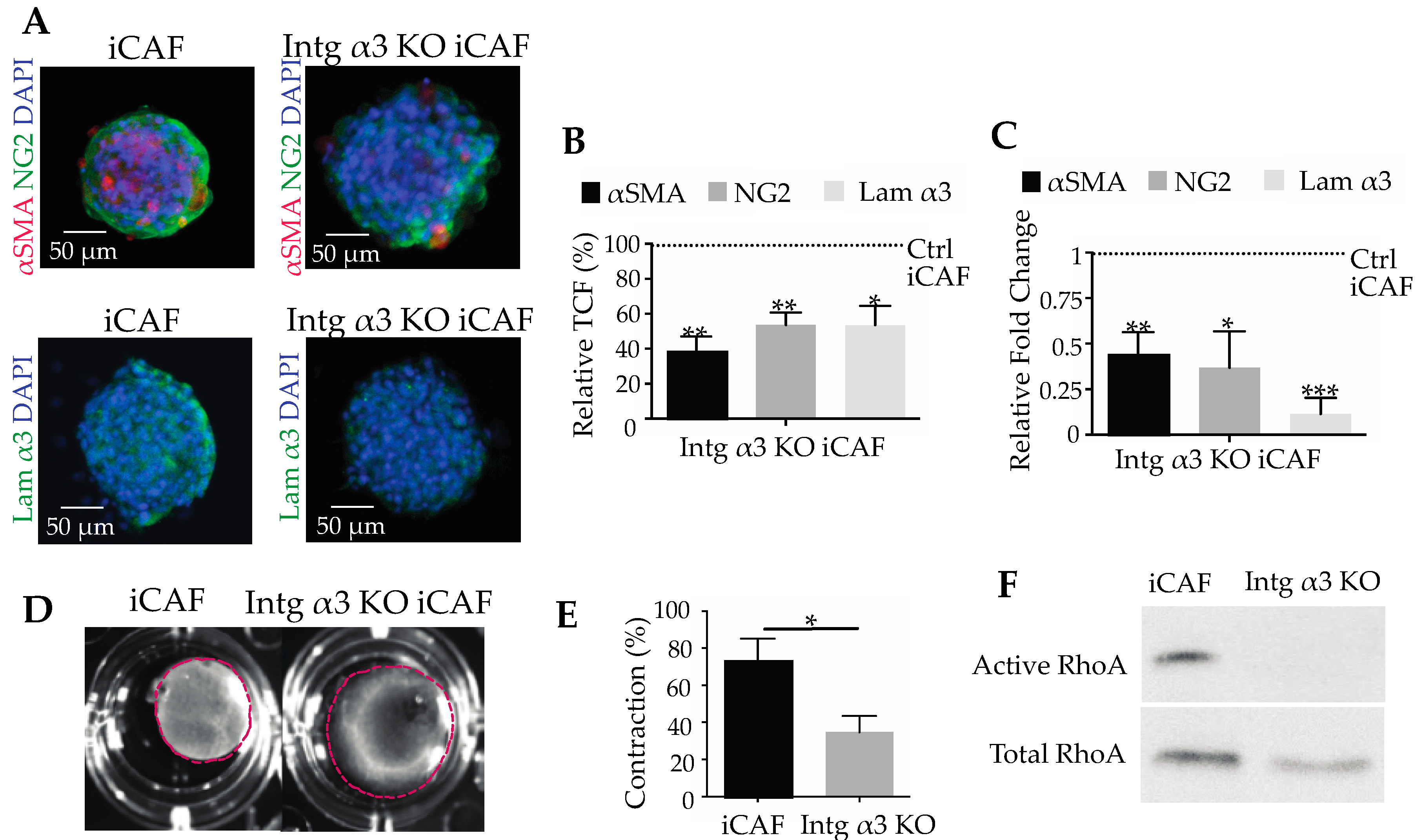
2.5. Interacting with Laminin-332, Integrin α3β1 Is Not Only a Marker, but also a Player on CAF Differentiation
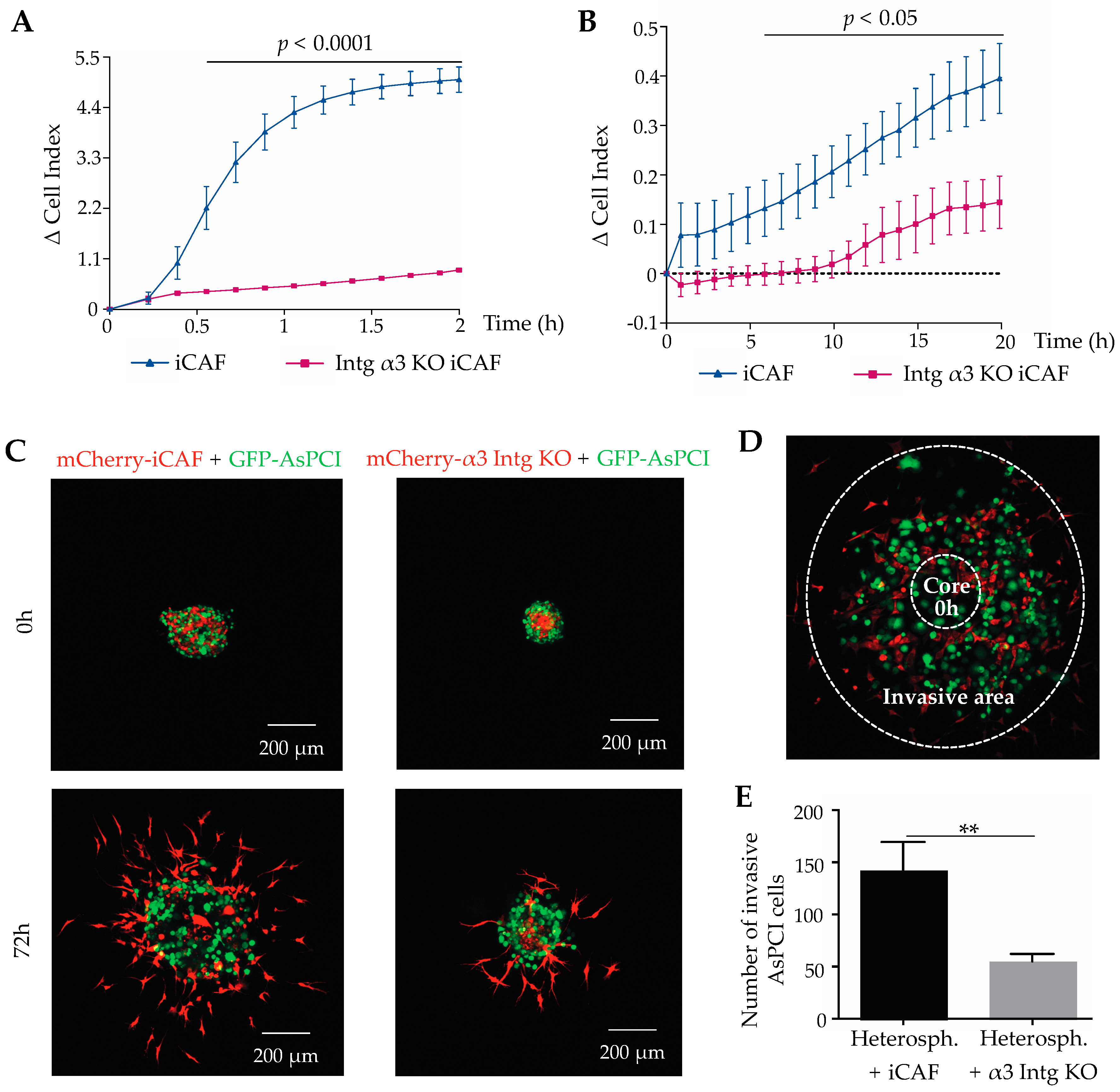
2.6. The Integrin α3β1-Mediated Interaction of CAFs with Laminin-332 Promote Invasion of Pancreatic Cancer Cells
3. Discussion
4. Materials and Methods
4.1. Cell Lines and Pancreatic Adenocarcinoma Sections
4.2. Immortalization of Human Pancreatic Fibroblasts and CAFs
4.3. Immunostaining of Human Pancreatic Adenocarcinoma Sections
4.4. 3D Spheroids as In Vitro Model for Fibroblast Differentiation and Their Immunofluorimetric Analysis
4.5. Quantification of Gene Expression in Homospheroids by RT q-PCR
4.6. Collagen I Gel Contraction Assay
4.7. Flow Cytometry Analysis of Integrin Expression in Human Pancreatic Fibroblasts and CAFs
4.8. Real-Time Adhesion and Migration of Fibroblasts and CAFs Using xCELLigence
4.9. Knocking out the Integrin α3 Subunit in Human Pancreatic CAFs
4.10. Determination of RhoA Activation in CAFs and Integrin α3 KO CAFs
4.11. Invasion Assay
4.12. Immunofluorescence Quantification
4.13. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| αSMA | α-Smooth Muscle Actin |
| CAF | Cancer-Associated Fibroblast |
| ECM | Extracellular Matrix |
| FBS | Fetal Bovine Serum |
| hTERT | Human Telomerase Reverse Transcriptase protein |
| iNF | Immortalized Normal Fibroblast |
| iCAF | Immortalized Cancer-Associated Fibroblast |
| α3KO-iCAF | Integrin α3 subunit Knock-Out immortalized Cancer-Associated Fibroblasts |
| Leb1 | Lebein-1 |
| MEM | Minimal Essential Medium |
| NG2 | Neural/Glial antigen 2 |
| RT | Room Temperature |
| TCF | Total Corrected Fluorescence |
| TCCF | Total Corrected Cell Fluorescence |
| TGF-β1 | Transforming Growth Factor-β1 |
References
- Bardeesy, N.; DePinho, R.A. Pancreatic cancer biology and genetics. Nat. Rev. Cancer 2002, 2, 897–909. [Google Scholar] [CrossRef]
- LeBleu, V.S.; Macdonald, B.; Kalluri, R. Structure and function of basement membranes. Exp. Biol. Med. 2007, 232, 1121–1129. [Google Scholar] [CrossRef]
- Domogatskaya, A.; Rodin, S.; Tryggvason, K. Functional diversity of laminins. Annu. Rev. Cell Dev. Biol. 2012, 28, 523–553. [Google Scholar] [CrossRef]
- McMillan, J.R.; Akiyama, M.; Shimizu, H. Ultrastructural orientation of laminin 5 in the epidermal basement membrane: an updated model for basement membrane organization. J. Histochem. Cytochem. 2003, 51, 1299–1306. [Google Scholar] [CrossRef]
- Miyazaki, K. Laminin-5 (laminin-332): Unique biological activity and role in tumor growth and invasion. Cancer Sci. 2006, 97, 91–98. [Google Scholar] [CrossRef]
- Tani, T.; Lumme, A.; Linnala, A.; Kivilaakso, E.; Kiviluoto, T.; Burgeson, R.E.; Kangas, L.; Leivo, I.; Virtanen, I. Pancreatic carcinomas deposit laminin-5, preferably adhere to laminin-5, and migrate on the newly deposited basement membrane. Am. J. Pathol. 1997, 151, 1289–1302. [Google Scholar]
- Guess, C.M.; Quaranta, V. Defining the role of laminin-332 in carcinoma. Matrix. Biol. 2009, 28, 445–455. [Google Scholar] [CrossRef]
- Rousselle, P.; Beck, K. Laminin 332 processing impacts cellular behavior. Cell Adh. Migr. 2013, 7, 122–134. [Google Scholar] [CrossRef]
- Kunneken, K.; Pohlentz, G.; Schmidt-Hederich, A.; Odenthal, U.; Smyth, N.; Peter-Katalinic, J.; Bruckner, P.; Eble, J.A. Recombinant human laminin-5 domains. Effects of heterotrimerization, proteolytic processing, and N-glycosylation on α3β1 integrin binding. J. Biol. Chem. 2004, 279, 5184–5193. [Google Scholar] [CrossRef]
- Barczyk, M.; Carracedo, S.; Gullberg, D. Integrins. Cell Tissue Res. 2010, 339, 269–280. [Google Scholar] [CrossRef]
- Niessen, C.M.; Hogervorst, F.; Jaspars, L.H.; de Melker, A.A.; Delwel, G.O.; Hulsman, E.H.; Kuikman, I.; Sonnenberg, A. The α6β4 integrin is a receptor for both laminin and kalinin. Exp. Cell Res. 1994, 211, 360–367. [Google Scholar] [CrossRef]
- Eble, J.A.; Wucherpfennig, K.W.; Gauthier, L.; Dersch, P.; Krukonis, E.; Isberg, R.R.; Hemler, M.E. Recombinant soluble human α3β1 integrin: purification, processing, regulation, and specific binding to laminin-5 and invasin in a mutually exclusive manner. Biochemistry 1998, 37, 10945–10955. [Google Scholar] [CrossRef]
- Sachs, N.; Secades, P.; van Hulst, L.; Kreft, M.; Song, J.Y.; Sonnenberg, A. Loss of integrin α3 prevents skin tumor formation by promoting epidermal turnover and depletion of slow-cycling cells. Proc. Natl. Acad. Sci. USA 2012, 109, 21468–21473. [Google Scholar] [CrossRef]
- Frijns, E.; Sachs, N.; Kreft, M.; Wilhelmsen, K.; Sonnenberg, A. EGF-induced MAPK signaling inhibits hemidesmosome formation through phosphorylation of the integrin β4. J. Biol. Chem. 2010, 285, 37650–37662. [Google Scholar] [CrossRef]
- Ramovs, V.; Te Molder, L.; Sonnenberg, A. The opposing roles of laminin-binding integrins in cancer. Matrix Biol. 2017, 57–58, 213–243. [Google Scholar] [CrossRef]
- Eble, J.A.; Bruckner, P.; Mayer, U. Vipera lebetina venom contains two disintegrins inhibiting laminin-binding β1 integrins. J. Biol. Chem. 2003, 278, 26488–26496. [Google Scholar] [CrossRef]
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef]
- Ostman, A.; Augsten, M. Cancer-associated fibroblasts and tumor growth--bystanders turning into key players. Curr. Opin. Genet. Dev. 2009, 19, 67–73. [Google Scholar] [CrossRef]
- Otranto, M.; Sarrazy, V.; Bonte, F.; Hinz, B.; Gabbiani, G.; Desmouliere, A. The role of the myofibroblast in tumor stroma remodeling. Cell Adh. Migr. 2012, 6, 203–219. [Google Scholar] [CrossRef]
- Gaggioli, C. Collective invasion of carcinoma cells: when the fibroblasts take the lead. Cell Adh. Migr. 2008, 2, 45–47. [Google Scholar] [CrossRef]
- Gabbiani, G. The myofibroblast in wound healing and fibrocontractive diseases. J. Pathol. 2003, 200, 500–503. [Google Scholar] [CrossRef]
- Stallcup, W.B. NG2 Proteoglycan Enhances Brain Tumor Progression by Promoting Β-1 Integrin Activation in both Cis and Trans Orientations. Cancers 2017, 9, 31. [Google Scholar] [CrossRef]
- Wells, R.G. The role of matrix stiffness in regulating cell behavior. Hepatology 2008, 47, 1394–1400. [Google Scholar] [CrossRef]
- Keely, P.J. Mechanisms by which the extracellular matrix and integrin signaling act to regulate the switch between tumor suppression and tumor promotion. J. Mammary Gland Biol. Neoplasia 2011, 16, 205–219. [Google Scholar] [CrossRef]
- Cavaco, A.; Rezaei, M.; Niland, S.; Eble, J.A. Collateral Damage Intended-Cancer-Associated Fibroblasts and Vasculature Are Potential Targets in Cancer Therapy. Int. J. Mol. Sci. 2017, 18, 2355. [Google Scholar] [CrossRef]
- Wipff, P.J.; Rifkin, D.B.; Meister, J.J.; Hinz, B. Myofibroblast contraction activates latent TGF-β1 from the extracellular matrix. J. Cell Biol. 2007, 179, 1311–1323. [Google Scholar] [CrossRef]
- Degen, M.; Brellier, F.; Kain, R.; Ruiz, C.; Terracciano, L.; Orend, G.; Chiquet-Ehrismann, R. Tenascin-W is a novel marker for activated tumor stroma in low-grade human breast cancer and influences cell behavior. Cancer Res. 2007, 67, 9169–9179. [Google Scholar] [CrossRef]
- Kim, B.G.; Gao, M.Q.; Choi, Y.P.; Kang, S.; Park, H.R.; Kang, K.S.; Cho, N.H. Invasive breast cancer induces laminin-332 upregulation and integrin β4 neoexpression in myofibroblasts to confer an anoikis-resistant phenotype during tissue remodeling. Breast Cancer Res. 2012, 14, R88. [Google Scholar] [CrossRef]
- Pyke, C.; Salo, S.; Ralfkiaer, E.; Romer, J.; Dano, K.; Tryggvason, K. Laminin-5 is a marker of invading cancer cells in some human carcinomas and is coexpressed with the receptor for urokinase plasminogen activator in budding cancer cells in colon adenocarcinomas. Cancer Res. 1995, 55, 4132–4139. [Google Scholar]
- Katayama, M.; Funakoshi, A.; Sumii, T.; Sanzen, N.; Sekiguchi, K. Laminin γ2-chain fragment circulating level increases in patients with metastatic pancreatic ductal cell adenocarcinomas. Cancer Lett. 2005, 225, 167–176. [Google Scholar] [CrossRef]
- Chen, J.; Wang, W.; Wei, J.; Zhou, D.; Zhao, X.; Song, W.; Sun, Q.; Huang, P.; Zheng, S. Overexpression of β3 Chains of Laminin-332 is Associated With Clinicopathologic Features and Decreased Survival in Patients With Pancreatic Adenocarcinoma. Appl. Immunohistochem. Mol. Morphol. 2015, 23, 516–521. [Google Scholar] [CrossRef]
- Tomasek, J.J.; Gabbiani, G.; Hinz, B.; Chaponnier, C.; Brown, R.A. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat. Rev. Mol. Cell Biol. 2002, 3, 349–363. [Google Scholar] [CrossRef]
- Winer, J.P.; Oake, S.; Janmey, P.A. Non-linear elasticity of extracellular matrices enables contractile cells to communicate local position and orientation. PLoS ONE 2009, 4, e6382. [Google Scholar] [CrossRef]
- Yeung, T.; Georges, P.C.; Flanagan, L.A.; Marg, B.; Ortiz, M.; Funaki, M.; Zahir, N.; Ming, W.; Weaver, V.; Janmey, P.A. Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion. Cell Motil. Cytoskeleton 2005, 60, 24–34. [Google Scholar] [CrossRef]
- Gehler, S.; Ponik, S.M.; Riching, K.M.; Keely, P.J. Bi-directional signaling: Extracellular matrix and integrin regulation of breast tumor progression. Crit. Rev. Eukaryot. Gene Expr. 2013, 23, 139–157. [Google Scholar] [CrossRef]
- Arora, P.D.; Narani, N.; McCulloch, C.A. The compliance of collagen gels regulates transforming growth factor-β induction of α-smooth muscle actin in fibroblasts. Am. J. Pathol. 1999, 154, 871–882. [Google Scholar] [CrossRef]
- Lin, R.Z.; Chang, H.Y. Recent advances in three-dimensional multicellular spheroid culture for biomedical research. Biotechnol. J. 2008, 3, 1172–1184. [Google Scholar] [CrossRef]
- Mehta, G.; Hsiao, A.Y.; Ingram, M.; Luker, G.D.; Takayama, S. Opportunities and challenges for use of tumor spheroids as models to test drug delivery and efficacy. J. Control Release 2012, 164, 192–204. [Google Scholar] [CrossRef]
- Morales, C.P.; Holt, S.E.; Ouellette, M.; Kaur, K.J.; Yan, Y.; Wilson, K.S.; White, M.A.; Wright, W.E.; Shay, J.W. Absence of cancer-associated changes in human fibroblasts immortalized with telomerase. Nat. Genet. 1999, 21, 115–118. [Google Scholar] [CrossRef]
- Mizushima, H.; Koshikawa, N.; Moriyama, K.; Takamura, H.; Nagashima, Y.; Hirahara, F.; Miyazaki, K. Wide distribution of laminin-5 γ 2 chain in basement membranes of various human tissues. Horm. Res. 1998, 50 (Suppl. 2), 7–14. [Google Scholar] [CrossRef]
- Rousselle, P.; Lunstrum, G.P.; Keene, D.R.; Burgeson, R.E. Kalinin: an epithelium-specific basement membrane adhesion molecule that is a component of anchoring filaments. J. Cell Biol. 1991, 114, 567–576. [Google Scholar] [CrossRef]
- Marinkovich, M.P. Tumour microenvironment: laminin 332 in squamous-cell carcinoma. Nat. Rev. Cancer 2007, 7, 370–380. [Google Scholar] [CrossRef]
- Hawinkels, L.J.; Paauwe, M.; Verspaget, H.W.; Wiercinska, E.; Van der Zon, J.M.; Van der Ploeg, K.; Koelink, P.J.; Lindeman, J.H.; Mesker, W.; Ten Dijke, P.; et al. Interaction with colon cancer cells hyperactivates TGF-β signaling in cancer-associated fibroblasts. Oncogene 2014, 33, 97–107. [Google Scholar] [CrossRef]
- Gagnoux-Palacios, L.; Allegra, M.; Spirito, F.; Pommeret, O.; Romero, C.; Ortonne, J.P.; Meneguzzi, G. The short arm of the laminin γ2 chain plays a pivotal role in the incorporation of laminin 5 into the extracellular matrix and in cell adhesion. J. Cell Biol. 2001, 153, 835–850. [Google Scholar] [CrossRef]
- Hendrix, M.J.; Seftor, E.A.; Kirschmann, D.A.; Quaranta, V.; Seftor, R.E. Remodeling of the microenvironment by aggressive melanoma tumor cells. Ann. N. Y. Acad. Sci. 2003, 995, 151–161. [Google Scholar] [CrossRef]
- Okamoto, O.; Bachy, S.; Odenthal, U.; Bernaud, J.; Rigal, D.; Lortat-Jacob, H.; Smyth, N.; Rousselle, P. Normal human keratinocytes bind to the α3LG4/5 domain of unprocessed laminin-5 through the receptor syndecan-1. J. Biol. Chem. 2003, 278, 44168–44177. [Google Scholar] [CrossRef]
- Decline, F.; Rousselle, P. Keratinocyte migration requires α2β1 integrin-mediated interaction with the laminin 5 γ2 chain. J. Cell Sci. 2001, 114, 811–823. [Google Scholar]
- Gaggioli, C.; Hooper, S.; Hidalgo-Carcedo, C.; Grosse, R.; Marshall, J.F.; Harrington, K.; Sahai, E. Fibroblast-led collective invasion of carcinoma cells with differing roles for RhoGTPases in leading and following cells. Nat. Cell Biol. 2007, 9, 1392–1400. [Google Scholar] [CrossRef]
- Zhang, A.; Qian, Y.; Ye, Z.; Chen, H.; Xie, H.; Zhou, L.; Shen, Y.; Zheng, S. Cancer-associated fibroblasts promote M2 polarization of macrophages in pancreatic ductal adenocarcinoma. Cancer Med. 2017, 6, 463–470. [Google Scholar] [CrossRef]
- Goldberg, M.T.; Han, Y.P.; Yan, C.; Shaw, M.C.; Garner, W.L. TNF-α suppresses α-smooth muscle actin expression in human dermal fibroblasts: an implication for abnormal wound healing. J. Investig. Dermatol. 2007, 127, 2645–2655. [Google Scholar] [CrossRef]
- Kim, B.G.; An, H.J.; Kang, S.; Choi, Y.P.; Gao, M.Q.; Park, H.; Cho, N.H. Laminin-332-rich tumor microenvironment for tumor invasion in the interface zone of breast cancer. Am. J. Pathol. 2011, 178, 373–381. [Google Scholar] [CrossRef]
- Huang, C.C.; Chen, D.Y.; Wei, H.J.; Lin, K.J.; Wu, C.T.; Lee, T.Y.; Hu, H.Y.; Hwang, S.M.; Chang, Y.; Sung, H.W. Hypoxia-induced therapeutic neovascularization in a mouse model of an ischemic limb using cell aggregates composed of HUVECs and cbMSCs. Biomaterials 2013, 34, 9441–9450. [Google Scholar] [CrossRef]
- Makizumi, R.; Yang, W.L.; Owen, R.P.; Sharma, R.R.; Ravikumar, T.S. Alteration of Drug Sensitivity in Human Colon Cancer Cells after Exposure to Heat: Implications for Liver Metastasis Therapy using RFA and Chemotherapy. Int. J. Clin. Exp. Med. 2008, 1, 117–129. [Google Scholar]
- Xi, B.; Yu, N.; Wang, X.; Xu, X.; Abassi, Y.A. The application of cell-based label-free technology in drug discovery. Biotechnol. J. 2008, 3, 484–495. [Google Scholar] [CrossRef]
- Ran, F.A.; Hsu, P.D.; Wright, J.; Agarwala, V.; Scott, D.A.; Zhang, F. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 2013, 8, 2281–2308. [Google Scholar] [CrossRef]
- Ansari, N.; Muller, S.; Stelzer, E.H.; Pampaloni, F. Quantitative 3D cell-based assay performed with cellular spheroids and fluorescence microscopy. Methods Cell Biol. 2013, 113, 295–309. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins Cavaco, A.C.; Rezaei, M.; Caliandro, M.F.; Martins Lima, A.; Stehling, M.; Dhayat, S.A.; Haier, J.; Brakebusch, C.; Eble, J.A. The Interaction between Laminin-332 and α3β1 Integrin Determines Differentiation and Maintenance of CAFs, and Supports Invasion of Pancreatic Duct Adenocarcinoma Cells. Cancers 2019, 11, 14. https://doi.org/10.3390/cancers11010014
Martins Cavaco AC, Rezaei M, Caliandro MF, Martins Lima A, Stehling M, Dhayat SA, Haier J, Brakebusch C, Eble JA. The Interaction between Laminin-332 and α3β1 Integrin Determines Differentiation and Maintenance of CAFs, and Supports Invasion of Pancreatic Duct Adenocarcinoma Cells. Cancers. 2019; 11(1):14. https://doi.org/10.3390/cancers11010014
Chicago/Turabian StyleMartins Cavaco, Ana C., Maryam Rezaei, Michele F. Caliandro, Augusto Martins Lima, Martin Stehling, Sameer A. Dhayat, Jörg Haier, Cord Brakebusch, and Johannes A. Eble. 2019. "The Interaction between Laminin-332 and α3β1 Integrin Determines Differentiation and Maintenance of CAFs, and Supports Invasion of Pancreatic Duct Adenocarcinoma Cells" Cancers 11, no. 1: 14. https://doi.org/10.3390/cancers11010014
APA StyleMartins Cavaco, A. C., Rezaei, M., Caliandro, M. F., Martins Lima, A., Stehling, M., Dhayat, S. A., Haier, J., Brakebusch, C., & Eble, J. A. (2019). The Interaction between Laminin-332 and α3β1 Integrin Determines Differentiation and Maintenance of CAFs, and Supports Invasion of Pancreatic Duct Adenocarcinoma Cells. Cancers, 11(1), 14. https://doi.org/10.3390/cancers11010014






