With Great Age Comes Great Metastatic Ability: Ovarian Cancer and the Appeal of the Aging Peritoneal Microenvironment
Abstract
1. Introduction
2. Aging Modifies the Metastatic Microenvironment
2.1. Mesothelial Cells
2.2. Extracellular Matrix
2.2.1. Collagen
2.2.2. Fibronectin
2.2.3. Basement Membrane
2.3. Fibroblasts
2.3.1. Senescent Fibroblasts
2.3.2. Fibroblast Activation
2.4. Immune Cells
2.4.1. Tumor Cells Preferentially Adhere to Immune Cell Clusters
2.4.2. Aging Affects Antitumor Macrophage Function in Peritoneum
2.4.3. Tumor Infiltrating Lymphocytes: B and T cells
2.5. Adipocytes
3. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Howlader, N.; Noore, A.M.; Krapcho, M.; Miller, D.; Bishop, K.; Kosary, C.L.; Yu, M.; Rahl, J.; Tatalovich, Z.; Mariotto, A.; et al. Seer Cancer Statistics Review, 1975–2014; National Cancer Institute: Bethesda, MD, USA, 2017. [Google Scholar]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [PubMed]
- Yancik, R. Ovarian cancer: Age contrasts in incidence, histology, disease stage at diagnosis, and mortality. Cancer 1993, 71, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Trillsch, F.; Woelber, L.; Eulenburg, C.; Braicu, I.; Lambrechts, S.; Chekerov, R.; van Nieuwenhuysen, E.; Speiser, P.; Zeimet, A.; Castillo-Tong, D.C.; et al. Treatment reality in elderly patients with advanced ovarian cancer: A prospective analysis of the ovcad consortium. J. Ovarian Res. 2013, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Mikuła-Pietrasik, J.; Uruski, P.; Sosińska, P.; Maksin, K.; Piotrowska-Kempisty, H.; Kucińska, M.; Murias, M.; Szubert, S.; Woźniak, A.; Szpurek, D.; et al. Senescent peritoneal mesothelium creates a niche for ovarian cancer metastases. Cell Death Dis. 2016, 7, e2565. [Google Scholar] [CrossRef] [PubMed]
- Kenny, H.A.; Krausz, T.; Yamada, S.D.; Lengyel, E. Use of a novel 3d culture model to elucidate the role of mesothelial cells, fibroblasts and extra-cellular matrices on adhesion and invasion of ovarian cancer cells to the omentum. Int. J. Cancer 2007, 121, 1463–1472. [Google Scholar] [CrossRef] [PubMed]
- Van Baal, J.O.A.M.; Van de Vijver, K.K.; Nieuwland, R.; van Noorden, C.J.F.; van Driel, W.J.; Sturk, A.; Kenter, G.G.; Rikkert, L.G.; Lok, C.A.R. The histophysiology and pathophysiology of the peritoneum. Tissue Cell 2017, 49, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Park, M.S.; Lee, H.A.; Chu, W.S.; Yang, D.H.; Hwang, S.D. Peritoneal accumulation of age and peritoneal membrane permeability. Perit. Dial. Int. 2000, 20, 452–460. [Google Scholar] [PubMed]
- Ulrich, T.A.; Pardo, E.M.D.J.; Kumar, S. The mechanical rigidity of the extracellular matrix regulates the structure, motility, and proliferation of glioma cells. Cancer Res. 2009, 69, 4167–4174. [Google Scholar] [CrossRef] [PubMed]
- Wells, R.G. The role of matrix stiffness in regulating cell behavior. Hepatology 2008, 47, 1394–1400. [Google Scholar] [CrossRef] [PubMed]
- Sprenger, C.C.; Plymate, S.R.; Reed, M.J. Aging-related alterations in the extracellular matrix modulate the microenvironment and influence tumor progression. Int. J. Cancer 2010, 127, 2739–2748. [Google Scholar] [CrossRef] [PubMed]
- Eyman, D.; Damodarasamy, M.; Plymate, S.R.; Reed, M.J. Ccl5 secreted by senescent aged fibroblasts induces proliferation of prostate epithelial cells and expression of genes that modulate angiogenesis. J. Cell. Physiol. 2009, 220, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Hayflick, L. Mortality and immortality at the cellular level. A review. Biochem. Mosc. 1997, 62, 1180–1190. [Google Scholar]
- Ksiazek, K.; Passos, J.F.; Olijslagers, S.; Saretzki, G.; Martin-Ruiz, C.; von Zglinicki, T. Premature senescence of mesothelial cells is associated with non-telomeric DNA damage. Biochem. Biophys. Res. Commun. 2007, 362, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Di Fagagna, F.D.A.; Reaper, P.M.; Clay-Farrace, L.; Fiegler, H.; Carr, P.; von Zglinicki, T.; Saretzki, G.; Carter, N.P.; Jackson, S.P. A DNA damage checkpoint response in telomere-initiated senescence. Nature 2003, 426, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Ksiazek, K.; Mikula-Pietrasik, J.; Korybalska, K.; Dworacki, G.; Jörres, A.; Witowski, J. Senescent peritoneal mesothelial cells promote ovarian cancer cell adhesion: The role of oxidative stress-induced fibronectin. Am. J. Pathol. 2009, 174, 1230–1240. [Google Scholar] [CrossRef] [PubMed]
- Ksiazek, K.; Passos, J.F.; Olijslagers, S.; von Zglinicki, T. Mitochondrial dysfunction is a possible cause of accelerated senescence of mesothelial cells exposed to high glucose. Biochem. Biophys. Res. Commun. 2008, 366, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Książek, K.; Korybalska, K.; Jörres, A.; Witowski, J. Accelerated senescence of human peritoneal mesothelial cells exposed to high glucose: The role of tgf-β1. Lab. Investig. 2007, 87, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Blazer, S.; Khankin, E.; Segev, Y.; Ofir, R.; Yalon-Hacohen, M.; Kra-Oz, Z.; Gottfried, Y.; Larisch, S.; Skorecki, K.L. High glucose-induced replicative senescence: Point of no return and effect of telomerase. Biochem. Biophys. Res. Commun. 2002, 296, 93–101. [Google Scholar] [CrossRef]
- Margetts, P.J.; Bonniaud, P.; Liu, L.; Hoff, C.M.; Holmes, C.J.; West-Mays, J.A.; Kelly, M.M. Transient overexpression of tgf-β1 induces epithelial mesenchymal transition in the rodent peritoneum. J. Am. Soc. Nephrol. 2005, 16, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Debacq-Chainiaux, F.; Borlon, C.; Pascal, T.; Royer, V.; Eliaers, F.; Ninane, N.; Carrard, G.; Friguet, B.; de Longueville, F.; Boffe, S.; et al. Repeated exposure of human skin fibroblasts to uvb at subcytotoxic level triggers premature senescence through the tgf-β1 signaling pathway. J. Cell Sci. 2005, 118, 743–758. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-H.; Park, G.-T.; Lim, Y.-B.; Rue, S.-W.; Jung, J.-C.; Sonn, J.-K.; Bae, Y.-S.; Park, J.-W.; Lee, Y.-S. Expression of connective tissue growth factor, a biomarker in senescence of human diploid fibroblasts, is up-regulated by a transforming growth factor-β-mediated signaling pathway. Biochem. Biophys. Res. Commun. 2004, 318, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Collado, M.; Gil, J.; Efeyan, A.; Guerra, C.; Schuhmacher, A.J.; Barradas, M.; Benguría, A.; Zaballos, A.; Flores, J.M.; Barbacid, M.; et al. Tumour biology: Senescence in premalignant tumours. Nature 2005, 436, 642. [Google Scholar] [CrossRef] [PubMed]
- Lawrenson, K.; Grun, B.; Benjamin, E.; Jacobs, I.J.; Dafou, D.; Gayther, S.A. Senescent fibroblasts promote neoplastic transformation of partially transformed ovarian epithelial cells in a three-dimensional model of early stage ovarian cancer. Neoplasia 2010, 12, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Krtolica, A.; Campisi, J. Cancer and aging: A model for the cancer promoting effects of the aging stroma. Int. J. Biochem. Biol. 2002, 34, 1401–1414. [Google Scholar] [CrossRef]
- Loughran, E.A.; Leonard, A.K.; Hilliard, T.S.; Phan, R.C.; Yemc, M.G.; Harper, E.; Sheedy, E.; Klymenko, Y.; Asem, M.; Liu, Y.; et al. Aging increases susceptibility to ovarian cancer metastasis in murine allograft models and alters immune composition of peritoneal adipose tissue. Neoplasia 2018, 20, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Sastre, J. The role of mitochondrial oxidative stress in aging. Free Radical Biol. Med. 2003, 35, 1–8. [Google Scholar] [CrossRef]
- Rodier, F.; Campisi, J.; Bhaumik, D. Two faces of p53: Aging and tumor suppression. Nucleic Acids Res. 2007, 35, 7475–7484. [Google Scholar] [CrossRef] [PubMed]
- Donehower, L.A. Using mice to examine p53 functions in cancer, aging, and longevity. Cold Spring Harb. Perspect. Biol. 2009, 1, a001081. [Google Scholar] [CrossRef] [PubMed]
- Yáñez-Mó, M.; Lara-Pezzi, E.; Selgas, R.; Ramírez-Huesca, M.; Domínguez-Jiménez, C.; Jiménez-Heffernan, J.A.; Aguilera, A.; Sánchez-Tomero, J.A.; Bajo, M.A.; Álvarez, V.; et al. Peritoneal dialysis and epithelial-to-mesenchymal transition of mesothelial cells. N. Engl. J. Med. 2003, 348, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Dimri, G.P.; Lee, X.; Basile, G.; Acosta, M.; Scott, G.; Roskelley, C.; Medrano, E.E.; Linskens, M.; Rubelj, I.; Pereira-Smith, O. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl. Acad. Sci. USA 1995, 92, 9363–9367. [Google Scholar] [CrossRef] [PubMed]
- Fodil-Bourahla, I.; Drubaix, I.; Robert, L. Effect of in vitro aging on the biosynthesis of glycosaminoglycans by human skin fibroblasts. Modulation by the elastin-laminin receptor. Mech. Ageing Dev. 1999, 106, 241–260. [Google Scholar] [CrossRef]
- Ksia̧żek, K.; Mikuła-Pietrasik, J.; Catar, R.; Dworacki, G.; Winckiewicz, M.; Frydrychowicz, M.; Dragun, D.; Staniszewski, R.; Jörres, A.; Witowski, J. Oxidative stress-dependent increase in icam-1 expression promotes adhesion of colorectal and pancreatic cancers to the senescent peritoneal mesothelium. Int. J. Cancer 2010, 127, 293–303. [Google Scholar] [PubMed]
- Debacq-Chainiaux, F.; Erusalimsky, J.D.; Campisi, J.; Toussaint, O. Protocols to detect senescence-associated beta-galactosidase (sa-βgal) activity, a biomarker of senescent cells in culture and in vivo. Nat. Protoc. 2009, 4, 1798–1806. [Google Scholar] [CrossRef] [PubMed]
- Vasile, E.; Tomita, Y.; Brown, L.F.; Kocher, O.; Dvorak, H.F. Differential expression of thymosin β-10 by early passage and senescent vascular endothelium is modulated by vpf/vegf: Evidence for senescent endothelial cells in vivo at sites of atherosclerosis. FASEB J. 2001, 15, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Labat-Robert, J. Fibronectin in malignancy. Semin. Cancer Biol. 2002, 12, 187–195. [Google Scholar] [CrossRef]
- Antia, M.; Baneyx, G.E.; Kubow, K.; Vogel, V. Fibronectin in aging extracellular matrix fibrils is progressively unfolded by cells and elicits an enhanced rigidity response. Faraday Discuss. 2008, 139, 229–249. [Google Scholar] [CrossRef] [PubMed]
- Nevado, J.; Vallejo, S.; El-Assar, M.; Peiró, C.; Sánchez-Ferrer, C.F.; Rodríguez-Mañas, L. Changes in the human peritoneal mesothelial cells during aging. Kidney Int. 2006, 69, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Colavitti, R.; Finkel, T. Reactive oxygen species as mediators of cellular senescence. IUBMB Life 2008, 57, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Mikuła-Pietrasik, J.; Sosińska, P.; Naumowicz, E.; Maksin, K.; Piotrowska, H.; Woźniak, A.; Szpurek, D.; Książek, K. Senescent peritoneal mesothelium induces a pro-angiogenic phenotype in ovarian cancer cells in vitro and in a mouse xenograft model in vivo. Clin. Exp. Metastasis 2016, 33, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Robert, L.; Robert, A.M.; Renard, G. Biological effects of hyaluronan in connective tissues, eye, skin, venous wall. Role in aging. Pathol. Biol. 2010, 58, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Young, V.J.; Brown, J.K.; Saunders, P.T.; Horne, A.W. The role of the peritoneum in the pathogenesis of endometriosis. Hum. Reprod. Update 2013, 19, 558–569. [Google Scholar] [CrossRef] [PubMed]
- Gallorini, M.; Berardi, A.C.; Berardocco, M.; Gissi, C.; Maffulli, N.; Cataldi, A.; Oliva, F. Hyaluronic acid increases tendon derived cell viability and proliferation in vitro: Comparative study of two different hyaluronic acid preparations by molecular weight. Muscles Ligaments Tendons J. 2017, 7, 208–214. [Google Scholar] [PubMed]
- Landau, M.; Fagien, S. Science of hyaluronic acid beyond filling: Fibroblasts and their response to the extracellular matrix. Plast. Reconstr. Surg. 2015, 136, 188S–195S. [Google Scholar] [CrossRef] [PubMed]
- Simpson, M.A.; Reiland, J.; Burger, S.R.; Furcht, L.T.; Spicer, A.P.; Oegema, T.R.; McCarthy, J.B. Hyaluronan synthase elevation in metastatic prostate carcinoma cells correlates with hyaluronan surface retention, a prerequisite for rapid adhesion to bone marrow endothelial cells. J. Biol. Chem. 2001, 276, 17949–17957. [Google Scholar] [CrossRef] [PubMed]
- Gardner, M.J.; Catterall, J.B.; Jones, L.M.H.; Turner, G.A. Human ovarian tumour cells can bind hyaluronic acid via membrane cd44: A possible step in peritoneal metastasis. Clin. Exp. Metastasis 1996, 14, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Dechaud, H.; Witz, C.A.; Montoya-Rodriguez, I.A.; Degraffenreid, L.A.; Schenken, R.S. Mesothelial cell–associated hyaluronic acid promotes adhesion of endometrial cells to mesothelium. Fert. Steril. 2001, 76, 1012–1018. [Google Scholar] [CrossRef]
- Jones, L.M.H.; Gardner, M.J.; Catterall, J.B.; Turner, G.A. Hyaluronic acid secreted by mesothelial cells: A natural barrier to ovarian cancer cell adhesion. Clin. Exp. Metastasis 1995, 13, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Wiig, M.; Abrahamsson, S.O.; Lundborg, G. Effects of hyaluronan on cell proliferation and collagen synthesis: A study of rabbit flexor tendons in vitro. J. Hand Surg. Am. 1996, 21, 599–604. [Google Scholar] [CrossRef]
- Klein, E.S.; Asculai, S.S.; Ben-Ari, G.Y. Effects of hyaluronic acid on fibroblast behavior in peritoneal injury. J. Surg. Res. 1996, 61, 473–476. [Google Scholar] [CrossRef] [PubMed]
- Anttila, M.A.; Tammi, R.H.; Tammi, M.I.; Syrjänen, K.J.; Saarikoski, S.V.; Kosma, V.-M. High levels of stromal hyaluronan predict poor disease outcome in epithelial ovarian cancer. Cancer Res. 2000, 60, 150–155. [Google Scholar] [PubMed]
- Hall, C.L.; Wang, C.; Lange, L.A.; Turley, E.A. Hyaluronan and the hyaluronan receptor rhamm promote focal adhesion turnover and transient tyrosine kinase activity. J. Cell Biol. 1994, 126, 575–588. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.Y.; Grant, M.E.; Schor, A.M.; Schor, S.L. Differences between adult and foetal fibroblasts in the regulation of hyaluronate synthesis: Correlation with migratory activity. J. Cell Sci. 1989, 94 Pt 3, 577–584. [Google Scholar] [PubMed]
- Melrose, J.; Numata, Y.; Ghosh, P. Biotinylated hyaluronan: A versatile and highly sensitive probe capable of detecting nanogram levels of hyaluronan binding proteins (hyaladherins) on electroblots by a novel affinity detection procedure. Electrophoresis 1996, 17, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Matuoka, K.; Namba, M.; Mitsui, Y. Hyaluronate synthetase inhibition by normal and transformed human fibroblasts during growth reduction. J. Cell Biol. 1987, 104, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Yeo, T.K.; Nagy, J.A.; Yeo, K.T.; Dvorak, H.F.; Toole, B.P. Increased hyaluronan at sites of attachment to mesentery by cd44-positive mouse ovarian and breast tumor cells. Am. J. Pathol. 1996, 148, 1733–1740. [Google Scholar] [PubMed]
- Bernard, E. Effect of hyaluronan on the elastase-type activity of human skin fibroblasts. Cell Biol. Int. 1994, 18, 967–972. [Google Scholar] [CrossRef] [PubMed]
- Breborowicz, A.; Polubinska, A.; Moberly, J.; Ogle, K.; Martis, L.; Oreopoulos, D. Hyaluronan modifies inflammatory response and peritoneal permeability during peritonitis in rats. Am. J. Kidney Dis. 2001, 37, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Terazawa, S.; Nakajima, H.; Tobita, K.; Imokawa, G. The decreased secretion of hyaluronan by older human fibroblasts under physiological conditions is mainly associated with the down-regulated expression of hyaluronan synthases but not with the expression levels of hyaluronidases. Cytotechnology 2015, 67, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Witz, C.A.; Montoya-Rodriguez, I.A.; Cho, S.; Centonze, V.E.; Bonewald, L.F.; Schenken, R.S. Composition of the extracellular matrix of the peritoneum. J. Soc. Gynecol. Investig. 2001, 8, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Iiyama, M.; Shimada, Y.; Kita, T.; Ito, H. Effect of aging on macrophage adherence to extracellular matrix proteins. Mech. Ageing Dev. 1992, 66, 149–158. [Google Scholar] [CrossRef]
- Sharaf, H.; Matou-Nasri, S.; Wang, Q.; Rabhan, Z.; Al-Eidi, H.; Al Abdulrahman, A.; Ahmed, N. Advanced glycation endproducts increase proliferation, migration and invasion of the breast cancer cell line mda-mb-231. BBA-Mol. Basis Dis. 2015, 1852, 429–441. [Google Scholar] [CrossRef] [PubMed]
- Lodish, H.; Berk, A.; Zipursky, S.L.; Matsudaira, P.; Baltimore, D.; Darnell, J. Collagen: The fibrous proteins of the matrix. Mol. Cell Biol. 2000, 4. [Google Scholar]
- Shuster, S.; Black, M.M.; McVitie, E. The influence of age and sex on skin thickness, skin collagen and density. Br. J. Dermatol. 1975, 93, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Rrong, Y.; Ning, F.; Zhang, G. The content and ratio of type i and iii collagen in skin differ with age and injury. Afr. J. Biotechnol. 2011, 10, 2524–2529. [Google Scholar]
- Damodarasamy, M.; Vernon, R.B.; Karres, N.; Chang, C.H.; Bianchi-Frias, D.; Nelson, P.S.; Reed, M.J. Collagen extracts derived from young and aged mice demonstrate different structural properties and cellular effects in three-dimensional gels. J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65A, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Bianchi-Frias, D.; Vakar-Lopez, F.; Coleman, I.M.; Plymate, S.R.; Reed, M.J.; Nelson, P.S. The effects of aging on the molecular and cellular composition of the prostate microenvironment. PLoS ONE 2010, 5, e12501. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Balimunkwe, R.M.; Quan, T. Age-related reduction of dermal fibroblast size upregulates multiple matrix metalloproteinases as observed in aged human skin in vivo. Brit. J. Dermatol. 2017, 177, 1337–1348. [Google Scholar] [CrossRef] [PubMed]
- Jabłońska-Trypuć, A.; Matejczyk, M.; Rosochacki, S. Matrix metalloproteinases (mmps), the main extracellular matrix (ecm) enzymes in collagen degradation, as a target for anticancer drugs. J. Enzym. Inhib. Med. Chem. 2016, 31, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Jacob, M.P. Extracellular matrix remodeling and matrix metalloproteinases in the vascular wall during aging and in pathological conditions. Biomed. Pharmacother. 2003, 57, 195–202. [Google Scholar] [CrossRef]
- Kamat, A.A.; Fletcher, M.; Gruman, L.M.; Mueller, P.; Lopez, A.; Landen, C.N.; Han, L.; Gershenson, D.M.; Sood, A.K. The clinical relevance of stromal matrix metalloproteinase expression in ovarian cancer. Clin. Cancer Res. 2006, 12, 1707–1714. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Hornsby, P.J. Senescent human fibroblasts increase the early growth of xenograft tumors via matrix metalloproteinase secretion. Cancer Res. 2007, 67, 3117–3126. [Google Scholar] [CrossRef] [PubMed]
- Verzijl, N.; DeGroot, J.; Thorpe, S.R.; Bank, R.A.; Shaw, J.N.; Lyons, T.J.; Bijlsma, J.W.J.; Lafeber, F.P.J.G.; Baynes, J.W.; TeKoppele, J.M. Effect of collagen turnover on the accumulation of advanced glycation end products. J. Biol. Chem. 2000, 275, 39027–39031. [Google Scholar] [CrossRef] [PubMed]
- Kagan, H.M.; Trackman, P.C. Properties and function of lysyl oxidase. Am. J. Respir. Cell Mol. Biol. 1991, 5, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Rossow, L.; Veitl, S.; Vorlová, S.; Wax, J.K.; Kuhn, A.E.; Maltzahn, V.; Upcin, B.; Karl, F.; Hoffmann, H.; Gätzner, S.; et al. Lox-catalyzed collagen stabilization is a proximal cause for intrinsic resistance to chemotherapy. Oncogene 2018, 1. [Google Scholar] [CrossRef] [PubMed]
- Panwar, P.; Lamour, G.; Mackenzie, N.C.W.; Yang, H.; Ko, F.; Li, H.; Brömme, D. Changes in structural-mechanical properties and degradability of collagen during aging-associated modifications. J. Biol. Chem. 2015, 290, 23291–23306. [Google Scholar] [CrossRef] [PubMed]
- Bailey, A.J.; Paul, R.G.; Knott, L. Mechanisms of maturation and ageing of collagen. Mech. Ageing Dev. 1998, 106, 1–56. [Google Scholar] [CrossRef]
- Unoki, H.; Bujo, H.; Yamagishi, S.-I.; Takeuchi, M.; Imaizumi, T.; Saito, Y. Advanced glycation end products attenuate cellular insulin sensitivity by increasing the generation of intracellular reactive oxygen species in adipocytes. Diabetes Res. Clin. Pract. 2007, 76, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Robert, L. Mechanisms of aging of the extracellular matrix: Role of the elastin-laminin receptor. Gerontology 1998, 44, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Liotta, L.A.; Tryggvason, K.; Garbisa, S.; Hart, I.; Foltz, C.M.; Shafie, S. Metastatic potential correlates with enzymatic degradation of basement membrane collagen. Nature 1980, 284, 67–68. [Google Scholar] [CrossRef] [PubMed]
- Hotary, K.; Li, X.Y.; Allen, E.; Stevens, S.L.; Weiss, S.J. A cancer cell metalloprotease triad regulates the basement membrane transmigration program. Gene Dev. 2006, 20, 2673–2686. [Google Scholar] [CrossRef] [PubMed]
- Richardson, L.L.; Kleinman, H.K.; Dym, M. The effects of aging on basement membrane in the testis. J. Androl. 1995, 16, 118–126. [Google Scholar] [PubMed]
- Vázquez, F.; Palacios, S.; Alemañ, N.; Guerrero, F. Changes of the basement membrane and type iv collagen in human skin during aging. Maturitas 1996, 25, 209–215. [Google Scholar] [CrossRef]
- Capo-Chichi, C.D.; Smith, E.R.; Yang, D.-H.; Roland, I.H.; Vanderveer, L.; Cohen, C.; Hamilton, T.C.; Godwin, A.K.; Xu, X.-X. Dynamic alterations of the extracellular environment of ovarian surface epithelial cells in premalignant transformation, tumorigenicity, and metastasis. Cancer 2002, 95, 1802–1815. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, E. Ovarian cancer development and metastasis. Am. J. Pathol. 2010, 177, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Bagadi, S.A.R.; Prasad, C.P.; Srivastava, A.; Prashad, R.; Gupta, S.D.; Ralhan, R. Frequent loss of dab2 protein and infrequent promoter hypermethylation in breast cancer. Breast Cancer Res. Treat. 2007, 104, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.A.; Akman, K.; Calimport, S.R.G.; Wuttke, D.; Stolzing, A.; de Magalhães, J.P. The role of DNA methylation in aging, rejuvenation, and age-related disease. Rejuv. Res. 2012, 15, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Zampieri, M.; Ciccarone, F.; Calabrese, R.; Franceschi, C.; Bürkle, A.; Caiafa, P. Reconfiguration of DNA methylation in aging. Mech. Ageing Dev. 2015, 151, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Rump, A.; Morikawa, Y.; Tanaka, M.; Minami, S.; Umesaki, N.; Takeuchi, M.; Miyajima, A. Binding of ovarian cancer antigen ca125/muc16 to mesothelin mediates cell adhesion. J. Biol. Chem. 2004, 279, 9190–9198. [Google Scholar] [CrossRef] [PubMed]
- Monaco, S.; Sparano, V.; Gioia, M.; Sbardella, D.; Di Pierro, D.; Marini, S.; Coletta, M. Enzymatic processing of collagen iv by mmp-2 (gelatinase a) affects neutrophil migration and it is modulated by extracatalytic domains. Protein Sci. 2006, 15, 2805–2815. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.H.; Wang, J.M.; Yang, S.S.; Wang, F.F.; Hu, J.L.; Xin, S.N.; Men, H.; Lu, G.F.; Lan, X.L.; Zhang, D.; et al. Down-regulation of dab2ip promotes colorectal cancer invasion and metastasis by translocating hnrnpk into nucleus to enhance the transcription of mmp2: Down-regulation of dab2ip. Int. J. Cancer 2017, 141, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Patarroyo, M.; Tryggvason, K.; Virtanen, I. Laminin isoforms in tumor invasion, angiogenesis and metastasis. Semin. Cancer Biol. 2002, 12, 197–207. [Google Scholar] [CrossRef]
- Brar, P.K.; Dalkin, B.L.; Weyer, C.; Sallam, K.; Virtanen, I.; Nagle, R.B. Laminin alpha-1, alpha-3, and alpha-5 chain expression in human prepubetal benign prostate glands and adult benign and malignant prostate glands. Prostate 2003, 55, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, M. Advanced protein glycosylation in diabetes and aging. Annu. Rev. Med. 1995, 46, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Karttunen, T.; Risteli, J.; Autio–Harmainen, H.; Risteli, L. Effect of age and diabetes on type iv collagen and laminin in human kidney cortex. Kidney Int. 1986, 30, 586–591. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.D. Effects of donor age on protein and collagen synthesis in vitro by human diploid fibroblasts. Lab. Investig. 1986, 55, 490–496. [Google Scholar] [PubMed]
- Coppé, J.-P.; Kauser, K.; Campisi, J.; Beauséjour, C.M. Secretion of vascular endothelial growth factor by primary human fibroblasts at senescence. J. Biol. Chem. 2006, 281, 29568–29574. [Google Scholar] [CrossRef] [PubMed]
- Schosserer, M.; Grillari, J.; Breitenbach, M. The dual role of cellular senescence in developing tumors and their response to cancer therapy. Front. Oncol. 2017, 7, 278. [Google Scholar] [CrossRef] [PubMed]
- Parrinello, S. Stromal-epithelial interactions in aging and cancer: Senescent fibroblasts alter epithelial cell differentiation. J. Cell Sci. 2005, 118, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Rosen, D.G.; Zhang, Z.; Bast, R.C.; Mills, G.B.; Colacino, J.A.; Mercado-Uribe, I.; Liu, J. The chemokine growth-regulated oncogene 1 (gro-1) links ras signaling to the senescence of stromal fibroblasts and ovarian tumorigenesis. Proc. Natl. Acad. Sci. USA 2006, 103, 16472–16477. [Google Scholar] [CrossRef] [PubMed]
- Krtolica, A.; Parrinello, S.; Lockett, S.; Desprez, P.Y.; Campisi, J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: A link between cancer and aging. Proc. Natl. Acad. Sci. USA 2001, 98, 12072–12077. [Google Scholar] [CrossRef] [PubMed]
- Campisi, J. Senescent cells, tumor suppression, and organismal aging: Good citizens, bad neighbors. Cell 2005, 120, 513–522. [Google Scholar] [CrossRef] [PubMed]
- García-Gómez, I.; Goldsmith, H.S.; Angulo, J.; Prados, A.; López-Hervás, P.; Cuevas, B.; Dujovny, M.; Cuevas, P. Angiogenic capacity of human omental stem cells. Neurol. Res. 2005, 27, 807–811. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Huang, G.; Song, H.; Chen, Y.; Chen, L. Cancer associated fibroblasts: An essential role in the tumor microenvironment. Oncol. Lett. 2017, 14, 2611–2620. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.; Banerjee, D.; Ben-Baruch, A. Chemokines at the crossroads of tumor-fibroblast interactions that promote malignancy. J. Leukoc. Biol. 2011, 89, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Notta, F.; Navab, R.; Joseph, J.; Ibrahimov, E.; Xu, J.; Zhu, C.-Q.; Borgida, A.; Gallinger, S.; Tsao, M.-S. Senescent carcinoma-associated fibroblasts upregulate il8 to enhance prometastatic phenotypes. Mol. Cancer Res. 2017, 15, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Meza-Perez, S.; Randall, T.D. Immunological functions of the omentum. Trends Immunol. 2017, 38, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Gerber, S.A.; Rybalko, V.Y.; Bigelow, C.E.; Lugade, A.A.; Foster, T.H.; Frelinger, J.G.; Lord, E.M. Preferential attachment of peritoneal tumor metastases to omental immune aggregates and possible role of a unique vascular microenvironment in metastatic survival and growth. Am. J. Pathol. 2006, 169, 1739–1752. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J. Role of angiogenesis in tumor growth and metastasis. Semin. Oncol. 2002, 29, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, S.; Kim, S.W.; Wu, S.Y.; Nishimura, M.; Chaluvally-Raghavan, P.; Miyake, T.; Pecot, C.V.; Kim, S.-J.; Choi, H.J.; Bischoff, F.Z.; et al. Hematogenous metastasis of ovarian cancer: Rethinking mode of spread. Cancer Cell 2014, 26, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Coffman, L.G.; Burgos-Ojeda, D.; Wu, R.; Cho, K.; Bai, S.; Buckanovich, R.J. New models of hematogenous ovarian cancer metastasis demonstrate preferential spread to the ovary and a requirement for the ovary for abdominal dissemination. Transl. Res. 2016, 175, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Krist, L.F.G.; Eestermans, I.L.; Steenbergen, J.J.E.; Hoefsmit, E.C.M.; Cuesta, M.A.; Meyer, S.; Beelen, R.H.J. Cellular composition of milky spots in the human greater omentum: An immunochemical and ultrastructural study. Anat. Rec. 1995, 241, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Stout, R.D.; Suttles, J. Functional plasticity of macrophages: Reversible adaptation to changing microenvironments. J. Leukoc. Biol. 2004, 76, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Sebastián, C.; Espia, M.; Serra, M.; Celada, A.; Lloberas, J. Macrophaging: A cellular and molecular review. Immunobiology 2005, 210, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Linehan, E.; Fitzgerald, D. Ageing and the immune system: Focus on macrophages. Eur. J. Microbiol. Immunol. 2015, 5, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Kohut, M.L.; Senchina, D.S.; Madden, K.S.; Martin, A.E.; Felten, D.L.; Moynihan, J.A. Age effects on macrophage function vary by tissue site, nature of stimulant, and exercise behavior. Exp. Gerontol. 2004, 39, 1347–1360. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, M.; Del Río, M.; Medina, S. Changes with aging in the modulation by neuropeptide y of murine peritoneal macrophage functions. J. Neuroimmunol. 2001, 116, 156–167. [Google Scholar] [CrossRef]
- Kulbe, H.; Thompson, R.; Wilson, J.L.; Robinson, S.; Hagemann, T.; Fatah, R.; Gould, D.; Ayhan, A.; Balkwill, F. The inflammatory cytokine tumor necrosis factor- generates an autocrine tumor-promoting network in epithelial ovarian cancer cells. Cancer Res. 2007, 67, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Wallace, P.K.; Eisenstein, T.K.; Meissler, J.J.; Morahan, P.S. Decreases in macrophage mediated antitumor activity with aging. Mech. Ageing Dev. 1995, 77, 169–184. [Google Scholar] [CrossRef]
- Linehan, E.; Dombrowski, Y.; Snoddy, R.; Fallon, P.G.; Kissenpfennig, A.; Fitzgerald, D.C. Aging impairs peritoneal but not bone marrow-derived macrophage phagocytosis. Aging Cell 2014, 13, 699–708. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Condeelis, J.; Pollard, J.W. Macrophages: Obligate partners for tumor cell migration, invasion, and metastasis. Cell 2006, 124, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Dhabekar, G.; Dandekar, R.; Kingaonkar, A. Role of macrophages in malignancy. Ann. Maxillofac. Surg. 2011, 1, 150. [Google Scholar] [CrossRef] [PubMed]
- Pollard, J.W. Tumour-educated macrophages promote tumour progression and metastasis. Nat. Rev. Cancer 2004, 4, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Santoiemma, P.P.; Powell, D.J. Tumor infiltrating lymphocytes in ovarian cancer. Cancer Biol. Ther. 2015, 16, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Salam, N.; Rane, S.; Das, R.; Faulkner, M.; Gund, R.; Kandpal, U.; Lewis, V.; Mattoo, H.; Prabhu, S.; Ranganathan, V.; et al. T cell ageing: Effects of age on development, survival & function. Indian J. Med. Res. 2013, 138, 595–608. [Google Scholar] [PubMed]
- Haynes, L.; Eaton, S.M.; Burns, E.M.; Randall, T.D.; Swain, S.L. Cd4 t cell memory derived from young naive cells functions well into old age, but memory generated from aged naive cells functions poorly. Proc. Natl. Acad. Sci. USA 2003, 100, 15053–15058. [Google Scholar] [CrossRef] [PubMed]
- Linton, P.J. Antigen-independent changes in naive cd4 t cells with aging. J. Exp. Med. 1996, 184, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J.S.; Nelson, B.H. Tumor-infiltrating b cells and t cells: Working together to promote patient survival. OncoImmunology 2012, 1, 1623–1625. [Google Scholar] [CrossRef] [PubMed]
- Montfort, A.; Pearce, O.; Maniati, E.; Vincent, B.G.; Bixby, L.; Böhm, S.; Dowe, T.; Wilkes, E.H.; Chakravarty, P.; Thompson, R.; et al. A strong b-cell response is part of the immune landscape in human high-grade serous ovarian metastases. Clin. Cancer Res. 2017, 23, 250–262. [Google Scholar] [CrossRef] [PubMed]
- Frasca, D.; Blomberg, B.B. Effects of aging on b cell function. Curr. Opin. Immunol. 2009, 21, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Lee-Chang, C.; Bodogai, M.; Moritoh, K.; Chen, X.; Wersto, R.; Sen, R.; Young, H.A.; Croft, M.; Ferrucci, L.; Biragyn, A. Aging converts innate b1a cells into potent cd8+ t cell inducers. J. Immunol. 2016, 196, 3385–3397. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, S.; Berntsson, J.; Nodin, B.; Micke, P.; Jirström, K. Prognostic impact of tumour-associated b cells and plasma cells in epithelial ovarian cancer. J. Ovarian Res. 2016, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.P.; Elstrand, M.B.; Holth, A.; Silins, I.; Berner, A.; Trope, C.G.; Davidson, B.; Risberg, B. Nk- and b-cell infiltration correlates with worse outcome in metastatic ovarian carcinoma. Am. J. Clin. Pathol. 2006, 125, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Nieman, K.M.; Kenny, H.A.; Penicka, C.V.; Ladanyi, A.; Buell-Gutbrod, R.; Zillhardt, M.R.; Romero, I.L.; Carey, M.S.; Mills, G.B.; Hotamisligil, G.S.; et al. Adipocytes promote ovarian cancer metastasis and provide energy for rapid tumor growth. Nat. Med. 2011, 17, 1498–1503. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.K.; Kirkland, J.L. Aging and adipose tissue: Potential interventions for diabetes and regenerative medicine. Exp. Gerontol. 2016, 86, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Cartwright, M.; Tchkonia, T.; Kirkland, J. Aging in adipocytes: Potential impact of inherent, depot-specific mechanisms. Exp. Gerontol. 2007, 42, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.; Marmol, P.; Moliner, A.; Björnholm, M.; Zhang, C.; Shokat, K.M.; Ibanez, C.F. Adipocyte alk7 links nutrient overload to catecholamine resistance in obesity. eLife 2014, 3, e03245. [Google Scholar] [CrossRef] [PubMed]
- Huffman, D.M.; Barzilai, N. Role of visceral adipose tissue in aging. BBA-Gen. Subj. 2009, 1790, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Gabriely, I.; Ma, X.H.; Yang, X.M.; Atzmon, G.; Rajala, M.W.; Berg, A.H.; Scherer, P.; Rossetti, L.; Barzilai, N. Removal of visceral fat prevents insulin resistance and glucose intolerance of aging: An adipokine-mediated process? Diabetes 2002, 51, 2951–2958. [Google Scholar] [CrossRef] [PubMed]
- Muzumdar, R.; Allison, D.B.; Huffman, D.M.; Ma, X.; Atzmon, G.; Einstein, F.H.; Fishman, S.; Poduval, A.D.; McVei, T.; Keith, S.W.; et al. Visceral adipose tissue modulates mammalian longevity. Aging Cell 2008, 7, 438–440. [Google Scholar] [CrossRef] [PubMed]
- Foong, K.W.; Bolton, H. Obesity and ovarian cancer risk: A systematic review. Post Reprod. Health 2017, 23, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Metzinger, M.N.; Lewellen, K.A.; Cripps, S.N.; Carey, K.D.; Harper, E.I.; Shi, Z.; Tarwater, L.; Grisoli, A.; Lee, E.; et al. Obesity contributes to ovarian cancer metastatic success through increased lipogenesis, enhanced vascularity, and decreased infiltration of m1 macrophages. Cancer Res. 2015, 75, 5046–5057. [Google Scholar] [CrossRef] [PubMed]
- Tchkonia, T.; Morbeck, D.E.; Zglinicki, T.V.; Deursen, J.V.; Lustgarten, J.; Scrable, H.; Khosla, S.; Jensen, M.D.; Kirkland, J.L. Fat tissue, aging, and cellular senescence. Aging Cell 2010, 9, 667–684. [Google Scholar] [CrossRef] [PubMed]
- Ahima, R.S. Connecting obesity, aging and diabetes. Nat. Med. 2009, 15, 996–997. [Google Scholar] [CrossRef] [PubMed]
- Gkogkolou, P.; Böhm, M. Advanced glycation end products. Dermatoendocrinol 2012, 4, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Naitoh, T.; Kitahara, M.; Tsuruzoe, N. Tumor necrosis factor-alpha is induced through phorbol ester--and glycated human albumin-dependent pathway in thp-1 cells. Cell. Signal. 2001, 13, 331–334. [Google Scholar] [CrossRef]
- Kirkland, J.L.; Tchkonia, T. Cellular senescence: A translational perspective. eBioMedicine 2017, 21, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Tchkonia, T.; Pirtskhalava, T.; Gower, A.C.; Ding, H.; Giorgadze, N.; Palmer, A.K.; Ikeno, Y.; Hubbard, G.B.; Lenburg, M.; et al. The achilles’ heel of senescent cells: From transcriptome to senolytic drugs. Aging Cell 2015, 14, 644–658. [Google Scholar] [CrossRef] [PubMed]
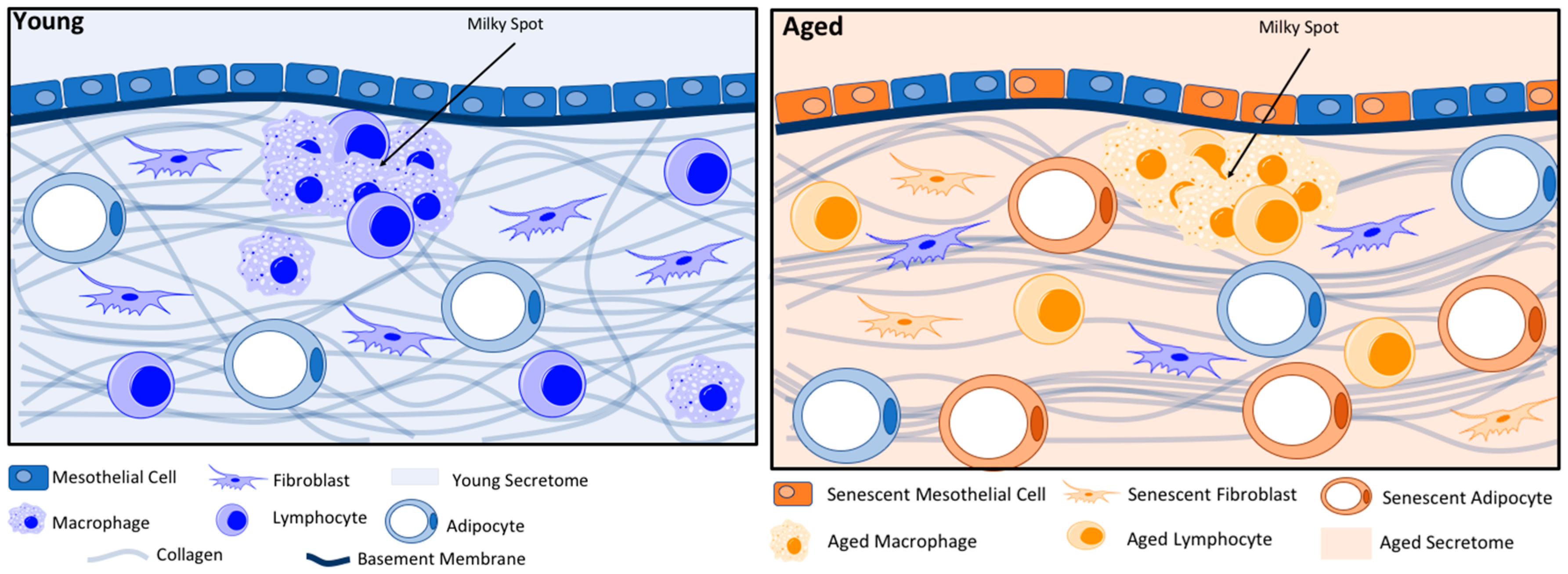
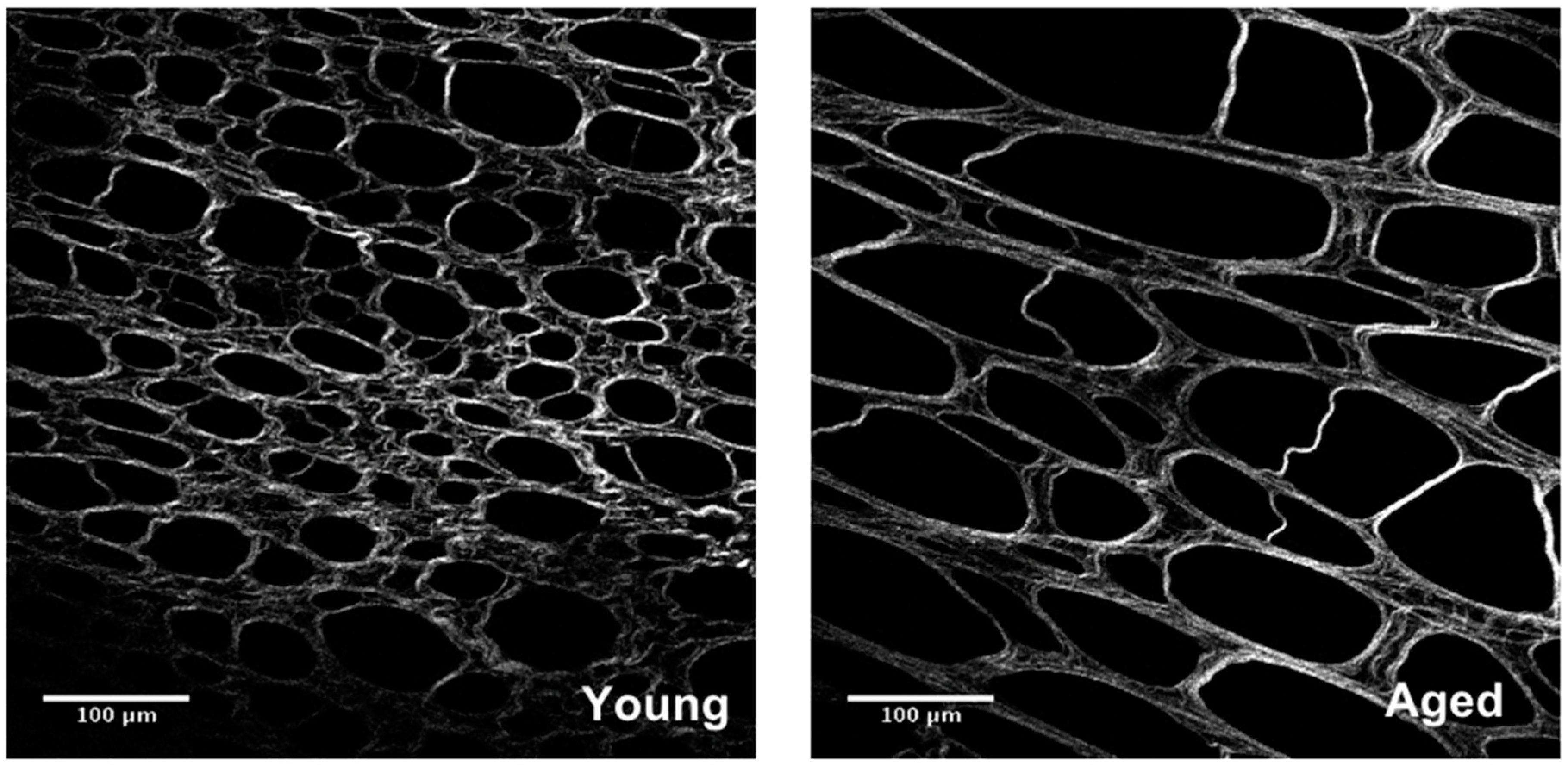
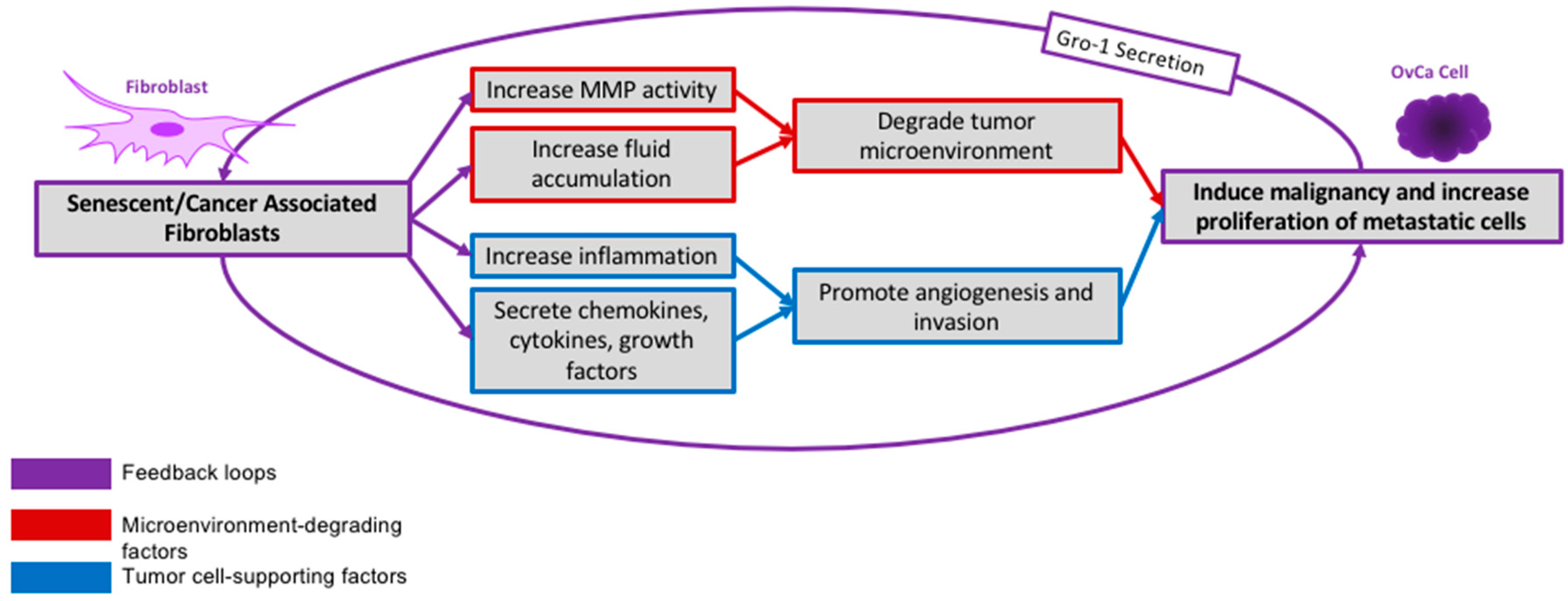
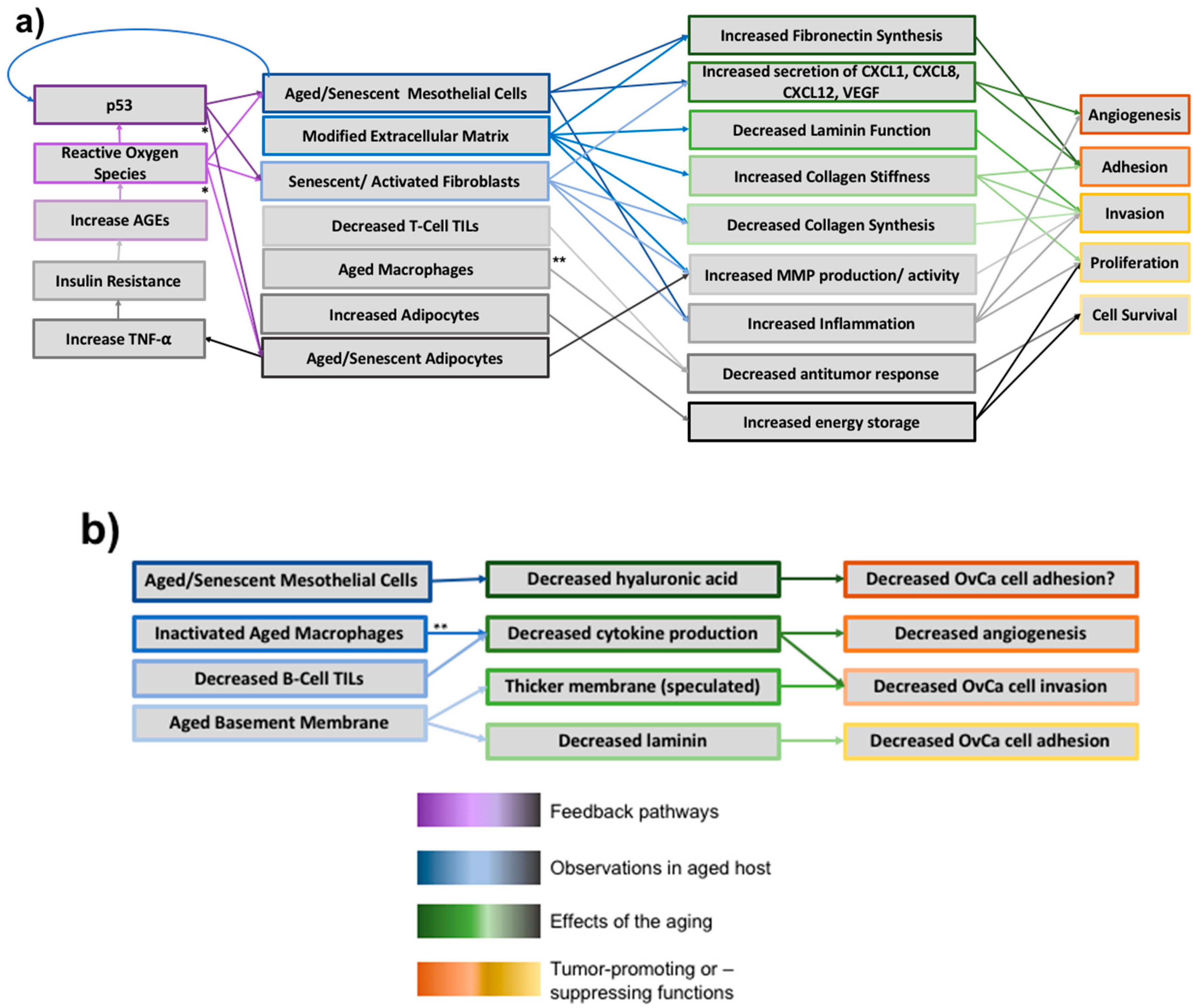
| Immune Cell Component | Effect of Aging | Effect on OvCa Metastasis |
|---|---|---|
| T-cell Tumor Infiltrating Lymphocytes |
| Decreased tumor lysis leads to increased proliferation |
| B-Cell tumor Infiltrating Lymphocytes |
| Possibly increased angiogenesis, possibly decreased tumor lysis |
| Pre-Activated Macrophages |
| Unknown, possibly mixed effects. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harper, E.I.; Sheedy, E.F.; Stack, M.S. With Great Age Comes Great Metastatic Ability: Ovarian Cancer and the Appeal of the Aging Peritoneal Microenvironment. Cancers 2018, 10, 230. https://doi.org/10.3390/cancers10070230
Harper EI, Sheedy EF, Stack MS. With Great Age Comes Great Metastatic Ability: Ovarian Cancer and the Appeal of the Aging Peritoneal Microenvironment. Cancers. 2018; 10(7):230. https://doi.org/10.3390/cancers10070230
Chicago/Turabian StyleHarper, Elizabeth I., Emma F. Sheedy, and M. Sharon Stack. 2018. "With Great Age Comes Great Metastatic Ability: Ovarian Cancer and the Appeal of the Aging Peritoneal Microenvironment" Cancers 10, no. 7: 230. https://doi.org/10.3390/cancers10070230
APA StyleHarper, E. I., Sheedy, E. F., & Stack, M. S. (2018). With Great Age Comes Great Metastatic Ability: Ovarian Cancer and the Appeal of the Aging Peritoneal Microenvironment. Cancers, 10(7), 230. https://doi.org/10.3390/cancers10070230





