Radiotherapy as a New Player in Immuno-Oncology
Abstract
1. Introduction
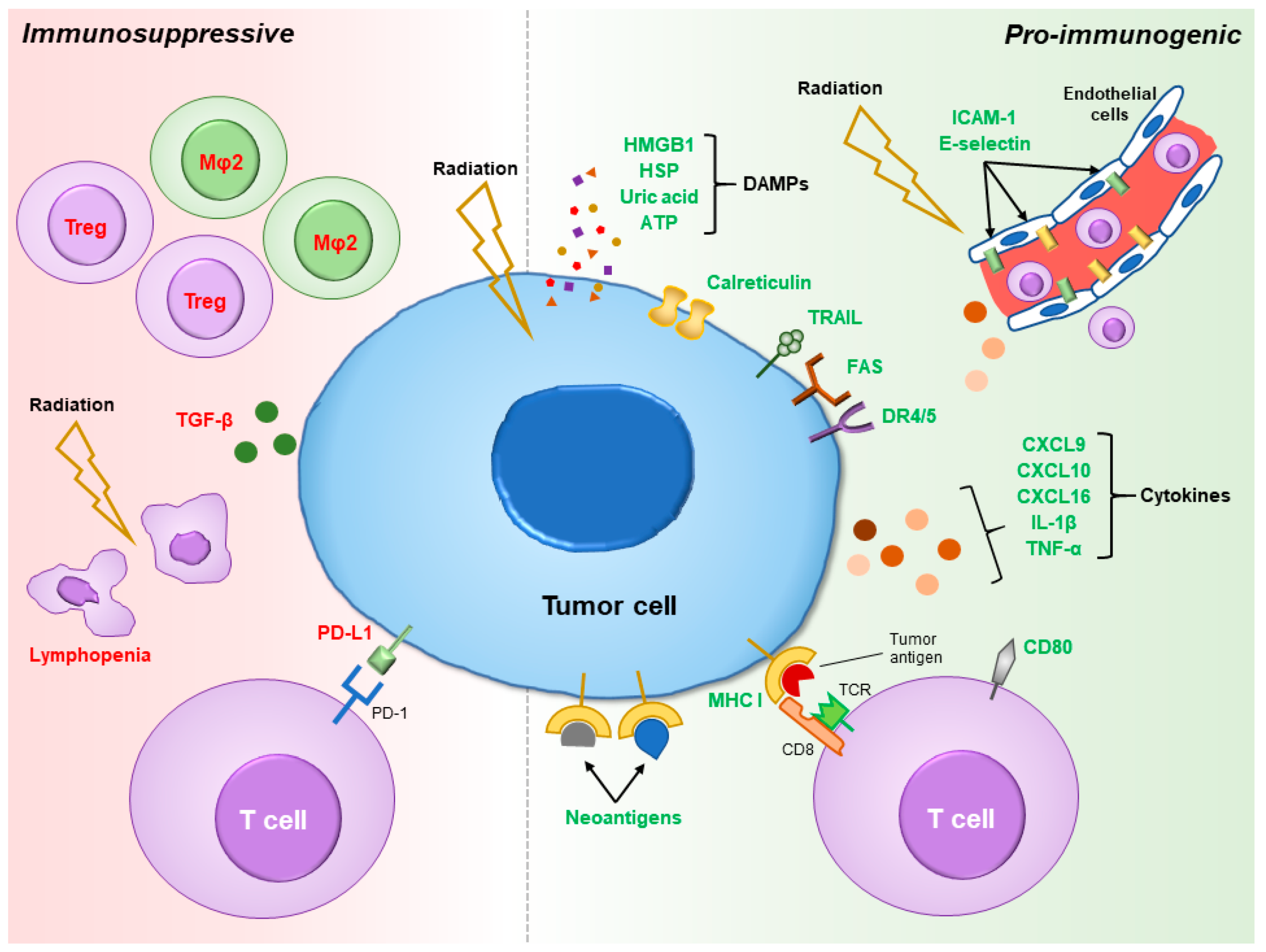
2. Pro-Immunogenic Effects of Radiation
3. Immunosuppressive Effects of Radiation
4. Immune-Mediated Systemic Effects of Radiotherapy
5. Synergy of Radiotherapy and Immunotherapy Combination
5.1. Toll-Like Receptor Agonists
5.2. Cytokines
5.3. Co-Stimulatory Molecules
5.4. Immune Checkpoint Inhibition
5.5. Macrophage Polarization
6. Effect of Radiation Dose and Timing on Immunogenicity
6.1. Dose per Fraction
6.2. Timing
7. Ongoing Clinical Trials Assessing Combination of Radiotherapy with Immunotherapy
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Golden, E.B.; Formenti, S.C. Is tumor (R)ejection by the immune system the “5th R” of radiobiology? Oncoimmunology 2014, 3, e28133. [Google Scholar] [CrossRef] [PubMed]
- Stone, H.B.; Peters, L.J.; Milas, L. Effect of host immune capability on radiocurability and subsequent transplantability of a murine fibrosarcoma. J. Natl. Cancer Inst. 1979, 63, 1229–1235. [Google Scholar] [PubMed]
- Lugade, A.A.; Moran, J.P.; Gerber, S.A.; Rose, R.C.; Frelinger, J.G.; Lord, E.M. Local radiation therapy of B16 melanoma tumors increases the generation of tumor antigen-specific effector cells that traffic to the tumor. J. Immunol. 2005, 174, 7516–7523. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Auh, S.L.; Wang, Y.; Burnette, B.; Wang, Y.; Meng, Y.; Beckett, M.; Sharma, R.; Chin, R.; Tu, T.; et al. Therapeutic effects of ablative radiation on local tumor require CD8+ T cells: Changing strategies for cancer treatment. Blood 2009, 114, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Demaria, S.; Ng, B.; Devitt, M.L.; Babb, J.S.; Kawashima, N.; Liebes, L.; Formenti, S.C. Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int. J. Radiat. Oncol. Biol. Phys. 2004, 58, 862–870. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, S.; Wang, B.; Kawashima, N.; Braunstein, S.; Badura, M.; Cameron, T.O.; Babb, J.S.; Schneider, R.J.; Formenti, S.C.; Dustin, M.L.; et al. Radiation-induced CXCL16 release by breast cancer cells attracts effector T cells. J. Immunol. 2008, 181, 3099–3107. [Google Scholar] [CrossRef] [PubMed]
- Dewan, M.Z.; Galloway, A.E.; Kawashima, N.; Dewyngaert, J.K.; Babb, J.S.; Formenti, S.C.; Demaria, S. Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti-CTLA-4 antibody. Clin. Cancer Res. 2009, 15, 5379–5388. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Liang, H.; Burnette, B.; Beckett, M.; Darga, T.; Weichselbaum, R.R.; Fu, Y.X. Irradiation and anti-PD-L1 treatment synergistically promote antitumor immunity in mice. J. Clin. Investig. 2014, 124, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Schuler, G.; Steinman, R.M. Dendritic cells as adjuvants for immune-mediated resistance to tumors. J. Exp. Med. 1997, 186, 1183–1187. [Google Scholar] [CrossRef] [PubMed]
- Albert, M.L.; Sauter, B.; Bhardwaj, N. Dendritic cells acquire antigen from apoptotic cells and induce class I-restricted CTLs. Nature 1998, 392, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Apetoh, L.; Ghiringhelli, F.; Tesniere, A.; Obeid, M.; Ortiz, C.; Criollo, A.; Mignot, G.; Maiuri, M.C.; Ullrich, E.; Saulnier, P.; et al. Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat. Med. 2007, 13, 1050–1059. [Google Scholar] [CrossRef] [PubMed]
- Zitvogel, L.; Kepp, O.; Kroemer, G. Decoding cell death signals in inflammation and immunity. Cell 2010, 140, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Golden, E.B.; Frances, D.; Pellicciotta, I.; Demaria, S.; Helen Barcellos-Hoff, M.; Formenti, S.C. Radiation fosters dose-dependent and chemotherapy-induced immunogenic cell death. Oncoimmunology 2014, 3, e28518. [Google Scholar] [CrossRef] [PubMed]
- Elliott, M.R.; Chekeni, F.B.; Trampont, P.C.; Lazarowski, E.R.; Kadl, A.; Walk, S.F.; Park, D.; Woodson, R.I.; Ostankovich, M.; Sharma, P.; et al. Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature 2009, 461, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Ghiringhelli, F.; Apetoh, L.; Tesniere, A.; Aymeric, L.; Ma, Y.; Ortiz, C.; Vermaelen, K.; Panaretakis, T.; Mignot, G.; Ullrich, E.; et al. Activation of the NLRP3 inflammasome in dendritic cells induces IL-1beta-dependent adaptive immunity against tumors. Nat. Med. 2009, 15, 1170–1178. [Google Scholar] [CrossRef] [PubMed]
- Obeid, M.; Panaretakis, T.; Joza, N.; Tufi, R.; Tesniere, A.; van Endert, P.; Zitvogel, L.; Kroemer, G. Calreticulin exposure is required for the immunogenicity of gamma-irradiation and UVC light-induced apoptosis. Cell Death Differ. 2007, 14, 1848–1850. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, M.; Abrams, S.I.; Camphausen, K.; Liu, K.; Scott, T.; Coleman, C.N.; Hodge, J.W. Irradiation of tumor cells up-regulates Fas and enhances CTL lytic activity and CTL adoptive immunotherapy. J. Immunol. 2003, 170, 6338–6347. [Google Scholar] [CrossRef] [PubMed]
- Kuwabara, M.; Takahashi, K.; Inanami, O. Induction of apoptosis through the activation of SAPK/JNK followed by the expression of death receptor Fas in X-irradiated cells. J. Radiat. Res. 2003, 44, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Chinnaiyan, A.M.; Prasad, U.; Shankar, S.; Hamstra, D.A.; Shanaiah, M.; Chenevert, T.L.; Ross, B.D.; Rehemtulla, A. Combined effect of tumor necrosis factor-related apoptosis-inducing ligand and ionizing radiation in breast cancer therapy. Proc. Natl. Acad. Sci. USA 2000, 97, 1754–1759. [Google Scholar] [CrossRef] [PubMed]
- Marini, P.; Schmid, A.; Jendrossek, V.; Faltin, H.; Daniel, P.T.; Budach, W.; Belka, C. Irradiation specifically sensitises solid tumour cell lines to TRAIL mediated apoptosis. BMC Cancer 2005, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Hori, T.; Kondo, T.; Kanamori, M.; Tabuchi, Y.; Ogawa, R.; Zhao, Q.L.; Ahmed, K.; Yasuda, T.; Seki, S.; Suzuki, K.; et al. Ionizing radiation enhances tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis through up-regulations of death receptor 4 (DR4) and death receptor 5 (DR5) in human osteosarcoma cells. J. Orthop. Res. 2010, 28, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Vereecque, R.; Buffenoir, G.; Gonzalez, R.; Cambier, N.; Hetuin, D.; Bauters, F.; Fenaux, P.; Quesnel, B. gamma-ray irradiation induces B7.1 expression in myeloid leukaemic cells. Br. J. Haematol. 2000, 108, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Gasser, S.; Orsulic, S.; Brown, E.J.; Raulet, D.H. The DNA damage pathway regulates innate immune system ligands of the NKG2D receptor. Nature 2005, 436, 1186–1190. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Son, Y.O.; Park, S.W.; Bae, J.H.; Chung, J.S.; Kim, H.H.; Chung, B.S.; Kim, S.H.; Kang, C.D. Increase of NKG2D ligands and sensitivity to NK cell-mediated cytotoxicity of tumor cells by heat shock and ionizing radiation. Exp. Mol. Med. 2006, 38, 474–484. [Google Scholar] [CrossRef] [PubMed]
- Bellone, M.; Calcinotto, A. Ways to enhance lymphocyte trafficking into tumors and fitness of tumor infiltrating lymphocytes. Front. Oncol. 2013, 3, 231. [Google Scholar] [CrossRef] [PubMed]
- Hallahan, D.; Kuchibhotla, J.; Wyble, C. Cell adhesion molecules mediate radiation-induced leukocyte adhesion to the vascular endothelium. Cancer Res. 1996, 56, 5150–5155. [Google Scholar] [PubMed]
- Quarmby, S.; Hunter, R.D.; Kumar, S. Irradiation induced expression of CD31, ICAM-1 and VCAM-1 in human microvascular endothelial cells. Anticancer Res. 2000, 20, 3375–3381. [Google Scholar] [PubMed]
- Matsumura, S.; Demaria, S. Up-regulation of the pro-inflammatory chemokine CXCL16 is a common response of tumor cells to ionizing radiation. Radiat. Res. 2010, 173, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Lugade, A.A.; Sorensen, E.W.; Gerber, S.A.; Moran, J.P.; Frelinger, J.G.; Lord, E.M. Radiation-induced IFN-gamma production within the tumor microenvironment influences antitumor immunity. J. Immunol. 2008, 180, 3132–3139. [Google Scholar] [CrossRef] [PubMed]
- Hallahan, D.E.; Spriggs, D.R.; Beckett, M.A.; Kufe, D.W.; Weichselbaum, R.R. Increased tumor necrosis factor alpha mRNA after cellular exposure to ionizing radiation. Proc. Natl. Acad. Sci. USA 1989, 86, 10104–10107. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, H.; Tsuneoka, K.; Dimchev, A.B.; Shikita, M. Induction of the expression of the interleukin-1 beta gene in mouse spleen by ionizing radiation. Radiat. Res. 1993, 133, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.H.; Chiang, C.S.; Tsao, C.Y.; Lin, P.Y.; McBride, W.H.; Wu, C.J. Rapid induction of cytokine gene expression in the lung after single and fractionated doses of radiation. Int. J. Radiat. Biol. 1999, 75, 1421–1427. [Google Scholar] [PubMed]
- Burnette, B.C.; Liang, H.; Lee, Y.; Chlewicki, L.; Khodarev, N.N.; Weichselbaum, R.R.; Fu, Y.X.; Auh, S.L. The efficacy of radiotherapy relies upon induction of type I interferon-dependent innate and adaptive immunity. Cancer Res. 2011, 71, 2488–2496. [Google Scholar] [CrossRef] [PubMed]
- Leone, P.; Shin, E.C.; Perosa, F.; Vacca, A.; Dammacco, F.; Racanelli, V. MHC class I antigen processing and presenting machinery: Organization, function, and defects in tumor cells. J. Natl. Cancer Inst. 2013, 105, 1172–1187. [Google Scholar] [CrossRef] [PubMed]
- Garnett, C.T.; Palena, C.; Chakraborty, M.; Tsang, K.Y.; Schlom, J.; Hodge, J.W. Sublethal irradiation of human tumor cells modulates phenotype resulting in enhanced killing by cytotoxic T lymphocytes. Cancer Res. 2004, 64, 7985–7994. [Google Scholar] [CrossRef] [PubMed]
- Reits, E.A.; Hodge, J.W.; Herberts, C.A.; Groothuis, T.A.; Chakraborty, M.; Wansley, E.K.; Camphausen, K.; Luiten, R.M.; de Ru, A.H.; Neijssen, J.; et al. Radiation modulates the peptide repertoire, enhances MHC class I expression, and induces successful antitumor immunotherapy. J. Exp. Med. 2006, 203, 1259–1271. [Google Scholar] [CrossRef] [PubMed]
- Newcomb, E.W.; Demaria, S.; Lukyanov, Y.; Shao, Y.; Schnee, T.; Kawashima, N.; Lan, L.; Dewyngaert, J.K.; Zagzag, D.; McBride, W.H.; et al. The combination of ionizing radiation and peripheral vaccination produces long-term survival of mice bearing established invasive GL261 gliomas. Clin. Cancer Res. 2006, 12, 4730–4737. [Google Scholar] [CrossRef] [PubMed]
- Formenti, S.C.; Demaria, S. Radiation therapy to convert the tumor into an in situ vaccine. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, 879–880. [Google Scholar] [CrossRef] [PubMed]
- Segal, N.H.; Parsons, D.W.; Peggs, K.S.; Velculescu, V.; Kinzler, K.W.; Vogelstein, B.; Allison, J.P. Epitope landscape in breast and colorectal cancer. Cancer Res. 2008, 68, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, T.N.; Schreiber, R.D. Neoantigens in cancer immunotherapy. Science 2015, 348, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, N.; Kusunoki, Y.; Akiyama, M. Radiosensitivity of CD4 or CD8 positive human T-lymphocytes by an in vitro colony formation assay. Radiat. Res. 1990, 123, 224–227. [Google Scholar] [CrossRef] [PubMed]
- Yovino, S.; Kleinberg, L.; Grossman, S.A.; Narayanan, M.; Ford, E. The etiology of treatment-related lymphopenia in patients with malignant gliomas: Modeling radiation dose to circulating lymphocytes explains clinical observations and suggests methods of modifying the impact of radiation on immune cells. Cancer Investig. 2013, 31, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Venkatesulu, B.P.; Mallick, S.; Lin, S.H.; Krishnan, S. A systematic review of the influence of radiation-induced lymphopenia on survival outcomes in solid tumors. Crit. Rev. Oncol./Hematol. 2018, 123, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Jin, S.; Zhang, A.; Zhang, B.; Shi, X.; Wang, J.; Zhao, Y. Gamma-ray resistance of regulatory CD4+CD25+Foxp3+ T cells in mice. Radiat. Res. 2010, 173, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Kachikwu, E.L.; Iwamoto, K.S.; Liao, Y.P.; DeMarco, J.J.; Agazaryan, N.; Economou, J.S.; McBride, W.H.; Schaue, D. Radiation enhances regulatory T cell representation. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 1128–1135. [Google Scholar] [CrossRef] [PubMed]
- Barcellos-Hoff, M.H.; Derynck, R.; Tsang, M.L.; Weatherbee, J.A. Transforming growth factor-beta activation in irradiated murine mammary gland. J. Clin. Investig. 1994, 93, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Jobling, M.F.; Mott, J.D.; Finnegan, M.T.; Jurukovski, V.; Erickson, A.C.; Walian, P.J.; Taylor, S.E.; Ledbetter, S.; Lawrence, C.M.; Rifkin, D.B.; et al. Isoform-specific activation of latent transforming growth factor beta (LTGF-beta) by reactive oxygen species. Radiat. Res. 2006, 166, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.S.; Chen, F.H.; Wang, C.C.; Huang, H.L.; Jung, S.M.; Wu, C.J.; Lee, C.C.; McBride, W.H.; Chiang, C.S.; Hong, J.H. Macrophages from irradiated tumors express higher levels of iNOS, arginase-I and COX-2, and promote tumor growth. Int. J. Radiat. Oncol. Biol. Phys. 2007, 68, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.S.; Fu, S.Y.; Wang, S.C.; Yu, C.F.; Chen, F.H.; Lin, C.M.; Hong, J.H. Irradiation promotes an m2 macrophage phenotype in tumor hypoxia. Front. Oncol. 2012, 2, 89. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Escamilla, J.; Mok, S.; David, J.; Priceman, S.; West, B.; Bollag, G.; McBride, W.; Wu, L. CSF1R signaling blockade stanches tumor-infiltrating myeloid cells and improves the efficacy of radiotherapy in prostate cancer. Cancer Res. 2013, 73, 2782–2794. [Google Scholar] [CrossRef] [PubMed]
- Dovedi, S.J.; Adlard, A.L.; Lipowska-Bhalla, G.; McKenna, C.; Jones, S.; Cheadle, E.J.; Stratford, I.J.; Poon, E.; Morrow, M.; Stewart, R.; et al. Acquired resistance to fractionated radiotherapy can be overcome by concurrent PD-L1 blockade. Cancer Res. 2014, 74, 5458–5468. [Google Scholar] [CrossRef] [PubMed]
- Parikh, F.; Duluc, D.; Imai, N.; Clark, A.; Misiukiewicz, K.; Bonomi, M.; Gupta, V.; Patsias, A.; Parides, M.; Demicco, E.G.; et al. Chemoradiotherapy-induced upregulation of PD-1 antagonizes immunity to HPV-related oropharyngeal cancer. Cancer Res. 2014, 74, 7205–7216. [Google Scholar] [CrossRef] [PubMed]
- Badoual, C.; Hans, S.; Merillon, N.; Van Ryswick, C.; Ravel, P.; Benhamouda, N.; Levionnois, E.; Nizard, M.; Si-Mohamed, A.; Besnier, N.; et al. PD-1-expressing tumor-infiltrating T cells are a favorable prognostic biomarker in HPV-associated head and neck cancer. Cancer Res. 2013, 73, 128–138. [Google Scholar] [CrossRef] [PubMed]
- McCulloch, H.D. On the analogy between spontaneous recoveries from cancer and the specific immunity induced by x ray irradiations of the lymphatic glands involved. Br. Med. J. 1908, 2, 1146–1148. [Google Scholar] [CrossRef] [PubMed]
- Brix, N.; Tiefenthaller, A.; Anders, H.; Belka, C.; Lauber, K. Abscopal, immunological effects of radiotherapy: Narrowing the gap between clinical and preclinical experiences. Immunol. Rev. 2017, 280, 249–279. [Google Scholar] [CrossRef] [PubMed]
- Budhu, S.; Loike, J.D.; Pandolfi, A.; Han, S.; Catalano, G.; Constantinescu, A.; Clynes, R.; Silverstein, S.C. CD8+ T cell concentration determines their efficiency in killing cognate antigen-expressing syngeneic mammalian cells in vitro and in mouse tissues. J. Exp. Med. 2010, 207, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Reynders, K.; Illidge, T.; Siva, S.; Chang, J.Y.; De Ruysscher, D. The abscopal effect of local radiotherapy: Using immunotherapy to make a rare event clinically relevant. Cancer Treat. Rev. 2015, 41, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Postow, M.A.; Callahan, M.K.; Barker, C.A.; Yamada, Y.; Yuan, J.; Kitano, S.; Mu, Z.; Rasalan, T.; Adamow, M.; Ritter, E.; et al. Immunologic correlates of the abscopal effect in a patient with melanoma. N. Engl. J. Med. 2012, 366, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Teitz-Tennenbaum, S.; Li, Q.; Rynkiewicz, S.; Ito, F.; Davis, M.A.; McGinn, C.J.; Chang, A.E. Radiotherapy potentiates the therapeutic efficacy of intratumoral dendritic cell administration. Cancer Res. 2003, 63, 8466–8475. [Google Scholar] [PubMed]
- Filatenkov, A.; Baker, J.; Mueller, A.M.; Kenkel, J.; Ahn, G.O.; Dutt, S.; Zhang, N.; Kohrt, H.; Jensen, K.; Dejbakhsh-Jones, S.; et al. Ablative Tumor Radiation Can Change the Tumor Immune Cell Microenvironment to Induce Durable Complete Remissions. Clin. Cancer Res. 2015, 21, 3727–3739. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, L.; Yu, D.; Kandimalla, E.R.; Sun, H.B.; Agrawal, S.; Guha, C. An in situ autologous tumor vaccination with combined radiation therapy and TLR9 agonist therapy. PLoS ONE 2012, 7, e38111. [Google Scholar] [CrossRef] [PubMed]
- Milas, L.; Mason, K.A.; Ariga, H.; Hunter, N.; Neal, R.; Valdecanas, D.; Krieg, A.M.; Whisnant, J.K. CpG oligodeoxynucleotide enhances tumor response to radiation. Cancer Res. 2004, 64, 5074–5077. [Google Scholar] [CrossRef] [PubMed]
- Mason, K.A.; Ariga, H.; Neal, R.; Valdecanas, D.; Hunter, N.; Krieg, A.M.; Whisnant, J.K.; Milas, L. Targeting toll-like receptor 9 with CpG oligodeoxynucleotides enhances tumor response to fractionated radiotherapy. Clin. Cancer Res. 2005, 11, 361–369. [Google Scholar] [PubMed]
- Chamoto, K.; Takeshima, T.; Wakita, D.; Ohkuri, T.; Ashino, S.; Omatsu, T.; Shirato, H.; Kitamura, H.; Togashi, Y.; Nishimura, T. Combination immunotherapy with radiation and CpG-based tumor vaccination for the eradication of radio- and immuno-resistant lung carcinoma cells. Cancer Sci. 2009, 100, 934–939. [Google Scholar] [CrossRef] [PubMed]
- Cerkovnik, P.; Novakovic, B.J.; Stegel, V.; Novakovic, S. Tumor vaccine composed of C-class CpG oligodeoxynucleotides and irradiated tumor cells induces long-term antitumor immunity. BMC Immunol. 2010, 11, 45. [Google Scholar] [CrossRef] [PubMed]
- Dewan, M.Z.; Vanpouille-Box, C.; Kawashima, N.; DiNapoli, S.; Babb, J.S.; Formenti, S.C.; Adams, S.; Demaria, S. Synergy of topical toll-like receptor 7 agonist with radiation and low-dose cyclophosphamide in a mouse model of cutaneous breast cancer. Clin. Cancer Res. 2012, 18, 6668–6678. [Google Scholar] [CrossRef] [PubMed]
- Dovedi, S.J.; Melis, M.H.; Wilkinson, R.W.; Adlard, A.L.; Stratford, I.J.; Honeychurch, J.; Illidge, T.M. Systemic delivery of a TLR7 agonist in combination with radiation primes durable antitumor immune responses in mouse models of lymphoma. Blood 2013, 121, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Adlard, A.L.; Dovedi, S.J.; Telfer, B.A.; Koga-Yamakawa, E.; Pollard, C.; Honeychurch, J.; Illidge, T.M.; Murata, M.; Robinson, D.T.; Jewsbury, P.J.; et al. A novel systemically administered Toll-like receptor 7 agonist potentiates the effect of ionizing radiation in murine solid tumor models. Int. J. Cancer 2014, 135, 820–829. [Google Scholar] [CrossRef] [PubMed]
- Scholch, S.; Rauber, C.; Tietz, A.; Rahbari, N.N.; Bork, U.; Schmidt, T.; Kahlert, C.; Haberkorn, U.; Tomai, M.A.; Lipson, K.E.; et al. Radiotherapy combined with TLR7/8 activation induces strong immune responses against gastrointestinal tumors. Oncotarget 2015, 6, 4663–4676. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.H.; Lee, H.J.; Ko, H.J.; Yoon, B.I.; Choe, J.; Kim, K.C.; Hahn, T.W.; Han, J.A.; Choi, S.S.; Jung, Y.M.; et al. The TLR7 agonist imiquimod induces anti-cancer effects via autophagic cell death and enhances anti-tumoral and systemic immunity during radiotherapy for melanoma. Oncotarget 2017, 8, 24932–24948. [Google Scholar] [CrossRef] [PubMed]
- Brody, J.D.; Ai, W.Z.; Czerwinski, D.K.; Torchia, J.A.; Levy, M.; Advani, R.H.; Kim, Y.H.; Hoppe, R.T.; Knox, S.J.; Shin, L.K.; et al. In situ vaccination with a TLR9 agonist induces systemic lymphoma regression: A phase I/II study. J. Clin. Oncol. 2010, 28, 4324–4332. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Gratzinger, D.; Harrison, C.; Brody, J.D.; Czerwinski, D.K.; Ai, W.Z.; Morales, A.; Abdulla, F.; Xing, L.; Navi, D.; et al. In situ vaccination against mycosis fungoides by intratumoral injection of a TLR9 agonist combined with radiation: A phase 1/2 study. Blood 2012, 119, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Seung, S.K.; Curti, B.D.; Crittenden, M.; Walker, E.; Coffey, T.; Siebert, J.C.; Miller, W.; Payne, R.; Glenn, L.; Bageac, A.; et al. Phase 1 study of stereotactic body radiotherapy and interleukin-2—Tumor and immunological responses. Sci. Transl. Med. 2012, 4, 137ra174. [Google Scholar] [CrossRef] [PubMed]
- Sedlar, A.; Kranjc, S.; Dolinsek, T.; Cemazar, M.; Coer, A.; Sersa, G. Radiosensitizing effect of intratumoral interleukin-12 gene electrotransfer in murine sarcoma. BMC Cancer 2013, 13, 38. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.Y.; Keng, P.C. Inhibition of cell growth in synchronous human hypernephroma cells by recombinant interferon alpha-D and irradiation. J. Interferon Res. 1983, 3, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Kardamakis, D.; Gillies, N.E.; Souhami, R.L.; Bewerley, P.C. Recombinant human interferon alpha-2b enhances the radiosensitivity of small cell lung cancer in vitro. Anticancer Res. 1989, 9, 1041–1044. [Google Scholar] [PubMed]
- Nukui, Y.; Picozzi, V.J.; Traverso, L.W. Interferon-based adjuvant chemoradiation therapy improves survival after pancreaticoduodenectomy for pancreatic adenocarcinoma. Am. J. Surg. 2000, 179, 367–371. [Google Scholar] [CrossRef]
- Picozzi, V.J.; Abrams, R.A.; Decker, P.A.; Traverso, W.; O’Reilly, E.M.; Greeno, E.; Martin, R.C.; Wilfong, L.S.; Rothenberg, M.L.; Posner, M.C.; et al. Multicenter phase II trial of adjuvant therapy for resected pancreatic cancer using cisplatin, 5-fluorouracil, and interferon-alfa-2b-based chemoradiation: ACOSOG Trial Z05031. Ann. Oncol. 2011, 22, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.; Abel, U.; Debus, J.; Harig, S.; Hoffmann, K.; Herrmann, T.; Bartsch, D.; Klein, J.; Mansmann, U.; Jager, D.; et al. Open-label, multicenter, randomized phase III trial of adjuvant chemoradiation plus interferon Alfa-2b versus fluorouracil and folinic acid for patients with resected pancreatic adenocarcinoma. J. Clin. Oncol. 2012, 30, 4077–4083. [Google Scholar] [CrossRef] [PubMed]
- Hallahan, D.E.; Vokes, E.E.; Rubin, S.J.; O’Brien, S.; Samuels, B.; Vijaykumar, S.; Kufe, D.W.; Phillips, R.; Weichselbaum, R.R. Phase I dose-escalation study of tumor necrosis factor-alpha and concomitant radiation therapy. Cancer J. Sci. Am. 1995, 1, 204–209. [Google Scholar] [PubMed]
- Mundt, A.J.; Vijayakumar, S.; Nemunaitis, J.; Sandler, A.; Schwartz, H.; Hanna, N.; Peabody, T.; Senzer, N.; Chu, K.; Rasmussen, C.S.; et al. A Phase I trial of TNFerade biologic in patients with soft tissue sarcoma in the extremities. Clin. Cancer Res. 2004, 10, 5747–5753. [Google Scholar] [CrossRef] [PubMed]
- Citrin, D.; Camphausen, K.; Wood, B.J.; Quezado, M.; Denobile, J.; Pingpank, J.F.; Royal, R.E.; Alexander, H.R.; Seidel, G.; Steinberg, S.M.; et al. A pilot feasibility study of TNFerade biologic with capecitabine and radiation therapy followed by surgical resection for the treatment of rectal cancer. Oncology 2010, 79, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Hecht, J.R.; Farrell, J.J.; Senzer, N.; Nemunaitis, J.; Rosemurgy, A.; Chung, T.; Hanna, N.; Chang, K.J.; Javle, M.; Posner, M.; et al. EUS or percutaneously guided intratumoral TNFerade biologic with 5-fluorouracil and radiotherapy for first-line treatment of locally advanced pancreatic cancer: A phase I/II study. Gastrointest. Endosc. 2012, 75, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.J.; Reid, T.; Senzer, N.; Swisher, S.; Pinto, H.; Hanna, N.; Chak, A.; Soetikno, R. Phase I evaluation of TNFerade biologic plus chemoradiotherapy before esophagectomy for locally advanced resectable esophageal cancer. Gastrointest. Endosc. 2012, 75, 1139–1146. [Google Scholar] [CrossRef] [PubMed]
- Seiwert, T.Y.; Darga, T.; Haraf, D.; Blair, E.A.; Stenson, K.; Cohen, E.E.; Salama, J.K.; Villaflor, V.; Witt, M.E.; Lingen, M.W.; et al. A phase I dose escalation study of Ad GV.EGR.TNF.11D (TNFerade Biologic) with concurrent chemoradiotherapy in patients with recurrent head and neck cancer undergoing reirradiation. Ann. Oncol. 2013, 24, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Herman, J.M.; Wild, A.T.; Wang, H.; Tran, P.T.; Chang, K.J.; Taylor, G.E.; Donehower, R.C.; Pawlik, T.M.; Ziegler, M.A.; Cai, H.; et al. Randomized phase III multi-institutional study of TNFerade biologic with fluorouracil and radiotherapy for locally advanced pancreatic cancer: Final results. J. Clin. Oncol. 2013, 31, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Neri, D.; Sondel, P.M. Immunocytokines for cancer treatment: Past, present and future. Curr. Opin. Immunol. 2016, 40, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Zegers, C.M.; Rekers, N.H.; Quaden, D.H.; Lieuwes, N.G.; Yaromina, A.; Germeraad, W.T.; Wieten, L.; Biessen, E.A.; Boon, L.; Neri, D.; et al. Radiotherapy combined with the immunocytokine L19-IL2 provides long-lasting antitumor effects. Clin. Cancer Res. 2015, 21, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Rekers, N.H.; Olivo Pimentel, V.; Yaromina, A.; Lieuwes, N.G.; Biemans, R.; Zegers, C.M.L.; Germeraad, W.T.V.; Van Limbergen, E.J.; Neri, D.; Dubois, L.J.; et al. The immunocytokine L19-IL2: An interplay between radiotherapy and long-lasting systemic anti-tumour immune responses. Oncoimmunology 2018, 7, e1414119. [Google Scholar] [CrossRef] [PubMed]
- Eckert, F.; Schmitt, J.; Zips, D.; Krueger, M.A.; Pichler, B.J.; Gillies, S.D.; Strittmatter, W.; Handgretinger, R.; Schilbach, K. Enhanced binding of necrosis-targeting immunocytokine NHS-IL12 after local tumour irradiation in murine xenograft models. Cancer Immunol. Immunother. 2016, 65, 1003–1013. [Google Scholar] [CrossRef] [PubMed]
- Eckert, F.; Jelas, I.; Oehme, M.; Huber, S.M.; Sonntag, K.; Welker, C.; Gillies, S.D.; Strittmatter, W.; Zips, D.; Handgretinger, R.; et al. Tumor-targeted IL-12 combined with local irradiation leads to systemic tumor control via abscopal effects in vivo. Oncoimmunology 2017, 6, e1323161. [Google Scholar] [CrossRef] [PubMed]
- Rech, A.J.; Dada, H.; Kotzin, J.J.; Henao-Mejia, J.; Minn, A.J.; Twyman-Saint Victor, C.; Vonderheide, R.H. Radiotherapy and CD40 Activation Separately Augment Immunity to Checkpoint Blockade in Cancer. Cancer Res. 2018, 78, 4282–4291. [Google Scholar] [CrossRef] [PubMed]
- Yasmin-Karim, S.; Bruck, P.T.; Moreau, M.; Kunjachan, S.; Chen, G.Z.; Kumar, R.; Grabow, S.; Dougan, S.K.; Ngwa, W. Radiation and Local Anti-CD40 Generate an Effective in situ Vaccine in Preclinical Models of Pancreatic Cancer. Front. Immunol. 2018, 9, 2030. [Google Scholar] [CrossRef] [PubMed]
- Yokouchi, H.; Yamazaki, K.; Chamoto, K.; Kikuchi, E.; Shinagawa, N.; Oizumi, S.; Hommura, F.; Nishimura, T.; Nishimura, M. Anti-OX40 monoclonal antibody therapy in combination with radiotherapy results in therapeutic antitumor immunity to murine lung cancer. Cancer Sci. 2008, 99, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Gough, M.J.; Crittenden, M.R.; Sarff, M.; Pang, P.; Seung, S.K.; Vetto, J.T.; Hu, H.M.; Redmond, W.L.; Holland, J.; Weinberg, A.D. Adjuvant therapy with agonistic antibodies to CD134 (OX40) increases local control after surgical or radiation therapy of cancer in mice. J. Immunother. 2010, 33, 798–809. [Google Scholar] [CrossRef] [PubMed]
- Niknam, S.; Barsoumian, H.B.; Schoenhals, J.E.; Jackson, H.L.; Yanamandra, N.; Caetano, M.S.; Li, A.; Younes, A.I.; Cadena, A.; Cushman, T.R.; et al. Radiation Followed by OX40 Stimulation Drives Local and Abscopal Antitumor Effects in an Anti-PD1-Resistant Lung Tumor Model. Clin. Cancer Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Moran, A.E.; Kovacsovics-Bankowski, M.; Weinberg, A.D. The TNFRs OX40, 4–1BB, and CD40 as targets for cancer immunotherapy. Curr. Opin. Immunol. 2013, 25, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Siemann, D.W. Augmented antitumor effects of radiation therapy by 4-1BB antibody (BMS-469492) treatment. Anticancer Res. 2006, 26, 3445–3453. [Google Scholar] [PubMed]
- Belcaid, Z.; Phallen, J.A.; Zeng, J.; See, A.P.; Mathios, D.; Gottschalk, C.; Nicholas, S.; Kellett, M.; Ruzevick, J.; Jackson, C.; et al. Focal radiation therapy combined with 4-1BB activation and CTLA-4 blockade yields long-term survival and a protective antigen-specific memory response in a murine glioma model. PLoS ONE 2014, 9, e101764. [Google Scholar] [CrossRef] [PubMed]
- Kroon, P.; Gadiot, J.; Peeters, M.; Gasparini, A.; Deken, M.A.; Yagita, H.; Verheij, M.; Borst, J.; Blank, C.U.; Verbrugge, I. Concomitant targeting of programmed death-1 (PD-1) and CD137 improves the efficacy of radiotherapy in a mouse model of human BRAFV600-mutant melanoma. Cancer Immunol. Immunother. 2016, 65, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Benaduce, A.P.; Brenneman, R.; Schrand, B.; Pollack, A.; Gilboa, E.; Ishkanian, A. 4-1BB Aptamer-Based Immunomodulation Enhances the Therapeutic Index of Radiation Therapy in Murine Tumor Models. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 458–461. [Google Scholar] [CrossRef] [PubMed]
- Demaria, S.; Kawashima, N.; Yang, A.M.; Devitt, M.L.; Babb, J.S.; Allison, J.P.; Formenti, S.C. Immune-mediated inhibition of metastases after treatment with local radiation and CTLA-4 blockade in a mouse model of breast cancer. Clin. Cancer Res. 2005, 11, 728–734. [Google Scholar] [PubMed]
- Slovin, S.F.; Higano, C.S.; Hamid, O.; Tejwani, S.; Harzstark, A.; Alumkal, J.J.; Scher, H.I.; Chin, K.; Gagnier, P.; McHenry, M.B.; et al. Ipilimumab alone or in combination with radiotherapy in metastatic castration-resistant prostate cancer: Results from an open-label, multicenter phase I/II study. Ann. Oncol. 2013, 24, 1813–1821. [Google Scholar] [CrossRef] [PubMed]
- Kwon, E.D.; Drake, C.G.; Scher, H.I.; Fizazi, K.; Bossi, A.; van den Eertwegh, A.J.; Krainer, M.; Houede, N.; Santos, R.; Mahammedi, H.; et al. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (CA184-043): A multicentre, randomised, double-blind, phase 3 trial. Lancet. Oncol. 2014, 15, 700–712. [Google Scholar] [CrossRef]
- Grimaldi, A.M.; Simeone, E.; Giannarelli, D.; Muto, P.; Falivene, S.; Borzillo, V.; Giugliano, F.M.; Sandomenico, F.; Petrillo, A.; Curvietto, M.; et al. Abscopal effects of radiotherapy on advanced melanoma patients who progressed after ipilimumab immunotherapy. Oncoimmunology 2014, 3, e28780. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.A.; Wilhite, T.J.; Balboni, T.A.; Alexander, B.M.; Spektor, A.; Ott, P.A.; Ng, A.K.; Hodi, F.S.; Schoenfeld, J.D. A systematic evaluation of abscopal responses following radiotherapy in patients with metastatic melanoma treated with ipilimumab. Oncoimmunology 2015, 4, e1046028. [Google Scholar] [CrossRef] [PubMed]
- Theurich, S.; Rothschild, S.I.; Hoffmann, M.; Fabri, M.; Sommer, A.; Garcia-Marquez, M.; Thelen, M.; Schill, C.; Merki, R.; Schmid, T.; et al. Local Tumor Treatment in Combination with Systemic Ipilimumab Immunotherapy Prolongs Overall Survival in Patients with Advanced Malignant Melanoma. Cancer Immunol. Res. 2016, 4, 744–754. [Google Scholar] [CrossRef] [PubMed]
- Knisely, J.P.; Yu, J.B.; Flanigan, J.; Sznol, M.; Kluger, H.M.; Chiang, V.L. Radiosurgery for melanoma brain metastases in the ipilimumab era and the possibility of longer survival. J. Neurosurg. 2012, 117, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Twyman-Saint Victor, C.; Rech, A.J.; Maity, A.; Rengan, R.; Pauken, K.E.; Stelekati, E.; Benci, J.L.; Xu, B.; Dada, H.; Odorizzi, P.M.; et al. Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature 2015, 520, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Formenti, S.C.; Rudqvist, N.P.; Golden, E.; Cooper, B.; Wennerberg, E.; Lhuillier, C.; Vanpouille-Box, C.; Friedman, K.; Ferrari de Andrade, L.; Wucherpfennig, K.W.; et al. Radiotherapy induces responses of lung cancer to CTLA-4 blockade. Nat. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; See, A.P.; Phallen, J.; Jackson, C.M.; Belcaid, Z.; Ruzevick, J.; Durham, N.; Meyer, C.; Harris, T.J.; Albesiano, E.; et al. Anti-PD-1 blockade and stereotactic radiation produce long-term survival in mice with intracranial gliomas. Int. J. Radiat. Oncol. Biol. Phys. 2013, 86, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Park, S.S.; Dong, H.; Liu, X.; Harrington, S.M.; Krco, C.J.; Grams, M.P.; Mansfield, A.S.; Furutani, K.M.; Olivier, K.R.; Kwon, E.D. PD-1 Restrains Radiotherapy-Induced Abscopal Effect. Cancer Immunol. Res. 2015, 3, 610–619. [Google Scholar] [CrossRef] [PubMed]
- Sharabi, A.B.; Nirschl, C.J.; Kochel, C.M.; Nirschl, T.R.; Francica, B.J.; Velarde, E.; Deweese, T.L.; Drake, C.G. Stereotactic Radiation Therapy Augments Antigen-Specific PD-1-Mediated Antitumor Immune Responses via Cross-Presentation of Tumor Antigen. Cancer Immunol. Res. 2015, 3, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Dovedi, S.J.; Cheadle, E.J.; Popple, A.L.; Poon, E.; Morrow, M.; Stewart, R.; Yusko, E.C.; Sanders, C.M.; Vignali, M.; Emerson, R.O.; et al. Fractionated Radiation Therapy Stimulates Antitumor Immunity Mediated by Both Resident and Infiltrating Polyclonal T-cell Populations when Combined with PD-1 Blockade. Clin. Cancer Res. 2017, 23, 5514–5526. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Schoenhals, J.E.; Li, A.; Valdecanas, D.R.; Ye, H.; Zang, F.; Tang, C.; Tang, M.; Liu, C.G.; Liu, X.; et al. Suppression of Type I IFN Signaling in Tumors Mediates Resistance to Anti-PD-1 Treatment That Can Be Overcome by Radiotherapy. Cancer Res. 2017, 77, 839–850. [Google Scholar] [CrossRef] [PubMed]
- Barker, C.A.; Postow, M.A.; Kronenberg, S.A.; Ma, J.; Yamada, Y.; Beal, K.; Chan, T.A.; Callahan, M.K.; Wolchok, J.D. Concurrent Radiation Therapy (RT), Ipilimumab (Ipi) and/or Nivolumab (Nivo) on a Phase 1 Clinical Trial. Int. J. Radiat. Oncol. Biol. Phys. 2015, 93, S210–S211. [Google Scholar] [CrossRef]
- Levy, A.; Massard, C.; Soria, J.C.; Deutsch, E. Concurrent irradiation with the anti-programmed cell death ligand-1 immune checkpoint blocker durvalumab: Single centre subset analysis from a phase 1/2 trial. Eur. J. Cancer 2016, 68, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Duffy, A.G.; Makarova-Rusher, O.V.; Kleiner, D.E.; Alewine, C.; Figg, W.D.; Steinberg, S.M.; Levy, E.B.; Krishnasamy, V.P.; Wood, B.J.; Citrin, D.E.; et al. A pilot study of immune checkpoint inhibition in combination with radiation therapy in patients with metastatic pancreatic cancer. J. Clin. Oncol. 2017, 35. [Google Scholar] [CrossRef]
- Lin, S.H.; Lin, Y.; Price, J.; Parker, M.; Gomez, D.R.; Welsh, J.W.; Komaki, R.; Kurie, J.M.; Simon, G.R.; Blumenschein, G.R.; et al. DETERRED: PD-L1 blockade to evaluate the safety of lung cancer therapy using carboplatin, paclitaxel, and radiation combined with MPDL3280A (atezolizumab). J. Clin. Oncol. 2017, 35, 3064. [Google Scholar] [CrossRef]
- Luke, J.J.; Lemons, J.M.; Karrison, T.G.; Pitroda, S.P.; Melotek, J.M.; Zha, Y.; Al-Hallaq, H.A.; Arina, A.; Khodarev, N.N.; Janisch, L.; et al. Safety and Clinical Activity of Pembrolizumab and Multisite Stereotactic Body Radiotherapy in Patients With Advanced Solid Tumors. J. Clin. Oncol. 2018, 36, 1611–1618. [Google Scholar] [CrossRef] [PubMed]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Yokoi, T.; Chiappori, A.; Lee, K.H.; de Wit, M.; et al. Durvalumab after Chemoradiotherapy in Stage III Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 377, 1919–1929. [Google Scholar] [CrossRef] [PubMed]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Kurata, T.; Chiappori, A.; Lee, K.H.; de Wit, M.; et al. Overall Survival with Durvalumab after Chemoradiotherapy in Stage III NSCLC. N. Engl. J. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Shaverdian, N.; Lisberg, A.E.; Bornazyan, K.; Veruttipong, D.; Goldman, J.W.; Formenti, S.C.; Garon, E.B.; Lee, P. Previous radiotherapy and the clinical activity and toxicity of pembrolizumab in the treatment of non-small-cell lung cancer: A secondary analysis of the KEYNOTE-001 phase 1 trial. Lancet Oncol. 2017, 18, 895–903. [Google Scholar] [CrossRef]
- Aras, S.; Zaidi, M.R. TAMeless traitors: Macrophages in cancer progression and metastasis. Br. J. Cancer 2017, 117, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- Pyonteck, S.M.; Akkari, L.; Schuhmacher, A.J.; Bowman, R.L.; Sevenich, L.; Quail, D.F.; Olson, O.C.; Quick, M.L.; Huse, J.T.; Teijeiro, V.; et al. CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nat. Med. 2013, 19, 1264–1272. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Knolhoff, B.L.; Meyer, M.A.; Nywening, T.M.; West, B.L.; Luo, J.; Wang-Gillam, A.; Goedegebuure, S.P.; Linehan, D.C.; DeNardo, D.G. CSF1/CSF1R blockade reprograms tumor-infiltrating macrophages and improves response to T-cell checkpoint immunotherapy in pancreatic cancer models. Cancer Res. 2014, 74, 5057–5069. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.R.; Maute, R.L.; Dulken, B.W.; Hutter, G.; George, B.M.; McCracken, M.N.; Gupta, R.; Tsai, J.M.; Sinha, R.; Corey, D.; et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature 2017, 545, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Hauser, S.H.; Calorini, L.; Wazer, D.E.; Gattoni-Celli, S. Radiation-enhanced expression of major histocompatibility complex class I antigen H-2Db in B16 melanoma cells. Cancer Res. 1993, 53, 1952–1955. [Google Scholar] [PubMed]
- Deng, L.; Liang, H.; Xu, M.; Yang, X.; Burnette, B.; Arina, A.; Li, X.D.; Mauceri, H.; Beckett, M.; Darga, T.; et al. STING-Dependent Cytosolic DNA Sensing Promotes Radiation-Induced Type I Interferon-Dependent Antitumor Immunity in Immunogenic Tumors. Immunity 2014, 41, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Schaue, D.; Ratikan, J.A.; Iwamoto, K.S.; McBride, W.H. Maximizing tumor immunity with fractionated radiation. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, 1306–1310. [Google Scholar] [CrossRef] [PubMed]
- Vanpouille-Box, C.; Alard, A.; Aryankalayil, M.J.; Sarfraz, Y.; Diamond, J.M.; Schneider, R.J.; Inghirami, G.; Coleman, C.N.; Formenti, S.C.; Demaria, S. DNA exonuclease Trex1 regulates radiotherapy-induced tumour immunogenicity. Nat. Commun. 2017, 8, 15618. [Google Scholar] [CrossRef] [PubMed]
- Wild, A.T.; Herman, J.M.; Dholakia, A.S.; Moningi, S.; Lu, Y.; Rosati, L.M.; Hacker-Prietz, A.; Assadi, R.K.; Saeed, A.M.; Pawlik, T.M.; et al. Lymphocyte-Sparing Effect of Stereotactic Body Radiation Therapy in Patients With Unresectable Pancreatic Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Wunderlich, R.; Ernst, A.; Rodel, F.; Fietkau, R.; Ott, O.; Lauber, K.; Frey, B.; Gaipl, U.S. Low and moderate doses of ionizing radiation up to 2 Gy modulate transmigration and chemotaxis of activated macrophages, provoke an anti-inflammatory cytokine milieu, but do not impact upon viability and phagocytic function. Clin. Exp. Immunol. 2015, 179, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.H.; Cook, J.A.; Chandramouli, G.V.; DeGraff, W.; Yan, H.; Zhao, S.; Coleman, C.N.; Mitchell, J.B.; Chuang, E.Y. Gene expression profiling of breast, prostate, and glioma cells following single versus fractionated doses of radiation. Cancer Res. 2007, 67, 3845–3852. [Google Scholar] [CrossRef] [PubMed]
- Klug, F.; Prakash, H.; Huber, P.E.; Seibel, T.; Bender, N.; Halama, N.; Pfirschke, C.; Voss, R.H.; Timke, C.; Umansky, L.; et al. Low-dose irradiation programs macrophage differentiation to an iNOS(+)/M1 phenotype that orchestrates effective T cell immunotherapy. Cancer Cell 2013, 24, 589–602. [Google Scholar] [CrossRef] [PubMed]
- Prakash, H.; Klug, F.; Nadella, V.; Mazumdar, V.; Schmitz-Winnenthal, H.; Umansky, L. Low doses of gamma irradiation potentially modifies immunosuppressive tumor microenvironment by retuning tumor-associated macrophages: Lesson from insulinoma. Carcinogenesis 2016, 37, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Brozyna, A.A.; Jozwicki, W.; Roszkowski, K.; Filipiak, J.; Slominski, A.T. Melanin content in melanoma metastases affects the outcome of radiotherapy. Oncotarget 2016, 7, 17844–17853. [Google Scholar] [CrossRef] [PubMed]
- Sniegocka, M.; Podgorska, E.; Plonka, P.M.; Elas, M.; Romanowska-Dixon, B.; Szczygiel, M.; Zmijewski, M.A.; Cichorek, M.; Markiewicz, A.; Brozyna, A.A.; et al. Transplantable Melanomas in Hamsters and Gerbils as Models for Human Melanoma. Sensitization in Melanoma Radiotherapy-From Animal Models to Clinical Trials. Int. J. Mol. Sci. 2018, 19, 1048. [Google Scholar] [CrossRef] [PubMed]
- Young, K.H.; Baird, J.R.; Savage, T.; Cottam, B.; Friedman, D.; Bambina, S.; Messenheimer, D.J.; Fox, B.; Newell, P.; Bahjat, K.S.; et al. Optimizing Timing of Immunotherapy Improves Control of Tumors by Hypofractionated Radiation Therapy. PLoS ONE 2016, 11, e0157164. [Google Scholar] [CrossRef] [PubMed]
- Simpson, T.R.; Li, F.; Montalvo-Ortiz, W.; Sepulveda, M.A.; Bergerhoff, K.; Arce, F.; Roddie, C.; Henry, J.Y.; Yagita, H.; Wolchok, J.D.; et al. Fc-dependent depletion of tumor-infiltrating regulatory T cells co-defines the efficacy of anti-CTLA-4 therapy against melanoma. J. Exp. Med. 2013, 210, 1695–1710. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.E.; Prell, R.A.; Thalhofer, C.J.; Hurwitz, A.A.; Weinberg, A.D. Engagement of OX40 enhances antigen-specific CD4(+) T cell mobilization/memory development and humoral immunity: Comparison of alphaOX-40 with alphaCTLA-4. J. Immunol. 2001, 167, 6804–6811. [Google Scholar] [CrossRef] [PubMed]
- Vacchelli, E.; Bloy, N.; Aranda, F.; Buque, A.; Cremer, I.; Demaria, S.; Eggermont, A.; Formenti, S.C.; Fridman, W.H.; Fucikova, J.; et al. Trial Watch: Immunotherapy plus radiation therapy for oncological indications. Oncoimmunology 2016, 5, e1214790. [Google Scholar] [CrossRef] [PubMed]
- Bloy, N.; Pol, J.; Manic, G.; Vitale, I.; Eggermont, A.; Galon, J.; Tartour, E.; Zitvogel, L.; Kroemer, G.; Galluzzi, L. Trial Watch: Radioimmunotherapy for oncological indications. Oncoimmunology 2014, 3, e954929. [Google Scholar] [CrossRef] [PubMed]
- Cannarile, M.A.; Weisser, M.; Jacob, W.; Jegg, A.M.; Ries, C.H.; Ruttinger, D. Colony-stimulating factor 1 receptor (CSF1R) inhibitors in cancer therapy. J. Immunother. Cancer 2017, 5, 53. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.D.; More, S.; Rufo, N.; Mece, O.; Sassano, M.L.; Agostinis, P.; Zitvogel, L.; Kroemer, G.; Galluzzi, L. Trial watch: Immunogenic cell death induction by anticancer chemotherapeutics. Oncoimmunology 2017, 6, e1386829. [Google Scholar] [CrossRef] [PubMed]
| Immune Target | Indication | Drug | Phase | Status | Type of RT | Combined with Other Therapies | Clinical Trial # |
|---|---|---|---|---|---|---|---|
| PD-1 | Breast | Pembrolizumab | II | R | EBRT | None | NCT03051672 |
| Carcinoma unknown primary | Pembrolizumab | II | R | HFRT | None | NCT03396471 | |
| Esophago-gastric | Nivolumab | I/II | R | CFRT | Carboplatin/paclitaxel | NCT03278626 | |
| Pembrolizumab | II | R | CFRT | Carboplatin/paclitaxel | NCT03064490 | ||
| Pembrolizumab | II | R | CFRT | Capecitabine | NCT03257163 | ||
| Pembrolizumab | II | R | EBRT | None | NCT02830594 | ||
| GBM | Pembrolizumab | II | NYR | CFRT | None | NCT03661723 | |
| Pembrolizumab | II | R | CFRT | Temozolomide/HSPPC-96 | NCT03018288 | ||
| REGN2810 | I/II | R | EBRT | Temozolomide/gene therapy | NCT03491683 | ||
| HNSCC | Nivolumab | II | NYR | CFRT | None | NCT03715946 | |
| Nivolumab | II | R | CFRT | None | NCT03521570 | ||
| Nivolumab | I/II | R | HFRT | None | NCT03247712 | ||
| Pembrolizumab | II | NYR | CFRT | None | NCT03383094 | ||
| Pembrolizumab | II | R | CFRT/HFRT | None | NCT03085719 d | ||
| Pembrolizumab | II | R | CFRT | None | NCT03057613 | ||
| Pembrolizumab | III | R | CFRT | Cisplatin | NCT03040999 | ||
| Pembrolizumab | II | R | CFRT | Cisplatin | NCT02777385 | ||
| Pembrolizumab | I/II | R | CFRT | Cisplatin | NCT02759575 | ||
| Hodgkin lymphoma | Pembrolizumab | II | R | CFRT | Multiple chemotherapy cocktails | NCT03407144 | |
| Pembrolizumab | II | R | CFRT | None | NCT03179917 | ||
| Kidney | Nivolumab | II | R | SBRT | None | NCT02781506 | |
| Melanoma | Pembrolizumab | I/II | R | CFRT | A-dmDT390-bisFv (UCHT1) immunotoxin | NCT02990416 | |
| Merkel Cell carcinoma | Pembrolizumab | II | R | SBRT | None | NCT03304639 | |
| Nasophary-ngeal carcinoma | Nivolumab | II | R | CFRT | Cisplatin | NCT03267498 | |
| Lymphoma | Pembrolizumab | II | R | CFRT | None | NCT03210662 | |
| NSCLC | Nivolumab | II | R | SBRT | None | NCT03110978 | |
| Pembrolizumab | II | R | CFRT | Carboplatin/paclitaxel/cisplatin/pemetrexed | NCT03631784 | ||
| Pembrolizumab | II | R | CFRT | None | NCT03523702 | ||
| Pembrolizumab | II | R | SBRT | None | NCT03217071 | ||
| Pembrolizumab | I | R | CFRT | Carboplatin/paclitaxel | NCT02621398 t | ||
| Pancreatic | Nivolumab | II | R | SBRT | Cyclophosphamide/GVAX pancreas vaccine | NCT03161379 | |
| Pediatric | REGN2810 | I/II | R | CFRT/HFRT | None | NCT03690869 | |
| Prostate | Nivolumab | I/II | R | CFRT + HDRB | ADT | NCT03543189 | |
| Rectal | Pembrolizumab | II | R | CFRT | Capecitabine/fluorouracil/leucovorin/oxaliplatin | NCT02921256 | |
| SCLC | Nivolumab | I/II | R | 177Lu-DOTA0-Tyr3-Octreotate | None | NCT03325816 m | |
| Pembrolizumab | II | R | CFRT | Cisplatin/carboplatin/etoposide | NCT02934503 | ||
| Sarcoma | Pembrolizumab | I/II | R | CFRT | None | NCT03338959 | |
| Pembrolizumab | II | R | CFRT | None | NCT03092323 | ||
| Urothelial carcinoma | Nivolumab | II | R | CFRT | None | NCT03421652 | |
| Pembrolizumab | II | NYR | Neutron EBRT | None | NCT03486197 m | ||
| Pembrolizumab | II | NYR | HFRT | None | NCT03419130 | ||
| PD-L1 | Cervical | Atezolizumab | II | NYR | SBRT | None | NCT03614949 |
| Esophago-gastric | Durvalumab | II | R | CFRT | Carboplatin/paclitaxel | NCT02962063 | |
| GBM | Atezolizumab | I/II | R | CFRT | Temozolomide | NCT03174197 | |
| Avelumab | II | R | HFRT | None | NCT02968940 | ||
| HNSCC | Avelumab | III | R | CBRT | Cisplatin | NCT02952586 | |
| Lymphoma | Atezolizumab | II | R | CFRT | None | NCT03465891 | |
| Mesothelioma | Avelumab | I/II | R | SBRT | None | NCT03399552 | |
| Metastatic brain (breast) | Atezolizumab | II | R | SRS | None | NCT03483012 | |
| NSCLC | Avelumab | I/II | R | SBRT | None | NCT03050554 | |
| Durvalumab | II | NYR | SBRT | None | NCT03589547 | ||
| Durvalumab | I/II | R | SBRT | None | NCT03148327 | ||
| Durvalumab | II | R | SBRT | None | NCT02904954 | ||
| Ovarian | Avelumab | II | R | SBRT | None | NCT03312114 | |
| Pancreatic | Durvalumab | I/II | R | SBRT | None | NCT03245541 | |
| Urothelial carcinoma | Durvalumab | I/II | R | HFRT | BCG | NCT03317158 | |
| Durvalumab | I/II | R | CFRT | None | NCT02891161 | ||
| Others | Lymphoma | TLR9 agonist SD-101 | I/II | R | CFRT | Ibrutinib | NCT02927964 |
| Combo | Esophago-gastric | Nivolumab Relatlimab | I/II | NYR | SBRT | None | NCT03610711 |
| GBM | Avelumab Epacadostat | I/II | NYR | CFRT | Bevacizumab | NCT03532295 | |
| Ipilimumab Nivolumab | II | R | HFRT | None | NCT03367715 | ||
| Hepato-biliary carcinoma | Durvalumab Tremelimumab | II | R | EBRT | None | NCT03482102 | |
| HNSCC | Durvalumab Tremelimumab | I/II | R | SBRT | None | NCT03618134 | |
| Durvalumab Tremelimumab | I/II | R | SBRT | None | NCT03522584 | ||
| Kidney | Ipilimumab Nivolumab | II | R | SBRT | None | NCT03065179 | |
| Lymphoma | Anti-OX40 antibody BMS 986178 SD-101 | I | R | CFRT | None | NCT03410901 | |
| Epacadostat SD-101 | I/II | R | EBRT | None | NCT03322384 | ||
| Melanoma | Ipilimumab Nivolumab | II | R | HFRT | None | NCT03646617 | |
| Merkel Cell carcinoma | Ipilimumab Nivolumab | II | R | SBRT | None | NCT03071406 | |
| NSCLC | Atezolizumab Nivolumab Pembrolizumab | II | R | HFRT/SBRT | None | NCT03176173 | |
| Durvalumab Tremelimumab | II | R | CFRT | None | NCT03237377 | ||
| Intralesional IL-2 Nivolumab Pembrolizumab | I | R | HFRT | None | NCT03224871 | ||
| Ipilimumab Nivolumab | I/II | NYR | CFRT | Platinum-based chemotherapy | NCT03663166 | ||
| Ipilimumab Nivolumab | I/II | R | HFRT | None | NCT03168464 | ||
| Pancreatic | Cabiralizumab Nivolumab | II | R | SBRT | None | NCT03599362 | |
| Prostate | Pembrolizumab SD-101 | II | R | SBRT | Leuprolide/abiraterone/prednisone | NCT03007732 | |
| Rectal | Epacadostat Pembrolizumab | I/II | NYR | CFRT | Capecitabine/oxaliplatin | NCT03516708 | |
| Sarcoma | Durvalumab Tremelimumab | I/II | R | CFRT | None | NCT03116529 | |
| Ipilimumab Nivolumab | II | R | CFRT | None | NCT03307616 | ||
| SCLC | Ipilimumab Nivolumab | I | R | SBRT | None | NCT03223155 t | |
| Urothelial carcinoma | Durvalumab Tremelimumab | II | NYR | CFRT | None | NCT03601455 | |
| Uveal melanoma | Ipilimumab Nivolumab | I/II | R | Yttrium 90 | None | NCT02913417 m | |
| Multiple sites | Atezolizumab Nivolumab | II | R | HFRT | None | NCT03115801 | |
| Atezolizumab Nivolumab Pembrolizumab | II | R | HFRT | Nelfinavir | NCT03050060 | ||
| Atezolizumab Nivolumab Pembrolizumab | II | R | SBRT | None | NCT03313804 | ||
| Cabiralizumab Nivolumab Urelumab | I | R | SBRT | None | NCT03431948 | ||
| Durvalumab Tremelimumab | II | R | CFRT/HFRT | None | NCT02888743 d | ||
| Ipilimumab Nivolumab | II | R | EBRT | None | NCT03104439 | ||
| Pembrolizumab ADV/HSV-tk | II | R | SBRT | Valacyclovir | NCT03004183 | ||
| Pembrolizumab IL-2 | I/II | NYR | HFRT | None | NCT03474497 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.-J.; Haffty, B. Radiotherapy as a New Player in Immuno-Oncology. Cancers 2018, 10, 515. https://doi.org/10.3390/cancers10120515
Wang S-J, Haffty B. Radiotherapy as a New Player in Immuno-Oncology. Cancers. 2018; 10(12):515. https://doi.org/10.3390/cancers10120515
Chicago/Turabian StyleWang, Shang-Jui, and Bruce Haffty. 2018. "Radiotherapy as a New Player in Immuno-Oncology" Cancers 10, no. 12: 515. https://doi.org/10.3390/cancers10120515
APA StyleWang, S.-J., & Haffty, B. (2018). Radiotherapy as a New Player in Immuno-Oncology. Cancers, 10(12), 515. https://doi.org/10.3390/cancers10120515




