Combination Therapy after TACE for Hepatocellular Carcinoma with Macroscopic Vascular Invasion: Stereotactic Body Radiotherapy versus Sorafenib
Abstract
1. Introduction
2. Results
2.1. Baseline Characteristics of the TACE–SBRT and TACE–Sorafenib Groups
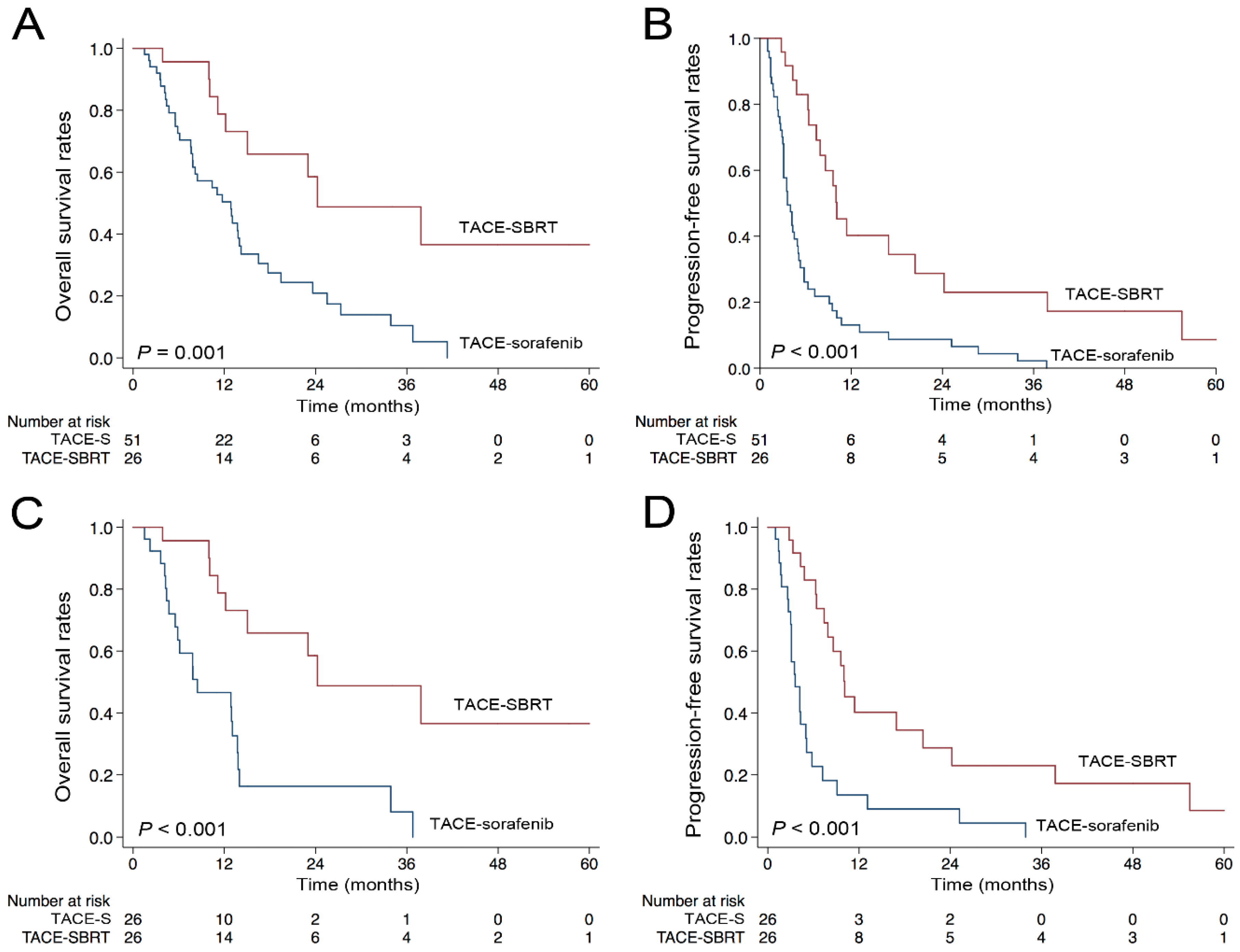
2.2. OS and PFS in the Whole Population
2.3. One-to-One Propensity Score Analysis
2.4. Follow-Up Treatments
2.5. Toxicity of SBRT
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Transarterial Chemoembolization
4.3. Stereotactic Body Radiotherapy
4.4. Sorafenib Administration
4.5. Follow-Up and Endpoints
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Llovet, J.M.; Bustamante, J.; Castells, A.; Vilana, R.; Ayuso Mdel, C.; Sala, M.; Bru, C.; Rodes, J.; Bruix, J. Natural history of untreated nonsurgical hepatocellular carcinoma: Rationale for the design and evaluation of therapeutic trials. Hepatology 1999, 29, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Chen, S.; Wei, M.; Lin, M.; Jiang, C.; Mei, J.; Li, B.; Wang, Y.; Li, J.; Xie, X.; et al. Advanced Recurrent Hepatocellular Carcinoma: Treatment with Sorafenib Alone or in Combination with Transarterial Chemoembolization and Radiofrequency Ablation. Radiology 2018, 287, 705–714. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver; European Organisation for Research and Treatment of Cancer. EASL-EORTC clinical practice guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2012, 56, 908–943. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Raoul, J.L.; Sherman, M.; Mazzaferro, V.; Bolondi, L.; Craxi, A.; Galle, P.R.; Santoro, A.; Beaugrand, M.; Sangiovanni, A.; et al. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma: Subanalyses of a phase III trial. J. Hepatol. 2012, 57, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Pawlik, T.M.; Reyes, D.K.; Cosgrove, D.; Kamel, I.R.; Bhagat, N.; Geschwind, J.F. Phase II trial of sorafenib combined with concurrent transarterial chemoembolization with drug-eluting beads for hepatocellular carcinoma. J. Clin. Oncol. 2011, 29, 3960–3967. [Google Scholar] [CrossRef] [PubMed]
- Abou-Alfa, G.K. TACE and sorafenib: A good marriage? J. Clin. Oncol. 2011, 29, 3949–3952. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Koh, Y.H.; Kim, H.B.; Kim, H.Y.; An, S.; Choi, J.I.; Woo, S.M.; Nam, B.H. Phase II study of concurrent transarterial chemoembolization and sorafenib in patients with unresectable hepatocellular carcinoma. J. Hepatol. 2012, 56, 1336–1342. [Google Scholar] [CrossRef]
- Choi, G.H.; Shim, J.H.; Kim, M.J.; Ryu, M.H.; Ryoo, B.Y.; Kang, Y.K.; Shin, Y.M.; Kim, K.M.; Lim, Y.S.; Lee, H.C. Sorafenib alone versus sorafenib combined with transarterial chemoembolization for advanced-stage hepatocellular carcinoma: Results of propensity score analyses. Radiology 2013, 269, 603–611. [Google Scholar] [CrossRef]
- Jiang, H.; Meng, Q.; Tan, H.; Pan, S.; Sun, B.; Xu, R.; Sun, X. Antiangiogenic therapy enhances the efficacy of transcatheter arterial embolization for hepatocellular carcinomas. Int. J. Cancer 2007, 121, 416–424. [Google Scholar] [CrossRef]
- Chino, F.; Stephens, S.J.; Choi, S.S.; Marin, D.; Kim, C.Y.; Morse, M.A.; Godfrey, D.J.; Czito, B.G.; Willett, C.G.; Palta, M. The role of external beam radiotherapy in the treatment of hepatocellular cancer. Cancer 2018. [Google Scholar] [CrossRef]
- Yoon, S.M.; Ryoo, B.Y.; Lee, S.J.; Kim, J.H.; Shin, J.H.; An, J.H.; Lee, H.C.; Lim, Y.S. Efficacy and Safety of Transarterial Chemoembolization Plus External Beam Radiotherapy vs. Sorafenib in Hepatocellular Carcinoma with Macroscopic Vascular Invasion: A Randomized Clinical Trial. JAMA Oncol. 2018, 4, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, Y.; Yoshida, K.; Nishimura, H.; Ejima, Y.; Miyawaki, D.; Uezono, H.; Ishihara, T.; Mayahara, H.; Fukumoto, T.; Ku, Y.; et al. Efficacy of stereotactic body radiotherapy for hepatocellular carcinoma with portal vein tumor thrombosis/inferior vena cava tumor thrombosis: Evaluation by comparison with conventional three-dimensional conformal radiotherapy. J. Radiat. Res. 2016, 57, 512–523. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Nie, Q.; Du, R.; Zhang, L.; Zhang, J.; Li, Q.; Li, J.; Qi, W. Stereotactic body radiotherapy combined with transarterial chemoembolization for hepatocellular carcinoma with portal vein tumor thrombosis. Mol. Clin. Oncol. 2014, 2, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.L.; Chong, C.C.; Chan, A.W.; Poon, D.M.; Chok, K.S. Management of hepatocellular carcinoma with portal vein tumor thrombosis: Review and update at 2016. World J. Gastroenterol. 2016, 22, 7289–7300. [Google Scholar] [CrossRef] [PubMed]
- Niu, Z.J.; Ma, Y.L.; Kang, P.; Ou, S.Q.; Meng, Z.B.; Li, Z.K.; Qi, F.; Zhao, C. Transarterial chemoembolization compared with conservative treatment for advanced hepatocellular carcinoma with portal vein tumor thrombus: Using a new classification. Med. Oncol. 2012, 29, 2992–2997. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Guo, R.P.; Lai, E.C.; Zhang, Y.J.; Lau, W.Y.; Chen, M.S.; Shi, M. Transarterial chemoembolization for unresectable hepatocellular carcinoma with portal vein tumor thrombosis: A prospective comparative study. Ann. Surg. Oncol. 2011, 18, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Katsanos, K.; Kitrou, P.; Spiliopoulos, S.; Maroulis, I.; Petsas, T.; Karnabatidis, D. Comparative effectiveness of different transarterial embolization therapies alone or in combination with local ablative or adjuvant systemic treatments for unresectable hepatocellular carcinoma: A network meta-analysis of randomized controlled trials. PLoS ONE 2017, 12, e0184597. [Google Scholar] [CrossRef]
- Kudo, M.; Matsui, O.; Izumi, N.; Iijima, H.; Kadoya, M.; Imai, Y.; Okusaka, T.; Miyayama, S.; Tsuchiya, K.; Ueshima, K.; et al. JSH Consensus-Based Clinical Practice Guidelines for the Management of Hepatocellular Carcinoma: 2014 Update by the Liver Cancer Study Group of Japan. Liver Cancer 2014, 3, 458–468. [Google Scholar] [CrossRef]
- Yim, H.J.; Suh, S.J.; Um, S.H. Current management of hepatocellular carcinoma: An Eastern perspective. World J. Gastroenterol. 2015, 21, 3826–3842. [Google Scholar] [CrossRef]
- Qu, X.D.; Chen, C.S.; Wang, J.H.; Yan, Z.P.; Chen, J.M.; Gong, G.Q.; Liu, Q.X.; Luo, J.J.; Liu, L.X.; Liu, R.; et al. The efficacy of TACE combined sorafenib in advanced stages hepatocellullar carcinoma. BMC Cancer 2012, 12, 263. [Google Scholar] [CrossRef]
- Cheng, A.L.; Kang, Y.K.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: A phase III randomised, double-blind, placebo-controlled trial. Lancet 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Zhu, K.; Chen, J.; Lai, L.; Meng, X.; Zhou, B.; Huang, W.; Cai, M.; Shan, H. Hepatocellular carcinoma with portal vein tumor thrombus: Treatment with transarterial chemoembolization combined with sorafenib—A retrospective controlled study. Radiology 2014, 272, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, K.; Wang, M.; Yang, G.; Ye, X.; Wu, M.; Cheng, S. Transarterial chemoembolization (TACE) combined with sorafenib versus TACE for hepatocellular carcinoma with portal vein tumor thrombus: A systematic review and meta-analysis. Oncotarget 2017, 8, 29416–29427. [Google Scholar] [CrossRef] [PubMed]
- Costentin, C.E.; Ferrone, C.R.; Arellano, R.S.; Ganguli, S.; Hong, T.S.; Zhu, A.X. Hepatocellular Carcinoma with Macrovascular Invasion: Defining the Optimal Treatment Strategy. Liver Cancer 2017, 6, 360–374. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, K.; Naqvi, I.H.; Mahmood, A.; Muhammad, S.; Muhammad, N. Deal a death blow! HCC in cirrhotics–thrombotic complications: Their frequency, characteristics, and risk factors. Prz. Gastroenterol. 2018, 13, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Izaki, K.; Sugimoto, K.; Mayahara, H.; Morita, Y.; Yoden, E.; Matsumoto, S.; Soejima, T.; Sugimura, K. Prospective trial of combined transcatheter arterial chemoembolization and three-dimensional conformal radiotherapy for portal vein tumor thrombus in patients with unresectable hepatocellular carcinoma. Int. J. Radiat. Oncol. Boil. Phys. 2003, 57, 113–119. [Google Scholar] [CrossRef]
- Zhang, R.; Shen, L.; Zhao, L.; Guan, Z.; Chen, Q.; Li, W. Combined transarterial chemoembolization and microwave ablation versus transarterial chemoembolization in BCLC stage B hepatocellular carcinoma. Diagn. Interv. Radiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Wei, M.; Chen, S.; Lin, M.; Jiang, C.; Mei, J.; Li, B.; Wang, Y.; Li, J.; Xie, X.; et al. Combined transcatheter arterial chemoembolization and radiofrequency ablation versus hepatectomy for recurrent hepatocellular carcinoma after initial surgery: A propensity score matching study. Eur. Radiol. 2018, 28, 3522–3531. [Google Scholar] [CrossRef]
- Kim, K.J.; Kim, J.H.; Lee, S.J.; Lee, E.J.; Shin, E.C.; Seong, J. Radiation improves antitumor effect of immune checkpoint inhibitor in murine hepatocellular carcinoma model. Oncotarget 2017, 8, 41242–41255. [Google Scholar] [CrossRef]
- Riou, O.; Llacer Moscardo, C.; Fenoglietto, P.; Deshayes, E.; Tetreau, R.; Molinier, J.; Lenglet, A.; Assenat, E.; Ychou, M.; Guiu, B.; et al. SBRT planning for liver metastases: A focus on immobilization, motion management and planning imaging techniques. Rep. Pract. Oncol. Radiother. 2017, 22, 103–110. [Google Scholar] [CrossRef]
- Sterzing, F.; Brunner, T.B.; Ernst, I.; Baus, W.W.; Greve, B.; Herfarth, K.; Guckenberger, M. Stereotactic body radiotherapy for liver tumors: Principles and practical guidelines of the DEGRO Working Group on Stereotactic Radiotherapy. Strahlentherapie und Onkologie 2014, 190, 872–881. [Google Scholar] [CrossRef] [PubMed]
- Xi, M.; Zhang, L.; Zhao, L.; Li, Q.Q.; Guo, S.P.; Feng, Z.Z.; Deng, X.W.; Huang, X.Y.; Liu, M.Z. Effectiveness of stereotactic body radiotherapy for hepatocellular carcinoma with portal vein and/or inferior vena cava tumor thrombosis. PLoS ONE 2013, 8, e63864. [Google Scholar] [CrossRef] [PubMed]
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin. Liver Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef] [PubMed]

| Variable | Before PSM | After PSM | ||||
|---|---|---|---|---|---|---|
| TACE–sorafenib (n = 51) | TACE–SBRT (n = 26) | p | TACE–sorafenib (n = 26) | TACE–SBRT (n = 26) | p | |
| Gender | 0.657 * | 1.000 * | ||||
| Male | 46 (90.2) | 25 (96.2) | 24 (92.3) | 25 (96.2) | ||
| Female | 5 (9.8) | 1 (3.8) | 2 (7.7) | 1 (3.8) | ||
| Age (years) | 0.012 | 0.532 | ||||
| <50 | 27 (52.9) | 6 (23.1) | 8 (26.7) | 6 (23.1) | ||
| ≥50 | 24 (47.1) | 20 (76.9) | 22 (73.3) | 20 (76.9) | ||
| Child–Pugh Class | 1.000 | 0.610 * | ||||
| A | 48 (94.1) | 25 (96.2) | 23 (88.5) | 25 (96.2) | ||
| B | 3 (5.9) | 1 (3.8) | 3 (11.5) | 1 (3.8) | ||
| Cause of disease | 0.338 * | 1.000 * | ||||
| HBV infection | 51 (100.0) | 25 (96.2) | 26 (100.0) | 25 (96.2) | ||
| HCV infection | 0 (0.0) | 1 (3.8) | 0 (0.0) | 1 (3.8) | ||
| Disease Onset | 0.026 * | 0.159 | ||||
| Initial | 48 (94.1) | 19 (73.1) | 23 (88.5) | 19 (73.1) | ||
| Recurrent | 3 (5.9) | 7 (26.9) | 3 (11.5) | 7 (26.9) | ||
| Tumor Size | 0.344 * | 0.465 * | ||||
| <5 cm | 7 (13.7) | 6 (23.1) | 3 (11.5) | 6 (23.1) | ||
| ≥5 cm | 44 (86.3) | 20 (76.9) | 23 (88.5) | 20 (76.9) | ||
| Number of Tumors | 0.005 | 0.548 | ||||
| Single | 20 (39.2) | 19 (73.1) | 17 (65.4) | 19 (73.1) | ||
| Multiple | 31 (60.8) | 7 (26.9) | 9 (34.6) | 7 (26.9) | ||
| AFP level (ng/mL) | 0.164 | 0.405 | ||||
| ≤400 | 19 (37.3) | 14 (53.8) | 11 (42.3) | 14 (53.8) | ||
| >400 | 7 (62.7) | 12 (46.2) | 15 (57.7) | 12 (46.2) | ||
| Type of PVTT | 0.449 ** | 0.864 ** | ||||
| Absent | 3 (5.9) | 2 (7.7) | 0 (0.0) | 2 (7.7) | ||
| Type I/II | 34 (66.7) | 14 (53.8) | 18 (69.2) | 14 (53.8) | ||
| Type III/IV | 14 (27.5) | 10 (38.5) | 8 (30.8) | 10 (38.5) | ||
| HV or IVC invasion | 0.526 * | 0.419 * | ||||
| Absent | 44 (86.3) | 21 (80.8) | 24 (92.3) | 21 (80.8) | ||
| Present | 7 (13.7) | 5 (19.2) | 2 (7.7) | 5 (19.2) | ||
| Variable | No. of Cases | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | ||
| Gender (Female vs. Male) | 6/71 | 0.92 (0.36–2.35) | 0.866 | – | – |
| Age (≥50 vs. <50) | 44/33 | 0.64 (0.36–1.15) | 0.136 | – | – |
| Child–Pugh Class (B vs. A) | 4/73 | 2.56 (0.77–8.49) | 0.125 | – | – |
| Disease Onset (Recurrent vs. Initial) | 10/67 | 0.42 (0.13–1.36) | 0.421 | – | – |
| Liver Tumor Size (≥5 cm vs. <5 cm) | 64/13 | 3.37 (1.20–9.43) | 0.021 | 2.57 (0.91–7.29) | 0.076 |
| Number of Tumors (Multiple vs. Single) | 39/38 | 0.99 (0.56–1.75) | 0.979 | – | – |
| AFP Level (>400 ng/mL vs. ≤400 ng/mL) | 44/33 | 1.62 (0.89–2.95) | 0.114 | – | – |
| Type of PVTT (Type III/IV vs. Type I/II/Absent) | 24/53 | 1.03 (0.56–1.91) | 0.917 | - | - |
| HV or IVC invasion (Present vs. Absent) | 12/65 | 0.70 (0.30–1.66) | 0.701 | - | - |
| Treatment (TACE–SBRT vs. TACE–sorafenib) | 26/51 | 0.30 (0.14–0.64) | 0.002 | 0.36 (0.17–0.75) | 0.007 |
| Variable | No. of Cases | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | ||
| Gender (Female vs. <Male) | 6/71 | 0.97 (0.39–2.44) | 0.948 | – | – |
| Age (≥50 vs. <50) | 44/33 | 0.74 (0.45–1.20) | 0.220 | – | – |
| Child–Pugh Class (B vs. A) | 4/73 | 1.90 (0.68–5.30) | 0.223 | – | – |
| Disease Onset (Recurrent vs. Initial) | 10/67 | 0.62 (0.28–1.37) | 0.234 | – | – |
| Liver Tumor Size (≥5 cm vs. <5 cm) | 64/13 | 2.55 (1.21–5.39) | 0.014 | 2.19 (1.03–4.64) | 0.041 |
| Number of Tumors (Multiple vs. Single) | 39/38 | 1.43 (0.88–2.32) | 0.152 | – | – |
| AFP Level (>400 ng/mL vs. ≤400 ng/mL) | 44/33 | 1.50 (0.88–2.38) | 0.141 | – | – |
| Type of PVTT (Type III/IV vs. Type I/II/Absent) | 24/53 | 0.75 (0.44–1.29) | 0.301 | – | – |
| HV or IVC invasion (Present vs. Absent) | 12/65 | 0.88 (0.45–1.72) | 0.702 | – | – |
| Treatment (TACE–SBRT vs. TACE–sorafenib) | 26/51 | 0.32 (0.18–0.58) | <0.001 | 0.35 (0.20–0.62) | <0.001 |
| Treatment | CR (n) | PR (n) | SD (n) | PD (n) | NA | RR (%) | p * |
|---|---|---|---|---|---|---|---|
| Overall Response | 0.139 | ||||||
| TACE–SBRT | 1 | 10 | 5 | 7 | 3 | 42.3 | |
| TACE–sorafenib | 1 | 5 | 6 | 8 | 5 | 23.1 | |
| Response of Hepatic Lesions | 0.575 | ||||||
| TACE–SBRT | 4 | 8 | 5 | 6 | 3 | 46.2 | |
| TACE–sorafenib | 2 | 8 | 3 | 7 | 5 | 38.5 | |
| Response of Macrovascular Invasion | / | ||||||
| TACE–SBRT | 3 | 18 a | 2 | 3 | / | ||
| TACE–sorafenib | 1 | 15 a | 4 | 5 | / | ||
| Categories | TACE–sorafenib (n, %) | TACE–SBRT (n, %) | p |
|---|---|---|---|
| Surgical resection | 0 (0.0) | 1 (3.8) | 1.000 * |
| Ablative therapies | 9 (34.6) | 6 (23.1) | 0.358 |
| Iodine 125 seed implantation | 1 (3.8) | 0 (0.0) | 1.000 * |
| Cytokine-induced killer cells infusion | 0 (0.0) | 1 (3.8) | 1.000 * |
| Intra-arterial infusion | 0 (0.0) | 1 (3.8) | 1.000 * |
| Category and Grade * | Number (%) |
|---|---|
| Liver enzyme | |
| 2 | 1 (3.8) |
| 3 | 1 (3.8) |
| Bilirubin | |
| 2 | 2 (7.7) |
| 3 | 1 (3.8) |
| Albumin | |
| 2 | 1 (3.8) |
| 3 | 0 (0.0) |
| Leukocyte | |
| 2 | 3 (11.5) |
| 3 | 3 (11.5) |
| Platelets | |
| 2 | 4 (15.4) |
| 3 | 3 (11.5) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, L.; Xi, M.; Zhao, L.; Zhang, X.; Wang, X.; Huang, Z.; Chen, Q.; Zhang, T.; Shen, J.; Liu, M.; et al. Combination Therapy after TACE for Hepatocellular Carcinoma with Macroscopic Vascular Invasion: Stereotactic Body Radiotherapy versus Sorafenib. Cancers 2018, 10, 516. https://doi.org/10.3390/cancers10120516
Shen L, Xi M, Zhao L, Zhang X, Wang X, Huang Z, Chen Q, Zhang T, Shen J, Liu M, et al. Combination Therapy after TACE for Hepatocellular Carcinoma with Macroscopic Vascular Invasion: Stereotactic Body Radiotherapy versus Sorafenib. Cancers. 2018; 10(12):516. https://doi.org/10.3390/cancers10120516
Chicago/Turabian StyleShen, Lujun, Mian Xi, Lei Zhao, Xuhui Zhang, Xiuchen Wang, Zhimei Huang, Qifeng Chen, Tianqi Zhang, Jingxian Shen, Mengzhong Liu, and et al. 2018. "Combination Therapy after TACE for Hepatocellular Carcinoma with Macroscopic Vascular Invasion: Stereotactic Body Radiotherapy versus Sorafenib" Cancers 10, no. 12: 516. https://doi.org/10.3390/cancers10120516
APA StyleShen, L., Xi, M., Zhao, L., Zhang, X., Wang, X., Huang, Z., Chen, Q., Zhang, T., Shen, J., Liu, M., & Huang, J. (2018). Combination Therapy after TACE for Hepatocellular Carcinoma with Macroscopic Vascular Invasion: Stereotactic Body Radiotherapy versus Sorafenib. Cancers, 10(12), 516. https://doi.org/10.3390/cancers10120516





