Effects of Annealing Temperature on Optical Band Gap of Sol-gel Tungsten Trioxide Films
Abstract
:1. Introduction
2. Materials and Methods
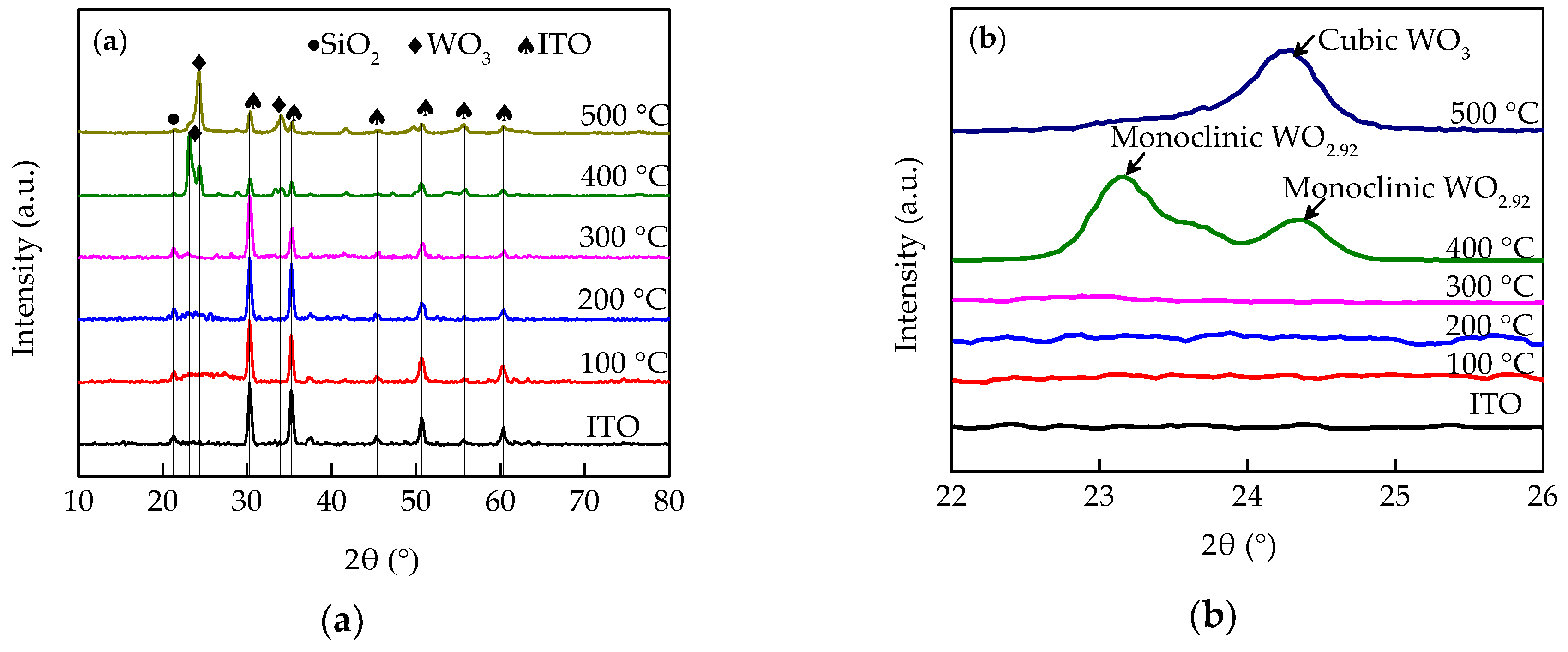
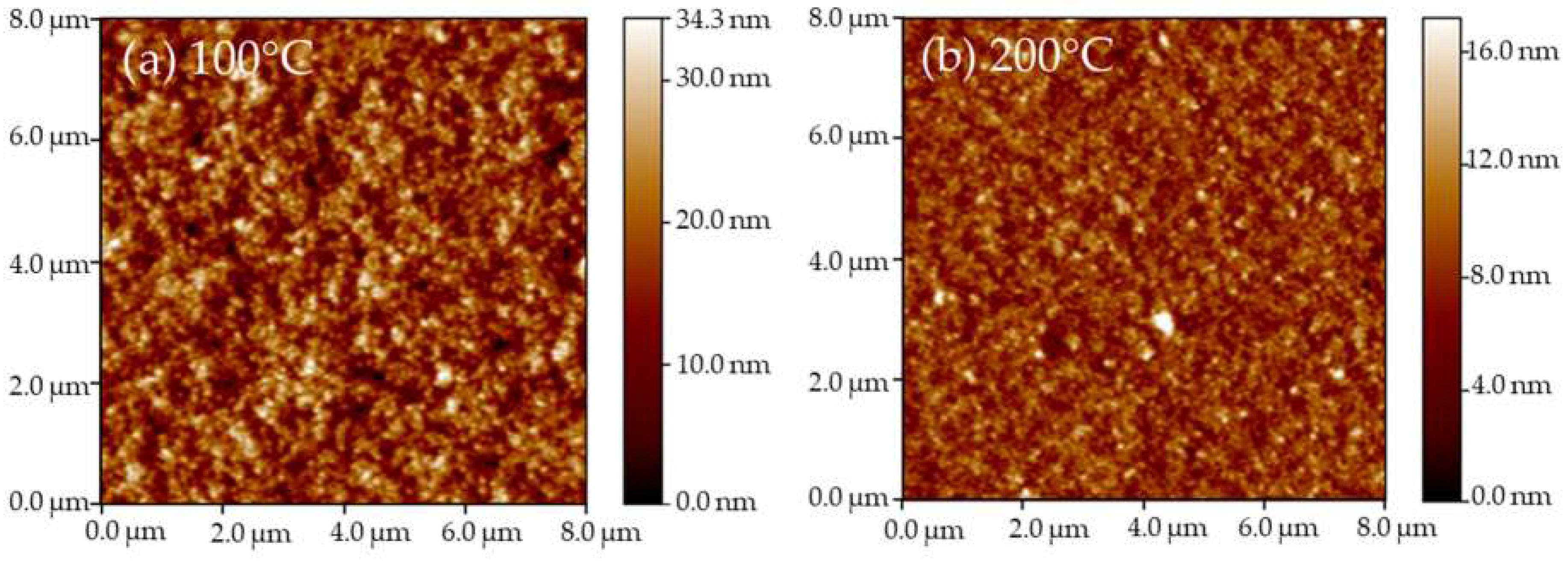
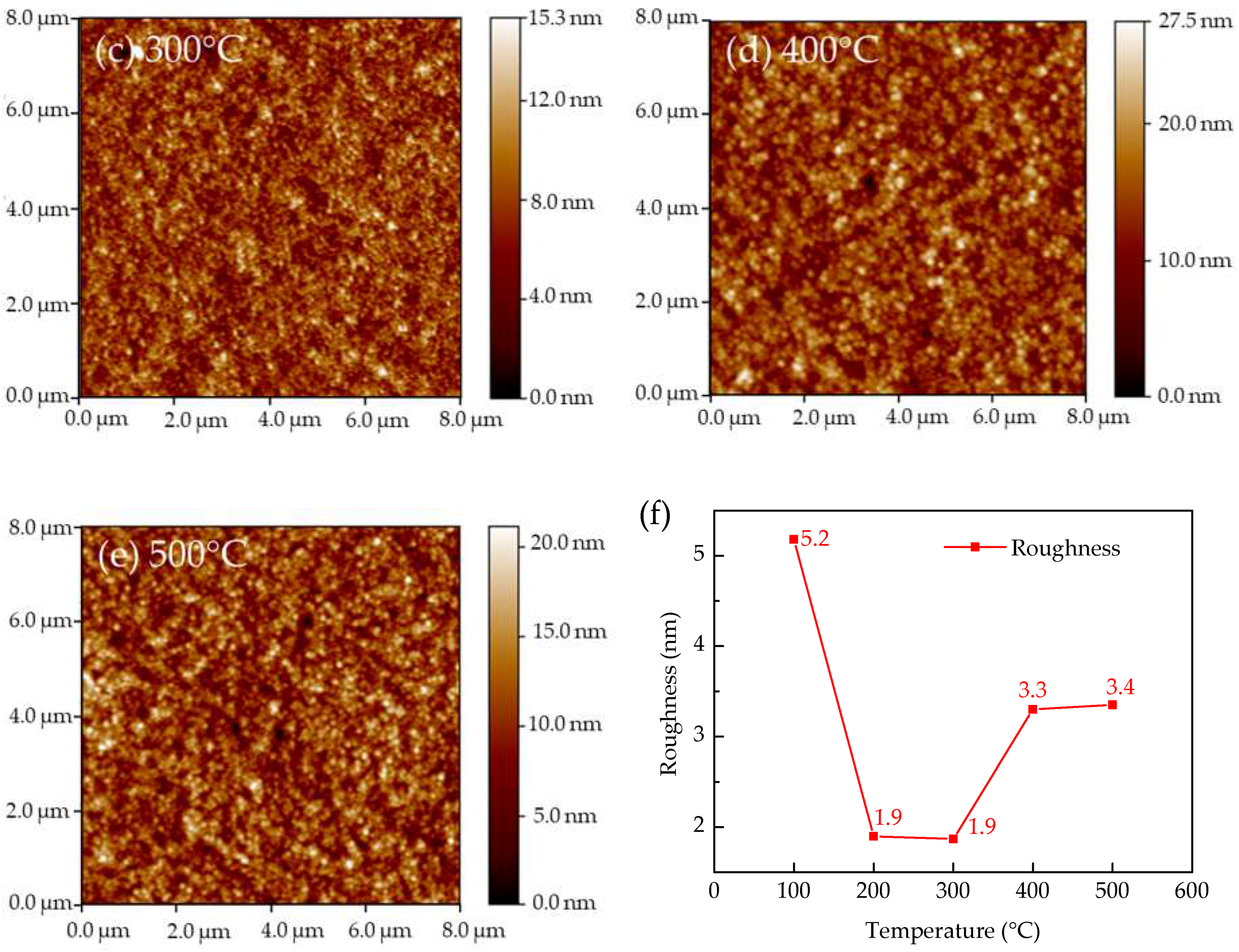
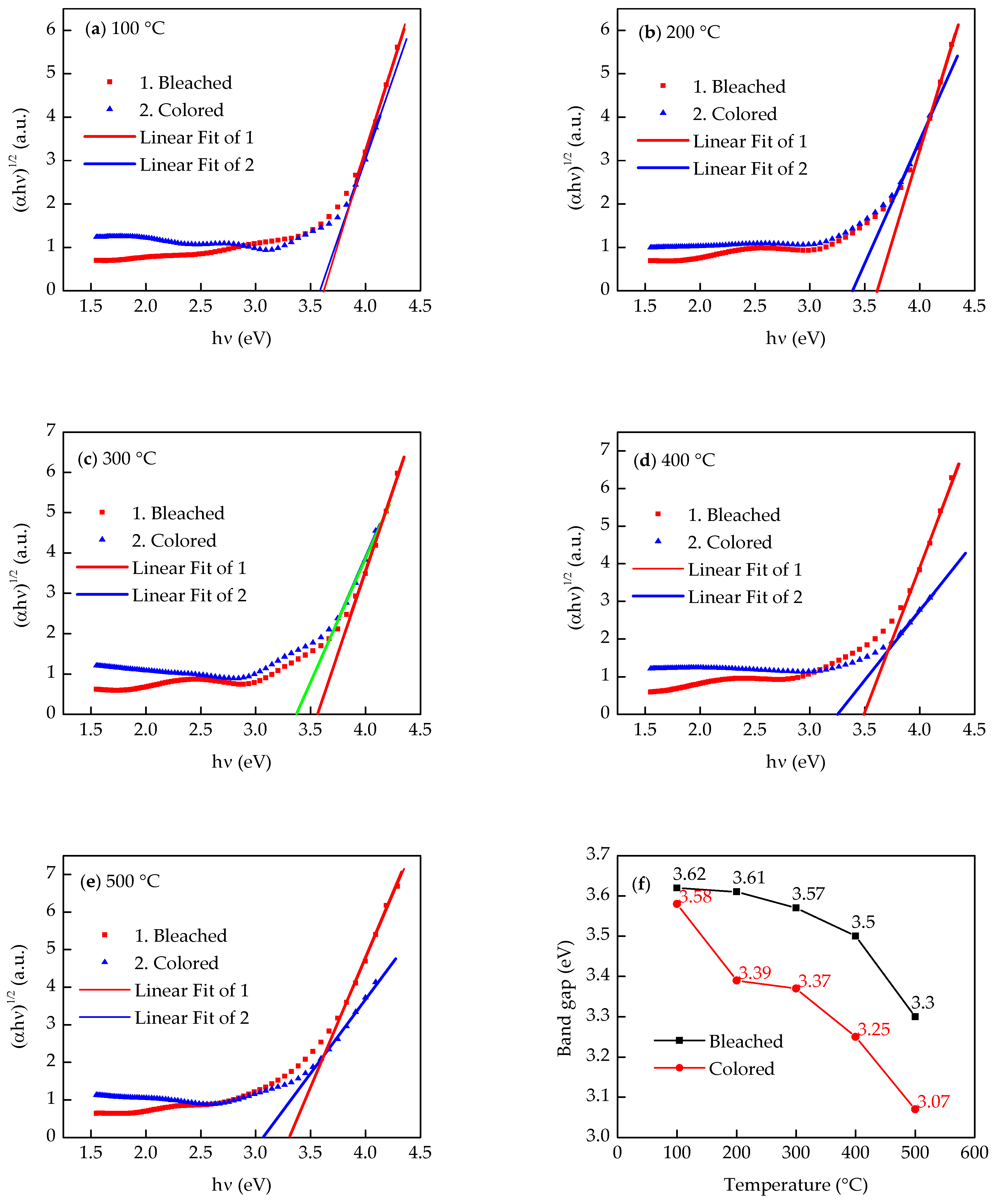
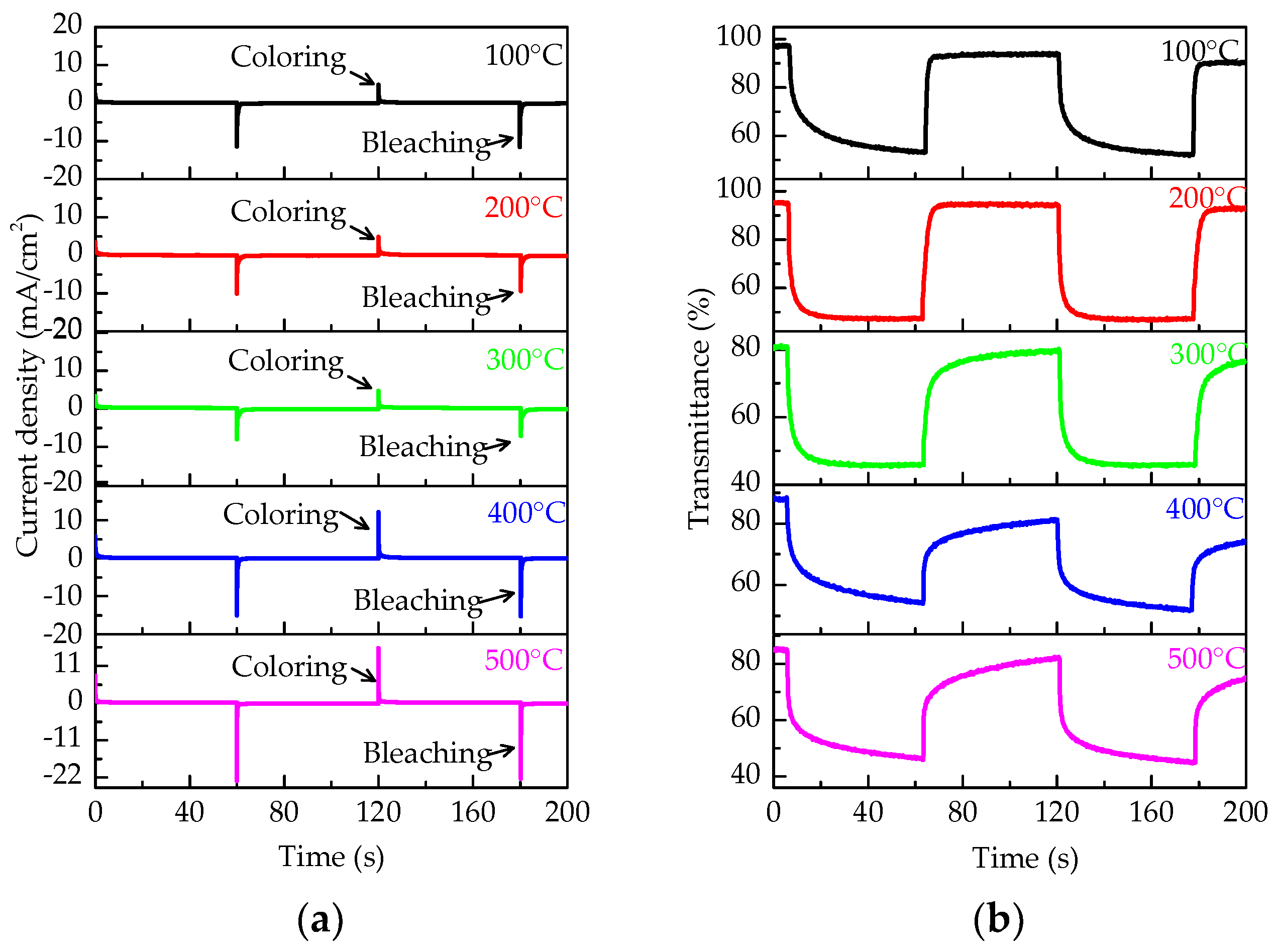
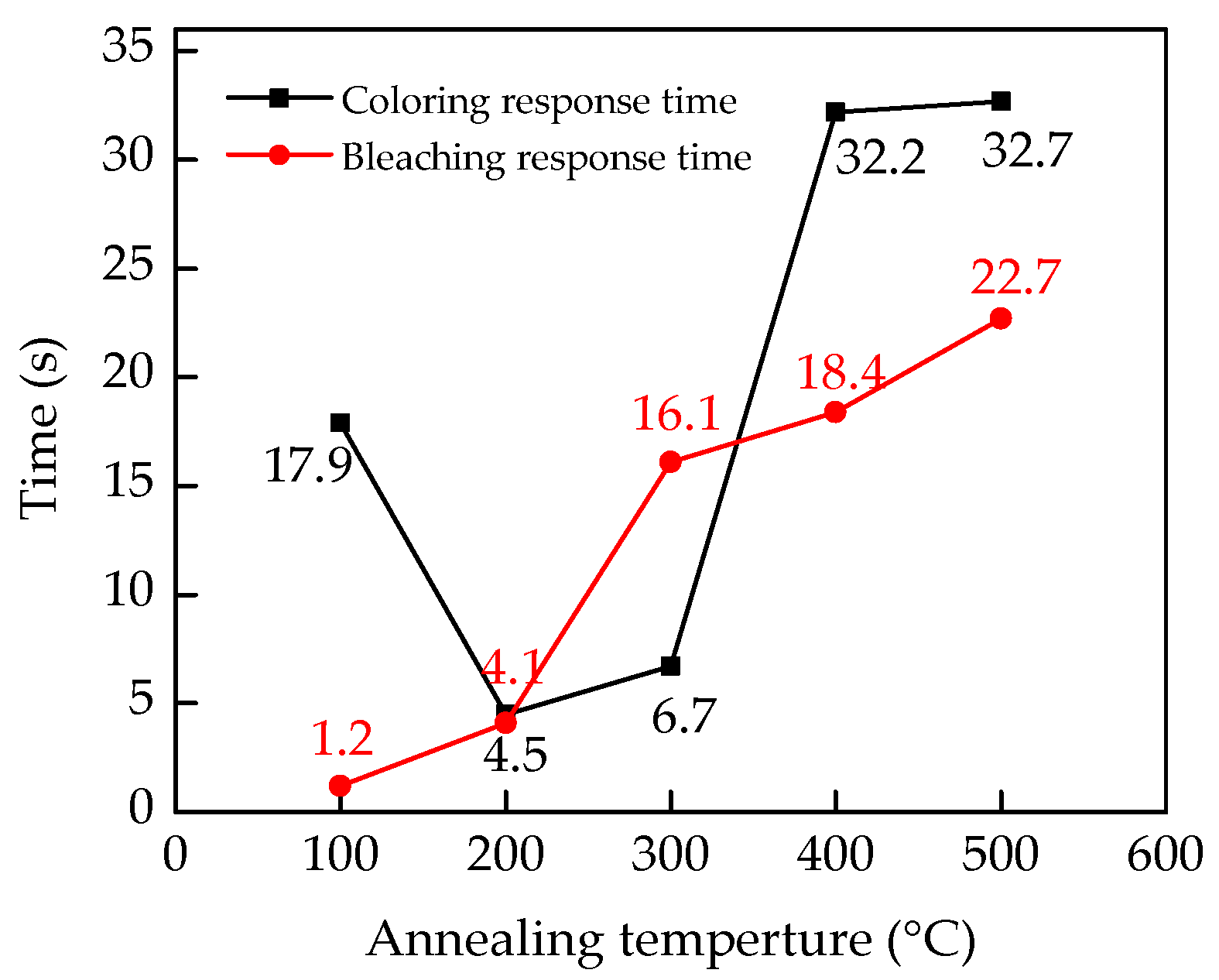
3. Results and Discussions
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Hill, J.C.; Choi, K.S. Effect of electrolytes on the selectivity and stability of n-type WO3 photoelectrodes for use in solar water oxidation. J. Phys. Chem. C 2012, 116, 7612–7620. [Google Scholar] [CrossRef]
- Leidinger, M.; Huotari, J.; Sauerwald, T.; Lappalainen, J.; Schütze, A. Selective detection of naphthalene with nanostructured WO3 gas sensors prepared by pulsed laser deposition. J. Sens. Sens. Syst. 2016, 5, 147–156. [Google Scholar] [CrossRef]
- Sotelo-Vazquez, C.; Quesada-Cabrera, R.; Ling, M.; Scanlon, D.O.; Kafizas, A.; Thakur, P.K.; Lee, T.L.; Taylor, A.; Watson, G.W.; Palgrave, R.G. Photocatalysis: Evidence and effect of photogenerated charge transfer for enhanced photocatalysis in WO3/TiO2 heterojunction films: A computational and experimental study. Adv. Funct. Mater. 2017, 27, 1605413. [Google Scholar] [CrossRef]
- Hara, K.; Zhao, Z.G.; Cui, Y.; Miyauchi, M.; Miyashita, M.; Mori, S. Nanocrystalline electrodes based on nanoporous-walled WO3 nanotubes for organic-dye-sensitized solar cells. Langmuir 2011, 27, 12730–12736. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Valentin, C.D.; Pacchioni, G. Doping of WO3 for photocatalytic water splitting: Hints from density functional theory. J. Phys. Chem. C 2012, 116, 8901–8909. [Google Scholar] [CrossRef]
- Cai, G.; Cui, M.; Kumar, V.; Darmawan, P.; Wang, J.; Wang, X.; Eh, L.S.; Qian, K.; Lee, P.S. Ultra large optical modulation of electrochromic porous WO3 film and the local monitoring of redox activity. Chem. Sci. 2016, 7, 1373–1382. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Runnerstrom, E.L.; Milliron, D.J. Switchable materials for smart windows. Annu. Rev. Chem. Biomol. Eng. 2016, 7, 283–304. [Google Scholar]
- Granqvist, C.G. Electrochromic tungsten oxide films: Review of progress 1993–1998. Sol. Energy Mat. Sol. Cells 2000, 60, 201–262. [Google Scholar] [CrossRef]
- Lemire, C.; Lollman, D.B.B.; Mohammad, A.A.; Gillet, E.; Aguir, K. Reactive R.F. magnetron sputtering deposition of WO3 thin films. Sens. Actuators B Chem. 2002, 84, 43–48. [Google Scholar] [CrossRef]
- Davazoglou, D.; Leveque, G.; Donnadieu, A. Study on the optical and electrochromic properties of polycrystalline WO3 thin films prepared by CVD. Sol. Energy Mater. 1988, 17, 379–390. [Google Scholar] [CrossRef]
- Reichman, B.; Bard, A.J. The Electrochromic process at WO3 electrodes prepared by vacuum evaporation and anodic oxidation of W. J. Electrochem. Soc. 2016, 126, 583–591. [Google Scholar] [CrossRef]
- Zayim, E.O. Optical and electrochromic properties of sol-gel made anti-reflective WO3-TiO2 films. Sol. Energy Mate. Sol. Cells 2005, 87, 695–703. [Google Scholar] [CrossRef]
- Wojcik, P.; Cruz, A.; Santos, L.; Pereira, L.; Martins, R.; Fortunato, E. Microstructure control of dual-phase inkjet-printed a-WO3/TiO2/WOX films for high-performance electrochromic applications. J. Mater. Chem. 2012, 22, 13268–13278. [Google Scholar] [CrossRef]
- Santos, L.; Wojcik, P.; Pinto, J.V.; Elangovan, E.; Viegas, J.; Pereira, L.; Martins, R.; Fortunato, E. Structure and morphologic influence of WO3 nanoparticles on the electrochromic performance of dual-phase a-WO3/WO3 inkjet printed films. Adv. Electron. Mater. 2015, 1, 1–2. [Google Scholar] [CrossRef]
- Mukherjee, R.; Sahay, P.P. Effect of precursors on the microstructural, optical, electrical and electrochromic properties of WO3 nanocrystalline thin films. J. Mater. Sci-Mater. Electron. 2015, 26, 1–13. [Google Scholar] [CrossRef]
- Chai, Y.N.; Razak, K.A.; Lockman, Z. Effect of annealing on acid-treated WO3·H2O nanoplates and their electrochromic properties. Electrochimica Acta 2015, 178, 673–681. [Google Scholar]
- Gonzalez-Borrero, P.P.; Sato, F.; Medina, A.N.; Baesso, M.L.; Bento, A.C.; Baldissera, G.; Persson, C.; Niklasson, G.A.; Granqvist, C.G.; Silva, A.J.D. Optical band-gap determination of nanostructured WO3 film. Appl. Phys. Lett. 2010, 96, 201. [Google Scholar] [CrossRef]
- Vemuri, R.S.; Engelhard, M.H.; Ramana, C.V. Correlation between surface chemistry, density, and band gap in nanocrystalline WO3 thin films. ACS Appl.Mater. Interfaces 2012, 4, 1371–1377. [Google Scholar] [CrossRef] [PubMed]
- Min, H.K.; Choi, H.W.; Kim, K.H. Thickness dependence of WO3-x thin films for electrochromic device application. Mol. Cryst. Liquid Cryst. 2014, 598, 54–61. [Google Scholar]
- Arfaoui, A.; Ouni, B.; Touihri, S.; Mannoubi, T. Investigation into the optoelectrical properties of tungsten oxide thin films annealed in an oxygen air. Mater. Res. Bull. 2014, 60, 719–729. [Google Scholar] [CrossRef]
- Szilágyi, I.M.; Santala, E.; Heikkilä, M.; Kemell, M.; Nikitin, T.; Khriachtchev, L.; Räsänen, M.; Ritala, M.; Leskelä, M. Thermal study on electrospun polyvinylpyrrolidone/ammonium metatungstate nanofibers: Optimising the annealing conditions for obtaining WO3 nanofibers. J. Therm. Anal. Calorim. 2011, 105, 73. [Google Scholar] [CrossRef]
- Badilescu, S.; Ashrit, P.V. Study of sol-gel prepared nanostructured WO3 thin films and composites for electrochromic applications. Solid State Ion. 2003, 158, 187–197. [Google Scholar] [CrossRef]
- Zou, Y.S.; Zhang, Y.C.; Lou, D.; Wang, H.P.; Gu, L.; Dong, Y.H.; Dou, K.; Song, X.F.; Zeng, H.B. Structural and optical properties of WO3 films deposited by pulsed laser deposition. J. Alloy Compd. 2014, 583, 465–470. [Google Scholar] [CrossRef]
- Haro-Poniatowski, E.; Jouanne, M.; Morhange, J.F.; Julien, C.; Diamant, R.; Fernández-Guasti, M.; Fuentes, G.A.; Alonso, J.C. Micro-raman characterization of WO3 and MoO3 thin films obtained by pulsed laser irradiation. Appl. Surf. Sci. 1998, 127–129, 674–678. [Google Scholar] [CrossRef]
- Chai, Y.; Tam, C.W.; Beh, K.P.; Yam, F.K.; Hassan, Z. Effects of thermal treatment on the anodic growth of tungsten oxide films. Thin Solid Films 2015, 588, 44–49. [Google Scholar] [CrossRef]
- Badot, J.C.; Beluze, L.; Dubrunfaut, O. Particle size effect on the electronic conductivity of electroactive LixWO3·H2O powders: A study from 103 to 1010 Hz. J. Phys. Chem. C 2009, 112, 14549–14554. [Google Scholar] [CrossRef]
- Faughnan, B.W.; Crandall, R.S.; Lampert, M.A. Model for the bleaching of WO3 electrochromic films by an electric field. Appl. Phys. Lett. 1975, 27, 275–277. [Google Scholar] [CrossRef]
- Srivastava, A.K.; Deepa, M.; Singh, S.; Kishore, R.; Agnihotry, S.A. Microstructural and electrochromic characteristics of electrodeposited and annealed WO3 films. Solid State Ion. 2005, 176, 1161–1168. [Google Scholar] [CrossRef]
- Koo, B.R.; Ahn, H.J. Fast-switching electrochromic properties of mesoporous WO3 films with oxygen vacancy defects. Nanoscale 2017, 9, 17788–17793. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, G.; Lu, K.; Zhang, X.; Yuan, W.; Shi, M.; Ning, H.; Tao, R.; Liu, X.; Yao, R.; Peng, J. Effects of Annealing Temperature on Optical Band Gap of Sol-gel Tungsten Trioxide Films. Micromachines 2018, 9, 377. https://doi.org/10.3390/mi9080377
Zhang G, Lu K, Zhang X, Yuan W, Shi M, Ning H, Tao R, Liu X, Yao R, Peng J. Effects of Annealing Temperature on Optical Band Gap of Sol-gel Tungsten Trioxide Films. Micromachines. 2018; 9(8):377. https://doi.org/10.3390/mi9080377
Chicago/Turabian StyleZhang, Guanguang, Kuankuan Lu, Xiaochen Zhang, Weijian Yuan, Muyang Shi, Honglong Ning, Ruiqiang Tao, Xianzhe Liu, Rihui Yao, and Junbiao Peng. 2018. "Effects of Annealing Temperature on Optical Band Gap of Sol-gel Tungsten Trioxide Films" Micromachines 9, no. 8: 377. https://doi.org/10.3390/mi9080377
APA StyleZhang, G., Lu, K., Zhang, X., Yuan, W., Shi, M., Ning, H., Tao, R., Liu, X., Yao, R., & Peng, J. (2018). Effects of Annealing Temperature on Optical Band Gap of Sol-gel Tungsten Trioxide Films. Micromachines, 9(8), 377. https://doi.org/10.3390/mi9080377






