1. Introduction
Retinal degenerative (RD) diseases are a clinically and genetically heterogeneous group of primary retinal abnormalities that cause degeneration of the photoreceptors (rods and cones) or the retinal pigment epithelium and lead to progressive loss of vision. Transcorneal electrical stimulation (TcES) is one of the therapies that promotes the survival of photoreceptors and preserves retinal functions [
1]. Previous electrical stimulation on the eye was performed through an ERG-Jet contact lens electrode with the stimulation current provided by external equipment through a metal wire [
2]. With this setup, the cornea has to be anesthetized for each treatment and is neither convenient nor portable for patients to use. Moreover, daily use of this TcES device has a high chance to induce tissue damage.
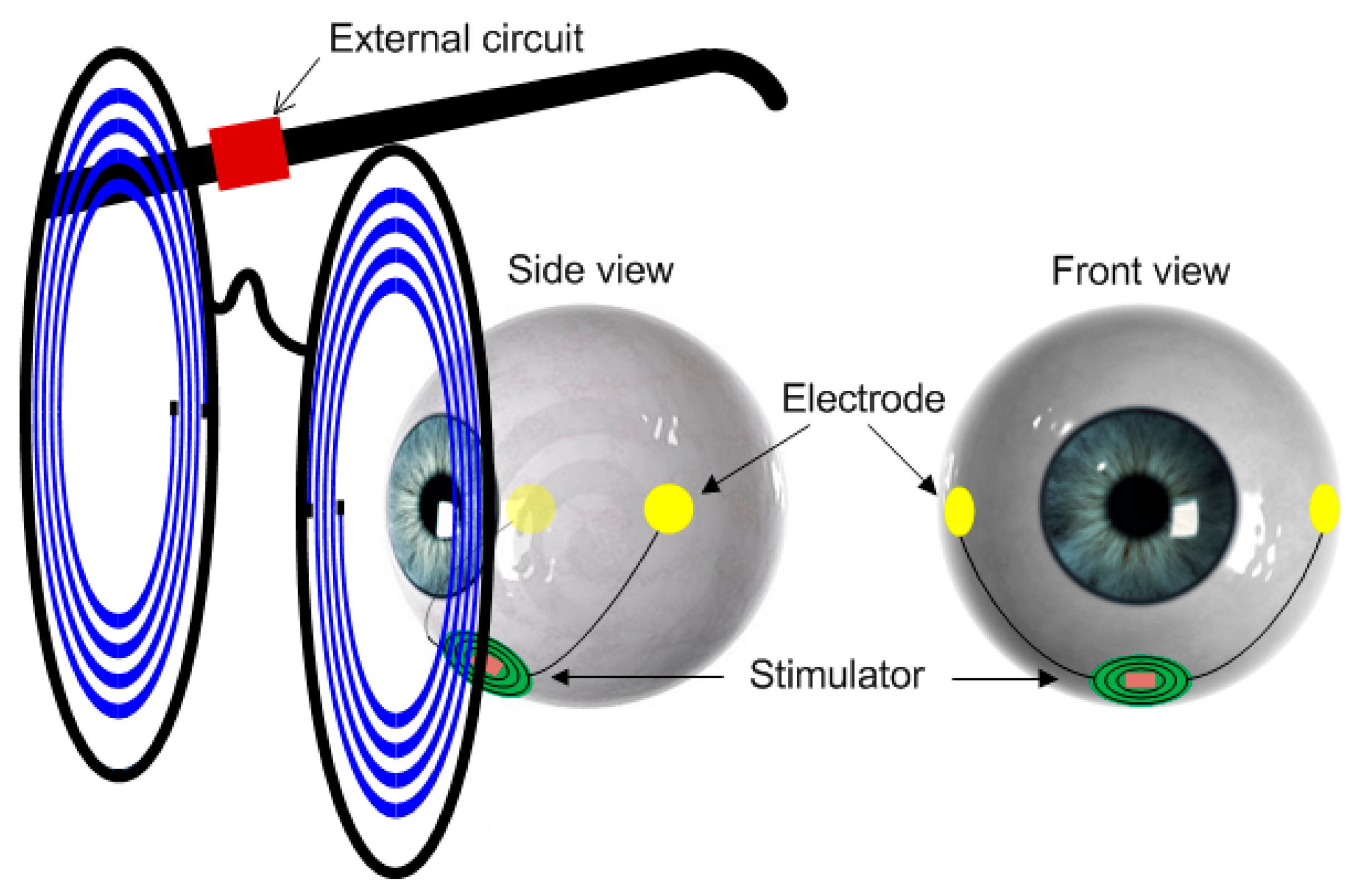
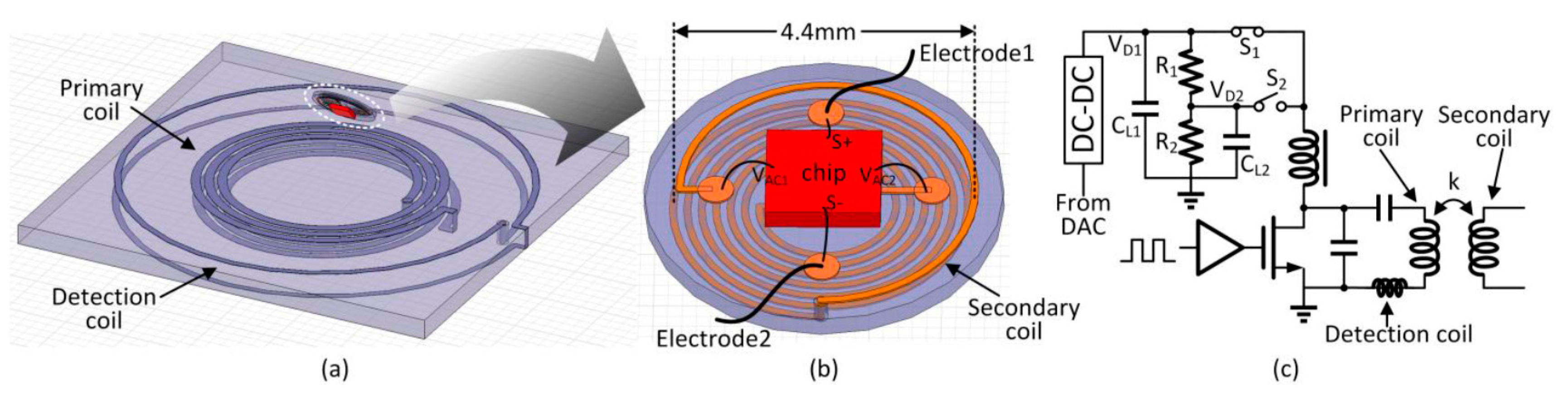
To enhance convenience and portability with a minimum invasive setup, one possible solution is to implant the whole stimulator underneath the eye. To reduce the side effects of tissue damage and invasive infection, and to reduce the complexity of the surgical operation so that it can be carried out in clinics instead of operation rooms, we propose to place this implantable stimulator beneath the eye’s conjunctiva but outside the eyeball on the sclera completely unexposed, as shown in
Figure 1. The two electrodes are placed on the sclera at the two sides of the eyeball. In this paper, we present the design of such an on-sclera implant-based TcES system.
An implantable medical device (IMD) has to be small and thin for the comfort of the patients. Battery has to be eliminated due to its bulky size and limited lifetime [
3]. Wireless power transfer using inductive coupling has been widely used for power transfer and data telemetry in IMD applications such as retinal prosthesis and neural stimulation and recording [
4,
5,
6,
7]. To achieve high power transfer efficiency and high data rate, multi-channels were proposed in [
8,
9,
10,
11] to transfer the power and data separately using two or more pairs of coils. In [
12] and [
13], 3-coil and 4-coil coupled power links were proposed, respectively, to enhance the coupling efficiency. For the TcES application, data rate does not need to be high and system volume has to be minimized. To reduce system volume, we propose that the power link and the data link should share only one pair of coils. On-chip coil [
14,
15] could be used to further reduce the size, but the power transfer efficiency and the voltage gain would be significantly deteriorated due to poor quality factor and weak coupling with the primary coil. Hence, off-chip coil is adopted.
The human tissue specific absorption rate (SAR) increases with the coupling frequency. Therefore, inductive power links with 13.56 MHz or lower ISM bands are commonly used [
16,
17]. However, lower coupling frequency means larger matching and filtering capacitors that lead to a larger implant size. In [
15], 40.68 MHz in the ISM band was proposed as a reasonable frequency in achieving good power transfer efficiency for the power link of IMDs. When compared with using 13.56 MHz, the matching and filtering capacitors at 40.68 MHz are reduced by 9 and 3 times, respectively. Hence, 40.68 MHz was finally chosen as the power coupling frequency.
This paper is organized as follows.
Section 2 gives an overview of the system and the design of the power and data links, including control methods for power boosting and regulation. Detailed circuit implementation is presented in
Section 3. Off-chip circuitry and coil design are discussed in
Section 4. Measurement results of the fabricated chip are given in
Section 5, and conclusions are drawn in
Section 6.
2. System Overview
2.1. System Functionality
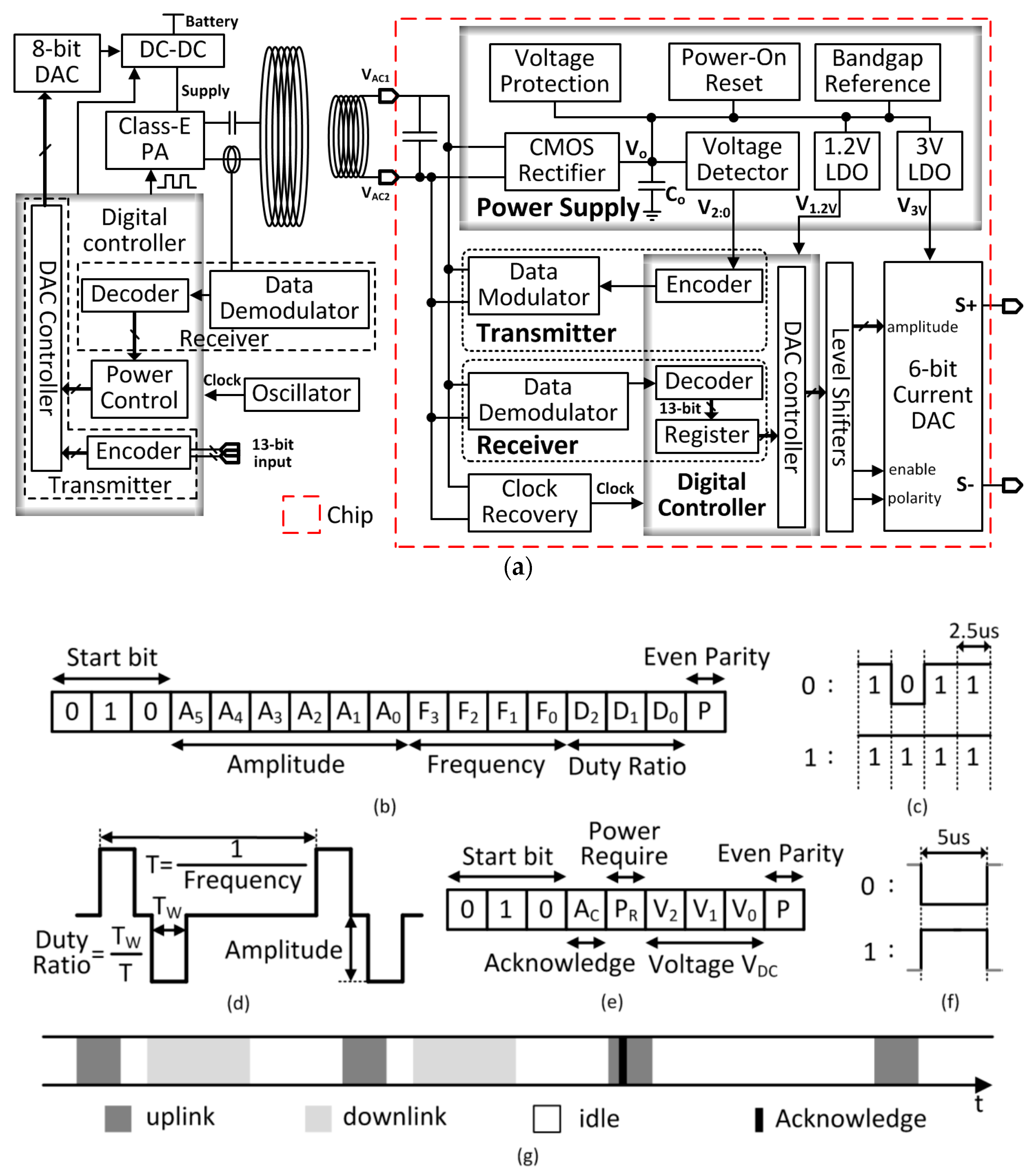
The TcES system is shown in
Figure 2a [
18]. It consists of an external control unit and an internal implant unit. Except for the secondary coil, the implant unit is fully integrated on-chip and is shown in the dash box in
Figure 2a. The internal implant unit consists of the power supply module, the data telemetry module, the digital controller and the stimulation unit. The inductively coupled AC power on the secondary coil is converted into DC power by the rectifier and stored in the on-chip filtering capacitor at V
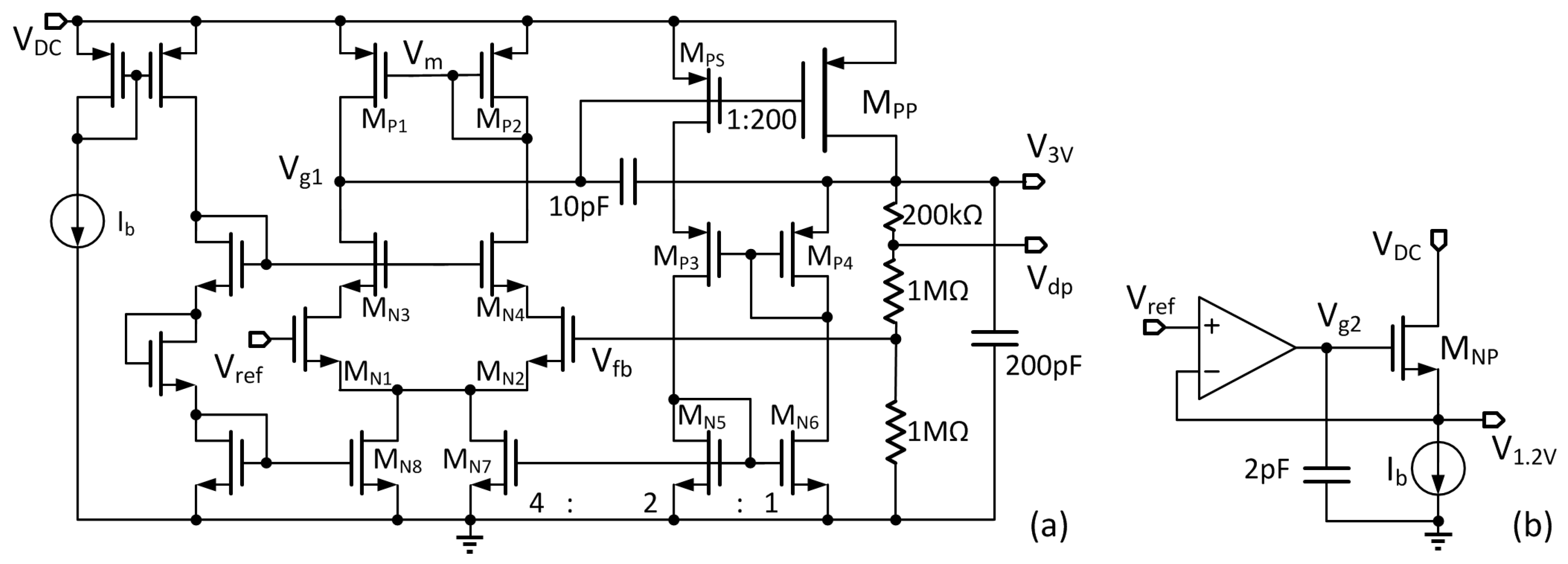
DC. A 1.2-V low dropout regulator (LDO) and a 3-V LDO powered by V
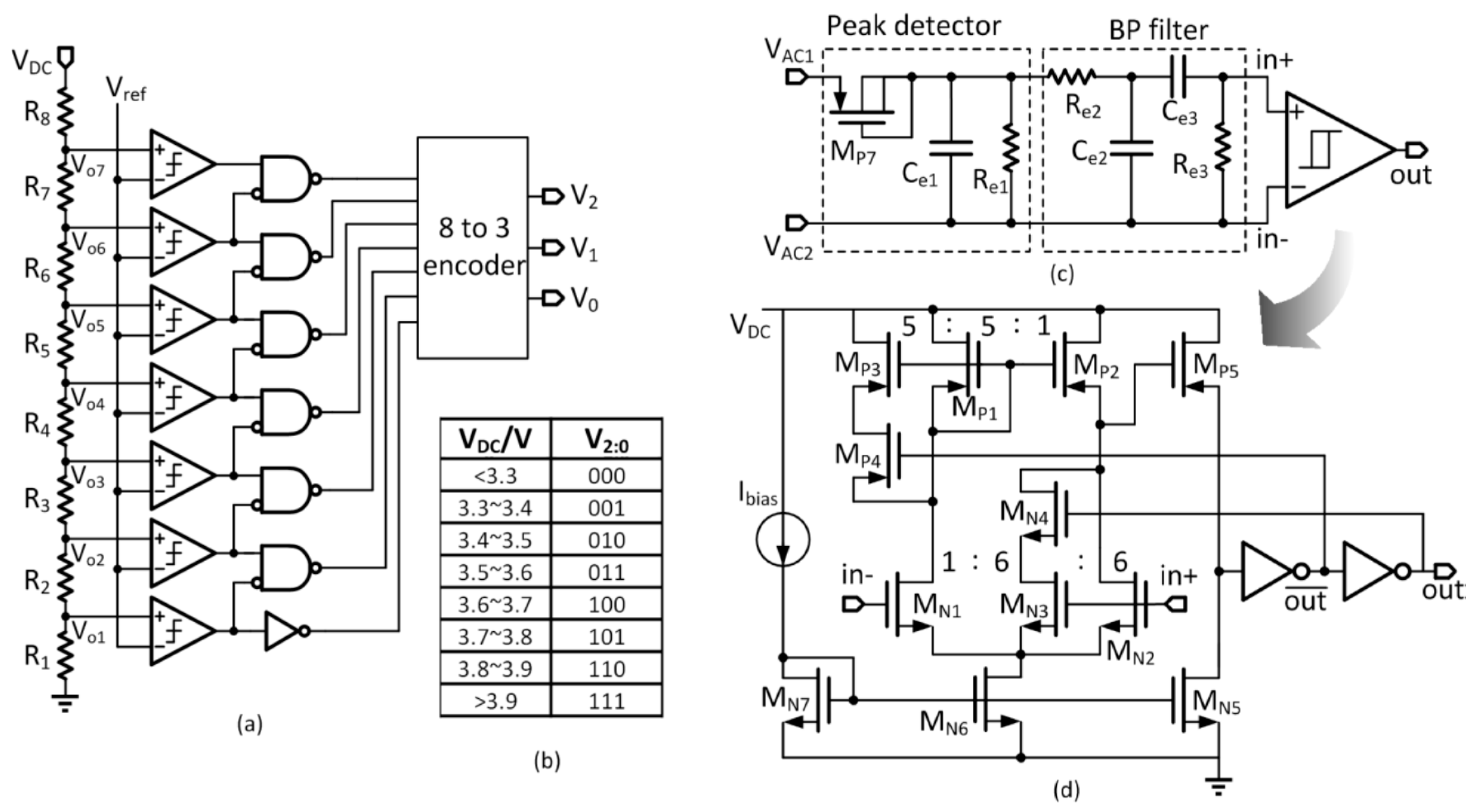
DC are used to supply power to the digital controller and the stimulation unit, respectively. The voltage detector monitors V
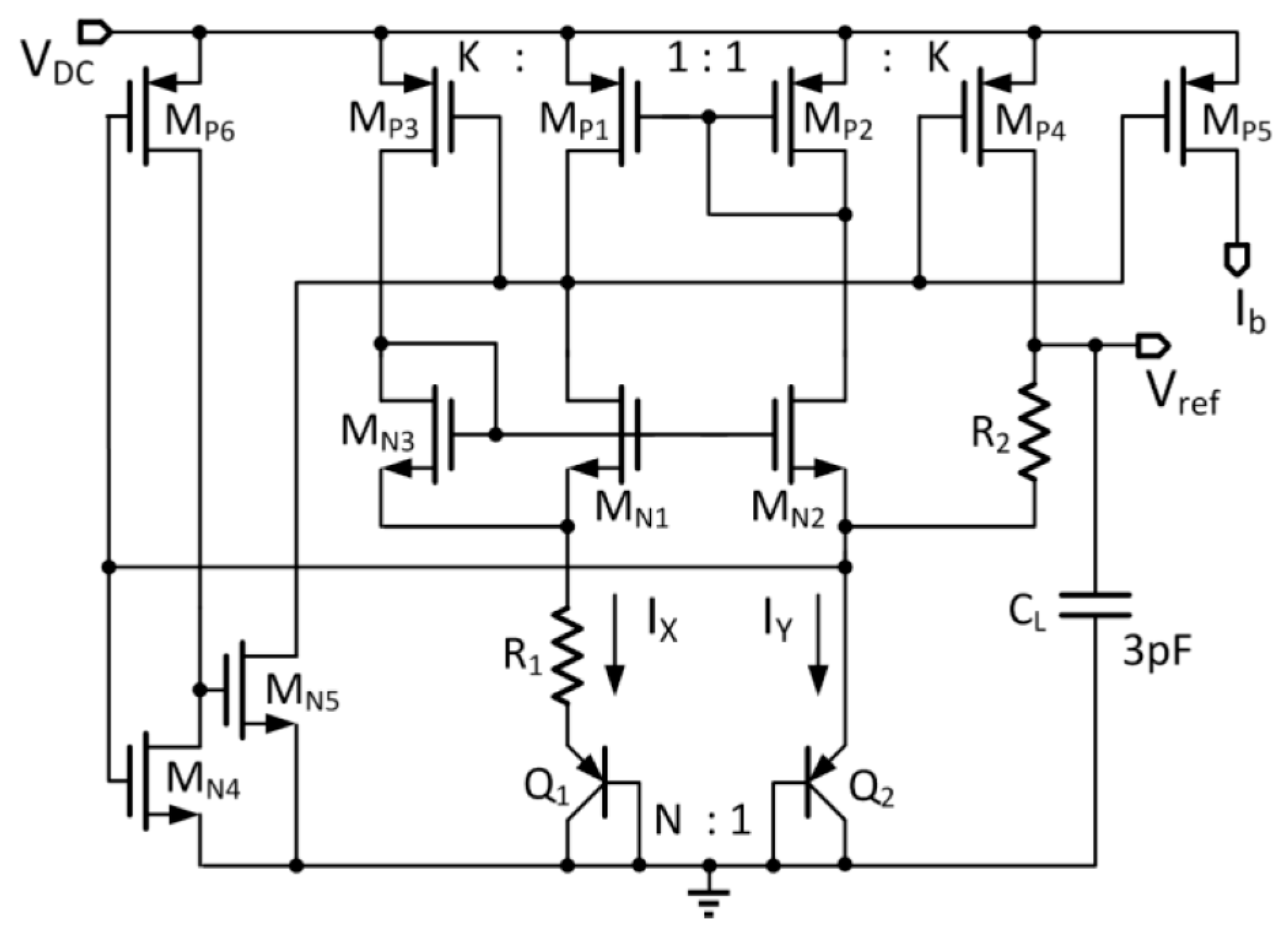
DC for controlling the power. Peripheral circuits include the voltage protection circuit and the power-on reset for over-voltage protection and initialization during startup, respectively. The bandgap voltage reference generates the reference voltages for the 1.2-V LDO and the 3-V LDO, and also the bias currents for the stimulation unit. The data telemetry circuitry consists of a transmitter that includes an encoder and a data modulator, and a receiver that includes a demodulator and a decoder. The clock for the digital block is recovered from the power coupling frequency by a clock recovery circuit.
The external control unit consists of the power transmission circuitry and the data telemetry circuitry. The DC-DC buck converter supplies power to the Class-E power amplifier that drives the series-resonant primary coil. An 8-bit digital-to-analog converter (DAC) is used to adjust the DC-DC output voltage to control the transmission power of the primary coil. A detection coil is used to receive the back-scattered data transmitted from the internal implant unit, and a digital controller is used to encode and decode the data.
For this TcES system, the power link and the data link share the same pair of coils. The output voltage of the rectifier VDC changes when the loading changes, as well as when the distance of the coils or their relative alignment changes. Adaptive power control is needed to dynamically adjust the emitted power to achieve a stable rectified DC voltage. It is divided into power regulation and power boosting. For power regulation, the voltage detector senses VDC periodically and the value is transmitted back to the external control unit through backscattering. By comparing with a reference voltage, the output of the DC-DC converter is adjusted to change the transmission power accordingly. Power boosting is activated to increase the transmission power prior to the scheduled task of electrical stimulation. With these proposed power control strategies, the rectified DC output is well regulated and achieves better immunity to coupling and loading variations.
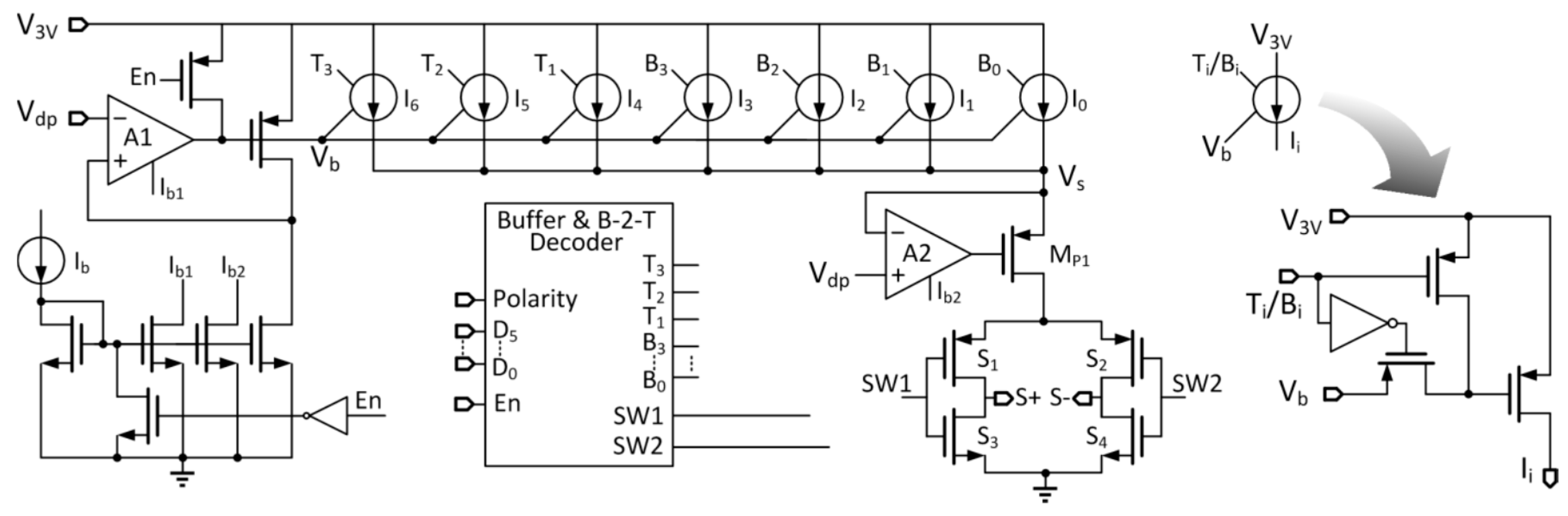
The implant unit generates a bi-polar current waveform for retina stimulation. For different patients or different therapy stages of the same patient, the stimulation profiles (current amplitude, stimulation frequency and duty ratio of current pulse) are different. The stimulation profile is programmed by the external control unit and sent through the data link. The digital controller reads the 13-bit configuration data, codes it, modulates it with amplitude shift keying (ASK) and then transmits it to the internal implant unit. The implant circuitry demodulates and decodes the 13-bit data, and stores it in a register that sets the DAC output with the stimulation profile. The current amplitude can be programmed to range from 5 μA to 320 μA, the stimulation frequency from 10 Hz to 160 Hz, and the current pulse duty ratio from 2.5% to 20%.
2.2. The Data Link
The downlink data package is shown in
Figure 2b. It contains the DAC controller configuration information. The 13 bits are organized as shown in
Figure 2c. The start bits “010” signifies the start of a data package, which also helps improve the bit error rate (BER). The current amplitude control bits A
5:0 corresponds to 5 μA to 320 μA with 6-bit quantization; the frequency control bits F
3:0 corresponds to 10 Hz to 160 Hz with 4-bit quantization; and the duty ratio control bits D
2:0 corresponds to 2.5% to 20% with 3-bit quantization. The final bit P is the even-parity check bit. ASK modulation is used for the downlink. In the idle state, the idle bit is “1”, and power but not data is transferred. If the data “1” is transmitted, power transfer is not affected. However, if the data “0” is transmitted, power transfer is reduced abruptly to generate a negative pulse, and the implant unit may not obtain enough power. Due to the limited bandwidth of both the primary and secondary
LC tanks, high data rate may not be easily achieved. The situation gets worse as the downlink data package has 17 bits in total, and the data package time-slot is quite long. In the worst case of transmitting a long chain of “0” s, the rectifier output would drop as the on-chip filtering capacitor is not too large. The solution is to use the coding scheme as shown in
Figure 2b: “0” is coded as “1011”; and “1” is coded as “1111”. Therefore, even a long chain of data “0” s are transmitted the implant unit could get 75% of full power, which is designed to be adequate. An additional benefit is the relaxed requirement of the internal decoder. The data are decoded as “0” s and “1” s with and without negative pulses, respectively. Both pulse width and pulse phase are not required to be accurate. With this coding scheme, the length of the downlink data package is 170 μs.
The 9-bit uplink data package that is sent to the external control unit is shown in
Figure 2e. The first 3 bits are the “010” start bits. The 4th bit A
C is the active-low acknowledgement bit. The 5th bit P
R is the power requirement bit for power boosting, which will be explained later. The next 3 bits V
2:0 indicate the quantized rectifier output voltage for power regulation; and the last bit P is the even-parity check bit. The uplink is based on backscattering that also uses ASK modulation. The uplink data transmission affects the power received, but as the data package has only 9 bits, the duration is much shorter. To balance the package duration and its effect on the received power, no special coding is used. The length of the uplink data package is 45 μs.
Both the downlink and the uplink are ASK modulated that share the same coupling channel, and they cannot work simultaneously. Time multiplexing is used as shown in
Figure 2f. The uplink data package is sent to the external control unit every 500 μs. The downlink data transmission interleaves with the uplink, but it only transmits data when there is a new configuration input data. When the new data is transmitted, the acknowledgement bit A
C will be checked in the next uplink data package. If there is no acknowledgement, this new data will be retransmitted until the external control unit receives A
C from the internal implant unit.
2.3. The Power Link
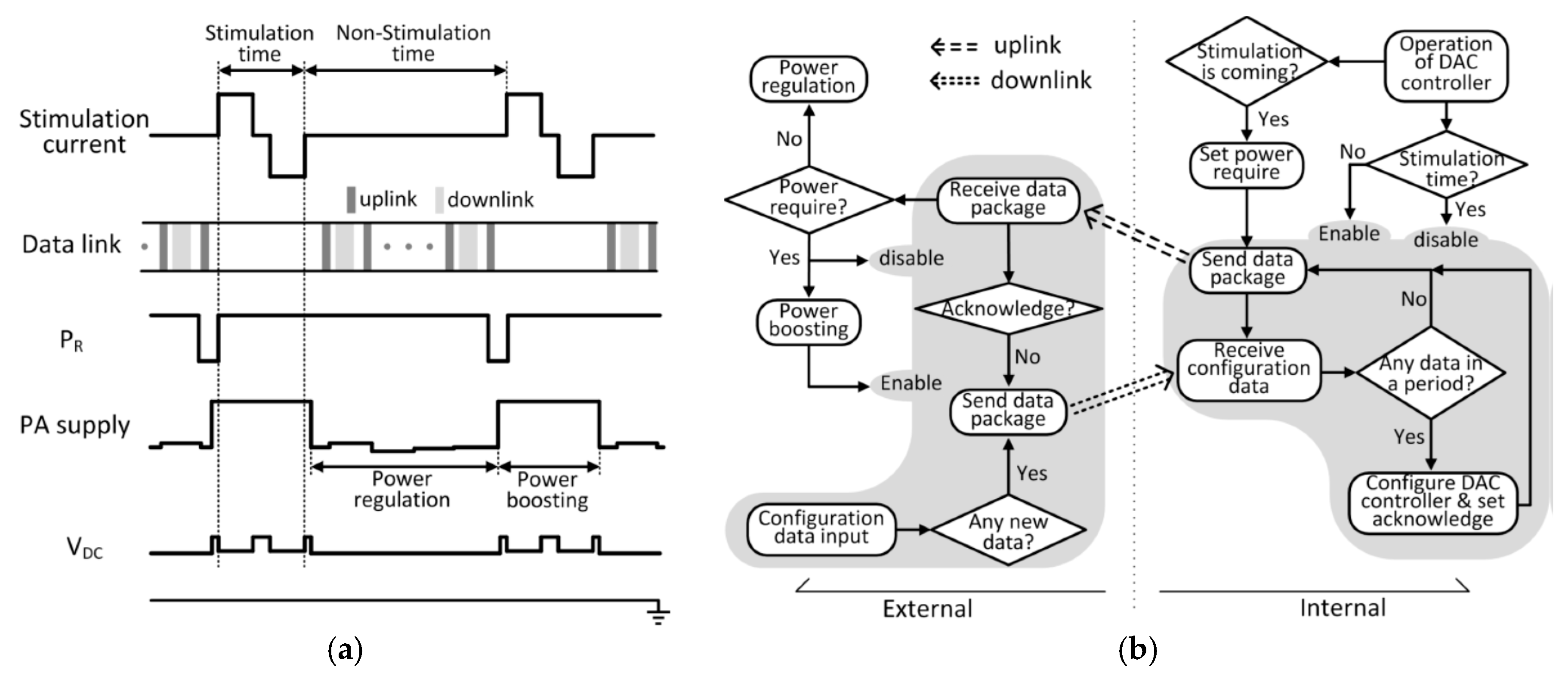
Power is wirelessly delivered from the external control unit to the internal implant unit using near-field coupling. For retina stimulation, the stimulation current is issued periodically as shown in
Figure 3a. During the non-stimulation time, the loading of the implant is light as it is only the total quiescent power of each block. During the stimulation time, the loading is heavy by issuing the stimulation current from the current DAC. Hence, power delivery is divided into the power-boosting phase and the power-regulation phase that are responsible for the stimulation time and the non-stimulation time, respectively.
The distance and the alignment of the two coupling coils may vary that affect the coupling coefficient and thus the power received. During the non-stimulation time, the power-regulation phase is activated, and the rectifier output VDC is periodically sensed and sent to the external control unit to adjust the transmitter power so that VDC could be regulated to 3.5 V. The movement of the human body is on the order of milliseconds, and so an uplink data package rate of 2 kHz is used.
During the stimulation time, the small on-chip filtering capacitor is drained to provide the stimulation current, and VDC will drop sharply. As the uplink package rate is low, it cannot adapt to this fast loading variation. To cater for this scheduled loading variation, the power requirement bit PR is set active (active low) just prior to the stimulation time, and is sent to the external control unit to activate the power-boosting phase, such that even VDC drops, it is still high enough for the downstream circuits to operate properly. During the stimulation time, the data link is disabled to avoid interference.
In the power-boosting phase, the transmitting power has to be increased to cover the whole stimulation time with adequate margin. For different coupling scenarios and stimulation profiles, the amount of increase in power is different (instead of using the maximum power) to avoid excessive radio frequency (RF) power that will be absorbed by the human body. Calibration is performed during the assembly phase of the implant unit. Load variation is related to the stimulation current amplitude, and coupling variation is related to the steady state PA voltage VDD,PA. By sweeping the above parameters for all possible cases and recording the needed power-boosting level to ensure adequate power to cover the whole stimulation time with margin, a look-up table is constructed to store the data of power increase required for normal operation.
2.4. The System State Diagram
The TcES system state diagram is shown in
Figure 3b. The shaded parts deal solely with the data link; and the non-shaded parts deal with the power link that also depends on the data link. The startup phase of the system will be discussed in
Section 2.5.
Let us consider the system operation after startup during the non-stimulation time, that is, during the power-regulation phase. The external control unit and the internal implant unit exchange roles as master and salve every 500 μs. Consider that the configuration data is inputted (through programming the external control unit), and when the external control unit resumes the master role it is then sent across the link. If the internal implant unit receives the data, it will be used to configure the DAC controller and the acknowledgement bit AC will be set active low to be sent back to the external control unit in the next data package. After the data package is sent, AC will then be reset. If there is no configuration data received within this period, the internal implant unit just sends back the uplink package that consists of the digitized rectifier output voltage VDC, so that the external control unit could adjust the transmitting power to regulate VDC. After receiving the data package, the external control unit becomes the master. If there is a new configuration data needed to be transmitted and there is no acknowledgement from the internal unit, the data will be sent and then the external unit shifts to be slave again waiting to receive data. If the above condition is not satisfied, no data will be sent and the external unit becomes slave after 500 μs.
For a certain stimulation profile, the internal implant unit knows the period of stimulation and knows exactly when the next stimulation slot will arrive. Just before the next scheduled stimulation time slot, the power-boosting phase has to be activated. Hence, when the internal implant unit becomes the master, the power require bit PR is set active low and sent to the external control unit to increase the transmitting power according to the look-up table. During the stimulation time, the data telemetry block of the internal implant unit is disabled. Note that the data telemetry block of the external control unit is disabled when the instruction for power boosting is received; and it is enabled after power boosting is completed.
2.5. The Startup
The power link relies on the data link to determine the transmitting power level, but the data link can function only if there is sufficient received power. During startup, the internal implant unit has no stored energy. To solve this problem, during the TcES system startup, the external control unit will issue a very high power to jump-start the internal implant unit. Once the data link is established, the transmitting power will decrease slowly. Eventually, the transmitting power will be controlled according to the power-regulation and the power-boosting mechanisms as previously discussed. The startup procedure will be repeated if the first trial fails. However, if it fails repeatedly due to weak coupling or misalignment between the coupling coils, the external control unit will be shut down for protection.
5. Experimental Results
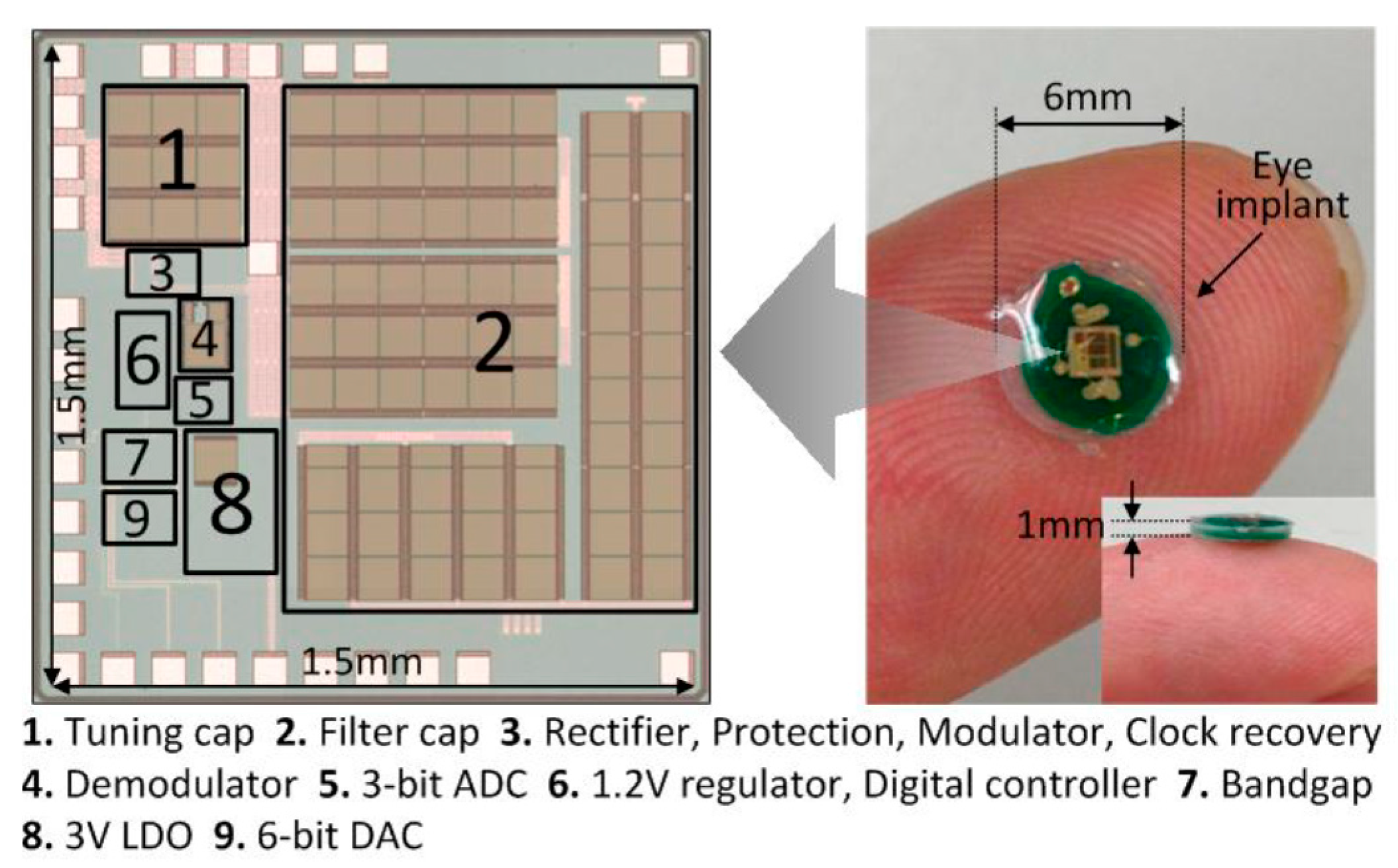
The internal implant unit is fabricated using UMC 0.13 μm CMOS process. The die photo is shown in
Figure 10 and the overall chip area is 2.25 mm
2 and the thickness is 0.2 mm. The filtering capacitor consists of stacked MOS, MIM and MOM capacitors to maximize the capacitance density. The tuning capacitor is made of MIM capacitors, and the bottom plate is connected to V
AC2 that is clamped at around V
DC/2. To save space, the MOS and MOM capacitors underneath the tuning capacitor are also used as part of the filtering capacitor, and simulations showed no observable effect on the tuning circuit. The chip is glued on the secondary PCB and bonded directly to the coil as shown in
Figure 9b. The whole implant unit is then encapsulated by biocompatible silicone. The overall thickness of this implant device is controlled to be less than 1 mm with an outer diameter of 6 mm, as shown in
Figure 10. The whole eye implant is much smaller than a contact lens and can be put beneath the eye’s conjunctiva easily. The external control unit is implemented using discrete components and a field-programmable gate array (FPGA) as shown in
Figure 11. A buzzer is added. When the distance of the two coils is too far apart, the buzzer will beep to alarm the patient to adjust the primary coil to make it closer to the eye implant. This external control unit will be integrated into a single chip in the future work.
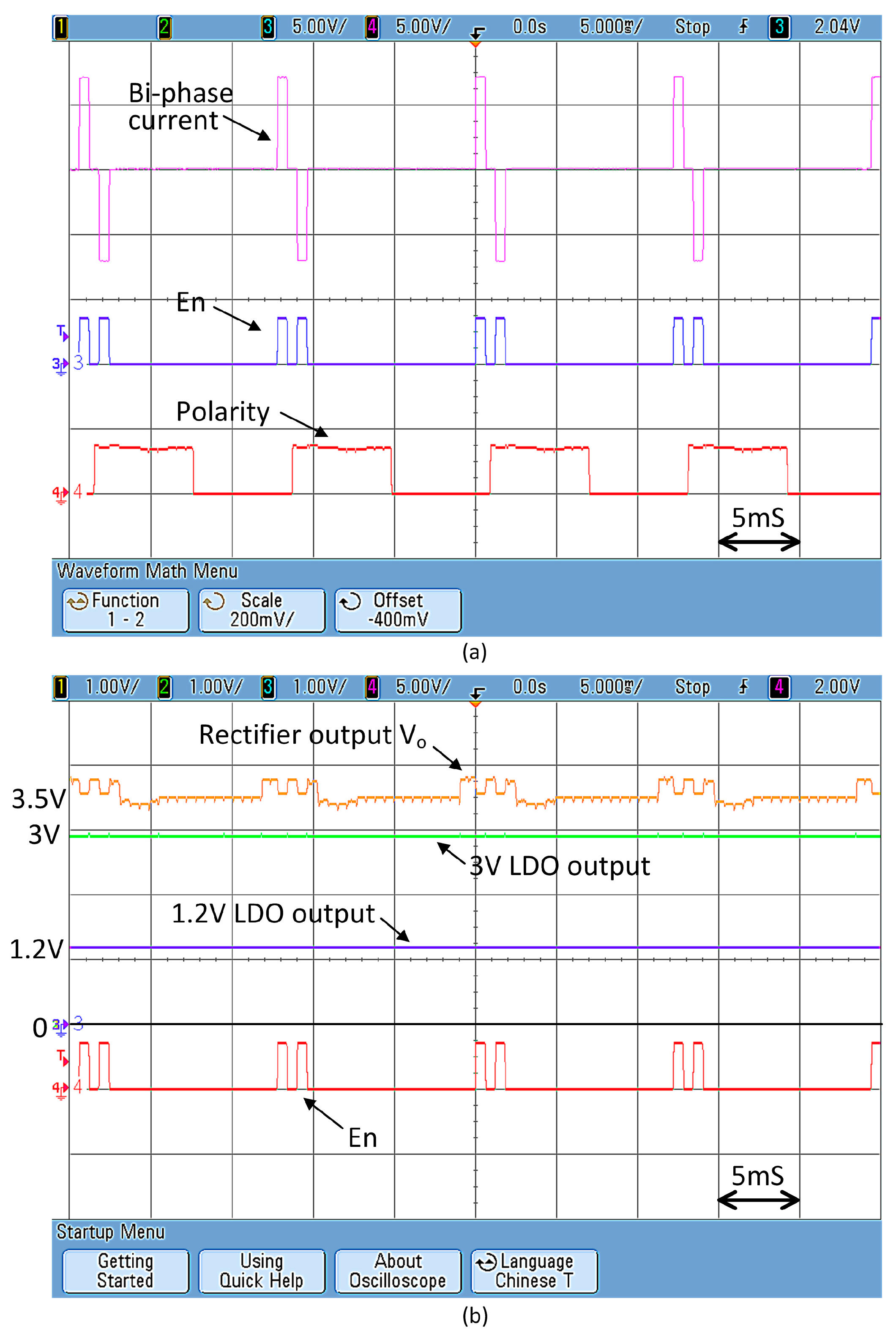
Figure 12a shows the measured waveform of the 6-bit current DAC with a 5 kΩ output load resistor. There is stimulation current when “En” = 1. The signal “Polarity” is a periodic square wave that changes the current direction. In
Figure 12b, it is shown that the output voltage of the rectifier V
DC is not very steady. The voltage ripple consists of two components: the first one is the large voltage ripple of around 400 mV that has a frequency of lower than 1 kHz due to power boosting; and the second is the smaller voltage ripple of 50 mV at 80 MHz due to backscattering. After filtering by the 3-V LDO and the 1.2-V LDO, both ripple components are significantly attenuated. The stable 3 V and 1.2 V outputs are then used to supply power to the DAC and the digital controller, respectively.
Figure 13 shows the action of power regulation through data backscattering in the non-stimulation time. V
DC is periodically detected and the equivalent digital value is backscattered to the external control unit. The external control unit then adjusts the transmitting power such that V
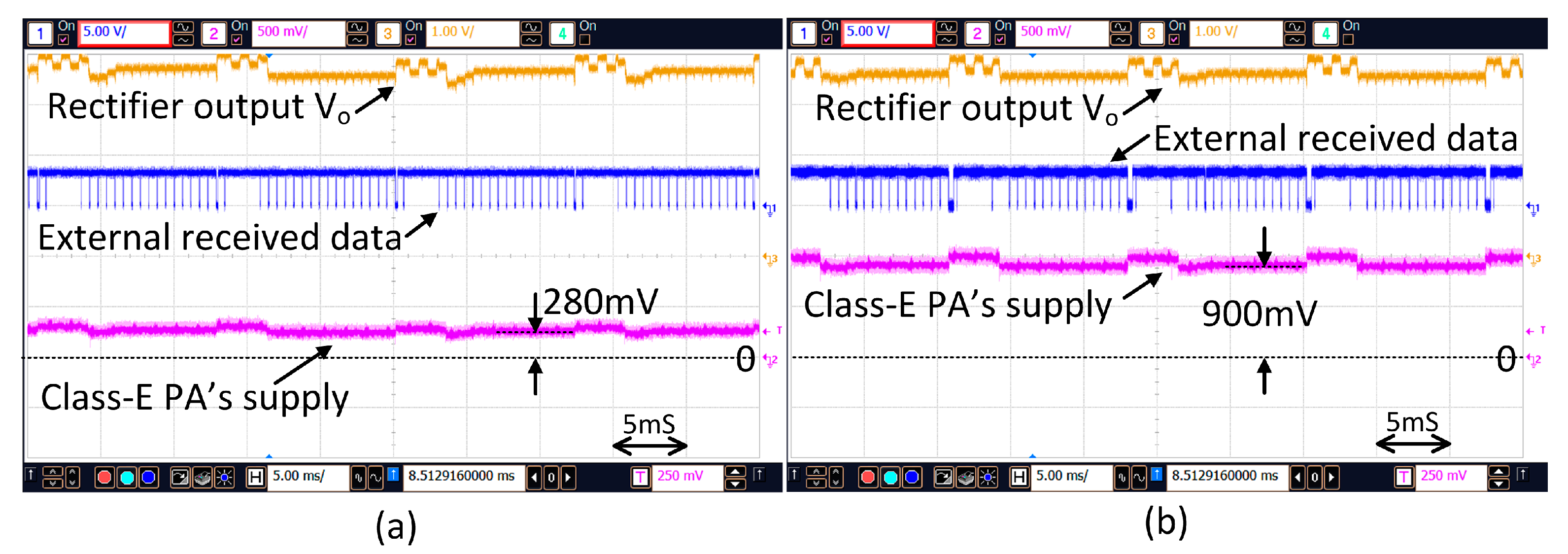
DC is regulated to the predefined value of 3.5 V. When the distance between the two coils changes the supply voltage of the PA will be adjusted adaptively. As shown in
Figure 14, when the distance changes from 4 mm to 13 mm, the supply voltage of the PA is increased from 280 mV to 900 mV such that V
DC is maintained at around 3.5 V.
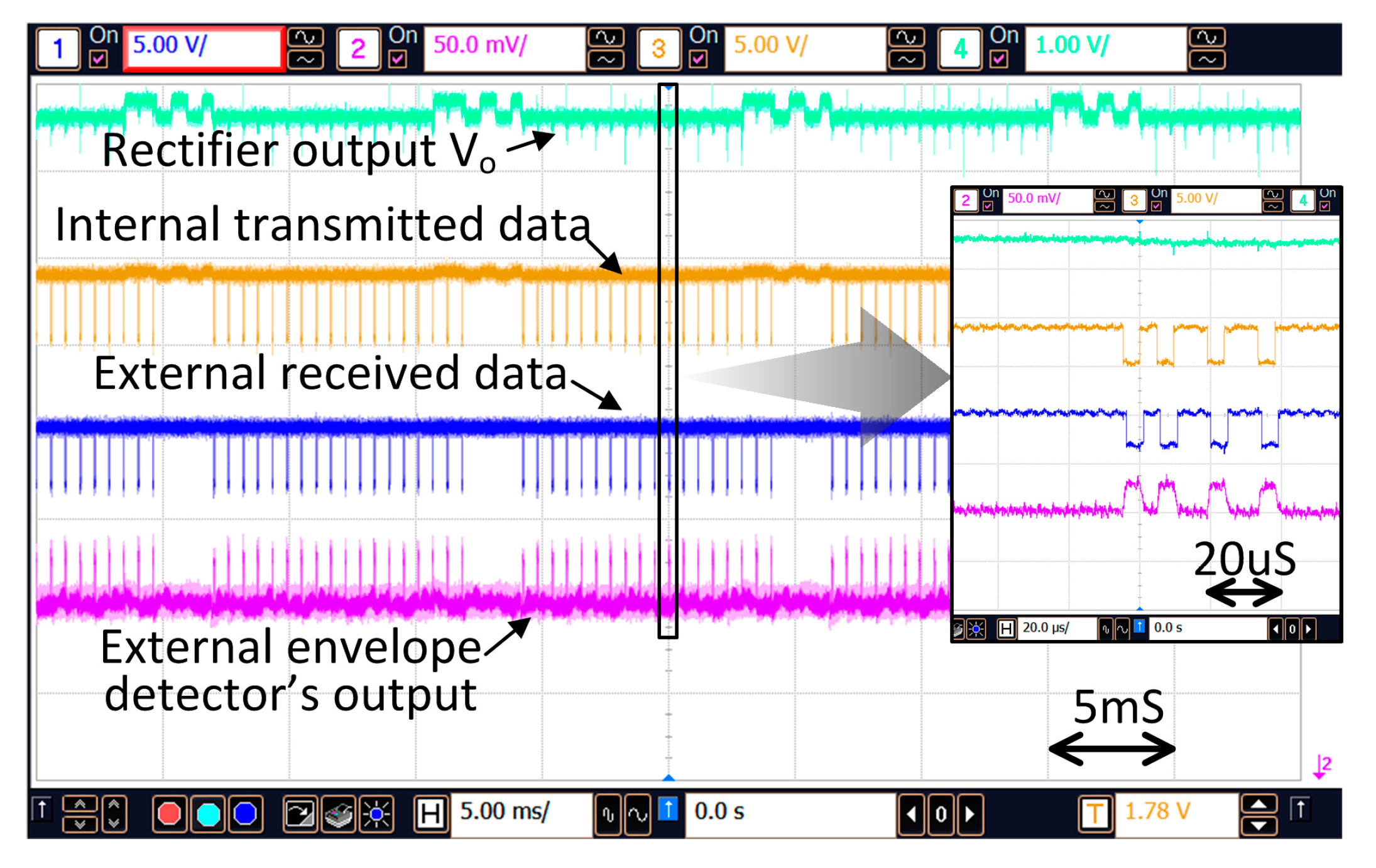
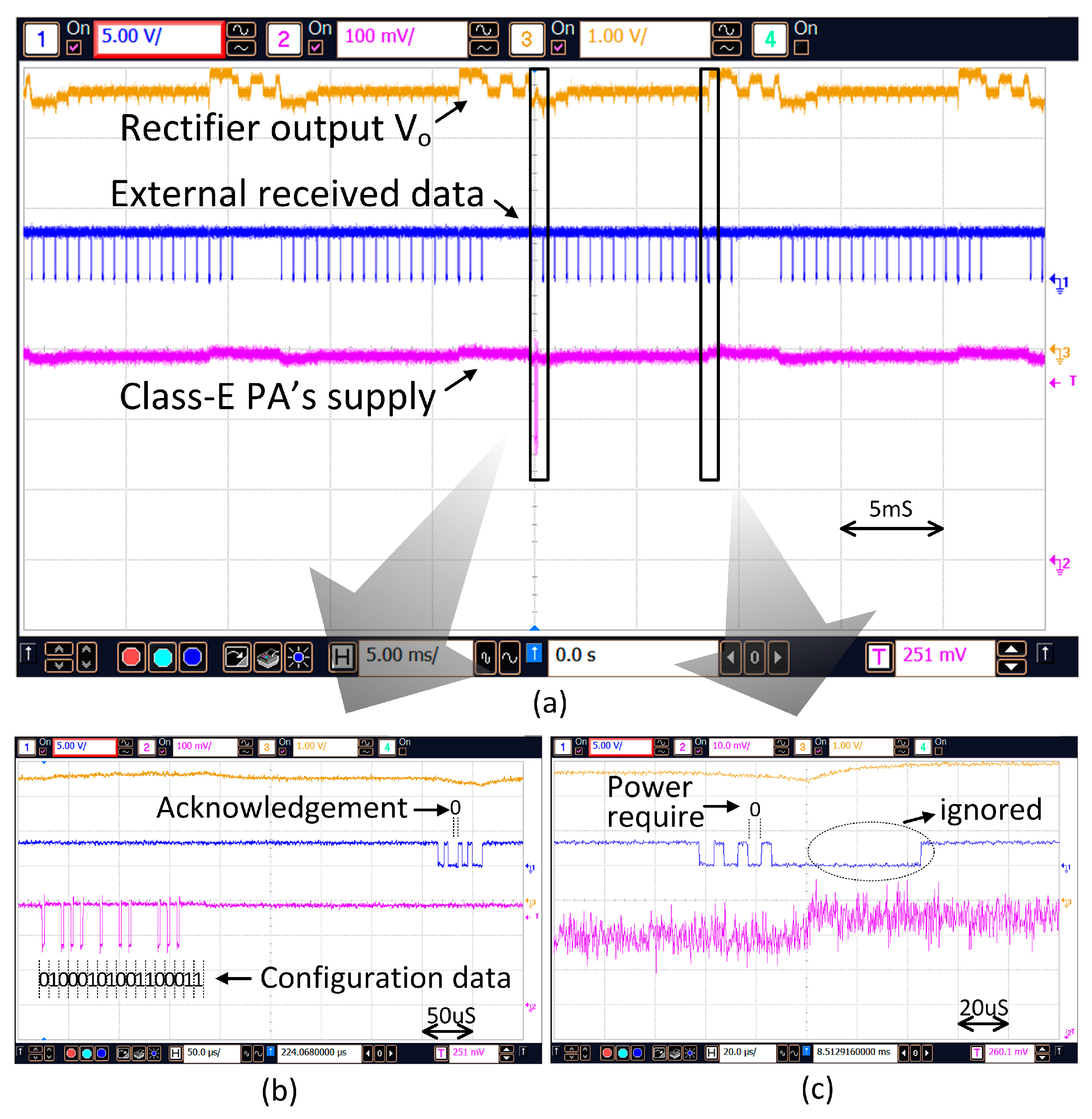
Figure 15a shows the transmitted configuration data and the accompanied power boosting during the stimulation time. When new configuration data is set and the primary side attains the status of “Master”, the data will be transmitted to the secondary side. As shown in
Figure 15b, for transmitting a “1”, the supply voltage of the PA maintains at V
D1; but for transmitting a “0”, it will drop temporarily to V
D2 as discussed in
Section 4.2. A short period after the configuration data is transmitted the internal implant unit becomes the master and sends back the acknowledgement bit such that transmission of the configuration data will not be repeated.
Prior to current stimulation, power boosting is activated such that the transmission power is increased to deliver adequate power for the stimulation. As shown in
Figure 15c, before the stimulation time, the Power Require bit P
R in the backscattered data is set to “0” to command the external control unit to increase the transmission power immediately.
The design parameters and measured data are summarized in
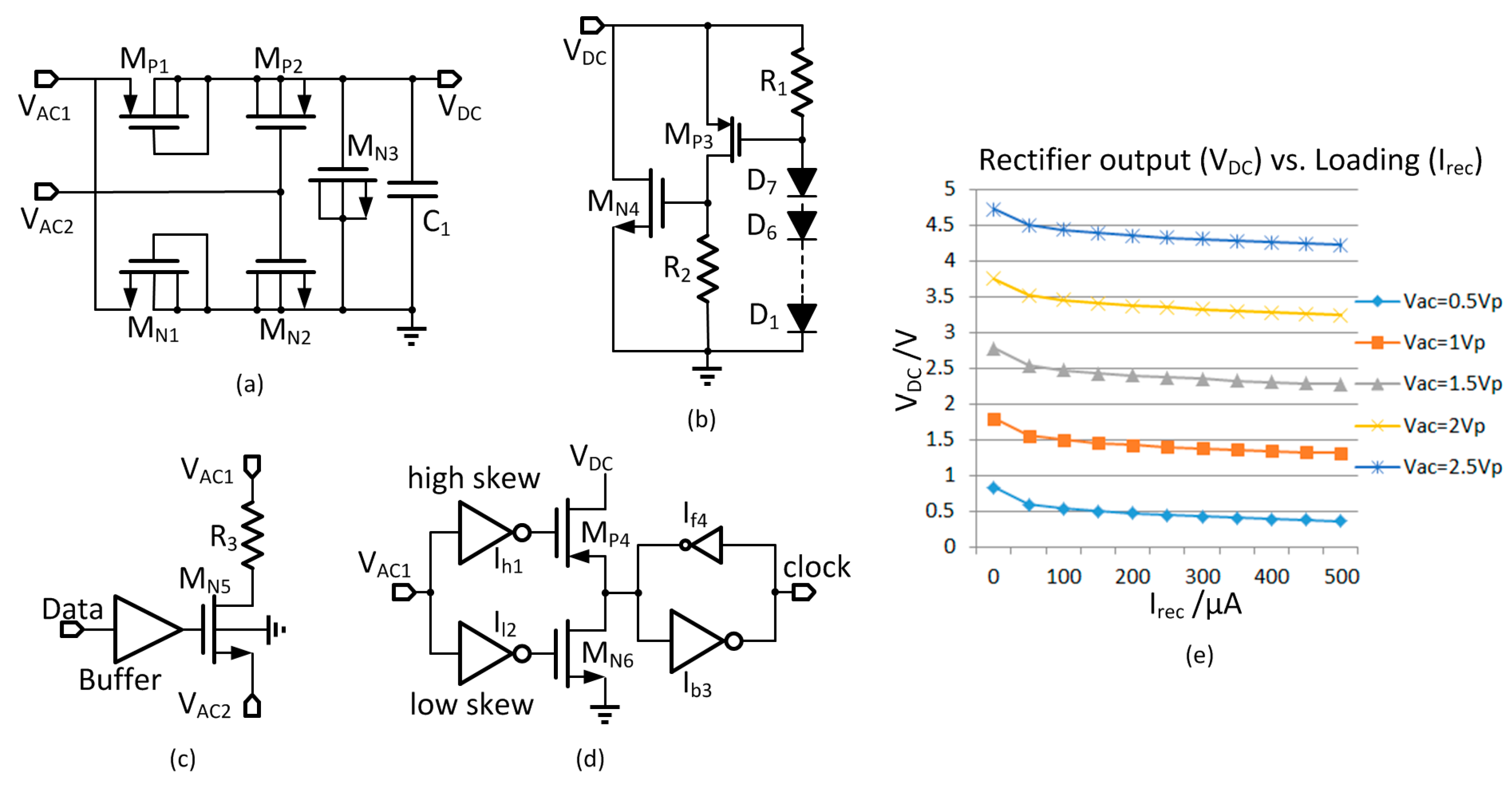
Table 1. The primary coil has good quality factor (98), but that of the secondary coil is not as high (41). The inductance of the detection coil is around 300 nH, and the coupling factor with the primary coil is around 0.3 that achieves good sensitivity for backscattered data detection and does not have excessive interference on the primary coil. The 3-V LDO and the 1.2-V LDO have good power supply rejection ratio (PSRR) at both <1 kHz and 80 MHz such that the two ripple components are rejected. The total quiescent current of the secondary chip is around 40 μA. The maximum power transfer efficiency from the PA input to the rectifier output is around 8% when the distance of the two coils is 1 cm and rectifier loading current is 300 μA.