Influence of Inner Lining Atoms of Multilayered Hexagonal Boron Nitride Porous Membrane on Desalination
Abstract
1. Introduction
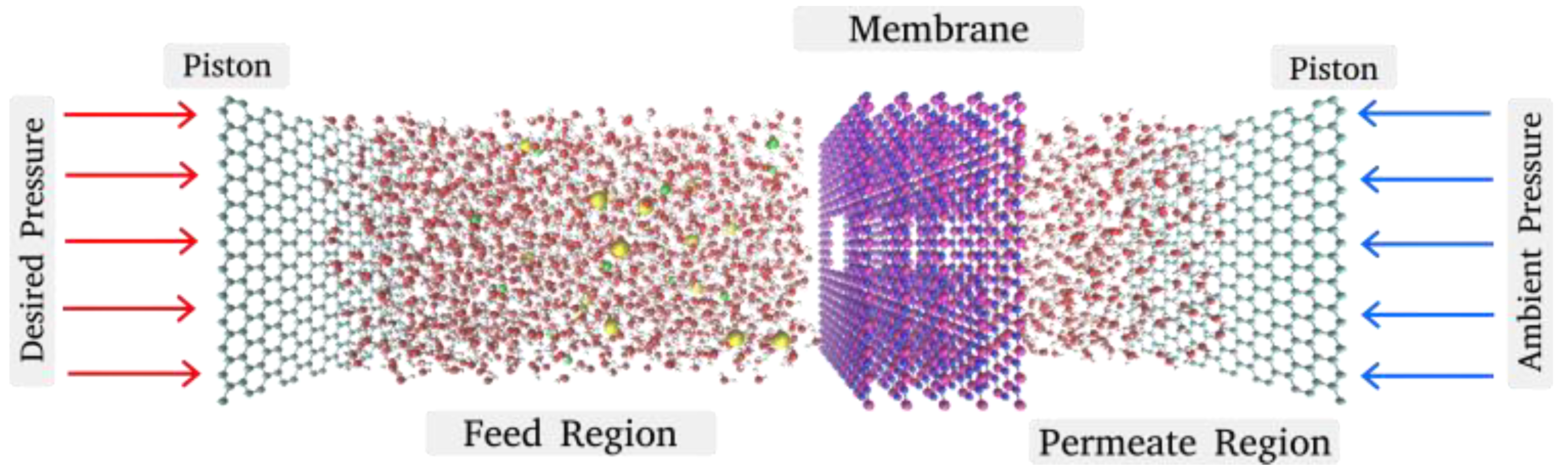
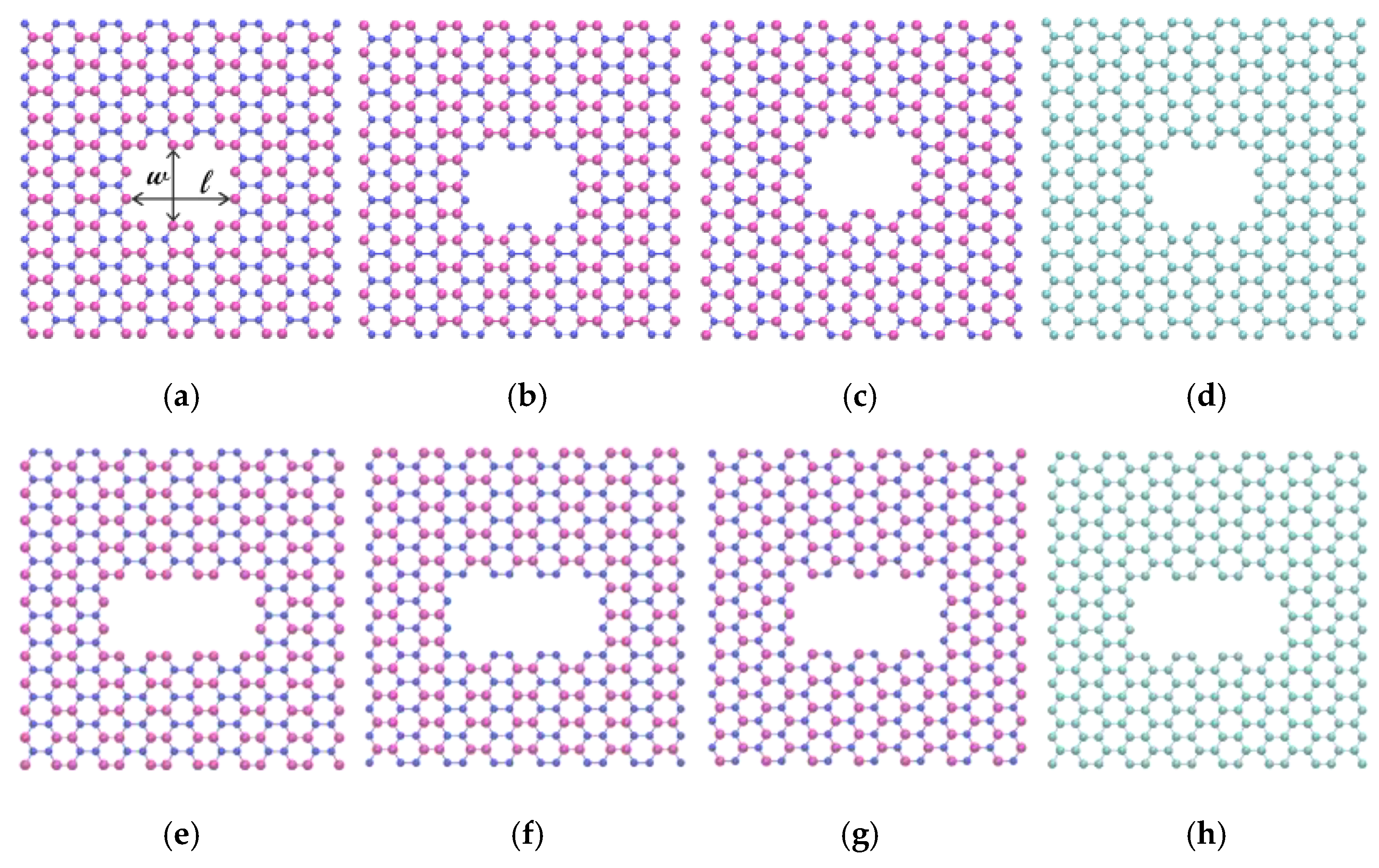
2. Simulation Model and Methods
3. Results and Discussion
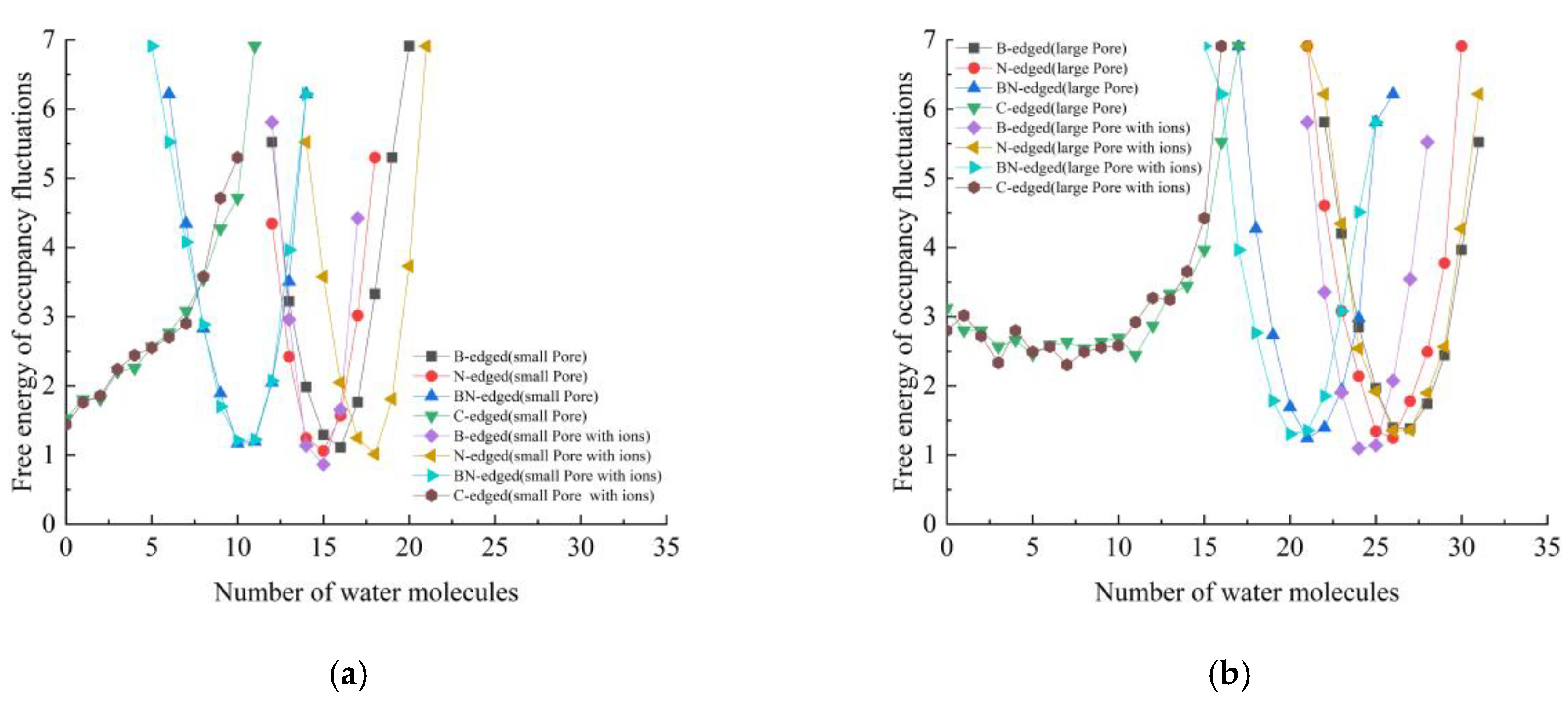
3.1. Occupancy and Free Energy of Occupancy Fluctuations
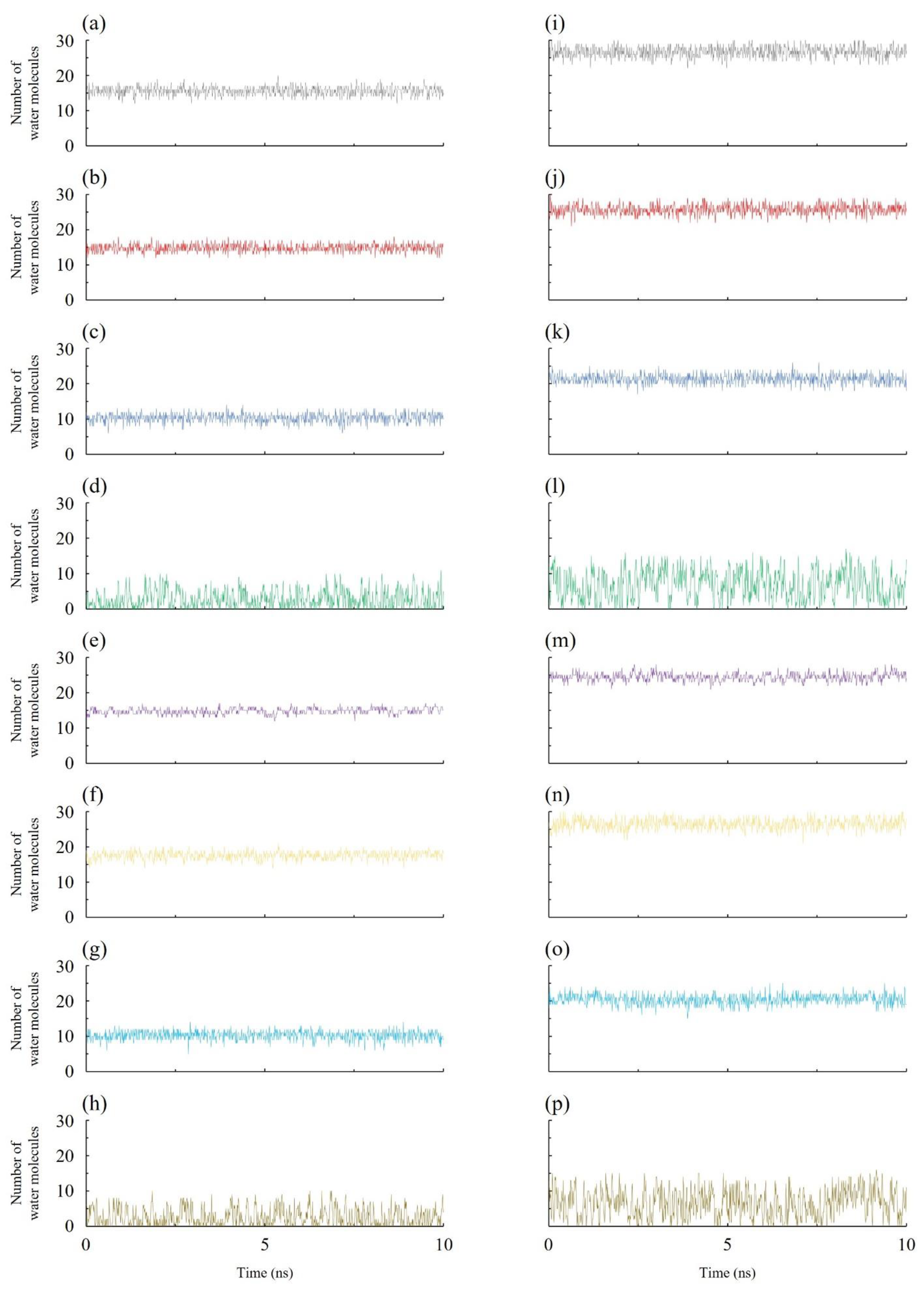
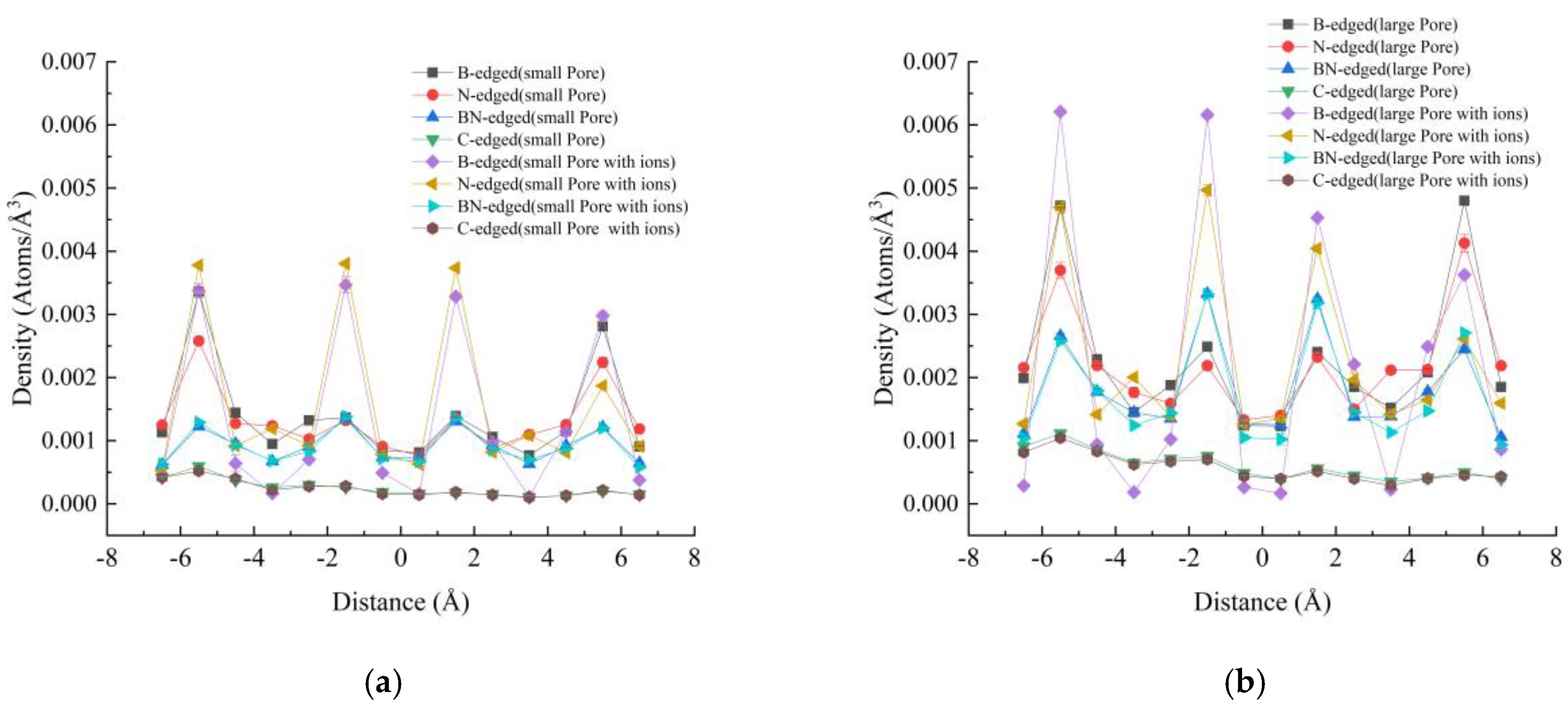
3.2. Density Profiles
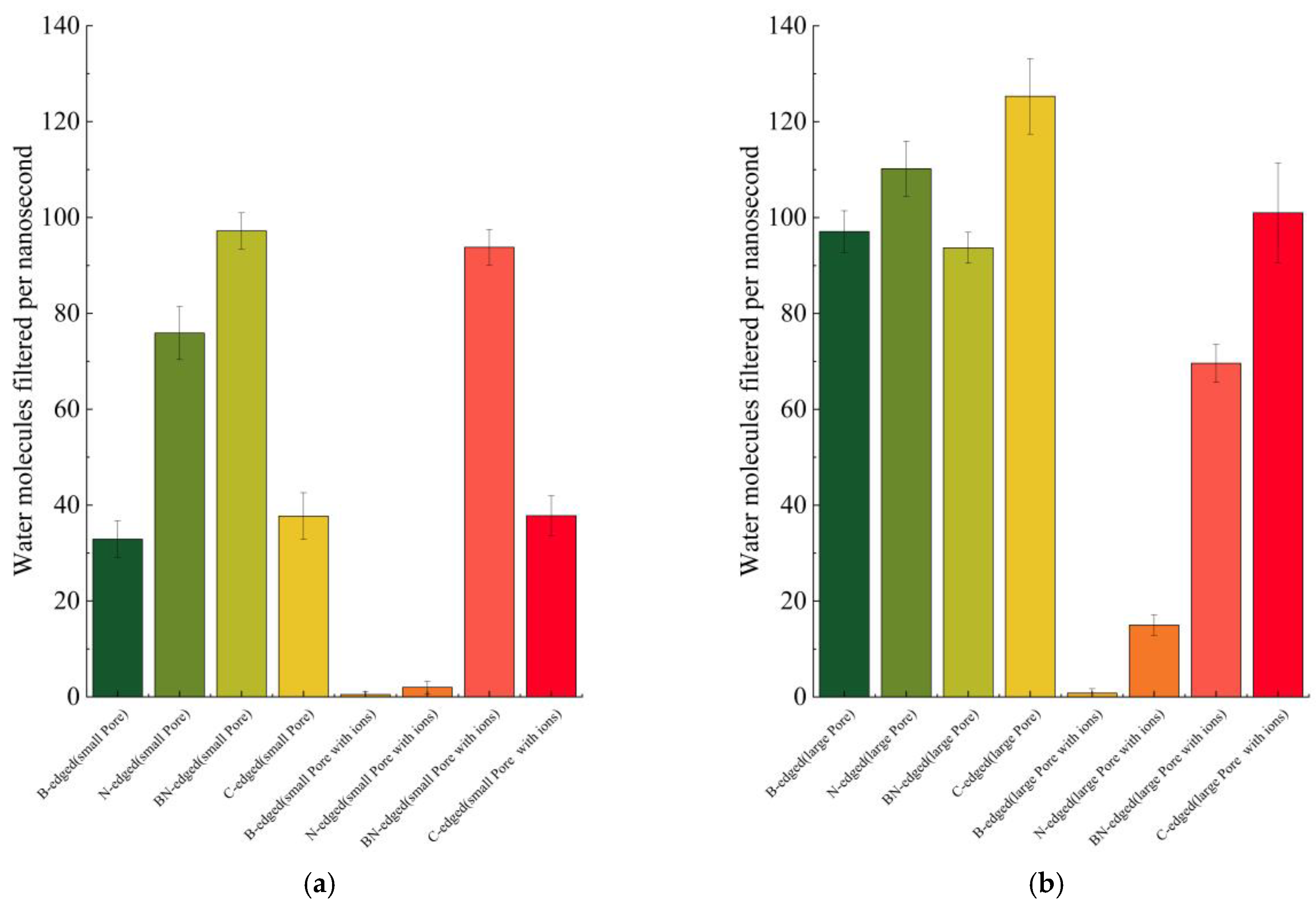
3.3. Water Conduction
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Alamaro, M. Water Politics Must Adapt to a Warming World. Nature 2014, 514, 7. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, J. The Rising Pressure of Global Water Shortages. Nature 2015, 517, 6. [Google Scholar] [CrossRef]
- Elimelech, M.; Phillip, W.A. The Future of Seawater Desalination: Energy, Technology, and the Environment. Science 2011, 333, 712–717. [Google Scholar] [CrossRef]
- Vörösmarty, C.J.; Green, P.; Salisbury, J.; Lammers, R.B. Global Water Resources: Vulnerability from Climate Change and Population Growth. Science 2000, 289, 284–288. [Google Scholar] [CrossRef]
- Elimelech, M. The Global Challenge for Adequate and Safe Water. J. Water Supply Res. Technol.-Aqua 2006, 55, 3–10. [Google Scholar] [CrossRef]
- Zhang, H.; Han, X.; Yu, H.; Zou, Y.; Dong, X. Enhanced Photocatalytic Performance of Boron and Phosphorous Co-Doped Graphitic Carbon Nitride Nanosheets for Removal of Organic Pollutants. Sep. Purif. Technol. 2019, 226, 128–137. [Google Scholar] [CrossRef]
- Manan, S.; Ullah, M.W.; Ul-Islam, M.; Atta, O.M.; Yang, G. Synthesis and Applications of Fungal Mycelium-Based Advanced Functional Materials. J. Bioresour. Bioprod. 2021, 6, 1–10. [Google Scholar] [CrossRef]
- Shao, J.; Ni, Y.; Yan, L. Oxidation of Furfural to Maleic Acid and Fumaric Acid in Deep Eutectic Solvent (DES) under Vanadium Pentoxide Catalysis. J. Bioresour. Bioprod. 2021, 6, 39–44. [Google Scholar] [CrossRef]
- Nasrollahi, N.; Ghalamchi, L.; Vatanpour, V.; Khataee, A. Photocatalytic-Membrane Technology: A Critical Review for Membrane Fouling Mitigation. J. Ind. Eng. Chem. 2021, 93, 101–116. [Google Scholar] [CrossRef]
- Li, Y.S.; Zhang, J.; Tian, G.H.; Shang, H.C.; Tang, H.B. Kirenol, Darutoside and Hesperidin Contribute to the Anti-inflammatory and Analgesic Activities of Siegesbeckia Pubescens Makino by Inhibiting COX-2 Expression and Inflammatory Cell Infiltration. J. Ethnophamacol. 2021, 268, 113547. [Google Scholar] [CrossRef]
- Yan, J.; Wei, D.; Zhao, Z.; Sun, K.; Sun, Y. Osteosarcoma-targeting PtIV Prodrug Amphiphile for Enhanced Chemo-immunotheraphy via Ca2+ Trapping. Acta Biomater. 2025, 193, 474–483. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, L.; Wang, J.; Pan, H.; Dou, M.; Teng, Y.; Fu, X.; Liu, Z.; Huang, X.; Wang, M. Bagasse-based Porous Flower-like MoS2/carbon Composites for Efficient Microwave Absorption. Carbon Lett. 2025, 35, 145–160. [Google Scholar] [CrossRef]
- Zhou, H.; Guo, J.; Zhu, G.; Xu, H.; Tang, X.; Luo, X. Flotation Behavior and Mechanism of Smithsonite under the System of Bidentate Ligand Sulfide Sodium Thiocyanate. Sep. Purif. Technol. 2024, 334, 126086. [Google Scholar] [CrossRef]
- Gao, C.; Jia, S.; Yin, X.; Li, Z.; Yang, G.; Chen, J.; Li, Z.; An, X. Enhancing Open-circuit Voltage in FAPbl3 Perovskite Solar Cells via Self-formation of Coherent Buried Interface FAPblxCl3-x. Chem. Commun. 2025, 61, 2758–2761. [Google Scholar] [CrossRef] [PubMed]
- Younos, T.; Tulou, K.E. Overview of Desalination Techniques. J. Contemp. Water Res. Educ. 2005, 132, 3–10. [Google Scholar] [CrossRef]
- Shon, H.K.; Phuntsho, S.; Vigneswaran, S.; Cho, J. Nanofiltration for Water and Wastewater Treatment-A Mini Review. Drink. Water Eng. Sci. 2013, 6, 47–53. [Google Scholar] [CrossRef]
- Chu, S.; Lin, M.; Li, D.; Lin, R.; Xiao, S. Adaptive Reward Shaping Based Reinforcement Learning for Docking Control of Autonomous Underwater Devices. Ocean Eng. 2025, 318, 120139. [Google Scholar] [CrossRef]
- Yao, Y.; Luo, Y.; Yang, Y.; Sheng, H.; Li, X.; Li, T.; Song, Y.; Zhang, H.; Chen, S.; He, W.; et al. Water Free Anaerobic Co-digestion of Vegetable Processing Waste with Cattle Slurry for Methane Production at High Total Solid Content. Energy 2014, 74, 309–313. [Google Scholar] [CrossRef]
- Yang, Z.Q.; Hou, K.P.; Guo, T.T. Study on the Effects of Different Water-Cement Ratios on the Flow Pattern Properties of Cement Grouts. Appl. Mech. Mater. 2011; 71–78, 1264–1267. [Google Scholar] [CrossRef]
- Shenvi, S.S.; Isloor, A.M.; Ismail, A.F. A Review on RO Membrane Technology: Developments and Challenges. Desalination 2015, 368, 10–26. [Google Scholar] [CrossRef]
- Ahmad, A.L.; Ooi, B.S.; Choudhury, J.P. Preparation and Characterization of Co-Polyamide Thin Film Composite Membrane from Piperazine and 3,5-Diaminobenzoic Acid. Desalination 2003, 158, 101–108. [Google Scholar] [CrossRef]
- Shawky, H.A. Performance of Aromatic Polyamide RO Membranes Synthesized by Interfacial Polycondensation Process in a Water–Tetrahydrofuran System. J. Membr. Sci. 2009, 339, 209–214. [Google Scholar] [CrossRef]
- Park, H.B.; Kamcev, J.; Robeson, L.M.; Elimelech, M.; Freeman, B.D. Maximizing the Right Stuff: The Trade-off between Membrane Permeability and Selectivity. Science 2017, 356, eaab0530. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, F.; Liu, Z.; Cheng, G.; Wang, X.; Ding, J. Molecular Dynamics Study on the Reverse Osmosis Using Multilayer Porous Graphene Membranes. Nanomaterials 2018, 8, 805. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; He, Z.; Gupta, K.M.; Shi, Q.; Lu, R. Molecular Dynamics Study on Water Desalination through Functionalized Nanoporous Graphene. Carbon 2017, 116, 120–127. [Google Scholar] [CrossRef]
- Konatham, D.; Yu, J.; Ho, T.A.; Striolo, A. Simulation Insights for Graphene-Based Water Desalination Membranes. Langmuir 2013, 29, 11884–11897. [Google Scholar] [CrossRef]
- Cohen-Tanugi, D.; Grossman, J.C. Water Desalination across Nanoporous Graphene. Nano Lett. 2012, 12, 3602–3608. [Google Scholar] [CrossRef] [PubMed]
- Madheswaran, D.K.; Thangavelu, P.; Krishna, R.; Thangamuthu, M.; Chandran, A.J.; Colak, I. Carbon-based Materials in Proton Exchange Membrane Fuel Cells: A Critical Review on Performance and Application. Carbon Lett. 2023, 33, 1495–1518. [Google Scholar] [CrossRef]
- Ning, G.; Fan, Z.; Wang, G.; Gao, J.; Qian, W.; Wei, F. Gram-Scale Synthesis of Nanomesh Graphene with High Surface Area and Its Application in Supercapacitor Electrodes. Chem. Commun. 2011, 47, 5976–5978. [Google Scholar] [CrossRef]
- Yan, J.; Fan, Z.; Wei, T.; Qian, W.; Zhang, M.; Wei, F. Fast and Reversible Surface Redox Reaction of Graphene–MnO2 Composites as Supercapacitor Electrodes. Carbon 2010, 48, 3825–3833. [Google Scholar] [CrossRef]
- Yang, R.; Wu, S.; Wang, D.; Xie, G.; Cheng, M.; Wang, G.; Yang, W.; Chen, P.; Shi, D.; Zhang, G. Fabrication of High-Quality All-Graphene Devices with Low Contact Resistances. Nano Res. 2014, 7, 1449–1456. [Google Scholar] [CrossRef]
- Liang, Y.; Qin, H.; Huang, J.; Huan, S.; Hui, D. Mechanical Properties of Boron Nitride Sheet with Randomly Distributed Vacancy Defects. Nanotechnol. Rev. 2019, 8, 210–217. [Google Scholar] [CrossRef]
- Schneider, G.F.; Xu, Q.; Hage, S.; Luik, S.; Spoor, J.N.H.; Malladi, S.; Zandbergen, H.; Dekker, C. Tailoring the Hydrophobicity of Graphene for Its Use as Nanopores for DNA Translocation. Nat. Commun. 2013, 4, 2619. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.Y.; Zulhairun, A.K.; Wong, T.W.; Alireza, S.; Marzuki, M.S.A.; Ismail, A.F. Water Transport Properties of Boron Nitride Nanosheets Mixed Matrix Membranes for Humic Acid Removal. Heliyon 2019, 5, e01142. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.B.; Govind Rajan, A. How Grain Boundaries and Interfacial Electrostatic Interactions Modulate Water Desalination via Nanoporous Hexagonal Boron Nitride. J. Phys. Chem. B 2022, 126, 1284–1300. [Google Scholar] [CrossRef] [PubMed]
- Jafarzadeh, R.; Azamat, J.; Erfan-Niya, H.; Hosseini, M. Molecular Insights into Effective Water Desalination through Functionalized Nanoporous Boron Nitride Nanosheet Membranes. Appl. Surf. Sci. 2019, 471, 921–928. [Google Scholar] [CrossRef]
- Loh, G.C. Fast Water Desalination by Carbon-Doped Boron Nitride Monolayer: Transport Assisted by Water Clustering at Pores. Nanotechnology 2018, 30, 055401. [Google Scholar] [CrossRef]
- Wang, X.; Zhi, C.; Weng, Q.; Bando, Y.; Golberg, D. Boron Nitride Nanosheets: Novel Syntheses and Applications in Polymeric Composites. J. Phys. Conf. Ser. 2013, 471, 012003. [Google Scholar] [CrossRef]
- Golberg, D.; Bando, Y.; Huang, Y.; Terao, T.; Mitome, M.; Tang, C.; Zhi, C. Boron Nitride Nanotubes and Nanosheets. ACS Nano 2010, 4, 2979–2993. [Google Scholar] [CrossRef]
- Geick, R.; Perry, C.H.; Rupprecht, G. Normal Modes in Hexagonal Boron Nitride. Phys. Rev. 1966, 146, 543–547. [Google Scholar] [CrossRef]
- Cartamil-Bueno, S.J.; Cavalieri, M.; Wang, R.; Houri, S.; Hofmann, S.; van der Zant, H.S.J. Mechanical Characterization and Cleaning of CVD Single-Layer h-BN Resonators. Npj 2D Mater. Appl. 2017, 1, 16. [Google Scholar] [CrossRef]
- Alem, N.; Erni, R.; Kisielowski, C.; Rossell, M.D.; Gannett, W.; Zettl, A. Atomically Thin Hexagonal Boron Nitride Probed by Ultrahigh-Resolution Transmission Electron Microscopy. Phys. Rev. B 2009, 80, 155425. [Google Scholar] [CrossRef]
- Zhang, H.; Qiu, Y.; Liu, T.; Yang, X.; Yan, R.; Wu, H.; Li, A.; Liu, J.; Wei, Y.; Yao, Y. H2 Production from Coal by Enriching Sugar Fermentation and Alkane Oxidation with Hyperthermophilic Resistance Microbes in Municipal Wastewater. Chem. Eng. J. 2024, 489, 151487. [Google Scholar] [CrossRef]
- Ma, C.; Yang, L.; Li, M.; He, J.; Hua, C.; Wang, L.; Li, G.; Liu, J.; Yang, J.; Liu, K.; et al. Closed-loop Two-phase Pulsating Heat Pipe towards Heat Export and Thermal Error Control for Spindle-bearing System of Large-size Vertical Machining Center. Appl. Therm. Eng. 2025, 269, 125993. [Google Scholar] [CrossRef]
- Yang, Z.; Zhao, Q.; Gan, J.; Zhang, J.; Chen, M.; Zhu, Y. Damage Evolution Characteristics of Siliceous Slate with Varying Initial Water Content During Freeze-thaw Cycles. Sci. Total Environ. 2024, 950, 175200. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Liu, Y.; Qi, Y.; Song, M.; Jiang, L.; Fu, G.; Li, J. Hexagonal Boron Nitride with Nanoslits as a Membrane for Water Desalination: A Molecular Dynamics Investigation. Sep. Purif. Technol. 2020, 251, 117409. [Google Scholar] [CrossRef]
- Gu, Z.; Liu, S.; Dai, X.; Chen, S.H.; Yang, Z.; Zhou, R. Nanoporous Boron Nitride for High Efficient Water Desalination. bioRxiv 2018, 500876. [Google Scholar] [CrossRef]
- Du, J.; Zhang, Y.; Han, L.; Ma, X.; Li, C.; Li, Q. Insights into Water Permeability and Hg2+ Removal Using Two-Dimensional Nanoporous Boron Nitride. New J. Chem. 2020, 44, 18084–18091. [Google Scholar] [CrossRef]
- SAMSON. OneAngstrom. 2020. Available online: https://www.samson-connect.net/ (accessed on 22 April 2025).
- Park, C.; Robinson, F.; Kim, D. On the Choice of Different Water Model in Molecular Dynamics Simulations of Nanopore Transport Phenomena. Membranes 2022, 12, 1109. [Google Scholar] [CrossRef]
- Hockney, R.W.; Eastwood, J.W. Computer Simulation Using Particles; Taylor & Francis Group: Boca Raton, FL, USA, 1988. [Google Scholar]
- Thompson, A.P.; Aktulga, H.M.; Berger, R.; Bolintineanu, D.S.; Brown, W.M.; Crozier, P.S.; in ’t Veld, P.J.; Kohlmeyer, A.; Moore, S.G.; Nguyen, T.D.; et al. LAMMPS—A Flexible Simulation Tool for Particle-Based Materials Modeling at the Atomic, Meso, and Continuum Scales. Comput. Phys. Commun. 2022, 271, 108171. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual Molecular Dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Evans, D.J.; Holian, B.L. The Nose–Hoover Thermostat. J. Chem. Phys. 1985, 83, 4069–4074. [Google Scholar] [CrossRef]
- Ryckaert, J.-P.; Ciccotti, G.; Berendsen, H.J.C. Numerical Integration of the Cartesian Equations of Motion of a System with Constraints: Molecular Dynamics of n-Alkanes. J. Comput. Phys. 1977, 23, 327–341. [Google Scholar] [CrossRef]
- Chogani, A.; Moosavi, A.; Bagheri Sarvestani, A.; Shariat, M. The Effect of Chemical Functional Groups and Salt Concentration on Performance of Single-Layer Graphene Membrane in Water Desalination Process: A Molecular Dynamics Simulation Study. J. Mol. Liq. 2020, 301, 112478. [Google Scholar] [CrossRef]
- Hummer, G.; Rasaiah, J.C.; Noworyta, J.P. Water Conduction through the Hydrophobic Channel of a Carbon Nanotube. Nature 2001, 414, 188–190. [Google Scholar] [CrossRef] [PubMed]
- Robinson, F.; Shahbabaei, M.; Kim, D. Deformation Effect on Water Transport through Nanotubes. Energies 2019, 12, 4424. [Google Scholar] [CrossRef]
- Robinson, F.; Park, C.; Kim, M.; Kim, D. Defect Induced Deformation Effect on Water Transport through (6, 6) Carbon Nanotube. Chem. Phys. Lett. 2021, 778, 138632. [Google Scholar] [CrossRef]
- Park, C.; Robinson, F.; Kim, D. Effect of Layer Orientation and Pore Morphology on Water Transport in Multilayered Porous Graphene. Micromachines 2022, 13, 1786. [Google Scholar] [CrossRef]
- Won, C.Y.; Aluru, N.R. Water Permeation through a Subnanometer Boron Nitride Nanotube. J. Am. Chem. Soc. 2007, 129, 2748–2749. [Google Scholar] [CrossRef]
- Davoy, X.; Gellé, A.; Lebreton, J.-C.; Tabuteau, H.; Soldera, A.; Szymczyk, A.; Ghoufi, A. High Water Flux with Ions Sieving in a Desalination 2D Sub-Nanoporous Boron Nitride Material. ACS Omega 2018, 3, 6305–6310. [Google Scholar] [CrossRef]
- Tsukanov, A.A.; Shilko, E.V. Computer-Aided Design of Boron Nitride-Based Membranes with Armchair and Zigzag Nanopores for Efficient Water Desalination. Materials 2020, 13, 5256. [Google Scholar] [CrossRef]
- Garnier, L.; Szymczyk, A.; Malfreyt, P.; Ghoufi, A. Physics behind Water Transport through Nanoporous Boron Nitride and Graphene. J. Phys. Chem. Lett. 2016, 7, 3371–3376. [Google Scholar] [CrossRef]
| σ (Å) | ε (kcal/mol) | |
|---|---|---|
| O | 3.178 | 0.15587 |
| B | 3.453 | 0.0949 |
| N | 3.365 | 0.1448 |
| C | 3.3997 | 0.0859 |
| Na | 2.217 | 0.3519 |
| Cl | 4.849 | 0.01838 |
| Type | Length (Å) | Width (Å) |
|---|---|---|
| B-edged (small pore) | 10.12 | 7.51 |
| N-edged (small pore) | 10.12 | 7.51 |
| BN-edged (small pore) | 10.12 | 7.51 |
| C-edged (small pore) | 9.93 | 7.37 |
| B-edged (large pore) | 14.46 | 7.51 |
| N-edged (large pore) | 14.46 | 7.51 |
| BN-edged (large pore) | 14.46 | 7.51 |
| C-edged (large pore) | 14.18 | 7.37 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, C.; Kim, D. Influence of Inner Lining Atoms of Multilayered Hexagonal Boron Nitride Porous Membrane on Desalination. Micromachines 2025, 16, 530. https://doi.org/10.3390/mi16050530
Park C, Kim D. Influence of Inner Lining Atoms of Multilayered Hexagonal Boron Nitride Porous Membrane on Desalination. Micromachines. 2025; 16(5):530. https://doi.org/10.3390/mi16050530
Chicago/Turabian StylePark, Chulwoo, and Daejoong Kim. 2025. "Influence of Inner Lining Atoms of Multilayered Hexagonal Boron Nitride Porous Membrane on Desalination" Micromachines 16, no. 5: 530. https://doi.org/10.3390/mi16050530
APA StylePark, C., & Kim, D. (2025). Influence of Inner Lining Atoms of Multilayered Hexagonal Boron Nitride Porous Membrane on Desalination. Micromachines, 16(5), 530. https://doi.org/10.3390/mi16050530






