Integration of Microfluidic Chip and Probe with a Dual Pump System for Measurement of Single Cells Transient Response
Abstract
1. Introduction
2. Materials and Methods
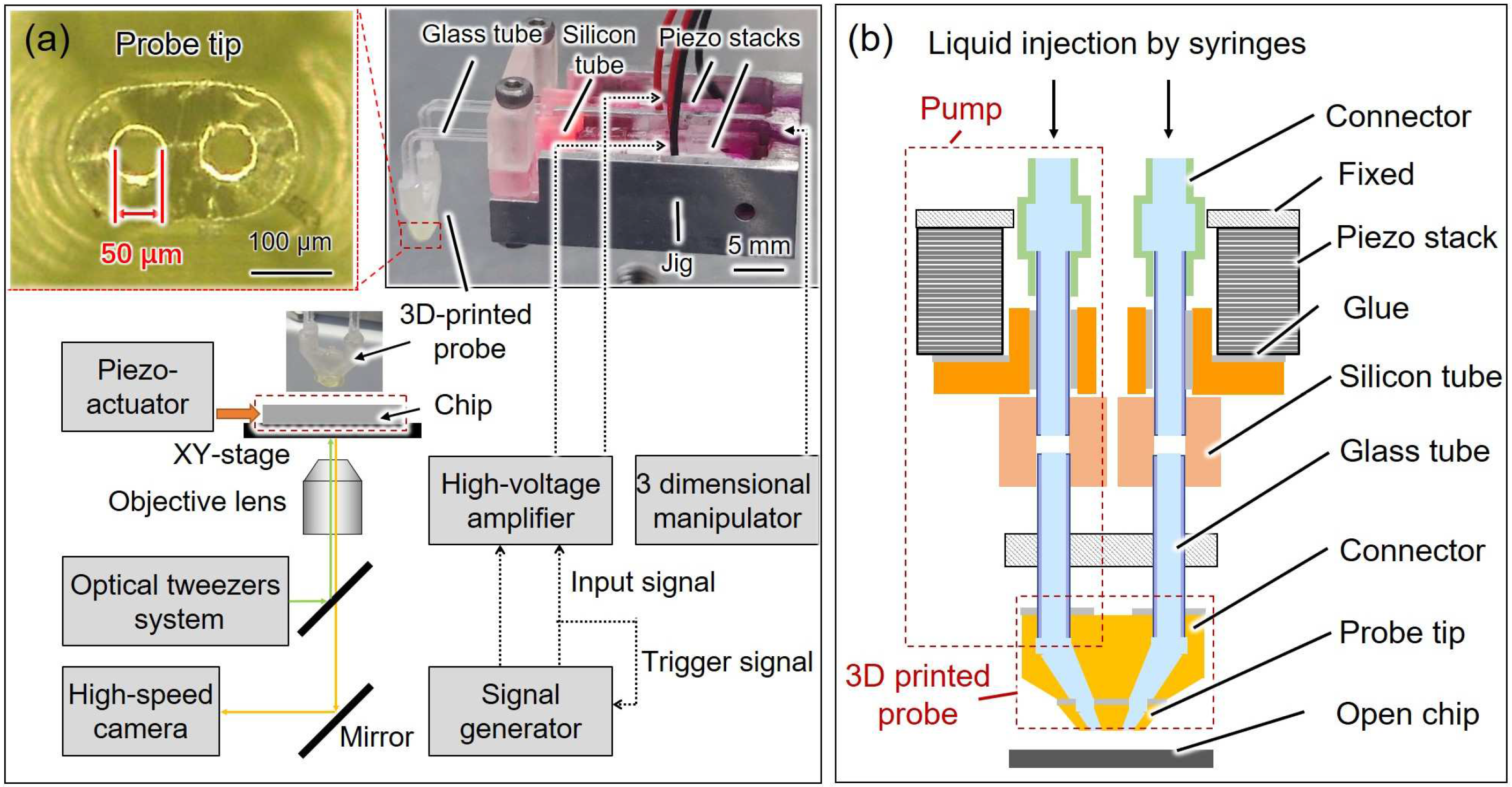
2.1. Overview of the On-Chip Cellular Measurement System
2.2. Probe with the Dual Pump System
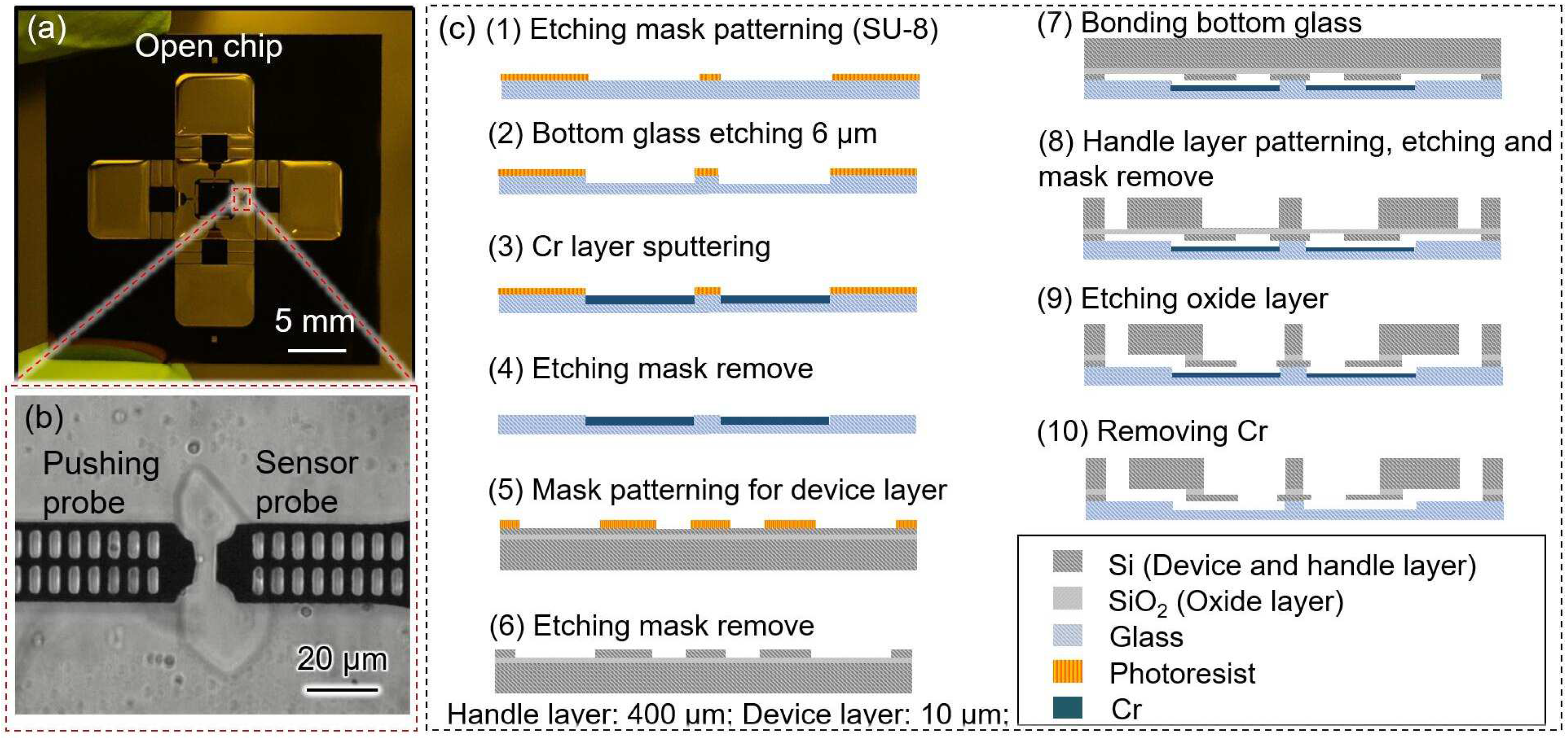
2.3. Robot-Integrated Microfluidic Chip
- (1)
- Spin-coating of SU-8 3010 (Nihon Kayaku Co. Ltd., Gumma, Japan) photoresist onto the glass surface, followed by patterning using a mask aligner.
- (2)
- Deep reactive ion etching (DRIE) of the glass to prevent friction between the on-chip probes and the glass.
- (3)
- Sputtering of a thin layer of Cr onto the etched side of the glass to protect the movable parts, followed by the removal of the photoresist and Cr using a piranha solution.
- (4)
- Spin-coating of the OFPR (Tokyo Ohka Co., Ltd., Tokyo, Japan) photoresist onto the device layer surface.
- (5)
- DRIE etching of the device layer, followed by the removal of the photoresist using a piranha solution.
- (6)
- Bonding of the glass with the device layer.
- (7)
- Spin-coating and patterning of SU-8 onto the handle layer surface.
- (8)
- DRIE etching of the handle layer, followed by the removal of SU-8 using oxygen plasma ashing.
- (9)
- Removal of the silicon dioxide layer using a buffered hydrogen fluoride solution, and cleaning of the chip with a piranha solution.
- (10)
- Removal of the Cr layers on the glass using chrome etchant.
2.4. Cell Culture and Preparation
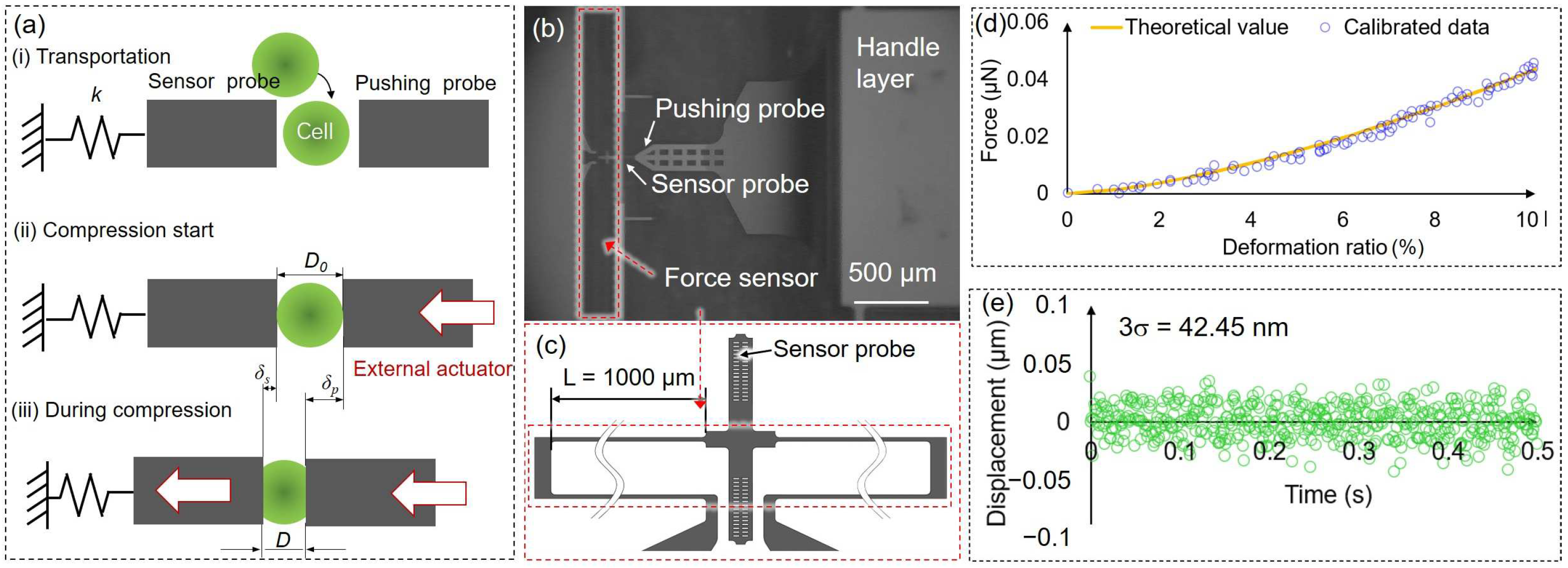
2.5. Compression Concept, Force Sensor Calibration, and Stability of Force Sensor
3. Results and Discussion
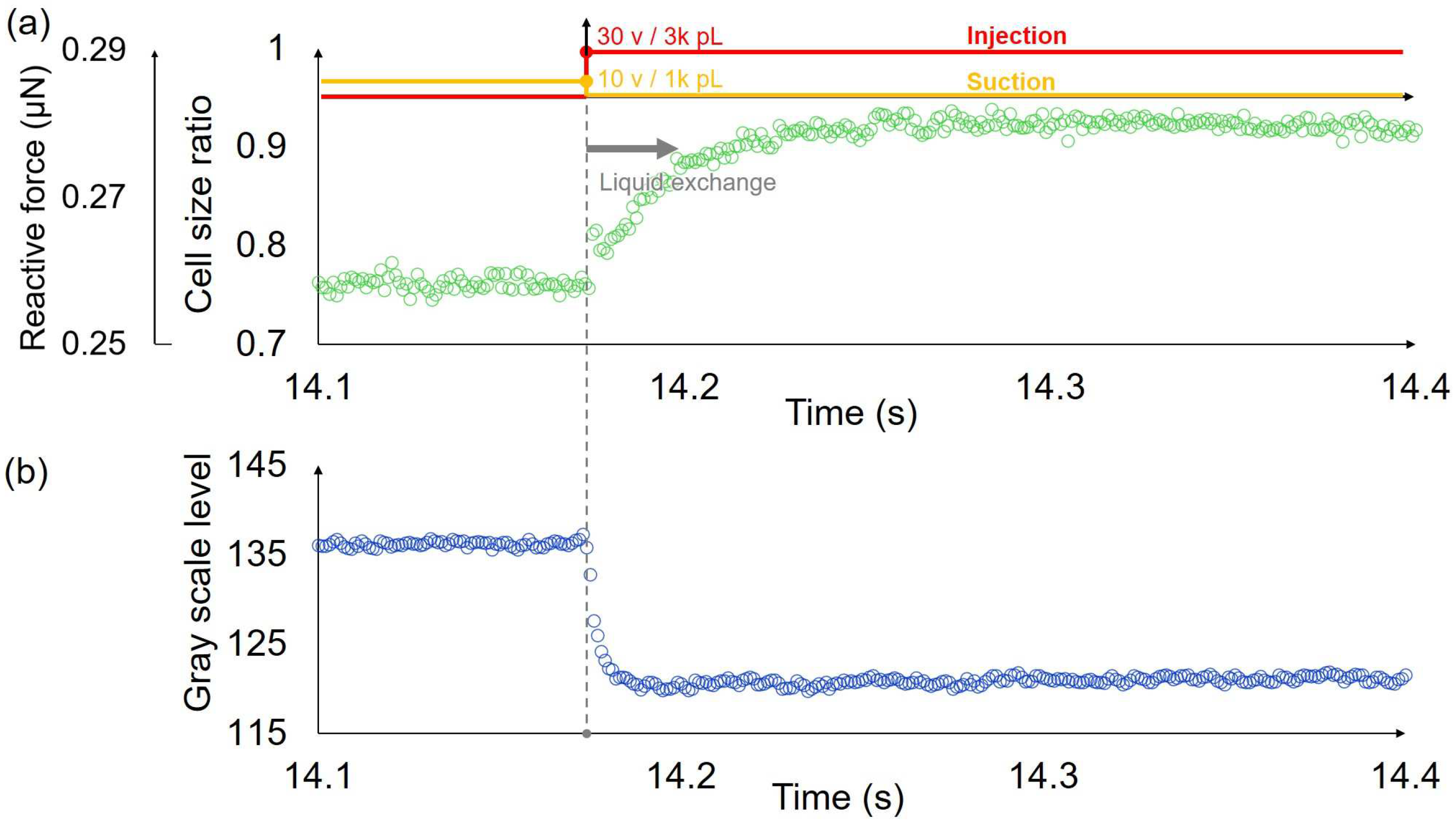
3.1. Liquid Exchange Process
3.2. Measurement of Synechocystis Cells
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Liu, Y.; Fan, Z.; Qiao, L.; Liu, B. Advances in microfluidic strategies for single-cell research. Trends Anal. Chem. 2022, 157, 116822. [Google Scholar] [CrossRef]
- Hagiwara, M.; Maruyama, H.; Akiyama, M.; Koh, I.; Arai, F. Weakening of resistance force by cell–ECM interactions regulate cell migration directionality and pattern formation. Commun. Biol. 2021, 4, 808. [Google Scholar] [CrossRef]
- Hagiwara, M.; Kawahara, T.; Yamanishi, Y.; Masuda, T.; Feng, L.; Arai, F. On-chip magnetically actuated robot with ultrasonic vibration for single cell manipulations. Lab Chip 2011, 11, 2049–2054. [Google Scholar] [CrossRef]
- Sugiura, H.; Sakuma, S.; Kaneko, M.; Arai, F. On-chip method to measure mechanical characteristics of a single cell by using moiré fringe. Micromachines 2015, 6, 660–673. [Google Scholar] [CrossRef]
- Ito, Y.; Nakamura, S.; Sugimoto, N.; Shigemori, T.; Kato, Y.; Ohno, M.; Eto, K. Turbulence activates platelet biogenesis to enable clinical scale ex vivo production. Cell 2018, 174, 636–648. [Google Scholar] [CrossRef]
- Chang, D.; Sakuma, S.; Kera, K.; Uozumi, N.; Arai, F. Measurement of the mechanical properties of single Synechocystis sp. strain PCC6803 cells in different osmotic concentrations using a robot-integrated microfluidic chip. Lab Chip 2018, 18, 1241–1249. [Google Scholar] [CrossRef]
- Du, X.; Chang, D.; Kaneko, S.; Maruyama, H.; Sugiura, H.; Tsujii, M.; Arai, F. Dynamic Deformation Measurement of an Intact Single Cell via Microfluidic Chip with Integrated Liquid Exchange. Engineering 2023, in press. [Google Scholar] [CrossRef]
- Huang, L.; Liang, F.; Feng, Y.; Zhao, P.; Wang, W. On-chip integrated optical stretching and electrorotation enabling single-cell biophysical analysis. Microsyst. Nanoeng. 2020, 6, 57. [Google Scholar] [CrossRef]
- Tarhan, M.C.; Lafitte, N.; Tauran, Y.; Jalabert, L.; Kumemura, M.; Perret, G.; Collard, D. A rapid and practical technique for real-time monitoring of biomolecular interactions using mechanical responses of macromolecules. Sci. Rep. 2016, 6, 28001. [Google Scholar] [CrossRef]
- Shakoor, A.; Xie, M.; Luo, T.; Hou, J.; Shen, Y.; Mills, J.K.; Sun, D. Achieving automated organelle biopsy on small single cells using a cell surgery robotic system. IEEE. Trans. Biomed. Eng. 2018, 66, 2210–2222. [Google Scholar] [CrossRef]
- Barazani, B.; Piercey, M.; Paulson, A.; Warnat, S.; Hubbard, T.; MacIntosh, A.J. Rehydration of active dried yeast: Impact on strength and stiffness of yeast cells measured using microelectromechanical systems. J. Inst. Brew. 2019, 125, 53–59. [Google Scholar] [CrossRef]
- Takayama, Y.; Perret, G.; Kumemura, M.; Ataka, M.; Meignan, S.; Karsten, S.L.; Tarhan, M.C. Developing a MEMS device with built-in microfluidics for biophysical single cell characterization. Micromachines 2018, 9, 275. [Google Scholar] [CrossRef]
- Yang, R.J.; Fu, L.M.; Hou, H.H. Review and perspectives on microfluidic flow cytometers. Sens. Actuators B Chem. 2018, 266, 26–45. [Google Scholar] [CrossRef]
- Takayama, S.; Ostuni, E.; LeDuc, P.; Naruse, K.; Ingber, D.E.; Whitesides, G.M. Subcellular positioning of small molecules. Nature 2001, 411, 1016. [Google Scholar] [CrossRef]
- Tarn, M.D.; Lopez-Martinez, M.J.; Pamme, N. On-chip processing of particles and cells via multilaminar flow streams. Anal. Bioanal. Chem. 2014, 406, 139–161. [Google Scholar] [CrossRef]
- Shinha, K.; Nihei, W.; Kimura, H. A Microfluidic Probe Integrated Device for Spatiotemporal 3D Chemical Stimulation in Cells. Micromachines 2020, 11, 691. [Google Scholar] [CrossRef]
- Ainla, A.; Jansson, E.T.; Stepanyants, N.; Orwar, O.; Jesorka, A. A microfluidic pipette for single-cell pharmacology. Anal. Chem. 2010, 82, 4529–4536. [Google Scholar] [CrossRef]
- Kaigala, G.V.; Lovchik, R.D.; Drechsler, U.; Delamarche, E. A vertical microfluidic probe. Langmuir 2011, 27, 5686–5693. [Google Scholar] [CrossRef]
- Ainla, A.; Jeffries, G.D.; Brune, R.; Orwar, O.; Jesorka, A. A multifunctional pipette. Lab Chip 2012, 12, 1255–1261. [Google Scholar] [CrossRef]
- Momotenko, D.; Cortes-Salazar, F.; Lesch, A.; Wittstock, G.; Girault, H.H. Microfluidic push–pull probe for scanning electrochemical microscopy. Anal. Chem. 2011, 83, 5275–5282. [Google Scholar] [CrossRef]
- Kaigala, G.V.; Lovchik, R.D.; Delamarche, E. Microfluidics in the “open space” for performing localized chemistry on biological interfaces. Angew. Chem. Int. Ed. Engl. 2012, 51, 11224–11240. [Google Scholar] [CrossRef]
- Chen, P.; Li, S.; Guo, Y.; Zeng, X.; Liu, B.F. A review on microfluidics manipulation of the extracellular chemical microenvironment and its emerging application to cell analysis. Anal. Chim. Acta 2020, 1125, 94–113. [Google Scholar] [CrossRef]
- Taylor, D.P.; Mathur, P.; Renaud, P.; Kaigala, G.V. Microscale hydrodynamic confinements: Shaping liquids across length scales as a toolbox in life sciences. Lab Chip 2022, 22, 1415–1437. [Google Scholar] [CrossRef]
- Delamarche, E.; Pereiro, I.; Kashyap, A.; Kaigala, G.V. Biopatterning: The art of patterning biomolecules on surfaces. Langmuir 2021, 37, 9637–9651. [Google Scholar] [CrossRef]
- Takahashi, K.; Kamiya, S.; Takao, H.; Shimokawa, F.; Terao, K. Stainless microfluidic probe with 2D-array microapertures. AIP Adv. 2021, 11, 015331. [Google Scholar] [CrossRef]
- Ajouz, B.; Berrier, C.; Garrigues, A.; Besnard, M.; Ghazi, A. Release of thioredoxin via the mechanosensitive channel MscL during osmotic downshock of Escherichia coli cells. J. Biol. Chem. 1998, 273, 26670–26674. [Google Scholar] [CrossRef]
- Nanatani, K.; Shijuku, T.; Akai, M.; Yukutake, Y.; Yasui, M.; Hamamoto, S.; Uozumi, N. Characterization of the role of a mechanosensitive channel in osmotic down shock adaptation in Synechocystis sp PCC 6803. Channels 2013, 7, 238–242. [Google Scholar] [CrossRef]
- McEvoy, E.; Han, Y.L.; Guo, M.; Shenoy, V.B. Gap junctions amplify spatial variations in cell volume in proliferating tumor spheroids. Nat. Commun. 2020, 11, 6148. [Google Scholar] [CrossRef]
- Veselovsky, N.S.; Engert, F.; Lux, H.D. Fast local superfusion technique. Pflügers Arch. 1996, 432, 351–354. [Google Scholar] [CrossRef]
- Kasai, Y.; Sakuma, S.; Arai, F. Isolation of single motile cells using a high-speed picoliter pipette. Microfluid. Nanofluidics 2019, 23, 18. [Google Scholar] [CrossRef]
- Hertz, H. On the contact of elastic solids. J. Reine Angew. Math. 1881, 92, 156–171. [Google Scholar]
- Johnson, K.L.; Johnson, K.L. Contact Mechanics; Cambridge University Press: Cambridge, UK, 1987. [Google Scholar]
- Du, X.; Kaneko, S.; Maruyama, H.; Sugiura, H.; Arai, F. High-Speed and Pinpoint Liquid Exchange on Microfluidic Chip using 3D Printed Double-Barreled Microprobe with the dual pumps. In Proceeding of the 2023 IEEE 36th International Conference on Micro Electro Mechanical Systems, Munich, Germany, 15–19 January 2023. [Google Scholar]
- Vitali, V.; Sutka, M.; Amodeo, G.; Chara, O.; Ozu, M. The water to solute permeability ratio governs the osmotic volume dynamics in beetroot vacuoles. Front. Plant Sci. 2016, 7, 1388. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, X.; Kaneko, S.; Maruyama, H.; Sugiura, H.; Tsujii, M.; Uozumi, N.; Arai, F. Integration of Microfluidic Chip and Probe with a Dual Pump System for Measurement of Single Cells Transient Response. Micromachines 2023, 14, 1210. https://doi.org/10.3390/mi14061210
Du X, Kaneko S, Maruyama H, Sugiura H, Tsujii M, Uozumi N, Arai F. Integration of Microfluidic Chip and Probe with a Dual Pump System for Measurement of Single Cells Transient Response. Micromachines. 2023; 14(6):1210. https://doi.org/10.3390/mi14061210
Chicago/Turabian StyleDu, Xu, Shingo Kaneko, Hisataka Maruyama, Hirotaka Sugiura, Masaru Tsujii, Nobuyuki Uozumi, and Fumihito Arai. 2023. "Integration of Microfluidic Chip and Probe with a Dual Pump System for Measurement of Single Cells Transient Response" Micromachines 14, no. 6: 1210. https://doi.org/10.3390/mi14061210
APA StyleDu, X., Kaneko, S., Maruyama, H., Sugiura, H., Tsujii, M., Uozumi, N., & Arai, F. (2023). Integration of Microfluidic Chip and Probe with a Dual Pump System for Measurement of Single Cells Transient Response. Micromachines, 14(6), 1210. https://doi.org/10.3390/mi14061210






