Bioengineering Outlook on Cultivated Meat Production
Abstract
1. Introduction
2. Major Cell Types with Potential for Use in Cultivated Meat (CM) Bioprocess
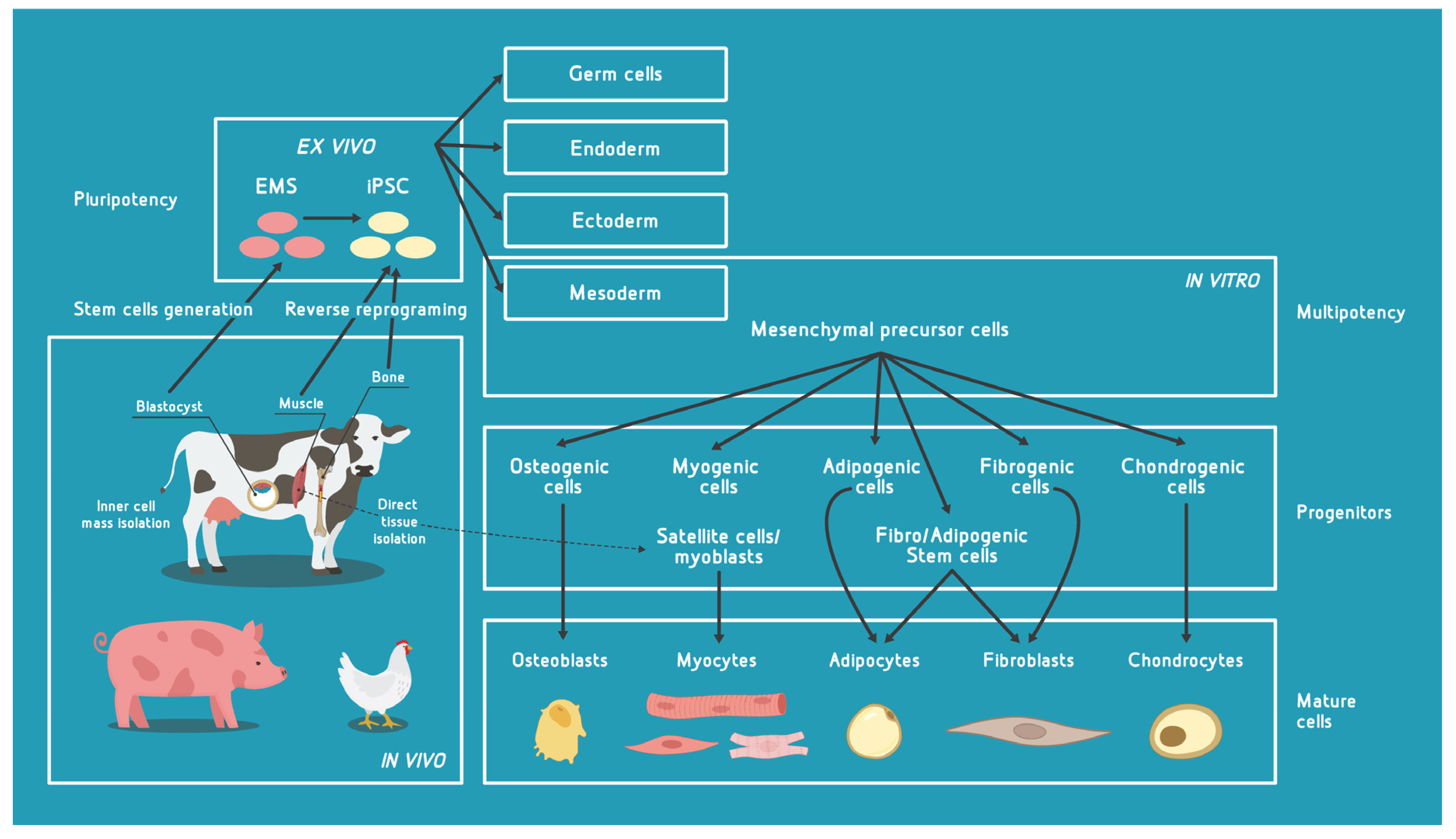
2.1. Pluripotent Cells
2.2. Multipotent Cells
2.3. Cell Immobilization
2.3.1. Microcarriers
2.3.2. Scaffolds
3. General Aspects of Bioreactors for Proliferation and Differentiation in CM Bioprocess
3.1. Two-Dimensional vs. Three-Dimensional Cell Culture for Cultivated Meat Production
3.2. Stirred-Tank Bioreactor
3.3. Rocking-Bed Bioreactor
3.4. Fluidized-Bed Bioreactor
3.5. Perfusion Bioreactor
3.5.1. Fixed-Bed (Packed-Bed) Bioreactor
3.5.2. Hollow-Fiber Bioreactor
3.6. Scale-Up, Scale-Down and Scale-Out Options
4. Bioprocess Conditions for Production of CM
4.1. Bioprocess Parameters
4.1.1. Temperature
4.1.2. pH Value
4.1.3. Aeration and Agitation—Mass and Energy Transfer
4.1.4. Duration
4.2. Media
4.3. Bioprocess Monitoring
4.3.1. Sensing Options
4.3.2. Sensors for Temperature, pH, DO, CO2 and Biomass
4.3.3. Commercially Available Sensors
4.4. Bioprocess Kinetics
4.5. Bioprocess Effluents
5. Bioengineering Aspects to Be Considered in Future Development of CM Industry
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ATF | alternating tangential flow system |
| BAMs | bio-artificial muscles |
| bASCs | adipose-derived stem cells |
| BMPs | morphogenetic proteins |
| CA | cellular agriculture |
| CFD | computational fluid dynamics |
| CHO | chinese hamster ovary cells |
| CM | cultured meat, cultivated meat |
| CMMC | cultivated meat modeling consortium |
| CIP | cleaning-in-place |
| CXCR4 | C-X-C chemokine receptor type 4 |
| DMEM | Dulbecco’s Modified Eagle Medium |
| DO | dissolved oxygen |
| ECM | extracellular matrix |
| EGF | epidermal growth factor |
| ESCs | embryonic stem cells |
| FACS | fluorescence-activated cell sorting |
| FAPs | fibro-adipogenic progenitors |
| FBM | fibroblast basal medium |
| FBR | fluidized-bed reactor |
| FBS | fetal bovine serum |
| FGF | fibroblast growth factor |
| FGF-2 | fibroblast growth factor-2 |
| GFs | growth factors |
| GMP | good manufacturing practice |
| HA | hyaluronic acid |
| HFR | hollow-fiber reactor |
| HGF | hepatocyte growth factor |
| hMSC | human mesenchymal stem cells |
| IGF | insulin-like growth factor |
| iPSC | induced pluripotent stem cells |
| LIF | leukemia inhibitory factor |
| LOC | Lab-on-a-Chip |
| MAPK | mitogen-activated protein kinase |
| MCs | microcarriers |
| MOs | microorganisms |
| MRF | myogenic regulatory factor |
| MSCs | mesenchymal stem cells |
| MyoD | myoblast determination protein 1 |
| PBR | packed-bed reactor |
| PDGF | platelet-derived growth factor |
| PDMS | polydimethylsiloxane |
| PEG | polyethylene glycol |
| PGA | polyglycolic acid |
| PLA | polylactic acid |
| PNIPAM-MCs | poly(N-isopropylacrylamide) grafted MCs |
| R&D | research and development |
| RBR | rocking-bed reactor |
| RTDs | resistance temperature devices |
| SCNT ES | somatic cell nuclear transfer embryonic stem cell |
| SDG | sustainable development goal |
| SIP | sterilization-in-place |
| STR | stirred-tank reactor |
| TE | tissue engineering |
| TFF | tangential flow filtration |
| TGF-β | transforming GF-β |
| TNF-α | tumor necrosis factor-α |
| VEGF | vascular endothelial growth factor |
| VC | venture capital |
References
- FAO. The Future of Food and Agriculture—Trends and Challenges; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2017; p. 180. [Google Scholar]
- Aimutis, W.R. Plant-Based Proteins: The Good, Bad, and Ugly. Annu. Rev. Food Sci. Technol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Godfray, H.C.J. Meat: The Future Series—Alternative Proteins|World Economic Forum; World Economic Forum: Cologny/Geneva, Switzerland, 2019. [Google Scholar]
- Lu, Y.; Nakicenovic, N.; Visbeck, M.; Stevance, A.-S. Policy: Five priorities for the UN Sustainable Development Goals. Nature 2015, 520, 432–433. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Oliveira, P.; Fraga-Corral, M.; Carpena, M.; Prieto, M.A.; Simal-Gandara, J. Approaches for sustainable food production and consumption systems. In Future Foods; Elsevier: Amsterdam, The Netherlands, 2022; pp. 23–38. ISBN 9780323910019. [Google Scholar]
- UN Transforming our World: The 2030 Agenda for Sustainable Development|Department of Economic and Social Affairs. Available online: https://sdgs.un.org/2030agenda (accessed on 19 January 2022).
- Jelinek, H.F.; Mousa, M.; Alefishat, E.; Osman, W.; Spence, I.; Bu, D.; Feng, S.F.; Byrd, J.; Magni, P.A.; Sahibzada, S.; et al. Evolution, ecology, and zoonotic transmission of betacoronaviruses: A review. Front. Vet. Sci. 2021, 8, 644414. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, X.; Zhao, W.; Yang, L.; Wang, Z.; Bi, H. Environmental factors and spatiotemporal distribution characteristics of the global outbreaks of the highly pathogenic avian influenza H5N1. Environ. Sci. Pollut. Res. Int. 2022. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Bai, T.; Shu, Y. Poultry to Human Passport: Cross-species Transmission of Zoonotic H7N9 Avian Influenza Virus to Humans. Zoonoses 2022, 2. [Google Scholar] [CrossRef]
- Nollet, L.M.L. (Ed.) Handbook of Meat, Poultry and Seafood Quality; Blackwell Publishing Ltd.: Oxford, UK, 2012; ISBN 9780470958322. [Google Scholar]
- Warriss, P.D.; Rhodes, D.N. Haemoglobin concentrations in beef. J. Sci. Food Agric. 1977, 28, 931–934. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.A.; Davis-Dusenbery, B.N.; Eggan, K.C. SnapShot: Directed differentiation of pluripotent stem cells. Cell 2012, 149, 1174-1174.e1. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.; Hunter, S. Source and nature of embryonic stem cells. C. R. Biol. 2002, 325, 1003–1007. [Google Scholar] [CrossRef]
- Orkin, S.H.; Hochedlinger, K. Chromatin connections to pluripotency and cellular reprogramming. Cell 2011, 145, 835–850. [Google Scholar] [CrossRef]
- Young, R.A. Control of the embryonic stem cell state. Cell 2011, 144, 940–954. [Google Scholar] [CrossRef] [PubMed]
- Nuschke, A.; Rodrigues, M.; Stolz, D.B.; Chu, C.T.; Griffith, L.; Wells, A. Human mesenchymal stem cells/multipotent stromal cells consume accumulated autophagosomes early in differentiation. Stem Cell Res. Ther. 2014, 5, 140. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Flores, L.; Madrid, J.F.; Gutiérrez, R.; Varela, H.; Valladares, F.; Alvarez-Argüelles, H.; Díaz-Flores, L. Adult stem and transit-amplifying cell location. Histol. Histopathol. 2006, 21, 995–1027. [Google Scholar] [CrossRef] [PubMed]
- Bonab, M.M.; Alimoghaddam, K.; Talebian, F.; Ghaffari, S.H.; Ghavamzadeh, A.; Nikbin, B. Aging of mesenchymal stem cell in vitro. BMC Cell Biol. 2006, 7, 14. [Google Scholar] [CrossRef]
- Post, M.J. Cultured meat from stem cells: Challenges and prospects. Meat Sci. 2012, 92, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Stephens, N.; Di Silvio, L.; Dunsford, I.; Ellis, M.; Glencross, A.; Sexton, A. Bringing cultured meat to market: Technical, socio-political, and regulatory challenges in cellular agriculture. Trends Food Sci. Technol. 2018, 78, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Bogliotti, Y.S.; Wu, J.; Vilarino, M.; Okamura, D.; Soto, D.A.; Zhong, C.; Sakurai, M.; Sampaio, R.V.; Suzuki, K.; Izpisua Belmonte, J.C.; et al. Efficient derivation of stable primed pluripotent embryonic stem cells from bovine blastocysts. Proc. Natl. Acad. Sci. USA 2018, 115, 2090–2095. [Google Scholar] [CrossRef]
- Yuan, Y. Capturing bovine pluripotency. Proc. Natl. Acad. Sci. USA 2018, 115, 1962–1963. [Google Scholar] [CrossRef]
- Loh, Y.-H.; Agarwal, S.; Park, I.-H.; Urbach, A.; Huo, H.; Heffner, G.C.; Kim, K.; Miller, J.D.; Ng, K.; Daley, G.Q. Generation of induced pluripotent stem cells from human blood. Blood 2009, 113, 5476–5479. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Colman, A. Profile of John Gurdon and Shinya Yamanaka, 2012 Nobel laureates in medicine or physiology. Proc. Natl. Acad. Sci. USA 2013, 110, 5740–5741. [Google Scholar] [CrossRef]
- Roberts, R.M.; Yuan, Y.; Genovese, N.; Ezashi, T. Livestock models for exploiting the promise of pluripotent stem cells. ILAR J. 2015, 56, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, S.; Blau, H.M. Nuclear reprogramming to a pluripotent state by three approaches. Nature 2010, 465, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Specht, E.A.; Welch, D.R.; Rees Clayton, E.M.; Lagally, C.D. Opportunities for applying biomedical production and manufacturing methods to the development of the clean meat industry. Biochem. Eng. J. 2018, 132, 161–168. [Google Scholar] [CrossRef]
- Post, M.J.; Levenberg, S.; Kaplan, D.L.; Genovese, N.; Fu, J.; Bryant, C.J.; Negowetti, N.; Verzijden, K.; Moutsatsou, P. Scientific, sustainability and regulatory challenges of cultured meat. Nat. Food 2020, 1, 403–415. [Google Scholar] [CrossRef]
- Chal, J.; Pourquié, O. Making muscle: Skeletal myogenesis in vivo and in vitro. Development 2017, 144, 2104–2122. [Google Scholar] [CrossRef]
- Rao, L.; Qian, Y.; Khodabukus, A.; Ribar, T.; Bursac, N. Engineering human pluripotent stem cells into a functional skeletal muscle tissue. Nat. Commun. 2018, 9, 126. [Google Scholar] [CrossRef]
- Genovese, N.J.; Domeier, T.L.; Telugu, B.P.V.L.; Roberts, R.M. Enhanced Development of Skeletal Myotubes from Porcine Induced Pluripotent Stem Cells. Sci. Rep. 2017, 7, 41833. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, T.; Thomson, J.A. Defined, feeder-independent medium for human embryonic stem cell culture. Curr. Protoc. Stem Cell Biol. 2007, 2, 1C.2. [Google Scholar] [CrossRef]
- Chen, G.; Gulbranson, D.R.; Hou, Z.; Bolin, J.M.; Ruotti, V.; Probasco, M.D.; Smuga-Otto, K.; Howden, S.E.; Diol, N.R.; Propson, N.E.; et al. Chemically defined conditions for human iPSC derivation and culture. Nat. Methods 2011, 8, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Burrell, K.; Dardari, R.; Goldsmith, T.; Toms, D.; Villagomez, D.A.F.; King, W.A.; Ungrin, M.; West, F.D.; Dobrinski, I. Stirred suspension bioreactor culture of porcine induced pluripotent stem cells. Stem. Cells Dev. 2019, 28, 1264–1275. [Google Scholar] [CrossRef]
- Reiss, J.; Robertson, S.; Suzuki, M. Cell Sources for Cultivated Meat: Applications and Considerations throughout the Production Workflow. Int. J. Mol. Sci. 2021, 22, 7513. [Google Scholar] [CrossRef] [PubMed]
- Asakura, A.; Komaki, M.; Rudnicki, M. Muscle satellite cells are multipotential stem cells that exhibit myogenic, osteogenic, and adipogenic differentiation. Differentiation 2001, 68, 245–253. [Google Scholar] [CrossRef]
- Shaikh, S.; Lee, E.; Ahmad, K.; Ahmad, S.-S.; Chun, H.; Lim, J.; Lee, Y.; Choi, I. Cell types used for cultured meat production and the importance of myokines. Foods 2021, 10, 2318. [Google Scholar] [CrossRef] [PubMed]
- Post, M.J. Cultured beef: Medical technology to produce food. J. Sci. Food Agric. 2014, 94, 1039–1041. [Google Scholar] [CrossRef]
- Joe, A.W.B.; Yi, L.; Natarajan, A.; Le Grand, F.; So, L.; Wang, J.; Rudnicki, M.A.; Rossi, F.M.V. Muscle injury activates resident fibro/adipogenic progenitors that facilitate myogenesis. Nat. Cell Biol. 2010, 12, 153–163. [Google Scholar] [CrossRef]
- Uezumi, A.; Fukada, S.; Yamamoto, N.; Takeda, S.; Tsuchida, K. Mesenchymal progenitors distinct from satellite cells contribute to ectopic fat cell formation in skeletal muscle. Nat. Cell Biol. 2010, 12, 143–152. [Google Scholar] [CrossRef]
- D’Angelo, W.; Chen, B.; Gurung, C.; Guo, Y.-L. Characterization of embryonic stem cell-differentiated fibroblasts as mesenchymal stem cells with robust expansion capacity and attenuated innate immunity. Stem Cell Res. Ther. 2018, 9, 278. [Google Scholar] [CrossRef] [PubMed]
- Okamura, L.H.; Cordero, P.; Palomino, J.; Parraguez, V.H.; Torres, C.G.; Peralta, O.A. Myogenic Differentiation Potential of Mesenchymal Stem Cells Derived from Fetal Bovine Bone Marrow. Anim. Biotechnol. 2018, 29, 1–11. [Google Scholar] [CrossRef]
- Ramírez-Espinosa, J.J.; González-Dávalos, L.; Shimada, A.; Piña, E.; Varela-Echavarria, A.; Mora, O. Bovine (Bos taurus) Bone Marrow Mesenchymal Cell Differentiation to Adipogenic and Myogenic Lineages. Cells Tissues Organs (Print) 2016, 201, 51–64. [Google Scholar] [CrossRef] [PubMed]
- King, J.A.; Miller, W.M. Bioreactor development for stem cell expansion and controlled differentiation. Curr. Opin. Chem. Biol. 2007, 11, 394–398. [Google Scholar] [CrossRef]
- Cardoso, T.C.; Ferrari, H.F.; Garcia, A.F.; Novais, J.B.; Silva-Frade, C.; Ferrarezi, M.C.; Andrade, A.L.; Gameiro, R. Isolation and characterization of Wharton’s jelly-derived multipotent mesenchymal stromal cells obtained from bovine umbilical cord and maintained in a defined serum-free three-dimensional system. BMC Biotechnol. 2012, 12, 18. [Google Scholar] [CrossRef]
- Gottipamula, S.; Muttigi, M.S.; Chaansa, S.; Ashwin, K.M.; Priya, N.; Kolkundkar, U.; SundarRaj, S.; Majumdar, A.S.; Seetharam, R.N. Large-scale expansion of pre-isolated bone marrow mesenchymal stromal cells in serum-free conditions. J. Tissue Eng. Regen. Med. 2016, 10, 108–119. [Google Scholar] [CrossRef]
- Du, M.; Huang, Y.; Das, A.K.; Yang, Q.; Duarte, M.S.; Dodson, M.V.; Zhu, M.J. Meat Science and Muscle Biology Symposium: Manipulating mesenchymal progenitor cell differentiation to optimize performance and carcass value of beef cattle. J. Anim. Sci. 2013, 91, 1419–1427. [Google Scholar] [CrossRef]
- Kim, M.; Choi, Y.S.; Yang, S.H.; Hong, H.-N.; Cho, S.-W.; Cha, S.M.; Pak, J.H.; Kim, C.W.; Kwon, S.W.; Park, C.J. Muscle regeneration by adipose tissue-derived adult stem cells attached to injectable PLGA spheres. Biochem. Biophys. Res. Commun. 2006, 348, 386–392. [Google Scholar] [CrossRef]
- Zidarič, T.; Milojević, M.; Vajda, J.; Vihar, B.; Maver, U. Cultured Meat: Meat Industry Hand in Hand with Biomedical Production Methods. Food Eng. Rev. 2020, 12, 498–519. [Google Scholar] [CrossRef]
- Dodson, M.; Martin, E.; Brannon, M.; Mathison, B.; McFarland, D. Optimization of bovine satellite cell-derived myotube formation in vitro. Tissue Cell. 1987, 19, 159–166. [Google Scholar] [CrossRef]
- Muroya, S.; Nakajima, I.; Chikuni, K. Bovine Skeletal Muscle Cells Predominantly Express a Vascular Cell Adhesion Molecule-1 Seven-Ig Domain Splice Form. Zool. Sci. 2001, 18, 797–805. [Google Scholar] [CrossRef][Green Version]
- Ding, S.; Swennen, G.N.M.; Messmer, T.; Gagliardi, M.; Molin, D.G.M.; Li, C.; Zhou, G.; Post, M.J. Maintaining bovine satellite cells stemness through p38 pathway. Sci. Rep. 2018, 8, 10808. [Google Scholar] [CrossRef] [PubMed]
- MacQueen, L.A.; Alver, C.G.; Chantre, C.O.; Ahn, S.; Cera, L.; Gonzalez, G.M.; O’Connor, B.B.; Drennan, D.J.; Peters, M.M.; Motta, S.E.; et al. Muscle tissue engineering in fibrous gelatin: Implications for meat analogs. NPJ Sci. Food 2019, 3, 20. [Google Scholar] [CrossRef]
- Furuhashi, M.; Morimoto, Y.; Shima, A.; Nakamura, F.; Ishikawa, H.; Takeuchi, S. Formation of contractile 3D bovine muscle tissue for construction of millimetre-thick cultured steak. NPJ Sci. Food 2021, 5, 6. [Google Scholar] [CrossRef]
- Yablonka-Reuveni, Z.; Quinn, L.S.; Nameroff, M. Isolation and clonal analysis of satellite cells from chicken pectoralis muscle. Dev. Biol. 1987, 119, 252–259. [Google Scholar] [CrossRef]
- Baquero-Perez, B.; Kuchipudi, S.V.; Nelli, R.K.; Chang, K.-C. A simplified but robust method for the isolation of avian and mammalian muscle satellite cells. BMC Cell Biol. 2012, 13, 16. [Google Scholar] [CrossRef]
- Jankowski, M.; Mozdziak, P.; Petitte, J.; Kulus, M.; Kempisty, B. Avian satellite cell plasticity. Animals 2020, 10, 1322. [Google Scholar] [CrossRef]
- Dodson, M.V.; McFarland, D.C.; Martin, E.L.; Brannon, M.A. Isolation of satellite cells from ovine skeletal muscle. J. Tissue Cult. Methods. 1986, 10, 233–237. [Google Scholar] [CrossRef]
- Doumit, M.E.; Merkel, R.A. Conditions for isolation and culture of porcine myogenic satellite cells. Tissue Cell. 1992, 24, 253–262. [Google Scholar] [CrossRef]
- Li, B.-J.; Li, P.-H.; Huang, R.-H.; Sun, W.-X.; Wang, H.; Li, Q.-F.; Chen, J.; Wu, W.-J.; Liu, H.-L. Isolation, culture and identification of porcine skeletal muscle satellite cells. Asian-Australas J Anim Sci. 2015, 28, 1171–1177. [Google Scholar] [CrossRef]
- Powell, R.L.; Dodson, M.V.; Cloud, J.G. Cultivation and differentiation of satellite cells from skeletal muscle of the rainbow trout Salmo gairdneri. J. Exp. Zool. 1989, 250, 333–338. [Google Scholar] [CrossRef]
- Matschak, T.W.; Stickland, N.C. The growth of Atlantic salmon (Salmo salar L.) myosatellite cells in culture at two different temperatures. Experientia 1995, 51, 260–266. [Google Scholar] [CrossRef]
- Millan-Cubillo, A.F.; Martin-Perez, M.; Ibarz, A.; Fernandez-Borras, J.; Gutiérrez, J.; Blasco, J. Proteomic characterization of primary cultured myocytes in a fish model at different myogenesis stages. Sci. Rep. 2019, 9, 14126. [Google Scholar] [CrossRef] [PubMed]
- Redondo, P.A.; Pavlou, M.; Loizidou, M.; Cheema, U. Elements of the niche for adult stem cell expansion. J. Tissue Eng. 2017, 8, 2041731417725464. [Google Scholar] [CrossRef]
- Ben-Arye, T.; Levenberg, S. Cultured Meat Compositions. U.S. Patent 16/631,217, 7 May 2020. [Google Scholar]
- Shima, A.; Itou, A.; Takeuchi, S. Cell fibers promote proliferation of co-cultured cells on a dish. Sci. Rep. 2020, 10, 288. [Google Scholar] [CrossRef]
- Jo, B.; Nie, M.; Shima, A.; Morimoto, Y.; Takeuchi, S. Micro Tissue Assembly for Co-Culturing 3D Skeletal Muscle and Adipose Tissues. In Proceedings of the 2020 IEEE 33rd International Conference on Micro Electro Mechanical Systems (MEMS), Vancouver, BC, Canada, 18–22 January 2020; pp. 459–460. [Google Scholar]
- Haraguchi, Y.; Shimizu, T. Three-dimensional tissue fabrication system by co-culture of microalgae and animal cells for production of thicker and healthy cultured food. Biotechnol. Lett. 2021, 43, 1117–1129. [Google Scholar] [CrossRef]
- Soice, E.; Johnston, J. Immortalizing cells for human consumption. Int. J. Mol. Sci. 2021, 22, 1660. [Google Scholar] [CrossRef] [PubMed]
- Bomkamp, C.; Skaalure, S.C.; Fernando, G.F.; Ben-Arye, T.; Swartz, E.W.; Specht, E.A. Scaffolding biomaterials for 3D cultivated meat: Prospects and challenges. Adv. Sci. 2021, 9, e2102908. [Google Scholar] [CrossRef]
- Bodiou, V.; Moutsatsou, P.; Post, M.J. Microcarriers for upscaling cultured meat production. Front. Nutr. 2020, 7, 10. [Google Scholar] [CrossRef]
- Djisalov, M.; Knežić, T.; Podunavac, I.; Živojević, K.; Radonic, V.; Knežević, N.Ž.; Bobrinetskiy, I.; Gadjanski, I. Cultivating multidisciplinarity: Manufacturing and sensing challenges in cultured meat production. Biology 2021, 10, 204. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.L.; Rodrigues, C.A.V.; Gomes, A.R.; Vieira, S.F.; Badenes, S.M.; Diogo, M.M.; Cabral, J.M.S. Dissolvable Microcarriers Allow Scalable Expansion and Harvesting Of Human Induced Pluripotent Stem Cells Under Xeno-Free Conditions. Biotechnol. J. 2019, 14, e1800461. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.K.W.; Chen, A.K.; Mok, Y.; Chen, X.; Lim, U.-M.; Chin, A.; Choo, A.B.H.; Reuveny, S. Long-term microcarrier suspension cultures of human embryonic stem cells. Stem Cell Res. 2009, 2, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Chen, Y.; Ordovas, L.; Verfaillie, C.M. Hepatic differentiation of human embryonic stem cells on microcarriers. J. Biotechnol. 2014, 174, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, C.J.; Lee, K.; Nienow, A.W.; Thomas, R.J.; Smith, M.; Thomas, C.R. Expansion of human mesenchymal stem cells on microcarriers. Biotechnol. Lett. 2011, 33, 2325–2335. [Google Scholar] [CrossRef] [PubMed]
- Rafiq, Q.A.; Brosnan, K.M.; Coopman, K.; Nienow, A.W.; Hewitt, C.J. Culture of human mesenchymal stem cells on microcarriers in a 5 l stirred-tank bioreactor. Biotechnol. Lett. 2013, 35, 1233–1245. [Google Scholar] [CrossRef]
- Tavassoli, H.; Alhosseini, S.N.; Tay, A.; Chan, P.P.Y.; Weng Oh, S.K.; Warkiani, M.E. Large-scale production of stem cells utilizing microcarriers: A biomaterials engineering perspective from academic research to commercialized products. Biomaterials 2018, 181, 333–346. [Google Scholar] [CrossRef] [PubMed]
- de Lucena-Thomas, J.P.; Boonprasirt, P.; Luetchford, K.; De Bank, P.; Ellis, M. Bed expansion properties of tissue engineering particles in a fluidised bed bioreactor. Biochem. Eng. J. 2020, 160, 107632. [Google Scholar] [CrossRef]
- Yuan, Y.; He, N.; Xue, Q.; Guo, Q.; Dong, L.; Haruna, M.H.; Zhang, X.; Li, B.; Li, L. Shellac: A promising natural polymer in the food industry. Trends Food Sci. Technol. 2021, 109, 139–153. [Google Scholar] [CrossRef]
- Ikeda, K.; Takeuchi, S. Anchorage-dependent cell expansion in fiber-shaped microcarrier aggregates. Biotechnol. Prog. 2019, 35, e2755. [Google Scholar] [CrossRef] [PubMed]
- Marga, F.; Forgacs, G.; Purcell, B.P.; Forgacs, A. Edible and Animal-Product-Free Microcarriers for Engineered Meat. U.S. Patent 9,752,122, 5 September 2017. [Google Scholar]
- Shit, S.C.; Shah, P.M. Edible polymers: Challenges and opportunities. J. Polym. 2014, 2014, 1–13. [Google Scholar] [CrossRef]
- Wu, C.-Y.; Stoecklein, D.; Kommajosula, A.; Lin, J.; Owsley, K.; Ganapathysubramanian, B.; Di Carlo, D. Shaped 3D microcarriers for adherent cell culture and analysis. Microsyst. Nanoeng. 2018, 4, 21. [Google Scholar] [CrossRef]
- Chang, C.; Zhang, L. Cellulose-based hydrogels: Present status and application prospects. Carbohydr. Polym. 2011, 84, 40–53. [Google Scholar] [CrossRef]
- Ahmadi, F.; Oveisi, Z.; Samani, S.M.; Amoozgar, Z. Chitosan based hydrogels: Characteristics and pharmaceutical applications. Res. Pharm. Sci. 2015, 10, 1–16. [Google Scholar] [PubMed]
- Ferrer, B. Cultured meat: Matrix Meats’ exec details nanofiber system that “mimics extracellular matrix found in living animals”. Food Ingred. First 2021. [Google Scholar]
- Hu, J.L.; Todhunter, M.E.; LaBarge, M.A.; Gartner, Z.J. Opportunities for organoids as new models of aging. J. Cell Biol. 2018, 217, 39–50. [Google Scholar] [CrossRef]
- Visconti, R.P.; Kasyanov, V.; Gentile, C.; Zhang, J.; Markwald, R.R.; Mironov, V. Towards organ printing: Engineering an intra-organ branched vascular tree. Expert Opin. Biol. Ther. 2010, 10, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Shahin-Shamsabadi, A.; Selvaganapathy, P.R. A rapid biofabrication technique for self-assembled collagen-based multicellular and heterogeneous 3D tissue constructs. Acta Biomater. 2019, 92, 172–183. [Google Scholar] [CrossRef]
- Koudan, E.V.; Gryadunova, A.A.; Karalkin, P.A.; Korneva, J.V.; Meteleva, N.Y.; Babichenko, I.I.; Volkov, A.V.; Rodionov, S.A.; Parfenov, V.A.; Pereira, F.D.A.S.; et al. Multiparametric Analysis of Tissue Spheroids Fabricated from Different Types of Cells. Biotechnol. J. 2020, 15, e1900217. [Google Scholar] [CrossRef] [PubMed]
- Milián, E.; Julien, T.; Biaggio, R.; Venereo-Sanchez, A.; Montes, J.; Manceur, A.P.; Ansorge, S.; Petiot, E.; Rosa-Calatrava, M.; Kamen, A. Accelerated mass production of influenza virus seed stocks in HEK-293 suspension cell cultures by reverse genetics. Vaccine 2017, 35, 3423–3430. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Tsai, A.-C.; Farrance, I.; Rowley, J.; Ma, T. Aggregation of Culture Expanded Human Mesenchymal Stem Cells in Microcarrier-based Bioreactor. Biochem. Eng. J. 2018, 131, 39–46. [Google Scholar] [CrossRef]
- Seah, J.S.H.; Singh, S.; Tan, L.P.; Choudhury, D. Scaffolds for the manufacture of cultured meat. Crit. Rev. Biotechnol. 2021, 1–13. [Google Scholar] [CrossRef]
- Datar, I.; Betti, M. Possibilities for an in vitro meat production system. Innov. Food Sci. Emerg. Technol. 2010, 11, 13–22. [Google Scholar] [CrossRef]
- Levy-Mishali, M.; Zoldan, J.; Levenberg, S. Effect of scaffold stiffness on myoblast differentiation. Tissue Eng. Part A 2009, 15, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Comley, K.; Fleck, N.A. The toughness of adipose tissue: Measurements and physical basis. J. Biomech. 2010, 43, 1823–1826. [Google Scholar] [CrossRef]
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar] [CrossRef]
- Ben-Arye, T.; Levenberg, S. Tissue engineering for clean meat production. Front. Sustain. Food Syst. 2019, 3, 46. [Google Scholar] [CrossRef]
- Post, M.; van der Weele, C. Principles of tissue engineering for food. In Principles of Tissue Engineering; Elsevier: Amsterdam, The Netherlands, 2014; pp. 1647–1662. ISBN 9780123983589. [Google Scholar]
- Ben-Arye, T.; Shandalov, Y.; Ben-Shaul, S.; Landau, S.; Zagury, Y.; Ianovici, I.; Lavon, N.; Levenberg, S. Textured soy protein scaffolds enable the generation of three-dimensional bovine skeletal muscle tissue for cell-based meat. Nat. Food 2020. [Google Scholar] [CrossRef]
- Ong, S.; Choudhury, D.; Naing, M.W. Cell-based meat: Current ambiguities with nomenclature. Trends Food Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Handral, H.K.; Hua Tay, S.; Wan Chan, W.; Choudhury, D. 3D Printing of cultured meat products. Crit. Rev. Food Sci. Nutr. 2020, 62, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bilirgen, A.C.; Toker, M.; Odabas, S.; Yetisen, A.K.; Garipcan, B.; Tasoglu, S. Plant-Based Scaffolds in Tissue Engineering. ACS Biomater. Sci. Eng. 2021, 7, 926–938. [Google Scholar] [CrossRef] [PubMed]
- Holmes, J.T.; Jaberansari, Z.; Collins, W.; Latour, M.L.; Modulevsky, D.J.; Pelling, A.E. Homemade bread: Repurposing an ancient technology for in vitro tissue engineering. Biomaterials 2022, 280, 121267. [Google Scholar] [CrossRef]
- Cunha, A.G.; Gandini, A. Turning polysaccharides into hydrophobic materials: A critical review. Part 2. Hemicelluloses, chitin/chitosan, starch, pectin and alginates. Cellulose 2010, 17, 1045–1065. [Google Scholar] [CrossRef]
- Fallahi, A.; Yazdi, I.K.; Serex, L.; Lesha, E.; Faramarzi, N.; Tarlan, F.; Avci, H.; Costa-Almeida, R.; Sharifi, F.; Rinoldi, C.; et al. Customizable composite fibers for engineering skeletal muscle models. ACS Biomater. Sci. Eng. 2020, 6, 1112–1123. [Google Scholar] [CrossRef]
- Powell, C.A.; Smiley, B.L.; Mills, J.; Vandenburgh, H.H. Mechanical stimulation improves tissue-engineered human skeletal muscle. Am. J. Physiol. Cell Physiol. 2002, 283, C1557–C1565. [Google Scholar] [CrossRef]
- Ahmed, T.A.E.; Dare, E.V.; Hincke, M. Fibrin: A versatile scaffold for tissue engineering applications. Tissue Eng. Part B Rev. 2008, 14, 199–215. [Google Scholar] [CrossRef]
- Gholobova, D.; Decroix, L.; Van Muylder, V.; Desender, L.; Gerard, M.; Carpentier, G.; Vandenburgh, H.; Thorrez, L. Endothelial Network Formation Within Human Tissue-Engineered Skeletal Muscle. Tissue Eng. Part A 2015, 21, 2548–2558. [Google Scholar] [CrossRef] [PubMed]
- Noori, A.; Ashrafi, S.J.; Vaez-Ghaemi, R.; Hatamian-Zaremi, A.; Webster, T.J. A review of fibrin and fibrin composites for bone tissue engineering. Int. J. Nanomed. 2017, 12, 4937–4961. [Google Scholar] [CrossRef]
- Sze, J.H.; Brownlie, J.C.; Love, C.A. Biotechnological production of hyaluronic acid: A mini review. 3 Biotech 2016, 6, 67. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, P.; Kundu, B.; Naskar, D.; Kim, H.-W.; Maiti, T.K.; Bhattacharya, D.; Kundu, S.C. Silk scaffolds in bone tissue engineering: An overview. Acta Biomater. 2017, 63, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-W.; Chen, Y.-K.; Tang, K.-C.; Yang, K.-C.; Cheng, N.-C.; Yu, J. Keratin scaffolds with human adipose stem cells: Physical and biological effects toward wound healing. J. Tissue Eng. Regen. Med. 2019, 13, 1044–1058. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, S.; Larsen, L.B.; Trifol, J.; Szabo, P.; Burri, H.V.R.; Canali, C.; Dufva, M.; Emnéus, J.; Wolff, A. Fabrication of scalable and structured tissue engineering scaffolds using water dissolvable sacrificial 3D printed moulds. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 55, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Qazi, T.H.; Mooney, D.J.; Pumberger, M.; Geissler, S.; Duda, G.N. Biomaterials based strategies for skeletal muscle tissue engineering: Existing technologies and future trends. Biomaterials 2015, 53, 502–521. [Google Scholar] [CrossRef] [PubMed]
- Amrita; Arora, A.; Sharma, P.; Katti, D.S. Pullulan-based composite scaffolds for bone tissue engineering: Improved osteoconductivity by pore wall mineralization. Carbohydr. Polym. 2015, 123, 180–189. [Google Scholar] [CrossRef]
- Huang, J.; Huang, K.; You, X.; Liu, G.; Hollett, G.; Kang, Y.; Gu, Z.; Wu, J. Evaluation of tofu as a potential tissue engineering scaffold. J. Mater. Chem. B 2018, 6, 1328–1334. [Google Scholar] [CrossRef]
- Reddy, N.; Yang, Y. Potential of plant proteins for medical applications. Trends Biotechnol. 2011, 29, 490–498. [Google Scholar] [CrossRef]
- Jahangirian, H.; Azizi, S.; Rafiee-Moghaddam, R.; Baratvand, B.; Webster, T.J. Status of Plant Protein-Based Green Scaffolds for Regenerative Medicine Applications. Biomolecules 2019, 9, 619. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Reddy, N.; Zhang, S.; Roscioli, N.; Yang, Y. Water-stable electrospun collagen fibers from a non-toxic solvent and crosslinking system. J. Biomed. Mater. Res. A 2013, 101, 1237–1247. [Google Scholar] [CrossRef]
- Qu, Z.-H.; Wang, H.-J.; Tang, T.-T.; Zhang, X.-L.; Wang, J.-Y.; Dai, K.-R. Evaluation of the zein/inorganics composite on biocompatibility and osteoblastic differentiation. Acta Biomater. 2008, 4, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, K.; Renneckar, S.; Gatenholm, P. Biomimetic calcium phosphate crystal mineralization on electrospun cellulose-based scaffolds. ACS Appl. Mater. Interfaces 2011, 3, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Courtenay, J.C.; Johns, M.A.; Galembeck, F.; Deneke, C.; Lanzoni, E.M.; Costa, C.A.; Scott, J.L.; Sharma, R.I. Surface modified cellulose scaffolds for tissue engineering. Cellulose 2017, 24, 253–267. [Google Scholar] [CrossRef]
- Ninan, N.; Muthiah, M.; Park, I.-K.; Elain, A.; Thomas, S.; Grohens, Y. Pectin/carboxymethyl cellulose/microfibrillated cellulose composite scaffolds for tissue engineering. Carbohydr. Polym. 2013, 98, 877–885. [Google Scholar] [CrossRef]
- Apsite, I.; Uribe, J.M.; Posada, A.F.; Rosenfeldt, S.; Salehi, S.; Ionov, L. 4D biofabrication of skeletal muscle microtissues. Biofabrication 2019, 12, 015016. [Google Scholar] [CrossRef]
- Antinori, M.E.; Contardi, M.; Suarato, G.; Armirotti, A.; Bertorelli, R.; Mancini, G.; Debellis, D.; Athanassiou, A. Advanced mycelium materials as potential self-growing biomedical scaffolds. Sci. Rep. 2021, 11, 12630. [Google Scholar] [CrossRef] [PubMed]
- Bar-Shai, N.; Sharabani-Yosef, O.; Zollmann, M.; Lesman, A.; Golberg, A. Seaweed cellulose scaffolds derived from green macroalgae for tissue engineering. Sci. Rep. 2021, 11, 11843. [Google Scholar] [CrossRef]
- Gershlak, J.R.; Hernandez, S.; Fontana, G.; Perreault, L.R.; Hansen, K.J.; Larson, S.A.; Binder, B.Y.K.; Dolivo, D.M.; Yang, T.; Dominko, T.; et al. Crossing kingdoms: Using decellularized plants as perfusable tissue engineering scaffolds. Biomaterials 2017, 125, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Modulevsky, D.J.; Cuerrier, C.M.; Pelling, A.E. Biocompatibility of Subcutaneously Implanted Plant-Derived Cellulose Biomaterials. PLoS ONE 2016, 11, e0157894. [Google Scholar] [CrossRef]
- Reddy, M.S.B.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A comparative review of natural and synthetic biopolymer composite scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef] [PubMed]
- Langelaan, M.L.P.; Boonen, K.J.M.; Polak, R.B.; Baaijens, F.P.T.; Post, M.J.; van der Schaft, D.W.J. Meet the new meat: Tissue engineered skeletal muscle. Trends Food Sci. Technol. 2010, 21, 59–66. [Google Scholar] [CrossRef]
- Prüller, J.; Mannhardt, I.; Eschenhagen, T.; Zammit, P.S.; Figeac, N. Satellite cells delivered in their niche efficiently generate functional myotubes in three-dimensional cell culture. PLoS ONE 2018, 13, e0202574. [Google Scholar] [CrossRef] [PubMed]
- Bettahalli, N.M.S.; Steg, H.; Wessling, M.; Stamatialis, D. Development of poly(l-lactic acid) hollow fiber membranes for artificial vasculature in tissue engineering scaffolds. J. Memb. Sci. 2011, 371, 117–126. [Google Scholar] [CrossRef]
- Shafa, M.; Sjonnesen, K.; Yamashita, A.; Liu, S.; Michalak, M.; Kallos, M.S.; Rancourt, D.E. Expansion and long-term maintenance of induced pluripotent stem cells in stirred suspension bioreactors. J. Tissue Eng. Regen. Med. 2012, 6, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Silva Couto, P.; Rotondi, M.C.; Bersenev, A.; Hewitt, C.J.; Nienow, A.W.; Verter, F.; Rafiq, Q.A. Expansion of human mesenchymal stem/stromal cells (hMSCs) in bioreactors using microcarriers: Lessons learnt and what the future holds. Biotechnol. Adv. 2020, 45, 107636. [Google Scholar] [CrossRef] [PubMed]
- Badenes, S.M.; Fernandes, T.G.; Rodrigues, C.A.V.; Diogo, M.M.; Cabral, J.M.S. Microcarrier-based platforms for in vitro expansion and differentiation of human pluripotent stem cells in bioreactor culture systems. J. Biotechnol. 2016, 234, 71–82. [Google Scholar] [CrossRef]
- Pasini, A.; Lovecchio, J.; Cortesi, M.; Liverani, C.; Spadazzi, C.; Mercatali, L.; Ibrahim, T.; Giordano, E. Perfusion Flow Enhances Viability and Migratory Phenotype in 3D-Cultured Breast Cancer Cells. Ann. Biomed. Eng. 2021, 49, 2103–2113. [Google Scholar] [CrossRef]
- Verbruggen, S.; Luining, D.; van Essen, A.; Post, M.J. Bovine myoblast cell production in a microcarriers-based system. Cytotechnology 2018, 70, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.; Mehan, L.; Kumar, R.; Kumar, A.; Srivastava, A. Computational fluid dynamic analysis of hydrodynamic shear stress generated by different impeller combinations in stirred bioreactor. Biochem. Eng. J. 2019, 151, 107312. [Google Scholar] [CrossRef]
- Specht, L. An Analysis of Culture Medium Costs and Production Volumes for Cultivated Meat; The Good Food Institute: Washington, DC, USA, 2020. [Google Scholar]
- Mandenius, C.-F.; Brundin, A. Bioprocess optimization using design-of-experiments methodology. Biotechnol. Prog. 2008, 24, 1191–1203. [Google Scholar] [CrossRef] [PubMed]
- Allan, S.J.; De Bank, P.A.; Ellis, M.J. Bioprocess design considerations for cultured meat production with a focus on the expansion bioreactor. Front. Sustain. Food Syst. 2019, 3, 44. [Google Scholar] [CrossRef]
- Bellani, C.F.; Ajeian, J.; Duffy, L.; Miotto, M.; Groenewegen, L.; Connon, C.J. Scale-Up Technologies for the Manufacture of Adherent Cells. Front. Nutr. 2020, 7, 575146. [Google Scholar] [CrossRef] [PubMed]
- Bardouille, C.; Lehmann, J.; Heimann, P.; Jockusch, H. Growth and differentiation of permanent and secondary mouse myogenic cell lines on microcarriers. Appl. Microbiol. Biotechnol. 2001, 55, 556–562. [Google Scholar] [CrossRef]
- Confalonieri, D.; La Marca, M.; van Dongen, E.M.W.M.; Walles, H.; Ehlicke, F. * An Injectable Recombinant Collagen I Peptide-Based Macroporous Microcarrier Allows Superior Expansion of C2C12 and Human Bone Marrow-Derived Mesenchymal Stromal Cells and Supports Deposition of Mineralized Matrix. Tissue Eng. Part A 2017, 23, 946–957. [Google Scholar] [CrossRef] [PubMed]
- Rozwadowska, N.; Malcher, A.; Baumann, E.; Kolanowski, T.J.; Rucinski, M.; Mietkiewski, T.; Fiedorowicz, K.; Kurpisz, M. In vitro culture of primary human myoblasts by using the dextran microcarriers Cytodex3®. Folia Histochem. Cytobiol. 2016, 54, 81–90. [Google Scholar] [CrossRef]
- Rafiq, Q.A.; Ruck, S.; Hanga, M.P.; Heathman, T.R.J.; Coopman, K.; Nienow, A.W.; Williams, D.J.; Hewitt, C.J. Qualitative and quantitative demonstration of bead-to-bead transfer with bone marrow-derived human mesenchymal stem cells on microcarriers: Utilising the phenomenon to improve culture performance. Biochem. Eng. J. 2018, 135, 11–21. [Google Scholar] [CrossRef]
- Leber, J.; Barekzai, J.; Blumenstock, M.; Pospisil, B.; Salzig, D.; Czermak, P. Microcarrier choice and bead-to-bead transfer for human mesenchymal stem cells in serum-containing and chemically defined media. Process Biochem. 2017, 59, 255–265. [Google Scholar] [CrossRef]
- Odeleye, A.O.O.; Baudequin, T.; Chui, C.-Y.; Cui, Z.; Ye, H. An additive manufacturing approach to bioreactor design for mesenchymal stem cell culture. Biochem. Eng. J. 2020, 156, 107515. [Google Scholar] [CrossRef]
- Lam, A.T.-L.; Li, J.; Toh, J.P.-W.; Sim, E.J.-H.; Chen, A.K.-L.; Chan, J.K.-Y.; Choolani, M.; Reuveny, S.; Birch, W.R.; Oh, S.K.-W. Biodegradable poly-ε-caprolactone microcarriers for efficient production of human mesenchymal stromal cells and secreted cytokines in batch and fed-batch bioreactors. Cytotherapy 2017, 19, 419–432. [Google Scholar] [CrossRef]
- Letti, L.A.J.; Karp, S.G.; Molento, C.F.M.; Colonia, B.S.O.; Boschero, R.A.; Soccol, V.T.; Herrmann, L.W.; Penha, R. de O.; Woiciechowski, A.L.; Soccol, C.R. Cultivated meat: Recent technological developments, current market and future challenges. Biotechnol. Res. Innov. 2021, 5, e2021001. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, X.; Li, X.; Du, G.; Zhou, J.; Chen, J. Challenges and possibilities for bio-manufacturing cultured meat. Trends Food Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Kumar, A.; Starly, B. Large scale industrialized cell expansion: Producing the critical raw material for biofabrication processes. Biofabrication 2015, 7, 044103. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-K.; Chen, C.S. Cell adhesion and mechanical stimulation in the regulation of mesenchymal stem cell differentiation. J. Cell. Mol. Med. 2013, 17, 823–832. [Google Scholar] [CrossRef]
- Rafiq, Q.A.; Hanga, M.P.; Heathman, T.R.J.; Coopman, K.; Nienow, A.W.; Williams, D.J.; Hewitt, C.J. Process development of human multipotent stromal cell microcarrier culture using an automated high-throughput microbioreactor. Biotechnol. Bioeng. 2017, 114, 2253–2266. [Google Scholar] [CrossRef]
- Jossen, V.; Schirmer, C.; Mostafa Sindi, D.; Eibl, R.; Kraume, M.; Pörtner, R.; Eibl, D. Theoretical and Practical Issues That Are Relevant When Scaling Up hMSC Microcarrier Production Processes. Stem Cells Int. 2016, 2016, 4760414. [Google Scholar] [CrossRef] [PubMed]
- Berry, J.D.; Liovic, P.; Šutalo, I.D.; Stewart, R.L.; Glattauer, V.; Meagher, L. Characterisation of stresses on microcarriers in a stirred bioreactor. Appl. Math. Model. 2016, 40, 6787–6804. [Google Scholar] [CrossRef]
- Ferrari, C.; Balandras, F.; Guedon, E.; Olmos, E.; Chevalot, I.; Marc, A. Limiting cell aggregation during mesenchymal stem cell expansion on microcarriers. Biotechnol. Prog. 2012, 28, 780–787. [Google Scholar] [CrossRef] [PubMed]
- Stolberg, S.; McCloskey, K.E. Can shear stress direct stem cell fate? Biotechnol. Prog. 2009, 25, 10–19. [Google Scholar] [CrossRef]
- Breese, T.W.; Admassu, W. Feasibility of culturing C2C12 mouse myoblasts on glass microcarriers in a continuous stirred tank bioreactor. Bioprocess Eng. 1999, 20, 463. [Google Scholar] [CrossRef]
- Rudolph, G.; Lindner, P.; Gierse, A.; Bluma, A.; Martinez, G.; Hitzmann, B.; Scheper, T. Online monitoring of microcarrier based fibroblast cultivations with in situ microscopy. Biotechnol. Bioeng. 2008, 99, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Eibl, R.; Kaiser, S.; Lombriser, R.; Eibl, D. Disposable bioreactors: The current state-of-the-art and recommended applications in biotechnology. Appl. Microbiol. Biotechnol. 2010, 86, 41–49. [Google Scholar] [CrossRef]
- Hähnel, A.; Pütz, B.; Iding, K.; Niediek, T.; Gudermann, F.; Lütkemeyer, D. Evaluation of a disposable stirred tank bioreactor for cultivation of mammalian cells. BMC Proc. 2011, 5 (Suppl. S8), P54. [Google Scholar] [CrossRef]
- Nogueira, D.E.S.; Cabral, J.M.S.; Rodrigues, C.A.V. Single-Use Bioreactors for Human Pluripotent and Adult Stem Cells: Towards Regenerative Medicine Applications. Bioengineering 2021, 8, 68. [Google Scholar] [CrossRef]
- Hanga, M.P.; de la Raga, F.A.; Moutsatsou, P.; Hewitt, C.J.; Nienow, A.W.; Wall, I. Scale-up of an intensified bioprocess for the expansion of bovine adipose-derived stem cells (bASCs) in stirred tank bioreactors. Biotechnol. Bioeng. 2021, 118, 3175–3186. [Google Scholar] [CrossRef]
- Singh, V. Disposable bioreactor for cell culture using wave-induced agitation. Cytotechnology 1999, 30, 149–158. [Google Scholar] [CrossRef]
- Jyothilekshmi, I.; Jayaprakash, N.S. Trends in monoclonal antibody production using various bioreactor syst. J. Microbiol. Biotechnol. 2021, 31, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Eibl, R.; Werner, S.; Eibl, D. Bag bioreactor based on wave-induced motion: Characteristics and applications. Adv. Biochem. Eng. Biotechnol. 2009, 115, 55–87. [Google Scholar] [CrossRef] [PubMed]
- Chotteau, V.; Zhang, Y.; Clincke, M.-F. Very High Cell Density in Perfusion of CHO Cells by ATF, TFF, Wave Bioreactor, and/or CellTank Technologies—Impact of Cell Density and Applications. In Continuous Processing in Pharmaceutical Manufacturing; Subramanian, G., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2014; pp. 339–356. ISBN 9783527335954. [Google Scholar]
- Clincke, M.-F.; Mölleryd, C.; Samani, P.K.; Lindskog, E.; Fäldt, E.; Walsh, K.; Chotteau, V. Very high density of Chinese hamster ovary cells in perfusion by alternating tangential flow or tangential flow filtration in WAVE BioreactorTM-part II: Applications for antibody production and cryopreservation. Biotechnol. Prog. 2013, 29, 768–777. [Google Scholar] [CrossRef] [PubMed]
- Mendonça da Silva, J.; Erro, E.; Awan, M.; Chalmers, S.-A.; Fuller, B.; Selden, C. Small-Scale Fluidized Bed Bioreactor for Long-Term Dynamic Culture of 3D Cell Constructs and in vitro Testing. Front. Bioeng. Biotechnol. 2020, 8, 895. [Google Scholar] [CrossRef]
- Ellis, M.; Jarman-Smith, M.; Chaudhuri, J.B. Bioreactor Systems for Tissue Engineering: A Four-Dimensional Challenge. In Bioreactors for Tissue Engineering; Chaudhuri, J., Al-Rubeai, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 1–18. ISBN 1-4020-3740-6. [Google Scholar]
- Van Wie, B.J.; Brouns, T.M.; Elliot, M.L.; Davis, W.C. A novel continuous centrifugal bioreactor for high-density cultivation of mammalian and microbial cells. Biotechnol. Bioeng. 1991, 38, 1190–1202. [Google Scholar] [CrossRef]
- Detzel, C.J.; Thorson, M.R.; Van Wie, B.J.; Ivory, C.F. A study of the Coriolis effect on the fluid flow profile in a centrifugal bioreactor. Biotechnol. Prog. 2009, 25, 1025–1034. [Google Scholar] [CrossRef] [PubMed]
- Detzel, C.J.; Van Wie, B.J.; Ivory, C.F. Fluid flow through a high cell density fluidized-bed during centrifugal bioreactor culture. Biotechnol. Prog. 2010, 26, 1014–1023. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, G.; Zhao, X.; Zhou, J.; Du, G.; Chen, J. A conceptual air-lift reactor design for large scale animal cell cultivation in the context of in vitro meat production. Chem. Eng. Sci. 2020, 211, 115269. [Google Scholar] [CrossRef]
- Meuwly, F.; von Stockar, U.; Kadouri, A. Optimization of the medium perfusion rate in a packed-bed bioreactor charged with CHO cells. Cytotechnology 2004, 46, 37–47. [Google Scholar] [CrossRef]
- Walther, J.; Godawat, R.; Hwang, C.; Abe, Y.; Sinclair, A.; Konstantinov, K. The business impact of an integrated continuous biomanufacturing platform for recombinant protein production. J. Biotechnol. 2015, 213, 3–12. [Google Scholar] [CrossRef]
- van der Weele, C.; Tramper, J. Cultured meat: Every village its own factory? Trends Biotechnol. 2014, 32, 294–296. [Google Scholar] [CrossRef] [PubMed]
- Karst, D.J.; Steinebach, F.; Morbidelli, M. Continuous integrated manufacturing of therapeutic proteins. Curr. Opin. Biotechnol. 2018, 53, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Fisher, A.C.; Kamga, M.-H.; Agarabi, C.; Brorson, K.; Lee, S.L.; Yoon, S. The current scientific and regulatory landscape in advancing integrated continuous biopharmaceutical manufacturing. Trends Biotechnol. 2019, 37, 253–267. [Google Scholar] [CrossRef]
- Xu, S.; Gavin, J.; Jiang, R.; Chen, H. Bioreactor productivity and media cost comparison for different intensified cell culture processes. Biotechnol. Prog. 2017, 33, 867–878. [Google Scholar] [CrossRef] [PubMed]
- Yongky, A.; Xu, J.; Tian, J.; Oliveira, C.; Zhao, J.; McFarland, K.; Borys, M.C.; Li, Z.J. Process intensification in fed-batch production bioreactors using non-perfusion seed cultures. MAbs 2019, 11, 1502–1514. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.C.; Minkler, D.F.; Kshirsagar, R.; Ryll, T.; Huang, Y.-M. Concentrated fed-batch cell culture increases manufacturing capacity without additional volumetric capacity. J. Biotechnol. 2016, 217, 1–11. [Google Scholar] [CrossRef]
- Pasini, A.; Lovecchio, J.; Ferretti, G.; Giordano, E. Medium perfusion flow improves osteogenic commitment of human stromal cells. Stem Cells Int. 2019, 2019, 1304194. [Google Scholar] [CrossRef] [PubMed]
- Konstantinov, K.; Goudar, C.; Ng, M.; Meneses, R.; Thrift, J.; Chuppa, S.; Matanguihan, C.; Michaels, J.; Naveh, D. The “Push-to-Low” Approach for Optimization of High-Density Perfusion Cultures of Animal Cells. In Cell Culture Engineering; Hu, W.-S., Ed.; Advances in Biochemical Engineering/Biotechnology; Springer: Berlin/Heidelberg, Germany, 2006; Volume 101, pp. 75–98. ISBN 978-3-540-34006-5. [Google Scholar]
- Voisard, D.; Meuwly, F.; Ruffieux, P.A.; Baer, G.; Kadouri, A. Potential of cell retention techniques for large-scale high-density perfusion culture of suspended mammalian cells. Biotechnol. Bioeng. 2003, 82, 751–765. [Google Scholar] [CrossRef] [PubMed]
- Stettler, M.; Monschois, V.; Jordan, V. Intensification of large-scale mammalian fed-batch processes. Pharm. Bioprocess 2018, 6, 1–2. [Google Scholar]
- Pohlscheidt, M.; Jacobs, M.; Wolf, S.; Thiele, J.; Jockwer, A.; Gabelsberger, J.; Jenzsch, M.; Tebbe, H.; Burg, J. Optimizing capacity utilization by large scale 3000 L perfusion in seed train bioreactors. Biotechnol. Prog. 2013, 29, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Jordan, M.; Mac Kinnon, N.; Monchois, V.; Stettler, M.; Broly, H. Intensification of large-scale cell culture processes. Curr. Opin. Chem. Eng. 2018, 22, 253–257. [Google Scholar] [CrossRef]
- Portner, R.; Platas, O.B.; Fassnacht, D.; Nehring, D.; Czermak, P.; Markl, H. Fixed Bed Reactors for the Cultivation of Mammalian Cells: Design, Performance and Scale-Up. Open Biotechnol. J. 2007, 1, 41–46. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Ito, A.; Jitsunobu, H.; Yamaguchi, K.; Kawabe, Y.; Mizumoto, H.; Kamihira, M. Hollow Fiber Bioreactor Perfusion Culture System for Magnetic Force-Based Skeletal Muscle Tissue Engineering. J. Chem. Eng. Japan 2012, 45, 348–354. [Google Scholar] [CrossRef]
- Delaine-Smith, R.M.; MacNeil, S.; Reilly, G.C. Matrix production and collagen structure are enhanced in two types of osteogenic progenitor cells by a simple fluid shear stress stimulus. Eur. Cell. Mater. 2012, 24, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, B.; Owen, R.; Bahmaee, H.; Rao, P.S.; Reilly, G.C. Design and assessment of a dynamic perfusion bioreactor for large bone tissue engineering scaffolds. Appl. Biochem. Biotechnol. 2018, 185, 555–563. [Google Scholar] [CrossRef]
- Yourek, G.; McCormick, S.M.; Mao, J.J.; Reilly, G.C. Shear stress induces osteogenic differentiation of human mesenchymal stem cells. Regen. Med. 2010, 5, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Della Porta, G.; Nguyen, B.-N.B.; Campardelli, R.; Reverchon, E.; Fisher, J.P. Synergistic effect of sustained release of growth factors and dynamic culture on osteoblastic differentiation of mesenchymal stem cells. J. Biomed. Mater. Res. A 2015, 103, 2161–2171. [Google Scholar] [CrossRef]
- Lovecchio, J.; Gargiulo, P.; Vargas Luna, J.L.; Giordano, E.; Sigurjónsson, Ó.E. A standalone bioreactor system to deliver compressive load under perfusion flow to hBMSC-seeded 3D chitosan-graphene templates. Sci. Rep. 2019, 9, 16854. [Google Scholar] [CrossRef]
- Yang, P.; Teo, W.-K.; Ting, Y.-P. Design and performance study of a novel immobilized hollow fiber membrane bioreactor. Bioresour. Technol. 2006, 97, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Meuwly, F.; Ruffieux, P.A.; Kadouri, A.; von Stockar, U. Packed-bed bioreactors for mammalian cell culture: Bioprocess and biomedical applications. Biotechnol. Adv. 2007, 25, 45–56. [Google Scholar] [CrossRef]
- Rafiq, Q.A.; Heathman, T.R.J.; Coopman, K.; Nienow, A.W.; Hewitt, C.J. Scalable manufacture for cell therapy needs. In Bioreactors: Design, Operation and Novel Applications; Mandenius, C.-F., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2016; pp. 113–146. ISBN 9783527337682. [Google Scholar]
- Gallo-Ramírez, L.E.; Nikolay, A.; Genzel, Y.; Reichl, U. Bioreactor concepts for cell culture-based viral vaccine production. Expert Rev. Vaccines 2015, 14, 1181–1195. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.; Kurisawa, M. Integrating biomaterials and food biopolymers for cultured meat production. Acta Biomater. 2021, 124, 108–129. [Google Scholar] [CrossRef]
- Preissmann, A.; Wiesmann, R.; Buchholz, R.; Werner, R.G.; Noé, W. Investigations on oxygen limitations of adherent cells growing on macroporous microcarriers. Cytotechnology 1997, 24, 121–134. [Google Scholar] [CrossRef]
- Osiecki, M.J.; Michl, T.D.; Kul Babur, B.; Kabiri, M.; Atkinson, K.; Lott, W.B.; Griesser, H.J.; Doran, M.R. Packed Bed Bioreactor for the Isolation and Expansion of Placental-Derived Mesenchymal Stromal Cells. PLoS ONE 2015, 10, e0144941. [Google Scholar] [CrossRef]
- Baba, K.; Sankai, Y. Development of biomimetic system for scale up of cell spheroids—Building blocks for cell transplantation. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2017, 2017, 1611–1616. [Google Scholar] [CrossRef]
- Knazek, R.A.; Gullino, P.M.; Kohler, P.O.; Dedrick, R.L. Cell culture on artificial capillaries: An approach to tissue growth in vitro. Science 1972, 178, 65–66. [Google Scholar] [CrossRef] [PubMed]
- Martin, Y.; Vermette, P. Bioreactors for tissue mass culture: Design, characterization, and recent advances. Biomaterials 2005, 26, 7481–7503. [Google Scholar] [CrossRef] [PubMed]
- Roberts, I.; Baila, S.; Rice, R.B.; Janssens, M.E.; Nguyen, K.; Moens, N.; Ruban, L.; Hernandez, D.; Coffey, P.; Mason, C. Scale-up of human embryonic stem cell culture using a hollow fibre bioreactor. Biotechnol. Lett. 2012, 34, 2307–2315. [Google Scholar] [CrossRef]
- Eghbali, H.; Nava, M.M.; Mohebbi-Kalhori, D.; Raimondi, M.T. Hollow fiber bioreactor technology for tissue engineering applications. Int. J. Artif. Organs 2016, 39, 1–15. [Google Scholar] [CrossRef]
- Wang, S.B.; Godfrey, S.; Radoniqi, F.; Lin, H.; Coffman, J. Larger Pore Size Hollow Fiber Membranes as a Solution to the Product Retention Issue in Filtration-Based Perfusion Bioreactors. Biotechnol. J. 2019, 14, e1800137. [Google Scholar] [CrossRef]
- Neubauer, P.; Junne, S. Scale-up and scale-down methodologies for bioreactors. In Bioreactors: Design, Operation and Novel Applications; Mandenius, C.-F., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2016; pp. 323–354. ISBN 9783527337682. [Google Scholar]
- Rafiq, Q.A.; Coopman, K.; Hewitt, C.J. Scale-up of human mesenchymal stem cell culture: Current technologies and future challenges. Curr. Opin. Chem. Eng. 2013, 2, 8–16. [Google Scholar] [CrossRef]
- Cunha, B.; Aguiar, T.; Carvalho, S.B.; Silva, M.M.; Gomes, R.A.; Carrondo, M.J.T.; Gomes-Alves, P.; Peixoto, C.; Serra, M.; Alves, P.M. Bioprocess integration for human mesenchymal stem cells: From up to downstream processing scale-up to cell proteome characterization. J. Biotechnol. 2017, 248, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Schirmaier, C.; Jossen, V.; Kaiser, S.C.; Jüngerkes, F.; Brill, S.; Safavi-Nab, A.; Siehoff, A.; van den Bos, C.; Eibl, D.; Eibl, R. Scale-up of adipose tissue-derived mesenchymal stem cell production in stirred single-use bioreactors under low-serum conditions. Eng. Life Sci. 2014, 14, 292–303. [Google Scholar] [CrossRef]
- Hewitt, C.J.; Nienow, A.W. The scale-up of microbial batch and fed-batch fermentation processes. In Advances in Applied Microbiology; Advances in Applied Microbiology; Elsevier: Amsterdam, The Netherlands, 2007; Volume 62, pp. 105–135. ISBN 9780123736697. [Google Scholar]
- Nienow, A.W.; Coopman, K.; Heathman, T.R.J.; Rafiq, Q.A.; Hewitt, C.J. Bioreactor engineering fundamentals for stem cell manufacturing. In Stem Cell Manufacturing; Elsevier: Amsterdam, The Netherlands, 2016; pp. 43–75. ISBN 9780444632654. [Google Scholar]
- Kang, D.-H.; Louis, F.; Liu, H.; Shimoda, H.; Nishiyama, Y.; Nozawa, H.; Kakitani, M.; Takagi, D.; Kasa, D.; Nagamori, E.; et al. Engineered whole cut meat-like tissue by the assembly of cell fibers using tendon-gel integrated bioprinting. Nat. Commun. 2021, 12, 5059. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Chen, H. High-density mammalian cell cultures in stirred-tank bioreactor without external pH control. J. Biotechnol. 2016, 231, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Nienow, A.W. Reactor engineering in large scale animal cell culture. Cytotechnology 2006, 50, 9–33. [Google Scholar] [CrossRef]
- Sieblist, C.; Hägeholz, O.; Aehle, M.; Jenzsch, M.; Pohlscheidt, M.; Lübbert, A. Insights into large-scale cell-culture reactors: II. Gas-phase mixing and CO₂ stripping. Biotechnol. J. 2011, 6, 1547–1556. [Google Scholar] [CrossRef]
- Konstantinov, K.B.; Tsai, Y.; Moles, D.; Matanguihan, R. Control of long-term perfusion Chinese hamster ovary cell culture by glucose auxostat. Biotechnol. Prog. 1996, 12, 100–109. [Google Scholar] [CrossRef]
- Lawson, T.; Kehoe, D.E.; Schnitzler, A.C.; Rapiejko, P.J.; Der, K.A.; Philbrick, K.; Punreddy, S.; Rigby, S.; Smith, R.; Feng, Q.; et al. Process development for expansion of human mesenchymal stromal cells in a 50L single-use stirred tank bioreactor. Biochem. Eng. J. 2017, 120, 49–62. [Google Scholar] [CrossRef]
- Yazdani, M. Technical aspects of oxygen level regulation in primary cell cultures: A review. Interdiscip. Toxicol. 2016, 9, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Chen, D.; Yu, J.; Liu, J.; Shi, X.; Sun, Y.; Pan, Q.; Luo, X.; Yang, J.; Li, Y.; et al. Impact of Oxygen Concentration on Metabolic Profile of Human Placenta-Derived Mesenchymal Stem Cells as Determined by Chemical Isotope Labeling LC-MS. J. Proteome Res. 2018, 17, 1866–1878. [Google Scholar] [CrossRef]
- Ivanovic, Z. Hypoxia or in situ normoxia: The stem cell paradigm. J. Cell. Physiol. 2009, 219, 271–275. [Google Scholar] [CrossRef]
- Ma, T.; Grayson, W.L.; Fröhlich, M.; Vunjak-Novakovic, G. Hypoxia and stem cell-based engineering of mesenchymal tissues. Biotechnol. Prog. 2009, 25, 32–42. [Google Scholar] [CrossRef]
- Heathman, T.R.J.; Nienow, A.W.; Rafiq, Q.A.; Coopman, K.; Kara, B.; Hewitt, C.J. Agitation and aeration of stirred-bioreactors for the microcarrier culture of human mesenchymal stem cells and potential implications for large-scale bioprocess development. Biochem. Eng. J. 2018, 136, 9–17. [Google Scholar] [CrossRef]
- Nienow, A.W. Mass transfer and mixing across the scales in animal cell culture. In Animal Cell Culture; Al-Rubeai, M., Ed.; Cell Engineering; Springer International Publishing: Cham, Switzerland, 2015; Volume 9, pp. 137–167. ISBN 978-3-319-10319-8. [Google Scholar]
- Bauer, P.; Hancock, L.; Rathman, J.; Chalmers, J.J. Cell-microcarrier adhesion to gas-liquid interfaces and foam. Biotechnol. Prog. 2000, 16, 125–132. [Google Scholar] [CrossRef]
- Fernandes-Platzgummer, A.; Diogo, M.M.; Baptista, R.P.; da Silva, C.L.; Cabral, J.M.S. Scale-up of mouse embryonic stem cell expansion in stirred bioreactors. Biotechnol. Prog. 2011, 27, 1421–1432. [Google Scholar] [CrossRef]
- Hu, W.; Berdugo, C.; Chalmers, J.J. The potential of hydrodynamic damage to animal cells of industrial relevance: Current understanding. Cytotechnology 2011, 63, 445–460. [Google Scholar] [CrossRef] [PubMed]
- Nienow, A.W. Aeration in Biotechnology. In Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons Inc, Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2000; pp. 1–23. ISBN 9780471238966. [Google Scholar]
- Ibrahim, S.; Nienow, A. Power curves and flow patterns for a range of impellers in newtonian fluids: 40 <Re <5 × 10(5). Chem. Eng. Res. Design 1995, 73, 485–491. [Google Scholar]
- Rameez, S.; Mostafa, S.S.; Miller, C.; Shukla, A.A. High-throughput miniaturized bioreactors for cell culture process development: Reproducibility, scalability, and control. Biotechnol. Prog. 2014, 30, 718–727. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.S.; El-Abd, Y.; Bui, K.; Hyun, Y.-E.; Hughes, R.H.; Kraus, W.E.; Truskey, G.A. Conditions that promote primary human skeletal myoblast culture and muscle differentiation in vitro. Am. J. Physiol. Cell Physiol. 2014, 306, C385–C395. [Google Scholar] [CrossRef]
- Shahini, A.; Vydiam, K.; Choudhury, D.; Rajabian, N.; Nguyen, T.; Lei, P.; Andreadis, S.T. Efficient and high yield isolation of myoblasts from skeletal muscle. Stem Cell Res. 2018, 30, 122–129. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, E.N.; Cosenza, Z.A.; Baar, K.; Block, D.E. Considerations for the development of cost-effective cell culture media for cultivated meat production. Comp. Rev. Food Sci. Food Safety 2021, 20, 686–709. [Google Scholar] [CrossRef] [PubMed]
- Agathocleous, M.; Harris, W.A. Metabolism in physiological cell proliferation and differentiation. Trends Cell Biol. 2013, 23, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Rumsey, J.W.; Bhargava, N.; Gregory, C.; Riedel, L.; Kang, J.F.; Hickman, J.J. Developing a novel serum-free cell culture model of skeletal muscle differentiation by systematically studying the role of different growth factors in myotube formation. In Vitro Cell Dev. Biol. Anim. 2009, 45, 378–387. [Google Scholar] [CrossRef]
- Mimura, S.; Kimura, N.; Hirata, M.; Tateyama, D.; Hayashida, M.; Umezawa, A.; Kohara, A.; Nikawa, H.; Okamoto, T.; Furue, M.K. Growth factor-defined culture medium for human mesenchymal stem cells. Int. J. Dev. Biol. 2011, 55, 181–187. [Google Scholar] [CrossRef]
- Chatterjee, I.; Li, F.; Kohler, E.E.; Rehman, J.; Malik, A.B.; Wary, K.K. Induced Pluripotent Stem (iPS) Cell Culture Methods and Induction of Differentiation into Endothelial Cells. Methods Mol. Biol. 2016, 1357, 311–327. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-Y.; Loh, H.-X.; Wan, A.C.A. Systems for muscle cell differentiation: From bioengineering to future food. Micromachines 2021, 13, 71. [Google Scholar] [CrossRef]
- Goonoo, N.; Bhaw-Luximon, A. Mimicking growth factors: Role of small molecule scaffold additives in promoting tissue regeneration and repair. RSC Adv. 2019, 9, 18124–18146. [Google Scholar] [CrossRef]
- Danoviz, M.E.; Yablonka-Reuveni, Z. Skeletal muscle satellite cells: Background and methods for isolation and analysis in a primary culture system. Methods Mol. Biol. 2012, 798, 21–52. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.E.; Boxhorn, L.K. Regulation of skeletal muscle satellite cell proliferation and differentiation by transforming growth factor-beta, insulin-like growth factor I, and fibroblast growth factor. J. Cell. Physiol. 1989, 138, 311–315. [Google Scholar] [CrossRef]
- Spangenburg, E.E.; Booth, F.W. Multiple signaling pathways mediate LIF-induced skeletal muscle satellite cell proliferation. Am. J. Physiol. Cell Physiol. 2002, 283, C204–C211. [Google Scholar] [CrossRef] [PubMed]
- Bentzinger, C.F.; von Maltzahn, J.; Rudnicki, M.A. Extrinsic regulation of satellite cell specification. Stem Cell Res. Ther. 2010, 1, 27. [Google Scholar] [CrossRef]
- Miller, K.J.; Thaloor, D.; Matteson, S.; Pavlath, G.K. Hepatocyte growth factor affects satellite cell activation and differentiation in regenerating skeletal muscle. Am. J. Physiol. Cell Physiol. 2000, 278, C174–C181. [Google Scholar] [CrossRef]
- Engert, J.C.; Berglund, E.B.; Rosenthal, N. Proliferation precedes differentiation in IGF-I-stimulated myogenesis. J. Cell Biol. 1996, 135, 431–440. [Google Scholar] [CrossRef]
- Musaro Á, A.; McCullagh, K.; Naya, F.; Olson, E.; Rosenthal, N. IGF-1 induces skeletal myocyte hypertrophy through calcineurin in association with GATA-2 and NF-ATc1. Nature 1999, 400, 581–585. [Google Scholar] [CrossRef] [PubMed]
- Carlson, M.E.; Conboy, M.J.; Hsu, M.; Barchas, L.; Jeong, J.; Agrawal, A.; Mikels, A.J.; Agrawal, S.; Schaffer, D.V.; Conboy, I.M. Relative roles of TGF-beta1 and Wnt in the systemic regulation and aging of satellite cell responses. Aging Cell 2009, 8, 676–689. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-J. Regulation of muscle mass by myostatin. Annu. Rev. Cell Dev. Biol. 2004, 20, 61–86. [Google Scholar] [CrossRef]
- Chen, S.-E.; Jin, B.; Li, Y.-P. TNF-alpha regulates myogenesis and muscle regeneration by activating p38 MAPK. Am. J. Physiol. Cell Physiol. 2007, 292, C1660–C1671. [Google Scholar] [CrossRef] [PubMed]
- Braga, M.; Simmons, Z.; Norris, K.C.; Ferrini, M.G.; Artaza, J.N. Vitamin D induces myogenic differentiation in skeletal muscle derived stem cells. Endocr. Connect. 2017, 6, 139–150. [Google Scholar] [CrossRef]
- Ryan, P.A.; Maher, V.M.; McCormick, J.J. Modification of MCDB 110 medium to support prolonged growth and consistent high cloning efficiency of diploid human fibroblasts. Exp. Cell Res. 1987, 172, 318–328. [Google Scholar] [CrossRef]
- Beers, J.; Gulbranson, D.R.; George, N.; Siniscalchi, L.I.; Jones, J.; Thomson, J.A.; Chen, G. Passaging and colony expansion of human pluripotent stem cells by enzyme-free dissociation in chemically defined culture conditions. Nat. Protoc. 2012, 7, 2029–2040. [Google Scholar] [CrossRef]
- Xu, C.; Rosler, E.; Jiang, J.; Lebkowski, J.S.; Gold, J.D.; O’Sullivan, C.; Delavan-Boorsma, K.; Mok, M.; Bronstein, A.; Carpenter, M.K. Basic fibroblast growth factor supports undifferentiated human embryonic stem cell growth without conditioned medium. Stem Cells 2005, 23, 315–323. [Google Scholar] [CrossRef]
- Amit, M.; Shariki, C.; Margulets, V.; Itskovitz-Eldor, J. Feeder layer- and serum-free culture of human embryonic stem cells. Biol. Reprod. 2004, 70, 837–845. [Google Scholar] [CrossRef]
- Kuo, H.-H.; Gao, X.; DeKeyser, J.-M.; Fetterman, K.A.; Pinheiro, E.A.; Weddle, C.J.; Fonoudi, H.; Orman, M.V.; Romero-Tejeda, M.; Jouni, M.; et al. Negligible-Cost and Weekend-Free Chemically Defined Human iPSC Culture. Stem Cell Rep. 2020, 14, 256–270. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Tristan, C.A.; Chen, L.; Jovanovic, V.M.; Malley, C.; Chu, P.-H.; Ryu, S.; Deng, T.; Ormanoglu, P.; Tao, D.; et al. A versatile polypharmacology platform promotes cytoprotection and viability of human pluripotent and differentiated cells. Nat. Methods 2021, 18, 528–541. [Google Scholar] [CrossRef]
- Watanabe, K.; Ueno, M.; Kamiya, D.; Nishiyama, A.; Matsumura, M.; Wataya, T.; Takahashi, J.B.; Nishikawa, S.; Nishikawa, S.; Muguruma, K.; et al. A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nat. Biotechnol. 2007, 25, 681–686. [Google Scholar] [CrossRef]
- Petitte, J.N. Isolation and maintenance of avian ES cells. In Handbook of Stem Cells; Elsevier: Amsterdam, The Netherlands, 2004; pp. 471–477. ISBN 9780124366435. [Google Scholar]
- Gstraunthaler, G.; Lindl, T.; van der Valk, J. A plea to reduce or replace fetal bovine serum in cell culture media. Cytotechnology 2013, 65, 791–793. [Google Scholar] [CrossRef]
- Van der Valk, J.; Brunner, D.; De Smet, K.; Fex Svenningsen, A.; Honegger, P.; Knudsen, L.E.; Lindl, T.; Noraberg, J.; Price, A.; Scarino, M.L.; et al. Optimization of chemically defined cell culture media—Replacing fetal bovine serum in mammalian in vitro methods. Toxicol. In Vitro 2010, 24, 1053–1063. [Google Scholar] [CrossRef]
- Mosa Meat Growth Medium without Fetal Bovine Serum (FBS)—Mosa Meat. Available online: https://mosameat.com/blog/growth-medium-without-fetal-bovine-serum-fbs (accessed on 10 January 2022).
- Merck Future Food Tech: Discussing Cell Culture Media for Cultured Meat—News—Innovation Center|Merck. Available online: https://www.merckgroup.com/en/research/innovation-center/news/all-news/future-food-tech.html (accessed on 10 January 2022).
- Messmer, T.; Klevernic, I.; Furquim, C.; Ovchinnikova, E.; Dogan, A.; Cruz, H.; Post, M.J.; Flack, J.E. A serum-free media formulation for cultured meat production supports bovine satellite cell differentiation in the absence of serum starvation. Nat. Food 2022. [Google Scholar] [CrossRef]
- Aleph Farms and WACKER Develop Open Supply Chain Solution for Proteins Used in Large-Scale Production of Cultivated Meat—Sustainability. Available online: https://www.aleph-farms.com/blog/open-supply-chain-solution (accessed on 10 January 2022).
- Kolkmann, A.M.; Post, M.J.; Rutjens, M.A.M.; van Essen, A.L.M.; Moutsatsou, P. Serum-free media for the growth of primary bovine myoblasts. Cytotechnology 2020, 72, 111–120. [Google Scholar] [CrossRef]
- Sinacore, M.S.; Drapeau, D.; Adamson, S.R. Adaptation of Mammalian Cells to Growth in Serum-Free Media. Mol. Biotechnol. 2000, 15, 249–258. [Google Scholar] [CrossRef]
- Heathman, T.R.J.; Nienow, A.W.; Rafiq, Q.A.; Coopman, K.; Kara, B.; Hewitt, C.J. Development of a process control strategy for the serum-free microcarrier expansion of human mesenchymal stem cells towards cost-effective and commercially viable manufacturing. Biochem. Eng. J. 2019, 141, 200–209. [Google Scholar] [CrossRef]
- Busse, C.; Biechele, P.; de Vries, I.; Reardon, K.F.; Solle, D.; Scheper, T. Sensors for disposable bioreactors. Eng. Life Sci. 2017, 17, 940–952. [Google Scholar] [CrossRef]
- Biechele, P.; Busse, C.; Solle, D.; Scheper, T.; Reardon, K. Sensor systems for bioprocess monitoring. Eng. Life Sci. 2015, 15, 469–488. [Google Scholar] [CrossRef]
- Zhao, L.; Fu, H.-Y.; Zhou, W.; Hu, W.-S. Advances in process monitoring tools for cell culture bioprocesses. Eng. Life Sci. 2015, 15, 459–468. [Google Scholar] [CrossRef]
- Kieninger, J.; Weltin, A.; Flamm, H.; Urban, G.A. Microsensor systems for cell metabolism—From 2D culture to organ-on-chip. Lab Chip 2018, 18, 1274–1291. [Google Scholar] [CrossRef]
- O’Mara, P.; Farrell, A.; Bones, J.; Twomey, K. Staying alive! Sensors used for monitoring cell health in bioreactors. Talanta 2018, 176, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Weidemann, R.; Ludwig, A.; Kretzmer, G. Low temperature cultivation—A step towards process optimisation. In Cell Culture Engineering IV: Improvements of Human Health; Current applications of cell culture, engineering; Buckland, B.C., Aunins, J.G., Bibila, T.A., Hu, W.-S., Robinson, D.K., Zhou, W., Eds.; Springer: Dordrecht, The Netherlands, 1995; Volume 1, pp. 111–116. ISBN 978-94-010-4114-0. [Google Scholar]
- Furukawa, K.; Ohsuye, K. Effect of culture temperature on a recombinant CHO cell line producing a C-terminal α-amidating enzyme. Cytotechnology 1998, 26, 153–164. [Google Scholar] [CrossRef]
- Mokhatab, S.; Poe, W.A.; Mak, J.Y. Gas processing plant automation. In Handbook of Natural Gas Transmission and Processing; Elsevier: Amsterdam, The Netherlands, 2019; pp. 615–642. ISBN 9780128158173. [Google Scholar]
- Maurer, D. The Significance of pH Stability for Cell Cultures|American Laboratory. Available online: https://www.americanlaboratory.com/914-Application-Notes/30798-The-Significance-of-pH-Stability-for-Cell-Cultures/ (accessed on 21 November 2021).
- Michl, J.; Park, K.C.; Swietach, P. Evidence-based guidelines for controlling pH in mammalian live-cell culture systems. Commun. Biol. 2019, 2, 144. [Google Scholar] [CrossRef] [PubMed]
- Naciri, M.; Kuystermans, D.; Al-Rubeai, M. Monitoring pH and dissolved oxygen in mammalian cell culture using optical sensors. Cytotechnology 2008, 57, 245–250. [Google Scholar] [CrossRef]
- Wei, Y.; Jiao, Y.; An, D.; Li, D.; Li, W.; Wei, Q. Review of dissolved oxygen detection technology: From laboratory analysis to online intelligent detection. Sensors 2019, 19, 3995. [Google Scholar] [CrossRef] [PubMed]
- Hamilton Should CO2 Be a Critical Process Parameter? Available online: https://www.primalab.hr/Portals/2/Hamilton%20Control/Hamilton_Should-CO2-Be-A-CPP.pdf (accessed on 21 November 2021).
- Verma, A.; Verma, M.; Singh, A. Animal tissue culture principles and applications. In Animal Biotechnology; Elsevier: Amsterdam, The Netherlands, 2020; pp. 269–293. ISBN 9780128117101. [Google Scholar]
- Ge, X.; Kostov, Y.; Rao, G. Low-cost noninvasive optical CO2 sensing system for fermentation and cell culture. Biotechnol. Bioeng. 2005, 89, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Mills, A. Optical sensors for carbon dioxide and their applications. In Sensors for Environment, Health and Security; Baraton, M.-I., Ed.; Springer: Dordrecht, The Netherlands, 2009; pp. 347–370. ISBN 978-1-4020-9009-7. [Google Scholar]
- Tamburini, E.; Marchetti, M.G.; Pedrini, P. Monitoring key parameters in bioprocesses using near-infrared technology. Sensors 2014, 14, 18941–18959. [Google Scholar] [CrossRef] [PubMed]
- Yardley, J.E.; Kell, D.B.; Barrett, J.; Davey, C.L. On-line, real-time measurements of cellular biomass using dielectric spectroscopy. Biotechnol. Genet. Eng. Rev. 2000, 17, 3–35. [Google Scholar] [CrossRef] [PubMed]
- Alpfmedical Standard Bioreactor Sensors 13221 Introduction—Cell Culture. Available online: https://www.alpfmedical.info/cell-culture/standard-bioreactor-sensors-13221-introduction.html (accessed on 23 November 2020).
- Ozturk, S.S.; Thrift, J.C.; Blackie, J.D.; Naveh, D. Real-time monitoring and control of glucose and lactate concentrations in a mammalian cell perfusion reactor. Biotechnol. Bioeng. 1997, 53, 372–378. [Google Scholar] [CrossRef]
- Eyer, K.; Heinzle, E. On-line estimation of viable cells in a hybridoma culture at various DO levels using ATP balancing and redox potential measurement. Biotechnol. Bioeng. 2000, 49, 277–283. [Google Scholar] [CrossRef]
- Fan, R.; Ebrahimi, M.; Quitmann, H.; Aden, M.; Czermak, P. An innovative optical sensor for the online monitoring and control of biomass concentration in a membrane bioreactor system for lactic acid production. Sensors 2016, 16, 411. [Google Scholar] [CrossRef] [PubMed]
- Afguard AFGUARD ǀ FAUDI aviation—Quality guarantees Safety. Available online: https://www.faudi-aviation.com/en/products/sensor-technology/afguard/ (accessed on 21 November 2020).
- Ude, C.; Schmidt-Hager, J.; Findeis, M.; John, G.T.; Scheper, T.; Beutel, S. Application of an online-biomass sensor in an optical multisensory platform prototype for growth monitoring of biotechnical relevant microorganism and cell lines in single-use shake flasks. Sensors 2014, 14, 17390–17405. [Google Scholar] [CrossRef]
- Direct Industry Reactor Temperature Sensor. Available online: https://www.directindustry.com/industrial-manufacturer/reactor-temperature-sensor-251712.html (accessed on 25 November 2020).
- Innovative Sensor Technology Customizable Platinum & Nickel RTD Temperature Sensors|IST AG. Available online: https://www.ist-ag.com/en/products-services/temperature-sensors (accessed on 11 January 2022).
- Rosemount RosemountTM Multipoint Thermocoupleand RTD Profiling Sensors. Available online: https://www.emerson.com/ (accessed on 11 January 2022).
- PyroScience Sensors—PyroScience GmbH. Available online: https://www.pyroscience.com/en/products/all-sensors/attributes/pH (accessed on 11 January 2022).
- Burns Engineering True Life-Cycle Cost of a Resistance Temperature Detector. Available online: https://burnsengineering.com/technical-paper/ (accessed on 11 January 2022).
- PreSens Optical pH Measurementfor Academic and Industrial Applications. Available online: https://www.presens.de/products/ph (accessed on 26 November 2020).
- METTLER TOLEDO pH Sensors, Probes & Electrodes. Available online: https://www.mt.com/int/en/home/perm-lp/product-organizations/pro/ph-sensors-probes-electrodes-2.html (accessed on 28 February 2021).
- Boqu China Bioreactor pH Probe—China High Temperature pH Sensor. Available online: https://shboqu.en.made-in-china.com/product/jKBxtdCycZWV/China-Bioreactor-pH-Probe.html (accessed on 11 January 2022).
- Mettler Toledo DO In-Line Dissolved Oxygen Sensor|In-Line DO Probe|In-Line DO Sensor. Available online: https://www.mt.com/int/en/home/products/Process-Analytics/DO-CO2-ozone-sensor/dissolved-oxygen-meter.html (accessed on 11 January 2022).
- Industrial Physics Paramagnetic Cells Technology for Our Paramagnetic O2 Analyser—Industrial Physics Paramagnetic Cells Technology For Our Paramagnetic O2 Analyser—Industrial Physics. Available online: https://industrialphysics.com/knowledgebase/paramagnetic-cells-technology-for-our-paramagnetic-o2-analyser/ (accessed on 11 January 2022).
- PreSens Single Use Single-Use Sensors for Biopharmaceutical Production. Available online: https://www.presens.de/products/oem-components/single-use-sensors-for-biopharmaceutical-production (accessed on 11 January 2022).
- Mettler Toledo CO2 Dissolved CO2 Sensor|Dissolved Carbon Dioxide Probe|In Situ CO2 Sensor. Available online: https://www.mt.com/ca/en/home/products/Process-Analytics/DO-CO2-ozone-sensor/dissolved-carbon-dioxide.html (accessed on 11 January 2022).
- Oyeleye, O.O.; Ogundeji, S.T.; Ola, S.I.; Omitogun, O.G. Basics of animal cell culture: Foundation for modern science. Biotechnol. Mol. Biol. Rev. 2016, 11, 6–16. [Google Scholar] [CrossRef]
- Galvanauskas, V.; Simutis, R.; Nath, S.C.; Kino-Oka, M. Kinetic modeling of human induced pluripotent stem cell expansion in suspension culture. Regen. Ther. 2019, 12, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Kyriakopoulos, S.; Ang, K.S.; Lakshmanan, M.; Huang, Z.; Yoon, S.; Gunawan, R.; Lee, D.-Y. Kinetic modeling of mammalian cell culture bioprocessing: The quest to advance biomanufacturing. Biotechnol. J. 2018, 13, e1700229. [Google Scholar] [CrossRef]
- Sartori, R.; Leme, J.; Caricati, C.P.; Tonso, A.; Núñez, E.G.F. Model comparison to describe bhk-21 cell growth and metabolism in stirred tank bioreactors operated in batch mode. Braz. J. Chem. Eng. 2018, 35, 441–458. [Google Scholar] [CrossRef]
- Sidoli, F.R.; Asprey, S.P.; Mantalaris, A. A Coupled Single Cell-Population-Balance Model for Mammalian Cell Cultures. Ind. Eng. Chem. Res. 2006, 45, 5801–5811. [Google Scholar] [CrossRef]
- Lynch, J.; Pierrehumbert, R. Climate impacts of cultured meat and beef cattle. Front. Sustain. Food Syst. 2019, 3. [Google Scholar] [CrossRef]
- Treich, N. Cultured meat: Promises and challenges. Environ. Resour. Econ. 2021, 79, 1–29. [Google Scholar] [CrossRef]
- Ghaffar, T.; Irshad, M.; Anwar, Z.; Aqil, T.; Zulifqar, Z.; Tariq, A.; Kamran, M.; Ehsan, N.; Mehmood, S. Recent trends in lactic acid biotechnology: A brief review on production to purification. J. Radiat. Res. Appl. Sci. 2014, 7, 222–229. [Google Scholar] [CrossRef]
- Warner, R.D. Review: Analysis of the process and drivers for cellular meat production. Animal 2019, 13, 3041–3058. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, Y.; Kagawa, Y.; Sakaguchi, K.; Matsuura, K.; Shimizu, T.; Okano, T. Thicker three-dimensional tissue from a “symbiotic recycling system” combining mammalian cells and algae. Sci. Rep. 2017, 7, 41594. [Google Scholar] [CrossRef] [PubMed]
- Humbird, D. Scale-up economics for cultured meat. Biotechnol. Bioeng. 2021, 118, 3239–3250. [Google Scholar] [CrossRef] [PubMed]
- Fassler, J.; Lab-Grown Meat Is Supposed to Be Inevitable. The Science Tells a Different Story. The Counter. 22 September 2021. Available online: https://thecounter.org/lab-grown-cultivated-meat-cost-at-scale/ (accessed on 11 January 2022).
- The Good Food Institute The Top 7 Alternative Protein Trends to Watch for in 2022—The Good Food Institute. Available online: https://gfi.org/blog/2022-alternative-protein-trends (accessed on 12 January 2022).
- Vergeer, R.; Sinke, P.; Odegard, I. TEA of Cultivated Meat. Future Projections for Different Scenarios—CE Delft—EN; CE Delft: Delft, The Netherlands, 2021; p. 45. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pajčin, I.; Knežić, T.; Savic Azoulay, I.; Vlajkov, V.; Djisalov, M.; Janjušević, L.; Grahovac, J.; Gadjanski, I. Bioengineering Outlook on Cultivated Meat Production. Micromachines 2022, 13, 402. https://doi.org/10.3390/mi13030402
Pajčin I, Knežić T, Savic Azoulay I, Vlajkov V, Djisalov M, Janjušević L, Grahovac J, Gadjanski I. Bioengineering Outlook on Cultivated Meat Production. Micromachines. 2022; 13(3):402. https://doi.org/10.3390/mi13030402
Chicago/Turabian StylePajčin, Ivana, Teodora Knežić, Ivana Savic Azoulay, Vanja Vlajkov, Mila Djisalov, Ljiljana Janjušević, Jovana Grahovac, and Ivana Gadjanski. 2022. "Bioengineering Outlook on Cultivated Meat Production" Micromachines 13, no. 3: 402. https://doi.org/10.3390/mi13030402
APA StylePajčin, I., Knežić, T., Savic Azoulay, I., Vlajkov, V., Djisalov, M., Janjušević, L., Grahovac, J., & Gadjanski, I. (2022). Bioengineering Outlook on Cultivated Meat Production. Micromachines, 13(3), 402. https://doi.org/10.3390/mi13030402









