Surfactant-Laden Janus Droplets with Tunable Morphologies and Enhanced Stability for Fabricating Lens-Shaped Polymeric Microparticles
Abstract
1. Introduction
2. Materials and Methods
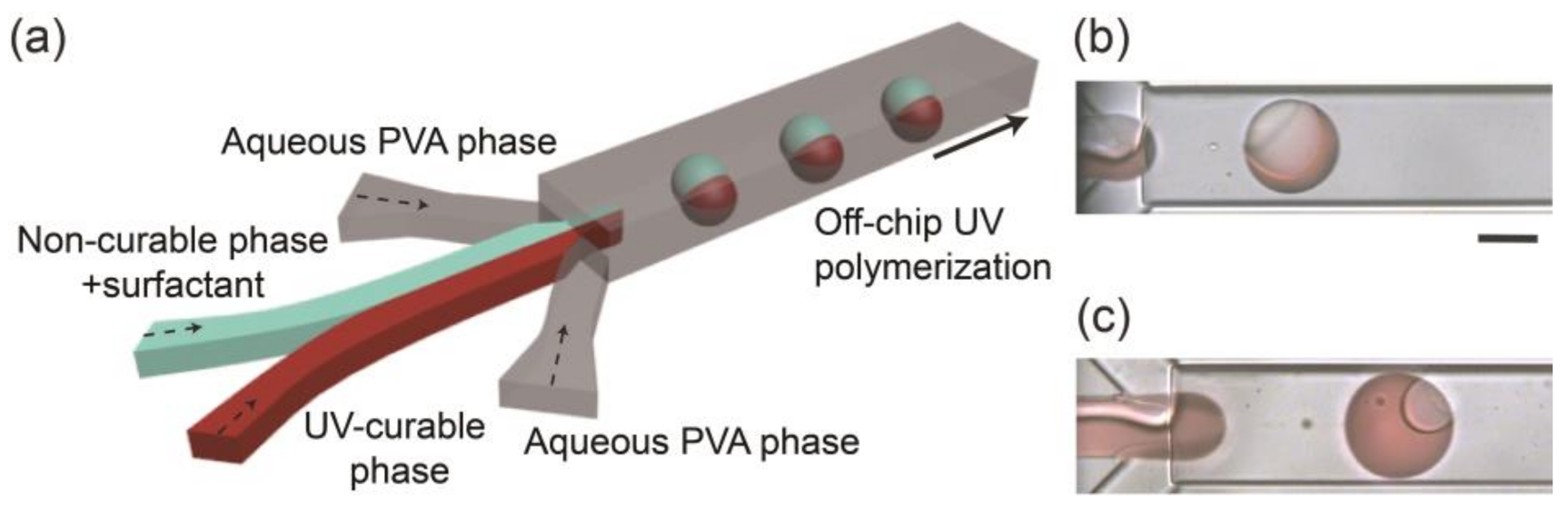
2.1. Microfluidic Device
2.2. Chemicals
2.3. Preparation of Droplets and Lens-Shaped Polymer Particles
2.4. Characterization
3. Results and Discussion
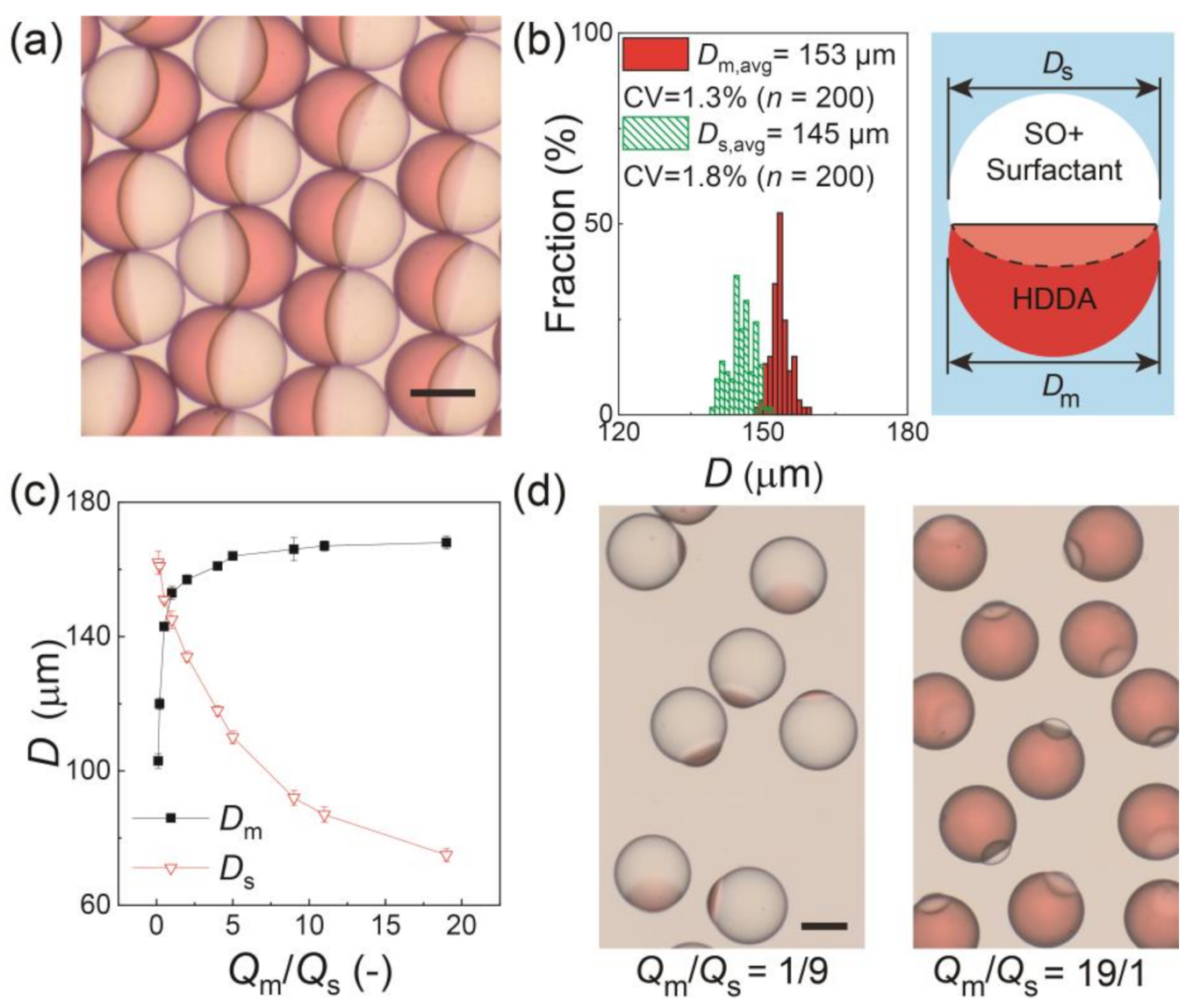
3.1. Generation of Biphasic Janus Droplets
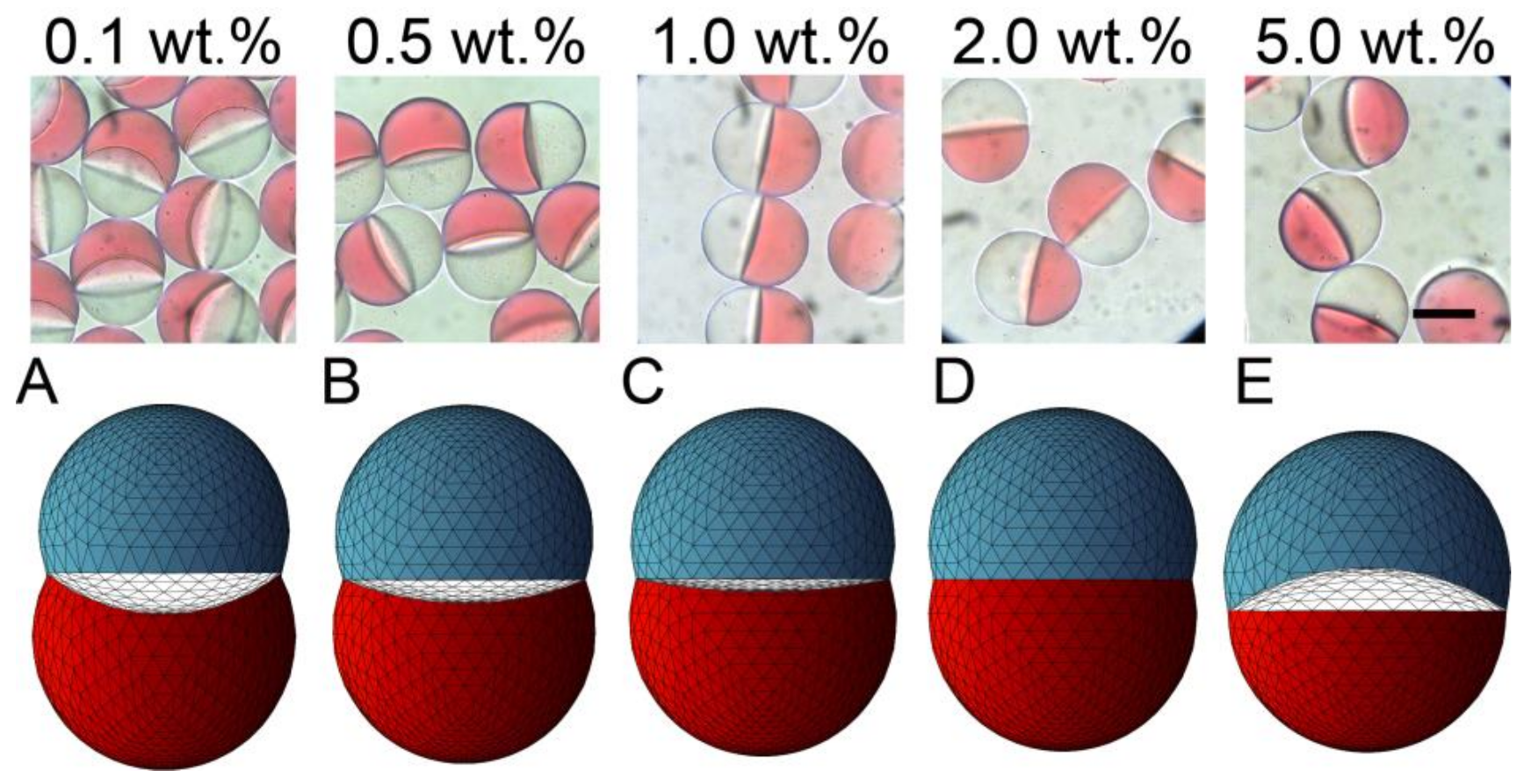
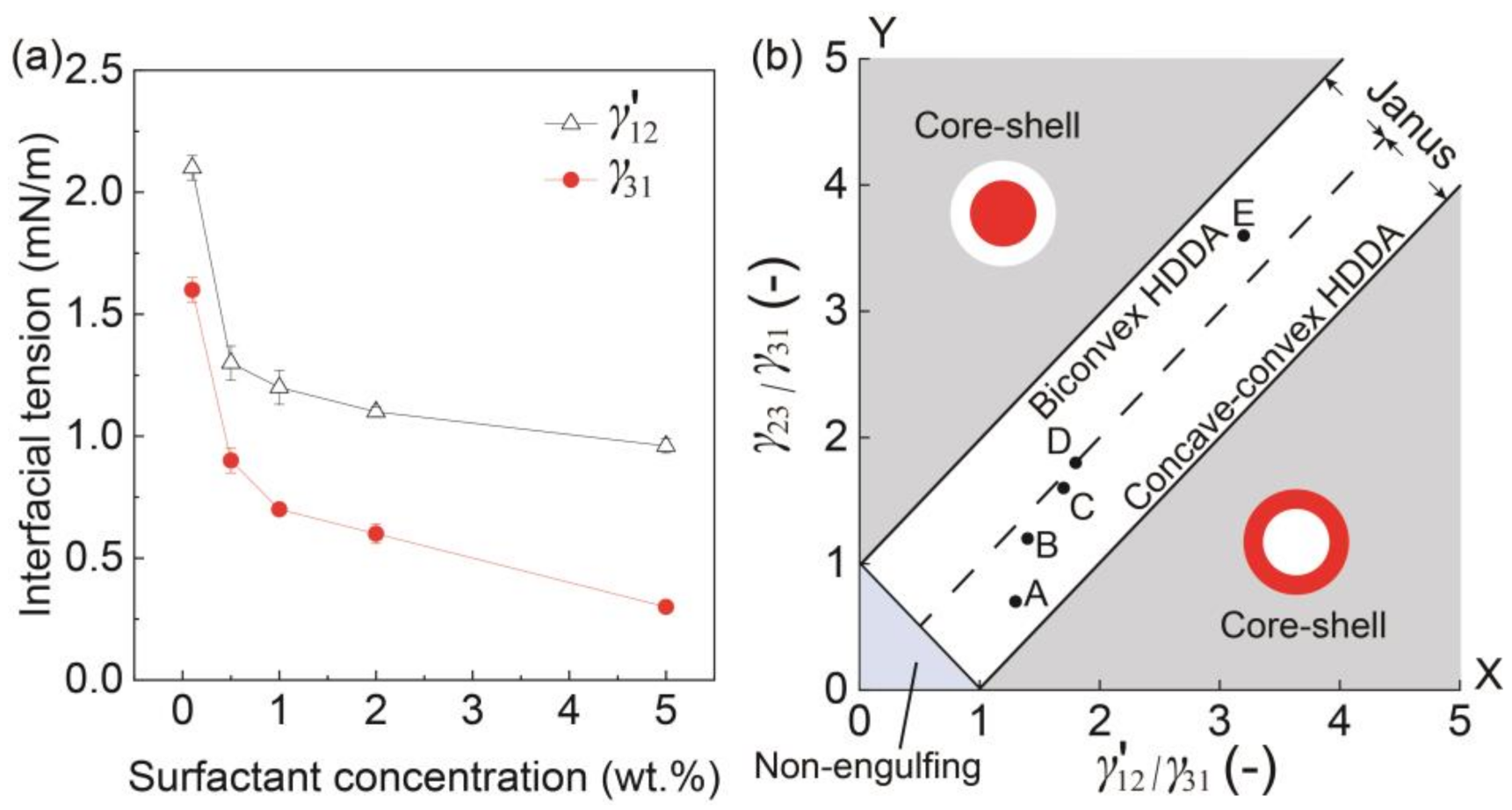
3.2. Effect of Inner Surfactant Concentration on Janus Morphology
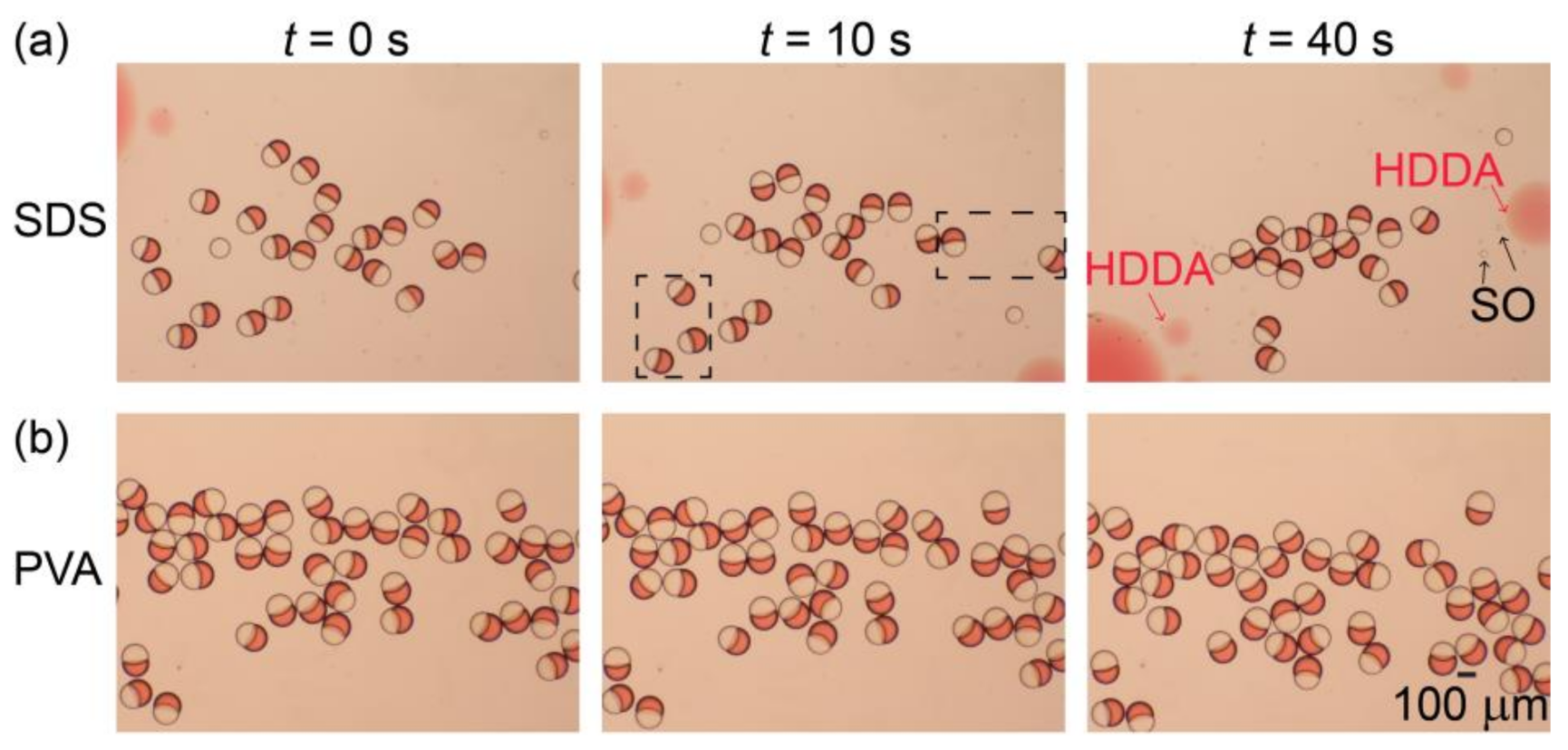
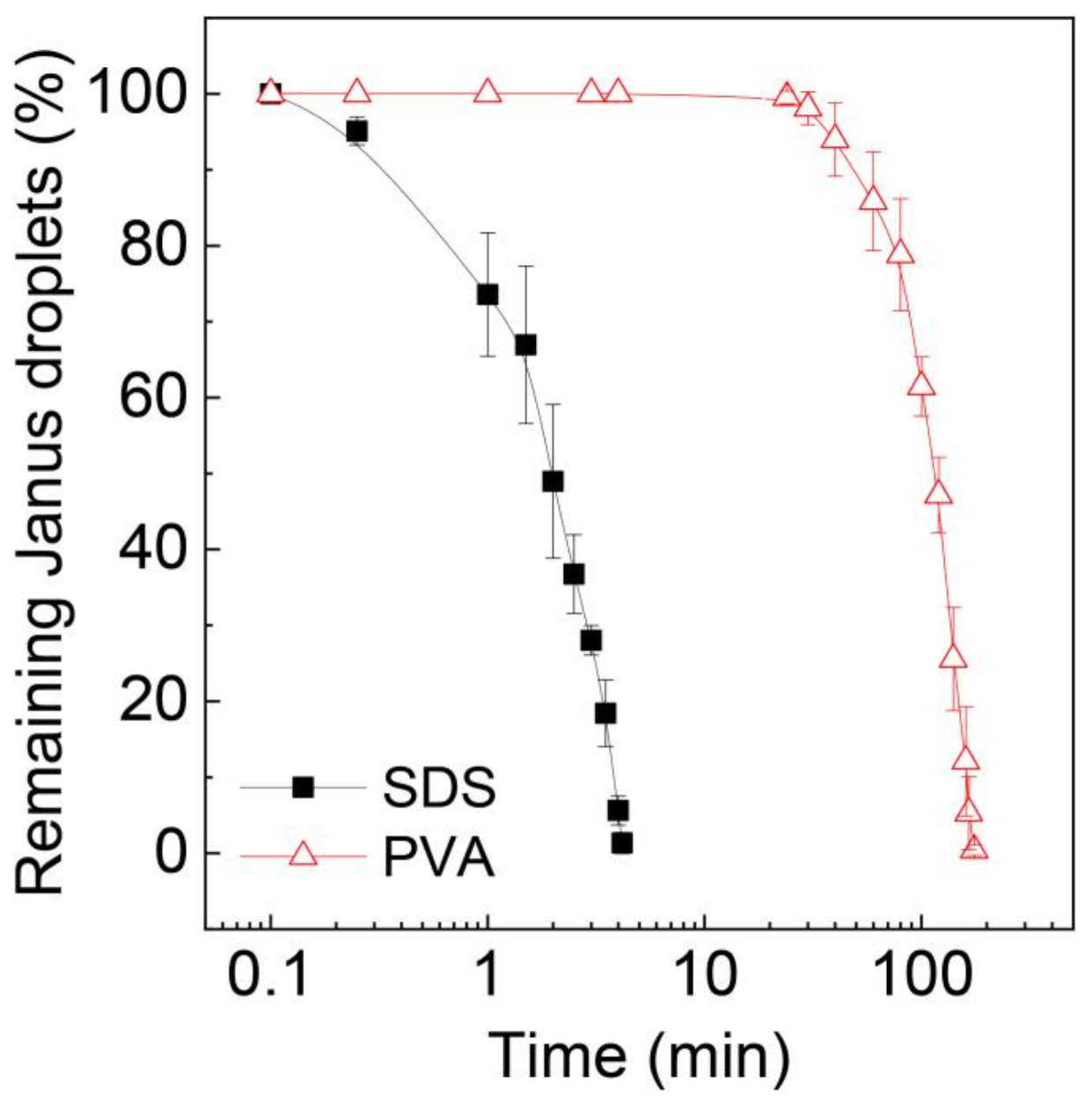
3.3. Stability Assessment of Janus Droplets
3.4. Fabrication and Characterization of Microlens-Shaped Particles
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Li, X.; Yang, Y.T.; Wu, L.J.; Li, Y.C.; Ye, M.Y.; Chang, Z.Q.; Meng, D.Q.; Serra, C.A. Fabrication of electro- and color-responsive CB/PTFE Janus beads in a simple microfluidic device. Mater. Lett. 2015, 142, 258–261. [Google Scholar] [CrossRef]
- Varma, V.B.; Wu, R.G.; Wang, Z.P.; Ramanujan, R.V. Magnetic Janus particles synthesized using droplet micro-magnetofluidic techniques for protein detection. Lab Chip 2017, 17, 3514–3525. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Ge, X.H.; Huang, J.P.; Dang, Z.X.; Xu, J.H.; Luo, G.S. A region-selective modified capillary microfluidic device for fabricating water–oil Janus droplets and hydrophilic–hydrophobic anisotropic microparticles. RSC Adv. 2015, 5, 46981–46988. [Google Scholar] [CrossRef]
- Gröschel, A.H.; Walther, A.; Löbling, T.I.; Schmelz, J.; Hanisch, A.; Schmalz, H.; Müller, A.H.E. Facile, solution-based synthesis of soft, nanoscale Janus particles with tunable Janus balance. J. Am. Chem. Soc. 2012, 134, 13850–13860. [Google Scholar] [CrossRef] [PubMed]
- Hasinovic, H.; Friberg, S.E. One-step inversion process to a Janus emulsion with two mutually insoluble oils. Langmuir 2011, 27, 6584–6588. [Google Scholar] [CrossRef]
- Nisisako, T. Recent advances in microfluidic production of Janus droplets and particles. Curr. Opin. Colloid Interface Sci. 2016, 25, 1–12. [Google Scholar] [CrossRef]
- Nisisako, T.; Torii, T.; Takahashi, T.; Takizawa, Y. Synthesis of monodisperse bicolored Janus particles with electrical anisotropy using a microfluidic co-flow system. Adv. Mater. 2006, 18, 1152–1156. [Google Scholar] [CrossRef]
- Nisisako, T.; Torii, T. Formation of biphasic Janus droplets in a microfabricated channel for the synthesis of shape-controlled polymer microparticles. Adv. Mater. 2007, 19, 1489–1493. [Google Scholar] [CrossRef]
- Nisisako, T.; Ando, T.; Hatsuzawa, T. Biconvex polymer microlenses fabricated from microfluidic Janus droplets. J. Jpn. Soc. Precis. Eng. 2013, 79, 460–466. [Google Scholar] [CrossRef]
- Nisisako, T.; Ando, T.; Hatsuzawa, T. Capillary-assisted fabrication of biconcave polymeric microlenses from microfluidic ternary emulsion droplets. Small 2014, 10, 5116–5125. [Google Scholar] [CrossRef]
- Nisisako, T.; Suzuki, H.; Hatsuzawa, T. Biconvex polymer microlenses with tunable imaging properties designed by Janus droplet microfluidics. Micromachines 2015, 6, 1435–1444. [Google Scholar] [CrossRef]
- Jeong, J.; Gross, A.; Wei, W.S.; Tu, F.; Lee, D.; Collings, P.J.; Yodh, A.G. Liquid crystal Janus emulsion droplets: Preparation, tumbling, and swimming. Soft Matter 2015, 11, 6747–6754. [Google Scholar] [CrossRef] [PubMed]
- Nisisako, T.; Hatsuzawa, T. A microfluidic cross-flowing emulsion generator for producing biphasic droplets and anisotropically shaped polymer particles. Microfluid. Nanofluid. 2010, 9, 427–437. [Google Scholar] [CrossRef]
- Yang, Y.T.; Wei, J.; Li, X.; Wu, L.J.; Chang, Z.Q.; Serra, C.A. A side-by-side capillaries-based microfluidic system for synthesizing size- and morphology-controlled magnetic anisotropy Janus beads. Adv. Powder Technol. 2015, 26, 156–162. [Google Scholar] [CrossRef]
- Seo, K.D.; Doh, J.; Kim, D.S. One-step microfluidic synthesis of Janus microhydrogels with anisotropic thermo-responsive behavior and organophilic/hydrophilic loading capability. Langmuir 2013, 29, 15137–15141. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Abbaspourrad, A.; Weitz, D.A. Amphiphilic crescent-moon-shaped microparticles formed by selective adsorption of colloids. J. Am. Chem. Soc. 2011, 133, 5516–5524. [Google Scholar] [CrossRef] [PubMed]
- Torza, S.; Mason, S.G. Coalescence of two immiscible liquid drops. Science 1969, 163, 813–814. [Google Scholar] [CrossRef]
- Kang, Z.; Kong, T.; Lei, L.; Zhu, P.; Tian, X.; Wang, L. Engineering particle morphology with microfluidic droplets. J. Micromech. Microeng. 2016, 26, 075011. [Google Scholar] [CrossRef]
- Ge, X.; Geng, Y.; Zhang, Q.; Shao, M.; Chen, J.; Luo, G.; Xu, J. Four reversible and reconfigurable structures for three-phase emulsions: Extended morphologies and applications. Sci. Rep. 2017, 7, 42738. [Google Scholar] [CrossRef]
- Zarzar, L.D.; Sresht, V.; Sletten, E.M.; Kalow, J.A.; Blankschtein, D.; Swager, T.M. Dynamically reconfigurable complex emulsions via tunable interfacial tensions. Nature 2015, 518, 520–524. [Google Scholar] [CrossRef]
- Nisisako, T.; Hatsuzawa, T. Microfluidic fabrication of oil-filled polymeric microcapsules with independently controllable size and shell thickness via Janus to core-shell evolution of biphasic droplets. Sens. Actuators B Chem. 2016, 223, 209–216. [Google Scholar] [CrossRef]
- Xu, S.; Nisisako, T. Polymer capsules with tunable shell thickness synthesized via Janus-to-core shell transition of biphasic droplets produced in a microfluidic flow-focusing device. Sci. Rep. 2020, 10, 4549. [Google Scholar] [CrossRef] [PubMed]
- Brakke, K. The surface evolver. Exp. Math. 1992, 1, 141. [Google Scholar] [CrossRef]
- Hui, C.Y.; Jagota, A. Planar equilibrium shapes of a liquid drop on a membrane. Soft Matter 2015, 11, 8960–8967. [Google Scholar] [CrossRef] [PubMed]
- Guzowski, J.; Korczyk, P.M.; Jakiela, S.; Garstecki, P. The structure and stability of multiple micro-droplets. Soft Matter 2012, 8, 7269–7278. [Google Scholar] [CrossRef]
- Baret, J.C. Surfactants in droplet-based microfluidics. Lab Chip 2012, 12, 422–433. [Google Scholar] [CrossRef]
- Raju, R.R.; Kosmella, S.; Friberg, S.E.; Koetz, J. Pickering Janus emulsions and polyelectrolyte complex-stabilized Janus gels. Colloids Surf. A 2017, 533, 241–248. [Google Scholar] [CrossRef]
- Milani, R.; Monogioudi, E.; Baldrighi, M.; Cavallo, G.; Arima, V.; Marra, L.; Zizzari, A.; Rinaldi, R.; Linder, M.; Resnati, G.; et al. Hydrophobin: Fluorosurfactant-like properties without fluorine. Soft Matter 2013, 9, 6505–6514. [Google Scholar] [CrossRef]
- Okada, M.; Sumino, Y.; Ito, H.; Kitahata, H. Spontaneous deformation and fission of oil droplets on an aqueous surfactant solution. Phys. Rev. E 2020, 102, 042603. [Google Scholar] [CrossRef]
- Kovach, I.; Koetz, J.; Friberg, S.E. Janus emulsions stabilized by phospholipids. Colloids Surf. A 2014, 441, 66–71. [Google Scholar] [CrossRef]
- Malloggi, F.; Pannacci, N.; Attia, R.; Monti, F.; Mary, P.; Willaime, H.; Poncet, P. Monodisperse colloids synthesized with nanofluidic technology. Langmuir 2010, 26, 2369–2373. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.K.; Dendukuri, D.; Doyle, P.S. Microfluidic-based synthesis of non-spherical magnetic hydrogel microparticles. Lab Chip 2008, 8, 1640–1647. [Google Scholar] [CrossRef] [PubMed]
- Nisisako, T.; Ando, T.; Hatsuzawa, T. High-volume production of single and compound emulsions in a microfluidic parallelization arrangement coupled with coaxial annular world-to-chip interfaces. Lab Chip 2012, 12, 3426–3435. [Google Scholar] [CrossRef] [PubMed]
- Lan, W.; Du, Y.; Guo, X.; Liu, A.; Jing, S.; Li, S. Flexible microfluidic fabrication of anisotropic polymer microfibers. Ind. Eng. Chem. Res. 2017, 57, 212–219. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, S.; Nisisako, T. Surfactant-Laden Janus Droplets with Tunable Morphologies and Enhanced Stability for Fabricating Lens-Shaped Polymeric Microparticles. Micromachines 2021, 12, 29. https://doi.org/10.3390/mi12010029
Xu S, Nisisako T. Surfactant-Laden Janus Droplets with Tunable Morphologies and Enhanced Stability for Fabricating Lens-Shaped Polymeric Microparticles. Micromachines. 2021; 12(1):29. https://doi.org/10.3390/mi12010029
Chicago/Turabian StyleXu, Siyuan, and Takasi Nisisako. 2021. "Surfactant-Laden Janus Droplets with Tunable Morphologies and Enhanced Stability for Fabricating Lens-Shaped Polymeric Microparticles" Micromachines 12, no. 1: 29. https://doi.org/10.3390/mi12010029
APA StyleXu, S., & Nisisako, T. (2021). Surfactant-Laden Janus Droplets with Tunable Morphologies and Enhanced Stability for Fabricating Lens-Shaped Polymeric Microparticles. Micromachines, 12(1), 29. https://doi.org/10.3390/mi12010029




