Microfluidic Magnetic Mixing at Low Reynolds Numbers and in Stagnant Fluids
Abstract
1. Introduction
2. Micromixing Methods
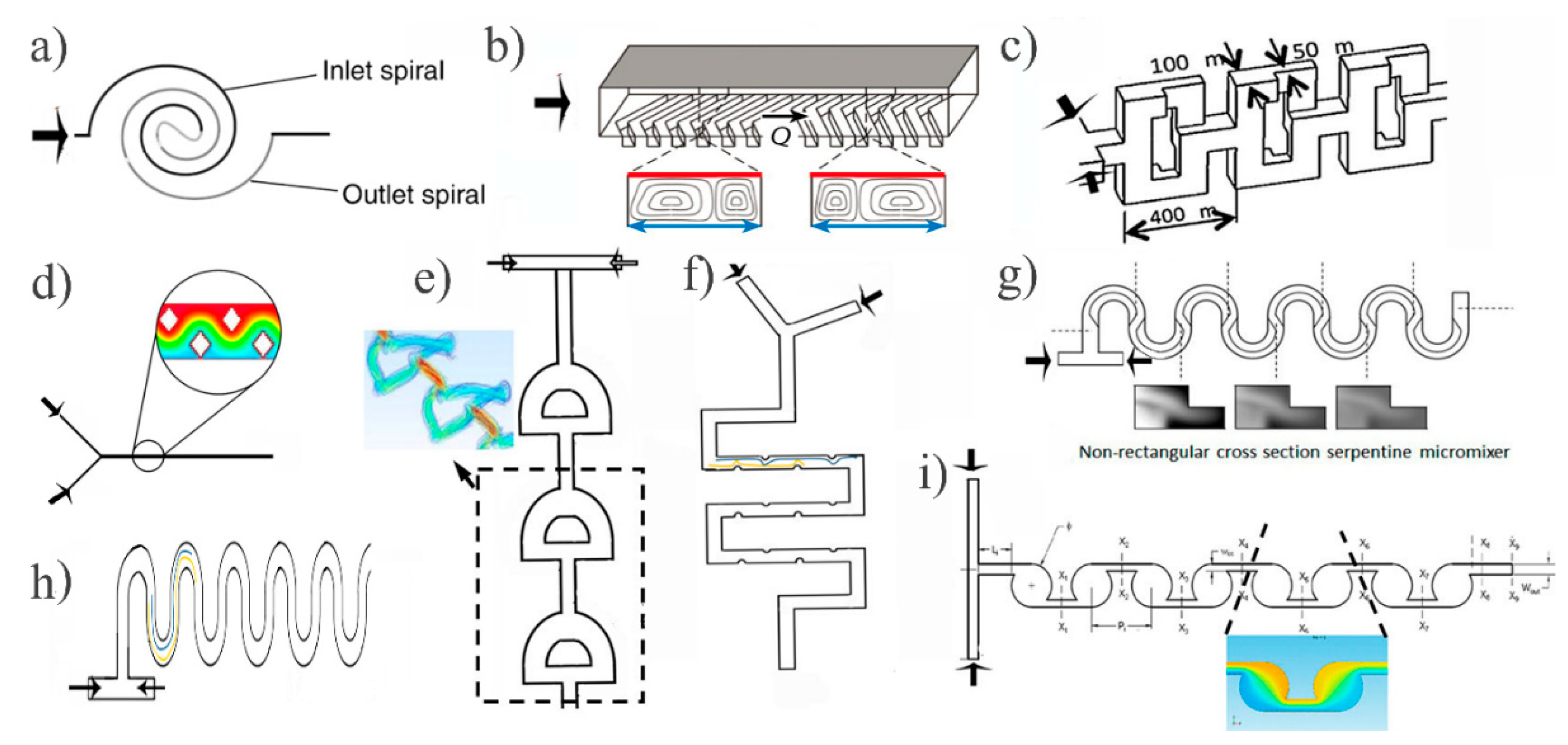
2.1. Passive Micromixers
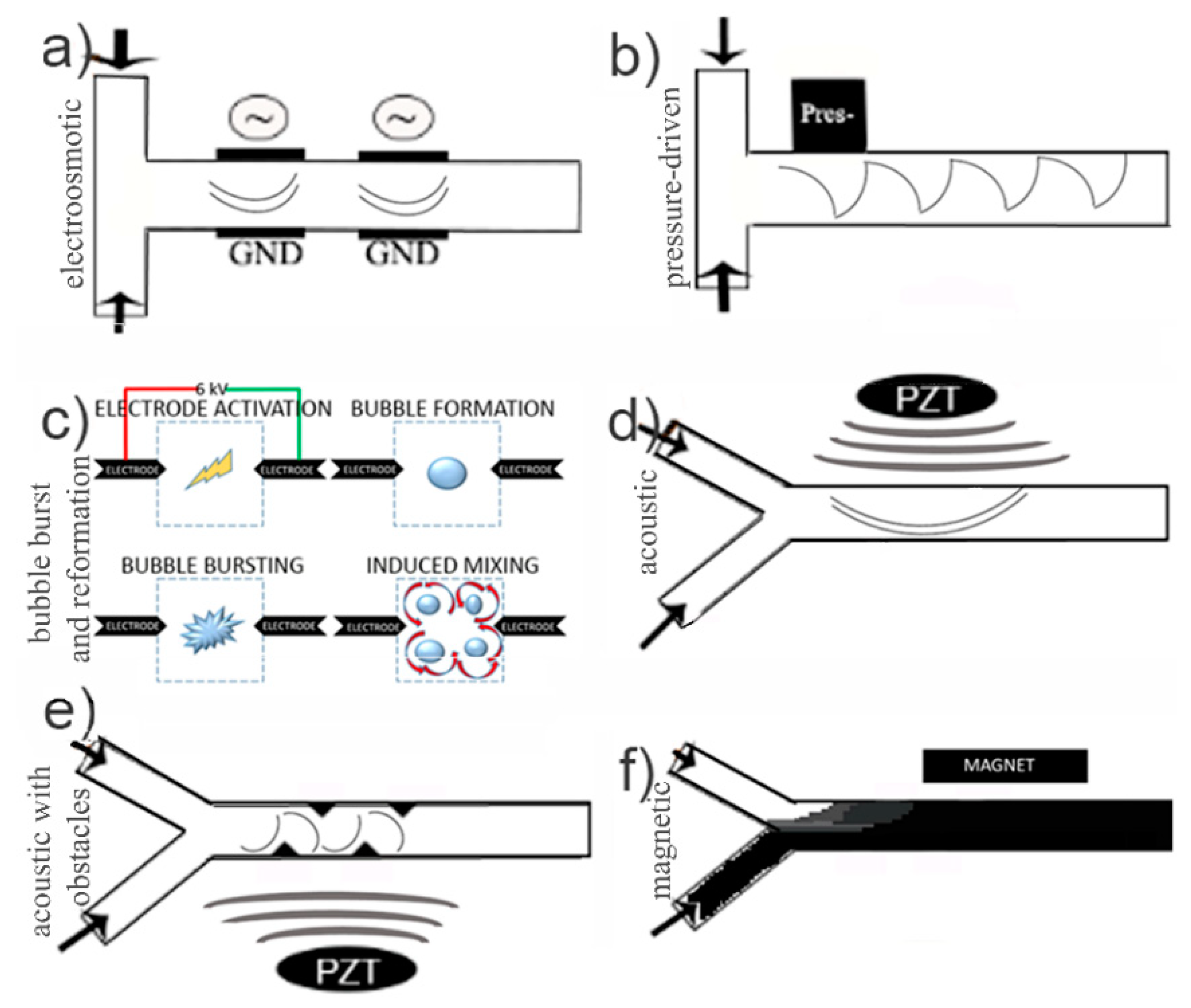
2.2. Active Micromixers
2.3. Active Micromixing Applications
3. Mechanisms of Magnetic Mixing
Magnetic Artificial Cilia
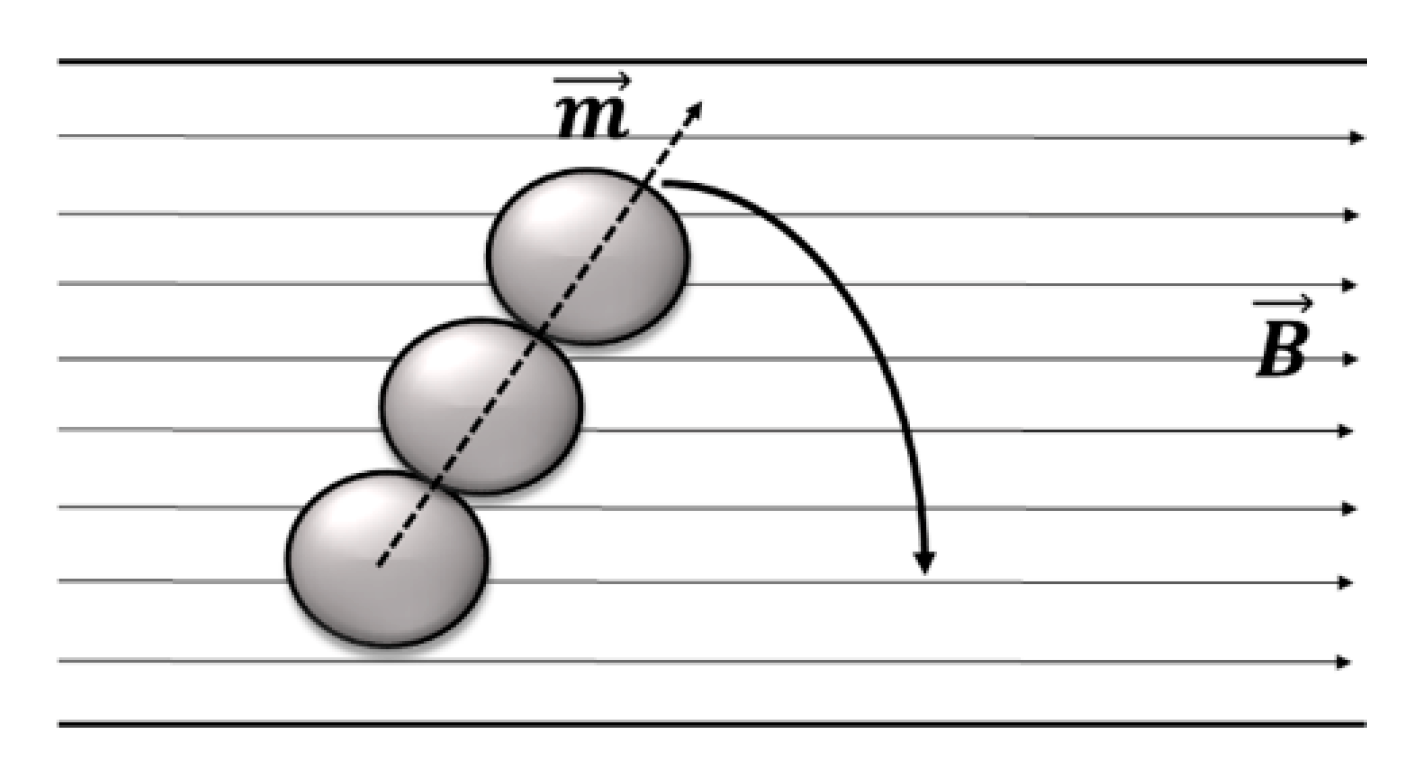
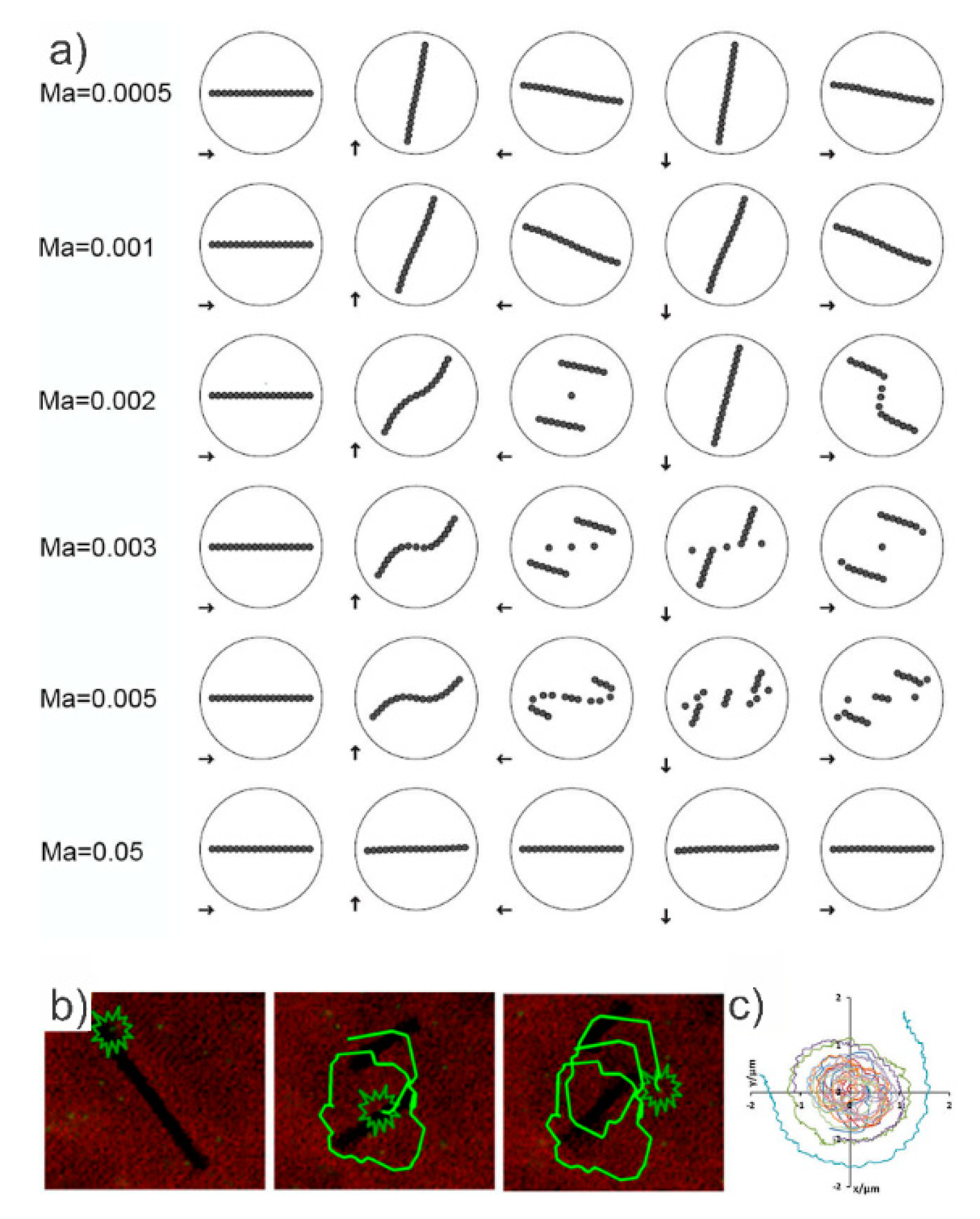
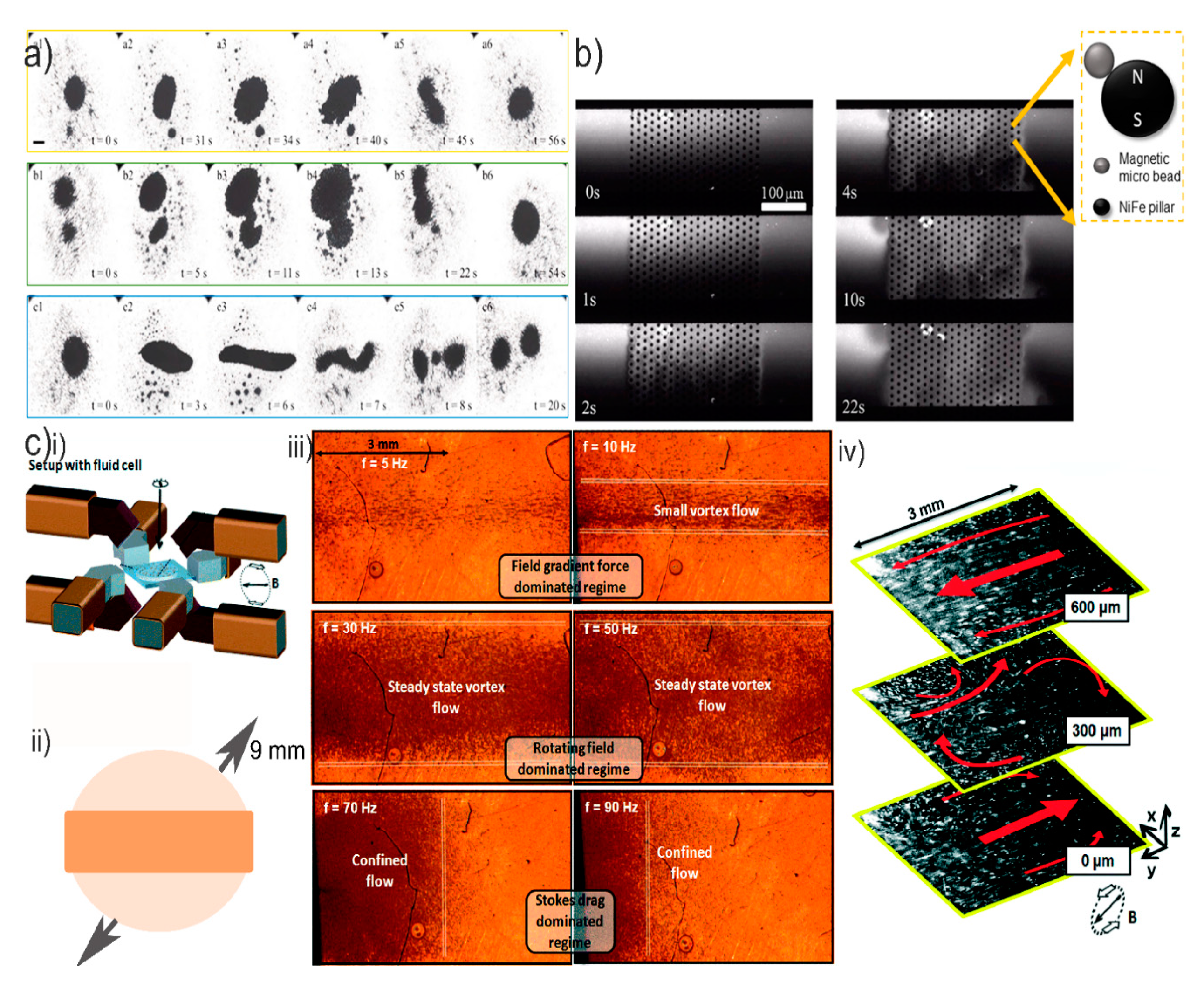
4. Magnetic Mixing Induced by Magnetic Bead Chains
Rotational Behavior of Magnetic Bead Chains and Effect on Mixing
5. Outlook on Magnetic Mixing
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Manz, A.; Graber, N.; Widmer, H.M. Miniaturized total chemical analysis systems: A novel concept for chemical sensing. Sens. Actuator B Chem. 1990, 1, 244–248. [Google Scholar] [CrossRef]
- Whitesides, G. Lab on a Chip 200th Issue. Lab Chip 2014, 14, 2880. [Google Scholar]
- Esmaeilsabzali, H.; Beischlag, T.V.; Cox, M.E.; Dechev, N.; Parameswaran, A.M.; Park, E.J. An integrated microfluidic chip for immunomagnetic detection and isolation of rare prostate cancer cells from blood. Biomed. Microdevices 2016, 18, 22. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhang, X.; Yao, Y.; Jing, W.; Liu, S.; Sui, G. A novel microfluidic module for rapid detection of airborne and waterborne pathogens. Sens. Actuator B Chem. 2018, 258, 1138–1145. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, D.; Feng, X.; Xu, Y.; Liu, B.-F. A microsecond microfluidic mixer for characterizing fast biochemical reactions. Talanta 2012, 88, 175–180. [Google Scholar] [CrossRef]
- Suh, Y.K.; Kang, S. A review on mixing in microfluidics. Micromachines 2010, 1, 82–111. [Google Scholar] [CrossRef]
- Stott, S.L.; Hsu, C.-H.C.-H.; Tsukrov, D.I.; Yu, M.; Miyamoto, D.T.; Waltman, B.A.; Rothenberg, S.M.; Shah, A.M.; Smas, M.E.; Korir, G.K.; et al. Isolation of circulating tumor cells using a microvortex-generating herringbone-chip. Proc. Natl. Acad. Sci. USA 2010, 107, 18392–18397. [Google Scholar] [CrossRef]
- Van Reenen, A.; De Jong, A.M.; Den Toonder, M.J. Integrated lab-on-chip biosensing systems based on magnetic particle actuation—A comprehensive review. Lab Chip 2014, 15, 9–15. [Google Scholar]
- Lee, C.Y.; Fu, L.M. Recent advances and applications of micromixers. Sens. Actuator B Chem. 2018, 259, 677–702. [Google Scholar] [CrossRef]
- Qiu, Z.; Zhao, L.; Weatherley, L. Process intensification technologies in continuous biodiesel production. Chem. Eng. Process. Process. Intensif. 2010, 49, 323–330. [Google Scholar] [CrossRef]
- Jeong, G.S.; Chung, S.; Kim, C.-B.; Lee, S.-H. Applications of micromixing technology. Analyst 2010, 135, 460. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Li, S.; Wu, Z.; Chen, Z. Diffusion and Mixing in Microfluidic Devices; Elsevier Inc.: Amsterdam, The Netherlands, 2019; ISBN 9780128126592. [Google Scholar]
- Capretto, L.; Cheng, W.; Hill, M.; Zhang, X. Micromixing within microfluidic devices. Micro Fluid Technol. Appl. 2011, 304, 27–68. [Google Scholar]
- Aref, H. Stirring by chaotic advection. J. Fluid Mech. 1984, 143, 1–21. [Google Scholar] [CrossRef]
- Chate, E.; Villermaux, E.; Chomaz, J.-M. Mixing: Chaos and Turbulence; Springer: Berlin, Germany, 1999; ISBN 9781461371274. [Google Scholar]
- Scherer, C. Figueiredo neto ferrofluids-properties and applications. Braz. J. Phys. 2005, 35, 718–728. [Google Scholar] [CrossRef]
- Kitenbergs, G.; Kaspars, E.; Perzynski, R.; Andrejs, C. Magnetic particle mixing with magnetic micro-convection for microfluidics. J. Magn. Magn. Mater. 2015, 380, 227–230. [Google Scholar] [CrossRef]
- Otieno, B.; Krauce, C.E.; Latus, A.; Chikkaveeraiah, B.V.; Faria, R.C.; Rusling, J.F. On-line protein capture on magnetic beads for ultrasensitive microfluidic immunoassays of cancer biomarkers. Biosens. Bioelectron. 2014, 53, 1883–1889. [Google Scholar] [CrossRef]
- Bohmer, N.; Demarmels, N.; Tsolaki, E.; Gerken, L.; Keevend, K.; Bertazzo, S.; Lattuada, M.; Herrmann, I.K. Removal of Cells from Body Fluids by Magnetic Separation in Batch and Continuous Mode: Influence of Bead Size, Concentration, and Contact Time. ACS Appl. Mater. Interfaces 2017, 9, 29571–29579. [Google Scholar] [CrossRef]
- Xiao, R.; Wang, C.W.; Zhu, A.N.; Long, F. Single functional magnetic-bead as universal biosensing platform for trace analyte detection using SERS-nanobioprobe. Biosens. Bioelectron. 2016, 79, 661–668. [Google Scholar] [CrossRef]
- Frohnmeyer, E.; Frisch, F.; Falke, S.; Betzel, C.; Fischer, M. Highly affine and selective aptamers against cholera toxin as capture elements in magnetic bead-based sandwich ELAA. J. Biotechnol. 2018, 269, 35–42. [Google Scholar] [CrossRef]
- Scherr, T.F.; Ryskoski, H.B.; Doyle, A.B.; Haselton, F.R. A two-magnet strategy for improved mixing and capture from biofluids. Biomicrofluidics 2016, 10, 1–15. [Google Scholar] [CrossRef]
- Armbrecht, L.; Dincer, C.; Kling, A.; Horak, J.; Kieninger, J.; Urban, G. Self-assembled magnetic bead chains for sensitivity enhancement of microfluidic electrochemical biosensor platforms. Lab Chip 2015, 15, 4314–4321. [Google Scholar] [CrossRef] [PubMed]
- Ottino, J.M.; Wiggins, S. Introduction: Mixing in microfluidics. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2004, 362, 923–935. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.T.; Hu, Y.C. Mixing of liquids using obstacles in Y-type microchannels. Tamkang J. Sci. Eng. 2010, 13, 385–394. [Google Scholar]
- Xu, Z.; Li, C.; Vadillo, D.; Ruan, X.; Fu, X. Numerical simulation on fluid mixing by effects of geometry in staggered oriented ridges micromixers. Sens. Actuator B Chem. 2011, 153, 284–292. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, S.K.; Kim, J.H.; Park, J.H. Separation of particles with bacterial size range using the control of sheath flow ratio in spiral microfluidic channel. Sens. Actuator. A Phys. 2019, 286, 211–219. [Google Scholar] [CrossRef]
- Chiu, P.L.; Chang, C.H.; Lin, Y.L.; Tsou, P.H.; Li, B.R. Rapid and safe isolation of human peripheral blood B and T lymphocytes through spiral microfluidic channels. Sci. Rep. 2019, 9, 8145. [Google Scholar] [CrossRef]
- Sudarsan, A.P.; Ugaz, V.M. Fluid mixing in planar spiral microchannels. Lab Chip 2006, 6, 74–82. [Google Scholar] [CrossRef]
- Duryodhan, V.S.; Chatterjee, R.; Govind Singh, S.; Agrawal, A. Mixing in planar spiral microchannel. Exp. Therm. Fluid Sci. 2017, 89, 119–127. [Google Scholar] [CrossRef]
- Al-Halhouli, A.; Alshare, A.; Mohsen, M.; Matar, M.; Dietzel, A.; Büttgenbach, S. Passive micromixers with interlocking semi-circle and omega-shaped modules: Experiments and simulations. Micromachines 2015, 6, 953–968. [Google Scholar] [CrossRef]
- Wang, H.; Iovenitti, P.; Harvey, E.; Masood, S. Optimizing layout of obstacles for enhanced mixing in microchannels. Smart Mater. Struct. 2002, 11, 662–667. [Google Scholar] [CrossRef]
- Bhagat, A.A.S.; Peterson, E.T.K.; Papautsky, I. A passive planar micromixer with obstructions for mixing at low Reynolds numbers. J. Micromech. Microeng. 2007, 17, 1017–1024. [Google Scholar] [CrossRef]
- Rahmannezhad, J.; Mirbozorgi, S.A. CFD analysis and RSM-based design optimization of novel grooved micromixers with obstructions. Int. J. Heat Mass Transf. 2019, 140, 483–497. [Google Scholar] [CrossRef]
- Franjione, A.J.G.; Ottino, J.M. Symmetry concepts for the geometric analysis of mixing flows. R. Soc. 1992, 338, 301–323. [Google Scholar]
- Hama, B.; Mahajan, G.; Fodor, P.S.; Kaufman, M.; Kothapalli, C.R. Evolution of mixing in a microfluidic reverse-staggered herringbone micromixer. Microfluid. Nanofluidics 2018, 22, 1–14. [Google Scholar] [CrossRef]
- Wangikar, S.S.; Patowari, P.K.; Misra, R.D. Numerical and experimental investigations on the performance of a serpentine microchannel with semicircular obstacles. Microsyst. Technol. 2018, 24, 3307–3320. [Google Scholar] [CrossRef]
- Gidde, R.R.; Pawar, P.M.; Ronge, B.P.; Misal, N.D.; Kapurkar, R.B.; Parkhe, A.K. Evaluation of the mixing performance in a planar passive micromixer with circular and square mixing chambers. Microsyst. Technol. 2018, 24, 2599–2610. [Google Scholar] [CrossRef]
- Sivashankar, S.; Agambayev, S.; Mashraei, Y.; Li, E.Q.; Thoroddsen, S.T.; Salama, K.N. A “twisted” microfluidic mixer suitable for a wide range of flow rate applications. Biomicrofluidics 2016, 10, 034120. [Google Scholar] [CrossRef]
- Huanming, X.; Jiawei, W.; Zhiping, W. A comparative discussion of different designs of passive micromixers: Specific sensitivities of mixing efficiency on Reynolds numbers and fluid properties. Microsyst. Technol. 2018, 24, 1253–1263. [Google Scholar] [CrossRef]
- Stroock, A.D.; McGraw, G.J. Investigation of the staggered herringbone mixer with a simple analytical model. Philos. Trans. R. Soc. Lond. 2004, 362, 971–986. [Google Scholar] [CrossRef]
- Ruijin, W.; Beiqi, L.; Dongdong, S.; Zefei, Z. Investigation on the splitting-merging passive micromixer based on Baker’s transformation. Sens. Actuator B Chem. 2017, 249, 395–404. [Google Scholar] [CrossRef]
- Clark, J.; Kaufman, M.; Fodor, P.S. Mixing enhancement in serpentine micromixers with a non-rectangular cross-section. Micromachines 2018, 9, 107. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, W.H. The Rheology of Saliva. J. Dent. Res. 1987, 66, 660–666. [Google Scholar] [CrossRef] [PubMed]
- Wood, A.W. Rheology of blood. Physiol. Biophys. Biomed. Eng. 2016, 49, 217–233. [Google Scholar]
- Brust, M.; Schaefer, C.; Doerr, R.; Pan, L.; Garcia, M.; Arratia, P.E.; Wagner, C. Rheology of human blood plasma: Viscoelastic versus Newtonian behavior. Phys. Rev. Lett. 2013, 110, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Ahmadian Yazdi, A.; Sadeghi, A.; Saidi, M.H. Rheology effects on cross-stream diffusion in a Y-shaped micromixer. Colloids Surfaces A Physicochem. Eng. Asp. 2014, 456, 296–306. [Google Scholar] [CrossRef]
- Lobasov, A.S.; Minakov, A.V. Analyzing mixing quality in a T-shaped micromixer for different fluids properties through numerical simulation. Chem. Eng. Process. Process. Intensif. 2018, 124, 11–23. [Google Scholar] [CrossRef]
- Vandenberg, A.; Wille, K. Evaluation of resonance acoustic mixing technology using ultra high performance concrete. Constr. Build. Mater. 2018, 164, 716–730. [Google Scholar] [CrossRef]
- Hin, S.; Paust, N.; Keller, M.; Rombach, M.; Strohmeier, O.; Zengerle, R.; Mitsakakis, K. Temperature change rate actuated bubble mixing for homogeneous rehydration of dry pre-stored reagents in centrifugal microfluidics. Lab Chip 2018, 18, 362–370. [Google Scholar] [CrossRef]
- Campos Domínguez, C.; Gamse, T. Process intensification by the use of micro devices for liquid fractionation with supercritical carbon dioxide. Chem. Eng. Res. Des. 2016, 108, 139–145. [Google Scholar] [CrossRef]
- Salmanzadeh, A.; Shafiee, H.; Davalos, R.V.; Stremler, M.A. Microfluidic mixing using contactless dielectrophoresis. Electrophoresis 2011, 32, 2569–2578. [Google Scholar] [CrossRef]
- Wuethrich, A.; Howard, C.B.; Trau, M. Geometric optimisation of electrohydrodynamic fluid flows for enhanced biosensing. Microchem. J. 2018, 137, 231–237. [Google Scholar] [CrossRef]
- Hayes, B.; Hayes, A.; Rolleston, M.; Ferreira, A.; Krisher, J. Pulsatory mixing of laminar flow using bubble-driven micropumps. In Proceedings of the International Mechanical Engineering Congress and Exposition, Pittsburgh, PA, USA, 9–15 November 2018; p. V007T09A091. [Google Scholar]
- Yang, F.; Kuang, C.; Zhao, W.; Wang, G. AC electrokinetic fast mixing in non-parallel microchannels. Chem. Eng. Commun. 2017, 204, 190–197. [Google Scholar] [CrossRef]
- Pelcová, M.; Řemínek, R.; Sandbaumhüter, F.A.; Mosher, R.A.; Glatz, Z.; Thormann, W. Simulation and experimental study of enzyme and reactant mixing in capillary electrophoresis based on-line methods. J. Chromatogr. A 2016, 1471, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, S.; Bafekr, H.; Valipour, M.S.; Esfahani, J.A. A review on the application, simulation, and experiment of the electrokinetic mixers. Chem. Eng. Process. Process. Intensif. 2018, 126, 108–122. [Google Scholar] [CrossRef]
- Nam, J.; Jang, W.S.; Lim, C.S. Micromixing using a conductive liquid-based focused surface acoustic wave (CL-FSAW). Sens. Actuator B Chem. 2018, 258, 991–997. [Google Scholar] [CrossRef]
- Nam, J.; Lim, C.S. Micromixing using swirling induced by three-dimensional dual surface acoustic waves (3D-dSAW). Sens. Actuators B Chem. 2018, 255, 3434–3440. [Google Scholar] [CrossRef]
- Charmiyan, M.; Moaveni, A. Optimization of an active electrokinetic micromixer based on the number and arrangement of microelectrodes. J. Appl. Fluid Mech. 2018, 11, 1531–1541. [Google Scholar]
- Kunti, G.; Das, S.S.; Pedireddi, V.M.; Bhattacharya, A.; Chakraborty, S. Paper based microfluidics for energy-efficient micromixing by harnessing the interplay of electrical and thermal fields. Analyst 2019, 7, 1800. [Google Scholar]
- Vafaie, R.H. A high-efficiency micromixing effect by pulsed AC electrothermal flow. Compel. Int. J. Comput. Math. Electr. Electron. Eng. 2018, 37, 418–431. [Google Scholar] [CrossRef]
- Lee, S.; Kim, J.; Wereley, S.T.; Kwon, J.S. Light-actuated electrothermal microfluidic flow for micro-mixing. J. Micromech. Microeng. 2019, 29, 017003. [Google Scholar] [CrossRef]
- Zhang, F.; Chen, H.; Chen, B.; Wu, J. Alternating current electrothermal micromixer with thin film resistive heaters. Adv. Mech. Eng. 2016, 8, 1–10. [Google Scholar] [CrossRef]
- Dong, X.; Zhang, L.; Fu, J. Laser-induced thermal bubble-mixing on a microfluidic platform for lab-on-a-chip applications. Adv. Mater. Res. 2012, 557–559, 2197–2201. [Google Scholar] [CrossRef]
- Meghdadi Isfahani, A.H.; Nasehi, R.; Shirani, E. Mixing enhancement in microchannels using thermo-viscous expansion by oscillating temperature wave. Chem. Eng. Process. Process. Intensif. 2018, 126, 123–131. [Google Scholar] [CrossRef]
- Wang, X.; Ma, X.; An, L.; Kong, X.; Xu, Z.; Wang, J. A pneumatic micromixer facilitating fluid mixing at a wide range flow rate for the preparation of quantum dots. Sci. China Chem. 2013, 56, 799–805. [Google Scholar] [CrossRef]
- Lee, Y.-K.; Deval, J.; Tabeling, P.; Ho, C.M. Chaotic mixing in electrokinetically and pressure driven micro flows. In Technical Digest. Proceedings of the 14th IEEE International Conference on Micro Electro Mechanical Systems (MEMS 2001)(Cat. No.01CH37090), Interlaken, Switzerland, 26 January 2001; IEEE: Piscataway, NJ, USA, 2001; pp. 483–486. [Google Scholar]
- Surdo, S.; Diaspro, A.; Duocastella, M. Micromixing with spark-generated cavitation bubbles. Microfluid. Nanofluidics 2017, 21, 1–10. [Google Scholar] [CrossRef]
- Shilton, R.J.; Yeo, L.Y.; Friend, J.R. Quantification of surface acoustic wave induced chaotic mixing-flows in microfluidic wells. Sensors Actuator. B Chem. 2011, 160, 1565–1572. [Google Scholar] [CrossRef]
- Cui, W.; Zhang, H.; Zhang, H.; Yang, Y.; He, M.; Qu, H.; Pang, W.; Zhang, D.; Duan, X. Localized ultrahigh frequency acoustic fields induced micro-vortices for submilliseconds microfluidic mixing. Appl. Phys. Lett. 2016, 109, 253503. [Google Scholar] [CrossRef]
- Huang, P.H.; Xie, Y.; Ahmed, D.; Nama, N.; Chao, Y.; Chan, C.Y.; Wang, L.; Huang, T.J. Acoustofluidic micromixer using acoustically oscillated sharp-edges. In Proceedings of the 17th International Conference on Miniaturized Systems for Chemistry and Life Sciences, µTAS, Freiburg, Germany, 27–31 October 2013; pp. 1227–1229. [Google Scholar]
- Ren, Y.; Liu, W.; Tao, Y.; Hui, M.; Wu, Q. On AC-field-induced nonlinear electroosmosis next to the sharp corner-field-singularity of leaky dielectric blocks and its application in on-chip micro-mixing. Micromachines 2018, 9, 102. [Google Scholar] [CrossRef]
- Nerem, R.M. Shear force and its effect on cell structure and function. ASGSB Bull. 1991, 4, 87–94. [Google Scholar]
- Iranmanesh, I.; Ohlin, M.; Ramachandraiah, H.; Ye, S.; Russom, A.; Wiklund, M. Acoustic micro-vortexing of fluids, particles and cells in disposable microfluidic chips. Biomed. Microdevices 2016, 18, 71. [Google Scholar] [CrossRef]
- Islam, M.S.; Aryasomayajula, A.; Selvaganapathy, P.R. A review on macroscale and microscale cell lysis methods. Micromachines 2017, 8, 83. [Google Scholar] [CrossRef]
- Wiklund, M. Acoustofluidics 12: Biocompatibility and cell viability in microfluidic acoustic resonators. Lab Chip 2012, 12, 2018–2028. [Google Scholar] [CrossRef] [PubMed]
- Hejazian, M.; Nguyen, N.T. A rapid magnetofluidic micromixer using diluted ferrofluid. Micromachines 2017, 8, 37. [Google Scholar] [CrossRef]
- Nouri, D.; Zabihi-Hesari, A.; Passandideh-Fard, M. Rapid mixing in micromixers using magnetic field. Sens. Actuator A Phys. 2017, 255, 79–86. [Google Scholar] [CrossRef]
- Ober, T.J.; Foresti, D.; Lewis, J.A. Active mixing of complex fluids at the microscale. Proc. Natl. Acad. Sci. USA 2015, 2015, 2–7. [Google Scholar] [CrossRef]
- Yang, R.-J.; Hou, H.-H.; Wang, Y.-N.; Fu, L.-M. Micro-magnetofluidics in microfluidic systems: A review. Sensors Actuators B Chem. 2016, 224, 1–15. [Google Scholar] [CrossRef]
- Fu, L.M.; Tsai, C.H.; Leong, K.P.; Wen, C.Y. Magnetic fluids rapid micromixer via ferrofluids. Phys. Procedia 2010, 9, 270–273. [Google Scholar] [CrossRef]
- Daniel, A.B.; Mohammad, S.A.; Miranda, M.A.; Aichele, C.P. Absorption and desorption mass transfer rates as a function of pressure and mixing in a simple hydrocarbon system. Chem. Eng. Res. Des. 2019, 144, 209–215. [Google Scholar] [CrossRef]
- Bastiaens, A.J.; Frimat, J.-P.; van Nunen, T.; Schurink, B.; Homburg, E.F.G.A.; Luttge, R. Advancing a MEMS-Based 3D Cell Culture System for in vitro Neuro-Electrophysiological Recordings. Front. Mech. Eng. 2018, 4, 1–10. [Google Scholar] [CrossRef]
- Frey, L.J.; Vorländer, D.; Rasch, D.; Ostsieker, H.; Müller, B.; Schulze, M.; Schenkendorf, R.; Mayr, T.; Grosch, J.H.; Krull, R. Novel electrodynamic oscillation technique enables enhanced mass transfer and mixing for cultivation in micro-bioreactor. Biotechnol. Prog. 2019, 1–12. [Google Scholar] [CrossRef]
- Wen, Y.; Zang, R.; Zhang, X.; Yang, S.T. A 24-microwell plate with improved mixing and scalable performance for high throughput cell cultures. Process. Biochem. 2012, 47, 612–618. [Google Scholar] [CrossRef]
- Dong, J.; Ueda, H. ELISA-type assays of trace biomarkers using microfluidic methods. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Wang, C.C.; Lei, K.F. Bubble-driven mixer integrated with a microfluidic bead-based ELISA for rapid bladder cancer biomarker detection. Biomed. Microdevices 2014, 16, 199–207. [Google Scholar] [CrossRef] [PubMed]
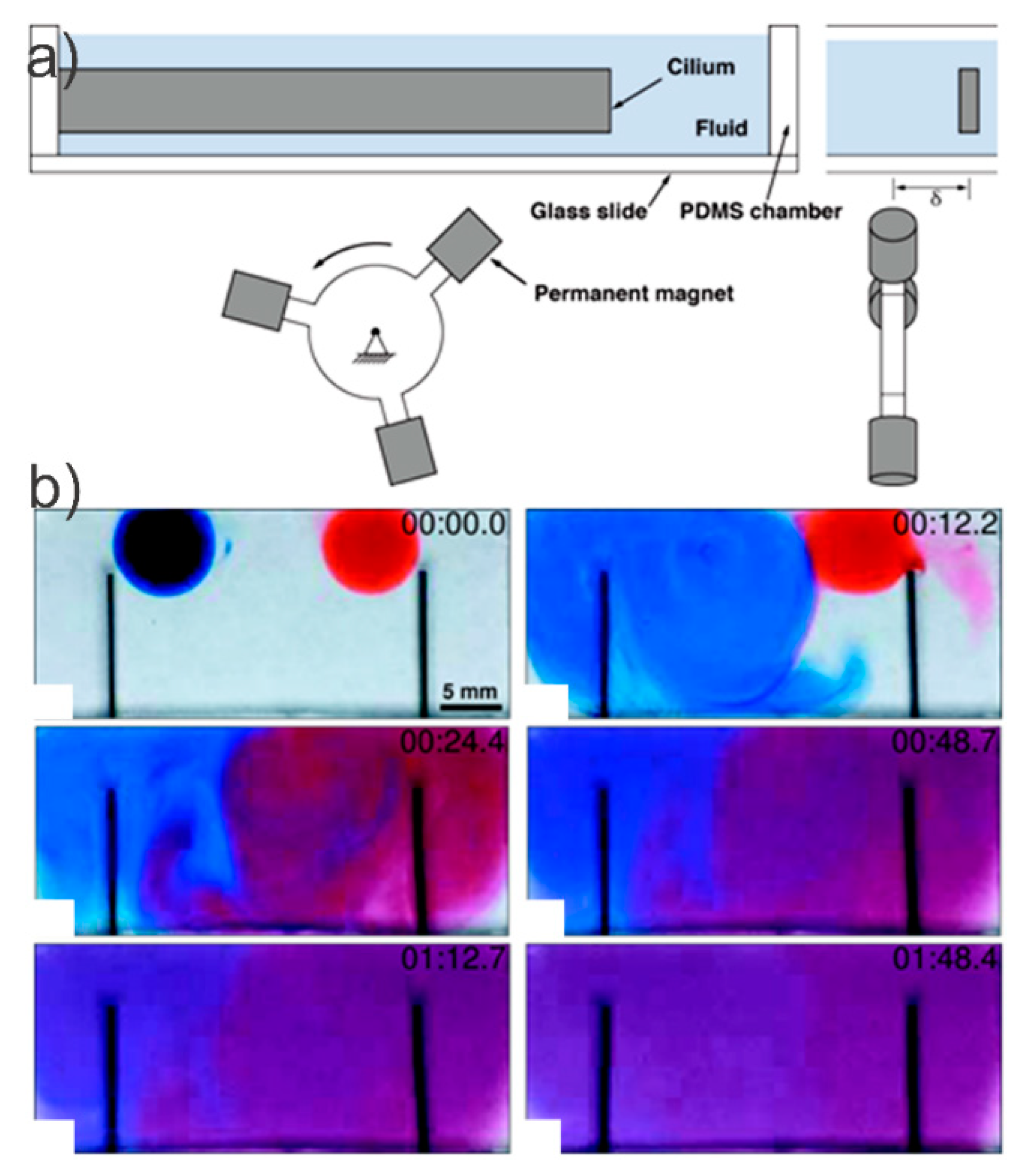
- Saberi, A.; Zhang, S.; van den Bersselaar, C.; Kandail, H.; den Toonder, J.M.J.; Kurniawan, N.A. A stirring system using suspended magnetically-actuated pillars for controlled cell clustering. Soft Matter 2019, 15, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Orbay, S.; Ozcelik, A.; Bachman, H.; Huang, T.J. Acoustic actuation of in situ fabricated artificial cilia. J. Micromech. Microeng. 2018, 28, 025012. [Google Scholar] [CrossRef]
- Den Toonder, J.M.J.; Onck, P.R. Microfluidic manipulation with artificial/bioinspired cilia. Trends Biotechnol. 2013, 31, 85–91. [Google Scholar] [CrossRef]
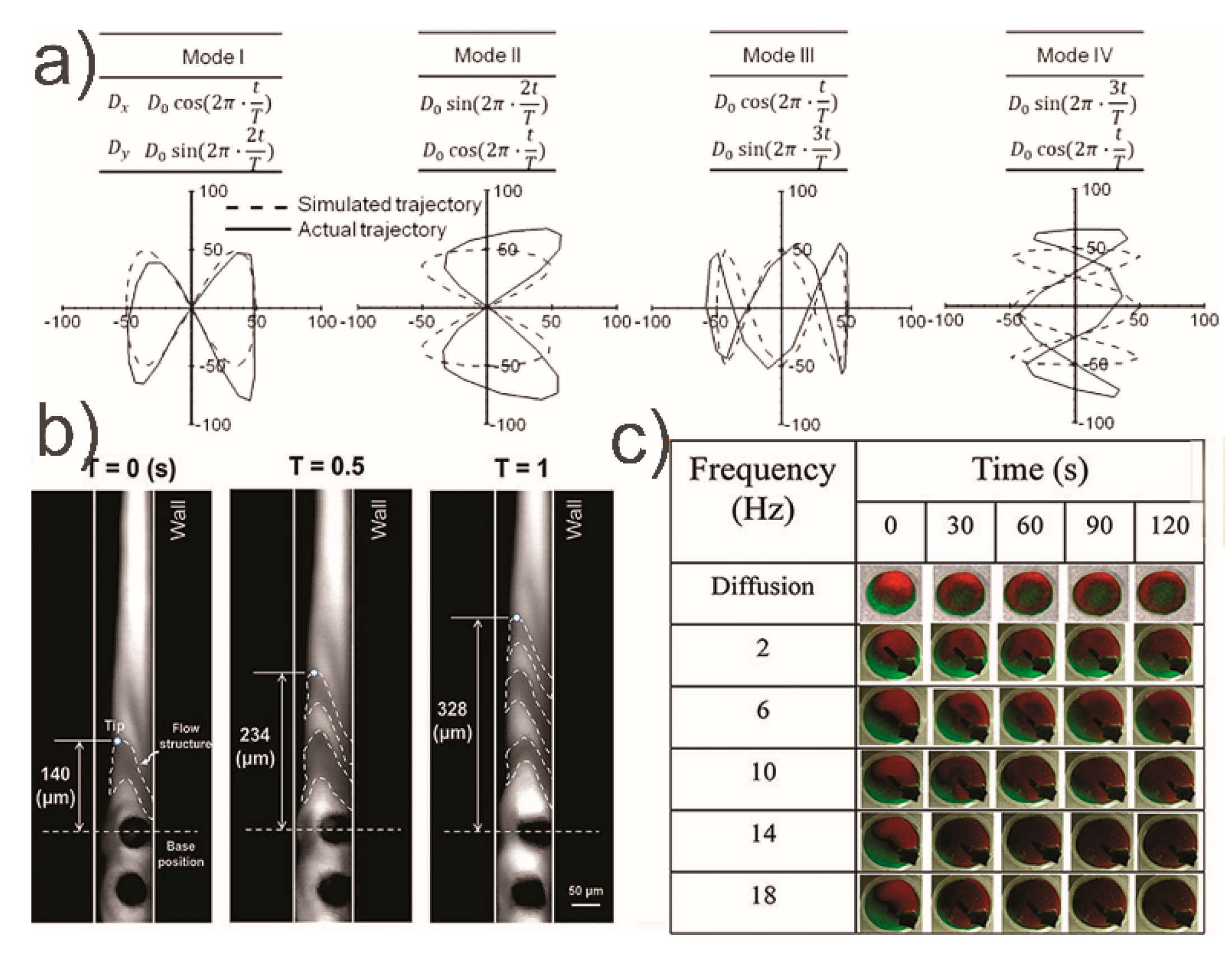
- Khaderi, S.N.; Craus, C.B.; Hussong, J.; Schorr, N.; Belardi, J.; Westerweel, J.; Prucker, O.; Rühe, J.; den Toonder, J.M.J.; Onck, P.R. Magnetically-actuated artificial cilia for microfluidic propulsion. Lab Chip 2011, 11, 2002. [Google Scholar] [CrossRef]
- Downton, M.T.; Stark, H. Beating kinematics of magnetically actuated cilia. Europhys. Lett. 2009, 85, 44002. [Google Scholar] [CrossRef]
- Vilfan, M.; Potocnik, A.; Kavcic, B.; Osterman, N.; Poberaj, I.; Vilfan, A.; Babic, D. Self-assembled artificial cilia. Proc. Natl. Acad. Sci. USA 2010, 107, 1844–1847. [Google Scholar] [CrossRef]
- Shields, A.R.; Fiser, B.L.; Evans, B.A.; Falvo, M.R.; Washburn, S.; Superfine, R. Biomimetic cilia arrays generate simultaneous pumping and mixing regimes. Proc. Natl. Acad. Sci. USA 2010, 107, 15670–15675. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Chen, C.-Y.; Lin, C.-Y.; Hu, Y.-T. Magnetically actuated artificial cilia for optimum mixing performance in microfluidics. Lab Chip 2013, 13, 2834. [Google Scholar] [CrossRef] [PubMed]
- Khaderi, S.; Hussong, J.; Westerweel, J.; den Toonder, J.; Onck, P. Fluid propulsion using magnetically-actuated artificial cilia–experiments and simulations. RSC Adv. 2013, 3, 12735. [Google Scholar] [CrossRef]
- Brennen, C.; Winet, H. Fluid mechanics of propulsion by cilia and flagella. Annu. Rev. Fluid Mech. 1977, 9, 339–398. [Google Scholar] [CrossRef]
- Khaderi, S.N.; Baltussen, M.G.H.M.; Anderson, P.D.; Den Toonder, J.M.J.; Onck, P.R. Breaking of symmetry in microfluidic propulsion driven by artificial cilia. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2010, 82, 027302. [Google Scholar] [CrossRef] [PubMed]
- Mayne, R.; den Toonder, J.M.J. (Eds.) Atlas of Cilia Bioengineering and Biocomputing Google Books; River Publishers: Gistrup, Denmark, 2018; ISBN 978-87-7022-002-06. [Google Scholar]
- Chen, C.Y.; Hsu, C.C.; Mani, K.; Panigrahi, B. Hydrodynamic influences of artificial cilia beating behaviors on micromixing. Chem. Eng. Process. Process. Intensif. 2016, 99, 33–40. [Google Scholar] [CrossRef]
- Wu, Y.-A.; Panigrahi, B.; Lu, Y.-H.; Chen, C.-Y. An integrated artificial cilia based microfluidic device for micropumping and micromixing applications. Micromachines 2017, 8, 260. [Google Scholar] [CrossRef]
- Banka, N.; Ng, Y.L.; Devasia, S. Individually Controllable Magnetic Cilia: Mixing Application. J. Med. Devices 2017, 11, 031003. [Google Scholar] [CrossRef]
- Liu, F.; Zhang, J.; Alici, G.; Yan, S.; Mutlu, R.; Li, W.; Yan, T. An inverted micro-mixer based on a magnetically-actuated cilium made of Fe doped PDMS. Smart Mater. Struct. 2016, 25, 095049. [Google Scholar] [CrossRef]
- Wang, Y.; den Toonder, J.; Cardinaels, R.; Anderson, P. A continuous roll-pulling approach for the fabrication of magnetic artificial cilia with microfluidic pumping capability. Lab Chip 2016, 16, 2277–2286. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, Y.; Lavrijsen, R.; Onck, P.R.; den Toonder, J.M.J. Versatile microfluidic flow generated by moulded magnetic artificial cilia. Sens. Actuators B Chem. 2018, 263, 614–624. [Google Scholar] [CrossRef]
- Wittbracht, F.; Weddemann, A.; Eickenberg, B.; Zahn, M.; Hu, A. Dipolar interaction in rotating magnetic fields Enhanced fluid mixing and separation of magnetic bead agglomerates based on dipolar interaction in rotating magnetic fields. Appl. Phys. Lett. 2012, 100, 123507. [Google Scholar] [CrossRef]
- Gao, Y.; van Reenen, A.; Hulsen, M.A.; de Jong, A.M.; Prins, M.W.J.; den Toonder, J.M.J.; Gijs, M.A.M.; Pamme, N.; Sherman, D.J.; Kenanova, V.E.; et al. Disaggregation of microparticle clusters by induced magnetic dipole–dipole repulsion near a surface. Lab Chip 2013, 13, 1394. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.; Han, X.; Li, L. An active microfluidic mixer utilizing a hybrid gradient magnetic field. Int. J. Appl. Electromagn. Mech. 2015, 47, 583–592. [Google Scholar] [CrossRef]
- Gon, T.; Gao, Y.; Hulsen, M.A.; Den Toonder, J.M.J.; Anderson, P.D. Direct simulation of the dynamics of two spherical particles actuated magnetically in a viscous fluid. Comput. Fluids 2013, 86, 569–581. [Google Scholar]
- Petousis, I.; Homburg, E.; Derks, R.; Dietzel, A. Transient behaviour of magnetic micro-bead chains rotating in a fluid by external fields. Lab Chip 2007, 12, 1746–1751. [Google Scholar] [CrossRef]
- Kang, T.G.; Hulsen, M.A.; Anderson, P.D.; Den Toonder, J.M.J.; Meijer, H.E.H. Chaotic mixing induced by a magnetic chain in a rotating magnetic field. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2007, 76, 1–11. [Google Scholar] [CrossRef]
- Kuei, S.; Garza, B.; Biswal, S.L. From strings to coils: Rotational dynamics of DNA-linked colloidal chains. Phys. Rev. Fluids 2017, 2, 1–11. [Google Scholar] [CrossRef]
- Biswal, S.L.; Gast, A.P. Mechanics of semiflexible chains formed by poly(ethylene glycol)-linked paramagnetic particles. Phys. Rev. E Stat. Phys. Plasmas Fluids Relat. Interdiscip. Top. 2003, 68, 9. [Google Scholar] [CrossRef]
- Benhal, P.; Broda, A.; Najafali, D.; Malik, P.; Mohammed, A.; Ramaswamy, B.; Depireux, D.A.; Shimoji, M.; Shukoor, M.; Shapiro, B. On-chip testing of the speed of magnetic nano-and micro-particles under a calibrated magnetic gradient. J. Magn. Magn. Mater. 2019, 474, 187–198. [Google Scholar] [CrossRef]
- Gao, Y.; Van Reenen, A.; Hulsen, M.A.; De Jong, A.M.; Prins, M.W.J.; Den Toonder, J.M.J. Chaotic fluid mixing by alternating microparticle topologies to enhance biochemical reactions. Microfluid. Nanofluidics 2014, 265–274. [Google Scholar] [CrossRef]
- Li, Y.H.; Lin, H.C.; Chen, C.Y. Steering of magnetic micro-swimmers. IEEE Trans. Magn. 2013, 49, 4120–4123. [Google Scholar] [CrossRef]
- Ido, Y.; Li, Y.H.; Tsutsumi, H.; Sumiyoshi, H.; Chen, C.Y. Magnetic microchains and microswimmers in an oscillating magnetic field. Biomicrofluidics 2016, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Byrom, J.; Han, P.; Savory, M.; Biswal, S.L. Directing assembly of DNA-coated colloids with magnetic fields to generate rigid, semiflexible, and flexible chains. Langmuir 2014, 30, 9045–9052. [Google Scholar] [CrossRef]
- Yu, J.; Yang, L.; Zhang, L. Pattern generation and motion control of a vortex-like paramagnetic nanoparticle swarm. Int. J. Rob. Res. 2018, 37, 912–930. [Google Scholar] [CrossRef]
- Regtmeier, A.; Wittbracht, F.; Rempel, T.; Mill, N.; Peter, M.; Weddemann, A.; Mattay, J.; Hütten, A. Uniform growth of clusters of magnetic nanoparticles in a rotating magnetic field. J. Nanopart. Res. 2012, 14, 1061. [Google Scholar] [CrossRef][Green Version]
- Owen, D.; Ballard, M.; Alexeev, A.; Hesketh, P.J. Rapid microfluidic mixing via rotating magnetic microbeads. Sens. Actuator A Phys. 2016, 251, 84–91. [Google Scholar] [CrossRef]
- Gao, Y.; Beerens, J.; Van Reenen, A.; Hulsen, M.A.; De Jong, A.M.; Prins, W.J.; Den Toonder, J.M.J. Strong vortical flows generated by the collective motion of magnetic particle chains rotating in a fluid cell. Lab Chip 2015, 15, 351–360. [Google Scholar] [CrossRef]
- Chong, W.H.; Huang, Y.; Wong, T.N.; Ooi, K.T.; Zhu, G.P. Magnetic nanorobots, generating vortexes inside nanoliter droplets for effective mixing. Adv. Mater. Technol. 2018, 3, 1700312. [Google Scholar] [CrossRef]
- Yu, J.; Wang, B.; Du, X.; Wang, Q.; Zhang, L. Ultra-extensible ribbon-like magnetic microswarm. Nat. Commun. 2018, 9, 3260. [Google Scholar] [CrossRef]
- Elgeti, J.; Winkler, R.G.; Gompper, G. Physics of microswimmers-single particle motion and collective behavior: A review. Rep. Prog. Phys. 2015, 78, 056601. [Google Scholar] [CrossRef]
- Guzmán-Lastra, F.; Kaiser, A.; Löwen, H. Fission and fusion scenarios for magnetic microswimmer clusters. Nat. Commun. 2016, 7, 13519. [Google Scholar] [CrossRef] [PubMed]
- Mairhofer, J.; Roppert, K.; Ertl, P.; Mairhofer, J.; Roppert, K.; Ertl, P. Microfluidic systems for pathogen sensing: A review. Sensors 2009, 9, 4804–4823. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, M.; Veres, T.; Tabrizian, M. Enzymatically-generated fluorescent detection in micro-channels with internal magnetic mixing for the development of parallel microfluidic ELISA. Lab Chip 2006, 6, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Zhi, S.; Sun, X.; Feng, Z.; Lei, C.; Zhou, Y. An innovative micro magnetic separator based on 3D micro-copper-coil exciting soft magnetic tips and FeNi wires for bio-target sorting. Microfluid. Nanofluid. 2019, 23, 43. [Google Scholar] [CrossRef]
- Rabehi, A.; Garlan, B.; Achtsnicht, S.; Krause, H.J.; Offenhäusser, A.; Ngo, K.; Neveu, S.; Graff-Dubois, S.; Kokabi, H. Magnetic detection structure for lab-on-chip applications based on the frequency mixing technique. Sensors 2018, 18, 1747. [Google Scholar] [CrossRef] [PubMed]
- Dong, T.; Su, Q.; Yang, Z.; Zhang, Y.; Egeland, E.B.; Gu, D.D.; Calabrese, P.; Kapiris, M.J.; Karlsen, F.; Minh, N.T.; et al. A smart fully integrated micromachined separator with soft magnetic micro-pillar arrays for cell isolation. J. Micromech. Microeng. 2010, 20, 115021. [Google Scholar] [CrossRef]
- Gooneratne, C.P.; Kodzius, R.; Li, F.; Foulds, I.G.; Kosel, J. On-chip magnetic bead manipulation and detection using a magnetoresistive sensor-based micro-chip: Design considerations and experimental characterization. Sensors 2016, 16, 1369. [Google Scholar] [CrossRef]
- Smistrup, K.; Hansen, O.; Bruus, H.; Hansen, M.F. Magnetic separation in microfluidic systems using microfabricated electromagnets - Experiments and simulations. J. Magn. Magn. Mater. 2005, 293, 597–604. [Google Scholar] [CrossRef]
- Royet, D.; Hériveaux, Y.; Marchalot, J.; Scorretti, R.; Dias, A.; Dempsey, N.M.; Bon, M.; Simonet, P.; Frénéa-robin, M. Using injection molding and reversible bonding for easy fabrication of magnetic cell trapping and sorting devices. J. Magn. Magn. Mater. 2017, 427, 306–313. [Google Scholar] [CrossRef]
- Van Pelt, S.; Frijns, A.; den Toonder, J. Microfluidic magnetic bead conveyor belt. Lab Chip 2017, 17, 3826–3840. [Google Scholar] [CrossRef]
- Liu, M.; Han, X.; Cao, Q.; Li, L. Performance analysis of a microfluidic mixer based on high gradient magnetic separation principles. J. Phys. D Appl. Phys. 2017, 50, aa7eb7. [Google Scholar] [CrossRef]
- Vemulkar, T.; Mansell, R.; Petit, D.C.M.C.; Cowburn, R.P.; Lesniak, M.S. Highly tunable perpendicularly magnetized synthetic antiferromagnets for biotechnology applications. Appl. Phys. Lett. 2015, 107, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, Y.; Onck, P.R.; den Toonder, J.M.J. Removal of Microparticles by Ciliated Surfaces—an Experimental Study. Adv. Funct. Mater. 2019, 29, 1806434. [Google Scholar] [CrossRef]
- Hernández-Neuta, I.; Pereiro, I.; Ahlford, A.; Ferraro, D.; Zhang, Q.; Viovy, J.L.; Descroix, S.; Nilsson, M. Microfluidic magnetic fluidized bed for DNA analysis in continuous flow mode. Biosens. Bioelectron. 2018, 102, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Hardt, S.; Drese, K.S.; Hessel, V.; Schönfeld, F. Passive micromixers for applications in the microreactor and μTAS fields. Microfluid. Nanofluidics 2005, 1, 108–118. [Google Scholar] [CrossRef]
| Mixing Mechanisms | Electro Osmotic | Pressure-Driven | Temperature-Driven | Acoustic | Cavitation Bubbles | Magnetofluids |
|---|---|---|---|---|---|---|
| Easy to fabricate | N | N | Y | N | N | Y |
| Low cost fabrication | N | N | Y | N | N | Y |
| Applicable to very low Re numbers | Y | Y | Y | Y | Y | Y |
| Applicable for stagnant flows | Y | Y | N | Y | Y | Y |
| Compatible with biological components | N | Y | N | N | N | Y |
| Sample preparation needed | Y | N | N | N | Y | N |
| Readout visibility | Y | Y | Y | Y | Y | N |
| MAC for Fluid Mixing | |||||
|---|---|---|---|---|---|
| Reference | Mixing Mechanism | Experimental (E)/ Simulation (S) | Overall Fluidic Dimensions | Re Number | Mixing Efficiency (%) |
| [101] | MAC | E and S | H = 400 µm, W = 114 µm, L = 152 µm | 3.18 × 10−3 | 91 |
| [102] | MAC | E and S | H = 500 µm, W = 100 µm, L = 500 µm | 1.16 × 10−3 | 84 |
| [103] | MAC | E | H = 4 mm, D = 5 mm | microwell | 80 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shanko, E.-S.; van de Burgt, Y.; Anderson, P.D.; den Toonder, J.M.J. Microfluidic Magnetic Mixing at Low Reynolds Numbers and in Stagnant Fluids. Micromachines 2019, 10, 731. https://doi.org/10.3390/mi10110731
Shanko E-S, van de Burgt Y, Anderson PD, den Toonder JMJ. Microfluidic Magnetic Mixing at Low Reynolds Numbers and in Stagnant Fluids. Micromachines. 2019; 10(11):731. https://doi.org/10.3390/mi10110731
Chicago/Turabian StyleShanko, Eriola-Sophia, Yoeri van de Burgt, Patrick D. Anderson, and Jaap M. J. den Toonder. 2019. "Microfluidic Magnetic Mixing at Low Reynolds Numbers and in Stagnant Fluids" Micromachines 10, no. 11: 731. https://doi.org/10.3390/mi10110731
APA StyleShanko, E.-S., van de Burgt, Y., Anderson, P. D., & den Toonder, J. M. J. (2019). Microfluidic Magnetic Mixing at Low Reynolds Numbers and in Stagnant Fluids. Micromachines, 10(11), 731. https://doi.org/10.3390/mi10110731







