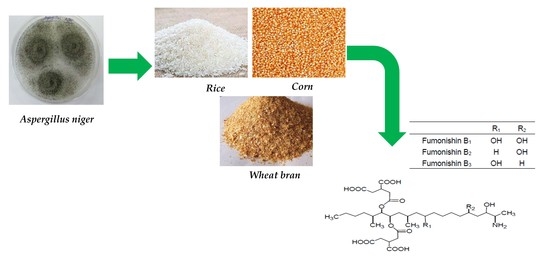
Dynamic Fumonisin B2 Production by Aspergillus niger Intented Used in Food Industry in China
Abstract
:1. Introduction
2. Results
2.1. Time Course of FB2 Production by A. niger
2.2. Effect of Media on Dynamic Production of FB2 for Strains of A. niger Intended Used in Different Functions
2.3. Distribution of FB2 Produced by A. niger Intended Used in Chinese Food Industry
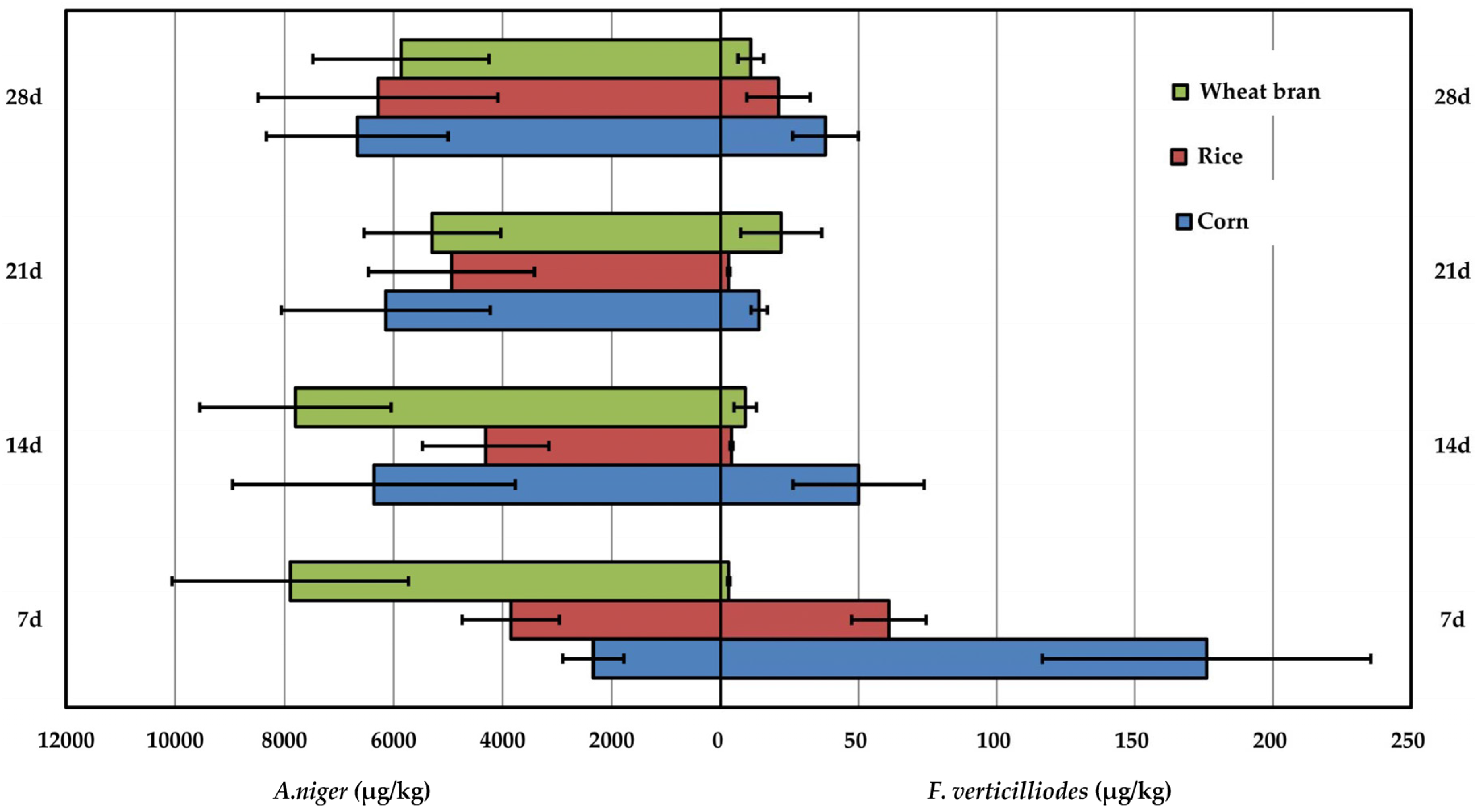
2.4. Comparison of Fumonision Production Produced by A. niger and F. verticilliodes
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Fungal Strains
4.3. Fumonisins Production
4.4. Fumonisin Extraction and Analysis
4.5. UPLC Conditions
4.6. MS/MS Conditions
4.7. Preparation of Standard Solutions
4.8. Method Validation
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gelderblom, W.C.; Marasas, W.F.O.; Steyn, P.; Wessels, P.L. Structure elucidation of fusarin C, a mutagen produced by Fusarium moniliforme. J. Chem. Soc. Chem. Commun. 1984, 2, 122–124. [Google Scholar] [CrossRef]
- Ross, P.F.; Rice, L.G.; Osweiler, G.D.; Nelson, P.E.; Richard, J.L.; Wilson, T.M. A review and update of animal toxicoses associated with fumonisin-contaminated feeds and production of fumonisins by Fusarium isolates. Mycopathologia 1992, 117, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Thiel, P.G.; Shephard, G.S.; Sydenham, E.W.; Marasas, W.F.O.; Nelson, P.E.; Wilson, T.M. Levels of fumonisins B1 and B2 in feeds associated with confirmed cases of equine leukoencephalomalacia. J. Agric. Food Chem. 1991, 39, 109–111. [Google Scholar] [CrossRef]
- Gelderblom, W.C.A.; Jaskiewicz, K.; Marasas, W.F.O.; Thiel, P.G.; Horak, R.M.; Vleggaar, R.; Kriek, N.P. Fumonisins-novel mycotoxins with cancer-promoting activity produced by Fusarium moniliforme. Appl. Environ. Microbiol. 1988, 54, 1806–1811. [Google Scholar] [PubMed]
- Yoshizawa, T.; Yamashita, A.; Luo, Y. Fumonisin occurrence in corn from high- and low-risk areas for human esophageal cancer in China. Appl. Environ. Microbiol. 1994, 60, 1626–1629. [Google Scholar] [PubMed]
- Wang, H.; Wei, H.; Ma, J.; Luo, X. The fumonisin B1 content in corn from North China, a high-risk area of esophageal cancer. J. Environ. Pathol. Toxicol. Oncol. 2000, 19, 139–141. [Google Scholar] [PubMed]
- Desjardins, A.E.; Manandhar, H.K.; Plattner, R.D.; Manandhar, G.G.; Poling, S.M.; Margos, C.M. Fusarium species from Nepalese rice and production of mycotoxin and gibberllic acid by selected species. Appl. Environ. Microbiol. 2000, 66, 1020–1025. [Google Scholar] [CrossRef] [PubMed]
- Fotso, J.; Leslie, J.F.; Smith, S. Production of beauvericin, moniliformin, fusaproliferin, and fumonisins B1, B2, and B3 by fifteen ex-type strains of Fusarium species. Appl. Environ. Microbiol. 2002, 68, 5195–5197. [Google Scholar] [CrossRef] [PubMed]
- Logrieco, A.; Ferracane, R.; Haidukowsky, M.; Cozzi, G.; Visconti, A.; Ritieni, A. Fumonisin B2 production by Aspergillus niger from grapes and natural occurrence in must. Food Addit. Contam. 2009, 26, 1495–1500. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, P.B.; Mogensen, J.M.; Larsen, T.O.; Nielson, K.F. Occurrence of fumonisins B2 and B4 in retail raisins. J. Agric. Food Chem. 2010, 59, 772–776. [Google Scholar] [CrossRef] [PubMed]
- Abrunhosa, L.; Calado, T.; Venâncio, A. Incidence of Fumonisin B2 Production by Aspergillus niger in Portuguese Wine Regions. J. Agric. Food Chem. 2011, 59, 7514–7518. [Google Scholar] [CrossRef] [PubMed]
- Noonim, P.; Mahakarnchanakul, W.; Nielsen, K.F.; Frisvad, J.C.; Samson, R.A. Fumonisin B2 production by Aspergillus niger in Thai coffee beans. Food Addit. Contam. 2009, 26, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Logrieco, A.F.; Haidukowski, M.; Susca, A.; Mulè, G.; Munkvold, G.P.; Moretti, A. Aspergillus section Nigri as contributor of fumonisin B(2) contamination in maize. Food Addit. Contam. Part A 2014, 31, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Susca, A.; Moretti, A.; Stea, G.; Villani, A.; Haidukowski, M.; Logrieco, A.; Munkvold, G. Comparison of species composition and fumonisin production in Aspergillus section Nigri populations in maize kernels from USA and Italy. Int. J. Food Microbiol. 2014, 188, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Frisvad, J.C.; Smedsgaard, J.; Samson, R.A.; Larsen, T.O.; Thrane, U. Fumonisin B2 production by Aspergillus niger. J. Agric. Food Chem. 2007, 55, 9727–9732. [Google Scholar] [CrossRef] [PubMed]
- Frisvad, J.C.; Larsen, T.O.; Thrane, U.; Meijer, M.; Varga, J.; Samon, R.A.; Nielsen, K. Fumonisin and ochratoxin production in industrial Aspergillus niger strains. PLoS ONE 2011, 6, e23496. [Google Scholar] [CrossRef] [PubMed]
- Alberts, J.F.; Gelderblom, W.C.A.; Thiel, P.G.; Marasas, W.F.O.; van Schalkwyk, D.J.; Behrend, Y. Effects of temperature and incubation period on production of fumonisin B1 by Fusarium moniliforme. Appl. Environ. Microbiol. 1990, 56, 1729–1733. [Google Scholar] [PubMed]
- Pel, H.J.; de Winde, J.H.; Archer, D.B.; Dyer, P.S.; Hofmann, G.; Schaap, P.J.; Turner, G.; de Vries, R.P.; Albang, R.; Albermann, K.; et al. Genome sequencing and analysis of the versatile cell factory Aspergillus niger. Nat. Biotechnol. 2007, 25, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Mogensen, J.M.; Nielsen, K.F.; Samson, R.A.; Frisvad, J.C.; Thrane, U. Effect of temperature and water activity on the production of fumonisins by Aspergillus niger, and different Fusarium species. BMC Microbiol. 2009, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kushiro, M.; Nakagawa, H.; Nagashima, H.; Saito, M. Fumonisin B2 production on agar media by Aspergillus niger of Japanese origin. JSM Mycotoxins 2011, 61, 47–52. [Google Scholar] [CrossRef]
- Mogensen, J.M.; Frisvad, J.C.; Thrane, U.; Nielsen, K.F. Production of Fumonisin B2 and B4 by Aspergillus niger on grapes and raisins. J. Agric. Food Chem. 2010, 58, 954–958. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, L.M.; Lametsch, R.; Andersen, M.R.; Nielsen, P.; Frisvad, J.C. Proteome analysis of Aspergillus niger, Lactate added in starch-containing medium can increase production of the mycotoxin fumonisin B2 by modifying acetyl-CoA metabolism. BMC Microbiol. 2009, 9, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Susca, A.; Proctor, R.H.; Morelli, M.; Haidukowski, M.; Gallo, A.; Lorgrieco, A.F.; Moretti, A. Variation in Fumonisin and Ochratoxin Production Associated with Differences in Biosynthetic Gene Content in Aspergillus niger and A. welwitschiae Isolates from Multiple Crop and Geographic Origins. Front. Microbiol. 2016, 7, 1412. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, J.D.; O’Keeffe, T.L.; Gorski, L. Multiplex PCR analysis of fumonisin biosynthetic genes in fumonisin-nonproducing Aspergillus niger and A. awamori strains. Mycologia 2013, 105, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Baird, R.; Abbas, H.K.; Windham, G.; Williams, P.; Baird, S.; Ma, P.; Kelley, R.; Hawkins, L.; Scruggs, M. Identification of select fumonisin forming Fusarium species using PCR applications of the polyketide synthase gene and its relationship to fumonisin production in vitro. Int. J. Mol. Sci. 2008, 9, 554–570. [Google Scholar] [CrossRef] [PubMed]
- Butchko, R.A.E.; Plattner, R.D.P.; Proctor, R.H. Deletion Analysis of FUM Genes Involved in Tricarballylic Ester Formation during Fumonisin Biosynthesis. J. Agric. Food Chem. 2006, 54, 9398–9404. [Google Scholar] [CrossRef] [PubMed]
- Glenn, A.E.; Zitomer, N.C.; Zimeri, A.M.; Riley, R.T.; Proctor, R.H. Transformation-mediated complementation of a FUM gene cluster deletion in Fusarium verticillioides restores both fumonisin production and pathogenicity on maize seedlings. MPMI 2008, 21, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.J.; Hu, X.; Shao, B.; Lin, X.H.; Yu, H.X.; Li, F.Q. Ultra performance liquid chromatography-tandem mass spectrometry method for determination of fumonisins in wheat flour, corn and corn-based products. J. Shandong Univ. Health Sci. 2012, 50, 115–120. [Google Scholar]
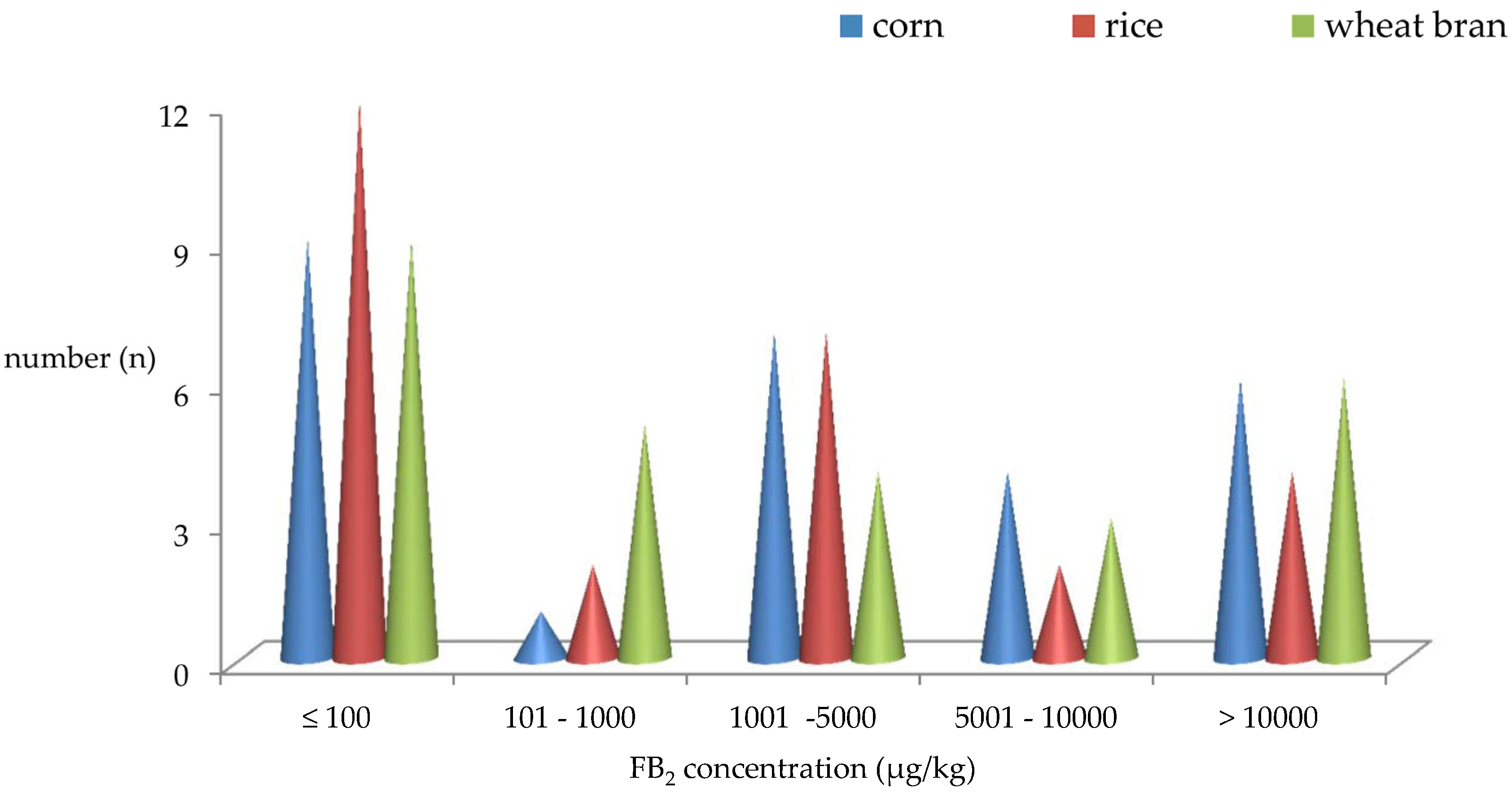
| Stain No 1 | Corn (μg/kg) | Rice (μg/kg) | Wheat Bran (μg/kg) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Day 7 | Day 14 | Day 21 | Day 28 | Day 7 | Day 14 | Day 21 | Day 28 | Day 7 | Day 14 | Day 21 | Day 28 | |
| SN-01 | 12,802 | 70,488 | 33165 | 32,910 | 11,449 | 15,716 | 6699 | 10,982 | 970 | 2562 | 3915 | 3048 |
| SN-02 | 6710 | 8166 | 6217.5 | 9442 | 3726 | 3116 | 2883 | 4462 | 3694 | 3442 | 4167.5 | 3490 |
| SN-03 | 4192 | 4540 | 3855 | 4552 | 10,160 | 6648 | 5665 | 8876 | 718 | 860 | 828 | 866 |
| SN-04 | 6304 | 8880 | 8482.5 | 8794 | 3518 | 4030 | 3640 | 5068 | 12,556 | 21,414 | 23,713 | 20,090 |
| SN-05 | 1282 | 850 | 860 | 3776 | 1570 | 2364 | 1078 | 2720 | 158 | 198 | 268 | 266 |
| SN-06 | 644 | 1970 | 3707.5 | 14,174 | 18,174 | 4252 | 34,994 | 54,284 | 150 | 240 | 175 | 186 |
| SN-07 | 942 | 2046 | 1916 | 1288 | 1926 | 5734 | 3376 | 3232 | 6640 | 9756 | 10,354 | 13,826 |
| SN-08 | 5264 | 11,978 | 7735 | 6592 | 4384 | 3036 | 3175 | 4892 | 10,558 | 18,148 | 12,355 | 15,360 |
| SN-09 | 2534 | 11,854 | 14,105 | 16,042 | 320 | 554 | 278 | 482 | 31,994 | 18,050 | 19,248 | 25,028 |
| SN-10 | 6 | 2 | 16 | 6 | nd 2 | 4 | 12 | 10 | 110 | 30 | 36 | 62 |
| SN-11 | 2336 | 3300 | 2694 | 4556 | 3968 | 4720 | 4448 | 3814 | 3052 | 8696 | 7232 | 6918 |
| SN-12 | 2716 | 7878 | 8474 | 7534 | 4644 | 20,178 | 16,744 | 20,906 | 5558 | 7858 | 8790 | 8160 |
| SN-13 | 458 | 1554 | 1416 | 1072 | 1786 | 568 | 324 | 410 | 1310 | 5304 | 3556 | 2922 |
| OA-01 | 958 | 2874 | 4952 | 3978 | 1784 | 54 | 40 | 34 | 230 | 928 | 624 | 500 |
| OA-02 | 608 | 220 | 78 | 98 | 250 | 246 | 230 | 4 | 7104 | 1212 | 180 | 48 |
| OA-03 | 22 | 262 | 78 | 24,240 | 18 | 12 | 15 | 6 | 7018 | 13,370 | 14,193 | 8740 |
| OA-04 | 332 | 7072 | 43,138 | 28,594 | 376 | 1990 | 9423 | 1996 | 34,250 | 22,326 | 13,735 | 23,498 |
| OA-05 | 3376 | 1258 | 10,600 | 3268 | 368 | 1576 | 3426 | 4776 | 38,094 | 34,506 | 18 26 | 28,826 |
| OA-06 | nd | nd | 123 | nd | nd | nd | 5 | nd | nd | nd | nd | 2 |
| TA-01 | 5 | 10 | 3 | 10 | nd | 2 | 8 | 4 | 14 | 20 | 28 | 30 |
| TA-02 | nd | nd | nd | 6 | nd | nd | nd | nd | nd | nd | 3 | 2 |
| TA-03 | nd | nd | 4 | 2 | nd | nd | nd | nd | nd | nd | nd | nd |
| TA-04 | nd | 4 | nd | nd | 4 | nd | nd | nd | nd | nd | 2 | 2 |
| TA-05 | nd | nd | nd | 4 | nd | 8 | nd | nd | nd | nd | 5 | 4 |
| TA-06 | nd | nd | nd | 6 | nd | nd | nd | 14 | nd | nd | 6 | 2 |
| TA-07 | nd | nd | nd | 10 | nd | nd | nd | nd | nd | nd | nd | nd |
| GA-01 | 2 | 4 | 4 | 2 | nd | 10 | 6 | 6 | 120 | 222 | 138 | 138 |
| ACCC30557 | 256 | 1558 | 553 | 2194 | 1226 | 5764 | 1223 | 78 | nd | 16 | 8 | 4 |
| ATCC16404 | 2 | 20 | 25 | 28 | 38 | 20 | 30 | 40 | nd | nd | nd | 2 |
| SI-01 | 2050 | 5828 | 1335 | 13,342 | 11,188 | 22,906 | 20,873 | 23,700 | 1422 | 2382 | 8718 | 2174 |
| Substrate | Time Intervals (day) | FB2 Production (µg/kg) | |||||
|---|---|---|---|---|---|---|---|
| Saccharifying Enzyme Producer (n = 13) | Organic Acid Producer (n = 6) | Tannase Producer (n = 7) | |||||
| No. of Positive (%) | Mean (Range) (μg/kg) | No. of Positive (%) | Mean (Range) (μg/kg) | No. of Positive (%) | Mean (Range) (μg/kg) | ||
| Corn | 7 | 13 (100) | 3553 (6–12802) | 5 (83) | 1059 (22–3376) | 1 (14) | 5 |
| 14 | 13 (100) | 10,270 (2–70,488) | 5 (83) | 2337 (220–7258) | 2 (29) | 7 (4–10) | |
| 21 | 13 (100) | 7126 (16–33,165) | 6 (100) | 9828 (78–43,138) | 2 (29) | 3 (3–4) | |
| 28 | 13 (100) | 8518 (6–32,910) | 5 (83) | 12,036 (98–28,594) | 5 (71) | 6 (2–10) | |
| Rice | 7 | 13 (100) | 5469 (320–18,174) | 5 (83) | 559 (18–1784) | 1 (14) | 4 |
| 14 | 13 (100) | 5455 (4–20178) | 5 (83) | 776 (12–1990) | 2 (29) | 5 (2–8) | |
| 21 | 13 (100) | 6409 (12–34,994) | 6 (100) | 2190 (5–9423) | 1 (14) | 8 | |
| 28 | 13 (100) | 9241 (10–54,284) | 5 (83) | 1363 (4–4776) | 1 (14) | 9 (4–14) | |
| Wheat bran | 7 | 13 (100) | 5959 (110–31,994) | 5 (83) | 17,339 (230–38,094) | 2 (29) | 14 |
| 14 | 13 (100) | 7428 (30–21,414) | 5 (83) | 14,468 (928–34,506) | 1 (14) | 20 | |
| 21 | 13 (100) | 7280 (36–23,713) | 5 (83) | 9491 (180–18,726) | 4 (57) | 9 (2–28) | |
| 28 | 13 (100) | 7709 (62–25,028) | 6 (100) | 10269 (2–28,826) | 5 (71) | 8 (2–30) | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, X.; Jiang, H.; Xu, J.; Zhang, J.; Li, F. Dynamic Fumonisin B2 Production by Aspergillus niger Intented Used in Food Industry in China. Toxins 2017, 9, 217. https://doi.org/10.3390/toxins9070217
Han X, Jiang H, Xu J, Zhang J, Li F. Dynamic Fumonisin B2 Production by Aspergillus niger Intented Used in Food Industry in China. Toxins. 2017; 9(7):217. https://doi.org/10.3390/toxins9070217
Chicago/Turabian StyleHan, Xiaomin, Hongru Jiang, Jin Xu, Jing Zhang, and Fengqin Li. 2017. "Dynamic Fumonisin B2 Production by Aspergillus niger Intented Used in Food Industry in China" Toxins 9, no. 7: 217. https://doi.org/10.3390/toxins9070217
APA StyleHan, X., Jiang, H., Xu, J., Zhang, J., & Li, F. (2017). Dynamic Fumonisin B2 Production by Aspergillus niger Intented Used in Food Industry in China. Toxins, 9(7), 217. https://doi.org/10.3390/toxins9070217