A Supercluster of Neutralizing Epitopes at the Interface of Ricin’s Enzymatic (RTA) and Binding (RTB) Subunits
Abstract
:1. Introduction
2. Results
2.1. Characterization of RTB- and Holotoxin-Specific VHHs that Compete with SyH7
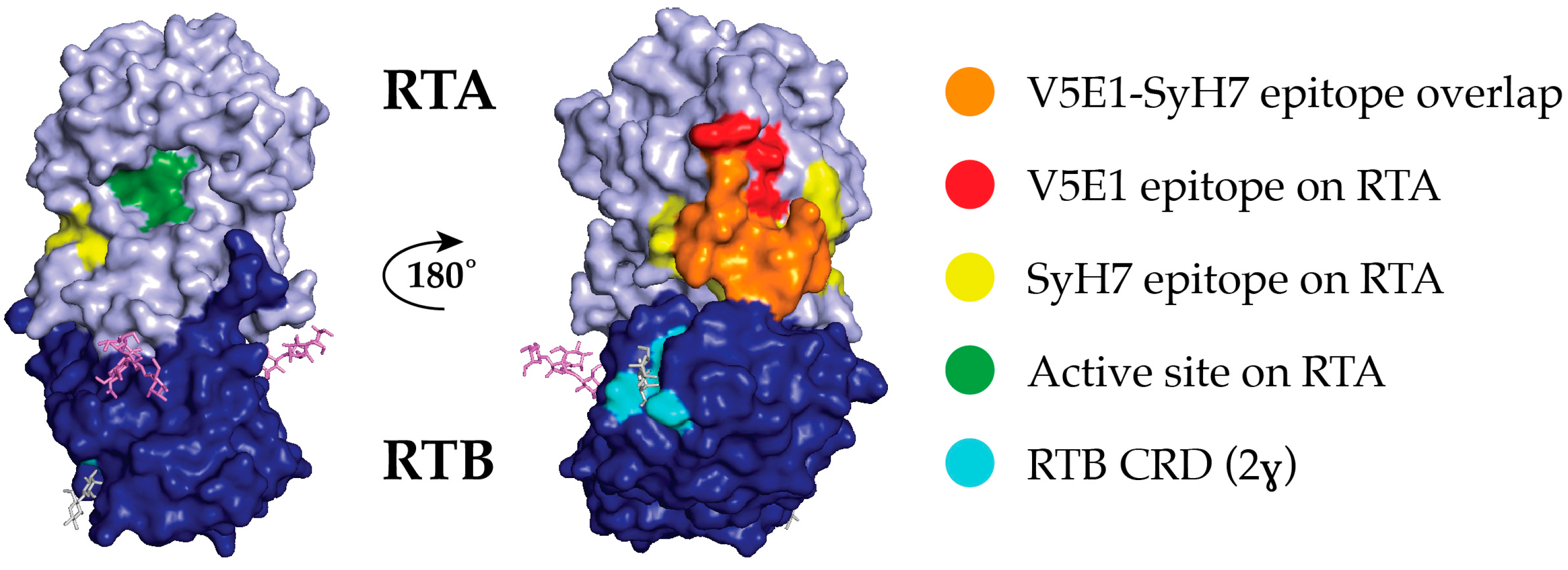
2.2. Epitope Positioning by V5E1 Competition
2.3. Cross-Competition ELISAs Indicate That Tier I VHHs Recognize Overlapping Epitopes on RTB
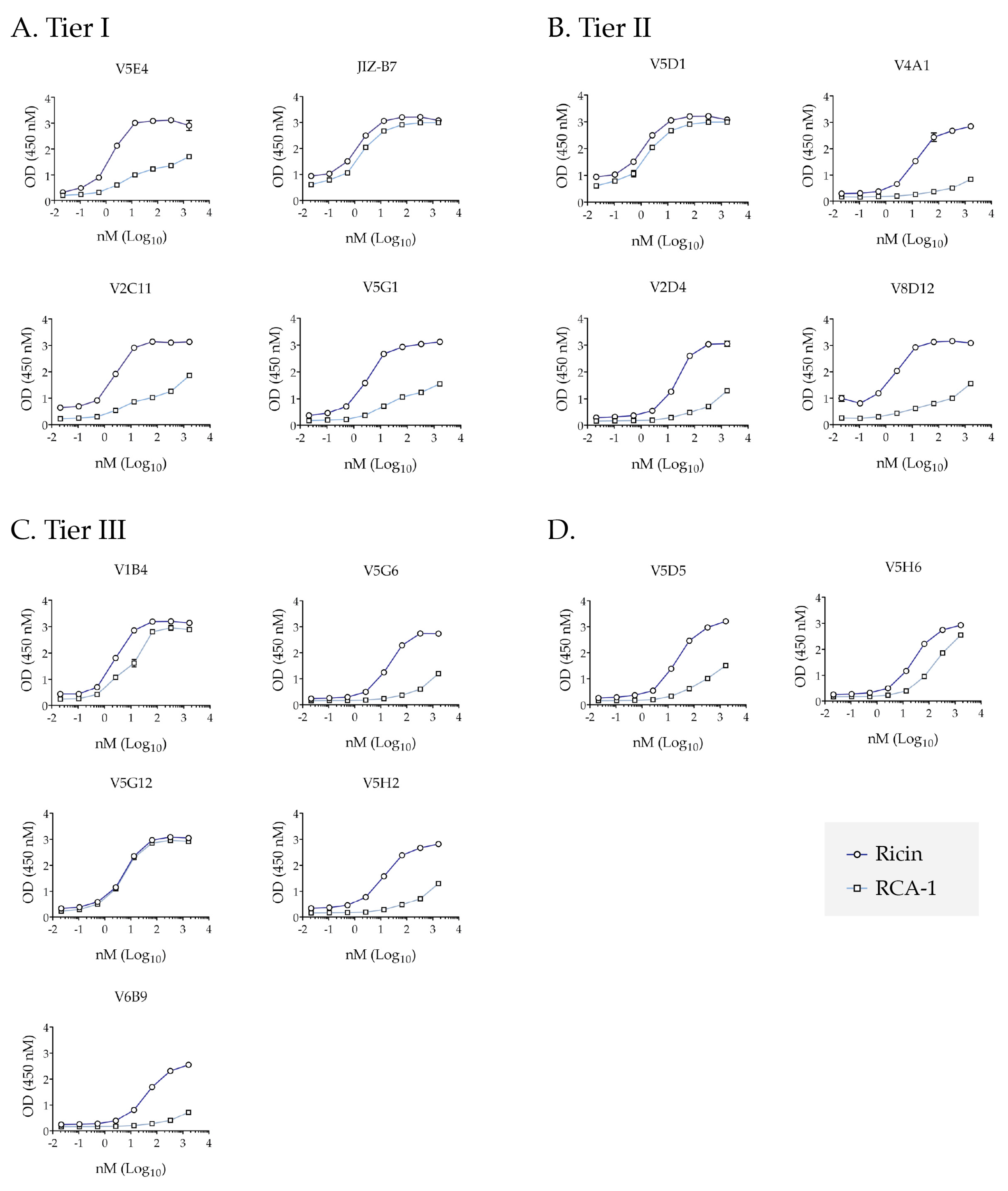
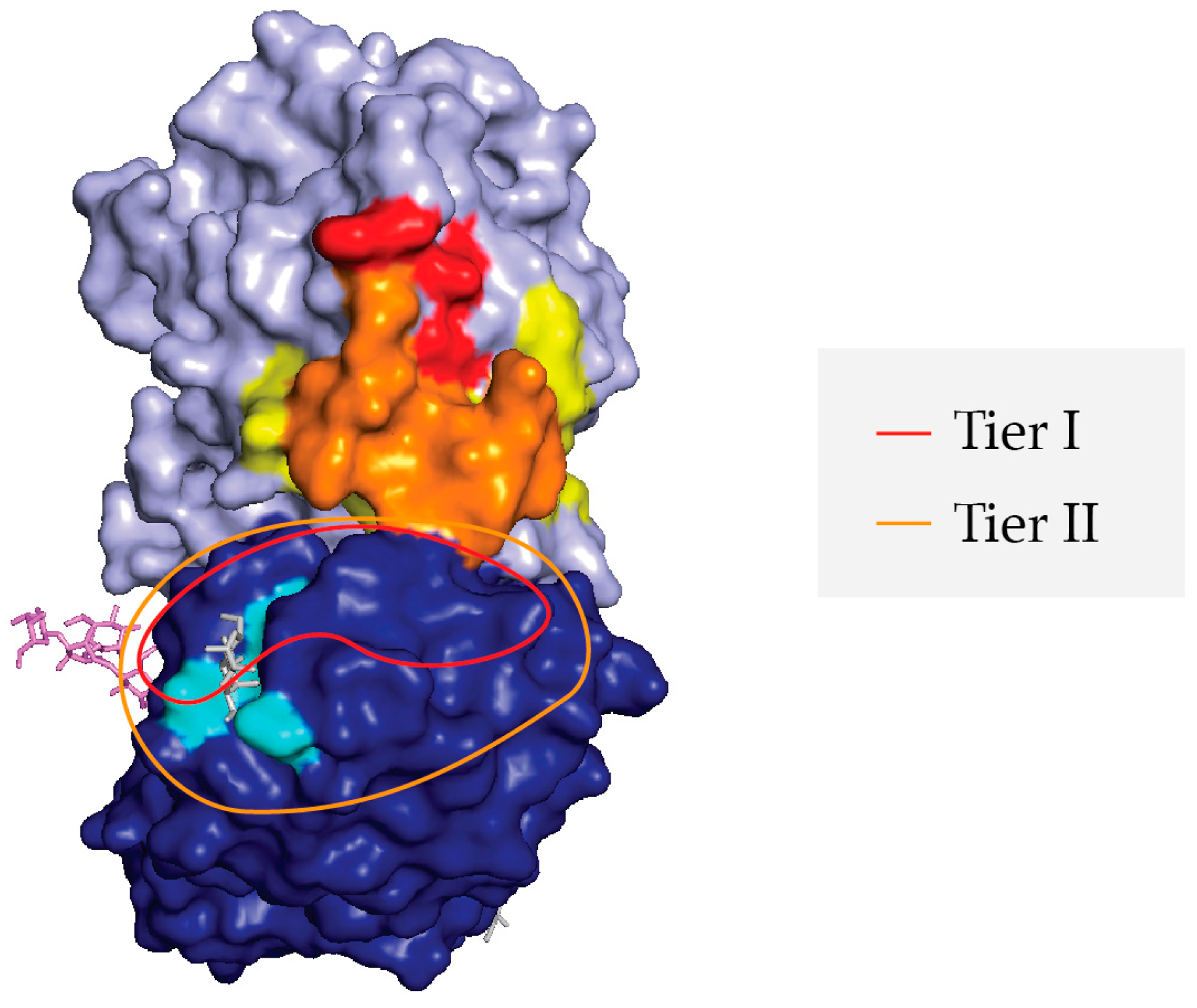
2.4. Differential Reactivity with RCA-1 Facilitates Epitope Localization
2.5. Relative Epitope Positioning within Supercluster II
2.6. TNA Does Not Correlate with VHHs’ Ability to Block Ricin Attachment to Receptors
3. Conclusions and Discussion
4. Methods
4.1. Chemicals, Biological Reagents and Cell Lines
4.2. VHHs and mAbs
4.3. ELISA
4.4. Vero Cell Cytotoxicity Assays
4.5. THP-1 Cell Attachment Assay
4.6. Statistical Analyses
4.7. Modeling of Ricin Toxin
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schrot, J.; Weng, A.; Melzig, M.F. Ribosome-inactivating and related proteins. Toxins (Basel) 2015, 7, 1556–1615. [Google Scholar] [CrossRef] [PubMed]
- Lamb, F.I.; Roberts, L.M.; Lord, J.M. Nucleotide sequence of cloned cdna coding for preproricin. Eur. J. Biochem. 1985, 148, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Lord, J.M. Precursors of ricin and ricinus communis agglutinin. Glycosylation and processing during synthesis and intracellular transport. Eur. J. Biochem. 1985, 146, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, N.A.; Craddock, C.P.; Frigerio, L. Pathways for protein transport to seed storage vacuoles. Biochem. Soc. Trans. 2005, 33, 1016–1018. [Google Scholar] [CrossRef] [PubMed]
- Endo, Y.; Mitsui, K.; Motizuki, M.; Tsurugi, K. The mechanism of action of ricin and related toxic lectins on eukaryotic ribosomes. The site and the characteristics of the modification in 28 s ribosomal rna caused by the toxins. J. Biol. Chem. 1987, 262, 5908–5912. [Google Scholar] [PubMed]
- Endo, Y.; Tsurugi, K. Rna n-glycosidase activity of ricin a-chain. Mechanism of action of the toxic lectin ricin on eukaryotic ribosomes. J. Biol. Chem. 1987, 262, 8128–8130. [Google Scholar] [PubMed]
- Tesh, V.L. The induction of apoptosis by shiga toxins and ricin. Curr. Top. Microbiol. Immunol. 2012, 357, 137–178. [Google Scholar] [PubMed]
- Montfort, W.; Villafranca, J.E.; Monzingo, A.F.; Ernst, S.R.; Katzin, B.; Rutenber, E.; Xuong, N.H.; Hamlin, R.; Robertus, J.D. The three-dimensional structure of ricin at 2.8 a. J. Biol. Chem. 1987, 262, 5398–5403. [Google Scholar] [PubMed]
- Rutenber, E.; Katzin, B.J.; Ernst, S.; Collins, E.J.; Mlsna, D.; Ready, M.P.; Robertus, J.D. Crystallographic refinement of ricin to 2.5 a. Proteins 1991, 10, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Rutenber, E.; Ready, M.; Robertus, J.D. Structure and evolution of ricin b chain. Nature 1987, 326, 624–626. [Google Scholar] [CrossRef] [PubMed]
- Sandvig, K.; Olsnes, S.; Pihl, A. Kinetics of binding of the toxic lectins abrin and ricin to surface receptors of human cells. J. Biol. Chem. 1976, 251, 3977–3984. [Google Scholar] [PubMed]
- Cummings, R.; Etzler, M. R-type lectins. In Essentials of Glycobiology; Varki, A., Cummings, R., Esko, J., Freeze, H., Stanley, P., Bertozzi, C., Hart, G., Etzler, M., Eds.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2009. [Google Scholar]
- Newton, D.L.; Wales, R.; Richardson, P.T.; Walbridge, S.; Saxena, S.K.; Ackerman, E.J.; Roberts, L.M.; Lord, J.M.; Youle, R.J. Cell surface and intracellular functions for ricin galactose binding. J. Biol. Chem. 1992, 267, 11917–11922. [Google Scholar] [PubMed]
- Zentz, C.; Frenoy, J.P.; Bourrillon, R. Binding of galactose and lactose to ricin. Equilibrium studies. Biochim. Biophys. Acta 1978, 536, 18–26. [Google Scholar] [CrossRef]
- Baenziger, J.U.; Fiete, D. Structural determinants of ricinus communis agglutinin and toxin specificity for oligosaccharides. J. Biol. Chem. 1979, 254, 9795–9799. [Google Scholar] [PubMed]
- Vance, D.J.; Tremblay, J.M.; Mantis, N.J.; Shoemaker, C.B. Stepwise engineering of heterodimeric single domain camelid vhh antibodies that passively protect mice from ricin toxin. J. Biol. Chem. 2013, 288, 36538–36547. [Google Scholar] [CrossRef] [PubMed]
- Vance, D.J.; Tremblay, J.M.; Rong, Y.; Angalakurthi, S.K.; Volkin, D.B.; Middaugh, C.R.; Weis, D.D.; Shoemaker, C.B.; Mantis, N.J. High-resolution epitope positioning of a large collection of neutralizing and non-neutralizing single domain antibodies on ricin toxin’s enzymatic and binding subunits. Clin. Vaccine Immunol. 2017. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, J.M.; Kasten-Jolly, J.C.; Reynolds, C.E.; Mantis, N.J. Localization of non-linear neutralizing b cell epitopes on ricin toxin’s enzymatic subunit (rta). Immunol. Lett. 2014, 158, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Toth, R.T.; Angalakurthi, S.K.; Van Slyke, G.; Vance, D.J.; Hickey, J.M.; Joshi, S.B.; Middaugh, C.R.; Volkin, D.B.; Weis, D.D.; Mantis, N.J. High-definition mapping of four spatially distinct neutralizing epitope clusters on rivax, a candidate ricin toxin subunit vaccine. Clin. Vaccine Immunol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Yermakova, A.; Vance, D.J.; Mantis, N.J. Sub-domains of ricin’s b subunit as targets of toxin neutralizing and non-neutralizing monoclonal antibodies. PLoS ONE 2012, 7, e44317. [Google Scholar] [CrossRef] [PubMed]
- Lebeda, F.J.; Olson, M.A. Prediction of a conserved, neutralizing epitope in ribosome-inactivating proteins. Int. J. Biol. Macromol. 1999, 24, 19–26. [Google Scholar] [CrossRef]
- O’Hara, J.M.; Neal, L.M.; McCarthy, E.A.; Kasten-Jolly, J.A.; Brey, R.N., 3rd; Mantis, N.J. Folding domains within the ricin toxin a subunit as targets of protective antibodies. Vaccine 2010, 28, 7035–7046. [Google Scholar]
- Vance, D.J.; Mantis, N.J. Resolution of two overlapping neutralizing b cell epitopes within a solvent exposed, immunodominant alpha-helix in ricin toxin’s enzymatic subunit. Toxicon 2012, 60, 874–877. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, M.J.; Vance, D.J.; Cassidy, M.S.; Rong, Y.; Mantis, N.J. Structural analysis of single domain antibodies bound to a second neutralizing hot spot on ricin toxin’s enzymatic subunit. J. Biol. Chem. 2017, 292, 872–883. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.; Vance, D.J.; Eisele, L.E.; Shoemaker, C.B.; Mantis, N.J. Differential neutralizing activities of a single domain camelid antibody (vhh) specific for ricin toxin’s binding subunit (rtb). PLoS ONE 2014, 9, e99788. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.; Tremblay, J.M.; Shoemaker, C.B.; Mantis, N.J. Mechanisms of ricin toxin neutralization revealed through engineered homodimeric and heterodimeric camelid antibodies. J. Biol. Chem. 2015, 290, 27880–27889. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.; Klokk, T.I.; Cole, R.; Sandvig, K.; Mantis, N.J. A bispecific antibody promotes aggregation of ricin toxin on cell surfaces and alters dynamics of toxin internalization and trafficking. PLoS ONE 2016, 11, e0156893. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, R.; Alcalay, R.; Mechaly, A.; Lapidoth, G.; Epstein, E.; Kronman, C.; Fleishman, S.J.; Mazor, O. Improved antibody-based ricin neutralization by affinity maturation is correlated with slower off-rate values. Protein Eng. Des. Sel. PEDS 2017, 30, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Derking, R.; Ozorowski, G.; Sliepen, K.; Yasmeen, A.; Cupo, A.; Torres, J.L.; Julien, J.P.; Lee, J.H.; van Montfort, T.; de Taeye, S.W.; et al. Comprehensive antigenic map of a cleaved soluble hiv-1 envelope trimer. PLoS Pathog. 2015, 11, e1004767. [Google Scholar] [CrossRef] [PubMed]
- Rong, Y.; Van Slyke, G.; Vance, D.J.; Westfall, J.; Ehrbar, D.; Mantis, N.J. Spatial location of neutralizing and non-neutralizing b cell epitopes on domain 1 of ricin toxin’s binding subunit. PLoS ONE 2017, 12, e0180999. [Google Scholar] [CrossRef] [PubMed]
- Yermakova, A.; Mantis, N.J. Protective immunity to ricin toxin conferred by antibodies against the toxin’s binding subunit (rtb). Vaccine 2011, 29, 7925–7935. [Google Scholar] [CrossRef] [PubMed]
- Bazzoli, A.; Vance, D.J.; Rudolph, M.J.; Rong, Y.; Angalakurthi, S.K.; Toth, R.T.T.; Middaugh, C.R.; Volkin, D.B.; Weis, D.D.; Karanicolas, J.; et al. Using homology modeling to interrogate binding affinity in neutralization of ricin toxin by a family of single domain antibodies. Proteins 2017, 85, 1994–2008. [Google Scholar] [CrossRef] [PubMed]
- Wales, R.; Richardson, P.T.; Roberts, L.M.; Woodland, H.R.; Lord, J.M. Mutational analysis of the galactose binding ability of recombinant ricin b chain. J. Biol. Chem. 1991, 266, 19172–19179. [Google Scholar] [PubMed]
- Green, E.D.; Adelt, G.; Baenziger, J.U.; Wilson, S.; Van Halbeek, H. The asparagine-linked oligosaccharides on bovine fetuin. Structural analysis of n-glycanase-released oligosaccharides by 500-megahertz 1 h nmr spectroscopy. J. Biol. Chem. 1988, 263, 18253–18268. [Google Scholar] [PubMed]
- Song, K.; Mize, R.R.; Marrero, L.; Corti, M.; Kirk, J.M.; Pincus, S.H. Antibody to ricin a chain hinders intracellular routing of toxin and protects cells even after toxin has been internalized. PLoS ONE 2013, 8, e62417. [Google Scholar] [CrossRef] [PubMed]
- Yermakova, A.; Klokk, T.I.; Cole, R.; Sandvig, K.; Mantis, N.J. Antibody-mediated inhibition of ricin toxin retrograde transport. MBio 2014, 5, e00995. [Google Scholar] [CrossRef] [PubMed]
- Yermakova, A.; Klokk, T.I.; O’Hara, J.M.; Cole, R.; Sandvig, K.; Mantis, N.J. Neutralizing monoclonal antibodies against disparate epitopes on ricin toxin’s enzymatic subunit interfere with intracellular toxin transport. Sci. Rep. 2016, 6, 22721. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Nellas, R.B.; Glover, M.M.; Shen, T. Stability and sugar recognition ability of ricin-like carbohydrate binding domains. Biochemistry 2011, 50, 4097–4104. [Google Scholar] [CrossRef] [PubMed]
- Chanh, T.C.; Romanowski, M.J.; Hewetson, J.F. Monoclonal antibody prophylaxis against the in vivo toxicity of ricin in mice. Immunol. Investig. 1993, 22, 63–72. [Google Scholar] [CrossRef]
- Colombatti, M.; Pezzini, A.; Colombatti, A. Monoclonal antibodies against ricin: Effects on toxin function. Hybridoma 1986, 5, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Maddaloni, M.; Cooke, C.; Wilkinson, R.; Stout, A.V.; Eng, L.; Pincus, S.H. Immunological characteristics associated with the protective efficacy of antibodies to ricin. J. Immunol. 2004, 172, 6221–6228. [Google Scholar] [CrossRef] [PubMed]
- Wahome, P.G.; Mantis, N.J. High-throughput, cell-based screens to identify small-molecule inhibitors of ricin toxin and related category b ribosome inactivating proteins (rips). Curr. Protoc. Toxicol. 2013. [Google Scholar] [CrossRef]




| Competitive Binding Assays | ||||||
|---|---|---|---|---|---|---|
| Tier a | VHH | Target | KD b [nM] | TNA c (IC50) | V5E1 d–f | Bin g |
| I | V5E4 | RTB | 0.006 | 0.3 | 87 ± 12 | 3 |
| JIZ-B7 | RTB | 0.0306 | 0.6 | 95 ± 2 | 4 | |
| V2C11 | RTB | 0.0154 | 0.8 | 88 ± 12 | 4 | |
| V5G1 | RTB | 0.106 | 5 | 86 ± 16 | 5 | |
| II | V5D1 | Holotoxin | 0.216 | 65 | 85 ± 5 | 1 |
| V2D4 | RTB | 3.2 | 130 | 64 ± 17 * | 6 | |
| V4A1 | RTB | 0.666 | 300 | 62 ± 16 * | 6 | |
| V8D12 | RTB | 0.194 | 300 | 62 ± 10 * | n.a. | |
| III | V1B4 | Holotoxin | 4.09 | - | 85 ± 7 | 2 |
| V5G6 | Holotoxin | 1.24 | - | 37 ± 6 ** | 7 | |
| V5G12 | Holotoxin | 0.993 | - | 42 ± 12 ** | 2 | |
| V5H2 | RTB | 2.17 | - | 38 ± 3 ** | 7 | |
| V6B9 | RTB | 1.61 | - | 40 ± 14 ** | n.a. | |
| Control | V5D5 | RTB | 5.54 | - | 7 ± 12 ** | 10 |
| V5H6 | RTB | 18.3 | - | 9 ±16 ** | 8 | |
| Capture VHHs | |||||
|---|---|---|---|---|---|
| Tier | Analyte a | V5E1 | JIZ-B7 | V2C11 | V5G1 |
| I | V5E4 | 96 ± 2 b | 48 ± 11 | 96 ± 1 | 95 ± 2 |
| JIZ-B7 | 96 ± 1 | 96 ± 1 | 96 ± 2 | 95 ± 2 | |
| V2C11 | 96 ± 2 | 42 ± 9 | 95 ± 3 | 95 ± 2 | |
| V5G1 | 94 ± 3 | 43 ± 4 | 94 ± 3 | 94 ± 3 | |
| II | V5D1 * | 80 ± 10 | 82 ± 8 | 79 ± 12 | 82 ± 12 |
| V2D4 | 83 ± 1 | 35 ± 11 | 82 ± 4 | 89 ± 2 | |
| V4A1 | 83 ± 3 | 29 ± 8 | 88 ± 1 | 88 ± 2 | |
| V8D12 | 84 ± 16 | 31 ± 10 | 89 ± 8 | 88 ± 10 | |
| III | V1B4 * | 43 ± 12 | 45 ± 4 | 40 ± 11 | 70 ± 6 |
| V5G6 * | 22 ± 2 | 10 ± 2 | 29 ± 7 | 44 ± 11 | |
| V5G12 * | 9 ± 3 | 13 ± 1 | 6 ± 5 | 29 ± 14 | |
| V5H2 | 47 ± 5 | 16 ± 6 | 53 ± 3 | 67 ± 7 | |
| V6B9 | 68 ± 4 | 27 ± 7 | 68 ± 13 | 79 ± 4 | |
| Controls | V5D5 | 2 ± 2 | 2 ± 1 | 3 ± 2 | 19 ± 14 |
| V5H2 | 2 ± 3 | 1 ± 2 | 4 ± 5 | 10 | |
| % Inhibition | |||
|---|---|---|---|
| Tier | VHH | ASF a | THP-1 b |
| I | V5E4 | 64 ± 8 | 45 ± 16 |
| JIZ-B7 | 48 ± 9 | 36 ± 11 | |
| V2C11 | 65 ± 11 | 50 ± 16 | |
| V5G1 | 60 ±12 | 44 ± 12 | |
| II | V5D1 * | 0 | 22 ± 17 |
| V2D4 | 62 ± 14 | 28 ± 12 | |
| V4A1 | 59 ± 9 | 40 ± 7 | |
| V8D12 | 57 ± 8 | 46 ± 10 | |
| III | V1B4 * | 19 ± 18 | 27 ± 18 |
| V5G6 * | 38 ± 13 | 16 ± 14 | |
| V5G12 * | 0 | 3 ± 14 | |
| V5H2 | 55 ± 2 | 40 ± 6 | |
| V6B9 | 57 ± 3 | 38 ± 8 | |
| Controls | V5D5 | 84 ± 10 | 90 ± 1 |
| V5H6 | 6 ± 11 | 18 ± 8 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poon, A.Y.; Vance, D.J.; Rong, Y.; Ehrbar, D.; Mantis, N.J. A Supercluster of Neutralizing Epitopes at the Interface of Ricin’s Enzymatic (RTA) and Binding (RTB) Subunits. Toxins 2017, 9, 378. https://doi.org/10.3390/toxins9120378
Poon AY, Vance DJ, Rong Y, Ehrbar D, Mantis NJ. A Supercluster of Neutralizing Epitopes at the Interface of Ricin’s Enzymatic (RTA) and Binding (RTB) Subunits. Toxins. 2017; 9(12):378. https://doi.org/10.3390/toxins9120378
Chicago/Turabian StylePoon, Amanda Y., David J. Vance, Yinghui Rong, Dylan Ehrbar, and Nicholas J. Mantis. 2017. "A Supercluster of Neutralizing Epitopes at the Interface of Ricin’s Enzymatic (RTA) and Binding (RTB) Subunits" Toxins 9, no. 12: 378. https://doi.org/10.3390/toxins9120378
APA StylePoon, A. Y., Vance, D. J., Rong, Y., Ehrbar, D., & Mantis, N. J. (2017). A Supercluster of Neutralizing Epitopes at the Interface of Ricin’s Enzymatic (RTA) and Binding (RTB) Subunits. Toxins, 9(12), 378. https://doi.org/10.3390/toxins9120378







