Lolitrem B and Indole Diterpene Alkaloids Produced by Endophytic Fungi of the Genus Epichloë and Their Toxic Effects in Livestock
Abstract
:1. Introduction
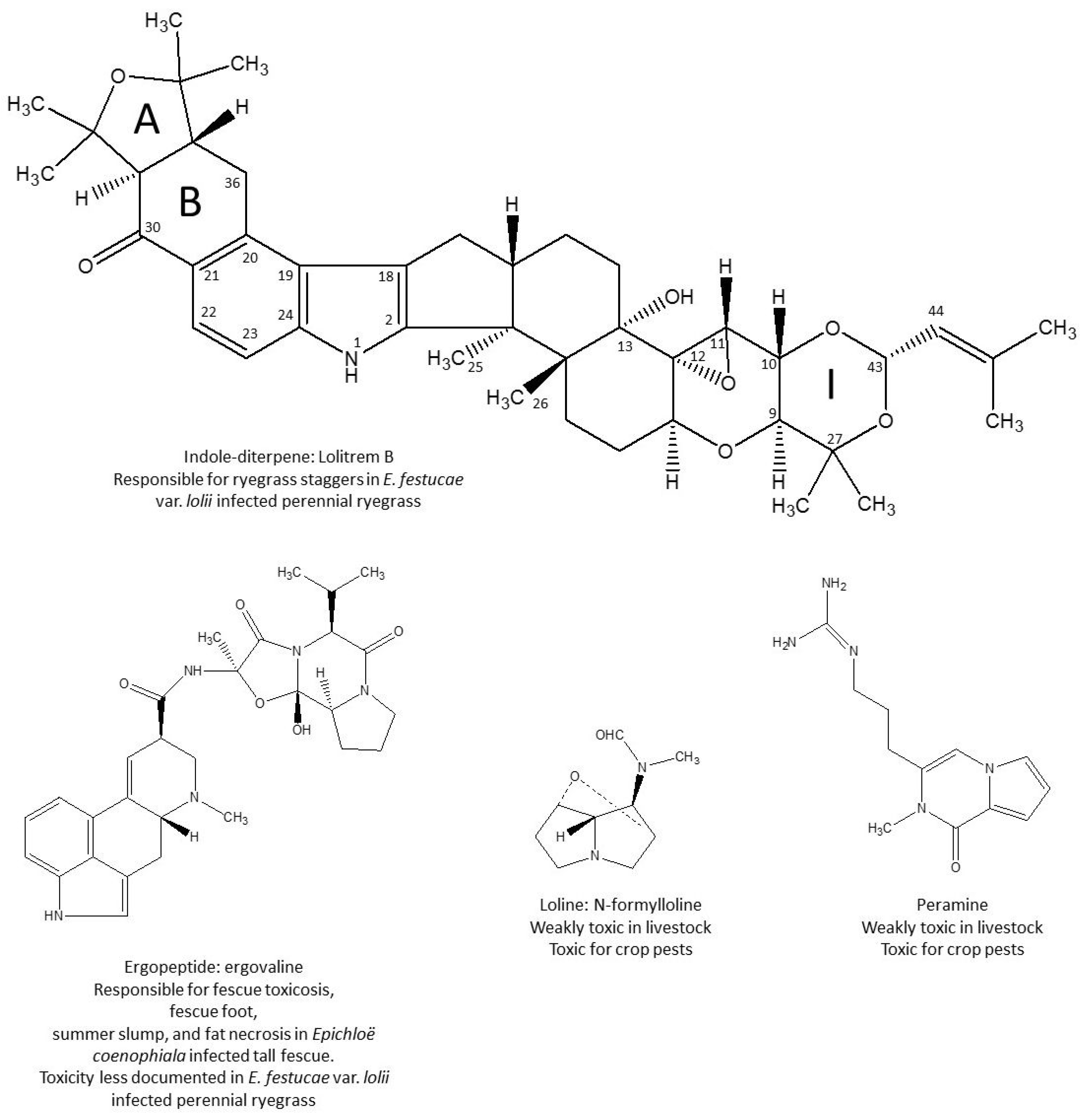
2. Ryegrass Staggers, from Its First Characterization to the Discovery of Lolitrem B
3. Lolitrem B, a Tremorgenic Mycotoxin
3.1. From Staggers to BK-Channels
3.2. Toxicokinetics
4. Lolitrem B in Plants and Ryegrass Staggers
4.1. Worldwide Distribution of the Disease
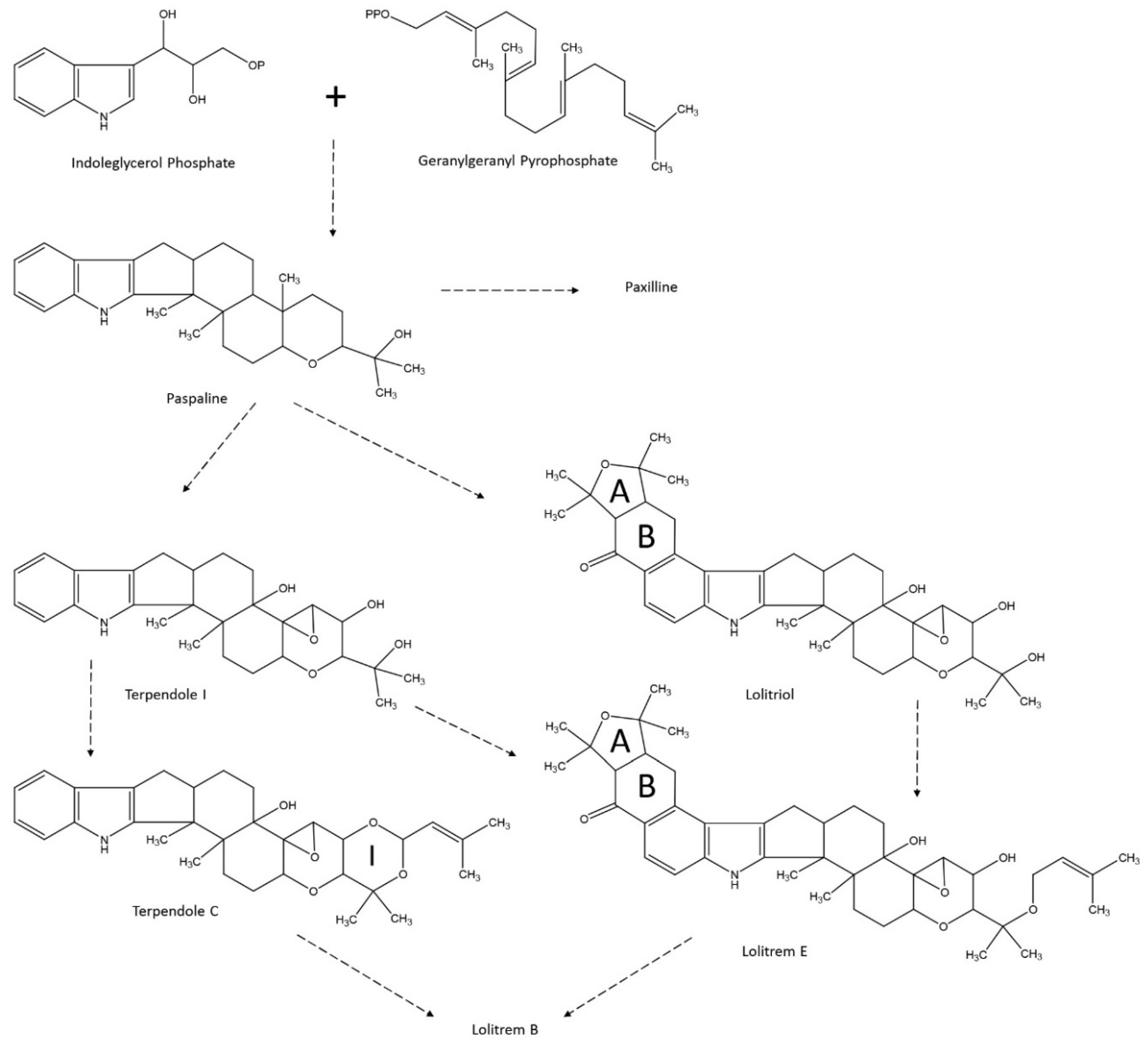
4.2. Lolitrem B Biosynthesis
4.3. Variations in Concentrations of Lolitrem B in Plants
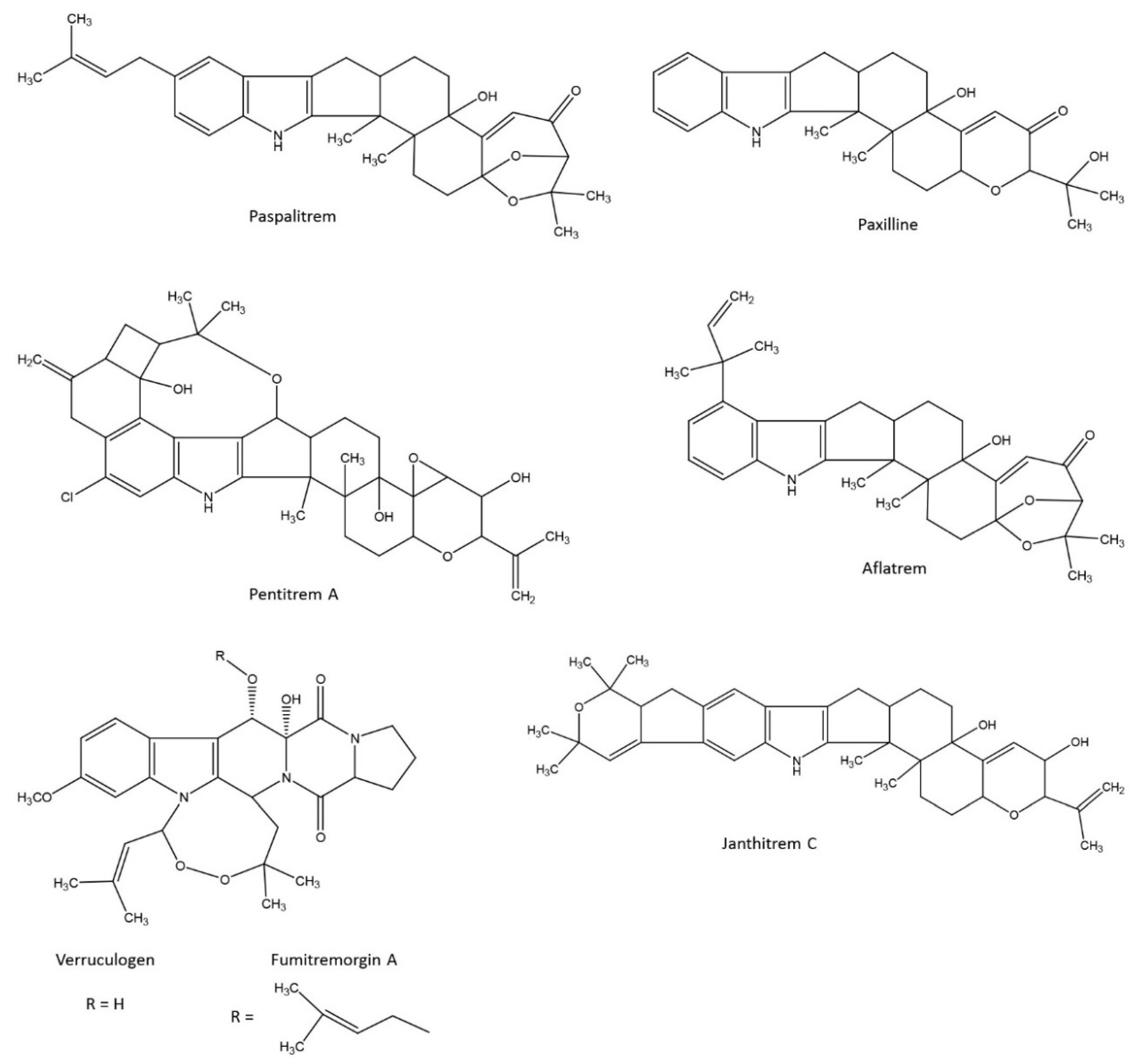
5. Indole-Diterpene Alkaloids, Not Only Lolitrem B
Acknowledgments
Conflicts of Interest
References
- Fletcher, L.R.; Harvey, I.C. An Association of a Lolium Endophyte with Ryegrass Staggers. N. Z. Vet. J. 1981, 29, 185–186. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, R.T.; White, E.P.; Mortimer, P.H. Ryegrass Staggers: Isolation of Potent Neurotoxins Lolitrem A and Lolitrem B from Staggers-Producing Pastures. N. Z. Vet. J. 1981, 29, 189–190. [Google Scholar] [CrossRef] [PubMed]
- Thom, E.R.; Waugh, C.D.; Minnee, E.M.K.; Waghorn, G.C. A New Generation Ryegrass Endophyte—the First Results from Dairy Cows Fed AR37. In Proceedings of the 6th International Symposium on Fungal Endophytes of Grasses’, Grassland Research and Practice Series, Christchurch, New Zealand, 25–28 March 2007; pp. 293–296.
- Schardl, C.L.; Young, C.A.; Faulkner, J.R.; Florea, S.; Pan, J. Chemotypic Diversity of Epichloae, Fungal Symbionts of Grasses. Fungal Ecol. 2012, 5, 331–344. [Google Scholar] [CrossRef]
- Guerre, P. Ergot Alkaloids Produced by Endophytic Fungi of the Genus Epichloë. Toxins 2015, 7, 773–790. [Google Scholar] [CrossRef] [PubMed]
- Easton, H.S.; Christensen, M.J.; Eerens, J.P.J.; Fletcher, L.R.; Hume, D.E.; Keogh, R.G.; Lane, G.A.; Latch, G.C.M.; Pennell, C.G.L.; Popay, A.J.; et al. Ryegrass Endophyte: A New Zealand Grassland Success Story. Proc. N. Z. Grassl. Assoc. 2001, 63, 37–46. [Google Scholar]
- Schardl, C.L.; Grossman, R.B.; Nagabhyru, P.; Faulkner, J.R.; Mallik, U.P. Loline Alkaloids: Currencies of Mutualism. Phytochemistry 2007, 68, 980–996. [Google Scholar] [CrossRef] [PubMed]
- McLennan, E. The Endophytic Fungus of Lolium. Part I. Proc. R. Soc. Victoria 1920, 11, 252–301. [Google Scholar]
- Sampson, K. The Systemic Infection of Grasses by Epichloe Typhina Pers. Tul. Trans. Br. Mycol. Soc. 1933, 18, 30–47. [Google Scholar] [CrossRef]
- Hopkirk, C.S.M. Staggers in Live-Stock. N. Z. J. Agric. 1935, 51, 18–22. [Google Scholar]
- Cunningham, I.J. Non-Toxicity to Animals of Ryegrass Endophyte and Other Endophytic Fungi of New Zealand Grasses. N. Z. J. Agric. Res. 1958, 1, 489–497. [Google Scholar] [CrossRef] [Green Version]
- Cunningham, I.J.; Harvey, W.J. Ryegrass Staggers. N. Z. Vet. J. 1959, 7, 1–7. [Google Scholar] [CrossRef]
- Thornton, R.H.T. An Examination of Ergot in Relation to Ryegrass Staggers. N. Z. Vet. J. 1964, 12, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Keogh, R.G. Induction and Prevention of Ryegrass Staggers in Grazing Sheep. N. Z. J. Exp. Agric. 1973, 1, 55–57. [Google Scholar] [CrossRef]
- Gallagher, R.T.; Keogh, R.G.; Latch, G.C.M.; Reid, C.S.W. The Role of Fungal Tremorgens in Ryegrass Staggers. N. Z. J. Agric. Res. 1977, 20, 431–440. [Google Scholar] [CrossRef]
- Di Menna, M.E.; Mantle, P.G.; Mortimer, P.H. Letter: Experimental Production of a Staggers Syndrome in Ruminants by a Tremorgenic Penicillium from Soil. N. Z. Vet. J. 1976, 24, 45–46. [Google Scholar] [PubMed]
- Gallagher, R.T.; Campbell, A.G.; Hawkes, A.D.; Holland, P.T.; McGaveston, D.A.; Pansier, E.A.; Harvey, I.C. Ryegrass Staggers: The Presence of Lolitrem Neurotoxins in Perennial Ryegrass Seed. N. Z. Vet. J. 1982, 30, 183–184. [Google Scholar] [CrossRef] [PubMed]
- Tor-Agbidye, J.; Blythe, L.L.; Craig, A.M. Correlation of Endophyte Toxins ergovaline and Lolitrem B. with Clinical Disease: Fescue Foot and Perennial Ryegrass Staggers. Vet. Hum. Toxicol. 2001, 43, 140–146. [Google Scholar] [PubMed]
- Miles, C.O.; Wilkins, A.L.; Gallagher, R.T.; Hawkes, A.D.; Munday, S.C.; Towers, N.R. Synthesis and Tremorgenicity of Paxitriols and Lolitriol: Possible Biosynthetic Precursors of Lolitrem B. J. Agric. Food Chem. 1992, 40, 234–238. [Google Scholar] [CrossRef]
- Easton, H.S.; Lane, G.A.; Tapper, B.A.; Keogh, R.G.; Cooper, B.M.; Blackwell, M.; Fletcher, L.R. Ryegrass Endophyte-Related Heat Stress in Cattle. Proc. N. Z. Grassland Assoc. 1995, 57, 37–41. [Google Scholar]
- Auldist, M.J.; Thom, E.R. Effects of Endophyte Infection of Perennial Ryegrass on Somatic Cell Counts, Mammary Inflammation, and Milk Protein Composition in Grazing Dairy Cattle. N. Z. J. Agric. Res. 2000, 43, 345–349. [Google Scholar] [CrossRef]
- Hovermale, J.T.; Craig, A.M. Correlation of Ergovaline and Lolitrem B Levels in Endophyte-Infected Perennial Ryegrass Lolium Perenne. J. Vet. Diagn. Investig. 2001, 13, 323–327. [Google Scholar] [CrossRef]
- Benkhelil, A.; Grancher, D.; Giraud, N.; Bezille, P.; Bony, S. Intoxication Par Des Toxines de Champignons Endophytes Chez Des Taureaux Reproducteurs. Rev. Méd. Vét. 2004, 156, 243–247. [Google Scholar]
- Zbib, N.; Repussard, C.; Tardieu, D.; Priymenko, N.; Domange, C.; Guerre, P. Ergovaline in Tall Fescue and Its Effect on Health, Milk Quality, Biochemical Parameters, Oxidative Status, and Drug Metabolizing Enzymes of Lactating Ewes. J. Anim. Sci. 2014, 92, 5112–5123. [Google Scholar] [CrossRef] [PubMed]
- Zbib, N.; Repussard, C.; Tardieu, D.; Priymenko, N.; Domange, C.; Guerre, P. Toxicity of Endophyte-Infected Ryegrass Hay Containing High Ergovaline Level in Lactating Ewes. J. Anim. Sci. 2015, 93, 4098–4109. [Google Scholar] [CrossRef] [PubMed]
- Gadberry, M.S.; Denard, T.M.; Spiers, D.E.; Piper, E.L. Effects of Feeding Ergovaline on Lamb Performance in a Heat Stress Environment. J. Anim. Sci. 2003, 81, 1538–1545. [Google Scholar] [PubMed]
- Dalziel, J.E.; Dunstan, K.E.; Finch, S.C. Combined Effects of Fungal Alkaloids on Intestinal Motility in an in Vitro Rat Model. J. Anim. Sci. 2013, 91, 5177–5182. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, L.R.; Easton, H.S. The Evaluation and Use of Endophytes for Pasture Improvement. In Neotyphodium/Grass Interactions; Bacon, C.W., Hill, N.S., Eds.; Springer: New York, NY, USA, 1997; pp. 209–227. [Google Scholar]
- Bluett, S.J.; Thom, E.R.; Clark, D.A.; Macdonald, K.A.; Minneé, E.M.K. Effects of Perennial Ryegrass Infected with Either AR1 or Wild Endophyte on Dairy Production in the Waikato. N. Z. J. Agric. Res. 2005, 48, 197–212. [Google Scholar] [CrossRef]
- Thom, E.R.; Waugh, C.D.; Minneé, E.M.K. Dairy Cow Responses to the Wild-Type Endophyte in Perennial Ryegrass. In Proceedings of the 4th Australasian Dairy Science Symposium 2010, Hamilton, New Zealand, 2010; pp. 370–375.
- Finch, S.; Thom, E.; Babu, J.; Hawkes, A.; Waugh, C. The Evaluation of Fungal Endophyte Toxin Residues in Milk. N. Z. Vet. J. 2013, 61, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Uhlig, S.; Botha, C.J.; Vrålstad, T.; Rolén, E.; Miles, C.O. Indole-diterpenes and Ergot Alkaloids in Cynodon Dactylon Bermuda Grass. Infected with Claviceps Cynodontis from an Outbreak of Tremors in Cattle. J. Agric. Food Chem. 2009, 57, 11112–11119. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Monahan, B.J.; Tkacz, J.S.; Scott, B. Indole-Diterpene Gene Cluster from Aspergillus Flavus. Appl. Environ. Microbiol. 2004, 70, 6875–6883. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.R.; Pratley, J.E.; Mace, W.J.; Weston, L.A. Variation in Alkaloid Production from Genetically Diverse Lolium Accessions Infected with Epichloë Species. J. Agric. Food Chem. 2015, 63, 10355–10365. [Google Scholar] [CrossRef] [PubMed]
- Zbib, N.; Repussard, C.; Tardieu, D.; Guerre, P. Toxicité Des Mycotoxines Produites Par Des Champignons Endophytes Du Genre Neotyphodium. Rev. Méd. Vét. 2014, 165, 116–135. [Google Scholar]
- Norris, P.J.; Smith, C.C.T.; De Belleroche, J.; Bradford, H.F.; Mantle, P.G.; Thomas, A.J.; Penny, R.H.C. Actions of Tremorgenic Fungal Toxins on Neurotransmitter Release. J. Neurochem. 1980, 34, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Mantle, P.G. Amino Acid Neurotransmitter Release from Cerebrocortical Synaptosomes of Sheep with Severe Ryegrass Staggers in New Zealand. Res. Vet. Sci. 1983, 34, 373–375. [Google Scholar] [PubMed]
- Gant, D.B.; Cole, R.J.; Valdes, J.J.; Eldefrawi, M.E.; Eldefrawi, A.T. Action of Tremorgenic Mycotoxins on GABAA Receptor. Life Sci. 1987, 41, 2207–2214. [Google Scholar] [CrossRef]
- Gallagher, R.T.; Hawkes, A.D. Estimation of Neurotoxin Levels in Perennial Ryegrass by Mouse Bioassay. N. Z. J. Agric. Res. 1985, 28, 427–431. [Google Scholar] [CrossRef]
- Gallagher, R.T.; Hawkes, A.D. The Potent Tremorgenic Neurotoxins Lolitrem B and Aflatrem: A Comparison of the Tremor Response in Mice. Experientia 1986, 42, 823–825. [Google Scholar] [CrossRef] [PubMed]
- Munday-Finch, S.C.; Garthwaite, I. Toxicology of Ryegrass Endophyte in Livestock. Ryegrass Endophyte Essent. N. Z. Symbiosis Grassl. Res. Pract. Ser. 1999, 7, 63–67. [Google Scholar]
- Smith, B.L.; McLeay, L.M.; Embling, P.P. Effect of the Mycotoxins Penitrem, Paxilline and Lolitrem B on the Electromyographic Activity of Skeletal and Gastrointestinal Smooth Muscle of Sheep. Res. Vet. Sci. 1997, 62, 111–116. [Google Scholar] [CrossRef]
- Selala, M.I.; Laekeman, G.M.; Loenders, B.; Musuku, A.; Herman, A.G.; Schepens, P. In Vitro Effects of Tremorgenic Mycotoxins. J. Nat. Prod. 1991, 54, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cross, A.L.; Allen, K.L.; Smith, B.L.; McLeay, L.M. Tremorgenic Mycotoxins Increase Gastric Smooth Muscle Activity of Sheep Reticulum and Rumen in Vitro. Res. Vet. Sci. 2003, 74, 93–100. [Google Scholar] [CrossRef]
- McLeay, L.M.; Smith, B.L. Effects of the Mycotoxins Lolitrem B and Paxilline on Gastrointestinal Smooth Muscle, the Cardiovascular and Respiratory Systems, and Temperature in Sheep. Ryegrass Endophyte: An Essential New Zealand Symbiosis. Grassland Res. Pract. Ser. 1999, 7, 69–76. [Google Scholar]
- Ruckebusch, Y.; Malbert, C.H.; Crichlow, E.C. Hexamethonium: A Probe to Assess Autonomic Nervous System Involvement in Upper Gastrointestinal Functions in Conscious Sheep. Vet. Res. Commun. 1987, 11, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Knaus, H.-G.; McManus, O.B.; Lee, S.H.; Schmalhofer, W.A.; Garcia-Calvo, M.; Helms, L.M.; Sanchez, M.; Giangiacomo, K.; Reuben, J.P. Tremorgenic Indole Alkaloids Potently Inhibit Smooth Muscle High-Conductance Calcium-Activated Potassium Channels. Biochem. Mosc. 1994, 33, 5819–5828. [Google Scholar] [CrossRef]
- Lee, U.S.; Cui, J. BK Channel Activation: Structural and Functional Insights. Trends Neurosci. 2010, 33, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Imlach, W.L.; Finch, S.C.; Dunlop, J.; Meredith, A.L.; Aldrich, R.W.; Dalziel, J.E. The Molecular Mechanism of “Ryegrass Staggers,” a Neurological Disorder of K+ Channels. J. Pharmacol. Exp. Ther. 2008, 327, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Imlach, W.L.; Finch, S.C.; Dunlop, J.; Dalziel, J.E. Structural Determinants of Lolitrems for Inhibition of BK Large Conductance Ca2+-Activated K+ Channels. Eur. J. Pharmacol. 2009, 605, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Munday-Finch, S.C.; Wilkins, A.L.; Miles, C.O.; Ede, R.M.; Thomson, R.A. Structure Elucidation of Lolitrem F, a Naturally Occurring Stereoisomer of the Tremorgenic Mycotoxin Lolitrem B, Isolated from Lolium Perenne Infected with Acremonium Lolii. J. Agric. Food Chem. 1996, 44, 2782–2788. [Google Scholar] [CrossRef]
- Dalziel, J.E.; Finch, S.C.; Dunlop, J. The Fungal Neurotoxin Lolitrem B Inhibits the Function of Human Large Conductance Calcium-Activated Potassium Channels. Toxicol. Lett. 2005, 155, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Grancher, D.; Durix, A.; Moulard, Y.; Bonnaire, Y.; Carcelen, M.; Camier, Y.; Bony, S. Distribution Du Lolitrème B et de L’ergovaline Après Un Bolus Intraruminal Chez La Chèvre En Lactation; Rencontres Recherches Ruminants: Paris, France, 2004; Volume 11, p. 37. [Google Scholar]
- Miyazaki, S.; Ishizaki, I.; Ishizaka, M.; Kanbara, T.; Ishiguro-Takeda, Y. Lolitrem B Residue in Fat Tissues of Cattle Consuming Endophyte-Infected Perennial Ryegrass Straw. J. Vet. Diagn. Invest. 2004, 16, 340–342. [Google Scholar] [CrossRef] [PubMed]
- Finch, S.; Fletcher, L.; Babu, J. The Evaluation of Endophyte Toxin Residues in Sheep Fat. N. Z. Vet. J. 2012, 60, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Shimada, N.; Yoshioka, M.; Mikami, O.; Tanimura, N.; Yamanaka, N.; Hanazumi, M.; Kojima, F.; Miyazaki, S. Toxicological Evaluation and Bioaccumulation Potential of Lolitrem B, Endophyte Mycotoxin in Japanese Black Steers. Food Addit. Contam. Part A 2013, 30, 1402–1406. [Google Scholar] [CrossRef] [PubMed]
- McMillan, L.K.; Carr, R.L.; Young, C.A.; Astin, J.W.; Lowe, R.G.T.; Parker, E.J.; Jameson, G.B.; Finch, S.C.; Miles, C.O.; McManus, O.B.; et al. Molecular Analysis of Two Cytochrome P450 Monooxygenase Genes Required for Paxilline Biosynthesis in Penicillium Paxilli, and Effects of Paxilline Intermediates on Mammalian Maxi-K Ion Channels. Mol. Genet. Genom. 2003, 270, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Saikia, S.; Takemoto, D.; Tapper, B.A.; Lane, G.A.; Fraser, K.; Scott, B. Functional Analysis of an Indole-Diterpene Gene Cluster for Lolitrem B Biosynthesis in the Grass Endosymbiont Epichloë Festucae. FEBS Lett. 2012, 586, 2563–2569. [Google Scholar] [CrossRef] [PubMed]
- Di Menna, M.; Finch, S.; Popay, A.; Smith, B. A Review of the Neotyphodium Lolii / Lolium Perenne Symbiosis and Its Associated Effects on Animal and Plant Health, with Particular Emphasis on Ryegrass Staggers. N. Z. Vet. J. 2012, 60, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Van Essen, G.J.; Blom, M.; Fink Gremmels-Gehrmann, J. Ryegrass cramps in horses. Tijdschr. Diergeneeskd. 1995, 120, 710–711. [Google Scholar] [PubMed]
- Bony, S.; Collin, E.; Perret du Cray, G.; Ravel, C.; Delatour, P. Endophyte Toxicosis: Observations of an Outbreack of “Ryegrass Staggers” in Dairy Cow Herd in France. Rev. Méd. Vét. 1998, 6, 149. [Google Scholar]
- Holzhauer, M.; Westerhout, A.C. Young perennial ryegrass staggers in a dairy herd. Tijdschr. Diergeneeskd. 2004, 129, 298–300. [Google Scholar] [PubMed]
- Nollet, H.; Vanschandevijl, K.; Lefère, L.; Deprez, P. First Confirmed Case of Ryegrass Staggers in Horses in Belgium. Vlaams Diergeneeskd. Tijdschr. 2007, 76, 355–358. [Google Scholar]
- Galey, F.D.; Tracy, M.L.; Craigmill, A.L.; Barr, B.C.; Markegard, G.; Peterson, R.; O’Connor, M. Staggers Induced by Consumption of Perennial Ryegrass in Cattle and Sheep from Northern California. J. Am. Vet. Med. Assoc. 1991, 199, 466–470. [Google Scholar] [PubMed]
- Miyazaki, S.; Fukumura, M.; Yoshioka, M.; Yamanaka, N. Detection of Endophyte Toxins in the Imported Perennial Ryegrass Straw. J. Vet. Med. Sci. 2001, 63, 1013–1015. [Google Scholar] [CrossRef] [PubMed]
- Odriozola, E.; Lopez, T.; Campero, C.; Gimenez Placeres, C. Ryegrass Staggers in Heifers: A New Mycotoxicosis in Argentina. Vet. Hum. Toxicol. 1993, 35, 144–146. [Google Scholar] [PubMed]
- Latch, G.C.M.; Falloon, R.E.; Christensen, M.J. Fungi and Ryegrass Staggers. N. Z. J. Agric. Res. 1976, 19, 233–242. [Google Scholar] [CrossRef]
- Cunningham, P.J.; Foot, J.Z.; Reed, K.F.M. Perennial Ryegrass Lolium Perenne. Endophyte Acremonium Lolii. Relationships: The Australian Experience. Agric. Ecosyst. Environ. 1993, 44, 157–168. [Google Scholar] [CrossRef]
- Reed, K.F.M.; Leonforte, A.; Cunningham, P.J.; Walsh, J.R.; Allen, D.I.; Johnstone, G.R.; Kearney, G. Incidence of Ryegrass Endophyte Neotyphodium Lolii. and Diversity of Associated Alkaloid Concentrations among Naturalised Populations of Perennial Ryegrass Lolium Perenne L. Aust. J. Agric. Res. 2000, 51, 569–578. [Google Scholar] [CrossRef]
- Van Zijll de Jong, E.; Dobrowolski, M.P.; Bannan, N.R.; Stewart, A.V.; Smith, K.F.; Spangenberg, G.C.; Forster, J.W. Global Genetic Diversity of the Perennial Ryegrass Fungal Endophyte. Crop Sci. 2008, 48, 1487. [Google Scholar] [CrossRef]
- Popay, A.J.; Thom, E.R. Endophyte Effects on Major Insect Pests in Waikato Dairy Pasture. Proc. N. Z. Grassl. Assoc. 2009, 71, 121–126. [Google Scholar]
- Popay, A.J.; Hume, D.E. Endophytes Improve Ryegrass Persistence by Controlling Insects. Pasture Persistence Grassl. Res. Pract. Ser. 2011, 15, 149–156. [Google Scholar]
- Thom, E.R.; Popay, A.J.; Waugh, C.D.; Minneé, E.M.K. Impact of Novel Endophytes in Perennial Ryegrass on Herbage Production and Insect Pests from Pastures under Dairy Cow Grazing in Northern New Zealand. Grass Forage Sci. 2014, 69, 191–204. [Google Scholar] [CrossRef]
- Oliveira, J.A.; Rottinghaus, G.E.; Collar, J.; Castro, P. Perennial Ryegrass Endophytes in Galicia, Northwest Spain. J. Agric. Sci. 1997, 129, 173–177. [Google Scholar] [CrossRef]
- Ribeiro, M.A.M.D.V.; Gurney, K.A.; Bush, L.P. Endophyte Acremonium Lolii in Ecotypes of Perennial Ryegrass Lolium Perenne L. Collected in Old Irish Pastures. Ir. J. Agric. Food Res. 1996, 35, 151–157. [Google Scholar]
- Latch, G.C.M.; Potter, L.R.; Tyler, B.F. Incidence of Endophytes in Seeds from Collections of Lolium and Festuca Species. Ann. Appl. Biol. 1987, 111, 59–64. [Google Scholar] [CrossRef]
- Lewis, G.C.; Ravel, C.; Naffaa, W.; Astier, C.; Charmet, G. Occurrence of Acremonium Endophytes in Wild Populations of Lolium Spp. in European Countries and a Relationship between Level of Infection and Climate in France. Ann. Appl. Biol. 1997, 130, 227–238. [Google Scholar] [CrossRef]
- Mette Dahl Jensen, A.; Roulund, N. Occurrence of Neotyphodium Endophytes in Permanent Grassland with Perennial Ryegrass Lolium Perenne. in Denmark. Agric. Ecosyst. Environ. 2004, 104, 419–427. [Google Scholar] [CrossRef]
- Malinowski, D.P.; Belesky, D.P. Ecological Importance of Neotyphodium Spp. Grass Endophytes in Agroecosystems. Grassl. Sci. 2006, 52, 1–14. [Google Scholar] [CrossRef]
- Young, C.A.; Tapper, B.A.; May, K.; Moon, C.D.; Schardl, C.L.; Scott, B. Indole-Diterpene Biosynthetic Capability of Epichloe Endophytes as Predicted by ltm Gene Analysis. Appl. Environ. Microbiol. 2009, 75, 2200–2211. [Google Scholar] [CrossRef] [PubMed]
- Hettiarachchige, I.K.; Ekanayake, P.N.; Mann, R.C.; Guthridge, K.M.; Sawbridge, T.I.; Spangenberg, G.C.; Forster, J.W. Phylogenomics of Asexual Epichloë Fungal Endophytes Forming Associations with Perennial Ryegrass. BMC Evol. Biol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Easton, H.S.; Latch, G.C.M.; Tapper, B.A.; Ball, O.-P. Ryegrass Host Genetic Control of Concentrations of Endophyte-Derived Alkaloids. Crop Sci. 2002, 42, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Young, C.A.; Felitti, S.; Shields, K.; Spangenberg, G.; Johnson, R.D.; Bryan, G.T.; Saikia, S.; Scott, B. A Complex Gene Cluster for Indole-Diterpene Biosynthesis in the Grass Endophyte Neotyphodium Lolii. Fungal Genet. Biol. 2006, 43, 679–693. [Google Scholar] [CrossRef] [PubMed]
- Young, C.A.; Bryant, M.K.; Christensen, M.J.; Tapper, B.A.; Bryan, G.T.; Scott, B. Molecular Cloning and Genetic Analysis of a Symbiosis-Expressed Gene Cluster for Lolitrem Biosynthesis from a Mutualistic Endophyte of Perennial Ryegrass. Mol. Genet. Genom. 2005, 274, 13–29. [Google Scholar] [CrossRef] [PubMed]
- Reed, K.F.M.; Walsh, J.R.; Cross, P.A.; McFarlane, N.M.; Sprague, M.A. Ryegrass Endophyte Neotyphodium Lolii. Alkaloids and Mineral Concentrations in Perennial Ryegrass Lolium Perenne. from Southwest Victorian Pasture. Aust. J. Exp. Agric. 2005, 44, 1185–1194. [Google Scholar] [CrossRef]
- Looper, M.L.; Aiken, G.E.; Rosenkrans, C.F., Jr. New Perspectives in Fescue Toxicosis and Ryegrass Staggers. In Proceedings of the 7th International Symposium on Fungal Endophytes of Grasses, Lexington, KY, USA, 28 June–1 July 2010; pp. 1–4.
- Fletcher, L.R. Novel Endophytes in New Zealand Grazing Systems: The Perfect Solution or a Compromise? In Epichloaëe, Endophytes of Cool Season Grasses: Implications, Utilization and Biology; Young, C.A., Aiken, G.E., McCulley, R.L., Strickland, J.R., Schardl, C.L., Eds.; Samuel Roberts Noble Foundation: Ardmore, OK, USA, 2012; pp. 5–13. [Google Scholar]
- Ravel, C.; Charmet, G.; Balfourier, F. Influence of the Fungal Endophyte Acremonium Lolii on Agronomic Traits of Perennial Ryegrass in France. Grass Forage Sci. 1995, 50, 75–80. [Google Scholar] [CrossRef]
- Ravel, C.; Courty, C.; Coudret, A.; Charmet, G. Beneficial Effects of Neotyphodium Lolii on the Growth and the Water Status in Perennial Ryegrass Cultivated under Nitrogen Deficiency or Drought Stress. Agronomie 1997, 17, 173–181. [Google Scholar] [CrossRef]
- Ravel, C.; Balfourier, F.; Guillaumin, J.J. Enhancement of Yield and Persistence of Perennial Ryegrass Inoculated with One Endophyte Isolate in France. Agronomie 1999, 19, 635–344. [Google Scholar] [CrossRef]
- Repussard, C.; Zbib, N.; Tardieu, D.; Guerre, P. Les Champignons Endophytes Du Genre Neotyphodium et Leurs Toxines: Généralités et Problématique Française. Rev. Méd. Vét. 2013, 164, 583–606. [Google Scholar]
- Oldenburg, E. Endophytic Fungi and Alkaloid Production in Perennial Ryegrass in Germany. Grass Forage Sci. 1997, 52, 425–431. [Google Scholar] [CrossRef]
- Durix, A.; Bony, S.; Ravel, C.; Balfourier, F.; Guillaumin, J.J.; Ghesquiere, M.; Chosson, J.F.; Charmet, G. The Influence of Toxic Neotyphodium Endophyte on Perennial Ryegrass (Lolium Perenne). Rev. Méd. Vét. 1998, 6, 528. [Google Scholar]
- Watson, R.H. Endophytic Perennial Ryegrass and Reproductive Performance of the Ewe. Ph.D. Thesis, Massey University, Auckland, New Zealand, 2000. [Google Scholar]
- Reed, K.F.M.; Nie, Z.N.; Walker, L.V.; Kearney, G. Fluctuations in the Concentration of Ergovaline and Lolitrem B Produced by the Wild-Type Endophyte Neotyphodium Lolii. in Perennial Ryegrass Lolium Perenne. Pasture. Anim. Prod. Sci. 2011, 51, 1098–1108. [Google Scholar] [CrossRef]
- Repussard, C.; Zbib, N.; Tardieu, D.; Guerre, P. Ergovaline and Lolitrem B Concentrations in Perennial Ryegrass in Field Culture in Southern France: Distribution in the Plant and Impact of Climatic Factors. J. Agric. Food Chem. 2014, 62, 12707–12712. [Google Scholar] [CrossRef] [PubMed]
- Cagaš, B.; Flieger, M.; Olšovská, J. Concentration of Ergot Alkaloids in Czech Ecotypes of Lolium Perenne and Festuca Pratensis. Grass Forage Sci. 1999, 54, 365–370. [Google Scholar] [CrossRef]
- Eerens, J.P.J.; Lucas, R.J.; Easton, H.S.; White, J.G.H. Influence of the Ryegrass Endophyte Neotyphodium Lolii. in a Cool Moist Environment. I. Pasture Production. N. Z. J. Agric. Res. 1998, 41, 39–48. [Google Scholar] [CrossRef]
- Keogh, R.G.; Tapper, B.A.; Fletcher, R.H. Distributions of the Fungal Endophyte Acremonium Lolii, and of the Alkaloids Lolitrem B and Peramine, within Perennial Ryegrass. N. Z. J. Agric. Res. 1996, 39, 121–127. [Google Scholar] [CrossRef]
- Spiering, M.J.; Lane, G.A.; Christensen, M.J.; Schmid, J. Distribution of the Fungal Endophyte Neotyphodium Lolii Is Not a Major Determinant of the Distribution of Fungal Alkaloids in Lolium Perenne Plants. Phytochemistry 2005, 66, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Lyons, P.C.; Plattner, R.D.; Bacon, C.W. Occurrence of Peptide and Clavine Ergot Alkaloids in Tall Fescue Grass. Science 1986, 232, 487–489. [Google Scholar] [CrossRef] [PubMed]
- Belesky, D.P.; Stuedemann, J.A.; Plattner, R.D.; Wilkinson, S.R. Ergopeptine Alkaloids in Grazed Tall Fescue. Agron. J. 1988, 80, 209. [Google Scholar] [CrossRef]
- Rottinghaus, G.E.; Garner, G.B.; Cornell, C.N.; Ellis, J.L. HPLC Method for Quantitating Ergovaline in Endophyte-Infested Tall Fescue: Seasonal Variation of Ergovaline Levels in Stems with Leaf Sheaths, Leaf Blades, and Seed Heads. J. Agric. Food Chem. 1991, 39, 112–115. [Google Scholar] [CrossRef]
- TePaske, M.R.; Powell, R.G.; Clement, S.L. Analyses of Selected Endophyte-Infected Grasses for the Presence of Loline-Type and Ergot-Type Alkaloids. J. Agric. Food Chem. 1993, 41, 2299–2303. [Google Scholar] [CrossRef]
- Arechavaleta, M.; Bacon, C.W.; Plattner, R.D.; Hoveland, C.S.; Radcliffe, D.E. Accumulation of Ergopeptide Alkaloids in Symbiotic Tall Fescue Grown under Deficits of Soil Water and Nitrogen Fertilizer. Appl. Environ. Microbiol. 1992, 58, 857–861. [Google Scholar] [PubMed]
- Repussard, C.; Zbib, N.; Tardieu, D.; Guerre, P. Endophyte Infection of Tall Fescue and the Impact of Climatic Factors on Ergovaline Concentrations in Field Crops Cultivated in Southern France. J. Agric. Food Chem. 2014, 62, 9609–9614. [Google Scholar] [CrossRef] [PubMed]
- Hunt, M.G.; Rasmussen, S.; Newton, P.C.D.; Parsons, A.J.; Newman, J.A. Near-Term Impacts of Elevated CO2, Nitrogen and Fungal Endophyte-Infection on Lolium Perenne L. Growth, Chemical Composition and Alkaloid Production. Plant Cell Environ. 2005, 28, 1345–1354. [Google Scholar] [CrossRef]
- Rasmussen, S.; Parsons, A.J.; Bassett, S.; Christensen, M.J.; Hume, D.E.; Johnson, L.J.; Johnson, R.D.; Simpson, W.R.; Stacke, C.; Voisey, C.R.; et al. High Nitrogen Supply and Carbohydrate Content Reduce Fungal Endophyte and Alkaloid Concentration in Lolium Perenne. New Phytol. 2007, 173, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Reed, K.F.M.; Nie, Z.N.; Walker, L.V.; Mace, W.J.; Clark, S.G. Weather and Pasture Characteristics Associated with Outbreaks of Perennial Ryegrass Toxicosis in Southern Australia. Anim. Prod. Sci. 2011, 51, 738–752. [Google Scholar] [CrossRef]
- Xu, W.; Gavia, D.J.; Tang, Y. Biosynthesis of Fungal Indole Alkaloids. Nat. Prod. Rep. 2014, 31, 1474–1487. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, M.; Eaton, C.; Stärkel, C.; Tapper, B.; Cox, M.; Scott, B. Molecular Cloning and Functional Analysis of Gene Clusters for the Biosynthesis of Indole-Diterpenes in Penicillium Crustosum and P. Janthinellum. Toxins 2015, 7, 2701–2722. [Google Scholar] [CrossRef] [PubMed]
- Cole, R.J.; Dorner, J.W.; Lansden, J.A.; Cox, R.H.; Pape, C.; Cunfer, B.; Nicholson, S.S.; Bedell, D.M. Paspalum Staggers: Isolation and Identification of Tremorgenic Metabolites from Sclerotia of Claviceps Paspali. J. Agric. Food Chem. 1977, 25, 1197–1201. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.B.; Cui, H. Indole-Diterpene Synthetic Studies: Total Synthesis of (—)-21-Isopentenylpaxilline. Helv. Chim. Acta 2003, 86, 3908–3938. [Google Scholar] [CrossRef]
- Weedon, C.M.; Mantle, P.G. Paxilline Biosynthesis by Acremonium Loliae; a Step towards Defining the Origin of Lolitrem Neurotoxins. Phytochemistry 1987, 26, 969–971. [Google Scholar] [CrossRef]
- Miles, C.O.; di Menna, M.E.; Jacobs, S.W.L.; Garthwaite, I.; Lane, G.A.; Prestidge, R.A.; Marshall, S.L.; Wilkinson, H.H.; Schardl, C.L.; Ball, O.J.-P.; et al. Endophytic Fungi in Indigenous Australasian Grasses Associated with Toxicity to Livestock. Appl. Environ. Microbiol. 1998, 64, 601–606. [Google Scholar] [PubMed]
- Munday-Finch, S.C.; Wilkins, A.L.; Miles, C.O.; Tomoda, H.; Ōmura, S. Isolation and Structure Elucidation of Lolilline, a Possible Biosynthetic Precursor of the Lolitrem Family of Tremorgenic Mycotoxins. J. Agric. Food Chem. 1997, 45, 199–204. [Google Scholar] [CrossRef]
- Miles, C.O.; Munday, S.C.; Wilkins, A.L.; Ede, R.M.; Towers, N.R. Large-Scale Isolation of Lolitrem B and Structure Determination of Lolitrem E. J. Agric. Food Chem. 1994, 42, 1488–1492. [Google Scholar] [CrossRef]
- Gallagher, R.T.; Latch, G.C.; Keogh, R.G. The Janthitrems: Fluorescent Tremorgenic Toxins Produced by Penicillium Janthinellum Isolates from Ryegrass Pastures. Appl. Environ. Microbiol. 1980, 39, 272–273. [Google Scholar] [PubMed]
- Penn, J.; Swift, R.; Wigley, L.J.; Mantle, P.G.; Bilton, J.N.; Sheppard, R.N. Janthitrems B and C, Two Principal Indole-Diterpenoids Produced by Penicillium Janthinellum. Phytochemistry 1993, 32, 1431–1434. [Google Scholar] [CrossRef]
- Caradus, J.; Lovatt, S.; Belgrave, B. Adoption of Forage Technologies by New Zealand Farmers-Case Studies. In Proceedings of the 22nd International Grassland Congress, Tauranga, New Zealand, 5–7 November 2013; pp. 1843–1845.
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Philippe, G. Lolitrem B and Indole Diterpene Alkaloids Produced by Endophytic Fungi of the Genus Epichloë and Their Toxic Effects in Livestock. Toxins 2016, 8, 47. https://doi.org/10.3390/toxins8020047
Philippe G. Lolitrem B and Indole Diterpene Alkaloids Produced by Endophytic Fungi of the Genus Epichloë and Their Toxic Effects in Livestock. Toxins. 2016; 8(2):47. https://doi.org/10.3390/toxins8020047
Chicago/Turabian StylePhilippe, Guerre. 2016. "Lolitrem B and Indole Diterpene Alkaloids Produced by Endophytic Fungi of the Genus Epichloë and Their Toxic Effects in Livestock" Toxins 8, no. 2: 47. https://doi.org/10.3390/toxins8020047
APA StylePhilippe, G. (2016). Lolitrem B and Indole Diterpene Alkaloids Produced by Endophytic Fungi of the Genus Epichloë and Their Toxic Effects in Livestock. Toxins, 8(2), 47. https://doi.org/10.3390/toxins8020047




