O’Leary-Sant Symptom Index Predicts the Treatment Outcome for OnabotulinumtoxinA Injections for Refractory Interstitial Cystitis/Bladder Pain Syndrome
Abstract
:1. Introduction
2. Results and Discussion
| Parameters | Total (n = 101) | Male (n = 13) | Female (n = 88) | p Value |
|---|---|---|---|---|
| Age (years) | 48.45 ± 11.97 | 46.00 ± 13.27 | 48.81 ± 11.81 | 0.433 |
| ICSI | 12.34 ± 3.42 | 11.08 ± 3.95 | 12.52 ± 3.32 | 0.156 |
| ICPI | 11.43 ± 3.03 | 11.77 ± 3.19 | 11.38 ± 3.02 | 0.664 |
| OSS (ICSI + ICPI) | 23.76 ± 6.14 | 22.85 ± 6.59 | 23.90 ± 6.09 | 0.567 |
| Pain VAS | 5.23 ± 2.37 | 4.23 ± 3.24 | 5.38 ± 2.20 | 0.105 |
| FBC | 128.86 ± 75.37 | 156.92 ± 58.93 | 124.72 ± 76.91 | 0.151 |
| Frequency | 15.39 ± 7.68 | 13.69 ± 6.10 | 15.64 ± 7.88 | 0.397 |
| Nocturia | 4.70 ± 4.67 | 3.38 ± 1.85 | 4.90 ± 4.93 | 0.278 |
| FSF | 115.96 ± 53.40 | 105.92 ± 55.63 | 117.33 ± 53.28 | 0.490 |
| FD | 171.19 ± 73.44 | 192.20 ± 72.48 | 168.53 ± 73.59 | 0.340 |
| SD | 202.46 ± 87.01 | 233.67 ± 79.81 | 197.45 ± 87.74 | 0.249 |
| CBC | 273.98 ± 109.32 | 268.83 ± 109.71 | 274.72 ± 109.92 | 0.863 |
| Pdet | 19.87 ± 10.82 | 24.33 ± 11.39 | 19.19 ± 10.65 | 0.126 |
| Qmax | 12.56 ± 5.38 | 12.17 ± 4.86 | 12.62 ± 5.48 | 0.787 |
| Volume | 241.18 ± 110.54 | 219.67 ± 101.13 | 244.41 ± 112.12 | 0.473 |
| PVR | 39.38 ± 94.08 | 49.17 ± 103.61 | 38.01 ± 93.26 | 0.703 |
| Positive PST | 98 (97.03) | 12 (92.31) | 86 (97.73) | 0.342 1 |
| Cystoscopic HD | ||||
| MBC (mL) | 570.00 ± 198.07 | 603.85 ± 145.00 | 681.82 ± 218.30 | 0.216 |
| Hunner’s ulcer | 10 (9.90) | 0 (0) | 10 (11.36) | 0.200 1 |
| Glomerulation | 0.386 1 | |||
| Grade 0 | 8 (7.92) | 1 (7.69) | 7 (7.95) | |
| Grade 1 | 39 (38.61) | 7 (53.85) | 32(36.36) | |
| Grade 2 | 35 (34.65) | 3 (23.08) | 32 (36.36) | |
| Grade 3 | 11 (10.89) | 0 (0) | 11 (12.50) | |
| Grade 4 | 8(7.92) | 2 (15.38) | 6 (6.82) |
| Parameters (N = 101) | Baseline | 3 Months | 6 Months | p Value 1 |
|---|---|---|---|---|
| ICSI | 12.34 ± 3.42 | 8.92 ± 4.42 | 8.79 ± 4.65 | 0.000 |
| ICPI | 11.43 ± 3.03 | 7.51 ± 4.30 | 7.75 ± 4.57 | 0.000 |
| OSS (ICSI+ICPI) | 23.76 ± 6.14 | 16.43 ± 8.51 | 16.54 ± 8.95 | 0.000 |
| Pain VAS | 5.23 ± 2.37 | 3.45 ± 2.47 | 3.50 ± 2.54 | 0.000 |
| FBC | 128.86 ± 75.37 | 180.00 ± 90.53 | 177.92 ± 86.39 | 0.000 |
| Frequency | 15.39 ± 7.68 | 11.79 ± 6.35 | 11.28 ± 6.29 | 0.000 |
| Nocturia | 4.70 ± 4.67 | 3.31 ± 2.86 | 3.52 ± 3.79 | 0.026 |
| Qmax | 12.56 ± 5.38 | 13.97 ± 6.07 | 14.53 ± 5.91 | 0.060 |
| Volume | 241.18 ± 110.54 | 236.66 ± 123.27 | 236.50 ± 121.70 | 0.955 |
| PVR | 39.38 ± 94.08 | 48.03 ± 79.09 | 48.09 ± 77.48 | 0.711 |
| CBC | 273.98 ± 109.32 | 285.64 ± 140.59 | 284.59 ± 142.12 | 0.799 |
| GRA | 0 | 1.27 ± 1.00 | 1.32 ± 0.97 | 0.000 |
| Parameters | Treatment Outcome | OR (95% CI) | p Value | |
|---|---|---|---|---|
| Successful | Failed | |||
| No. patients | 46 | 55 | ||
| Gender | ||||
| Male (%) | 5 (38.46) | 8 (61.54) | 1.396 (0.423–4.603) | 0.584 |
| Female (%) | 41 (46.59) | 47 (53.41) | ||
| Age (years) | 46.91 ± 12.00 | 49.73 ± 11.91 | 0.980 (0.948–1.014) | 0.240 |
| ICSI | 11.15 ± 3.47 | 13.33 ± 3.07 | 0.811 (0.709–0.928) | 0.002 |
| ICPI | 10.76 ± 3.34 | 11.98 ± 2.65 | 0.869 (0.756–0.999) | 0.048 |
| OSS (ICSI+ICPI) | 21.91 ± 6.56 | 25.31 ± 5.35 | 0.905 (0.756–0.999) | 0.008 |
| Pain VAS | 4.80 ± 2.25 | 5.58 ± 2.43 | 0.866 (0.729–1.030) | 0.104 |
| FBC | 151.96 ± 82.75 | 109.55 ± 63.09 | 1.008 (1.002–1.014) | 0.007 |
| Frequency | 13.37 ± 5.02 | 17.07 ± 9.04 | 0.922 (0.861–0.989) | 0.023 |
| Nocturia | 3.59 ± 2.54 | 5.64 ± 5.75 | 0.852 (0.724–1.002) | 0.052 |
| VUDS parameters | ||||
| FSF (mL) | 123.93 ± 49.57 | 109.17 ± 56.03 | 1.005 (0.998–1.013) | 0.169 |
| FD (mL) | 191.12 ± 68.71 | 154.17 ± 73.74 | 1.007 (1.001–1.014) | 0.021 |
| SD (mL) | 222.64 ± 83.10 | 181.66 ± 87.31 | 1.006 (1.000–1.012) | 0.062 |
| CBC (mL) | 289.50 ± 107.99 | 259.41 ± 109.66 | 1.003 (0.999–1.006) | 0.181 |
| Pdet (cm H2O) | 19.51 ± 10.39 | 20.19 ± 11.30 | 0.994 (0.957–1.033) | 0.765 |
| Qmax (mL/s) | 13.03 ± 4.81 | 12.11 ± 5.89 | 1.033 (0.955–1.117) | 0.416 |
| Voided volume (mL) | 264.82 ± 110.28 | 219.52 ± 107.38 | 1.004 (1.000–1.008) | 0.052 |
| PVR (mL) | 32.60 ± 91.79 | 42.19 ± 96.87 | 0.999 (0.995–1.004) | 0.752 |
| PST | ||||
| Negative (%) | 1 (33.33) | 2 (66.67) | ||
| Positive (%) | 45 (45.92) | 53 (54.08) | 1.698 (0.149–19.349) | 0.670 |
| Cystoscopic HD | ||||
| MBC (mL) | 688.04 ± 199.77 | 658.18 ± 221.48 | 1.001 (0.999–1.003) | 0.478 |
| Hunner’s ulcer | ||||
| Negative (%) | 43 (47.25) | 48 (52.75) | ||
| Positive (%) | 3 (30.00) | 7 (70.00) | 0.307 (0.756–0.999) | 0.307 |
| Glomerulation | ||||
| Grade 0 (%) | 6 (75.00) | 2 (25.00) | ||
| Grade 1 (%) | 15 (38.46) | 24 (61.54) | 0.208 (0.037–1.170) | 0.075 |
| Grade 2 (%) | 18 (51.43) | 17 (48.57) | 0.353 (0.062–1.995) | 0.239 |
| Grade 3 (%) | 4 (36.36) | 7 (63.64) | 0.190 (0.025–1.432) | 0.107 |
| Grade 4 (%) | 3 (37.50) | 5 (62.50) | 0.200 (0.023–1.712) | 0.142 |
| Parameters | Odds Ratio | 95% CI | p Value |
|---|---|---|---|
| ICSI | 0.770 | 0.601–0.989 | 0.040 |
| ICPI | 1.106 | 0.853–1.435 | 0.447 |
| FBC (mL) | 1.003 | 0.996–1.010 | 0.389 |
| Frequency | 1.011 | 0.904–1.053 | 0.531 |
| FD (mL) | 1.005 | 0.998–1.012 | 0.171 |
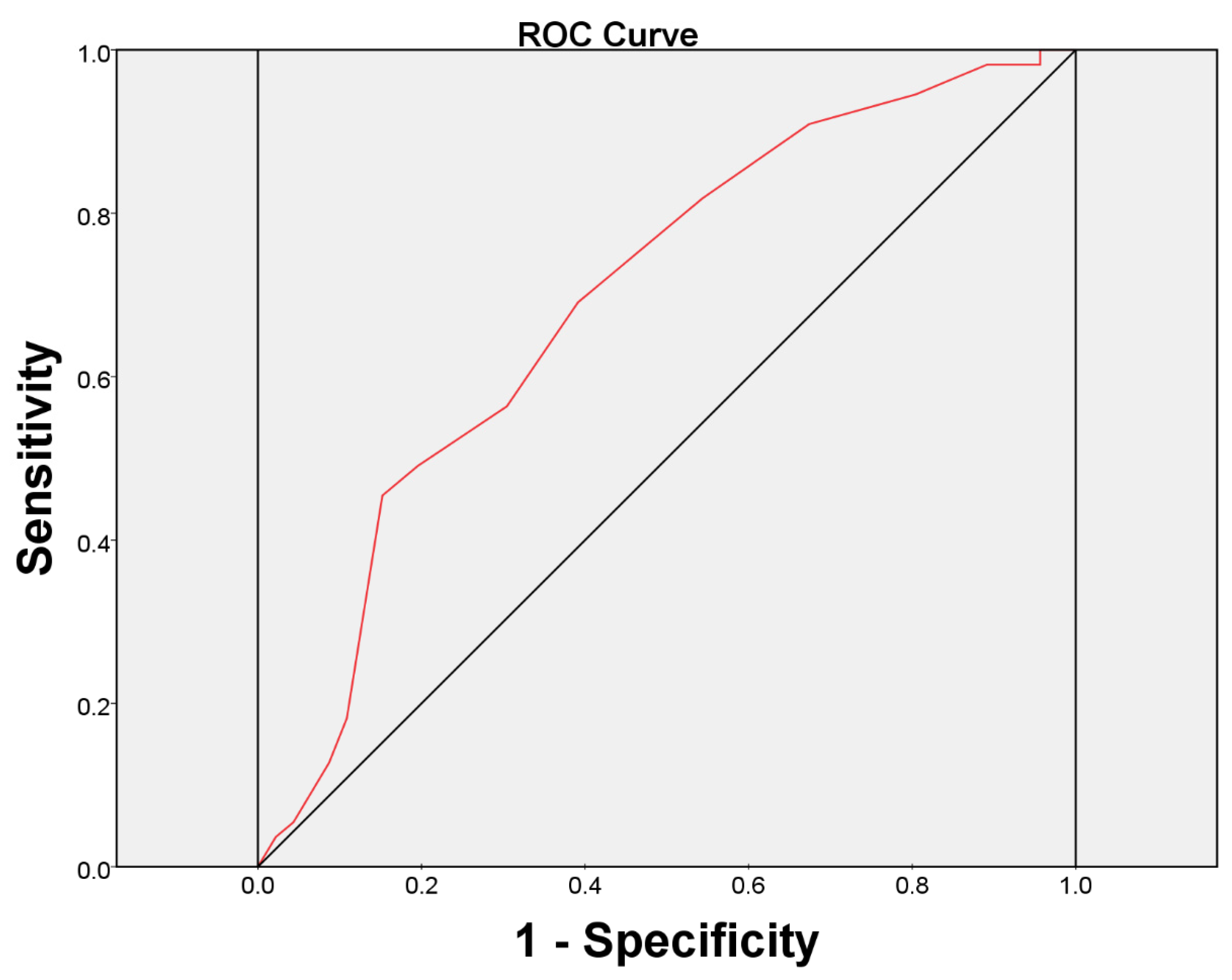
3. Experimental Section
3.1. Patient Enrollment
3.2. Outcome Assessment
3.3. Urodynamic Study
3.4. Botulinum Toxin Injections
3.5. Patient Follow-up
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Hanno, P.M.; Sant, G.R. Clinical highlights of the national institute of diabetes and digestive and kidney diseases/interstitial cystitis association scientific conference on interstitial cystitis. Urology 2001, 57, 2–6. [Google Scholar] [CrossRef]
- Payne, C.K.; Mosbaugh, P.G.; Forrest, J.B.; Evans, R.J.; Whitmore, K.E.; Antoci, J.P.; Perez-Marrero, R.; Jacoby, K.; Diokno, A.C.; O’Reilly, K.J.; et al. Intravesical resiniferatoxin for the treatment of interstitial cystitis: A randomized, double-blind, placebo controlled trial. J. Urol. 2005, 173, 1590–1594. [Google Scholar] [CrossRef] [PubMed]
- Nickel, J.C.; Barkin, J.; Forrest, J.; Mosbaugh, P.G.; Hernandez-Graulau, J.; Kaufman, D.; Lloyd, K.; Evans, R.J.; Parsons, C.L.; Atkinson, L.E. Randomized, double-blind, dose-ranging study of pentosan polysulfate sodium for interstitial cystitis. Urology 2005, 65, 654–658. [Google Scholar] [CrossRef] [PubMed]
- Sant, G.R.; Propert, K.J.; Hanno, P.M.; Burks, D.; Culkin, D.; Diokno, A.C.; Hardy, C.; Landis, J.R.; Mayer, R.; Madigan, R.; et al. A pilot clinical trial of oral pentosan polysulfate and oral hydroxyzine in patients with interstitial cystitis. J. Urol. 2003, 170, 810–815. [Google Scholar] [CrossRef] [PubMed]
- Hanno, P.M.; Buehler, J.; Wein, A.J. Use of amitriptyline in the treatment of interstitial cystitis. J. Urol. 1989, 141, 846–848. [Google Scholar] [PubMed]
- Sairanen, J.; Forsell, T.; Ruutu, M. Long-term outcome of patients with interstitial cystitis treated with low dose cyclosporine A. J. Urol. 2004, 171, 2138–2141. [Google Scholar] [CrossRef] [PubMed]
- Rapp, D.E.; Turk, K.W.; Bales, G.T.; Cook, S.P. Botulinum toxin type a inhibits calcitonin gene-related peptide release from isolated rat bladder. J. Urol. 2006, 175, 1138–1142. [Google Scholar] [CrossRef]
- Chuang, Y.C.; Yoshimura, N.; Huang, C.C.; Chiang, P.H.; Chancellor, M.B. Intravesical botulinum toxin a administration produces analgesia against acetic acid induced bladder pain responses in rats. J. Urol. 2004, 172, 1529–1532. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.P.; Radziszewski, P.; Borkowski, A.; Somogyi, G.T.; Boone, T.B.; Chancellor, M.B. Botulinum toxin a has antinociceptive effects in treating interstitial cystitis. Urology 2004, 64, 871–875. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.C. Preliminary results of suburothelial injection of botulinum a toxin in the treatment of chronic interstitial cystitis. Urol. Int. 2005, 75, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Giannantoni, A.; Costantini, E.; Di Stasi, S.M.; Tascini, M.C.; Bini, V.; Porena, M. Botulinum a toxin intravesical injections in the treatment of painful bladder syndrome: A pilot study. Eur. Urol. 2006, 49, 704–709. [Google Scholar] [CrossRef] [PubMed]
- Giannantoni, A.; Porena, M.; Costantini, E.; Zucchi, A.; Mearini, L.; Mearini, E. Botulinum a toxin intravesical injection in patients with painful bladder syndrome: 1-year followup. J. Urol. 2008, 179, 1031–1034. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.D.; Kuo, Y.C.; Kuo, H.C. Intravesical onabotulinumtoxin A injections for refractory painful bladder syndrome. Pain Phys. 2012, 15, 197–202. [Google Scholar]
- Chrysanthopoulou, E.L.; Doumouchtsis, S.K. Challenges and current evidence on the management of bladder pain syndrome. Neurourol. Urodyn. 2014, 33, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.; Lopes, T.; Frias, B.; Silva, A.; Silva, J.A.; Silva, C.M.; Cruz, C.; Cruz, F.; Dinis, P. Trigonal injection of botulinum toxin a in patients with refractory bladder pain syndrome/interstitial cystitis. Eur. Urol. 2010, 58, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Duggan, M.J.; Quinn, C.P.; Chaddock, J.A.; Purkiss, J.R.; Alexander, F.C.; Doward, S.; Fooks, S.J.; Friis, L.M.; Hall, Y.H.; Kirby, E.R.; et al. Inhibition of release of neurotransmitters from rat dorsal root ganglia by a novel conjugate of a clostridium botulinum toxin a endopeptidase fragment and erythrina cristagalli lectin. J. Biol. Chem. 2002, 277, 34846–34852. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Wang, J.; Lawrence, G.; Dolly, J.O. Synaptobrevin I mediates exocytosis of CGRP from sensory neurons and inhibition by botulinum toxins reflects their anti-nociceptive potential. J. Cell Sci. 2007, 120, 2864–2874. [Google Scholar] [CrossRef] [PubMed]
- Lucioni, A.; Bales, G.T.; Lotan, T.L.; McGehee, D.S.; Cook, S.P.; Rapp, D.E. Botulinum toxin type a inhibits sensory neuropeptide release in rat bladder models of acute injury and chronic inflammation. BJU Int. 2008, 101, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.T.; Kuo, H.C. Intravesical botulinum toxin a injections plus hydrodistension can reduce nerve growth factor production and control bladder pain in interstitial cystitis. Urology 2007, 70, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.T.; Tyagi, P.; Chancellor, M.B.; Kuo, H.C. Urinary nerve growth factor level is increased in patients with interstitial cystitis/bladder pain syndrome and decreased in responders to treatment. BJU Int. 2009, 104, 1476–1481. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.C.; Chancellor, M.B. Comparison of intravesical botulinum toxin type a injections plus hydrodistention with hydrodistention alone for the treatment of refractory interstitial cystitis/painful bladder syndrome. BJU Int. 2009, 104, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.C. Repeated intravesical onabotulinumtoxin A injections are effective in treatment of refractory interstitial cystitis/bladder pain syndrome. Int. J. Clin. Pract. 2013, 67, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.C. Repeated onabotulinumtoxin A injections provide better results than single injection in treatment of painful bladder syndrome. Pain Phys. 2013, 16, E15–E23. [Google Scholar]
- Denys, P.; Dmochowski, R.; Aliotta, P.; Castro-Diaz, D.; Blok, B.; Ethans, K.; Joshi, M.; Ni, Q.; Kennelly, M. Positive response to first onabotulinumtoxin A treatment persists long-term with repeattreatments in patients with neurogenic detrusor overactivity. Eur. Urol. Suppl. 2015, 14, e1092. [Google Scholar] [CrossRef]
- Peyronnet, B.; Roumiguié, M.; Castel-Lacanal, E.; Guillotreau, J.; Malavaud, B.; Marque, P.; Rischmann, P.; Gamé, X. Preliminary results of botulinum toxin A switch after first detrusor injection failure as a treatment of neurogenic detrusor overactivity. Neurourol. Urodyn. 2014. [Google Scholar] [CrossRef] [PubMed]
- Camprubi-Robles, M.; Planells-Cases, R.; Ferrer-Montiel, A. Differential contribution of SNARE-dependent exocytosis to inflammatory potentiation of TRPV1 in nociceptors. FASEB J. 2009, 23, 3722–3733. [Google Scholar] [CrossRef] [PubMed]
- Morenilla-Palao, C.; Planells-Cases, R.; Garcia-Sanz, N.; Ferrer-Montiel, A. Regulated exocytosis contributes to protein kinase C potentiation of vanilloid receptor activity. J Biol. Chem. 2004, 279, 25665–25672. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.P.; Vemulakonda, V.M.; Kiss, S.; Boone, T.B.; Somogyi, G.T. Enhanced ATP release from rat bladder urothelium during chronic bladder inflammation: Effect of botulinum toxin A. Neurochem. Int. 2005, 47, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Vemulakonda, V.M.; Somogyi, G.T.; Kiss, S.; Salas, N.A.; Boone, T.B.; Smith, C.P. Inhibitory effect on intravesically applied botulinum toxin A in chronic bladder inflammation. J. Urol. 2005, 173, 621–624. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.L.; Zheng, C.X.; Sui, B.D.; Li, Y.Q.; Wang, Y.Y.; Yang, Y.L. A closer look to botulinum neurotoxin type A-induced analgesia. Toxicon 2013, 71, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.C.; Jiang, Y.H.; Tsai, Y.C.; Kuo, Y.C. Intravesical botulinum toxin-A injections reduce bladder pain of interstitial cystitis/bladder pain syndrome refractory to conventional treatment—A prospective, multicenter, randomized, double-blind, placebo-controlled clinical trial. Neurourol. Urodyn. 2015. [Google Scholar] [CrossRef] [PubMed]
- Homma, Y.; Ueda, T.; Tomoe, H.; Lin, A.T.; Kuo, H.C.; Lee, M.H.; Lee, J.G.; Kim, D.Y.; Lee, K.S. Clinical guidelines for interstitial cystitis and hypersensitive bladder syndrome. Int. J. Urol. 2009, 16, 597–615. [Google Scholar] [CrossRef] [PubMed]
- Lubeck, D.P.; Whitmore, K.; Sant, G.R.; Alvarez-Horine, S.; Lai, C. Psychometric validation of the O’Leary-Sant interstitial cystitis symptom index in a clinical trial of pentosan polysulfate sodium. Urology 2001, 57, 62–66. [Google Scholar] [CrossRef]
- Propert, K.J.; Mayer, R.D.; Wang, Y.; Sant, G.R.; Hanno, P.M.; Peters, K.M.; Kusek, J.W. Responsiveness of symptom scales for interstitial cystitis. Urology 2006, 67, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Abrams, P.; Cardozo, L.; Fall, M.; Griffiths, D.; Rosier, P.; Ulmsten, U.; van Kerrebroeck, P.; Victor, A.; Wein, A. The standardisation of terminology of lower urinary tract function: Report from the standardisation sub-committee of the international continence society. Neurourol. Urodyn. 2002, 21, 167–178. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuo, Y.-C.; Kuo, H.-C. O’Leary-Sant Symptom Index Predicts the Treatment Outcome for OnabotulinumtoxinA Injections for Refractory Interstitial Cystitis/Bladder Pain Syndrome. Toxins 2015, 7, 2860-2871. https://doi.org/10.3390/toxins7082860
Kuo Y-C, Kuo H-C. O’Leary-Sant Symptom Index Predicts the Treatment Outcome for OnabotulinumtoxinA Injections for Refractory Interstitial Cystitis/Bladder Pain Syndrome. Toxins. 2015; 7(8):2860-2871. https://doi.org/10.3390/toxins7082860
Chicago/Turabian StyleKuo, Yuh-Chen, and Hann-Chorng Kuo. 2015. "O’Leary-Sant Symptom Index Predicts the Treatment Outcome for OnabotulinumtoxinA Injections for Refractory Interstitial Cystitis/Bladder Pain Syndrome" Toxins 7, no. 8: 2860-2871. https://doi.org/10.3390/toxins7082860
APA StyleKuo, Y.-C., & Kuo, H.-C. (2015). O’Leary-Sant Symptom Index Predicts the Treatment Outcome for OnabotulinumtoxinA Injections for Refractory Interstitial Cystitis/Bladder Pain Syndrome. Toxins, 7(8), 2860-2871. https://doi.org/10.3390/toxins7082860







